The Role of Wnt Signalling in Chronic Kidney Disease (CKD)
Abstract
1. Chronic Kidney Disease (CKD)
2. The Clinical Symptoms of Genetically Inherited Chronic Kidney Diseases
3. The Role of Upstream Wnt Components in Genetic CKD
4. Wnt Signalling Receptor Involvement in CKD
5. The Role of β-Catenin in Genetic CKD
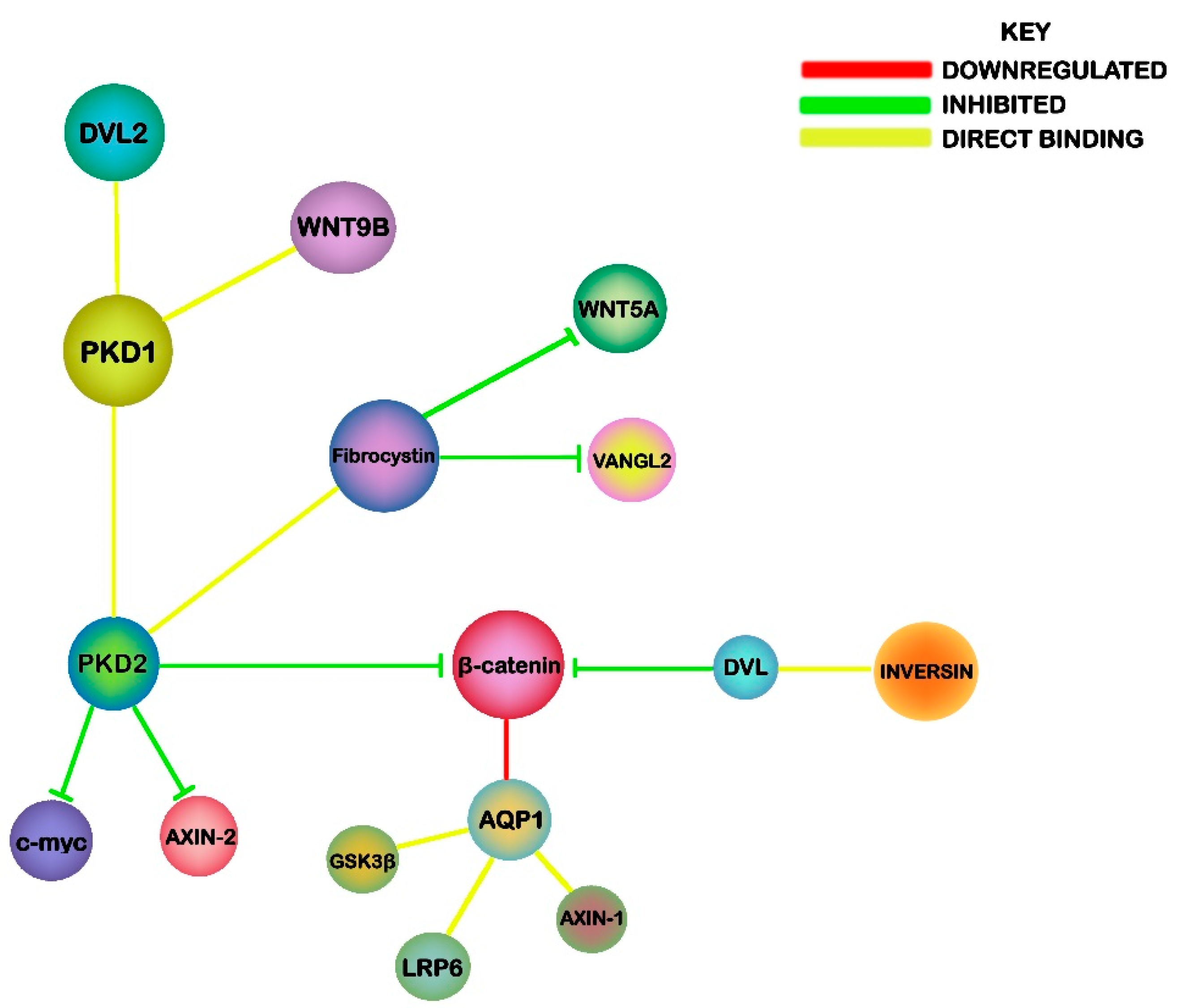
6. Links between Wnt Signalling, PKD and NPHP
Author Contributions
Acknowledgments
Conflicts of Interest
Abbreviation
| ADPKD | Autosomal Dominant Polycystic Kidney Disease |
| ADR | Adriamycin |
| AKI | Acute kidney injury |
| APC | Adenomatous polyposis coli |
| AQP1 | Aquaporin-1 |
| ARPKD | Autosomal Recessive Polycystic Kidney Disease |
| AXIN | Axis inhibitor |
| CK | Casein kinase |
| CKD | Chronic Kidney Disease |
| DKK | Dickkopf |
| DN | Diabetic nephropathy |
| Dvl | Dishevelled |
| E | Embryonic day |
| ESRD | End-stage renal disease |
| FSGS | Focal and segmental glomerulosclerosis |
| Fz | Frizzled |
| GFR | Glomerular Filtration Rate |
| GSK | Glycogen synthase kinase |
| HEK293 | Human embryonic kidney 293 cells |
| IgAN | IgA nephropathy |
| Inv | Inversin |
| IRI | Ischaemia reperfusion injury |
| Jbn | Jouberin |
| JS | Joubert syndrome |
| JSRD | Joubert syndrome and related disorders |
| KO | Knockout |
| LOF | Loss of function |
| Lp | Loop-tail |
| LRP | Lipoprotein receptor related protein |
| MDCK | Madin-Darby Canine Kidney |
| MEF | Mouse embryonic fibroblast |
| MGS | Meckel-Gruber Syndrome |
| MM | Metanephric mesenchyme |
| MMP-7 | Matrix metalloproteinase-7 |
| NPHP | Nephronophthisis |
| PAI-1 | Plasminogen activator inhibitor-1 |
| PC | Polycystin |
| PCP | Planar Cell Polarity |
| PKD | Polycystic Kidney Disease |
| PKHD1 | Polycystic Kidney and Hepatic Disease 1 |
| sFRP | Soluble Frizzled-related protein |
| TRPC | Transient receptor potential cation channel |
| UUO | Unilateral Ureteral Obstruction |
References
- Thomas, R.; Kanso, A.; Sedor, J. Chronic Kidney Disease and Its Complications. Prim. Care. 2008, 35, 329–344. [Google Scholar] [CrossRef]
- Chapman, A.; Devuyst, O.; Eckardt, K.; Gansevoort, R.; Harris, T.; Horie, S.; Kasiske, B.; Odland, D.; Pei, Y.; Perrone, R.; et al. Autosomal-dominant polycystic kidney disease (ADPKD): Executive summary from a Kidney Disease: Improving Global Outcomes (KDIGO) Controversies Conference. Kidney Int. 2015, 88, 17–27. [Google Scholar] [CrossRef] [PubMed]
- Halvorson, C.; Bremmer, M.; Jacobs, S. Polycystic kidney disease: Inheritance, pathophysiology, prognosis, and treatment. Int. J. Nephrol. Renov. Dis. 2010, 3, 69–83. [Google Scholar] [CrossRef]
- Torra, R.; Badenas, C.; Darnell, A.; Nicolau, C.; Volpini, C.; Revert, L.; Estivill, X. Linkage, clinical features, and prognosis of autosomal dominant polycystic kidney disease types 1 and 2. J. Am. Soc. Nephrol. 1996, 7, 2142–2151. [Google Scholar] [PubMed]
- Harris, P.; Bae, K.; Rossetti, S.; Torres, V.; Grantham, J.; Chapman, A.; Guay-Woodford, L.; King, B.; Wetzel, L.; Baumgarten, D.; et al. Cyst Number but Not the Rate of Cystic Growth Is Associated with the Mutated Gene in Autosomal Dominant Polycystic Kidney Disease. J. Am. Soc. Nephrol. 2006, 17, 3013–3019. [Google Scholar] [CrossRef]
- Hartung, E.; Guay-Woodford, L. Autosomal Recessive Polycystic Kidney Disease: A Hepatorenal Fibrocystic Disorder with Pleiotropic Effects. Pediatrics 2014, 134, e833–e845. [Google Scholar] [CrossRef]
- Al-Bhalal, L.; Akhtar, M. Molecular Basis of Autosomal Recessive Polycystic Kidney Disease (ARPKD). Adv. Anat. Pathol. 2008, 15, 54–58. [Google Scholar] [CrossRef]
- Zhang, J.; Wu, M.; Wang, S.; Shah, J.; Wilson, P.; Zhou, J. Polycystic kidney disease protein fibrocystin localizes to the mitotic spindle and regulates spindle bipolarity. Hum. Mol. Genet. 2010, 19, 3306–3319. [Google Scholar] [CrossRef]
- Goggolidou, P. Wnt and planar cell polarity signaling in cystic renal disease. Organogenesis 2014, 10, 86–95. [Google Scholar] [CrossRef]
- Lu, H.; Galeano, M.; Ott, E.; Kaeslin, G.; Kausalya, P.; Kramer, C.; Ortiz-Brüchle, N.; Hilger, N.; Metzis, V.; Hiersche, M.; et al. Mutations in DZIP1L, which encodes a ciliary-transition-zone protein, cause autosomal recessive polycystic kidney disease. Nat. Genet. 2017, 49, 1025–1034. [Google Scholar] [CrossRef]
- Wolf, M. Nephronophthisis and related syndromes. Curr. Opin. Pediatr. 2015, 27, 201–211. [Google Scholar] [CrossRef] [PubMed]
- Simms, R.; Eley, L.; Sayer, J. Nephronophthisis. Eur. J. Hum. Genet. 2008, 17, 406–416. [Google Scholar] [CrossRef] [PubMed]
- Hildebrandt, F.; Otto, E.; Rensing, C.; Nothwang, H.; Vollmer, M.; Adolphs, J.; Hanusch, H.; Brandis, M. A novel gene encoding an SH3 domain protein is mutated in nephronophthisis type 1. Nat. Genet. 1997, 17, 149–153. [Google Scholar] [CrossRef]
- Gagnadoux, M.; Bacri, J.; Broyer, M.; Habib, R. Infantile chronic tubulo-interstitial nephritis with cortical microcysts: Variant of nephronophthisis or new disease entity? Pediatr. Nephrol. 1989, 3, 50–55. [Google Scholar] [CrossRef] [PubMed]
- Omran, H.; Fernandez, C.; Jung, M.; Häffner, K.; Fargier, B.; Villaquiran, A.; Waldherr, R.; Gretz, N.; Brandis, M.; Rüschendorf, F. Identification of a New Gene Locus for Adolescent Nephronophthisis, on Chromosome 3q22 in a Large Venezuelan Pedigree. Am. J. Hum. Genet. 2000, 66, 118–127. [Google Scholar] [CrossRef] [PubMed]
- Srivastava, S.; Molinari, E.; Raman, S.; Sayer, J. Many Genes—One Disease? Genetics of Nephronophthisis (NPHP) and NPHP-Associated Disorders. Front. Pediatr. 2018, 5, 287. [Google Scholar] [CrossRef] [PubMed]
- Sayer, J.; Otto, E.; O’Toole, J.; Nurnberg, G.; Kennedy, M.; Becker, C.; Hennies, H.; Helou, J.; Attanasio, M.; Fausett, B.; et al. The centrosomal protein nephrocystin-6 is mutated in Joubert syndrome and activates transcription factor ATF4. Nat. Genet. 2006, 38, 674–681. [Google Scholar] [CrossRef]
- Frank, V.; den Hollander, A.; Brüchle, N.; Zonneveld, M.; Nürnberg, G.; Becker, C.; Du Bois, G.; Kendziorra, H.; Roosing, S.; Senderek, J.; et al. Mutations of theCEP290gene encoding a centrosomal protein cause Meckel-Gruber syndrome. Hum. Mutat. 2008, 29, 45–52. [Google Scholar] [CrossRef]
- Romani, M.; Micalizzi, A.; Valente, E. Joubert syndrome: Congenital cerebellar ataxia with the molar tooth. Lancet Neurol. 2013, 12, 894–905. [Google Scholar] [CrossRef]
- Parisi, M. Clinical and molecular features of Joubert syndrome and related disorders. Am. J. Med. Genet. C 2009, 151, 326–340. [Google Scholar] [CrossRef]
- Kheir, A.; Imam, A.; Omer, I.; Hassan, I.; Elamin, S.; Awadalla, E.; Gadalla, M.; Hamdoon, T. Meckel-Gruber syndrome: A rare and lethal anomaly. Sudan. J. Paediatr. 2012, 12, 93–96. [Google Scholar] [PubMed]
- Dell, K. The role of cilia in the pathogenesis of cystic kidney disease. Curr. Opin. Pediatr. 2015, 27, 212–218. [Google Scholar] [CrossRef] [PubMed]
- Hildebrandt, F.; Benzing, T.; Katsanis, N. Ciliopathies. N. Engl. J. Med. 2011, 364, 1533–1543. [Google Scholar] [CrossRef] [PubMed]
- McGrogan, A.; Franssen, C.; de Vries, C. The incidence of primary glomerulonephritis worldwide: A systematic review of the literature. Nephrol. Dial. Transplant. 2010, 26, 414–430. [Google Scholar] [CrossRef] [PubMed]
- Penfold, R.; Prendecki, M.; McAdoo, S.; Tam, F. Primary IgA nephropathy: Current challenges and future prospects. Int. J. Nephrol. Renov. Dis. 2018, 11, 137–148. [Google Scholar] [CrossRef]
- Feehally, J.; Barratt, J. The Genetics of IgA Nephropathy: An Overview from Western Countries. Kidney Dis. 2015, 1, 33–41. [Google Scholar] [CrossRef]
- Schena, F.; Scivittaro, V.; Ranieri, E.; Sinico, R.; Benuzzi, S.; Cillo, M.; Aventaggiato, L. Abnormalities of the IgA immune system in members of unrelated pedigrees from patients with IgA nephropathy. Clin. Exp. Immunol. 2008, 92, 139–144. [Google Scholar] [CrossRef]
- Gharavi, A.; Kiryluk, K.; Choi, M.; Li, Y.; Hou, P.; Xie, J.; Sanna-Cherchi, S.; Men, C.; Julian, B.; Wyatt, R.; et al. Genome-wide association study identifies susceptibility loci for IgA nephropathy. Nat. Genet. 2011, 43, 321–327. [Google Scholar] [CrossRef]
- Rosenberg, A.; Kopp, J. Focal Segmental Glomerulosclerosis. Clin. J. Am. Soc. Nephrol. 2017, 12, 502–517. [Google Scholar] [CrossRef]
- Kitiyakara, C.; Eggers, P.; Kopp, J. Twenty-one-year trend in ESRD due to focal segmental glomerulosclerosis in the United States. Am. J. Kidney Dis. 2004, 44, 815–825. [Google Scholar] [CrossRef]
- D’Agati, V.; Fogo, A.; Bruijn, J.; Jennette, J. Pathologic classification of focal segmental glomerulosclerosis: A working proposal. Am. J. Kidney Dis. 2004, 43, 368–382. [Google Scholar] [CrossRef] [PubMed]
- Stokes, M.; Markowitz, G.; Lin, J.; Valeri, A. D’Agati, V. Glomerular tip lesion: A distinct entity within the minimal change disease/focal segmental glomerulosclerosis spectrum. Kidney Int. 2004, 65, 1690–1702. [Google Scholar] [CrossRef] [PubMed]
- Stokes, M.; Valeri, A.; Markowitz, G.; D’Agati, V. Cellular focal segmental glomerulosclerosis: Clinical and pathologic features. Kidney Int. 2006, 70, 1783–1792. [Google Scholar] [CrossRef] [PubMed]
- Markowitz, G.; Nasr, S.; Stokes, M.; D’Agati, V. Treatment with IFN-α, -β, or -γ Is Associated with Collapsing Focal Segmental Glomerulosclerosis. Clin. J. Am. Soc. Nephrol. 2010, 5, 607–615. [Google Scholar] [CrossRef]
- Wiggins, R. The spectrum of podocytopathies: A unifying view of glomerular diseases. Kidney Int. 2007, 71, 1205–1214. [Google Scholar] [CrossRef]
- Genovese, G.; Friedman, D.; Ross, M.; Lecordier, L.; Uzureau, P.; Freedman, B.; Bowden, D.; Langefeld, C.; Oleksyk, T.; Knob, A.U. Association of Trypanolytic ApoL1 Variants with Kidney Disease in African Americans. Science 2010, 329, 841–845. [Google Scholar] [CrossRef]
- Zhou, D.; Fu, H.; Zhang, L.; Zhang, K.; Min, Y.; Xiao, L.; Lin, L.; Bastacky, S.; Liu, Y. Tubule-Derived Wnts Are Required for Fibroblast Activation and Kidney Fibrosis. J. Am. Soc. Nephrol. 2017, 28, 2322–2336. [Google Scholar] [CrossRef]
- He, S.; Lu, Y.; Liu, X.; Huang, X.; Keller, E.; Qian, C.; Zhang, J. Wnt3a: Functions and implications in cancer. Chin. J. Cancer. 2015, 34, 554–562. [Google Scholar] [CrossRef]
- Fu, H.; Wang, B.; Wan, Z.; Lin, H.; Chang, M.; Han, G. Wnt5a mediated canonical Wnt signaling pathway activation in orthodontic tooth movement: Possible role in the tension force-induced bone formation. J. Mol. Histol. 2016, 47, 455–466. [Google Scholar] [CrossRef]
- Tu, X.; Joeng, K.; Nakayama, K.; Nakayama, K.; Rajagopal, J.; Carroll, T.; McMahon, A.; Long, F. Noncanonical Wnt Signaling through G Protein-Linked PKCδ Activation Promotes Bone Formation. Dev. Cell 2007, 12, 113–127. [Google Scholar] [CrossRef]
- Kestler, H.; Kühl, M. From individual Wnt pathways towards a Wnt signalling network. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2008, 363, 1333–1347. [Google Scholar] [CrossRef] [PubMed]
- Dai, C.; Stolz, D.; Kiss, L.; Monga, S.; Holzman, L.; Liu, Y. Wnt/β-Catenin Signaling Promotes Podocyte Dysfunction and Albuminuria. J. Am. Soc. Nephrol. 2009, 20, 1997–2008. [Google Scholar] [CrossRef] [PubMed]
- Wachi, T.; Yoshida, N.; Funae, Y.; Ueno, M.; Germino, G.; Hirotsune, S.; and Deguchi, N. Progesterone induced mesenchymal differentiation and rescued cystic dilation of renal tubules of Pkd1−/−mice. Biochem. Biophys. Res. Commun. 2012, 425, 212–218. [Google Scholar] [CrossRef] [PubMed]
- Kiewisz, J.; Skowronska, A.; Winiarska, A.; Pawlowska, A.; Kiezun, J.; Rozicka, A.; Perkowska-Ptasinska, A.; Kmiec, Z.; Stompor, T. WNT4 Expression in Primary and Secondary Kidney Diseases: Dependence on Staging. Kidney Blood Press. Res. 2019, 44, 200–210. [Google Scholar] [CrossRef] [PubMed]
- Goggolidou, P.; Hadjirin, N.; Bak, A.; Papakrivopoulou, E.; Hilton, H.; Norris, D.; Dean, C. Atmin mediates kidney morphogenesis by modulating Wnt signaling. Hum. Mol. Genet. 2014, 23, 5303–5316. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Zhang, L.; Zhou, Y.; Zhu, D.; Wang, Q.; Hao, L. Aberrant activation of Wnt pathways in arteries associates with vascular calcification in chronic kidney disease. Int. Urol. Nephrol. 2016, 48, 1313–1319. [Google Scholar] [CrossRef] [PubMed]
- Richards, T.; Modarage, K.; Dean, C.; McCarthy-Boxer, A.; Hilton, H.; Esapa, C.; Norman, J.; Wilson, P.; Goggolidou, P. Atmin modulates Pkhd1 expression and may mediate Autosomal Recessive Polycystic Kidney Disease (ARPKD) through altered non-canonical Wnt/Planar Cell Polarity (PCP) signalling. Biochim. Biophys. Acta. Mol. Basis Dis. 2019, 1865, 378–390. [Google Scholar] [CrossRef]
- Kiefer, S.; Robbins, L.; Rauchman, M. Conditional Expression of Wnt9b in Six2-Positive Cells Disrupts Stomach and Kidney Function. PLoS ONE 2012, 7, e43098. [Google Scholar] [CrossRef]
- Karner, C.; Chirumamilla, R.; Aoki, S.; Igarashi, P.; Wallingford, J.; Carroll, T. Wnt9b signaling regulates planar cell polarity and kidney tubule morphogenesis. Nat. Genet. 2009, 41, 793–799. [Google Scholar] [CrossRef]
- O’Brien, L.; Combes, A.; Short, K.; Lindström, N.; Whitney, P.; Cullen-McEwen, L.; Ju, A.; Abdelhalim, A.; Michos, O.; Bertram, J.; et al. Wnt11 directs nephron progenitor polarity and motile behavior ultimately determining nephron endowment. eLife 2018, 7. [Google Scholar] [CrossRef]
- Cnossen, W.; te Morsche, R.; Hoischen, A.; Gilissen, C.; Venselaar, H.; Mehdi, S.; Bergmann, C.; Losekoot, M.; Breuning, M.; Peters, D.; et al. LRP5 variants may contribute to ADPKD. Eur. J. Hum. Genet. 2015, 24, 237–242. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Stokes, A.; Duan, Z.; Hui, J.; Xu, Y.; Chen, Y.; Chen, H.; Lam, K.; Zhou, C. LDL Receptor–Related Protein 6 Modulates Ret Proto-Oncogene Signaling in Renal Development and Cystic Dysplasia. J. Am. Soc. Nephrol. 2015, 27, 417–427. [Google Scholar] [CrossRef] [PubMed]
- Cox, S.; Sallustio, F.; Serino, G.; Pontrelli, P.; Verrienti, R.; Pesce, F.; Torres, D.; Ancona, N.; Stifanelli, P.; Zaza, G.; et al. Altered modulation of WNT–β-catenin and PI3K/Akt pathways in IgA nephropathy. Kidney Int. 2010, 78, 396–407. [Google Scholar] [CrossRef] [PubMed]
- Saadi-Kheddouci, S.; Berrebi, D.; Romagnolo, B.; Cluzeaud, F.; Peuchmaur, M.; Kahn, A.; Vandewalle, A.; Perret, C. Early development of polycystic kidney disease in transgenic mice expressing an activated mutant of the β-catenin gene. Oncogene 2001, 20, 5972–5981. [Google Scholar] [CrossRef] [PubMed]
- Kim, I.; Ding, T.; Fu, Y.; Li, C.; Cui, L.; Li, A.; Lian, P.; Liang, D.; Wang, D.; Guo, C.; et al. Conditional Mutation of Pkd2 Causes Cystogenesis and Upregulates β-Catenin. J. Am. Soc. Nephrol. 2009, 20, 2556–2569. [Google Scholar] [CrossRef] [PubMed]
- Vilboux, T.; Doherty, D.; Glass, I.; Parisi, M.; Phelps, I.; Cullinane, A.; Zein, W.; Brooks, B.; Heller, T.; Soldatos, A.; et al. Molecular genetic findings and clinical correlations in 100 patients with Joubert syndrome and related disorders prospectively evaluated at a single center. Genet. Med. 2017, 19, 875–882. [Google Scholar] [CrossRef] [PubMed]
- Niehrs, C. Function and biological roles of the Dickkopf family of Wnt modulators. Oncogene 2006, 25, 7469–7481. [Google Scholar] [CrossRef]
- Tanigawa, S.; Wang, H.; Yang, Y.; Sharma, N.; Tarasova, N.; Ajima, R.; Yamaguchi, T.; Rodriguez, L.; Perantoni, A. Wnt4 induces nephronic tubules in metanephric mesenchyme by a non-canonical mechanism. Dev. Biol. 2011, 352, 58–69. [Google Scholar] [CrossRef]
- Zhang, B.; Tran, U.; Wessely, O. Expression of Wnt Signaling Components during Xenopus Pronephros Development. PLoS ONE 2011, 6, e26533. [Google Scholar] [CrossRef]
- Li, B.; Rauhauser, A.; Dai, J.; Sakthivel, R.; Igarashi, P.; Jetten, A.; Attanasio, M. Increased hedgehog signaling in postnatal kidney results in aberrant activation of nephron developmental programs. Hum. Mol. Genet. 2011, 20, 4155–4166. [Google Scholar] [CrossRef]
- Yun, K.; Ajima, R.; Sharma, N.; Costantini, F.; Mackem, S.; Lewandoski, M.; Yamaguchi, T.; Perantoni, A. Non-canonical Wnt5a/Ror2 signaling regulates kidney morphogenesis by controlling intermediate mesoderm extension. Hum. Mol. Genet. 2014, 23, 6807–6814. [Google Scholar] [CrossRef]
- Huang, L.; Xiao, A.; Wecker, A.; McBride, D.; Choi, S.; Zhou, W.; Lipschutz, J. A Possible Zebrafish Model of Polycystic Kidney Disease: Knockdown of wnt5a causes cysts in Zebrafish Kidneys. J. Vis. Exp. 2014. [Google Scholar] [CrossRef] [PubMed]
- Sanna-Cherchi, S.; Khan, K.; Westland, R.; Krithivasan, P.; Fievet, L.; Rasouly, H.; Ionita-Laza, I.; Capone, V.; Fasel, D.; Kiryluk, K.; et al. Exome-wide Association Study Identifies GREB1L Mutations in Congenital Kidney Malformations. Am. J. Hum. Genet. 2017, 101, 789–802. [Google Scholar] [CrossRef] [PubMed]
- Ye, X.; Wang, Y.; Rattner, A.; Nathans, J. Genetic mosaic analysis reveals a major role for frizzled 4 and frizzled 8 in controlling ureteric growth in the developing kidney. Development 2011, 138, 1161–1172. [Google Scholar] [CrossRef] [PubMed]
- Nagy, I.; Xu, Q.; Naillat, F.; Ali, N.; Miinalainen, I.; Samoylenko, A.; Vainio, S. Impairment of Wnt11 function leads to kidney tubular abnormalities and secondary glomerular cystogenesis. BMC Dev. Biol. 2016, 16. [Google Scholar] [CrossRef]
- Sagara, N.; Toda, G.; Hirai, M.; Terada, M.; Katoh, M. Molecular Cloning, Differential Expression, and Chromosomal Localization of HumanFrizzled-1, Frizzled-2, and Frizzled-7. Biochem. Biophys. Res. Commun. 1998, 252, 117–122. [Google Scholar] [CrossRef]
- Sala, C.; Formenti, E.; Terstappen, G.; Caricasole, A. Identification, Gene Structure, and Expression of Human Frizzled-3 (FZD3). Biochem. Biophys. Res. Commun. 2000, 273, 27–34. [Google Scholar] [CrossRef]
- Kirikoshi, H.; Sagara, N.; Koike, J.; Tanaka, K.; Sekihara, H.; Hirai, M.; Katoh, M. Molecular Cloning and Characterization of Human Frizzled-4 on Chromosome 11q14-q21. Biochem. Biophys. Res. Commun. 1999, 264, 955–961. [Google Scholar] [CrossRef]
- Saitoh, T.; Hirai, M.; Katoh, M. Molecular cloning and characterization of human Frizzled-8 gene on chromosome 10p11.2. Int. J. Oncol. 2001, 18, 991–996. [Google Scholar] [CrossRef]
- Tokuhara, M.; Hirai, M.; Atomi, Y.; Terada, M.; Katoh, M. Molecular Cloning of HumanFrizzled-6. Biochem. Biophys. Res. Commun. 1998, 243, 622–627. [Google Scholar] [CrossRef]
- Saitoh, T.; Hirai, M.; Katoh, M. Molecular cloning and characterization of human Frizzled-5 gene on chromosome 2q33.3-q34 region. Int. J. Oncol. 2001, 19, 105–110. [Google Scholar] [CrossRef] [PubMed]
- Chailangkarn, T.; Trujillo, C.; Freitas, B.; Hrvoj-Mihic, B.; Herai, R.; Yu, D.; Brown, T.; Marchetto, M.; Bardy, C.; McHenry, L.; et al. A human neurodevelopmental model for Williams syndrome. Nature 2016, 536, 338–343. [Google Scholar] [CrossRef] [PubMed]
- Koike, J.; Takagi, A.; Miwa, T.; Hirai, M.; Terada, M.; Katoh, M. Molecular Cloning of Frizzled-10, a Novel Member of the Frizzled Gene Family. Biochem. Biophys. Res. Commun. 1999, 262, 39–43. [Google Scholar] [CrossRef]
- Luyten, A.; Su, X.; Gondela, S.; Chen, Y.; Rompani, S.; Takakura, A.; Zhou, J. Aberrant Regulation of Planar Cell Polarity in Polycystic Kidney Disease. J. Am. Soc. Nephrol. 2010, 21, 1521–1532. [Google Scholar] [CrossRef] [PubMed]
- Komiya, Y.; Habas, R. Wnt signal transduction pathways. Organogenesis 2008, 4, 68–75. [Google Scholar] [CrossRef] [PubMed]
- Cnossen, W.; te Morsche, R.; Hoischen, A.; Gilissen, C.; Chrispijn, M.; Venselaar, H.; Mehdi, S.; Bergmann, C.; Veltman, J.; Drenth, J. Whole-exome sequencing reveals LRP5 mutations and canonical Wnt signaling associated with hepatic cystogenesis. Proc. Natl. Acad. Sci. USA 2014, 111, 5343–5348. [Google Scholar] [CrossRef]
- Pinson, K.; Brennan, J.; Monkley, S.; Avery, B.; Skarnes, W. An LDL-receptor-related protein mediates Wnt signalling in mice. Nature 2000, 407, 535–538. [Google Scholar] [CrossRef]
- Lancaster, M.; Louie, C.; Silhavy, J.; Sintasath, L.; DeCambre, M.; Nigam, S.; Willert, K.; Gleeson, J. Impaired Wnt–β-catenin signaling disrupts adult renal homeostasis and leads to cystic kidney ciliopathy. Nat. Med. 2009, 15, 1046–1054. [Google Scholar] [CrossRef]
- Walz, G.; Kim, E. Wnt signaling and rejuvenation of the adult kidney. Nephrol. Dial. Transplant. 2009, 25, 34–36. [Google Scholar] [CrossRef]
- Ossipova, O.; Chu, C.; Fillatre, J.; Brott, B.; Itoh, K.; Sokol, S. The involvement of PCP proteins in radial cell intercalations during Xenopus embryonic development. Dev. Biol. 2015, 408, 316–327. [Google Scholar] [CrossRef]
- Papakrivopoulou, E.; Vasilopoulou, E.; Lindenmeyer, M.; Pacheco, S.; Brzóska, H.; Price, K.; Kolatsi-Joannou, M.; White, K.; Henderson, D.; Dean, C.; et al. Vangl2, a planar cell polarity molecule, is implicated in irreversible and reversible kidney glomerular injury. J. Pathol. 2018, 246, 485–496. [Google Scholar] [CrossRef] [PubMed]
- Yates, L.; Papakrivopoulou, J.; Long, D.; Goggolidou, P.; Connolly, J.; Woolf, A.; Dean, C. The planar cell polarity gene Vangl2 is required for mammalian kidney-branching morphogenesis and glomerular maturation. Hum. Mol. Genet. 2010, 19, 4663–4676. [Google Scholar] [CrossRef] [PubMed]
- Goffinet, A.; Tissir, F. Seven pass Cadherins CELSR1-3. Semin. Cell Dev. Biol. 2017, 69, 102–110. [Google Scholar] [CrossRef] [PubMed]
- Brzóska, H.; d’Esposito, A.; Kolatsi-Joannou, M.; Patel, V.; Igarashi, P.; Lei, Y.; Finnell, R.; Lythgoe, M.; Woolf, A.; Papakrivopoulou, E.; et al. Planar cell polarity genes Celsr1 and Vangl2 are necessary for kidney growth, differentiation, and rostrocaudal patterning. Kidney Int. 2016, 90, 1274–1284. [Google Scholar] [CrossRef]
- Xu, D.; Lv, J.; He, L.; Fu, L.; Hu, R.; Cao, Y.; Mei, C. Scribble influences cyst formation in autosomal-dominant polycystic kidney disease by regulating Hippo signaling pathway. FASEB J. 2018, 32, 4394–4407. [Google Scholar] [CrossRef]
- Skouloudaki, K.; Puetz, M.; Simons, M.; Courbard, J.; Boehlke, C.; Hartleben, B.; Engel, C.; Moeller, M.; Englert, C.; Bollig, F.; et al. Scribble participates in Hippo signaling and is required for normal zebrafish pronephros development. Proc. Natl. Acad. Sci. USA 2009, 106, 8579–8584. [Google Scholar] [CrossRef]
- Zuo, Y.; Liu, Y. New insights into the role and mechanism of Wnt/β-catenin signalling in kidney fibrosis. Nephrology 2018, 23, 38–43. [Google Scholar] [CrossRef]
- Zhou, D.; Tan, R.; Fu, H.; Liu, Y. Wnt/β-catenin signaling in kidney injury and repair: A double-edged sword. Lab. Investig. 2015, 96, 156–167. [Google Scholar] [CrossRef]
- Gewin, L. Renal Tubule Repair: Is Wnt/β-Catenin a Friend or Foe? Genes 2018, 9, 58. [Google Scholar] [CrossRef]
- Xiao, L.; Zhou, D.; Tan, R.; Fu, H.; Zhou, L.; Hou, F.; Liu, Y. Sustained Activation of Wnt/β-Catenin Signaling Drives AKI to CKD Progression. J. Am. Soc. Nephrol. 2015, 27, 1727–1740. [Google Scholar] [CrossRef]
- Tan, R.; Zhou, D.; Zhou, L.; Liu, Y. Wnt/β-catenin signaling and kidney fibrosis. Kidney Int. Suppl. 2014, 4, 84–90. [Google Scholar] [CrossRef] [PubMed]
- He, W.; Dai, C.; Li, Y.; Zeng, G.; Monga, S.; Liu, Y. Wnt/β-Catenin Signaling Promotes Renal Interstitial Fibrosis. J. Am. Soc. Nephrol. 2009, 20, 765–776. [Google Scholar] [CrossRef] [PubMed]
- Surendran, K.; Schiavi, S.; Hruska, K. Wnt-Dependent β-Catenin Signaling Is Activated after Unilateral Ureteral Obstruction, and Recombinant Secreted Frizzled-Related Protein 4 Alters the Progression of Renal Fibrosis. J. Am. Soc. Nephrol. 2005, 16, 2373–2384. [Google Scholar] [CrossRef] [PubMed]
- Kato, H.; Gruenwald, A.; Suh, J.; Miner, J.; Barisoni-Thomas, L.; Taketo, M.; Faul, C.; Millar, S.; Holzman, L.; Susztak, K. Wnt/β-Catenin Pathway in Podocytes Integrates Cell Adhesion, Differentiation, and Survival. J. Biol. Chem. 2011, 286, 26003–26015. [Google Scholar] [CrossRef] [PubMed]
- von Toerne, C.; Schmidt, C.; Adams, J.; Kiss, E.; Bedke, J.; Porubsky, S.; Gretz, N.; Lindenmeyer, M.; Cohen, C.; Gröne, H.; et al. Wnt Pathway Regulation in Chronic Renal Allograft Damage. Am. J. Transplant. 2009, 9, 2223–2239. [Google Scholar] [CrossRef]
- Wang, X.; Huang, X.; Yan, Q.; Bao, C. Aberrant Activation of the WNT/β-Catenin Signaling Pathway in Lupus Nephritis. PLoS ONE 2014, 9, e84852. [Google Scholar] [CrossRef] [PubMed]
- He, W.; Tan, R.; Li, Y.; Wang, D.; Nie, J.; Hou, F.; Liu, Y. Matrix Metalloproteinase-7 as a Surrogate Marker Predicts Renal Wnt/β-Catenin Activity in CKD. J. Am. Soc. Nephrol. 2011, 23, 294–304. [Google Scholar] [CrossRef]
- Hao, S.; He, W.; Li, Y.; Ding, H.; Hou, Y.; Nie, J.; Hou, F.; Kahn, M.; Liu, Y. Targeted inhibition of β-catenin/CBP signaling ameliorates renal interstitial fibrosis. J. Am. Soc. Nephrol. 2011, 22, 1642–1653. [Google Scholar] [CrossRef]
- Hamano, K.; Iwano, M.; Akai, Y.; Sato, H.; Kubo, A.; Nishitani, Y.; Uyama, H.; Yoshida, Y.; Miyazaki, M.; Shiiki, H.; et al. Expression of glomerular plasminogen activator inhibitor type 1 in glomerulonephritis. Am. J. Kidney Dis. 2002, 39, 695–705. [Google Scholar] [CrossRef]
- Li, Z.; Xu, J.; Xu, P.; Liu, S.; Yang, Z. Wnt/β-catenin signalling pathway mediates high glucose induced cell injury through activation of TRPC6 in podocytes. Cell Prolif. 2013, 46, 76–85. [Google Scholar] [CrossRef]
- Winn, M.; Conlon, P.; Lynn, K.; Farrington, M.; Creazzo, T.; Hawkins, A.; Daskalakis, N.; Kwan, S.; Ebersviller, S.; Burchette, J.; et al. A Mutation in the TRPC6 Cation Channel Causes Familial Focal Segmental Glomerulosclerosis. Science 2005, 308, 1801–1804. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Zhou, C.; Liu, Y. Wnt Signaling in Kidney Development and Disease. Prog. Mol. Biol. Transl. Sci. 2018, 153, 181–207. [Google Scholar] [CrossRef] [PubMed]
- Zschiedrich, S.; Budde, K.; Nürnberger, J.; Wanner, C.; Sommerer, C.; Kunzendorf, U.; Banas, B.; Hoerl, W.; Obermüller, N.; Arns, W.; et al. Secreted frizzled-related protein 4 predicts progression of autosomal dominant polycystic kidney disease. Nephrol. Dial. Transplant. 2015, 31, 284–289. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Romaker, D.; Puetz, M.; Teschner, S.; Donauer, J.; Geyer, M.; Gerke, P.; Rumberger, B.; Dworniczak, B.; Pennekamp, P.; Buchholz, B.; et al. Increased Expression of Secreted Frizzled-Related Protein 4 in Polycystic Kidneys. J. Am. Soc. Nephrol. 2008, 20, 48–56. [Google Scholar] [CrossRef]
- Cheyette, B.; Waxman, J.; Miller, J.; Takemaru, K.; Sheldahl, L.; Khlebtsova, N.; Fox, E.; Earnest, T.; Moon, R. Dapper, a Dishevelled-Associated Antagonist of β-Catenin and JNK Signaling, Is Required for Notochord Formation. Dev. Cell. 2002, 2, 449–461. [Google Scholar] [CrossRef]
- Xue, H.; Xiao, Z.; Zhang, J.; Wen, J.; Wang, Y.; Chang, Z.; Zhao, J.; Gao, X.; Du, J.; Chen, Y. Disruption of theDapper3Gene Aggravates Ureteral Obstruction-mediated Renal Fibrosis by Amplifying Wnt/β-catenin Signaling. J. Biol. Chem. 2013, 288, 15006–15014. [Google Scholar] [CrossRef]
- Zhou, L.; Li, Y.; Hao, S.; Zhou, D.; Tan, R.; Nie, J.; Hou, F.; Kahn, M.; Liu, Y. Multiple Genes of the Renin-Angiotensin System Are Novel Targets of Wnt/β-Catenin Signaling. J. Am. Soc. Nephrol. 2014, 26, 107–120. [Google Scholar] [CrossRef]
- Li, A.; Xu, Y.; Fan, S.; Meng, J.; Shen, X.; Xiao, Q.; Li, Y.; Zhang, L.; Zhang, X.; Wu, G.; et al. Canonical Wnt inhibitors ameliorate cystogenesis in a mouse ortholog of human ADPKD. JCI Insight 2018, 3, e95874. [Google Scholar] [CrossRef]
- Qian, C.; Knol, J.; Igarashi, P.; Lin, F.; Zylstra, U.; Teh, B.; Williams, B. Cystic Renal Neoplasia Following Conditional Inactivation of Apc in Mouse Renal Tubular Epithelium. J. Biol. Chem. 2004, 280, 3938–3945. [Google Scholar] [CrossRef]
- Miller, M.; Iglesias, D.; Zhang, Z.; Corsini, R.; Chu, L.; Murawski, I.; Gupta, I.; Somlo, S.; Germino, G.; Goodyer, P. T-cell factor/β-catenin activity is suppressed in two different models of autosomal dominant polycystic kidney disease. Kidney Int. 2011, 80, 146–153. [Google Scholar] [CrossRef]
- Lustig, B.; Jerchow, B.; Sachs, M.; Weiler, S.; Pietsch, T.; Karsten, U.; van de Wetering, M.; Clevers, H.; Schlag, P.; Birchmeier, W.; et al. Negative Feedback Loop of Wnt Signaling through Upregulation of Conductin/Axin2 in Colorectal and Liver Tumors. Mol. Cell. Biol. 2002, 22, 1184–1193. [Google Scholar] [CrossRef] [PubMed]
- He, T. Identification of c-MYC as a Target of the APC Pathway. Science 1998, 281, 1509–1512. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Li, F.; Sun, Y.; Lei, L.; Zhou, H.; Lei, T.; Xia, Y.; Verkman, A.; Yang, B. Aquaporin-1 retards renal cyst development in polycystic kidney disease by inhibition of Wnt signaling. FASEB J. 2015, 29, 1551–1563. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.; Nie, H.; Nesin, V.; Tran, U.; Outeda, P.; Bai, C.; Keeling, J.; Maskey, D.; Watnick, T.; Wessely, O.; et al. The polycystin complex mediates Wnt/Ca2+ signalling. Nat. Cell Biol. 2016, 18, 752–764. [Google Scholar] [CrossRef] [PubMed]
- McCoy, K.; Zhou, X.; Vize, P. Non-canonical wnt signals antagonize and canonical wnt signals promote cell proliferation in early kidney development. Dev. Dyn. 2011, 240, 1558–1566. [Google Scholar] [CrossRef] [PubMed]
- Simons, M.; Gloy, J.; Ganner, A.; Bullerkotte, A.; Bashkurov, M.; Krönig, C.; Schermer, B.; Benzing, T.; Cabello, O.; Jenny, A.; et al. Inversin, the gene product mutated in nephronophthisis type II, functions as a molecular switch between Wnt signaling pathways. Nat. Genet. 2005, 37, 537–543. [Google Scholar] [CrossRef] [PubMed]
- Lienkamp, S.; Ganner, A.; Boehlke, C.; Schmidt, T.; Arnold, S.; Schafer, T.; Romaker, D.; Schuler, J.; Hoff, S.; Powelske, C.; et al. Inversin relays Frizzled-8 signals to promote proximal pronephros development. Proc. Natl. Acad. Sci. USA 2010, 107, 20388–20393. [Google Scholar] [CrossRef]
- Veland, I.; Montjean, R.; Eley, L.; Pedersen, L.; Schwab, A.; Goodship, J.; Kristiansen, K.; Pedersen, S.; Saunier, S.; Christensen, S. Inversin/Nephrocystin-2 Is Required for Fibroblast Polarity and Directional Cell Migration. PLoS ONE 2013, 8, e60193. [Google Scholar] [CrossRef]
- Otto, E.; Schermer, B.; Obara, T.; O’Toole, J.; Hiller, K.; Mueller, A.; Ruf, R.; Hoefele, J.; Beekmann, F.; Landau, D.; et al. Mutations in INVS encoding inversin cause nephronophthisis type 2, linking renal cystic disease to the function of primary cilia and left-right axis determination. Nat. Genet. 2003, 34, 413–420. [Google Scholar] [CrossRef]
- Watanabe, D. The left-right determinant Inversin is a component of node monocilia and other 9+0 cilia. Development 2003, 130, 1725–1734. [Google Scholar] [CrossRef]
- Tory, K.; Rousset-Rouvière, C.; Gubler, M.; Morinière, V.; Pawtowski, A.; Becker, C.; Guyot, C.; Gié, S.; Frishberg, Y.; Nivet, H.; et al. Mutations of NPHP2 and NPHP3 in infantile nephronophthisis. Kidney Int. 2009, 75, 839–847. [Google Scholar] [CrossRef] [PubMed]
- Bellavia, S.; Dahan, K.; Terryn, S.; Cosyns, J.; Devuyst, O.; Pirson, Y. A homozygous mutation in INVS causing juvenile nephronophthisis with abnormal reactivity of the Wnt/β-catenin pathway. Nephrol. Dial. Transplant. 2010, 25, 4097–4102. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Borgal, L.; Habbig, S.; Hatzold, J.; Liebau, M.; Dafinger, C.; Sacarea, I.; Hammerschmidt, M.; Benzing, T.; Schermer, B. The Ciliary Protein Nephrocystin-4 Translocates the Canonical Wnt Regulator Jade-1 to the Nucleus to Negatively Regulate β-Catenin Signaling. J. Biol. Chem. 2012, 287, 25370–25380. [Google Scholar] [CrossRef] [PubMed]
- Burcklé, C.; Gaudé, H.; Vesque, C.; Silbermann, F.; Salomon, R.; Jeanpierre, C.; Antignac, C.; Saunier, S.; Schneider-Maunoury, S. Control of the Wnt pathways by nephrocystin-4 is required for morphogenesis of the zebrafish pronephros. Hum. Mol. Genet. 2011, 20, 2611–2627. [Google Scholar] [CrossRef]
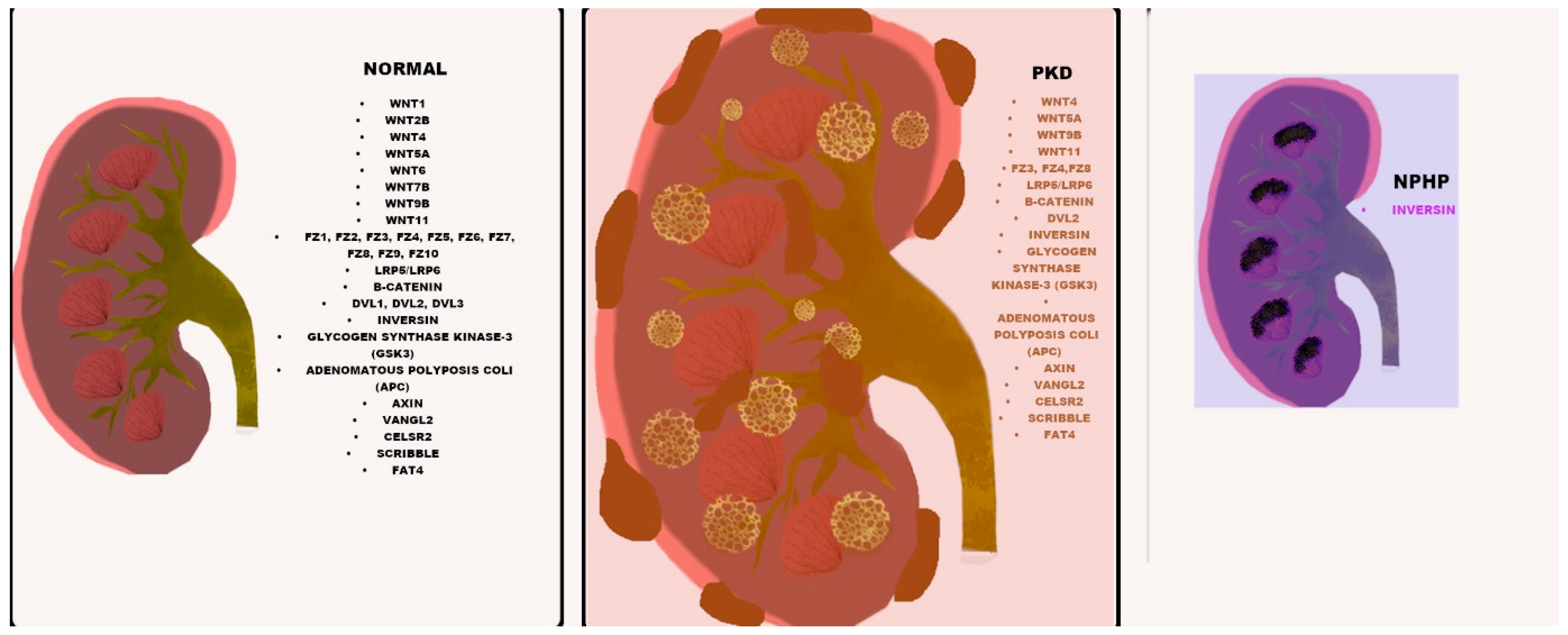
| Disease | Brief Description | Associated Genes | References |
|---|---|---|---|
| Autosomal Dominant Polycystic Kidney Disease (ADPKD) |
|
| [2,3,4,5] |
| Autosomal Recessive Polycystic Kidney Disease (ARPKD) |
|
| [6,7,8,9,10] |
| Nephronophthisis (NPHP) |
|
| [11,12,13,14,15,16] |
| Joubert Syndrome (JS) |
|
| [16,17,18,19,20] |
| Meckel-Gruber syndrome (MGS) |
|
| [16,17,18,19,21,22,23] |
| IgA nephropathy (IgAN) |
|
| [24,25,26,27,28] |
| Focal and segmental glomerulosclerosis (FSGS) |
|
| [29,30,31,32,33,34,35,36] |
| Wnt Component | Disease | Model | References |
|---|---|---|---|
| WNT1 | FSGS | Human | [42] |
| WNT4 | PKD, Membranous Nephropathy | Human, mouse | [43,44] |
| WNT5A | ARPKD, ESRD | Human | [45,46,47] |
| WNT9B | PKD | Mouse | [48,49] |
| WNT11 | PKD | Mouse | [50] |
| LRP5 | ADPKD | Human, cell line (HEK293) | [51] |
| LRP6 | PKD | Mouse | [52] |
| β-catenin | ADPKD, IgAN, FSGS | Human, mouse, cell line (renal collecting duct cell line) | [42,53,54,55] |
| NPHP2/Inversin | ADPKD, IgAN, NPHP | Human | [53] |
| VANGL2 | ARPKD | Human | [47] |
| CELSR2 | JS | Human | [56] |
| SCRIBBLE | ARPKD | Human | [47] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Malik, S.A.; Modarage, K.; Goggolidou, P. The Role of Wnt Signalling in Chronic Kidney Disease (CKD). Genes 2020, 11, 496. https://doi.org/10.3390/genes11050496
Malik SA, Modarage K, Goggolidou P. The Role of Wnt Signalling in Chronic Kidney Disease (CKD). Genes. 2020; 11(5):496. https://doi.org/10.3390/genes11050496
Chicago/Turabian StyleMalik, Soniya A., Kavindiya Modarage, and Paraskevi Goggolidou. 2020. "The Role of Wnt Signalling in Chronic Kidney Disease (CKD)" Genes 11, no. 5: 496. https://doi.org/10.3390/genes11050496
APA StyleMalik, S. A., Modarage, K., & Goggolidou, P. (2020). The Role of Wnt Signalling in Chronic Kidney Disease (CKD). Genes, 11(5), 496. https://doi.org/10.3390/genes11050496





