Assigning the Sex-Specific Markers via Genotyping-by-Sequencing onto the Y Chromosome for a Torrent Frog Amolops mantzorum
Abstract
1. Introduction
2. Material and Methods
2.1. Sampling and Preparation
2.2. DNA Extraction and Genotyping-by-Sequencing
2.3. Filtering and SNP Calling
2.4. Sex-Specific Locus Discovery and Isolation
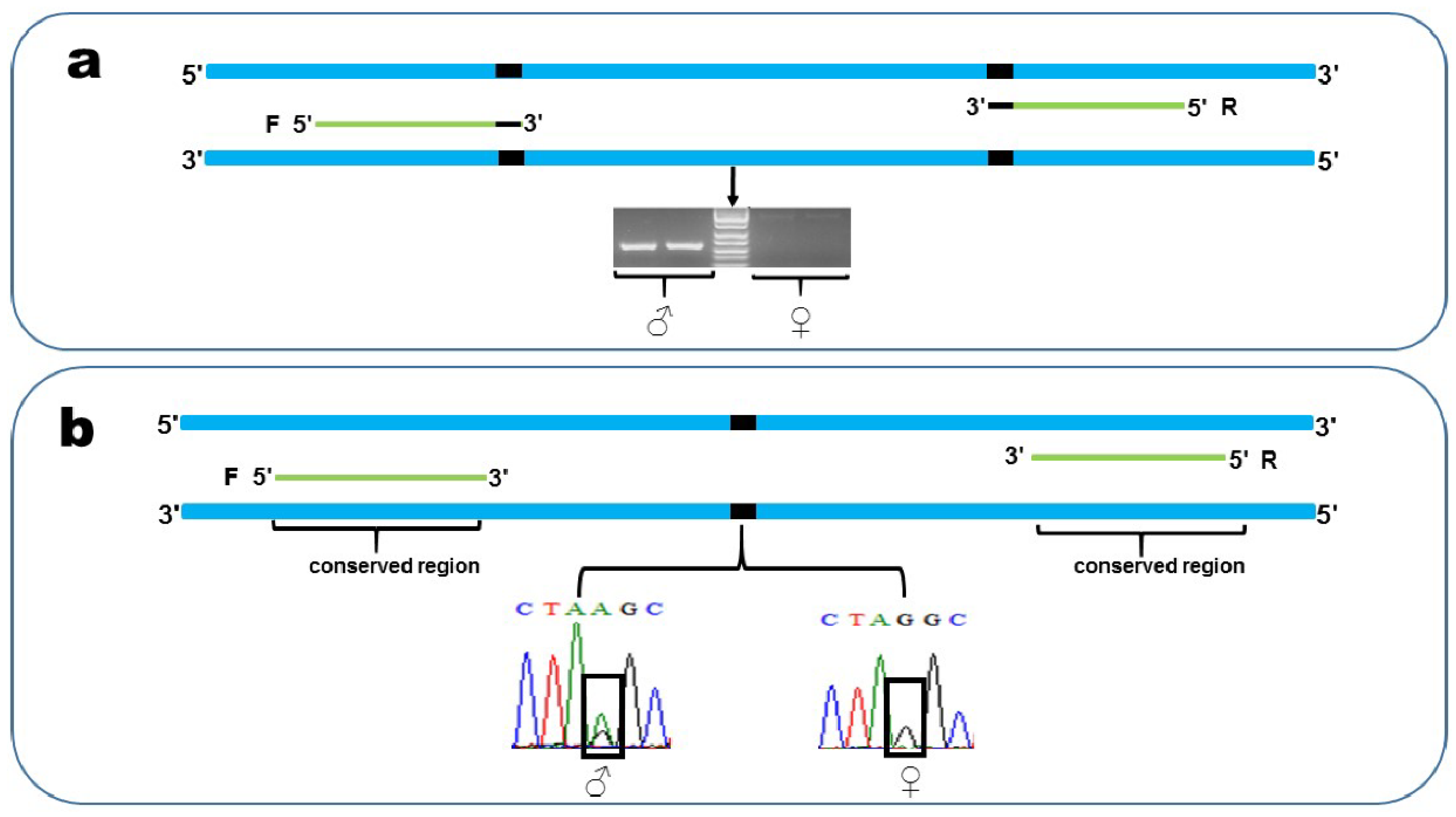
2.5. Validation of Sex-Linked Markers
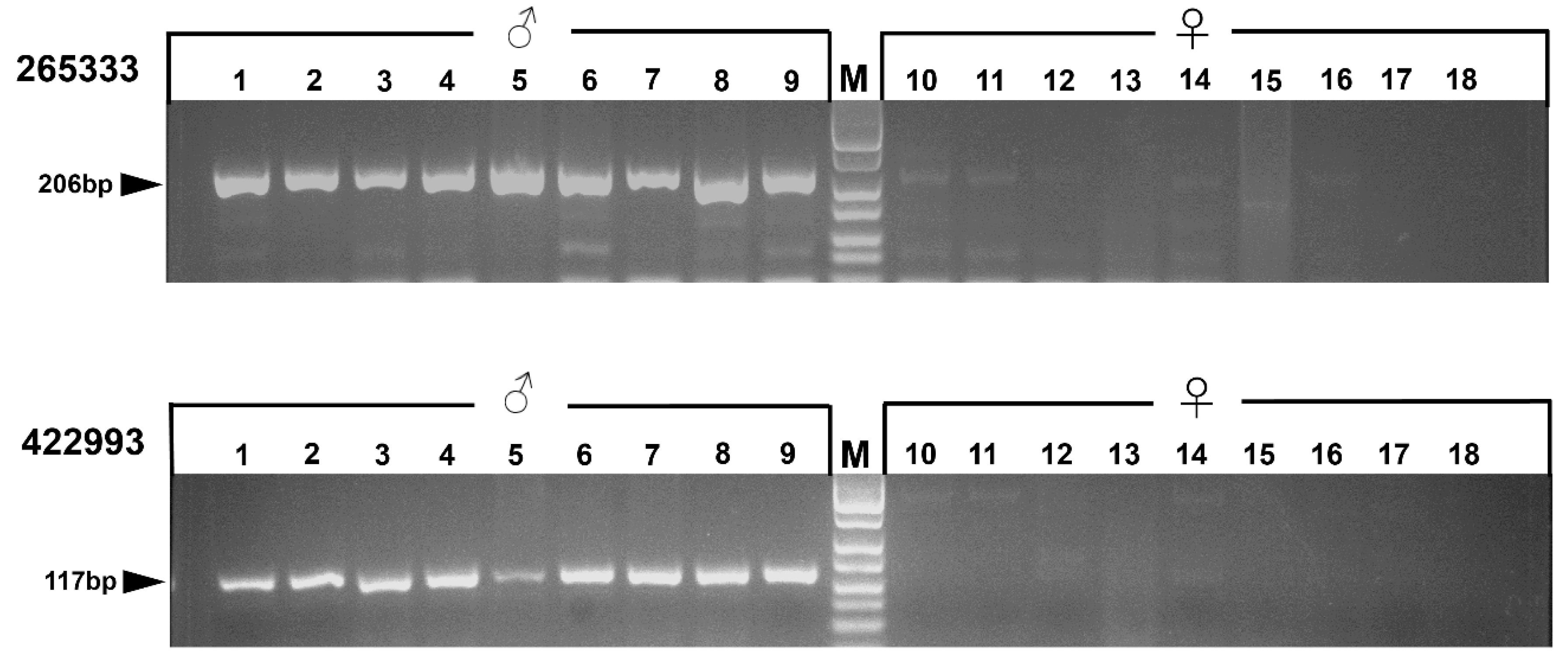
2.6. PCR Verification
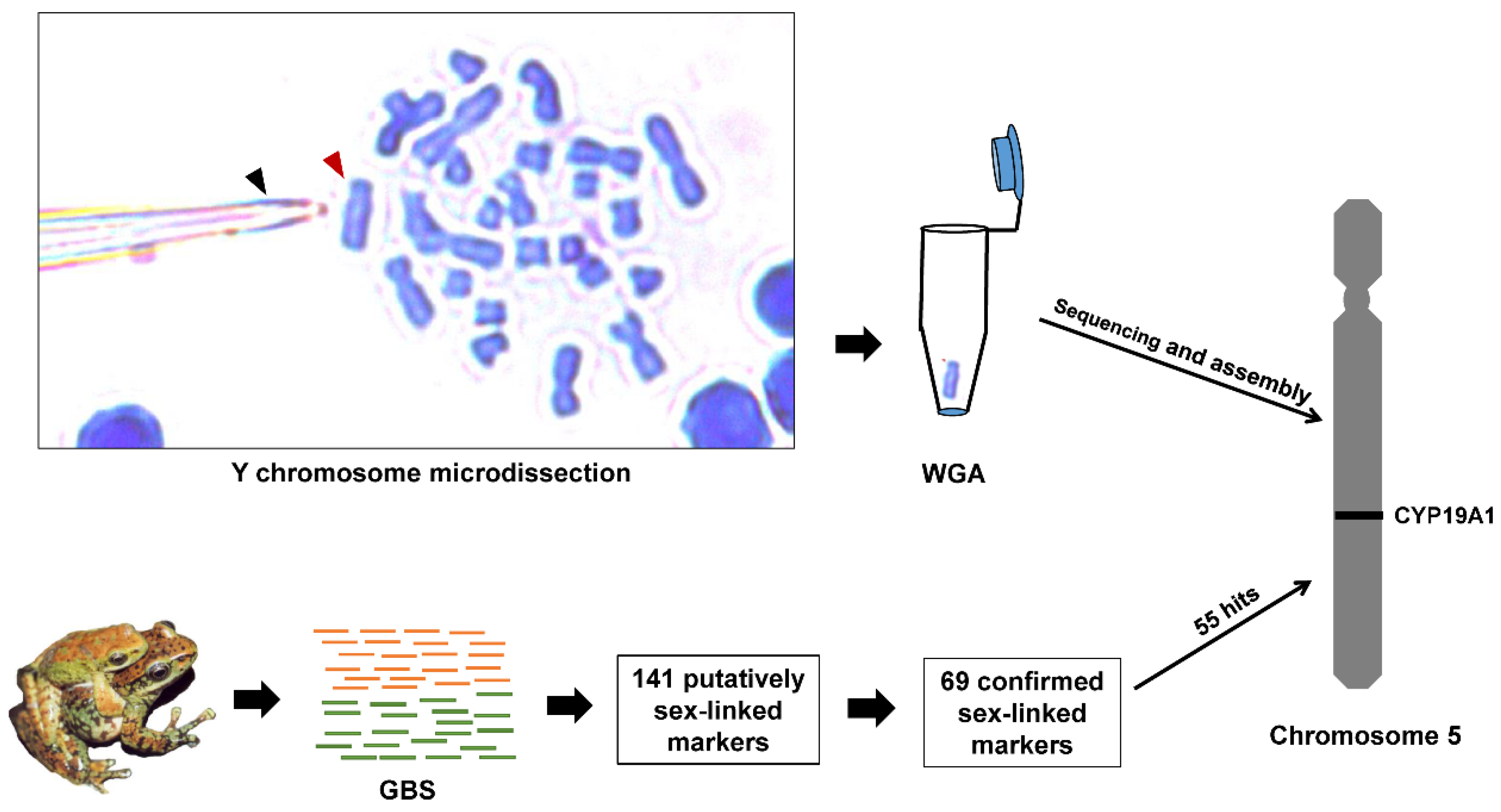
2.7. Y Chromosome Assembly and Sex-Linked Markers Assignment
2.8. Genes Involved in Sex-Determination or Sex Differentiation on Y Chromosome
3. Results
3.1. Genotyping-by-Sequencing
3.2. Sex-Specific Locus Discovery and Isolation
3.3. Validation of Sex-Linked Markers
3.4. PCR Verification
3.5. Y Chromosome Assembly and Sex-Linked Markers Assignment
3.6. Identifying Potential Sex-Determining Genes
4. Discussion
4.1. Chromosome 5 is the Sex Chromosome Pair
4.2. Particular Chromosomes Make Better Sex Chromosomes
4.3. Sex-Determination and Differentiation Genes Involved in A. Mantzorum
Supplementary Materials
Author Contributions
Acknowledgments
Data Availability
Conflicts of Interest
References
- Kashimada, K.; Koopman, P. Sry: The master switch in mammalian sex-determination. Development 2010, 137, 3921–3930. [Google Scholar] [CrossRef] [PubMed]
- Smith, C.A.; Roeszler, K.N.; Ohnesorg, T.; Cummins, D.M.; Farlie, P.G.; Doran, T.J.; Sinclair, A.H. The avian Z-linked gene DMRT1 is required for male sex-determination in the chicken. Nature 2009, 461, 267–271. [Google Scholar] [CrossRef] [PubMed]
- Hillis, D.M.; Green, D.M. Evolutionary changes of heterogametic sex in the phylogenetic history of amphibians. J. Evol. Biol. 1990, 3, 49–64. [Google Scholar] [CrossRef]
- Nakamura, M. Sex-determination in amphibians. Semin. Cell Dev. Biol. 2009, 20, 271–282. [Google Scholar] [CrossRef]
- Uno, Y.; Nishida, C.; Yoshimoto, S.; Ito, M.; Oshima, Y.; Yokoyama, S.; Nakamura, M.; Matsuda, Y. Diversity in the origins of sex chromosomes in anurans inferred from comparative mapping of sexual differentiation genes for three species of the Raninae and Xenopodinae. Chromosome Res. 2008, 16, 999–1011. [Google Scholar] [CrossRef]
- Uno, Y.; Nishida, C.; Takagi, C.; Igawa, T.; Ueno, N.; Sumida, M.; Matsuda, Y. Extraordinary Diversity in the Origins of Sex Chromosomes in Anurans Inferred from Comparative Gene Mapping. Cytogenet. Genome Res. 2015, 145, 218–229. [Google Scholar] [CrossRef]
- Marshall Graves, J.A.; Peichel, C.L. Are homologies in vertebrate sex-determination due to shared ancestry or to limited options? Genome Biol. 2010, 11, 205. [Google Scholar] [CrossRef]
- Miura, I. An evolutionary witness: The frog Rana rugosa underwent change of heterogametic sex from XY male to ZW female. Sex. Dev. 2007, 1, 323–331. [Google Scholar] [CrossRef]
- Miura, I. Sex-determination and Sex Chromosomes in Amphibia. Sex. Dev. 2017, 11, 298–306. [Google Scholar] [CrossRef]
- Bewick, A.J.; Anderson, D.W.; Evans, B.J. Evolution of the closely related, sex-related genes DM-W and DMRT1 in African clawed frogs (Xenopus). Evolution 2011, 65, 698–712. [Google Scholar] [CrossRef]
- Yoshimoto, S.; Okada, E.; Umemoto, H.; Tamura, K.; Uno, Y.; Nishida-Umehara, C.; Matsuda, Y.; Takamatsu, N.; Shiba, T.; Ito, M. A W-linked DM-domain gene, DM-W, participates in primary ovary development in Xenopus laevis. Proc. Natl. Acad. Sci. USA 2008, 105, 2469–2474. [Google Scholar] [CrossRef]
- Fei, L.; Hu, S.Q.; Ye, C.Y.; Huang, Y.Z. Amphibia (vol3), Anura ranidae. In Fauna Sinica; Science Press: Beijing, China, 2009; pp. 1–22. [Google Scholar]
- Wu, G.F.; Zhao, E.M. Two rare karyotypes in Anura-the karyotypes of Staurois mantzorum and Staurois liangshanensis. Acta Herpetol. Sin. 1984, 3, 4–9. [Google Scholar]
- Schmid, M.; Steinlein, C.; Bogart, J.P.; Feichtinger, W.; León, P.; La Marca, E.; Diaz, L.M.; Sanz, A.; Chen, S.H.; Hedges, S.B. The Chromosomes of Terraranan Frogs. Insights into Vertebrate Cytogenetics. Cytogenet. Genome Res. 2010, 130–131, 1–14. [Google Scholar] [CrossRef]
- Elshire, R.J.; Glaubitz, J.C.; Sun, Q.; Poland, J.A.; Kawamoto, K.; Buckler, E.S.; Mitchell, S.E. A Robust, Simple Genotyping-by-Sequencing (GBS) Approach for High Diversity Species. PLoS ONE 2011, 6, e19379. [Google Scholar] [CrossRef] [PubMed]
- Catchen, J.M.; Amores, A.; Hohenlohe, P.; Cresko, W.; Postlethwait, J.H. Stacks: Building and Genotyping Loci De Novo From Short-Read Sequences. G3 Genes Genomes Genet. 2011, 1, 171–182. [Google Scholar] [CrossRef] [PubMed]
- Rochette, N.C.; Catchen, J.M. Deriving genotypes from RAD-seq short-read data using Stacks. Nat. Protoc. 2017, 12, 2640–2659. [Google Scholar] [CrossRef]
- Brelsford, A.; Lavanchy, G.; Sermier, R.; Rausch, A.; Perrin, N. Identifying homomorphic sex chromosomes from wild-caught adults with limited genomic resources. Mol. Ecol. Resour. 2017, 17, 752–759. [Google Scholar] [CrossRef] [PubMed]
- Fowler, B.L.; Buonaccorsi, V.P. Genomic characterization of sex-identification markers in Sebastes carnatus and Sebastes chrysomelas rockfishes. Mol. Ecol. 2016, 25, 2165–2175. [Google Scholar] [CrossRef]
- Xia, Y.; Luo, W.; Yuan, S.; Zheng, Y.; Zeng, X. Microsatellite development from genome skimming and transcriptome sequencing: Comparison of strategies and lessons from frog species. BMC Genom. 2018, 19, 886. [Google Scholar] [CrossRef]
- Zhang, Z.; Schwartz, S.; Wagner, L.; Miller, W. A greedy algorithm for aligning DNA sequences. J. Comput. Biol. 2000, 7, 203–214. [Google Scholar] [CrossRef]
- Liu, Y.; Song, M.; Luo, W.; Xia, Y.; Zeng, X. Chromosomal Evolution in the Amolops mantzorum Species Group (Ranidae; Anura) Narrated by Repetitive DNAs. Cytogenet. Genome Res. 2019, 157, 172–178. [Google Scholar] [CrossRef] [PubMed]
- Liu, T.; Song, M.; Xia, Y.; Zeng, X. A tandemly arranged pattern of two 5S rDNA arrays in Amolops mantzorum (Anura, Ranidae). Cytogenet. Genome Res. 2017, 151, 161–170. [Google Scholar] [CrossRef] [PubMed]
- Zimmer, R.; King, W.A.; Verrinder Gibbins, A.M. Generation of chicken Z-chromosome painting probes by microdissection for screening large-insert genomic libraries. Cytogenet. Cell Genet. 1997, 78, 124–130. [Google Scholar] [CrossRef] [PubMed]
- Yuan, X.; Yuan, S.; Liu, Y.; Xia, Y.; Zeng, X. Microsatellites mapping for non-model species with chromosomal rearrangement: A case study in the frog Quasipaa boulengeri (Anura: Dicroglossidae). Genome 2017, 60, 707–711. [Google Scholar] [CrossRef] [PubMed]
- Bolger, A.M.; Lohse, M.; Usadel, B. Trimmomatic: A flexible trimmer for Illumina sequence data. Bioinformatics 2014, 30, 2114–2120. [Google Scholar] [CrossRef] [PubMed]
- Luo, R.; Liu, B.; Xie, Y.; Li, Z.; Huang, W.; Yuan, J.; He, G.; Chen, Y.; Pan, Q.; Liu, Y. SOAPdenovo2: An empirically improved memory-efficient short-read de novo assembler. Gigascience 2012, 1, 18. [Google Scholar] [CrossRef] [PubMed]
- Wu, Z.; Tan, A.; Zhao, E. Cytological evidence on four species of Amolops in the Hengduan Range. Acta Genet. Sin. 1987, 14, 63–68. [Google Scholar]
- Volff, J.N.; Nanda, I.; Schmid, M.; Schartl, M. Governing Sex-determination in Fish: Regulatory Putsches and Ephemeral Dictators. Sex. Dev. 2007, 1, 85–99. [Google Scholar] [CrossRef]
- Ezaz, T.; Stiglec, R.; Veyrunes, F.; Marshall Graves, J.A. Relationships between vertebrate ZW and XY sex chromosome systems. Curr. Biol. 2006, 16, R736–R743. [Google Scholar] [CrossRef]
- Jeffries, D.L.; Lavanchy, G.; Sermier, R.; Sredl, M.J.; Miura, I.; Borzee, A.; Barrow, L.N.; Canestrelli, D.; Crochet, P.A.; Dufresnes, C.; et al. A rapid rate of sex-chromosome turnover and non-random transitions in true frogs. Nat. Commun. 2018, 9, 4088. [Google Scholar] [CrossRef]
- Pyron, R.A.; Wiens, J.J. A large-scale phylogeny of Amphibia including over 2800 species, and a revised classification of extant frogs, salamanders, and caecilians. Mol. Phylogenet. Evol. 2011, 61, 543–583. [Google Scholar] [CrossRef] [PubMed]
- Ohtani, H.; Miura, I.; Ichikawa, Y. Role of aromatase and androgen receptor expression in gonadal sex differentiation of ZW/ZZ-type frogs, Rana rugosa. Comp. Biochem. Physiol. C-Toxicol. 2003, 134, 215–225. [Google Scholar] [CrossRef]
- Crews, D. Sex-determination: Where environment and genetics meet. Evol. Dev. 2003, 5, 50–55. [Google Scholar] [CrossRef] [PubMed]
- Abinawanto; Shimada, K.; Yoshida, K.; Saito, N. Effects of Aromatase Inhibitor on Sex Differentiation and Levels of P45017α and P450 arom Messenger Ribonucleic Acid of Gonads in Chicken Embryos. Gen. Comp. Endocrinol. 1996, 102, 241–246. [Google Scholar] [CrossRef]
- Kato, T.; Matsui, K.; Takase, M.; Kobayashi, M.; Nakamura, M. Expression of P450 aromatase protein in developing and in sex-reversed gonads of the XX/XY type of the frog Rana rugosa. Gen. Comp. Endocrinol. 2004, 137, 227–236. [Google Scholar] [CrossRef]
- Maruo, K.; Suda, M.; Yokoyama, S.; Oshima, Y.; Nakamura, M. Steroidogenic gene expression during sex-determination in the frog Rana rugosa. Gen. Comp. Endocrinol. 2008, 158, 87–94. [Google Scholar] [CrossRef]
- Mawaribuchi, S.; Ikeda, N.; Fujitani, K.; Ito, Y.; Onuma, Y.; Komiya, T.; Takamatsu, N.; Ito, M. Cell-Mass Structures Expressing the Aromatase Gene Cyp19a1 Lead to Ovarian Cavities in Xenopus laevis. Endocrinology 2014, 155, 3996–4005. [Google Scholar] [CrossRef]
- Ramsey, M.; Shoemaker, C.; Crews, D. Gonadal expression of Sf1 and aromatase during sex-determination in the red-eared slider turtle (Trachemys scripta), a reptile with temperature-dependent sex-determination. Differentiation 2007, 75, 978–991. [Google Scholar] [CrossRef]
- Matsumoto, Y.; Buemio, A.; Chu, R.; Vafaee, M.; Crews, D. Epigenetic control of gonadal aromatase (cyp19a1) in temperature-dependent sex-determination of red-eared slider turtles. PLoS ONE 2013, 8, e63599. [Google Scholar] [CrossRef]
- Matsumoto, Y.; Hannigan, B.; Crews, D. Temperature Shift Alters DNA Methylation and Histone Modification Patterns in Gonadal Aromatase (cyp19a1) Gene in Species with Temperature-Dependent Sex-determination. PLoS ONE 2016, 11, e0167362. [Google Scholar] [CrossRef]
- Navarro-Martín, L.; Viñas, J.; Ribas, L.; Díaz, N.; Gutiérrez, A.; Di Croce, L.; Piferrer, F. DNA Methylation of the Gonadal Aromatase (cyp19a) Promoter Is Involved in Temperature-Dependent Sex Ratio Shifts in the European Sea Bass. PLoS Genet. 2011, 7, e1002447. [Google Scholar] [CrossRef] [PubMed]
- Yokoyama, S.; Oshima, Y.; Tokita, J.; Suda, M.; Shinozuka, T.; Nakamura, M. Androgen receptor of the frog Rana rugosa: Molecular cloning and its characterization. J. Exp. Zool. Part A Ecol. Genet. Physiol. 2009, 311A, 796–812. [Google Scholar] [CrossRef] [PubMed]
- Fujii, J.; Kodama, M.; Oike, A.; Matsuo, Y.; Min, M.-S.; Hasebe, T.; Ishizuya-Oka, A.; Kawakami, K.; Nakamura, M. Involvement of Androgen Receptor in Sex-determination in an Amphibian Species. PLoS ONE 2014, 9, e93655. [Google Scholar] [CrossRef] [PubMed]
| Locus | Primer sequence (5′–3′) | T (°C) | Length (bp) |
|---|---|---|---|
| 265333 * | Forward: GTGCTGCCTCTCGCTTCCCGAT | 55 | 117 |
| Reverse: AAATGGTCTAAGCTCGGGGTCG | |||
| 422993 * | Forward: TAAAGGCACGGTGTTCG | 55 | 206 |
| Reverse: TTCCTCCTGACATAAGAGCTA | |||
| 3662 | Forward: TCGACGTGAGCCTTAGTCAT | 55 | 258 |
| Reverse: AAAGCAATTCAAACGAGCAT | |||
| 37926 | Forward: TTGATGGTGAATTGATTGGG | 55 | 200 |
| Reverse: TCACAAATTCCAGGTGCTTG | |||
| 52538 | Forward: CTCCTGGCAACTCCTTTCTG | 55 | 189 |
| Reverse: GAGCCCATATTCAATTCACC | |||
| 116241 | Forward: GTGTCAGCACAAGAGGTAGG | 55 | 214 |
| Reverse: ATCTGGTATCATCCGAGAAA | |||
| 144173 | Forward: AAAGCAATTCAAACGAGCAT | 55 | 256 |
| Reverse: CGACGTGAGCCTTAGTCATAG | |||
| 181302 | Forward: GGGACAGAGTGAGGCTCGCTAA | 55 | 160 |
| Reverse: CTGAGGATATGCAATCCCGTTG |
| Locus | Length (bp) | NCBI Hit | Query Cover | E-Value | Identity | Start | End |
|---|---|---|---|---|---|---|---|
| 174,608 | 283 | Rana rugosa CYP19A1 gene for cytochrome P-450 aromatase, promoter region and partial cds (AB379847.1) | 97% | 2e−49 | 81.72% | 12,255 | 12,505 |
| 201,126 | 280 | 95% | 2e−53 | 81.82% | 11,730 | 11,997 | |
| 408,566 | 299 | 21% | 2e−13 | 92.06% | 14,151 | 14,212 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Luo, W.; Xia, Y.; Yue, B.; Zeng, X. Assigning the Sex-Specific Markers via Genotyping-by-Sequencing onto the Y Chromosome for a Torrent Frog Amolops mantzorum. Genes 2020, 11, 727. https://doi.org/10.3390/genes11070727
Luo W, Xia Y, Yue B, Zeng X. Assigning the Sex-Specific Markers via Genotyping-by-Sequencing onto the Y Chromosome for a Torrent Frog Amolops mantzorum. Genes. 2020; 11(7):727. https://doi.org/10.3390/genes11070727
Chicago/Turabian StyleLuo, Wei, Yun Xia, Bisong Yue, and Xiaomao Zeng. 2020. "Assigning the Sex-Specific Markers via Genotyping-by-Sequencing onto the Y Chromosome for a Torrent Frog Amolops mantzorum" Genes 11, no. 7: 727. https://doi.org/10.3390/genes11070727
APA StyleLuo, W., Xia, Y., Yue, B., & Zeng, X. (2020). Assigning the Sex-Specific Markers via Genotyping-by-Sequencing onto the Y Chromosome for a Torrent Frog Amolops mantzorum. Genes, 11(7), 727. https://doi.org/10.3390/genes11070727





