Transcription Analysis of the Chemerin Impact on Gene Expression Profile in the Luteal Cells of Gilts
Abstract
1. Introduction
2. Materials and Methods
2.1. Experimental Animals and Tissue Collection
2.2. Isolation of the Luteal Cells and in Vitro Cell Cultures
2.3. RNA Isolation and High-Throughput Sequencing
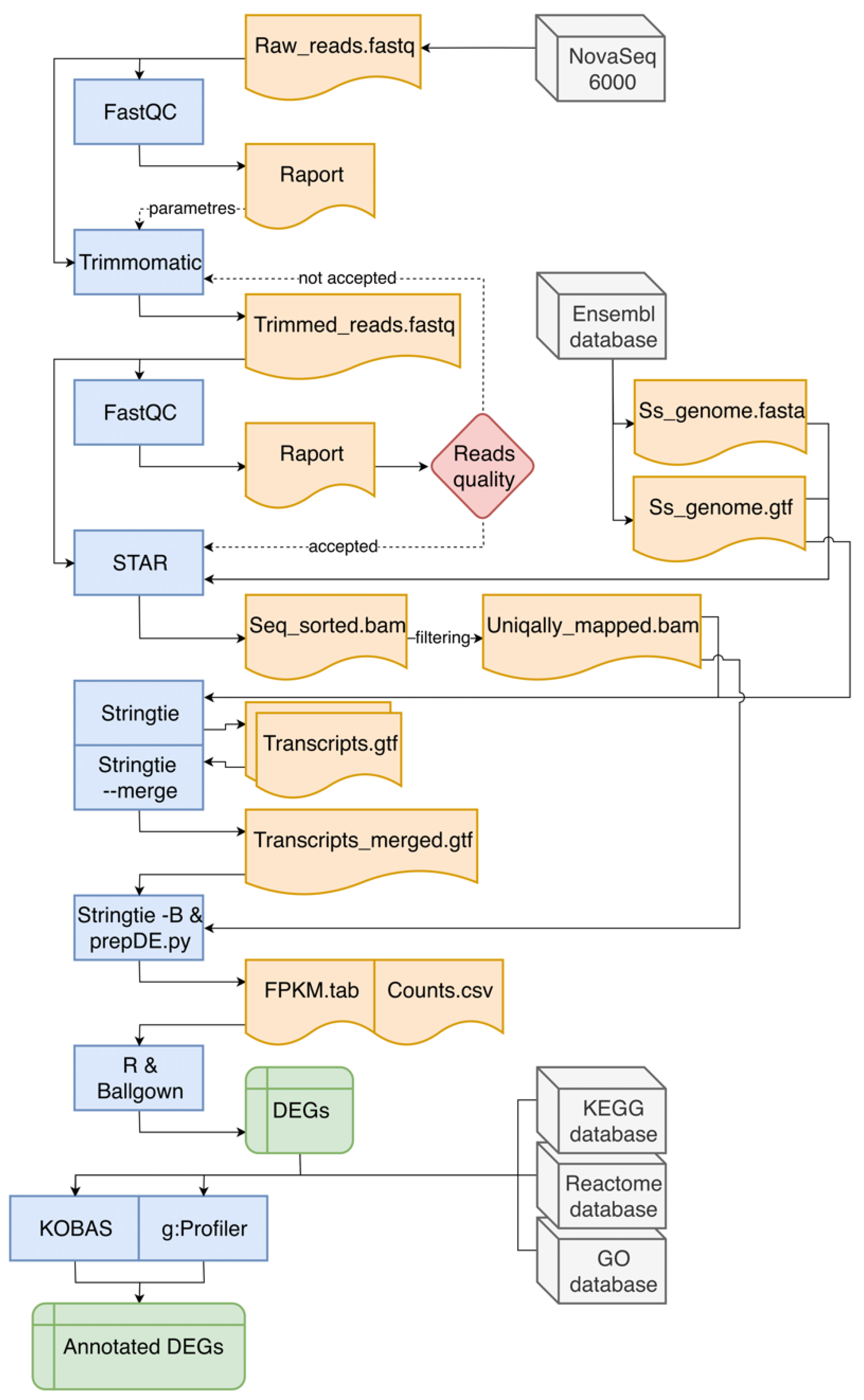
2.4. In Silico Analyses
2.4.1. Row Reads Pre-Processing and Differentially Expressed Genes Processing
2.4.2. Functional Annotation of Differentially Expressed Genes
2.5. Quantitative Real-Time PCR Validations
3. Results
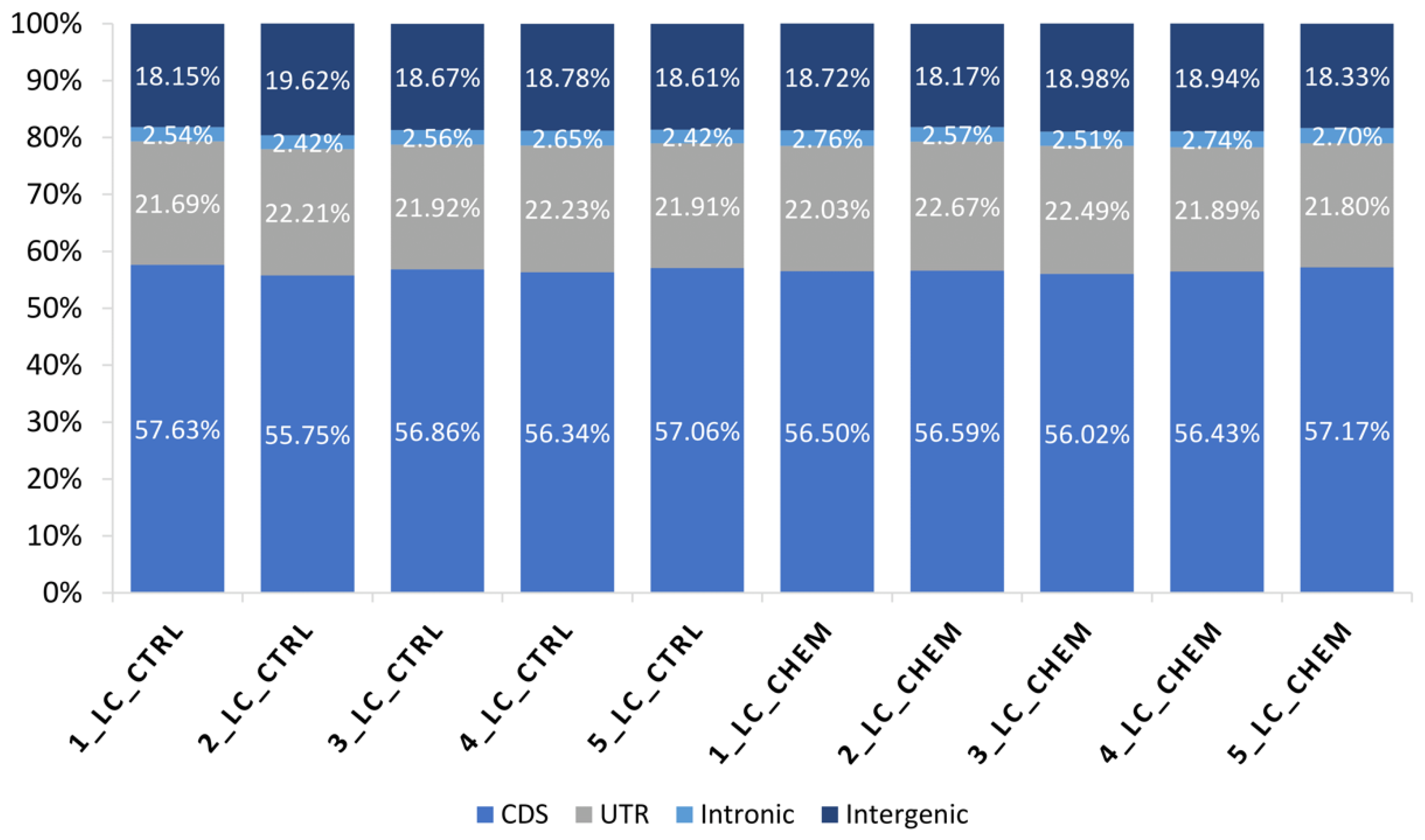
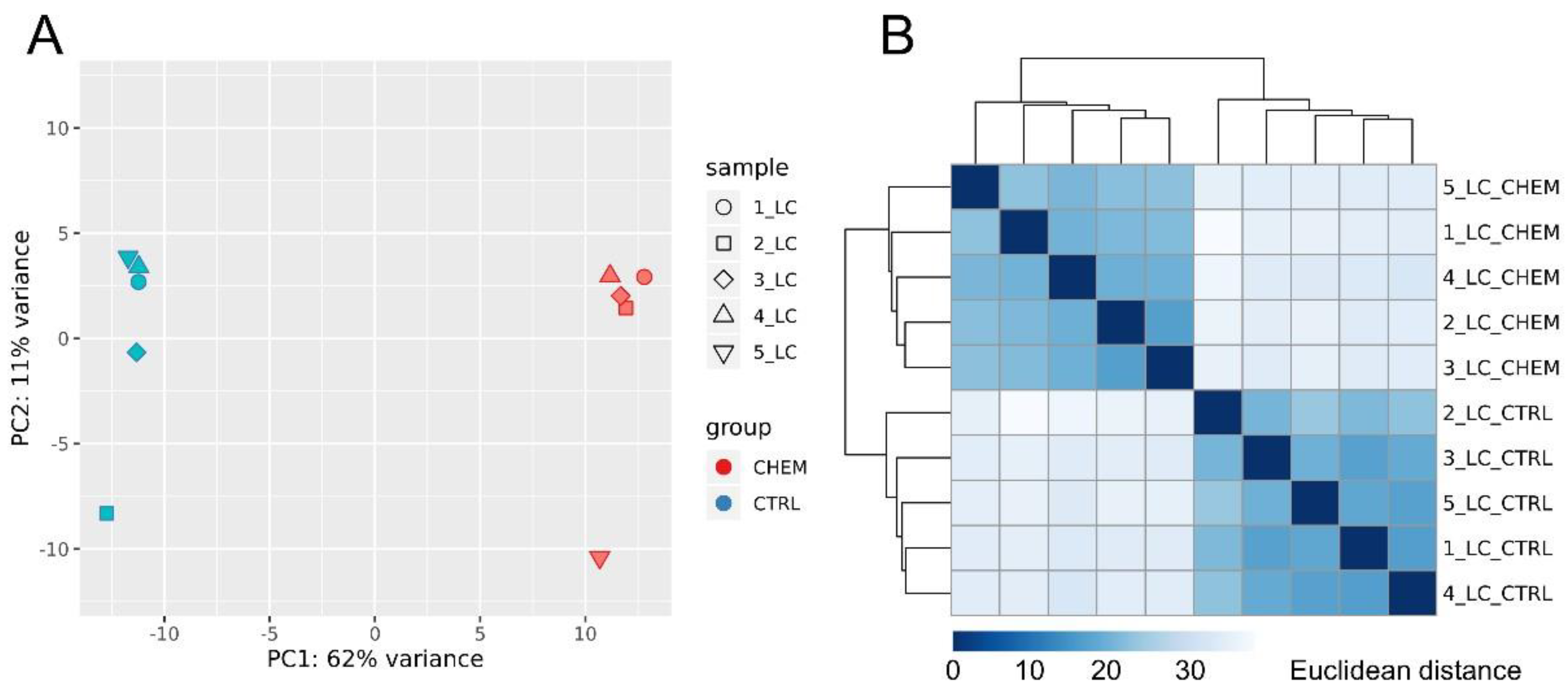
3.1. Overall Statistics of RNA-Seq Data and Mapping Results
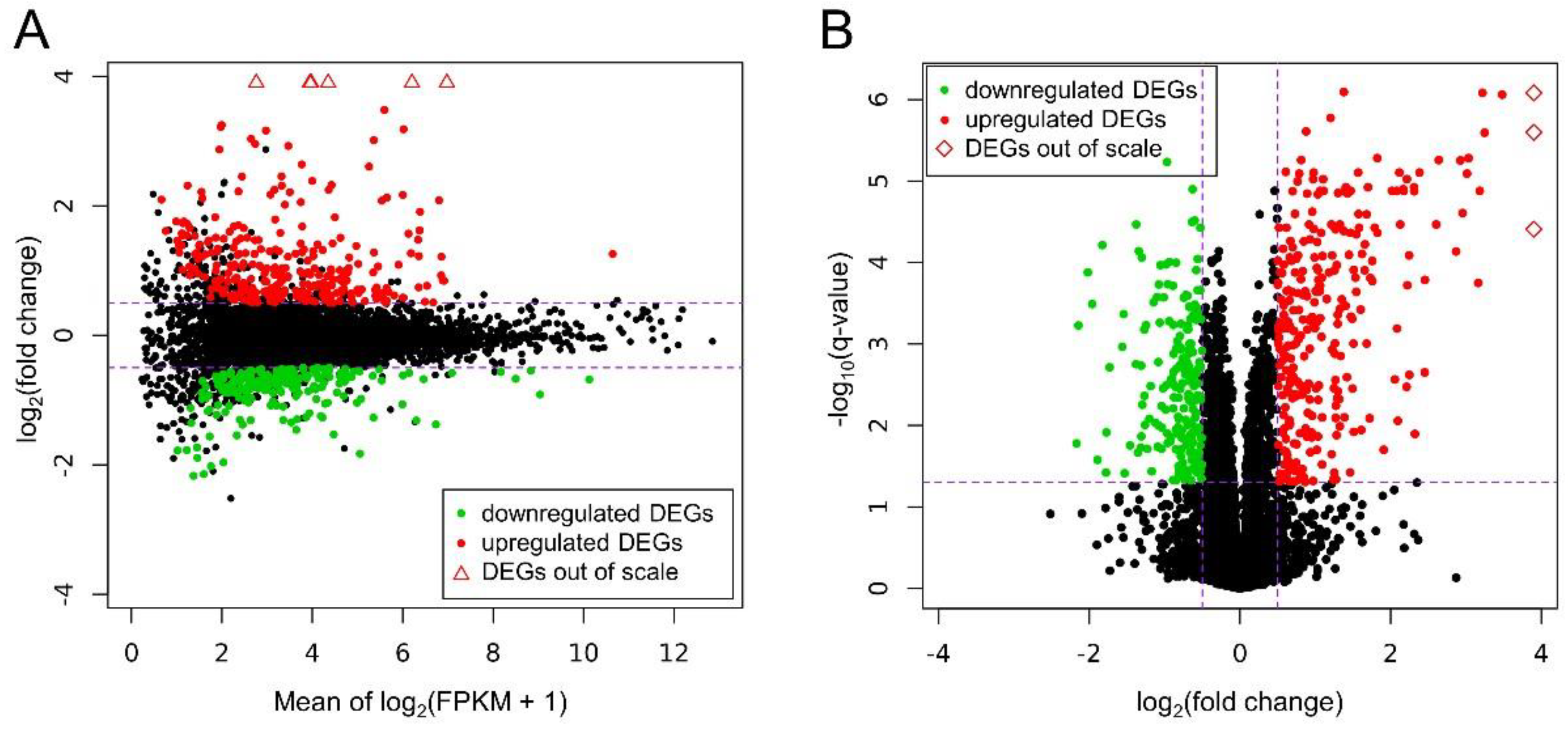
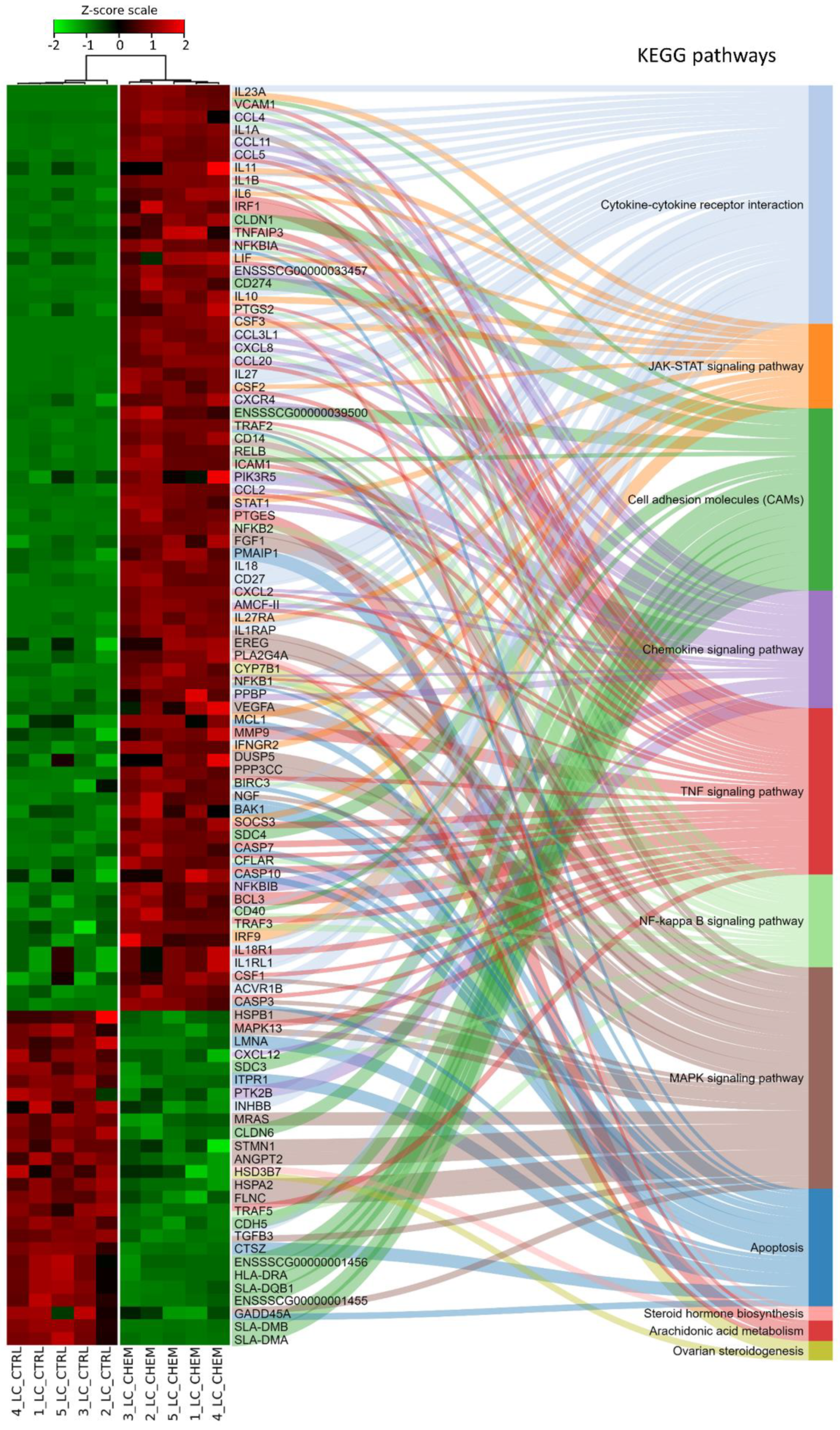
3.2. Differentially Expressed Genes
3.3. Functional Genes Analysis
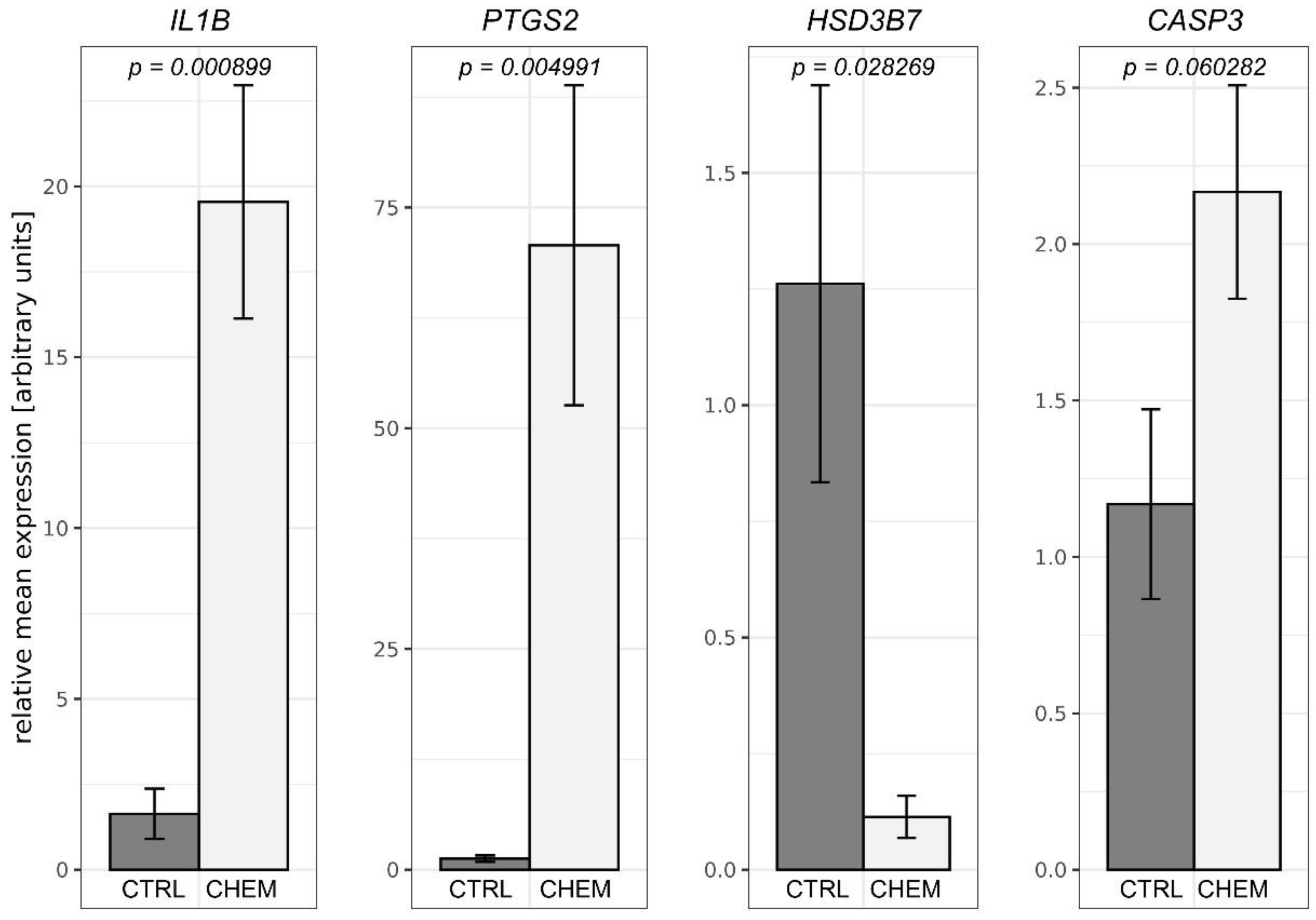
3.4. Quantitative Real-Time PCR Validations
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Nagpal, S.; Patel, S.; Jacobe, H.; Disepio, D.; Ghosn, C.; Malhotra, M.; Teng, M.; Duvic, M.; Chandraratna, R.A. Tazarotene-induced Gene 2 (TIG2), a Novel Retinoid-Responsive Gene in Skin. J. Investig. Dermatol. 1997, 109, 91–95. [Google Scholar] [CrossRef] [PubMed]
- Reverchon, M.; Cornuau, M.; Rame, C.; Guerif, F.; Royère, D.; Dupont, J. Chemerin inhibits IGF-1-induced progesterone and estradiol secretion in human granulosa cells. Hum. Reprod. 2012, 27, 1790–1800. [Google Scholar] [CrossRef] [PubMed]
- Goralski, K.B.; McCarthy, T.C.; Hanniman, E.A.; Zabel, B.A.; Butcher, E.C.; Parlee, S.D.; Muruganandan, S.; Sinal, C.J. Chemerin, a Novel Adipokine That Regulates Adipogenesis and Adipocyte Metabolism. J. Boil. Chem. 2007, 282, 28175–28188. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Kim, J.Y.; Xue, K.; Liu, J.-Y.; Leader, A.; Tsang, B.K. Chemerin, a Novel Regulator of Follicular Steroidogenesis and Its Potential Involvement in Polycystic Ovarian Syndrome. Endocrinology 2012, 153, 5600–5611. [Google Scholar] [CrossRef] [PubMed]
- Du, X.-Y.; Leung, L.L. Proteolytic regulatory mechanism of chemerin bioactivity. Acta Biochim. Biophys. Sin. 2009, 41, 973–979. [Google Scholar] [CrossRef]
- De Henau, O.; De Groot, G.-N.; Imbault, V.; Robert, V.; De Poorter, C.; Mcheik, S.; Gales, C.; Parmentier, M.; Springael, J.-Y. Signaling Properties of Chemerin Receptors CMKLR1, GPR1 and CCRL2. PLoS ONE 2016, 11, e0164179. [Google Scholar] [CrossRef]
- Yang, Y.-L.; Ren, L.-R.; Sun, L.-F.; Huang, C.; Xiao, T.-X.; Wang, B.-B.; Chen, J.; Zabel, B.A.; Ren, P.; Zhang, J. The role of GPR1 signaling in mice corpus luteum. J. Endocrinol. 2016, 230, 55–65. [Google Scholar] [CrossRef][Green Version]
- Monnier, J.; Lewén, S.; O’Hara, E.; Huang, K.; Tu, H.; Butcher, E.C.; Zabel, B.A. Expression, regulation, and function of atypical chemerin receptor CCRL2 on endothelial cells. J. Immunol. 2012, 189, 956–967. [Google Scholar] [CrossRef]
- Mazzotti, C.; Gagliostro, V.; Bosisio, D.; Del Prete, A.; Tiberio, L.; Thelen, M.; Sozzani, S. The Atypical Receptor CCRL2 (C-C Chemokine Receptor-Like 2) Does Not Act as a Decoy Receptor in Endothelial Cells. Front. Immunol. 2017, 8. [Google Scholar] [CrossRef]
- Mazaki-Tovi, S.; Kasher-Meron, M.; Hemi, R.; Haas, J.; Gat, I.; Lantsberg, D.; Hendler, I.; Kanety, H. Chemerin is present in human cord blood and is positively correlated with birthweight. Am. J. Obstet. Gynecol. 2012, 207, 412.e1–412.e10. [Google Scholar] [CrossRef]
- Garces, M.; Sánchez, E.; Acosta, B.; Angel, E.; Ruiz, A.; Rubio-Romero, J.; Diéguez, C.; Nogueiras, R.; Caminos, J. Expression and regulation of chemerin during rat pregnancy. Placenta 2012, 33, 373–378. [Google Scholar] [CrossRef] [PubMed]
- Reverchon, M.; Bertoldo, M.J.; Ramé, C.; Froment, P.; Dupont, J. CHEMERIN (RARRES2) Decreases In Vitro Granulosa Cell Steroidogenesis and Blocks Oocyte Meiotic Progression in Bovine Species1. Boil. Reprod. 2014, 90, 102. [Google Scholar] [CrossRef] [PubMed]
- Margioris, A.N.; Katrinaki, M.; Tsatsanis, C.; Giwercman, A.; Bobjer, J.; Dermitzaki, E.; Katrinaki, M.; Dermitzaki, E.; Margioris, A.N.; Giwercman, A.; et al. Serum chemerin levels are negatively associated with male fertility and reproductive hormones. Hum. Reprod. 2018, 33, 2168–2174. [Google Scholar]
- Yao, J.; Li, Z.; Fu, Y.; Wu, R.; Wang, Y.; Liu, C.; Yang, L.; Zhang, H.-W. Involvement of obesity-associated upregulation of chemerin/chemokine-like receptor 1 in oxidative stress and apoptosis in ovaries and granulosa cells. Biochem. Biophys. Res. Commun. 2019, 510, 449–455. [Google Scholar] [CrossRef]
- Kim, J.Y.; Xue, K.; Cao, M.; Wang, Q.; Liu, J.-Y.; Leader, A.; Han, J.Y.; Tsang, B.K. Chemerin Suppresses Ovarian Follicular Development and Its Potential Involvement in Follicular Arrest in Rats Treated Chronically With Dihydrotestosterone. Endocrinology 2013, 154, 2912–2923. [Google Scholar] [CrossRef]
- Smolinska, N.; Kiezun, M.; Dobrzyn, K.; Rytelewska, E.; Kisielewska, K.; Gudelska, M.; Zaobidna, E.; Bogus-Nowakowska, K.; Wyrebek, J.; Bors, K.; et al. Expression of Chemerin and Its Receptors in the Porcine Hypothalamus and Plasma Chemerin Levels during the Oestrous Cycle and Early Pregnancy. Int. J. Mol. Sci. 2019, 20, 3887. [Google Scholar] [CrossRef]
- Rytelewska, E.; Kisielewska, K.; Kiezun, M.; Dobrzyn, K.; Gudelska, M.; Rak-Mardyla, A.; Dupont, J.; Kaminska, B.; Kaminski, T.; Smolinska, N. Expression of chemerin and its receptors in the ovaries of prepubertal and mature gilts. Mol. Reprod. Dev. 2020. accepted. [Google Scholar] [CrossRef]
- Akins, E.L.; Morrissette, M.C. Gross ovarian changes during estrous cycle of swine. Am. J. Veter Res. 1968, 29. [Google Scholar]
- Kaminski, T.; Siawrys, G.; Okrasa, S.; Przala, J. Action of the opioid agonist FK 33-824 on porcine small and large luteal cells from the mid-luteal phase: Effect on progesterone, cAMP, cGMP and inositol phosphate release. Anim. Reprod. Sci. 1999, 56, 245–257. [Google Scholar] [CrossRef]
- Bozaoglu, K.; Curran, J.E.; Stocker, C.J.; Zaibi, M.S.; Segal, D.; Konstantopoulos, N.; Morrison, S.; Carless, M.; Dyer, T.D.; Cole, S.A.; et al. Chemerin, a novel adipokine in the regulation of angiogenesis. J. Clin. Endocrinol. Metab. 2010, 95, 2476–2485. [Google Scholar] [CrossRef]
- Shen, Y.; Mao, H.; Huang, M.; Chen, L.; Chen, J.; Cai, Z.; Wang, Y.; Xu, N. Long Noncoding RNA and mRNA Expression Profiles in the Thyroid Gland of Two Phenotypically Extreme Pig Breeds Using Ribo-Zero RNA Sequencing. Genes 2016, 7, 34. [Google Scholar] [CrossRef] [PubMed]
- Pertea, M.; Kim, D.; Pertea, G.M.; Leek, J.T.; Salzberg, S.L. Transcript-level expression analysis of RNA-seq experiments with HISAT, StringTie and Ballgown. Nat. Protoc. 2016, 11, 1650–1667. [Google Scholar] [CrossRef] [PubMed]
- Andrews, S. FastQC: A Quality Control Tool for High Throughput Sequence Data. Available online: http://www.bioinformatics.babraham.ac.uk/projects/fastqc (accessed on 10 June 2020).
- Bolger, A.M.; Lohse, M.; Usadel, B. Trimmomatic: A flexible trimmer for Illumina sequence data. Bioinformatics 2014, 30, 2114–2120. [Google Scholar] [CrossRef] [PubMed]
- Dobin, A.; Davis, C.A.; Schlesinger, F.; Drenkow, J.; Zaleski, C.; Jha, S.; Batut, P.; Chaisson, M.; Gingeras, T.R. STAR: Ultrafast universal RNA-seq aligner. Bioinformatics 2012, 29, 15–21. [Google Scholar] [CrossRef]
- Zerbino, D.R.; Achuthan, P.; Akanni, W.; Amode, M.R.; Barrell, D.; Bhai, J.; Billis, K.; Cummins, C.; Gall, A.; Girón, C.G.; et al. Ensembl 2018. Nucleic Acids Res. 2018, 46, 754–761. [Google Scholar] [CrossRef]
- Broad Institute. Picard Tools. Available online: http://broadinstitute.github.io/picard/ (accessed on 10 June 2020).
- Wickham, H. ggplot2: Elegant Graphics for Data Analysis; Springer: New York, NY, USA, 2016; ISBN 978-3-319-24277-4. [Google Scholar]
- Paukszto, L.; Mikolajczyk, A.; Szeszko, K.; Smolinska, N.; Jastrzebski, J.P.; Kaminski, T. Transcription analysis of the response of the porcine adrenal cortex to a single subclinical dose of lipopolysaccharide from Salmonella Enteritidis. Int. J. Boil. Macromol. 2019, 141, 1228–1245. [Google Scholar] [CrossRef]
- Pertea, M.; Pertea, G.M.; Antonescu, C.M.; Chang, T.-C.; Mendell, J.T.; Salzberg, S.L. StringTie enables improved reconstruction of a transcriptome from RNA-seq reads. Nat. Biotechnol. 2015, 33, 290–295. [Google Scholar] [CrossRef]
- Frazee, A.C.; Pertea, G.; Jaffe, A.E.; Langmead, B.; Salzberg, S.L.; Leek, J.T. Flexible analysis of transcriptome assemblies with Ballgown. BioRxiv 2014, 003665. [Google Scholar]
- Ramos, M.; Schiffer, L.; Re, A.; Azhar, R.; Basunia, A.; Rodriguez, C.; Chan, T.; Chapman, P.; Davis, S.R.; Gomez-Cabrero, D.; et al. Software for the Integration of Multiomics Experiments in Bioconductor. Cancer Res. 2017, 77, e39–e42. [Google Scholar] [CrossRef]
- Ihaka, R.; Gentleman, R.R. A Language for Data Analysis and Graphics. J. Comput. Graph. Stat. 1996, 5, 299–314. [Google Scholar]
- Reimand, J.; Arak, T.; Adler, P.; Kolberg, L.; Reisberg, S.; Peterson, H.; Vilo, J. g: Profiler-a web server for functional interpretation of gene lists (2016 update). Nucleic Acids Res. 2016, 44, 83–89. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Mao, X.; Cai, T.; Luo, J.; Wei, L. KOBAS server: A web-based platform for automated annotation and pathway identification. Nucleic Acids Res. 2006, 34, 720–724. [Google Scholar] [CrossRef] [PubMed]
- Kanehisa, M.; Furumichi, M.; Tanabe, M.; Sato, Y.; Morishima, K. KEGG: New perspectives on genomes, pathways, diseases and drugs. Nucleic Acids Res. 2016, 45, 353–361. [Google Scholar] [CrossRef] [PubMed]
- Ashburner, M.; Ball, C.A.; A Blake, J.; Botstein, D.; Butler, H.; Cherry, J.M.; Davis, A.P.; Dolinski, K.; Dwight, S.S.; Eppig, J.T.; et al. Gene Ontology: Tool for the unification of biology. Nat. Genet. 2000, 25, 25–29. [Google Scholar] [CrossRef] [PubMed]
- The Gene Ontology Consortium. Expansion of the Gene Ontology knowledgebase and resources. Nucleic Acids Res. 2016, 45, D331–D338. [Google Scholar] [CrossRef]
- Fabregat, A.; Jupe, S.; Matthews, L.; Sidiropoulos, K.; Gillespie, M.; Garapati, P.; Haw, R.; Jassal, B.; Korninger, F.; May, B.; et al. The Reactome Pathway Knowledgebase. Nucleic Acids Res. 2018, 46, D649–D655. [Google Scholar] [CrossRef]
- Livak, K.J.; Schmittgen, T.D. Analysis of Relative Gene Expression Data Using Real-Time Quantitative PCR and the 2−ΔΔCT Method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef]
- Spagnuolo-Weaver, M.; Fuerst, R.; Campbell, S.T.; Meehan, B.M.; McNeilly, F.; Adair, B.; Allan, G. A fluorimeter-based RT-PCR method for the detection and quantitation of porcine cytokines. J. Immunol. Methods 1999, 230, 19–27. [Google Scholar] [CrossRef]
- Nitkiewicz, A.; Smolinska, N.; Przala, J.; Kaminski, T. Expression of orexin receptors 1 (OX1R) and 2 (OX2R) in the porcine ovary during the oestrous cycle. Regul. Pept. 2010, 165, 186–190. [Google Scholar] [CrossRef]
- Li, X.; Zhu, Q.; Wang, W.; Qi, J.; He, Y.; Wang, Y.; Lu, Y.; Wu, H.; Ding, Y.; Sun, Y. Elevated Chemerin Induces Insulin Resistance in Human Granulosa-Lutein Cells from Polycystic Ovary Syndrome Patients. SSRN Electron. J. 2018. [Google Scholar] [CrossRef]
- Wang, Q.; Leader, A.; Tsang, B.K. Inhibitory Roles of Prohibitin and Chemerin in FSH-Induced Rat Granulosa Cell Steroidogenesis. Endocrinology 2013, 154, 956–967. [Google Scholar] [CrossRef] [PubMed]
- Abreu, R.D.S.; Penalva, L.O.; Salemi, M.; Vogel, C. Global signatures of protein and mRNA expression levels. Mol. BioSyst. 2009, 5, 1512–1526. [Google Scholar] [CrossRef]
- Schwanhäusser, B.; Busse, D.; Li, N.; Dittmar, G.; Schuchhardt, J.; Wolf, J.; Chen, W.; Selbach, M. Global quantification of mammalian gene expression control. Nature 2011, 473, 337–342. [Google Scholar] [CrossRef] [PubMed]
- Li, J.J.; Bickel, P.J.; Biggin, M.D. System wide analyses have underestimated protein abundances and the importance of transcription in mammals. Peer J. 2014, 2, e270. [Google Scholar] [CrossRef]
- Lawrence, T. The Nuclear Factor NF-kB Pathway in Inflammation. Cold Spring Harb. Perspect. Biol. 2009, 1, a001651. [Google Scholar] [CrossRef] [PubMed]
- Zmijewska, A.; Franczak, A.; Kotwica, G. Role of interleukin-1β in the regulation of porcine corpora lutea during the late luteal phase of the cycle and during pregnancy. Acta Veter Hung. 2012, 60, 395–407. [Google Scholar] [CrossRef][Green Version]
- Lin, S.-C.; Lo, Y.-C.; Wu, H. Helical assembly in the MyD88–IRAK4–IRAK2 complex in TLR/IL-1R signalling. Nature 2010, 465, 885–890. [Google Scholar] [CrossRef]
- Xia, Y.; Chen, S.; Wang, Y.; Mackman, N.; Ku, G.; Lo, D.; Feng, L. RelB Modulation of IκBα Stability as a Mechanism of Transcription Suppression of Interleukin-1α (IL-1α), IL-1β, and Tumor Necrosis Factor Alpha in Fibroblasts. Mol. Cell. Boil. 1999, 19, 7688–7696. [Google Scholar] [CrossRef]
- Hayden, M.; West, A.P.; Ghosh, S. NF-κB and the immune response. Oncogene 2006, 25, 6758–6780. [Google Scholar] [CrossRef]
- Luo, W.; Diaz, F.J.; Wiltbank, M.C. Induction of mRNA for chemokines and chemokine receptors by prostaglandin F2α is dependent upon stage of the porcine corpus luteum and intraluteal progesterone. Endocrinology 2011, 152, 2797–2805. [Google Scholar] [CrossRef]
- Przygrodzka, E.; Kaczmarek, M.M.; Kaczyński, P.; Ziecik, A.J.; Ski, P.K. Steroid hormones, prostanoids, and angiogenic systems during rescue of the corpus luteum in pigs. Reproduction 2016, 151, 135–147. [Google Scholar] [CrossRef] [PubMed]
- Vince, J.E.; Pantaki, D.; Feltham, R.; Mace, P.D.; Cordier, S.M.; Schmukle, A.C.; Davidson, A.J.; Callus, B.A.; Wong, W.W.-L.; E Gentle, I.; et al. TRAF2 Must Bind to Cellular Inhibitors of Apoptosis for Tumor Necrosis Factor (TNF) to Efficiently Activate NF-κB and to Prevent TNF-induced Apoptosis. J. Boil. Chem. 2009, 284, 35906–35915. [Google Scholar] [CrossRef] [PubMed]
- Rawlings, J.S.; Rosler, K.M.; Harrison, D. The JAK/STAT signaling pathway. J. Cell Sci. 2004, 117, 1281–1283. [Google Scholar] [CrossRef] [PubMed]
- Heinrich, P.C.; Behrmann, I.; Haan, S.; Hermanns, H.M.; Müller-Newen, G.; Schaper, F. Principles of interleukin (IL)-6-type cytokine signalling and its regulation. Biochem. J. 2003, 374, 1–20. [Google Scholar] [CrossRef]
- Kumar, A.; Commane, M.; Flickinger, T.W.; Horvath, C.M.; Stark, G.R. Defective TNF-α-Induced Apoptosis in STAT1-Null Cells Due to Low Constitutive Levels of Caspases. Science 1997, 278, 1630–1632. [Google Scholar] [CrossRef]
- Kaplan, D.H.; Shankaran, V.; Dighe, A.S.; Stockert, E.; Aguet, M.; Old, L.J.; Schreiber, R.D. Demonstration of an interferon γ-dependent tumor surveillance system in immunocompetent mice. Proc. Natl. Acad. Sci. USA 1998, 95, 7556–7561. [Google Scholar] [CrossRef]
- Tsuji, Y.; Adachi, S.; Iemoto, A.; Hasegawa, A.; Koyama, K.; Tamaoki, T.H.; Kashiwamura, S.-I.; Ueda, H.; Muranaka, J.; Furuyama, J.-I.; et al. Expression of interleukin-18 and its receptor in mouse ovary. Am. J. Reprod. Immunol. 2001, 46, 349–357. [Google Scholar] [CrossRef]
- Prakash, B.S.; Pedina, J.; Steiner, A.; Wuttke, W. Demonstration of luteotrophic responses of human recombinant γ interferon in porcine corpora lutea using an in-vivo microdialysis system. J. Steroid Biochem. Mol. Biol. 1997, 63, 189–194. [Google Scholar] [CrossRef]
- Przygrodzka, E.; Witek, K.J.; Kaczmarek, M.M.; Andronowska, A.; Ziecik, A.J. Expression of factors associated with apoptosis in the porcine corpus luteum throughout the luteal phase of the estrous cycle and early pregnancy: Their possible involvement in acquisition of luteolytic sensitivity. Theriogenology 2015, 83, 535–545. [Google Scholar] [CrossRef]
- Rytelewska, E.; Kiezun, M.; Kisielewska, K.; Gudelska, M.; Dobrzyn, K.; Kaminska, B.; Kaminski, T.; Smolinska, N. Chemerin as a modulator of ovarian steroidogenesis in pigs: An in vitro study. Theriogenology. under review.
- Ruiz de Galarreta, C.M.; Fanjul, L.F.; Hsueh, A.J.W. Progestin regulation of progesterone biosynthetic enzymes in cultured rat granulosa cells. Steroids 1985, 46, 987–1002. [Google Scholar] [CrossRef]
- Tanaka, N.; Iwamasa, J.; Matsuura, K.; Okamura, H. Effects of progesterone and anti-progesterone RU486 on ovarian 3β-hydroxysteroid dehydrogenase activity during ovulation in the gonadotrophin-primed immature rat. J. Reprod. Fertil. 1993, 97, 167–172. [Google Scholar] [CrossRef] [PubMed]
- Ruiz de Galarreta, C.M.; Fanjul, L.F.; Meidan, R.; Hsueh, A.J.W. Regulation of 3β-hydroxysteroid dehydrogenase activity by human chorionic gonadotropin, androgens, and anti-androgens in cultured testicular cells. J. Biol. Chem. 1983, 438, 663–665. [Google Scholar] [CrossRef]
- Fanjul, L.F.; Quintana, J.; González, J.; Santana-Delgado, P.; Estévez, F.; De Galarreta, C.M.R. Testicular 3β-hydroxysteroid dehydrogenase/Δ5–4 isomerase in the hypophysectomized rat: Effect of treatment with 5α-dihydrotestosterone. J. Endocrinol. 1992, 133, 237–243. [Google Scholar] [CrossRef] [PubMed]
- Park, J.Y.; Pillinger, M.H.; Abramson, S.B. Prostaglandin E2 synthesis and secretion: The role of PGE2 synthases. Clin. Immunol. 2006, 119, 229–240. [Google Scholar] [CrossRef]
- Kim, S.O.; Harris, S.M.; Duffy, D.M. Prostaglandin E2 (EP) receptors mediate PGE2-specific events in ovulation and luteinization within primate ovarian follicles. Endocrinology 2014, 155, 1466–1475. [Google Scholar] [CrossRef]
- Li, X.M. Prostaglandins alter the abundance of messenger ribonucleic acid for steroidogenic enzymes in cultured porcine granulosa cells. Boil. Reprod. 1993, 48, 1360–1366. [Google Scholar] [CrossRef]
- Wiesak, T.; Hunter, M.G.; Foxcroft, G.R. Effect of prostaglandins on luteal function during early pregnancy in pigs. Reproduction 1992, 95, 831–840. [Google Scholar] [CrossRef][Green Version]
- Jimbo, K.; Park, J.S.; Yokosuka, K.; Sato, K.; Nagata, K. Positive feedback loop of interleukin-1β upregulating production of inflammatory mediators in human intervertebral disc cells in vitro. J. Neurosurg. Spine 2005, 2, 589–595. [Google Scholar] [CrossRef]
- Ziecik, A.J.; Przygrodzka, E.; Kaczmarek, M.M. Corpus Luteum Regression and Early Pregnancy Maintenance in Pigs. In The Life Cycle of the Corpus Luteum; Springer International Publishing: Cham, Switzerland, 2017; pp. 227–248. ISBN 9783319432380. [Google Scholar]
- Maekawa, R.; Okuda, K. Species-related differences in the mechanism of apoptosis during structural luteolysis. J. Reprod. Dev. 2007, 53, 977–986. [Google Scholar] [CrossRef]
- Levine, B.; Sinha, S.C.; Kroemer, G. Bcl-2 family members: Dual regulators of apoptosis and autophagy. Autophagy 2008, 4, 600–606. [Google Scholar] [CrossRef] [PubMed]
- Lavitrano, M.; Barazzoni, A.; Forni, M.; Costerbosa, G.L. In situ detection of apoptosis in regressing corpus luteum of pregnant sow: Evidence of an early presence of DNA fragmentation. Domest. Anim. Endocrinol. 1996, 13, 361–372. [Google Scholar] [CrossRef]
- Safa, A.R. Roles of c-FLIP in Apoptosis, Necroptosis, and Autophagy. J. Carcinog. Mutagen. 2013, 4, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Graber, T.E.; Holcik, M. Distinct roles for the cellular inhibitors of apoptosis proteins 1 and 2. Cell Death Dis. 2011, 2, e135. [Google Scholar] [CrossRef]
- Yalcin, A.; Clem, B.F.; Imbert-Fernandez, Y.; Ozcan, S.C.; Peker, S.; O’Neal, J.; Klarer, A.C.; Clem, A.L.; Telang, S.; Chesney, J. 6-Phosphofructo-2-kinase (PFKFB3) promotes cell cycle progression and suppresses apoptosis via Cdk1-mediated phosphorylation of p27. Cell Death Dis. 2014, 5, e1337. [Google Scholar] [CrossRef] [PubMed]

| Gene Symbol | Gene Description | Primers Sequences | Product Length | Reference |
|---|---|---|---|---|
| IL1B | Interleukin 1β | F: TTTGAAGAAGAGCCCATCATCC R: CCAGCCAGCACTAGAGATTTG | 119 bp | [The present study] |
| CASP3 | Caspase 3 | F: GTGCTTCTAAGCCATGGTGAA R: CGGCAGGCCTGAATTATGAA | 143 bp | [The present study] |
| PTGS2 | Prostaglandin-endoperoxide synthase 2 | F: ATGGGTGTGAAAGGGAGGAAA R: AAACTGATGGGTGAAGTGCTG | 141 bp | [The present study] |
| HSD3B7 | Hydroxy-δ-5-steroid dehydrogenase, 3 β- and steroid δ-isomerase 7 | F: CTCGAAGCCAACGGAAGGA R: CCACGTTACCCACGTAGACC | 193 bp | [The present study] |
| ACTB | β-actin | F: ACATCAAGGAGAAGCTCTGCTACG R: GAGGGGCGATGATCTTGATCTTCA | 366 bp | [41] |
| GAPDH | Glyceraldehyde-3- phosphate dehydrogenase | F: CCTTCATTGACCTCCACTACATGG R: CCACAACATACGTAGCACCAGCATC | 183 bp | [42] |
| Treatment | CTRL | CHEM | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Samples | 1_LC | 2_LC | 3_LC | 4_LC | 5_LC | 1_LC | 2_LC | 3_LC | 4_LC | 5_LC |
| Row reads | 110.458 | 125.606 | 121.017 | 115.746 | 108.415 | 109.209 | 115.928 | 112.795 | 122.503 | 112.816 |
| Processed reads | 97.605 | 111.356 | 106.973 | 102.611 | 96.010 | 97.413 | 102.895 | 99.857 | 109.165 | 100.500 |
| Mapped reads | 97.057 | 110.805 | 106.363 | 102.009 | 95.54 | 96.93 | 102.244 | 99.362 | 108.613 | 99.97 |
| Uniquely mapped | 94.274 | 106.514 | 102.766 | 98.577 | 92.284 | 93.337 | 99.22 | 95.599 | 104.365 | 96.408 |
| % of uniquely mapped | 97.13% | 96.13% | 96.62% | 96.64% | 96.59% | 96.29% | 97.04% | 96.21% | 96.09% | 96.44% |
| Multi-mapped | 2.783 | 4.291 | 3.597 | 3.432 | 3.256 | 3.593 | 3.024 | 3.763 | 4.248 | 3.562 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Makowczenko, K.G.; Jastrzebski, J.P.; Szeszko, K.; Smolinska, N.; Paukszto, L.; Dobrzyn, K.; Kiezun, M.; Rytelewska, E.; Kaminska, B.; Kaminski, T. Transcription Analysis of the Chemerin Impact on Gene Expression Profile in the Luteal Cells of Gilts. Genes 2020, 11, 651. https://doi.org/10.3390/genes11060651
Makowczenko KG, Jastrzebski JP, Szeszko K, Smolinska N, Paukszto L, Dobrzyn K, Kiezun M, Rytelewska E, Kaminska B, Kaminski T. Transcription Analysis of the Chemerin Impact on Gene Expression Profile in the Luteal Cells of Gilts. Genes. 2020; 11(6):651. https://doi.org/10.3390/genes11060651
Chicago/Turabian StyleMakowczenko, Karol G., Jan P. Jastrzebski, Karol Szeszko, Nina Smolinska, Lukasz Paukszto, Kamil Dobrzyn, Marta Kiezun, Edyta Rytelewska, Barbara Kaminska, and Tadeusz Kaminski. 2020. "Transcription Analysis of the Chemerin Impact on Gene Expression Profile in the Luteal Cells of Gilts" Genes 11, no. 6: 651. https://doi.org/10.3390/genes11060651
APA StyleMakowczenko, K. G., Jastrzebski, J. P., Szeszko, K., Smolinska, N., Paukszto, L., Dobrzyn, K., Kiezun, M., Rytelewska, E., Kaminska, B., & Kaminski, T. (2020). Transcription Analysis of the Chemerin Impact on Gene Expression Profile in the Luteal Cells of Gilts. Genes, 11(6), 651. https://doi.org/10.3390/genes11060651








