A Simple Method to Detect the Inhibition of Transcription Factor-DNA Binding Due to Protein–Protein Interactions In Vivo
Abstract
1. Introduction
2. Materials and Methods
2.1. Modification of pGBKT7 Vector
2.2. Vector Construction
2.3. Yeast Experiments
3. Results
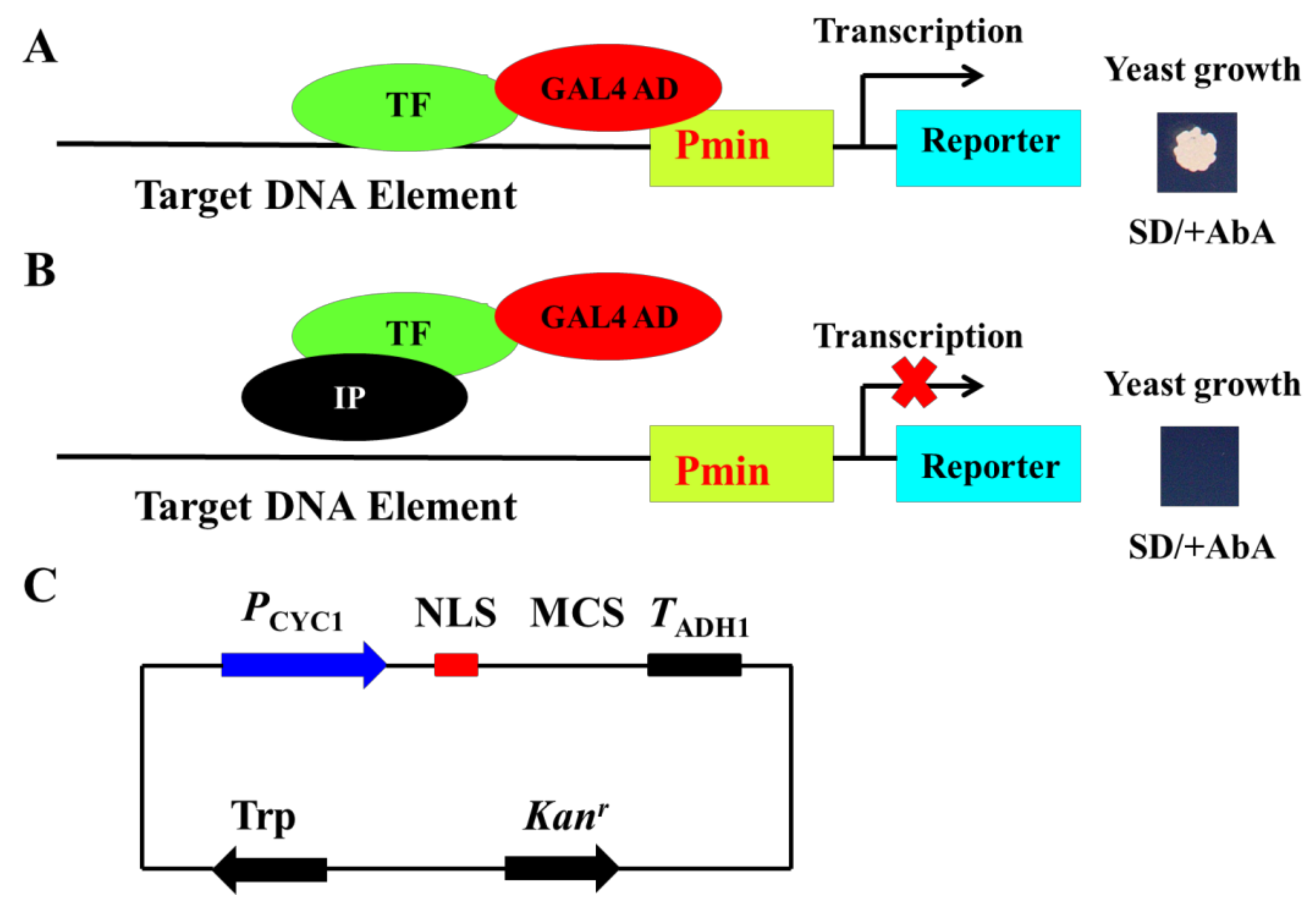
3.1. The Principle of the Newly Developed Method
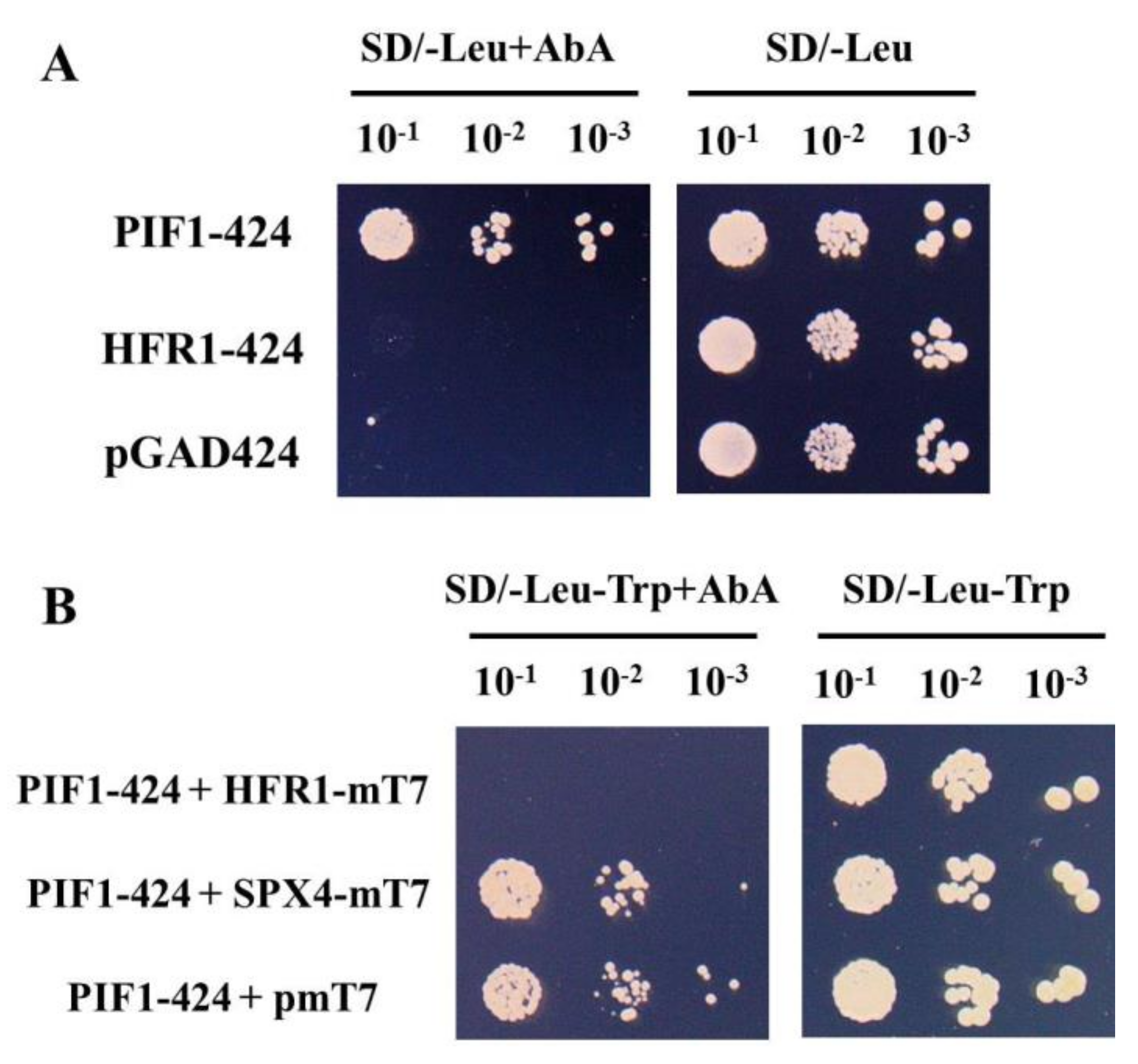
3.2. The New Method was used to Verify the Result that HFR1-PIF1 Interaction Inhibited PIF1 Binding to G-box Element
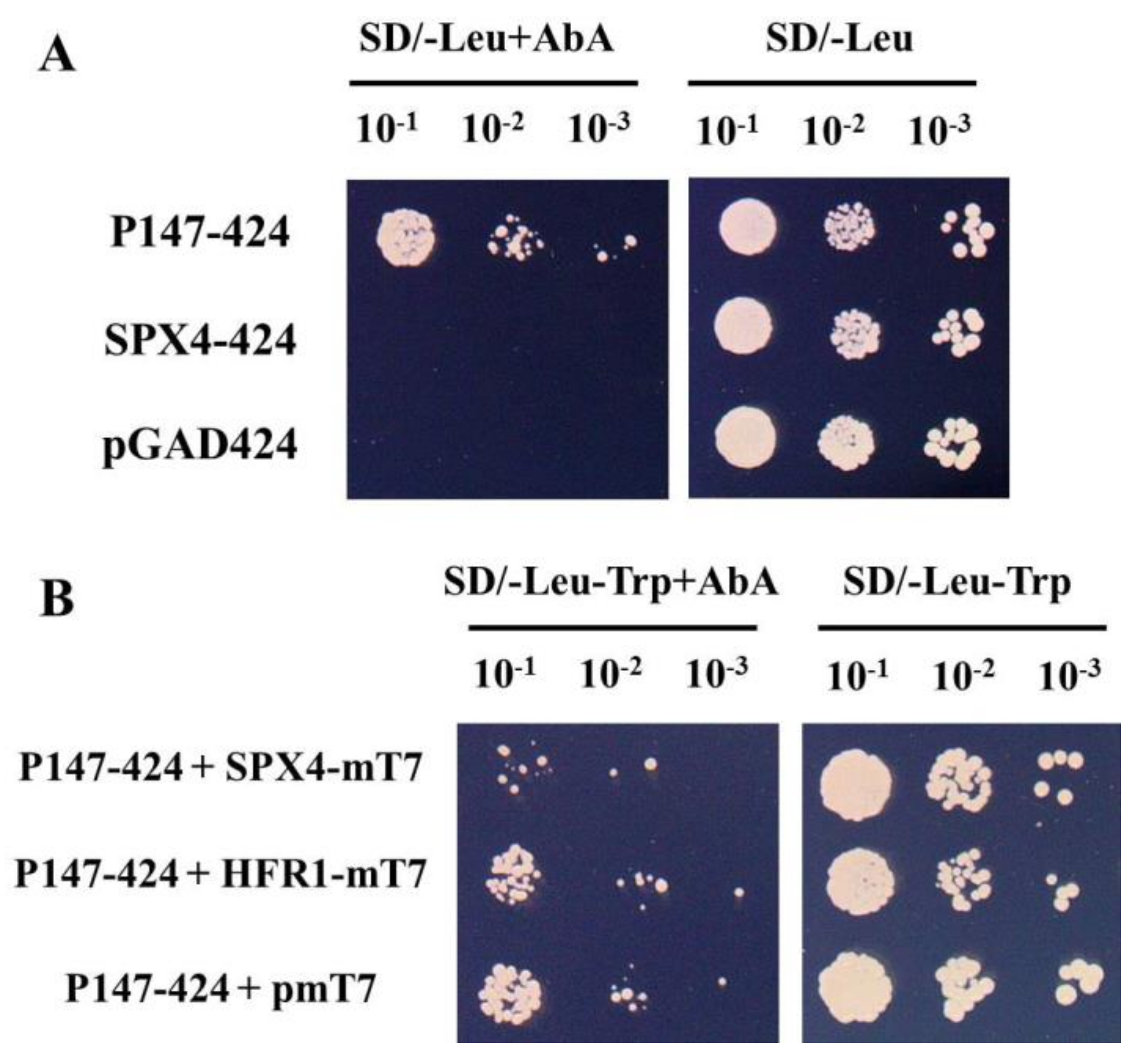
3.3. The New Method was used to Verify the Result that SPX4–PHR2 Interaction Inhibited PHR2 Binding to P1BS Element
4. Discussion
Supplementary Materials
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Husbands, A.; Bell, E.M.; Shuai, B.; Smith, H.M.; Springer, P.S. LATERAL ORGAN BOUNDARIES defines a new family of DNA-binding transcription factors and can interact with specific bHLH proteins. Nucleic Acids Res. 2007, 35, 6663–6671. [Google Scholar] [CrossRef]
- Shi, H.; Zhong, S.; Mo, X.; Liu, N.; Nezames, C.D.; Deng, X.W. HFR1 sequesters PIF1 to govern the transcriptional network underlying light-initiated seed germination in Arabidopsis. Plant Cell 2013, 25, 3770–3784. [Google Scholar] [CrossRef]
- Lv, Q.; Zhong, Y.J.; Wang, Y.G.; Wang, Z.Y.; Zhang, L.; Shi, J.; Wu, Z.; Liu, Y.; Mao, C.Z.; Yi, K.K.; et al. SPX4 negatively regulates phosphate signaling and homeostasis through its interaction with PHR2 in rice. Plant Cell 2014, 26, 1586–1597. [Google Scholar] [CrossRef]
- Job, N.; Yadukrishnan, P.; Bursch, K.; Datta, S.; Johansson, H. Two B-box proteins regulate photomorphogenesis by oppositely modulating HY5 through their diverse C-terminal domains. Plant Physiol. 2018, 176, 2963–2976. [Google Scholar] [CrossRef]
- Song, C.; Lee, J.; Kim, T.; Hong, J.C.; Lim, C.O. VOZ1, a transcriptional repressor of DREB2C, mediates heat stress responses in Arabidopsis. Planta 2018, 247, 1439–1448. [Google Scholar] [CrossRef]
- Oh, E.; Yamaguchi, S.; Hu, J.H.; Yusuke, J.; Jung, B.; Paik, I.; Lee, H.S.; Sun, T.P.; Kamiya, Y.; Choi, G. PIL5, a phytochrome-interacting bHLH protein, regulates gibberellin responsiveness by binding directly to the GAI and RGA promoters in Arabidopsis seeds. Plant Cell 2007, 19, 1192–1208. [Google Scholar] [CrossRef]
- Mumberg, D.; Muller, R.; Funk, M. Yeast vectors for the controlled expression of heterologous proteins in different genetic backgrounds. Gene 1995, 156, 119–122. [Google Scholar] [CrossRef]
- Peng, B.; Williams, T.C.; Henry, M.; Nielsen, L.K.; Vickers, C.E. Controlling heterologous gene expression in yeast cell factories on different carbon substrates and across the diauxic shift: a comparison of yeast promoter activities. Microb. Cell Fact. 2015, 14, 91. [Google Scholar] [CrossRef]
- Rubio, V.; Linhares, F.; Solano, R.; Martin, A.C.; Iglesias, J.; Leyva, A.; Paz-Ares, J. A conserved MYB transcription factor involved in phosphate starvation signaling both in vascular plants and in unicellular algae. Genes Dev. 2001, 15, 2122–2133. [Google Scholar] [CrossRef]
- Bustos, R.; Castrillo, G.; Linhares, F.; Puga, M.I.; Rubio, V.; Perez-Perez, J.; Solano, R.; Leyva, A.; Paz-Ares, J. A central regulatory system largely controls transcriptional activation and repression responses to phosphate starvation in Arabidopsis. PLoS Genet. 2010, 6, e1001102. [Google Scholar] [CrossRef]
- Reece-Hoyes, J.S; Marian Walhout, A.J. Yeast one-hybrid assays: A historical and technical perspective. Methods 2012, 57, 441–447. [Google Scholar] [CrossRef]
- Ji, X.; Wang, L.; Zang, D.; Wang, Y. Transcription Factor-Centered Yeast One-Hybrid Assay. In Two-Hybrid Systems: Methods and Protocols, Methods in Molecular Biology; Ji, X.; Wang, L.; Zang, D.; Wang, Y. Humana Press: New York, NY, USA, 2018; Volume 1794, p. 18194. [Google Scholar]
- Bonaldi, K.; Li, Z.; Kang, S.E.; Breton, G.; Pruneda-Paz, J.L. Novel cell surface luciferase reporter for high-throughput yeast one-hybrid screens. Nucleic Acids Res. 2017, 45, e157. [Google Scholar] [CrossRef]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yang, G.; Chao, D.; Ming, Z.; Xia, J. A Simple Method to Detect the Inhibition of Transcription Factor-DNA Binding Due to Protein–Protein Interactions In Vivo. Genes 2019, 10, 684. https://doi.org/10.3390/genes10090684
Yang G, Chao D, Ming Z, Xia J. A Simple Method to Detect the Inhibition of Transcription Factor-DNA Binding Due to Protein–Protein Interactions In Vivo. Genes. 2019; 10(9):684. https://doi.org/10.3390/genes10090684
Chicago/Turabian StyleYang, Guangzhe, Dong Chao, Zhenhua Ming, and Jixing Xia. 2019. "A Simple Method to Detect the Inhibition of Transcription Factor-DNA Binding Due to Protein–Protein Interactions In Vivo" Genes 10, no. 9: 684. https://doi.org/10.3390/genes10090684
APA StyleYang, G., Chao, D., Ming, Z., & Xia, J. (2019). A Simple Method to Detect the Inhibition of Transcription Factor-DNA Binding Due to Protein–Protein Interactions In Vivo. Genes, 10(9), 684. https://doi.org/10.3390/genes10090684




