The Caribou (Rangifer tarandus) Genome
Abstract
1. Introduction
2. Materials and Methods
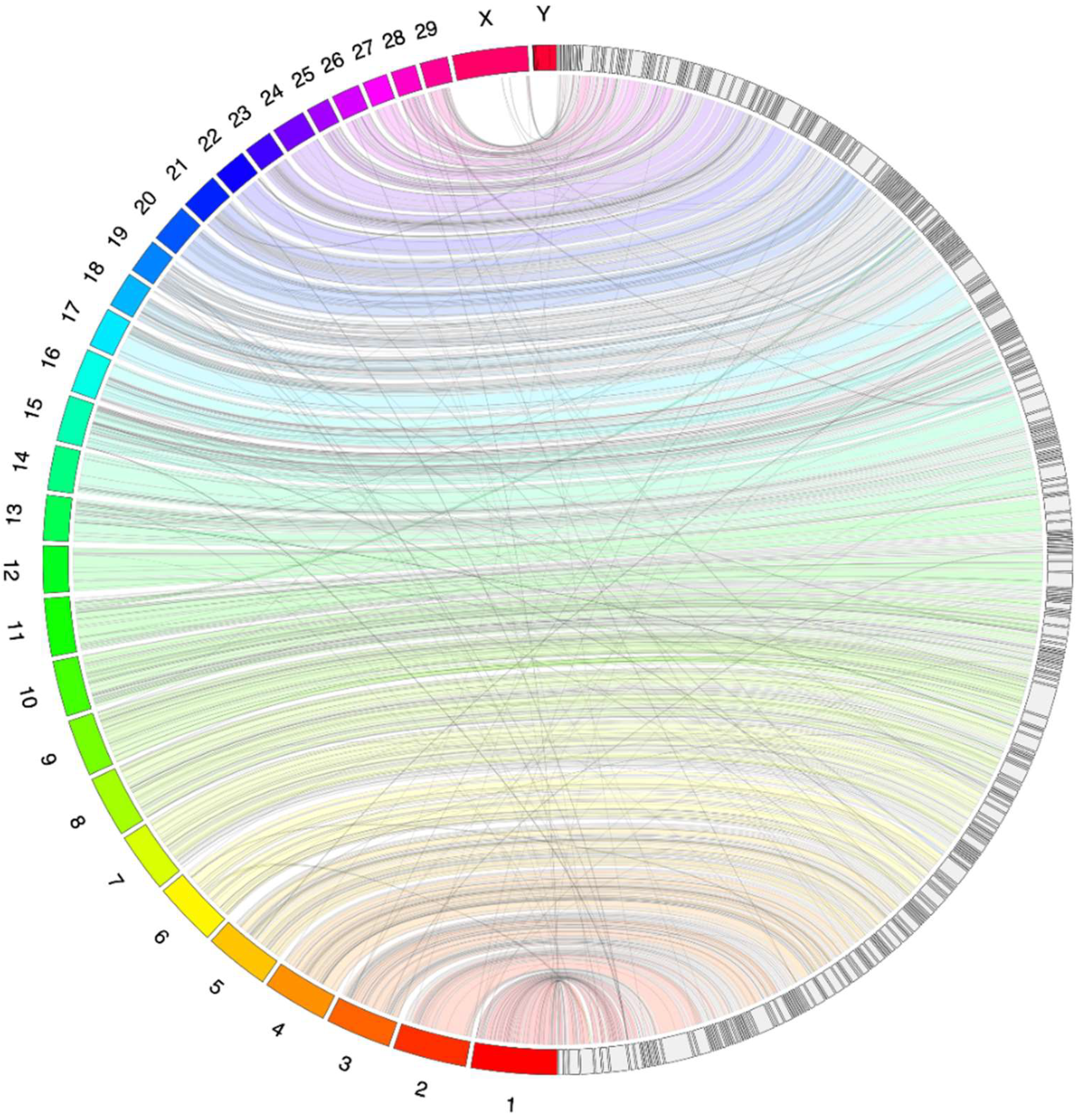
3. Results and Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Banfield, A.W.F. A Revision of the Reindeer and Caribou, Genus Rangifer; National Museum of Canada, Bulletin No. 177, Queen’s Printer: Ottawa, ON, Canada, 1967. [Google Scholar]
- Vors, L.S.; Boyce, M.S. Global declines of caribou and reindeer. Glob. Change Biol. 2009, 15, 2626–2633. [Google Scholar] [CrossRef]
- Festa-Bianchet, M.; Ray, J.C.; Boutin, S.; Côté, S.D.; Gunn, A. Conservation of caribou (Rangifer tarandus) in Canada: An uncertain future. Can. J. Zool. 2011, 89, 419–434. [Google Scholar] [CrossRef]
- COSEWIC. Designatable Units for Caribou (Rangifer tarandus) in Canada; Committee on the Status of Endangered Wildlife in Canada: Ottawa, ON, Canada, 2011. [Google Scholar]
- Yannic, G.; Pellissier, L.; Ortego, J.; Lecomte, N.; Couturier, S.; Cuyler, C.; Dussault, C.; Hundertmark, K.J.; Irvine, R.J.; Jenkins, D.A.; et al. Genetic diversity in caribou linked to past and future climate change. Nat. Clim. Chang. 2014, 4, 132–137. [Google Scholar] [CrossRef]
- Klütsch, C.F.C.; Manseau, M.; Anderson, M.; Sinkins, P.; Wilson, P.J. Evolutionary reconstruction supports the presence of a Pleistocene Arctic refugium for a large mammal species. J. Biogeogr. 2017, 44, 2729–2739. [Google Scholar] [CrossRef]
- Maniatis, T.; Fritch, E.F.; Sambrook, J. Molecular Cloning: A Laboratory Manual; Cold Spring Harbor Laboratory: New York, NY, USA, 1982. [Google Scholar]
- Putnam, N.H.; O’Connell, B.L.; Stites, J.C.; Rice, B.J.; Blanchette, M.; Calef, R.; Troll, C.J.; Fields, A.; Hartley, P.D.; Sugnet, C.W.; et al. Chromosome-scale shotgun assembly using an in vitro method for long-range linkage. Genome Res. 2016, 26, 342–350. [Google Scholar] [CrossRef] [PubMed]
- Chapman, J.A.; Ho, I.; Sunkara, S.; Luo, S.; Schroth, G.P.; Rokhsar, D.S. Meraculous: De novo genome assembly with short paired-end reads. PLoS ONE 2011, 6, e23501. [Google Scholar] [CrossRef] [PubMed]
- Bolger, A.M.; Lohse, M.; Usadel, B. Trimmomatic: A flexible trimmer for Illumina sequence data. Bioinformatics 2014, 30, 2114–2120. [Google Scholar] [CrossRef]
- Stanke, M.; Diekhans, M.; Baertsch, R.; Haussler, D. Using native and syntenically mapped cDNA alignments to improve de novo gene finding. Bioinformatics 2008, 24, 637–644. [Google Scholar] [CrossRef]
- Smit, A.F.A.; Hubley, R.; Green, P. Repeat Masker. Available online: http://repeatmasker.org (accessed on 30 June 2018).
- Bushnell, B.; Rood, J.; Singer, E. BBMerge—Accurate paired shotgun read merging via overlap. PLoS ONE 2017, 12, e0185056. [Google Scholar] [CrossRef]
- Waterhouse, R.M.; Seppey, M.; Simao, F.A.; Manni, M.; Ioannidis, P.; Klioutchnikov, G.; Kriventseva, E.V.; Zdobnov, E.M. BUSCO applications from quality assessments to gene prediction and phylogenomics. Mol. Biol. Evol. 2018, 35, 543–554. [Google Scholar] [CrossRef]
- Lorenzini, R.; Garofalo, L. Insights into the evolutionary history of Cervus (Cervidae, tribe Cervini) based on Bayesian analysis of mitochondrial marker sequences, with first indications for a new species. J. Zoolog. Syst. Evol. Res. 2015, 53, 340–349. [Google Scholar] [CrossRef]
- Elsik, C.G.; Unni, D.R.; Diesh, C.M.; Tayal, A.; Emery, M.L.; Nguyen, H.N.; Hagen, D.E. Bovine Genome Database: New tools for gleaning function from the Bos taurus genome. Nucleic Acids Res. 2016, 44, D834–D839. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Durbin, R. Fast and accurate short read alignment with Burrows-Wheeler transform. Bioinformatics 2009, 25, 1754–1760. [Google Scholar] [CrossRef] [PubMed]
- Chu, J. Jupiter Plot: A Circos-Based Tool to Visualize Genome Assembly Consistency (Version 1.0). Zenodo. Available online: https://zenodo.org/record/1241235#.XA92q2hKiUk (accessed on 15 March 2019).
- Krzywinski, M.; Schein, J.; Birol, I.; Connors, J.; Gascoyne, R.; Horsman, D.; Jones, S.J.; Marra, M.A. Circos: An information aesthetic for comparative genomics. Genome Res. 2009, 19, 1639–1645. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Lin, Z.; Ba, H.; Chen, L.; Yang, Y.; Wang, K.; Qiu, Q.; Wang, W.; Li, G. Draft genome of the reindeer (Rangifer tarandus). GigaScience 2017, 6, 1–5. [Google Scholar] [CrossRef]
- Mikheenko, A.; Prjibelski, A.; Saveliev, V.; Antipov, D.; Gurevich, A. Versatile genome assembly evaluation with QUAST-LG. Bioinformatics 2018, 34, i142–i150. [Google Scholar] [CrossRef] [PubMed]
- Taylor, G.A.; Kirk, K.; Coombe, L.; Jackman, S.D.; Chu, J.; Tse, K.; Cheng, D.; Chuah, E.; Pandoh, P.; Carlson, R.; et al. The genome of the North American brown bear or grizzly: Ursos arctos ssp. horribilis. Genes 2018, 9, 598. [Google Scholar] [CrossRef] [PubMed]
- Jones, S.J.M.; Taylor, G.A.; Chan, S.; Warren, R.L.; Austin Hammond, S.; Bilobram, S.; Mordecai, G.; Suttle, C.A.; Miller, K.M.; Schulze, A.; et al. The genome of the Beluga whale (Delphinapterus leucas). Genes 2017, 8, 378. [Google Scholar] [CrossRef] [PubMed]
- Jones, S.J.; Haulena, M.; Taylor, G.A.; Chan, S.; Bilbobram, S.; Warren, R.L.; Austin Hammond, S.; Mungall, K.L.; Choo, C.; Kirk, H.; et al. The genome of the Northern Sea Otter (Enhydra lutris kenyoni). Genes 2017, 8, 379. [Google Scholar] [CrossRef] [PubMed]
- Kharzinova, V.R.; Dotsev, A.V.; Deniskova, T.E.; Solovieva, A.D.; Fedorov, V.I.; Layshev, K.A.; Romanenko, T.M.; Okhlopkov, I.M.; Wimmers, K.; Reyer, H.; et al. Genetic diversity and population structure of domestic and wild reindeer (Rangifer tarandus L. 1758): A novel approach using BovineHD BeadChip. PLoS ONE 2018, 13, e0207944. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Ajimura, M.; Chen, Z.; Liu, J.; Chen, E.; Guo, H.; Tadapatri, V.; Reddy, C.G.; Zhang, J.; Kishino, H.; et al. A new approach for comprehensively describing heterogametic sex chromosomes. DNA Res. 2018, 25, 375–382. [Google Scholar] [CrossRef] [PubMed]
- Primmer, C.R. From conservation genetics to conservation genomics. Ann. N. Y. Acad. Sci. 2009, 1162, 357–368. [Google Scholar] [CrossRef] [PubMed]
| Statistic | Rangifer tarandus genome |
|---|---|
| Scaffold sequence total (bp) | 22.052 × 108 |
| Number scaffolds | 4699 |
| Scaffold N50 (bp) | 11.765 × 106 |
| Scaffold L50 | 52 |
| Scaffold N90 (bp) | 89.704 × 104 |
| Scaffold L90 | 289 |
| Contig sequence total (bp) | 21.893 × 108 |
| Number contigs | 146,562 |
| Contig N50 (bp) | 32.819 × 103 |
| Contig L50 | 19,701 |
| Contig N90 (bp) | 89.140 × 102 |
| Contig L90 | 68,199 |
| A | C | G | T | N |
|---|---|---|---|---|
| 29.27% | 20.72% | 20.73% | 29.28% | 0.72% |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Taylor, R.S.; Horn, R.L.; Zhang, X.; Golding, G.B.; Manseau, M.; Wilson, P.J. The Caribou (Rangifer tarandus) Genome. Genes 2019, 10, 540. https://doi.org/10.3390/genes10070540
Taylor RS, Horn RL, Zhang X, Golding GB, Manseau M, Wilson PJ. The Caribou (Rangifer tarandus) Genome. Genes. 2019; 10(7):540. https://doi.org/10.3390/genes10070540
Chicago/Turabian StyleTaylor, Rebecca S., Rebekah L. Horn, Xi Zhang, G. Brian Golding, Micheline Manseau, and Paul J. Wilson. 2019. "The Caribou (Rangifer tarandus) Genome" Genes 10, no. 7: 540. https://doi.org/10.3390/genes10070540
APA StyleTaylor, R. S., Horn, R. L., Zhang, X., Golding, G. B., Manseau, M., & Wilson, P. J. (2019). The Caribou (Rangifer tarandus) Genome. Genes, 10(7), 540. https://doi.org/10.3390/genes10070540





