Analysis of CFTR Mutation Spectrum in Ethnic Russian Cystic Fibrosis Patients
Abstract
1. Introduction
2. Materials and Methods
3. Results
4. Discussion
4.1. Similarity and Difference of Frequency Profiles of Common CF Variants in Two Samples of Russian Patients and the Data of CFTR2
4.2. Sanger Sequencing Detection of the CFTR Gene Variants
4.3. CNV in Russian CF Patients Detected by MLPA
4.4. Detection Rate of Three-Stage Analysis of CFTR Gene in Russian CF Patients
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Sosnay, P.R.; Raraigh, K.S.; Gibson, R.L. Molecular Genetics of Cystic Fibrosis Transmembrane Conductance Regulator: Genotype and Phenotype. Pediatr. Clin. North Am. 2016, 63, 585–598. [Google Scholar] [CrossRef] [PubMed]
- Farrell, P.M.; White, T.B.; Howenstine, M.S.; Munck, A.; Parad, R.B.; Rosenfeld, M.; Sommerburg, O.; Accurso, F.J.; Davies, J.C.; Rock, M.J.; et al. Diagnosis of Cystic Fibrosis in Screened Populations. J. Pediatr. 2017, 181S, S33–S44. [Google Scholar] [CrossRef] [PubMed]
- Castellani, C.; Cuppens, H.; Macek, M., Jr.; Cassiman, J.J.; Kerem, E.; Durie, P.; Tullis, E.; Assael, B.M.; Bombieri, C.; Brown, A.; et al. Consensus on the use and interpretation of cystic fibrosis mutation analysis in clinical practice. J. Cyst. Fibros. 2008, 7, 179–196. [Google Scholar] [CrossRef] [PubMed]
- De Boeck, K.; Vermeulen, F.; Dupont, L. The diagnosis of cystic fibrosis. Presse Med. 2017, 46, e97–e108. [Google Scholar] [CrossRef] [PubMed]
- Castellani, C.; Duff, A.J.A.; Bell, S.C.; Heijerman, H.G.M.; Munck, A.; Ratjen, F.; Sermet-Gaudelus, I.; Southern, K.W.; Barben, J.; Flume, P.A.; et al. ECFS best practice guidelines: The 2018 revision. J. Cyst. Fibros. 2018, 17, 153–178. [Google Scholar] [CrossRef] [PubMed]
- National Consensus Project. Cystic Fibrosis: Definition, Diagnostic Criteria, Treatment 2018; Kondratyeva, E.I., Kashirskaya, N.Y., Kaprqanov, N.I., Eds.; Company BORGES Ldt.: Moscow, Russia, 2018; p. 356. [Google Scholar]
- MEDPRAKTIKA-M. Registry of Patients with Cystic Fibrosis in the Russian Federation. 2017; Voronkova, A.Y., Amelina, E.A., Kashirskaya, N.Y., Kondratyeva, E.I., Krasovsky, S.A., Starinova, M.A., Kapranov, N.I., Eds.; MEDPRAKTIKA-M: Moscow, Russia, 2019; p. 68. (in Russian) [Google Scholar]
- Petrova, N.V.; Kashirskaya, N.Y.; Vasilieva, T.A.; Timkovskaya, E.E.; Voronkova, A.Y.; Shabalova, L.A.; Kondrateva, E.I.; Sherman, V.D.; Kapranov, N.I.; Zinchenko, R.A.; et al. High proportion of W1282X mutation in CF patients from Karachai-Cherkessia. J. Cyst. Fibros. 2016, 15, e28–e32. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Petrova, N.V.; Kashirskaya, N.Y.; Saydaeva, D.K.; Polyakov, A.V.; Adyan, T.A.; Simonova, O.I.; Gorinova, Y.V.; Kondratyeva, E.I.; Sherman, V.D.; Novoselova, O.G.; et al. Spectrum of CFTR mutations in Chechen cystic fibrosis patients: High frequency of c.1545_1546delTA (p.Tyr515X; 1677delTA) and c.274G>A (p.Glu92Lys, E92K) mutations in North Caucasus. BMC Med. Genet. 2019, 20, 44. [Google Scholar] [CrossRef]
- Stepanova, A.A.; Abrukova, A.V.; Savaskina, E.N.; Poliakov, A.V. Mutation p.E92K is the primary cause of cystic fibrosis in Chuvashes. Russ. J. Genet. 2012, 48, 731–737. [Google Scholar] [CrossRef]
- StatData.ru. Available online: http://www.statdata.ru/nacionalnyj-sostav-rossii (accessed on 8 February 2020).
- Petrova, N.V.; Kondratyeva, E.I.; Krasovsky, S.A.; Polyakov, A.V.; Ivachshenko, T.E.; Pavlov, A.E.; Zinchenko, R.A.; Ginter, E.K.; Kutsev, S.I.; Odinokova, O.N.; et al. National Consensus Project «Cystic fibrosis: Definition, diagnostic criteria, treatment» Section «Genetics of Cystic Fibrosis. Molecular genetic diagnosis of cystic fibrosis». Meditsinskaya Genet. 2016, 11, 29–45. (in Russian). [Google Scholar]
- Hantash, F.M.; Redman, J.B.; Goos, D.; Kammesheidt, A.; McGinniss, M.J.; Sun, W.; Strom, C.M. Characterization of a recurrent novel large duplication in the cystic fibrosis transmembrane conductance regulator gene. J. Mol. Diagn. 2007, 9, 556–560. [Google Scholar] [CrossRef][Green Version]
- Petrova, N.V.; Timkovskaya, E.E.; Zinchenko, R.A. The analysis of CFTR mutations frequencies in different populations of Russia. Eur. J. Hum. Genet. 2008, 16, 387. [Google Scholar]
- Verlingue, C.; Kapranov, N.I.; Mercier, B.; Ginter, E.K.; Petrova, N.V.; Audrezet, M.P.; Ferec, C. Complete screening of mutations in the coding sequence of the CFTR gene in a sample of CF patients from Russia: Identification of three novel alleles. Hum. Mutat. 1995, 5, 205–209. [Google Scholar] [CrossRef] [PubMed]
- Ivaschenko, T.E.; Baranov, V.S. Biochemical and Molecular Genetic Basis of Cystic Fibrosis Pathogenesis; Intermedika: Saint-Petersburg, Russia, 2002; p. 256. (in Russian) [Google Scholar]
- Korytina, G.F.; Victorova, T.V.; Baykova, G.V.; Khusnutdinova, E.K. Analysis of the Spectra of Mutations and Polymorphic Loci of Cystic Fibrosis Transmembrane Conductance Regulator in the Population of Bashkortostan. Rus. J. Genet. 2002, 38, 1076–1081. [Google Scholar] [CrossRef]
- WHO Human Genetics Programme. The Molecular Genetic Epidemiology of Cystic Fibrosis. Report of a Joint Meeting of WHO/ECFTN/ICF(M)/ECFS/World Health Organization (WHO); WHO: Genoa, Italy, 2002; Available online: https://www.cfww.org/docs/who/2002/who_hgn_cf_wg_04.02.pdf (accessed on 8 February 2020).
- Cystic Fibrosis Mutation Database. Available online: http://www.genet.sickkids.on.ca (accessed on 5 February 2020).
- Bobadilla, J.L.; Macek, M., Jr.; Fine, J.P.; Farrell, P.M. Cystic fibrosis: A worldwide analysis of CFTR mutations—Correlation with incidence data and application to screening. Hum. Mutat. 2002, 19, 575–606. [Google Scholar] [CrossRef] [PubMed]
- Dörk, T.; Macek, M., Jr.; Mekus, F.; Tümmler, B.; Tzountzouris, J.; Casals, T.; Krebsová, A.; Koudová, M.; Sakmaryová, I.; Macek, M.S.; et al. Characterization of a novel 21-kb deletion, CFTRdele2,3(21kb), in the CFTR gene: A cystic fibrosis mutation of Slavic origin common in Central and East Europe. Hum. Genet. 2000, 106, 259–268. [Google Scholar] [CrossRef] [PubMed]
- CFTR2. Clinical and Functional Translation of CFTR. Available online: https://www.cftr2.org/ (accessed on 5 February 2020).
- Petrova, N.V.; Marakhonov, A.V.; Vasilyeva, T.A.; Kashirskaya, N.Y.; Ginter, E.K.; Kutsev, S.I.; Zinchenko, R.A. Comprehensive genotyping reveals novel CFTR variants in cystic fibrosis patients from the Russian Federation. Clin. Genet. 2018, 95, 1–4. [Google Scholar] [CrossRef] [PubMed]
- Richards, S.; Aziz, N.; Bale, S.; Bick, D.; Das, S.; Gastier-Foster, J.; Grody, W.W.; Hegde, M.; Lyon, E.; Spector, E.; et al. Standards and guidelines for the interpretation of sequence variants: A joint consensus recommendation of the American College of Medical Genetics and Genomics and the Association for Molecular Pathology. Genet. Med. 2015, 17, 405–424. [Google Scholar] [CrossRef] [PubMed]
- National Center for Biotechnology Information (NCBI-ClinVar). Available online: https://www.ncbi.nlm.nih.gov/clinvar/ (accessed on 5 February 2020).
- Lee, J.H.; Choi, J.H.; Namkung, W.; Hanrahan, J.W.; Chang, J.; Song, S.Y.; Park, S.W.; Kim, D.S.; Yoon, J.-H.; Suh, Y.; et al. A haplotype-based molecular analysis of CFTR mutations associated with respiratory and pancreatic diseases. Hum. Mol. Genet. 2003, 12, 2321–2332. [Google Scholar] [CrossRef] [PubMed]
- Bergougnoux, A.; Délétang, K.; Pommier, A.; Varilh, J.; Houriez, F.; Altieri, J.P.; Koenig, M.; Férec, C.; Claustres, M.; Lalau, G.; et al. Functional characterization and phenotypic spectrum of three recurrent disease-causing deep intronic variants of the CFTR gene. J. Cyst. Fibros. 2019, 18, 468–475. [Google Scholar] [CrossRef] [PubMed]
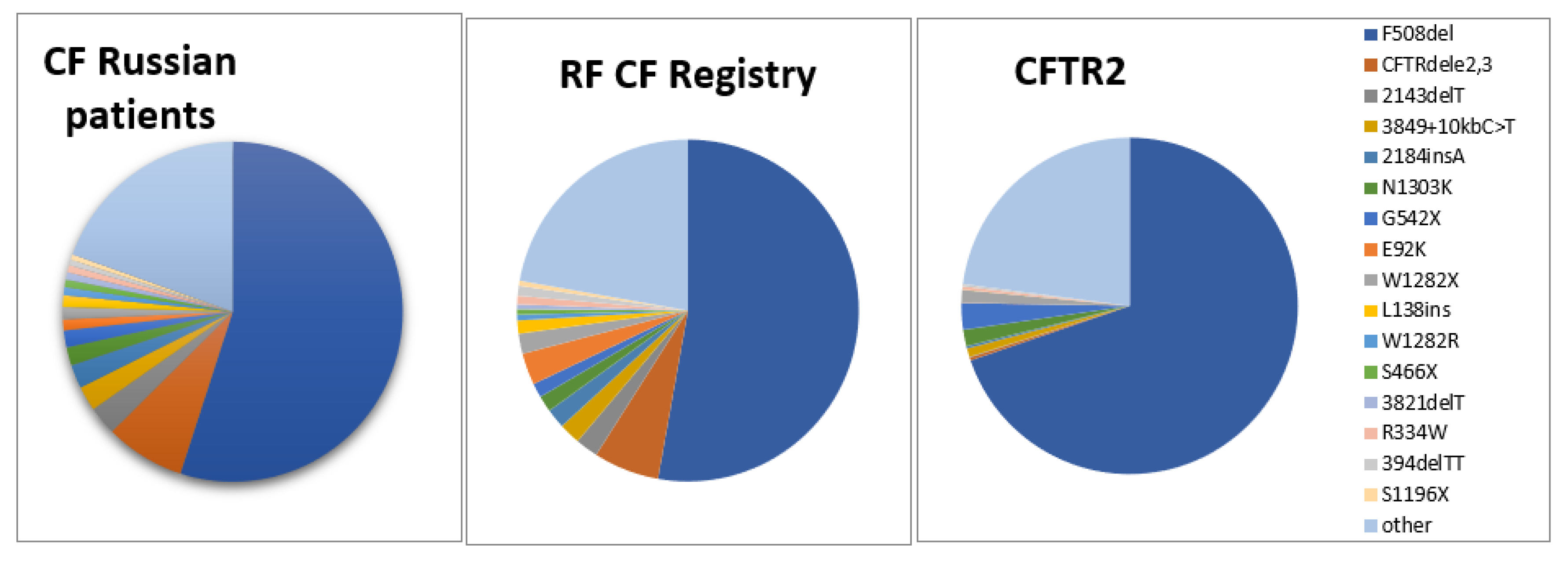
| No. | Variants | Number | % | % in RF CF Registry |
|---|---|---|---|---|
| 1 | c.1521_1523delCTT (p.Phe508del, F508del) | 1522 | 54.99 | 52.81 |
| 2 | c.54-5940_273+10250del21kb (p.Ser18Argfs*16, CFTRdele2,3) | 210 | 7.59 | 6.21 |
| 3 | c.2012delT (p.Leu671X, 2143delT) | 75 | 2.71 | 2.15 |
| 4 | c.3718-2477C>T (3849+10kbC-T) | 65 | 2.35 | 2.02 |
| 5 | c.2052_2053insA (p.Gln685ThrfsX4, 2184insA) | 62 | 2.24 | 1.85 |
| 6 | c.3909C>G (p.Asn1303Lys, N1303K) | 48 | 1.73 | 1.54 |
| 7 | c.1624G>T (p.Gly542X, G542X) | 44 | 1.59 | 1.35 |
| 8 | c.274G>A (p.Glu92Lys, E92K) | 29 | 1.05 | 3.00 |
| 9 | c.3846G>A (p.Trp1282X, W1282X) | 32 | 1.16 | 1.90 |
| 10 | c.413_415dupTAC (p.Leu138dup; L138ins) | 31 | 1.12 | 1.24 |
| 11 | c.3844T>C (p.Trp1282Arg, W1282R) | 21 | 0.76 | 0.55 |
| 12 | c.1397C>G (p.Ser466X, Ser466X) | 20 | 0.72 | 0.50 |
| 13 | c.3691delT (p.Ser1231ProfsX4, 3821delT) | 19 | 0.69 | 0.46 |
| 14 | c.1000C>T (p.Arg334Trp, R334W) | 19 | 0.69 | 0.80 |
| 15 | c.262_263delTT (p.Leu88IlefsX22, 394delTT) | 15 | 0.54 | 0.94 |
| 16 | c.3587C>G (p.Ser1196X, S1196X) | 14 | 0.51 | 0.48 |
| 17 | c.3816_3817delGT (p.Ser1273LeufsX28, 3944delGT) | 12 | 0.43 | 0.27 |
| 18 | c.2657+5G>A (2789+5A>G) | 10 | 0.36 | 0.48 |
| 19 | c.489+1G>T (621+1G>T) | 7 | 0.25 | 0.18 |
| 20 | c.3140-16T>A (3272-16T>A) | 6 | 0.22 | 0.34 |
| 21 | c.1657C>T (p.Arg553X, R553X) | 5 | 0.18 | 0.18 |
| 22 | c.1545_1546delTA (p.Tyr515X, 1677delTA) | 5 | 0.18 | 1.81 |
| 23 | c.3535_3536insTCAA (p.Thr1179IlefsX17, 3667ins4) | 4 | 0.14 | 0.10 |
| 24 | c.254G>A (p.Gly85Glu, G85E) | 4 | 0.14 | 0.10 |
| 25 | c.472dupA (p.Ser158LysfsX5, 604insA) | 3 | 0.11 | 0.10 |
| 26 | c.2051_2052delAAinsG (p.Lys684SerfsX38, 2183AA>G) | 3 | 0.11 | 0.04 |
| 27 | c.3475T>C (p.Ser1159Pro; S1159P) | 3 | 0.11 | 0.10 |
| 28 | c.1040G>C (p.Arg347Pro, R347P) | 2 | 0.07 | 0.10 |
| 29 | c.350G>A (p.Arg117His, R117H) | 1 | 0.04 | 0.04 |
| 30 | c.1519_1521delATC (p.Ile507del, I507del) | 0 | - | 0 |
| 31 | c.1585-1G>A (1717-1G>A) | 0 | - | 0.04 |
| 32 | c.1652G>A (p.Gly551Asp, G551D) | 0 | - | 0.04 |
| 33 | c.3476C>T (p.Ser1159Phe, S1159F) | 0 | - | 0.11 |
| Identified | 2290 | 82.78 | ||
| Not identified | 478 | 17.22 | ||
| Total | 2768 |
| No. | Variant According to cDNA | Protein Change | Legacy Name | Exon/Intron 1 | Number | Mutation Type |
|---|---|---|---|---|---|---|
| 1 | c.43delC | p.Leu15PhefsX1 | 175delC | 1e | 2 | sd |
| 2 | c.53+1G>T | 185+1G->T | 1i | 2 | s | |
| 3 | c.79G>T | p.Gly27X | G27X | 2e | 1 | n |
| 4 | c.115C>T | p.Gln39X | Q39X | 2e | 1 | n |
| 5 | c.223C>T | p.Arg75X | R75X | 3e | 1 | n |
| 6 | c.252T>A | p.Tyr84X | 3e | 2 | n | |
| 7 | c.264_268delATATT | p.Leu88PhefsX21 | 3e | 1 | sd | |
| 8 | c.274-6T>C | 406-6T>C | 3i | 1 | s | |
| 9 | c.274G>T | p.Glu92X | E92X | 4e | 1 | n |
| 10 | c.293A>G | p.Gln98Arg | Q98R | 4e | 1 | m |
| 11 | c.358G>C | p.Ala120Pro | 4e | 1 | m | |
| 12 | c.422C>A | p.Ala141Asp | A141D | 4e | 1 | m |
| 13 | c.490-1G>C | 4i | 1 | s | ||
| 14 | c.580-1G>T | 712-1G->T | 5i | 8 | s | |
| 15 | c.613C>A | p.Pro205Thr | 6a e | 1 | m | |
| 16 | c.650A>G | p.Glu217Gly | E217G | 6a e | 1 | m |
| 17 | c.831G>A | p.Trp277X | 6b e | 1 | n | |
| 18 | c.940G>A | p.Gly314Arg | G314R | 7e | 1 | m |
| 19 | c.[1075C>A;1079C>A] | p.[Gln359Lys;Thr360Lys] | Q359K/T360K | 7e | 1 | m |
| 20 | c.1083G>A | p.Trp361X | 7e | 2 | n | |
| 21 | c.1086T>A | p.Tyr362X | Y362X | 7e | 1 | n |
| 22 | c.1204G>T | p.Glu402X | 8e | 1 | n | |
| 23 | c.1209G>C | p.Glu403Asp | E403D | 8e | 3 | m |
| 24 | c.[1210−12[5];1210-34TG[12]] | 5T;TG12 | 7i | 1 | s | |
| 25 | c.1219delG | p.Glu407AsnfsX35 | 9e | 1 | sd | |
| 26 | c.1352G>T | p.Gly451Val | 9e | 1 | m | |
| 27 | c.1240_1244delCAAAA | p.Asn415X | 1365del5 | 9e | 11 | sd |
| 28 | c.1364C>A | p.Ala455Glu | A455E | 9e | 1 | m |
| 29 | c.1382G>A | p.Gly461Glu | 9e | 1 | m | |
| 30 | c.1438G>T | p.Gly480Cys | G480C | 10e | 1 | m |
| 31 | c.1501A>G | p.Thr501Ala | T501A | 10e | 1 | m |
| 32 | c.1513A>C | p.Asn505His | 10e | 1 | m | |
| 33 | c.1525G>C | p.Gly509Arg | 10e | 1 | m | |
| 34 | c.1528delG | p.Val510PhefsX17 | 1660delG | 10e | 1 | sd |
| 35 | c.1584+1G>A | 1716+1G>A | 10i | 1 | s | |
| 36 | c.1589T>C | p.Ile530Thr | 11e | 1 | m | |
| 37 | c.1608delA | p.Asp537ThrfsX3 | 11e | 2 | sd | |
| 38 | c.1646G>A | p.Ser549Asn | S549N | 11e | 1 | m |
| 39 | c.1705T>C | p.Tyr569His | Y569H | 12e | 1 | m |
| 40 | c.1735G>T | p.Asp579Tyr | D579Y | 12e | 2 | m |
| 41 | c.1766+2T>C | 12i | 2 | s | ||
| 42 | c.1766+1G>A | 1898+1G>A | 12i | 4 | s | |
| 43 | c.1766+1G>C | 1898+1G>C | 12i | 5 | s | |
| 44 | c.1792_1793insAAA | p.Lys598dup | K598ins | 13e | 1 | i |
| 45 | c.1795dupA | p.Thr599AsnfsX2 | 13e | 1 | si | |
| 46 | c.1911delG | p.Gln637HisfsX26 | 2043delG | 13e | 2 | sd |
| 47 | c.2128A>T | p.Lys710X | K710X | 13e | 3 | n |
| 48 | c.2195T>G | p.Leu732X | L732X | 13e | 1 | n |
| 49 | c.2290C>T | p.Arg764X | R764X | 13e | 1 | n |
| 50 | c.2312delA | p.Asn771ThrfsX2 | 13e | 1 | sd | |
| 51 | c.2353C>T | p.Arg785X | R785X | 13e | 6 | n |
| 52 | c.2374C>T | p.Arg792X | R792X | 13e | 1 | n |
| 53 | c.2417A>G | p.Asp806Gly | D806G | 13e | 1 | m |
| 54 | c.2589_2599delAATTTGGTGCT | p.Ile864SerfsX28 | 2721del11 | 14a e | 2 | sd |
| 55 | c.2617G>T | p.Glu873X | 14a e | 1 | n | |
| 56 | c.2658-2A>G | 2790-2A->G | 14b i | 1 | s | |
| 57 | c.2780T>C | p.Leu927Pro | L927P | 15e | 1 | m |
| 58 | c.2834C>T | p.Ser945Leu | S945L | 15e | 4 | m |
| 59 | c.2909G>A | p.Gly970Asp | G970D | 16e | 1 | m |
| 60 | c.2936A>T | p.Asp979Val | D979V | 16e | 1 | m |
| 61 | c.2988+1G>A | 3120+1G->A | 16i | 1 | s | |
| 62 | c.2989-2A>C | 16i | 1 | s | ||
| 63 | c.2989-2A>G | 3121-2A->G | 16i | 1 | s | |
| 64 | c.3107C>A | p.Thr1036Asn | 17a e | 1 | m | |
| 65 | c.3112C>T | p.Gln1038X | 17a e | 1 | n | |
| 66 | c.3189delG | p.Trp1063X | 17b e | 1 | n | |
| 67 | c.3472C>T | p.Arg1158X | R1158X | 19e | 2 | n |
| 68 | c.3484C>T | p.Arg1162X | R1162X | 19e | 2 | n |
| 69 | c.3528delC | p.Lys1177SerfsX15 | 3659delC | 19e | 2 | sd |
| 70 | c.3763T>C | p.Ser1255Pro | S1255P | 20e | 2 | m |
| 71 | c.3775A>T | p.Arg1259X | 20e | 1 | n | |
| 72 | c.3872A>G | p.Gln1291Arg | Q1291R | 20e | 1 | m |
| 73 | c.3874-2A>G | 4006-2A->G | 20i | 1 | s | |
| 74 | c.3883delA | p.Ile1295PhefsX33 | 4015delA | 21e | 3 | sd |
| 75 | c.3884_3885insT | p.Ser1297PhefsX5 | 4016insT | 21e | 3 | si |
| 76 | c.3929G>A | p.Trp1310X | W1310X | 21e | 7 | n |
| 77 | c.3963+1G>T | 4095+1G->T | 21i | 1 | s | |
| 78 | c.4004T>C | p.Leu1335Pro | L1335P | 22e | 6 | m |
| 79 | c.4242+1G>A | 4374+G->A | 23i | 1 | s | |
| 80 | c.4296_4297insGA | p.Ser1435GlyfsX14 | 4428insGA | 24e | 2 | si |
| 81 | c.(?-1)_(1584+1_1585-1)del | CFTRdele1-10 | 1 | CNV | ||
| 82 | c.(53+2-54-1)_(273+1_274-1)del | CFTRdele2,3(non 21kb)1 | 1 | CNV | ||
| 83 | c.(273+1_274-1)_(743+1_744-1)del | CFTRdele4-6a | 1 | CNV | ||
| 84 | c.(273-1_274+1)_(869+1_870-1)del(1209-1_1210+1)_(1392+1_1393+1)del | CFTRdel4-7;del9-10 | 2 | CNV | ||
| 85 | c.(489+1_490-1)_(1392+1_1393-1)del | CFTRdele5-10 | 1 | CNV | ||
| 86 | c.(53+1_54-1)_(164+1_165+1)del | CFTRdele21 | 2 | CNV | ||
| 87 | c.(53+1_54-1)_(869+1_870+1)del | CFTRdele2-7 | 1 | CNV | ||
| 88 | c.(1679+1_1680-1)_(2490+1_2491-1)del(2908+1_2909-1)del | CFTRdele12,13;del161 | 2 | CNV | ||
| 89 | c.(743+1_744-1)_(1584+1_1585-1)dup | CFTRdup6b-10 (gIVS6a+415_IVS10+2987Dup26817bp) | 5 | CNV | ||
| 90 | c.(743-1_744+1)_(869+1_870-1)dup | CFTRdup6b,7 | 1 | CNV | ||
| 91 | c.(4136+1_4137-1)_(*1_?)dup | CFTRdup23,24 | 1 | CNV |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Petrova, N.V.; Kashirskaya, N.Y.; Vasilyeva, T.A.; Kondratyeva, E.I.; Zhekaite, E.K.; Voronkova, A.Y.; Sherman, V.D.; Galkina, V.A.; Ginter, E.K.; Kutsev, S.I.; et al. Analysis of CFTR Mutation Spectrum in Ethnic Russian Cystic Fibrosis Patients. Genes 2020, 11, 554. https://doi.org/10.3390/genes11050554
Petrova NV, Kashirskaya NY, Vasilyeva TA, Kondratyeva EI, Zhekaite EK, Voronkova AY, Sherman VD, Galkina VA, Ginter EK, Kutsev SI, et al. Analysis of CFTR Mutation Spectrum in Ethnic Russian Cystic Fibrosis Patients. Genes. 2020; 11(5):554. https://doi.org/10.3390/genes11050554
Chicago/Turabian StylePetrova, Nika V., Nataliya Y. Kashirskaya, Tatyana A. Vasilyeva, Elena I. Kondratyeva, Elena K. Zhekaite, Anna Y. Voronkova, Victoria D. Sherman, Varvara A. Galkina, Eugeny K. Ginter, Sergey I. Kutsev, and et al. 2020. "Analysis of CFTR Mutation Spectrum in Ethnic Russian Cystic Fibrosis Patients" Genes 11, no. 5: 554. https://doi.org/10.3390/genes11050554
APA StylePetrova, N. V., Kashirskaya, N. Y., Vasilyeva, T. A., Kondratyeva, E. I., Zhekaite, E. K., Voronkova, A. Y., Sherman, V. D., Galkina, V. A., Ginter, E. K., Kutsev, S. I., Marakhonov, A. V., & Zinchenko, R. A. (2020). Analysis of CFTR Mutation Spectrum in Ethnic Russian Cystic Fibrosis Patients. Genes, 11(5), 554. https://doi.org/10.3390/genes11050554





