Microfluidic Quantitative PCR Detection of 12 Transgenes from Horse Plasma for Gene Doping Control
Abstract
1. Introduction
2. Materials and Methods
2.1. Ethical Considerations
2.2. Blood Collection and DNA Extraction
2.3. Animal Genomic DNA Preparations
2.4. Preparation of Cloned Horse Transgenes as Reference Material
2.5. Quantitation of Transgenes Cloned into Plasmid Vector
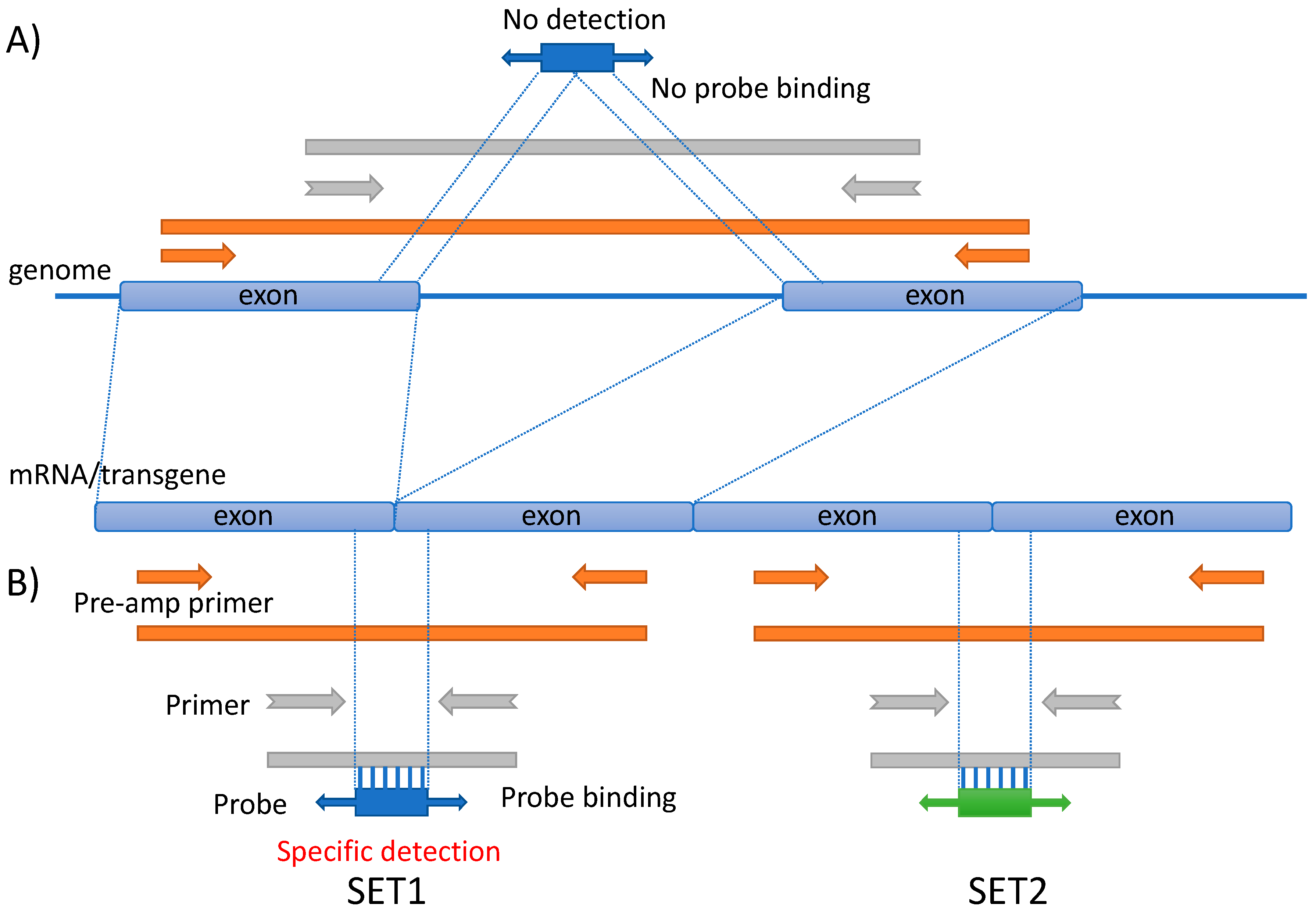
2.6. Designing Primers and Probes for MFQPCR Detection
2.7. Preparation and Quantification of Control Samples
2.8. MFQPCR Detection Using Dynamic Array Integrated Fluid Circuits (IFCs)
2.9. Administration of EPO Transgene
3. Results and Discussion
3.1. Targeted Genes for Gene-Doping Control
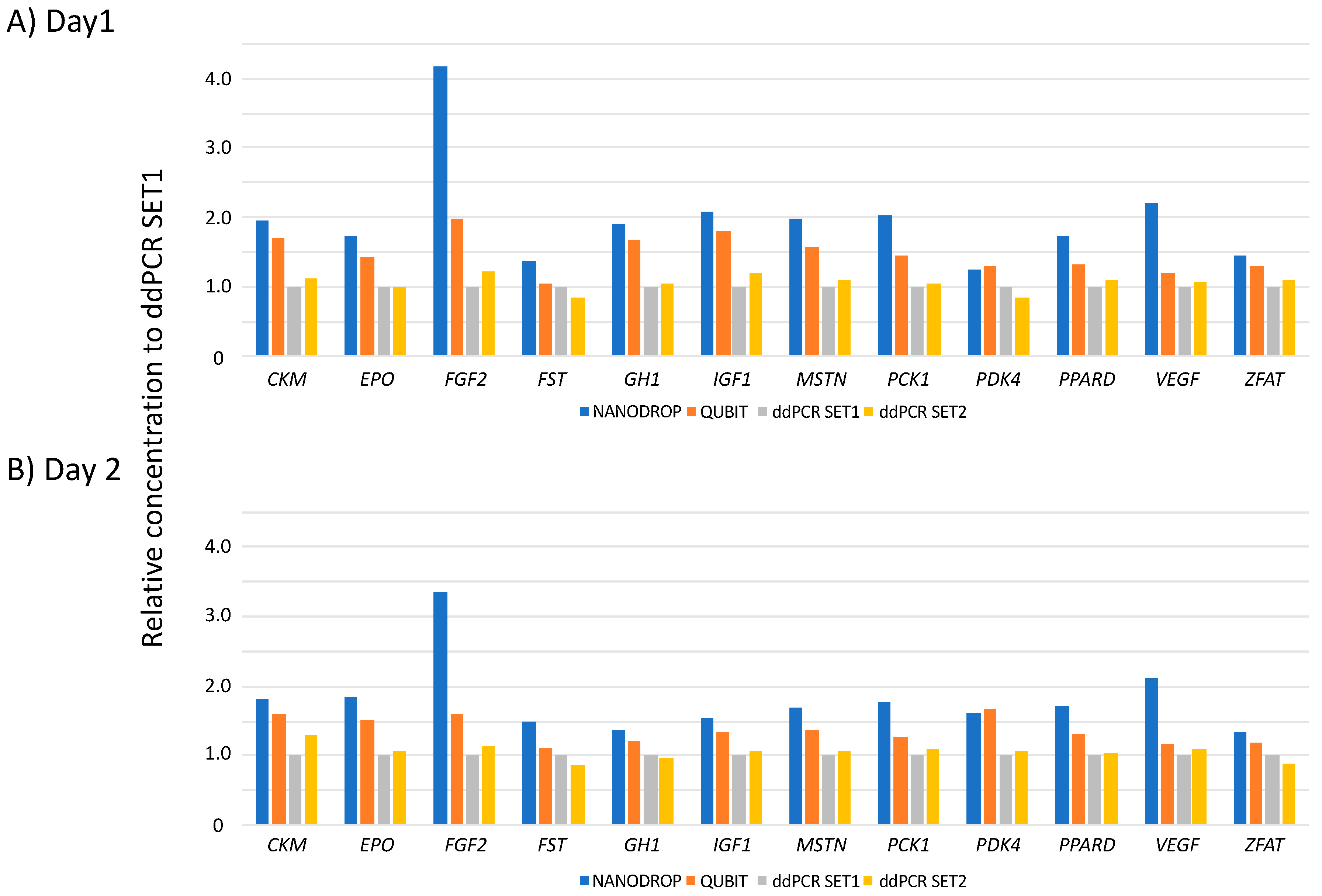
3.2. Quantitation of Reference Materials (RMs)
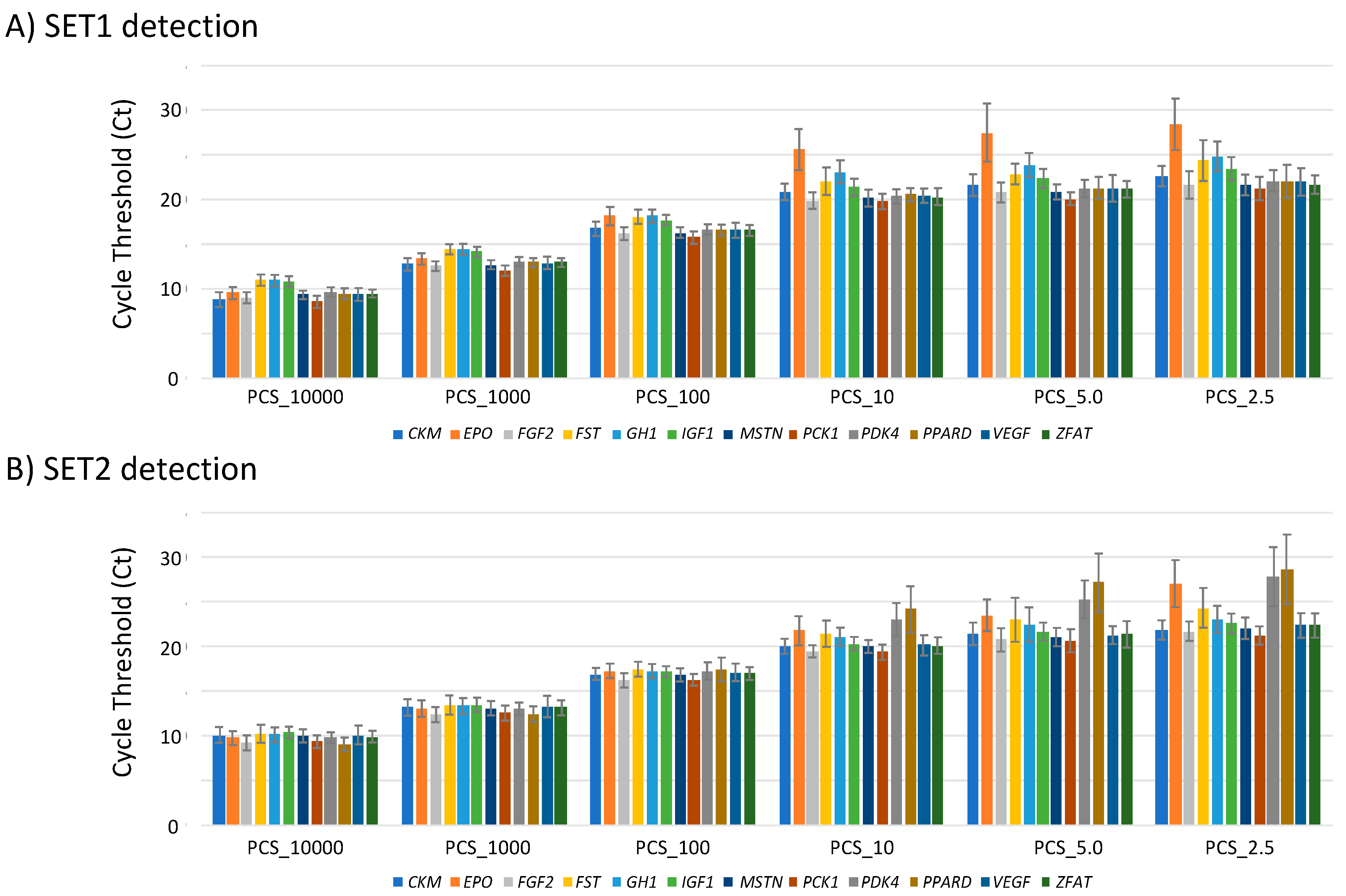
3.3. Limit of Detection (LOD) of MFQPCR
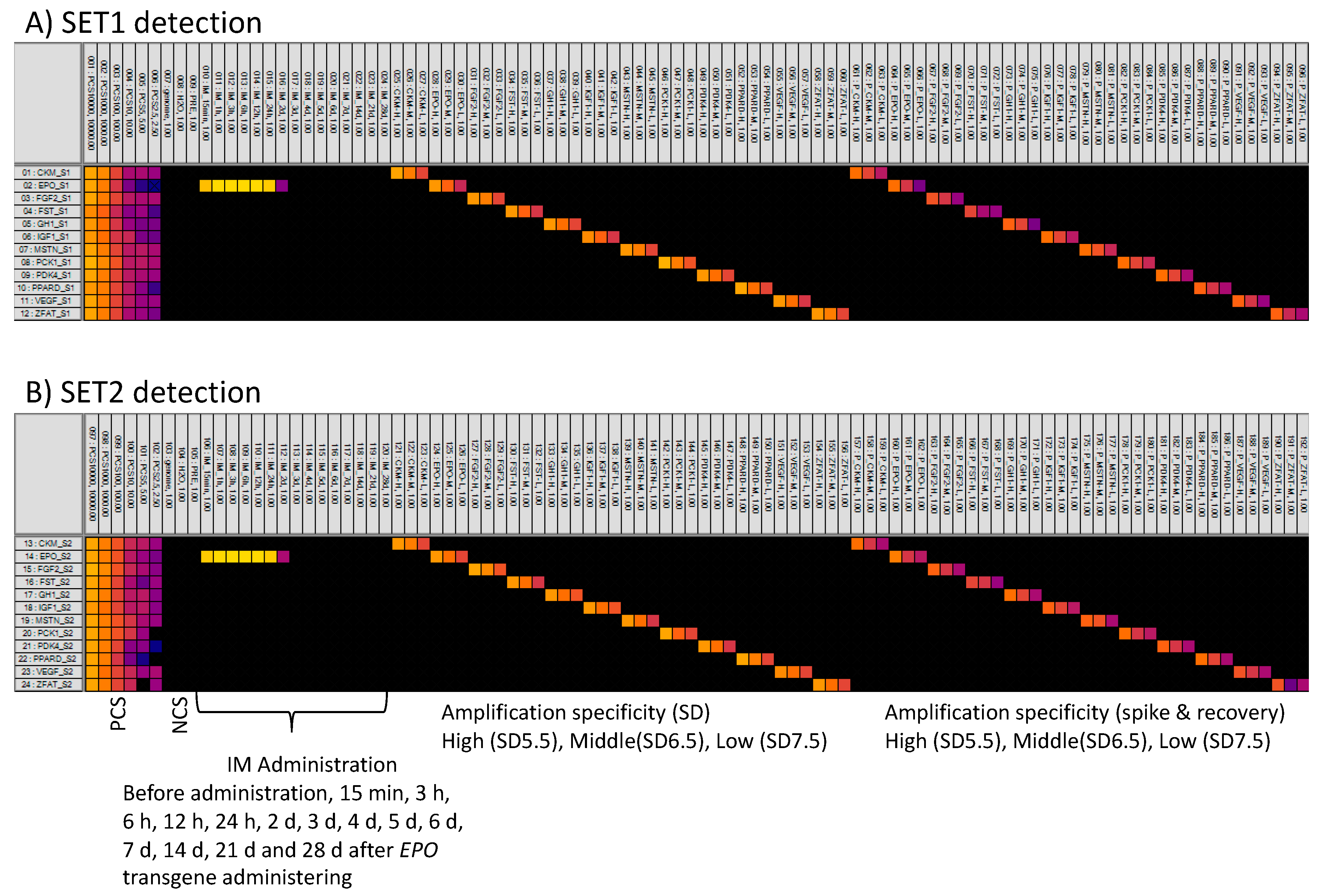
3.4. MFQPCR-Amplification Specificity
3.5. Detection of the EPO Transgene After It Was Administered to a Horse
3.6. Casework Example
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Wong, J.K.; Wan, T.S. Doping control analyses in horseracing: A clinician’s guide. Vet. J. 2014, 200, 8–16. [Google Scholar] [CrossRef] [PubMed]
- Eenoo, P.V.; Delbeke, F.T. Detection of inhaled salbutamol in equine urine by ELISA and GC/MS2. Biomed. Chromatogr. 2002, 16, 513–516. [Google Scholar] [CrossRef] [PubMed]
- Wong, C.H.; Leung, D.K.; Tang, F.P.; Wong, J.K.; Yu, N.H.; Wan, T.S. Rapid screening of anabolic steroids in horse urine with ultra-high-performance liquid chromatography/tandem mass spectrometry after chemical derivatisation. J. Chromatogr. A 2012, 1232, 257–265. [Google Scholar] [CrossRef] [PubMed]
- Kovac, M.; Litvin, Y.A.; Aliev, R.O.; Zakirova, E.Y.; Rutland, C.S.; Kiyasov, A.P.; Rizvanov, A.A. Gene therapy using plasmid DNA encoding VEGF164 and FGF2 genes: A novel treatment of naturally occurring tendinitis and desmitis in horses. Front. Pharmacol. 2018, 9, 978. [Google Scholar] [CrossRef]
- Ishihara, A.; Shields, K.M.; Litsky, A.S.; Mattoon, J.S.; Weisbrode, S.E.; Bartlett, J.S.; Bertone, A.L. Osteogenic gene regulation and relative acceleration of healing by adenoviral-mediated transfer of human BMP-2 or -6 in equine osteotomy and ostectomy models. J. Orthop. Res. 2008, 26, 764–771. [Google Scholar] [CrossRef]
- Moss, K.L.; Jiang, Z.; Dodson, M.E.; Linardi, R.L.; Haughan, J.; Gale, A.L.; Grzybowski, C.; Engiles, J.E.; Stefanovski, D.; Robinson, M.A.; et al. Sustained Interleukin-10 Transgene Expression Following Intra-Articular AAV5-IL-10 Administration to Horses. Hum. Gene Ther. 2020, 31, 110–118. [Google Scholar] [CrossRef]
- Nathwani, A.C.; Davidoff, A.M.; Tuddenham, E.G.D. Gene Therapy for Hemophilia. Hematol. Oncol. Clin. N. Am. 2017, 31, 853–868. [Google Scholar] [CrossRef]
- Kokkinos, J.; Ignacio, R.M.C.; Sharbeen, G.; Boyer, C.; Gonzales-Aloy, E.; Goldstein, D.; Australian Pancreatic Cancer Genome Initiative (APGI). Targeting the undruggable in pancreatic cancer using nano-based gene silencing drugs. Biomaterials 2020, 240, 119742. [Google Scholar] [CrossRef]
- Skipper, K.A.; Mikkelsen, J.G. Toward In Vivo Gene Therapy Using CRISPR. Methods Mol. Biol. 2019, 1961, 293–306. [Google Scholar]
- Wilkin, T.; Baoutina, A.; Hamilton, N. Equine performance genes and the future of doping in horseracing. Drug Test. Anal. 2017, 9, 1456–1471. [Google Scholar] [CrossRef]
- Tozaki, T.; Karasawa, K.; Minamijima, Y.; Ishii, H.; Kikuchi, M.; Kakoi, H.; Hirota, K.; Kusano, K.; Nagata, S. Detection of phosphorothioated (PS) oligonucleotides in horse plasma using a product ion (m/z 94.9362) derived from the PS moiety for doping control. BMC Res. Notes 2018, 11, 770. [Google Scholar] [CrossRef]
- Neuberger, E.W.; Perez, I.; Le Guiner, C.; Moser, D.; Ehlert, T.; Allais, M.; Moullier, P.; Simon, P.; Snyder, R.O. Establishment of two quantitative nested qPCR assays targeting the human EPO transgene. Gene Ther. 2016, 23, 330–339. [Google Scholar] [CrossRef] [PubMed]
- Perez, I.C.; Le Guiner, C.; Ni, W.; Lyles, J.; Moullier, P.; Snyder, R.O. PCR-based detection of gene transfer vectors: Application to gene doping surveillance. Anal. Bioanal. Chem. 2013, 405, 9641–9653. [Google Scholar] [CrossRef]
- Sugasawa, T.; Aoki, K.; Watanabe, K.; Yanazawa, K.; Natsume, T.; Takemasa, T.; Yamaguchi, K.; Takeuchi, Y.; Aita, Y.; Yahagi, N.; et al. Detection of transgenes in gene delivery model mice by adenoviral vector using ddPCR. Genes 2019, 10, 436. [Google Scholar] [CrossRef] [PubMed]
- Tozaki, T.; Gamo, S.; Takasu, M.; Kikuchi, M.; Kakoi, H.; Hirota, K.; Kusano, K.; Nagata, S. Digital PCR detection of plasmid DNA administered to the skeletal muscle of a microminipig: A model case study for gene doping detection. BMC Res. Notes 2018, 11, 708. [Google Scholar] [CrossRef] [PubMed]
- Tozaki, T.; Ohnuma, A.; Takasu, M.; Kikuchi, M.; Kakoi, H.; Hirota, K.; Kusano, K.; Nagata, S. Droplet digital PCR Detection of the erythropoietin transgene from horse plasma and urine for gene-doping control. Genes 2019, 10, 243. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.J.; Xu, J.F.; Shen, Y.W.; Ma, S.J.; Zhang, T.T.; Meng, Q.L.; Lan, W.J.; Zhang, C.; Liu, X.M. Detection of exogenous gene doping of IGF-I by a real-time quantitative PCR assay. Biotechnol. Appl. Biochem. 2017, 64, 549–554. [Google Scholar] [CrossRef]
- de Boer, E.N.; van der Wouden, P.E.; Johansson, L.F.; van Diemen, C.C.; Haisma, H.J. A next-generation sequencing method for gene doping detection that distinguishes low levels of plasmid DNA against a background of genomic DNA. Gene Ther. 2019, 26, 338–346. [Google Scholar] [CrossRef]
- Ishii, S.; Segawa, T.; Okabe, S. Simultaneous quantification of multiple food- and waterborne pathogens by use of microfluidic quantitative PCR. Appl. Environ. Microbiol. 2013, 79, 2891–2898. [Google Scholar] [CrossRef]
- Echegaray, M.; Rivera, M.A. Role of creatine kinase isoenzymes on muscular and cardiorespiratory endurance: Genetic and molecular evidence. Sports Med. 2001, 31, 919–934. [Google Scholar] [CrossRef]
- Latorre-Muro, P.; Baeza, J.; Armstrong, E.A.; Hurtado-Guerrero, R.; Corzana, F.; Wu, L.E.; Sinclair, D.A.; López-Buesa, P.; Carrodeguas, J.A.; Denu, J.M. Dynamic Acetylation of phosphoenolpyruvate carboxykinase toggles enzyme activity between gluconeogenic and anaplerotic reactions. Mol. Cell. 2018, 71, 718–732. [Google Scholar] [CrossRef] [PubMed]
- Connaughton, S.; Chowdhury, F.; Attia, R.R.; Song, S.; Zhang, Y.; Elam, M.B.; Cook, G.A.; Park, E.A. Regulation of pyruvate dehydrogenase kinase isoform 4 (PDK4) gene expression by glucocorticoids and insulin. Mol. Cell Endocrinol. 2010, 315, 159–167. [Google Scholar] [CrossRef]
- Liu, Y.; Colby, J.K.; Zuo, X.; Jaoude, J.; Wei, D.; Shureiqi, I. The role of PPAR-δ in metabolism, inflammation, and cancer: Many characters of a critical transcription factor. Int. J. Mol. Sci. 2018, 19, E3339. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez, S.; Gaunt, T.R.; Day, I.N. Molecular genetics of human growth hormone, insulin-like growth factors and their pathways in common disease. Hum. Genet. 2007, 122, 1–21. [Google Scholar] [CrossRef] [PubMed]
- Juul, S.; Felderhoff-Mueser, U. Epo and other hematopoietic factors. Semin. Fetal Neonatal Med. 2007, 12, 250–258. [Google Scholar] [CrossRef] [PubMed]
- Moser, D.A.; Neuberger, E.W.; Simon, P. A quick one-tube nested PCR-protocol for EPO transgene detection. Drug. Test. Anal. 2012, 4, 870–875. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.J.; Lee, Y.S.; Zimmers, T.A.; Soleimani, A.; Matzuk, M.M.; Tsuchida, K.; Cohn, R.D.; Barton, E.R. Regulation of muscle mass by follistatin and activins. Mol. Endocrinol. 2010, 24, 1998–2008. [Google Scholar] [CrossRef]
- Tozaki, T.; Kikuchi, M.; Kakoi, H.; Hirota, K.I.; Nagata, S.I. A genome-wide association study for body weight in Japanese Thoroughbred racehorses clarifies candidate regions on chromosomes 3, 9, 15, and 18. J. Equine Sci. 2017, 28, 127–134. [Google Scholar] [CrossRef]
- Gu, J.; MacHugh, D.E.; McGivney, B.A.; Park, S.D.; Katz, L.M.; Hill, E.W. Association of sequence variants in CKM (creatine kinase, muscle) and COX4I2 (cytochrome c oxidase, subunit 4, isoform 2) genes with racing performance in Thoroughbred horses. Equine Vet. J. Suppl. 2010, 38, 569–575. [Google Scholar] [CrossRef]
- Hill, E.W.; Gu, J.; Eivers, S.S.; Fonseca, R.G.; McGivney, B.A.; Govindarajan, P.; Orr, N.; Katz, L.M.; MacHugh, D.E. A sequence polymorphism in MSTN predicts sprinting ability and racing stamina in thoroughbred horses. PLoS ONE 2010, 5, e8645. [Google Scholar] [CrossRef]
- Tozaki, T.; Miyake, T.; Kakoi, H.; Gawahara, H.; Sugita, S.; Hasegawa, T.; Ishida, N.; Hirota, K.; Nakano, Y. A genome-wide association study for racing performances in Thoroughbreds clarifies a candidate region near the MSTN gene. Anim. Genet. 2010, 41 (Suppl. 2), 28–35. [Google Scholar] [CrossRef] [PubMed]
- Tozaki, T.; Hill, E.W.; Hirota, K.; Kakoi, H.; Gawahara, H.; Miyake, T.; Sugita, S.; Hasegawa, T.; Ishida, N.; Nakano, Y.; et al. A cohort study of racing performance in Japanese Thoroughbred racehorses using genome information on ECA18. Anim. Genet. 2012, 43, 42–52. [Google Scholar] [CrossRef] [PubMed]
- Wade, C.M.; Giulotto, E.; Sigurdsson, S.; Zoli, M.; Gnerre, S.; Imsland, F.; Lear, T.L.; Adelson, D.L.; Bailey, E.; Bellone, R.R.; et al. Genome sequence, comparative analysis, and population genetics of the domestic horse. Science 2009, 326, 865–867. [Google Scholar] [CrossRef] [PubMed]
- Sarzynski, M.A.; Loos, R.J.; Lucia, A.; Pérusse, L.; Roth, S.M.; Wolfarth, B.; Rankinen, T.; Bouchard, C. Advances in Exercise, Fitness, and Performance Genomics in 2015. Med. Sci. Sports Exerc. 2016, 48, 1906–1916. [Google Scholar] [CrossRef] [PubMed]
| Gene Name | Symbol | Function or Related Traits |
|---|---|---|
| creatine kinase, muscle | CKM | energy transduction |
| erythropoietin | EPO | hematopoiesis |
| fibroblast growth factor 2 | FGF2 | cell growth, morphogenesis, tissue repair |
| follistatin | FST | muscle growth (antagonist of MSTN) |
| growth hormone 1 | GH1 | growth control |
| insulin like growth factor 1 | IGF1 | systemic body growth stimulation |
| myostatin | MSTN | muscle growth (negative regulator) |
| phosphoenolpyruvate carboxykinase 1 | PCK1 | regulation of gluconeogenesis |
| pyruvate dehydrogenase kinase 4 | PDK4 | regulation of glucose and fatty acid metabolism |
| peroxisome proliferator activated receptor delta | PPARD | oxidative (fat-burning) capacity |
| vascular endothelial growth factor | VEGF | vasculogenesis, angiogenesis |
| zinc finger and AT-hook domain containing | ZFAT | effect on body weight in thoroughbreds |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tozaki, T.; Ohnuma, A.; Kikuchi, M.; Ishige, T.; Kakoi, H.; Hirota, K.-i.; Kusano, K.; Nagata, S.-i. Microfluidic Quantitative PCR Detection of 12 Transgenes from Horse Plasma for Gene Doping Control. Genes 2020, 11, 457. https://doi.org/10.3390/genes11040457
Tozaki T, Ohnuma A, Kikuchi M, Ishige T, Kakoi H, Hirota K-i, Kusano K, Nagata S-i. Microfluidic Quantitative PCR Detection of 12 Transgenes from Horse Plasma for Gene Doping Control. Genes. 2020; 11(4):457. https://doi.org/10.3390/genes11040457
Chicago/Turabian StyleTozaki, Teruaki, Aoi Ohnuma, Mio Kikuchi, Taichiro Ishige, Hironaga Kakoi, Kei-ichi Hirota, Kanichi Kusano, and Shun-ichi Nagata. 2020. "Microfluidic Quantitative PCR Detection of 12 Transgenes from Horse Plasma for Gene Doping Control" Genes 11, no. 4: 457. https://doi.org/10.3390/genes11040457
APA StyleTozaki, T., Ohnuma, A., Kikuchi, M., Ishige, T., Kakoi, H., Hirota, K.-i., Kusano, K., & Nagata, S.-i. (2020). Microfluidic Quantitative PCR Detection of 12 Transgenes from Horse Plasma for Gene Doping Control. Genes, 11(4), 457. https://doi.org/10.3390/genes11040457






