Abstract
Plastics have become an important environmental concern due to their durability and resistance to degradation. Out of all plastic materials, polyesters such as polyethylene terephthalate (PET) are amenable to biological degradation due to the action of microbial polyester hydrolases. The hydrolysis products obtained from PET can thereby be used for the synthesis of novel PET as well as become a potential carbon source for microorganisms. In addition, microorganisms and biomass can be used for the synthesis of the constituent monomers of PET from renewable sources. The combination of both biodegradation and biosynthesis would enable a completely circular bio-PET economy beyond the conventional recycling processes. Circular strategies like this could contribute to significantly decreasing the environmental impact of our dependence on this polymer. Here we review the efforts made towards turning PET into a viable feedstock for microbial transformations. We highlight current bottlenecks in degradation of the polymer and metabolism of the monomers, and we showcase fully biological or semisynthetic processes leading to the synthesis of PET from sustainable substrates.
1. Introduction
Thermoplastic polymers, some of which constitute the majority of the commonly known plastics, are extremely useful materials endowed with properties that make them ideal for applications such as insulation and packaging [1,2]. They are durable, water-proof and versatile materials that have become almost essential in our lives. In fact, in 2017 the contribution of plastics to the European economy reached a market size of EUR 355 billion while employing 1.5 million people [3]. Plastics are light-weight and have significantly contributed to decreasing transportation costs and extending the shelf life of food [4]. Their success as a material is only comparable to their detrimental environmental impact. The accumulation of plastic waste in the environment has become an extremely serious concern [5,6]. Plastic pollution is present in every single niche of the planet, with dramatic effects on ecosystems, especially in marine environments, affecting equally large and small fauna and flora [6,7].
Plastics possess two key features: they are barely degradable by environmental physical, chemical and especially by biological processes [8], and they have low production costs, which make their reuse not economically competitive. While these individual properties are desirable, when combined they lead to the current problem we are facing: the accumulation of recalcitrant and polymers in the environment that can degrade into microplastics with potential toxic effects [9]. The story of plastic pollution is a story of mismanagement of an otherwise valuable resource. Numerous recent studies have highlighted the poor recycling rates of plastics compared to other materials. For instance, a recent report estimates the amount of virgin plastics produced from oil of over 8 billion metric tons, out of which only 9% have been recycled [10]. This reflects a saturated traditional recycling industry and emphasises the need for novel approaches to plastic management, including the possibility of harnessing microbial activities to use plastic waste as a feedstock for biotransformations [11,12,13,14].
Out of all plastics, polyesters such as polyethylene terephthalate (PET) are in a good position for becoming a sustainable polymer compared to other oil-derived counterparts. PET is obtained from the polymerisation of the constituent monomers terephthalic acid (TPA) and ethylene glycol (EG) (Figure 1). It is durable, relatively easy to mould by blowing, which results in an almost inert, hard and stiff polymer that has been adopted by the beverage industry as the main material for the production of bottles [15,16]. PET has, in addition, the highest collection rates of all plastics even though reused PET is only a small fraction of the total PET consumed: The US National Association for PET Container Resources (NAPCOR) reported that out of the approximately 3 million tons of new PET bottles reaching the market in 2017, only 29% of them were made from collected and recycled PET, a nearly 5% decrease compared to recycling rates of the previous year [17].
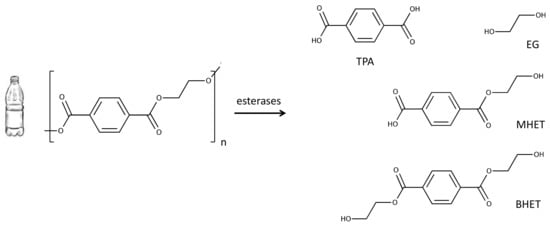
Figure 1.
Enzymatic hydrolysis of polyethylene terephthalate (PET) results in a mixture of terephthalic acid (TPA) and ethylene glycol (EG) and, to a lesser extent, the incomplete hydrolysis products bis-(2-hydroxyethyl) terephthalate (BHET) and mono-(2-hydroxyethyl) terephthalate (MHET).
As a polyester, PET can be depolymerized as a more effective alternative to mechanical recycling [18]. Methods of depolymerization include glycolysis, methanolysis, hydrolysis, aminolysis and ammonolysis [19]. Among them, glycolysis has recently emerged as a key technology for recycling PET waste. Glycolysis is the process of PET degradation by glycols at high temperatures and in the presence of catalysts such as metal acetates [20]. Compared to other methods, glycolysis has the great advantage of enabling the recycling of coloured and opaque PET that cannot be otherwise recycled due to the presence of the pigments. The resulting monomers TPA and EG can be re-used to produce PET, as well as other polymers of interest [21]. Glycolysis and related methods contribute to a more sustainable PET economy, although they also have drawbacks such as the energy cost of the high temperatures required and the long reaction times needed for effective depolymerization [21].
Biological activities capable of catalysing PET hydrolysis under mild reaction conditions are emerging as an alternative to chemical PET depolymerization methods [22]. As a result, a number of enzymes from different microorganisms have been characterised [23,24,25], facilitating the implementation of PET as a biotechnological feedstock [11,26]. We argue that this strategy is more versatile than chemical methods because, if funnelled to the central microbial metabolism, the monomers obtained can be transformed into a plethora of molecules by harnessing advances in synthetic biology and metabolic engineering. This would contribute to creating a path for revenue from PET waste beyond current recycling activities. It could thereby help to mitigate the impact of PET environmental release and promote the competitive development of a next generation of environmentally friendly materials.
Given the interesting physicochemical properties of PET and its potential use as a substrate in biotechnology, in this article we review the genes that are required for a sustainable and circular PET economy. In our view, to accomplish this goal it is required to (i) improve the kinetics of PET enzymatic hydrolysis; (ii) link the metabolism of the resulting monomers to relevant biosynthetic pathways and (iii) engineer biological systems for the production of PET monomers TPA and EG from renewable sources.
2. PET Metabolism
The enzymatic hydrolysis of PET involves the release of constituent monomers TPA and EG due to the action of esterases. The resulting monomers can be degraded by microorganisms endowed with the appropriate metabolic pathways for these compounds. TPA is converted into protocatechuate (PCA) that will undergo dioxygenolytic cleavage and degradation through different routes prior to reaching the central metabolism [27,28,29,30]. Similarly, EG is assimilated through different pathways depending on the microorganism. For instance, it can be transformed into acetate via acetyl-CoA in Acetobacterium woodii [31], whereas in some strains in Pseudomonas putida it is funnelled directly to the Krebs cycle via isocitrate [32]. In this section we will focus on the genes responsible for these activities and their (co)occurrence in different bacterial taxa.
2.1. Enzymatic Hydrolysis of PET
Different types of hydrolases have shown to be active against the PET polymer. These enzymes are lipases, esterases, cutinases and carboxylesterases isolated from fungi and bacteria (see [14,25] for recent reviews on this topic). They belong to the α/β hydrolase superfamily and have evolved in a different context and for a different function [33]. For instance, the original role of the cutinases from the genus Thermobifida is to hydrolyse the plant polyester cutin. Among the different variants of these enzymes, the ones endowed with certain properties (e.g., a more accessible active site) display the highest activity against PET [34]. A recent bioinformatic analysis has investigated the distribution of genes encoding for homologs of these esterases in terrestrial and marine metagenomes and has allowed to identify 504 new hydrolases [35]. The two main conclusions of this study are: (i) genes potentially encoding polyester hydrolases are rare, and (ii) their taxonomic distribution seems to be related to the niche studied, with Actinobacteria or Proteobacteria being more prominent hosts in terrestrial environments, whereas Bacteroidetes are the most frequent hosts in marine metagenomes [35].
As a new-to-nature polymer, PET constitutes a challenge for any of the hydrolases that are active against it. In this sense, it is worth highlighting that not all types of PET are equally susceptible to microbial degradation. Depending on processing and thermal treatments, PET can occur in an amorphous form or a semi-crystalline form [36]. It has been shown that the extent of enzymatic polyester hydrolysis depends on the degree of its crystallinity and chain orientation [37]. In the amorphous regions, the polymer chains are less densely packed and are more susceptible to hydrolytic attack compared to the crystalline regions. The enzymatic degradation rate of the polyester correlates with the temperature difference between the melting temperature of the polymer and the hydrolysis temperature. The polymer chain can be considered to be more mobile and accessible to enzymatic attack when close to the glass transition temperature (Tg) of amorphous PET [38]. Therefore, increased enzymatic hydrolysis rates of PET are expected when performing the reaction at temperatures near the Tg of the amorphous polyester (67–71 °C). This suggests that efficient PET hydrolysis needs to be conducted by thermostable polyester hydrolases such as the cutinases TfCut2 and HiC isolated, respectively, from the thermophilic actinomycete Thermobifida fusca [23] or the fungus Thermomyces insolens [37], both of which, especially the latter, have been reported to be active for long periods of time at temperatures of up to 70 °C. Engineered post-translational modifications (e.g., glycosylation) can then be used on these polyester hydrolases to improve thermal properties of the enzymes further [39]. Hydrolysis at those temperatures is obviously not compatible with most bioprocesses using whole-cell catalysts, especially those involving engineered mesophilic organisms such as Escherichia coli which can grow up to a maximum temperature of 48–50 °C only after evolutionary adaptation and at a fitness cost [40,41]. The bacterium Ideonella sakaiensis has been reported to be capable of growing on PET as a sole carbon source due to the secretion of a PET hydrolase [24]. When tested in vitro and in mesophilic conditions (below the Tg of PET), this enzyme shows very low degradation rates of PET and, even though this activity could be increased somewhat by directed evolution [42], potential hydrolysis yields are far from being able to sustain industrial bioprocesses.
Another important factor affecting the performance of the enzymes hydrolysing PET is their inhibition mediated by mono-(2-hydroxyethyl) terephthalate (MHET) and bis-(2-hydroxyethyl) terephthalate (BHET), by-products of an incomplete hydrolysis [43]. These molecules are oligomers of TPA and EG that act as competitive inhibitors of the enzymes [44]. Even though it is possible to design reactors that allow for a continuous removal of MHET and BHET [45], this is likely to pose a challenge for the biodegradation of PET using whole cells. Other solutions have been tested such as the use of mixtures of hydrolases that act synergistically [46], or the selective modification of amino acid residues of the polyester hydrolase involved in the interaction with the inhibitors [47]. These factors emphasise the need for obtaining enzymes, either by direct screening or by modification of existing ones, which are not susceptible to inhibition by MHET and BHET and can therefore be used to develop efficient bioprocesses using PET as the substrate.
2.2. Metabolism of TPA
TPA is transformed into PCA by the pathway encoded by the tph genes. These genes encode two sequential catabolic steps: the addition of two hydroxyl groups in positions 4 and 5 of TPA by the activity of the TPA dioxygenase TphA1A2A3 producing 1,6-dihydroxycyclohexa-2,4-diene-dicarboxylate (DCD), and the removal of the carboxyl group in position 6 by the action of the 1,2-dihydroxy-3,5-cyclohexadiene-1,4-dicarboxylate dehydrogenase TphB (Figure 2A). The genes responsible for those activities have been characterised in the actinomycete Rhodococcus sp. strain DK17 [48], in the β-proteobacteria Comamonas testosteroni YZW-D [49], and in Comamonas sp. strain E6 [50]. In addition to the catabolic tph genes, both organisms encode within this cluster the transcriptional regulator TphR (Figure 2B). TphR has been described as an IclR-type activator that responds to the inducer TPA [51]. Comamonas sp. strain E6 also contains the extra gene tphC, which encodes a permease involved in the uptake of TPA using the tripartite aromatic acid transporter [52].
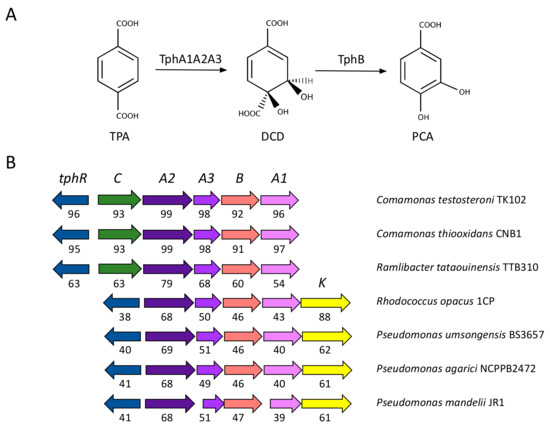
Figure 2.
(a) TPA metabolism reported in bacteria. The names of the molecules and abbreviations are: terephthalic acid, TPA; 1,6-dihydroxycyclohexa-2,4-diene-dicarboxylate, DCD; protocatechuate, PCA. (b) Genetic organisation of the tph genes identified in several genomes available in databases. Numbers below arrows indicate the percentage of identity compared to the orthologous genes present in Comamonas sp. E6 (accession: AB238679; [50]) with the exception of the tphK genes that were compared to the ortholog present in the plasmid pDK3 of Rhodococcus sp. DK17 (accession: AY502076; [48]). Plots were produced with SyntTax (http://archaea.u-psud.fr/SyntTax; [55]).
We conducted a systematic analysis of the presence of the tph genes in the genomes available in public databases. As a result, we identified genes sharing a significant identity (greater than 35% for all the genes in the cluster with tphA2 greater than 65%) and similar genetic organisation in only a limited number of organisms, which are representative of β-proteobacteria (Comamonas, Ideonella and Ramlibacter) and γ-proteobacteria (Pseudomonas), as well as of actinomycetes (Rhodococcus). In the genus Rhodococcus the tph genes are associated with plasmids with the exception of Rhodococcus opacus 1CP in which the cluster of genes was identified in the chromosome. In all the genomes investigated, the four catabolic genes were conserved in the same order. All clusters contain a regulatory gene encoding an IclR-type transcriptional regulator upstream the catabolic genes and in a divergent orientation. More diversity was observed in the putative transport of TPA inside the cell: all the β-proteobacteria utilized the transporter tphC, whereas the rest of the organisms contained a previously unidentified MFS transporter of the AAHS family (aromatic acid:H+ symporter; named tphK) homologous to the p-hydroxybenzoate transporter pcaK [53] (Figure 2B).
2.3. Metabolism of PCA
The PCA resulting from the activity of the Tph enzymes follows different pathways depending on the organism. This suggests that the tph genes can act as an independent metabolic module regardless of the type of PCA metabolism present in the TPA degrading strain. In fact, two copies of this cluster of genes are harboured by two different plasmids in Rhodococcus sp. strain DK17, indicating that this pathway can be mobilised by horizontal gene transfer into species containing one of the widespread PCA degradation pathways [48]. All PCA pathways share an initial dioxygenolytic step in which the aromatic ring is cleaved. Until now, three different pathways have been reported depending on the cleavage position in the aromatic ring. They are known as the ortho-, meta- and para-cleavage pathways and their initial reaction is catalysed by a PCA-3,4-, 4,5- and 2,3-dioxygenase, respectively (Figure 3) [27,29,54]. For simplicity, we will refer from now on to the nomenclature of the enzymes to discriminate between the pathways.
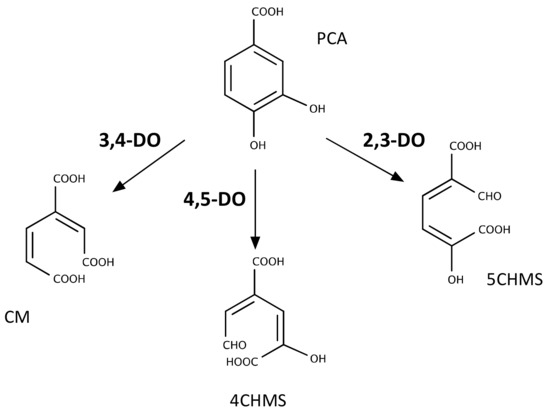
Figure 3.
Types of dioxygenase-mediated reactions involved in PCA cleavage by bacteria. DO: dioxygenase; CM: 3-carboxy-cis,cis-muconate; 4CHMS: 4-carboxy-2-hydroxymuconate semialdehyde; 5CHMS: 5-carboxy-2-hydroxymuconate-6-semialdehyde.
Using the sequences of characterised PCA dioxygenases, we conducted a bioinformatics search of the pathways likely involved in the metabolism of PCA that are present in the genomes in which we had previously identified the genes responsible for the conversion of TPA into PCA. Out of the three pathways, the PCA-2,3-dioxygenase was not present in any of them. Among the β-proteobacteria, C. testosteroni, C. thiooxydans and R. tataouinensis have homologs of the PCA-4,5-dioxygenase in their genomes, whereas I. sakaiensis, the different species of Pseudomonas and R. opacus contain the PCA-3,4-dioxygenase pathway. These results are consistent with previous observations showing that a PCA-3,4-dioxygenase activity is present in cells of Rhodococcus sp. strain DK17 growing on TPA [48], whereas a PCA-4,5-dioxygenase activity was identified in Comamonas sp. strain E6 [50]. Likewise, I. sakaiensis has been reported to contain a tph cluster and PCA-3,4-pathway [24].
The diversity of PCA metabolic pathways is an important factor when considering developing bioprocesses based on PET. Depending on the pathway used, a range of metabolites can be produced with different applications in mind. Out of them, the PCA-3,4-dioxygenolytic pathway has been thoroughly studied. This route is one of the branches of the β-ketoadipate pathway that connects the metabolism of aromatics converging on either catechol (e.g., benzoate) or PCA (e.g., 4-hydrozybenzoate) with the central metabolism of certain bacterial species [30]. The β-ketoadipate pathway has traditionally been used as a way of incorporating toxic and recalcitrant aromatic molecules in the central metabolism of bacteria, including nitrophenols and polychlorinated arenes. It is also an important path for funnelling the degradation products of lignocellulosic waste that could be used for the synthesis of other molecules of interest [56]. Strikingly, despite the metabolic diversity of the pathways involved which could allow for the production of molecules with interesting properties (e.g., functionalised lactones), complete mineralization of PCA continues to be the main application of the PCA metabolism. Only recently, PCA obtained from lignin-derived aromatics has been used for synthesis of the industrially relevant metabolite adipic acid [57]. This has not been achieved by the action of any of the described PCA pathways, but by the conversion of PCA into catechol catalysed by a PCA decarboxylase. Catechol is then transformed into cis,cis-muconate by the action of a catechol-1,2-dioxygenase, and the latter is hydrogenated abiotically to adipic acid in the presence of a catalyst [58].
2.4. Metabolism of EG
The metabolism of EG is more diverse compared to TPA. In acetogens, EG is oxidised to ethanol and acetaldehyde that is eventually converted to acetate via acetyl-CoA [31]. In other bacterial species, however, EG is degraded via the formation of glyoxylate (GLA) (Figure 4A) [59,60]. Activities responsible for the conversion of EG into GLA have been identified in multiple organisms. These initial steps are catalysed by dehydrogenases with broad specificity involved in the metabolism of short-chain alcohols and aldehydes such as the propanediol oxidoreductase of E. coli (also known as lactaldehyde reductase AldA) [61]. In Pseudomonas aeruginosa and P. putida, the initial reaction is carried out by periplasmic alcohol dehydrogenases that depend on pyrroloquinoline quinone for their activity [32,62]. Once GLA is produced, the pathway proceeds to intermediates of the central metabolism through different routes depending on the organism. For instance, whereas in Escherichia coli the pathway continues to acetyl-CoA via 3-phosphoglycerate—this is called the “canonical” pathway [63]—it has been proposed that some strains of P. putida make use of the shunt that funnels GLA to the Krebs cycle via isocitrate or malate [32,64]. The genetic determinants of the canonical GLA pathway have been identified in different microorganisms. The reactions are catalysed by the enzymes GLA carboligase (Gcl), tartronate semialdehyde reductase (GlxR) and glycerate-2-kinase (GlxK), all of which are encoded in the same cluster of genes in E. coli K12 and Pseudonocardia dioxanivorans strain CB1190 [65,66].
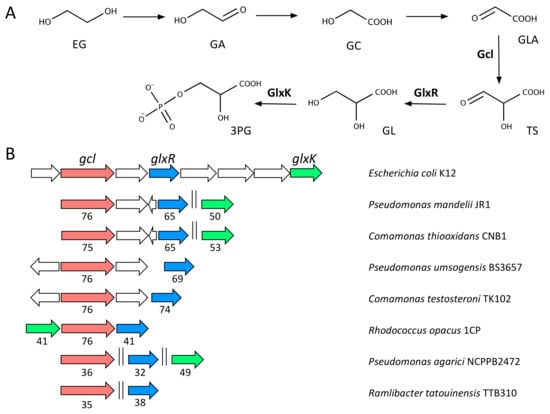
Figure 4.
(a) EG metabolism via GLA. The GLA canonical pathway described in the text is shown. The 3-phosphoglycerate (3PG) produced is later funnelled into the central metabolism via acetyl-CoA. The names of the molecules and abbreviations are: ethylene glycol, EG; glycoladehyde, GA; glycolate, GC; glyoxylate, GLA; tartronate semialdehyde, TS; glycerate, GL. (b) Genetic organisation of the genes involved in GLA metabolism identified in several genomes available in databases. Numbers below arrows indicate the percentage of identity compared to the orthologous genes present in E. coli K12 (accession: AP009048; [67]). Plots were produced with SyntTax (http://archaea.u-psud.fr/SyntTax; [55]).
Using the sequences of FucO and Gcl from E. coli as probes, we conducted an analysis of the likelihood of the occurrence of activities for EG degradation in different bacteria. Homologs to fucO are widespread and present in all organisms investigated (not shown). Added to the broad substrate specificity of the enzymes active against EG, this suggests that EG degradation is a relatively common feature in bacteria. Likewise, the canonical pathway for GLA degradation seems ubiquitous as gcl is conserved in a very large number of bacterial species (not shown). As TPA degradation genes are not as frequently present in bacterial genomes, next we investigated the presence of activities for EG degradation in the strains that we had previously identified as carriers of the tph genes for TPA mineralisation (Figure 4B). All of them contain homologs to fucO or alcohol dehydrogenases similar to pedE described in Pseudomonas species. Moreover, all of them contain homologs to gcl, glxR and glxK, although only the genetic organization of these genes in R. opacus resembles that of E. coli. Contrary to the case of TPA, our synteny search did not identify conserved transporters involved in the uptake of EG or GLA. Likewise, no regulatory elements controlling the expression of the genes responsible for GLA degradation could be found.
Taken all together, these results indicate that most organisms capable of degrading TPA are also likely able to degrade EG, thereby enabling a more efficient usage of the products resulting from PET hydrolysis. In this sense, it has been recently demonstrated that EG can be readily transformed into the bioplastic polyhydroxyalkanoate in an engineered strain of P. putida KT2440 [64], underlining the usability of microorganisms for the conversion of oil-derived plastics into bioplastics.
3. Anabolism of Monomers Used for Bio-PET Synthesis
Bio-based PET, also known as bio-PET, is the common term used to refer to a PET polymer in which at least a fraction of the constituent monomers is obtained from biological—and therefore renewable—sources. In this section we will review recent efforts to produce TPA and EG involving microorganisms at any step (Figure 5). These methods can be fully or at least partially biotic and may involve abiotic physico-chemical steps. Even if not completely green, these synthetic processes promise to decrease the dependence on virgin PET derived from fossil feedstocks and may certainly contribute to a fully circular and sustainable PET economy.
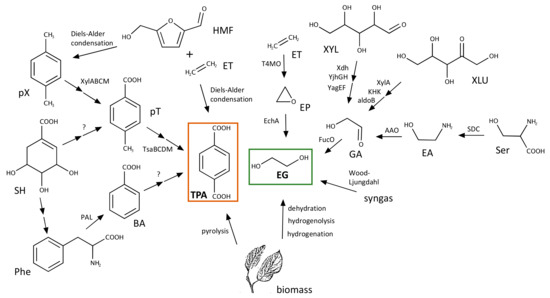
Figure 5.
Selected pathways and processes used to produce TPA and EG from renewable sources. The names of the molecules and abbreviations are: p-xylene, pX; p-toluate, pT; shikimate, SH; phenylalanine, Phe; benzoate, BA; 5-(hydromethyl)furfural, HMF; ethylene, ET; ethylene oxide, EP; xylose, XYL; xylulose, XLU; glycoaldehyde, GA; serine, Ser; ethanolamine, EA. If known, the names of enzymes/processes responsible for the different conversions are shown next to the arrows.
3.1. Biosynthesis of TPA
The microbial biosynthesis of aromatic compounds has not been characterised with the same level of detail as their degradation. Despite this, there are a number of pathways that render aromatic compounds and generally involve the metabolism of aromatic amino acids and the shikimate pathway, or the condensation of molecules such as cis,cis-muconate [68]. Unfortunately, none of the currently known pathways are likely to allow for the direct production of TPA from central intermediates. It has been proposed, however, that the shikimate pathway could be used to produce p-toluate that could later be transformed into TPA, although the activities required for this pathway have not been identified [69]. Inspired by this, we have conducted a retrosynthesis analysis of plausible biochemical reactions that could render TPA using as substrates molecules present in the metabolism of E. coli. This allows for the formulations of reactions that are chemically plausible (e.g., because the mechanism involves reactive groups following known mechanistic rules), even though this might be in the absence of any biochemical evidence [70]. This method is particularly useful for guiding the screening of genomic or metagenomic libraries in search of genes coding for enzymes capable of catalysing a proposed reaction, as well as for the lab-directed evolution of known enzymes for the efficient catalysis of novel reactions. Our analysis resulted in a number of pathways leading to benzoate that can be obtained from phenylalanine, which itself is produced from shikimic acid [71]. The last step, however, will be more difficult to take place biotically as it would involve the conversion of benzoate into TPA by direct incorporation in the aromatic ring of a carboxylic group coming from bicarbonate, a step that is typically conducted at high temperatures and in the presence of metal catalysts [72,73].
Another possibility for the sustainable production of TPA is to use aromatics obtained from renewable sources such as lignin [74]. A recent work shows that TPA biosynthesis can be achieved from p-xylene [75]. This process was successfully implemented in E. coli by the heterologous expression of segments of two different pathways. In this process, p-xylene is first converted into toluic acid by the action of the xylene monooxygenase (XylMA), benzyl alcohol dehydrogenase (XylB) and the benzaldehyde dehydrogenase (XylC) of the TOL pathway for the degradation of toluene and xylene encoded in the pWW0 plasmid of P. putida mt-2 [76]. These enzymes oxidise, respectively, one of the methyl groups of xylene to a carboxylic group via formation of the corresponding alcohol and aldehyde [77]. Toluic acid is later transformed into TPA by the action of a toluene sulfate monooxygenase (TsaMB), a 4-carboxybenzaldehyde dehydrogenase (TsaC) and a 4-carboxybenzalcohol dehydrogenase (TsaD) present in C. testosteroni T2 [78].
This biosynthetic pathway poses a significant improvement in terms of sustainability compared to conventional chemical methods [75], but obtaining p-xylene from renewable sources also poses a considerable challenge. This has been solved by using isobutanol [79,80] or biomass as substrates for different chemical transformations. Pyrolysis of biomass [81], as well as the Diels-Alders condensation of ethylene with different types of biomass-derived molecules (e.g., furans) can be used to produce p-xylene or TPA [82,83,84,85]. Ethylene itself can be produced by different biosynthetic pathways, some of which have been harnessed to produce high levels of this molecule in engineered bacteria [86,87].
3.2. Biosynthesis of EG
Given the difficulties of obtaining TPA from sources other than fossil feedstocks, bio-PET typically refers to a PET polymer in which only EG is obtained from renewable sources [88]. EG accounts for 30% of the mass of the polymer and, therefore, this is usually the maximum percentage of bio components encountered in bio-PET. As recently reviewed in [89], there are a number of artificial pathways that have been engineered to obtain EG from renewable plant feedstocks using microorganisms. Among them, biosynthesis of EG in bacteria can be achieved in high yields by a pentose pathway that uses xylose as a substrate (Figure 5). Xylose is first transformed into xylonate by the action of a dehydrogenase. After the subsequent action of a dehydratase and an aldolase, glycoaldehyde is obtained, which is finally reduced to EG by a reductase [90,91,92]. This pathway has been extensively engineered to increase production yields that currently reach a 98% of the theoretical maximum and constitute a promising alternative for the synthesis of EG [93].
The engineered xylose pathway is not the only way of obtaining EG. It can also be produced from glucose in Saccharomyces cerevisiae using glycolytic enzymes [94] and via the synthesis of serine in an engineered pathway in E. coli [95]. Serine is transformed into ethanolamine by a plant serine decarboxylase. Ethanolamine is later transformed into glycolaldehyde by an oxidase and the latter reduced to EG by a reductase (Figure 5). The pathway has been artificially reconstituted in E. coli and is also amenable to metabolic engineering efforts to improve production yields. More recent work has shown the feasibility of using synthesis gas (syngas) for the production of EG harnessing the Wood-Ljungdahl pathway of carbon fixation present in acetogenic bacterial species such as Moorella thermoacetica and Clostridium ljungdahlii [96]. In another approach, EG was obtained from gaseous alkenes by a strain of E. coli that expresses recombinantly a monooxygenase and an epoxide hydrolase [97].
Similarly to TPA, EG can also be directly obtained from biomass. This can be achieved through the dehydration of cellulosic ethanol [98], the hydrogenolysis of xylitol [99] and the hydrogenation of corn stalk [100]. This reflects a wide diversity of options for the production of EG that could be used to replace the chemical procedures relying on fossil feedstocks.
4. Future Prospects and Concluding Remarks
Here we have reviewed the potential use of PET as a feedstock for microbial biotransformations. We have identified the challenges of large-scale PET enzymatic hydrolysis and proposed strategies for the enhancement of this process using enzymes—and possibly organisms—capable of being stable and active near the Tg of the polymer.
Similarly to the case of glycolytic procedures, TPA and EG resulting from hydrolysis could be used for the synthesis of fresh PET, but we also advocate for their biotransformation into molecules or processes with added value. An example of this could be their use in microbial fuel cells for the production of electricity that has been achieved using TPA as a carbon source [101]. TPA metabolism is neither widespread nor diverse in the genomes currently available. This could constitute a bottleneck for the development of future applications that currently have adipic acid as the main target of molecules funnelled through the PCA pathways. EG metabolic genes, on the contrary, are found in numerous organisms and encode a more diverse metabolism, likely enabling a variety of applications.
Hand in hand with an efficient degradation of PET, a circular economy of this polymer requires a sustainable large-scale synthesis of TPA and EG. We have reviewed a number of efforts made for the biosynthesis of bio-PET using renewable sources. On this front a milestone seems to have been reached recently with the production of the first bottle that is completely made of monomers obtained from biological sources [102]. Any method, including those reaching a maximum of 30% bio-PET, have a significantly lower carbon footprint compared with the synthesis of oil-derived plastics and are worth pursuing. By using plant biomass, it is possible to contribute to CO2 fixation, although a major breakthrough would be to obtain TPA or EG with engineered microorganisms directly from CO2.
Overall, the prospects for a circular bio-based economy of PET are encouraging, and most of the technological hurdles for either biodegradation or biosynthesis have already been overcome, or there are alternatives or clear strategies to overcome them. Although bio-approaches to the PET economy might not be as profitable as the current status quo in the short term, there is an undeniable pressure from the general public to manage PET differently, and this is already producing changes in policies and regulations. In our view, this will at least partially bridge the strict financial gap compared to chemical processes, which will enable itself the diversification of applications of PET including its upcycling in other molecules. In the long run, this will have a positive impact on recycling rates and will also lower the environmental release of PET waste, therefore contributing to solving an imperative environmental concern.
Funding
J.I.J., W.Z., M.S., A.A.S., J.K. and R.W. would like to acknowledge the support received from the European Union’s Horizon 2020 research and innovation programme under grant agreement no. 633962 for the project P4SB. J.I.J., A.A.S. and J.K. acknowledge the support from the Biotechnology and Biological Sciences Research Council (BBSRC) (grant BB/M009769/1). J.L.F. acknowledges the support from the Engineering and Physical Sciences Research Council (EPSRC) (grant EP/N025504/1). U.A. is the recipient of a PhD studentship from the Petroleum Technology Development Fund of Nigeria.
Conflicts of Interest
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, or in the decision to publish the results.
References
- Andrady, A.L.; Neal, M.A. Applications and societal benefits of plastics. Philos. Trans. R. Soc. B Biol. Sci. 2009, 364, 1977–1984. [Google Scholar] [CrossRef] [PubMed]
- Scalenghe, R. Resource or waste? A perspective of plastics degradation in soil with a focus on end-of-life options. Heliyon 2018, 4, e00941. [Google Scholar] [CrossRef]
- PlasticsEurope: Plastics—The Facts 2018. Available online: https://www.plasticseurope.org/en/resources/publications/619-plastics-facts-2018 (accessed on 16 May 2019).
- Upasen, S.; Wattanachai, P. Packaging to prolong shelf life of preservative-free white bread. Heliyon 2018, 4, e00802. [Google Scholar] [CrossRef] [PubMed]
- Ryan, P.G.; Moore, C.J.; van Franeker, J.A.; Moloney, C.L. Monitoring the abundance of plastic debris in the marine environment. Philos. Trans. R. Soc. B Biol. Sci. 2009, 364, 1999–2012. [Google Scholar] [CrossRef] [PubMed]
- Barnes, D.K.A.; Galgani, F.; Thompson, R.C.; Barlaz, M. Accumulation and fragmentation of plastic debris in global environments. Philos. Trans. R. Soc. B Biol. Sci. 2009, 364, 1985–1998. [Google Scholar] [CrossRef] [PubMed]
- Derraik, J.G. The pollution of the marine environment by plastic debris: A review. Mar. Pollut. Bull. 2002, 44, 842–852. [Google Scholar] [CrossRef]
- Kubowicz, S.; Booth, A.M. Biodegradability of plastics: Challenges and misconceptions. Environ. Sci. Technol. 2017, 51, 12058–12060. [Google Scholar] [CrossRef]
- Alimba, C.G.; Faggio, C. Microplastics in the marine environment: Current trends in environmental pollution and mechanisms of toxicological profile. Environ. Toxicol. Pharmacol. 2019, 68, 61–74. [Google Scholar] [CrossRef]
- Geyer, R.; Jambeck, J.R.; Law, K.L. Production, use, and fate of all plastics ever made. Sci. Adv. 2017, 3, e1700782. [Google Scholar] [CrossRef]
- Wierckx, N.; Prieto, M.A.; Pomposiello, P.; de Lorenzo, V.; O’Connor, K.; Blank, L.M. Plastic waste as a novel substrate for industrial biotechnology. Microb. Biotechnol. 2015, 8, 900–903. [Google Scholar] [CrossRef] [PubMed]
- Wierckx, N.; Narancic, T.; Eberlein, C.; Wei, R.; Drzyzga, O.; Magnin, A.; Ballerstedt, H.; Kenny, S.T.; Pollet, E.; Avérous, L.; et al. Plastic biodegradation: Challenges and opportunities. In Consequences of Microbial Interactions with Hydrocarbons, Oils, and Lipids: Biodegradation and Bioremediation; Springer: Cham, Switzerland, 2018; pp. 1–29. [Google Scholar]
- Narancic, T.; O’Connor, K.E. Microbial biotechnology addressing the plastic waste disaster. Microb. Biotechnol. 2017, 10, 1232–1235. [Google Scholar] [CrossRef] [PubMed]
- Koshti, R.; Mehta, L.; Samarth, N. Biological recycling of polyethylene terephthalate: A mini-review. J. Polym. Environ. 2018, 26, 3520–3529. [Google Scholar] [CrossRef]
- Arroyo, M. Thermoplastic polyesters. In Handbook of Thermoplastics; Olabisi, O., Ed.; Marcel Dekker: New York, NY, USA, 1997; pp. 417–448. [Google Scholar]
- Ji, L.N. Study on preparation process and properties of polyethylene terephthalate (PET). Appl. Mech. Mater. 2013, 312, 406–410. [Google Scholar] [CrossRef]
- NAPCOR. Report on Postconsumer PET Container Recycling Activity in 2017. Available online: https://napcor.com/wp-content/uploads/2018/11/NAPCOR_2017RateReport_FINAL.pdf (accessed on 16 May 2019).
- Nikles, D.E.; Farahat, M.S. New motivation for the depolymerization products derived from poly(ethylene terephthalate) (PET) waste: A review. Macromol. Mater. Eng. 2005, 290, 13–30. [Google Scholar] [CrossRef]
- Al-Sabagh, A.M.; Yehia, F.Z.; Eshaq, G.; Rabie, A.M.; ElMetwally, A.E. Greener routes for recycling of polyethylene terephthalate. Egypt. J. Pet. 2016, 25, 53–64. [Google Scholar] [CrossRef]
- Sinha, V.; Patel, M.; Patel, J. Pet Waste Management by Chemical Recycling: A Review. J. Polym. Environ. 2010, 18, 8–25. [Google Scholar] [CrossRef]
- Furtwengler, P.; Avérous, L. Renewable polyols for advanced polyurethane foams from diverse biomass resources. Polym. Chem. 2018, 9, 4258–4287. [Google Scholar] [CrossRef]
- Webb, H.K.; Arnott, J.; Crawford, R.J.; Ivanova, E.P. Plastic degradation and its environmental implications with special reference to poly(ethylene terephthalate). Polymers 2013, 5, 1–18. [Google Scholar] [CrossRef]
- Roth, C.; Wei, R.; Oeser, T.; Then, J.; Föllner, C.; Zimmermann, W.; Sträter, N. Structural and functional studies on a thermostable polyethylene terephthalate degrading hydrolase from Thermobifida fusca. Appl. Microbiol. Biotechnol. 2014, 98, 7815–7823. [Google Scholar] [CrossRef]
- Yoshida, S.; Hiraga, K.; Takehana, T.; Taniguchi, I.; Yamaji, H.; Maeda, Y.; Toyohara, K.; Miyamoto, K.; Kimura, Y.; Oda, K. A bacterium that degrades and assimilates poly(ethylene terephthalate). Science 2016, 351, 1196–1199. [Google Scholar] [CrossRef] [PubMed]
- Wei, R.; Oeser, T.; Zimmermann, W. Synthetic polyester-hydrolyzing enzymes from thermophilic actinomycetes. Adv. Appl. Microbiol. 2014, 89, 267–305. [Google Scholar]
- Wei, R.; Zimmermann, W. Biocatalysis as a green route for recycling the recalcitrant plastic polyethylene terephthalate. Microb. Biotechnol. 2017, 10, 1302–1307. [Google Scholar] [CrossRef] [PubMed]
- Kasai, D.; Fujinami, T.; Abe, T.; Mase, K.; Katayama, Y.; Fukuda, M.; Masai, E. Uncovering the protocatechuate 2,3-cleavage pathway genes. J. Bacteriol. 2009, 191, 6758–6768. [Google Scholar] [CrossRef] [PubMed]
- Maruyama, K.; Shibayama, T.; Ichikawa, A.; Sakou, Y.; Yamada, S.; Sugisaki, H. Cloning and characterization of the genes encoding enzymes for the protocatechuate meta-degradation pathway of Pseudomonas ochraceae NGJ1. Biosci. Biotechnol. Biochem. 2004, 68, 1434–1441. [Google Scholar] [CrossRef]
- Frazee, R.W.; Livingston, D.M.; LaPorte, D.C.; Lipscomb, J.D. Cloning, sequencing, and expression of the Pseudomonas putida protocatechuate 3,4-dioxygenase genes. J. Bacteriol. 1993, 175, 6194–6202. [Google Scholar] [CrossRef]
- Harwood, C.S.; Parales, R.E. The β-ketoadipate pathway and the biology of self-identity. Annu. Rev. Microbiol. 1996, 50, 553–590. [Google Scholar] [CrossRef]
- Trifunović, D.; Schuchmann, K.; Müller, V. Ethylene glycol metabolism in the acetogen Acetobacterium woodii. J. Bacteriol. 2016, 198, 1058–1065. [Google Scholar] [CrossRef]
- Mückschel, B.; Simon, O.; Klebensberger, J.; Graf, N.; Rosche, B.; Altenbuchner, J.; Pfannstiel, J.; Huber, A.; Hauer, B. Ethylene glycol metabolism by Pseudomonas putida. Appl. Environ. Microbiol. 2012, 78, 8531–8539. [Google Scholar] [CrossRef] [PubMed]
- Zimmermann, W.; Billig, S. Enzymes for the biofunctionalization of poly(ethylene terephthalate). In Biofunctionalization of Polymers and Their Applications; Nyanhongo, G.S., Steiner, W., Gübitz, G., Eds.; Springer: Berlin/Heidelberg, Germany, 2011; pp. 97–120. [Google Scholar]
- Herrero Acero, E.; Ribitsch, D.; Steinkellner, G.; Gruber, K.; Greimel, K.; Eiteljoerg, I.; Trotscha, E.; Wei, R.; Zimmermann, W.; Zinn, M.; et al. Enzymatic surface hydrolysis of PET: Effect of structural diversity on kinetic properties of cutinases from Thermobifida. Macromolecules 2011, 44, 4632–4640. [Google Scholar] [CrossRef]
- Danso, D.; Schmeisser, C.; Chow, J.; Zimmermann, W.; Wei, R.; Leggewie, C.; Li, X.; Hazen, T.; Streit, W.R. New insights into the function and global distribution of polyethylene terephthalate (PET)-Degrading bacteria and enzymes in marine and terrestrial metagenomes. Appl. Environ. Microbiol. 2018, 84, e02773-17. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Ma, Z.; Li, L. Flow-induced crystallization of polymers: Molecular and thermodynamic considerations. Macromolecules 2016, 49, 1505–1517. [Google Scholar] [CrossRef]
- Ronkvist, Å.M.; Xie, W.; Lu, W.; Gross, R.A. Cutinase-catalyzed hydrolysis of poly(ethylene terephthalate). Macromolecules 2009, 42, 5128–5138. [Google Scholar] [CrossRef]
- Marten, E.; Müller, R.-J.; Deckwer, W.-D. Studies on the enzymatic hydrolysis of polyesters. II. Aliphatic–aromatic copolyesters. Polym. Degrad. Stab. 2005, 88, 371–381. [Google Scholar] [CrossRef]
- Shirke, A.N.; White, C.; Englaender, J.A.; Zwarycz, A.; Butterfoss, G.L.; Linhardt, R.J.; Gross, R.A. Stabilizing Leaf and branch Compost Cutinase (LCC) with glycosylation: Mechanism and effect on PET hydrolysis. Biochemistry 2018, 57, 1190–1200. [Google Scholar] [CrossRef]
- Guyot, S.; Pottier, L.; Hartmann, A.; Ragon, M.; Hauck Tiburski, J.; Molin, P.; Ferret, E.; Gervais, P. Extremely rapid acclimation of Escherichia coli to high temperature over a few generations of a fed-batch culture during slow warming. Microbiologyopen 2014, 3, 52–63. [Google Scholar] [CrossRef]
- Rudolph, B.; Gebendorfer, K.M.; Buchner, J.; Winter, J. Evolution of Escherichia coli for growth at high temperatures. J. Biol. Chem. 2010, 285, 19029–19034. [Google Scholar] [CrossRef]
- Austin, H.P.; Allen, M.D.; Donohoe, B.S.; Rorrer, N.A.; Kearns, F.L.; Silveira, R.L.; Pollard, B.C.; Dominick, G.; Duman, R.; El Omari, K.; et al. Characterization and engineering of a plastic-degrading aromatic polyesterase. Proc. Natl. Acad. Sci. USA 2018, 115, E4350–E4357. [Google Scholar] [CrossRef]
- Wei, R.; Zimmermann, W. Microbial enzymes for the recycling of recalcitrant petroleum-based plastics: How far are we? Microb. Biotechnol. 2017, 10, 1308–1322. [Google Scholar] [CrossRef] [PubMed]
- Barth, M.; Oeser, T.; Wei, R.; Then, J.; Schmidt, J.; Zimmermann, W. Effect of hydrolysis products on the enzymatic degradation of polyethylene terephthalate nanoparticles by a polyester hydrolase from Thermobifida fusca. Biochem. Eng. J. 2015, 93, 222–228. [Google Scholar] [CrossRef]
- Barth, M.; Wei, R.; Oeser, T.; Then, J.; Schmidt, J.; Wohlgemuth, F.; Zimmermann, W. Enzymatic hydrolysis of polyethylene terephthalate films in an ultrafiltration membrane reactor. J. Membr. Sci. 2015, 494, 182–187. [Google Scholar] [CrossRef]
- Carniel, A.; Valoni, É.; Nicomedes, J.; Gomes, A.d.C.; Castro, A.M.d. Lipase from Candida antarctica (CALB) and cutinase from Humicola insolens act synergistically for PET hydrolysis to terephthalic acid. Process Biochem. 2017, 59, 84–90. [Google Scholar] [CrossRef]
- Wei, R.; Oeser, T.; Schmidt, J.; Meier, R.; Barth, M.; Then, J.; Zimmermann, W. Engineered bacterial polyester hydrolases efficiently degrade polyethylene terephthalate due to relieved product inhibition. Biotechnol. Bioeng. 2016, 113, 1658–1665. [Google Scholar] [CrossRef]
- Choi, K.Y.; Sul, W.J.; Kim, Y.M.; Kim, E.; Kim, D.; Zylstra, G.J.; Chae, J.-C. Molecular and biochemical analysis of phthalate and terephthalate degradation by Rhodococcus sp. strain DK17. FEMS Microbiol. Lett. 2005, 252, 207–213. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.Z.; Zhou, Y.; Zylstra, G.J. Molecular analysis of isophthalate and terephthalate degradation by Comamonas testosteroni YZW-D. Environ. Health Perspect. 1995, 103, 9–12. [Google Scholar] [PubMed]
- Sasoh, M.; Masai, E.; Ishibashi, S.; Hara, H.; Kamimura, N.; Miyauchi, K.; Fukuda, M. Characterization of the terephthalate degradation genes of Comamonas sp. strain E6. Appl. Environ. Microbiol. 2006, 72, 1825–1832. [Google Scholar] [CrossRef] [PubMed]
- Kasai, D.; Kitajima, M.; Fukuda, M.; Masai, E. Transcriptional regulation of the terephthalate catabolism operon in Comamonas sp. strain E6. Appl. Environ. Microbiol. 2010, 76, 6047–6055. [Google Scholar] [CrossRef] [PubMed]
- Hosaka, M.; Kamimura, N.; Toribami, S.; Mori, K.; Kasai, D.; Fukuda, M.; Masai, E. Novel tripartite Aromatic Acid Transporter Essential for Terephthalate Uptake in Comamonas sp. Strain E6. Appl. Environ. Microbiol. 2013, 79, 6148–6155. [Google Scholar] [CrossRef] [PubMed]
- Nichols, N.N.; Harwood, C.S. PcaK, a high-affinity permease for the aromatic compounds 4-hydroxybenzoate and protocatechuate from Pseudomonas putida. J. Bacteriol. 1997, 179, 5056–5061. [Google Scholar] [CrossRef] [PubMed]
- Noda, Y.; Nishikawa, S.; Shiozuka, K.; Kadokura, H.; Nakajima, H.; Yoda, K.; Katayama, Y.; Morohoshi, N.; Haraguchi, T.; Yamasaki, M. Molecular cloning of the protocatechuate 4,5-dioxygenase genes of Pseudomonas paucimobilis. J. Bacteriol. 1990, 172, 2704–2709. [Google Scholar] [CrossRef]
- Oberto, J. SyntTax: A web server linking synteny to prokaryotic taxonomy. BMC Bioinf. 2013, 14, 4. [Google Scholar] [CrossRef] [PubMed]
- Wells Jr, T.; Ragauskas, A.J. Biotechnological opportunities with the b-ketoadipate pathway. Trends Biotechnol. 2012, 30, 627–637. [Google Scholar] [CrossRef]
- Johnson, C.W.; Salvachúa, D.; Khanna, P.; Smith, H.; Peterson, D.J.; Beckham, G.T. Enhancing muconic acid production from glucose and lignin-derived aromatic compounds via increased protocatechuate decarboxylase activity. Metab. Eng. Commun. 2016, 3, 111–119. [Google Scholar] [CrossRef] [PubMed]
- Vardon, D.R.; Franden, M.A.; Johnson, C.W.; Karp, E.M.; Guarnieri, M.T.; Linger, J.G.; Salm, M.J.; Strathmann, T.J.; Beckham, G.T. Adipic acid production from lignin. Energy Environ. Sci. 2015, 8, 617–628. [Google Scholar] [CrossRef]
- Child, J.; Willetts, A. Microbial metabolism of aliphatic glycols bacterial metabolism of ethylene glycol. Biochim. Biophys. Acta Gen. Subj. 1978, 538, 316–327. [Google Scholar] [CrossRef]
- Kataoka, M.; Sasaki, M.; Hidalgo, A.-R.G.D.; Nakano, M.; Shimizu, S. Glycolic acid production using ethylene glycol-oxidizing microorganisms. Biosci. Biotechnol. Biochem. 2001, 65, 2265–2270. [Google Scholar] [CrossRef] [PubMed]
- Boronat, A.; Aguilar, J. Rhamnose-induced propanediol oxidoreductase in Escherichia coli: Purification, properties, and comparison with the fucose-induced enzyme. J. Bacteriol. 1979, 140, 320–326. [Google Scholar]
- Wehrmann, M.; Billard, P.; Martin-Meriadec, A.; Zegeye, A.; Klebensberger, J. Functional role of lanthanides in enzymatic activity and transcriptional regulation of pyrroloquinoline quinone-dependent alcohol dehydrogenases in Pseudomonas putida KT2440. MBio 2017, 8, e00570-17. [Google Scholar] [CrossRef]
- Boronat, A.; Caballero, E.; Aguilar, J. Experimental evolution of a metabolic pathway for ethylene glycol utilization by Escherichia coli. J. Bacteriol. 1983, 153, 134–139. [Google Scholar]
- Franden, M.A.; Jayakody, L.N.; Li, W.-J.; Wagner, N.J.; Cleveland, N.S.; Michener, W.E.; Hauer, B.; Blank, L.M.; Wierckx, N.; Klebensberger, J.; et al. Engineering Pseudomonas putida KT2440 for efficient ethylene glycol utilization. Metab. Eng. 2018, 48, 197–207. [Google Scholar] [CrossRef]
- Cusa, E.; Obradors, N.; Baldomà, L.; Badía, J.; Aguilar, J. Genetic analysis of a chromosomal region containing genes required for assimilation of allantoin nitrogen and linked glyoxylate metabolism in Escherichia coli. J. Bacteriol. 1999, 181, 7479–7484. [Google Scholar] [PubMed]
- Grostern, A.; Sales, C.M.; Zhuang, W.-Q.; Erbilgin, O.; Alvarez-Cohen, L. Glyoxylate metabolism Is a key feature of the metabolic degradation of 1,4-dioxane by Pseudonocardia dioxanivorans strain CB1190. Appl. Environ. Microbiol. 2012, 78, 3298–3308. [Google Scholar] [CrossRef] [PubMed]
- Wanner, B.L.; Wishart, D.; Blattner, F.R.; Thomas, G.H.; Plunkett Guy, I.I.I.; Mori, H.; Keseler, I.M.; Glasner, J.D.; Rudd, K.E.; Serres, M.H.; et al. Escherichia coli K-12: A cooperatively developed annotation snapshot—2005. Nucleic Acids Res. 2006, 34, 1–9. [Google Scholar]
- Huccetogullari, D.; Luo, Z.W.; Lee, S.Y. Metabolic engineering of microorganisms for production of aromatic compounds. Microb. Cell Fact. 2019, 18, 41. [Google Scholar] [CrossRef]
- Osterhout, R.E.; Burgard, A.P.; Pharkya, P.; Burk, P. Microorganisms and Methods for the Biosynthesis of Aromatics, 2,4-Pentadienoate and 1,3-Butadiene. U.S. Patent 8715957 B2, 26 July 2011. [Google Scholar]
- Delépine, B.; Duigou, T.; Carbonell, P.; Faulon, J.-L. RetroPath2.0: A retrosynthesis workflow for metabolic engineers. Metab. Eng. 2018, 45, 158–170. [Google Scholar] [CrossRef] [PubMed]
- Moore, B.S.; Hertweck, C.; Hopke, J.N.; Izumikawa, M.; Kalaitzis, J.A.; Nilsen, G.; O’Hare, T.; Piel, J.; Shipley, P.R.; Xiang, L.; et al. Plant-like biosynthetic pathways in bacteria: From benzoic acid to chalcone. J. Nat. Prod. 2002, 65, 1956–1962. [Google Scholar] [CrossRef]
- Lind, W.; Campbell, R. Preparation of Potassium Terephthalate. U.S. Patent 3761515 A, 21 October 1971. [Google Scholar]
- Bernhard, R. Production of Terephthalic Acid. U.S. Patent 2823229 A, 20 June 1956. [Google Scholar]
- Graglia, M.; Kanna, N.; Esposito, D. Lignin refinery: Towards the preparation of renewable aromatic building blocks. ChemBioEng Rev. 2015, 2, 377–392. [Google Scholar] [CrossRef]
- Luo, Z.W.; Lee, S.Y. Biotransformation of p-xylene into terephthalic acid by engineered Escherichia coli. Nat. Commun. 2017, 8, 15689. [Google Scholar] [CrossRef] [PubMed]
- Franklin, F.C.; Bagdasarian, M.; Bagdasarian, M.M.; Timmis, K.N. Molecular and functional analysis of the TOL plasmid pWWO from Pseudomonas putida and cloning of genes for the entire regulated aromatic ring meta cleavage pathway. Proc. Natl. Acad. Sci. USA 1981, 78, 7458–7462. [Google Scholar] [CrossRef] [PubMed]
- Harayama, S.; Rekik, M.; Wubbolts, M.; Rose, K.; Leppik, R.A.; Timmis, K.N. Characterization of five genes in the upper-pathway operon of TOL plasmid pWW0 from Pseudomonas putida and identification of the gene products. J. Bacteriol. 1989, 171, 5048–5055. [Google Scholar] [CrossRef] [PubMed]
- Junker, F.; Kiewitz, R.; Cook, A.M. Characterization of the p-toluenesulfonate operon tsaMBCD and tsaR in Comamonas testosteroni T-2. J. Bacteriol. 1997, 179, 919–927. [Google Scholar] [CrossRef]
- Dedov, A.G.; Loktev, A.S.; Karavaev, A.A.; Moiseev, I.I. A novel direct catalytic production of p-xylene from isobutanol. Mendeleev Commun. 2018, 28, 352–353. [Google Scholar] [CrossRef]
- Peters, M.; Taylor, J.; Jenni, M.; Manzer, L.; Henton, D. Integrated process to selectively convert renewable isobutanol to p-xylene. U.S. Patent US 20110087000 A1, 6 October 2010. [Google Scholar]
- Chang, R.; Zhu, L.; Jin, F.; Fan, M.; Liu, J.; Jia, Q.; Tang, C.; Li, Q. Production of bio-based p-xylene via catalytic pyrolysis of biomass over metal oxide-modified HZSM-5 zeolites. J. Chem. Technol. Biotechnol. 2018, 93, 3292–3301. [Google Scholar] [CrossRef]
- Pacheco, J.J.; Davis, M.E. Synthesis of terephthalic acid via Diels-Alder reactions with ethylene and oxidized variants of 5-hydroxymethylfurfural. Proc. Natl. Acad. Sci. USA 2014, 111, 8363–8367. [Google Scholar] [CrossRef] [PubMed]
- Shiramizu, M.; Toste, F.D. On the Diels–Alder approach to solely biomass-derived polyethylene terephthalate (PET): Conversion of 2,5-dimethylfuran and acrolein into p-xylene. Chem. A Eur. J. 2011, 17, 12452–12457. [Google Scholar] [CrossRef] [PubMed]
- Maneffa, A.; Priecel, P.; Lopez-Sanchez, J.A. Biomass-derived renewable aromatics: Selective routes and outlook for p-xylene commercialisation. ChemSusChem 2016, 9, 2736–2748. [Google Scholar] [CrossRef] [PubMed]
- Williams, C.L.; Chang, C.-C.; Do, P.; Nikbin, N.; Caratzoulas, S.; Vlachos, D.G.; Lobo, R.F.; Fan, W.; Dauenhauer, P.J. Cycloaddition of biomass-derived furans for catalytic production of renewable p-xylene. ACS Catal. 2012, 2, 935–939. [Google Scholar] [CrossRef]
- Van de Poel, B.; Cooper, E.D.; Delwiche, C.F.; Chang, C. An evolutionary perspective on the plant hormone ethylene. In Ethylene in Plants; Springer: Dordrecht, The Netherlands; pp. 109–134.
- Digiacomo, F.; Girelli, G.; Aor, B.; Marchioretti, C.; Pedrotti, M.; Perli, T.; Tonon, E.; Valentini, V.; Avi, D.; Ferrentino, G.; et al. Ethylene-producing bacteria that ripen fruit. ACS Synth. Biol. 2014, 3, 935–938. [Google Scholar] [CrossRef] [PubMed]
- Steeman, A. PET is PET—Petro-PET or Bio-PET. Available online: https://bestinpackaging.com/2011/07/13/pet-is-pet-petro-pet-or-bio-pet/ (accessed on 3 March 2019).
- Salusjärvi, L.; Havukainen, S.; Koivistoinen, O.; Toivari, M. Biotechnological production of glycolic acid and ethylene glycol: Current state and perspectives. Appl. Microbiol. Biotechnol. 2019, 103, 2525–2535. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Ramos, K.R.M.; Valdehuesa, K.N.G.; Nisola, G.M.; Lee, W.-K.; Chung, W.-J. Biosynthesis of ethylene glycol in Escherichia coli. Appl. Microbiol. Biotechnol. 2013, 97, 3409–3417. [Google Scholar] [CrossRef] [PubMed]
- Alkim, C.; Cam, Y.; Trichez, D.; Auriol, C.; Spina, L.; Vax, A.; Bartolo, F.; Besse, P.; François, J.M.; Walther, T. Optimization of ethylene glycol production from (d)-xylose via a synthetic pathway implemented in Escherichia coli. Microb. Cell Fact. 2015, 14, 127. [Google Scholar] [CrossRef]
- Cam, Y.; Alkim, C.; Trichez, D.; Trebosc, V.; Vax, A.; Bartolo, F.; Besse, P.; François, J.M.; Walther, T. Engineering of a synthetic metabolic pathway for the assimilation of (d)-xylose into value-added chemicals. ACS Synth. Biol. 2016, 5, 607–618. [Google Scholar] [CrossRef] [PubMed]
- Cabulong, R.B.; Valdehuesa, K.N.G.; Ramos, K.R.M.; Nisola, G.M.; Lee, W.-K.; Lee, C.R.; Chung, W.-J. Enhanced yield of ethylene glycol production from d-xylose by pathway optimization in Escherichia coli. Enzyme Microb. Technol. 2017, 97, 11–20. [Google Scholar] [CrossRef] [PubMed]
- Uranukul, B.; Woolston, B.M.; Fink, G.R.; Stephanopoulos, G. Biosynthesis of monoethylene glycol in Saccharomyces cerevisiae utilizing native glycolytic enzymes. Metab. Eng. 2019, 51, 20–31. [Google Scholar] [CrossRef] [PubMed]
- Pereira, B.; Zhang, H.; De Mey, M.; Lim, C.G.; Li, Z.-J.; Stephanopoulos, G. Engineering a novel biosynthetic pathway in Escherichia coli for production of renewable ethylene glycol. Biotechnol. Bioeng. 2016, 113, 376–383. [Google Scholar] [CrossRef]
- Islam, M.A.; Hadadi, N.; Ataman, M.; Hatzimanikatis, V.; Stephanopoulos, G. Exploring biochemical pathways for mono-ethylene glycol (MEG) synthesis from synthesis gas. Metab. Eng. 2017, 41, 173–181. [Google Scholar] [CrossRef] [PubMed]
- Desai, S.H.; Koryakina, I.; Case, A.E.; Toney, M.D.; Atsumi, S. Biological conversion of gaseous alkenes to liquid chemicals. Metab. Eng. 2016, 38, 98–104. [Google Scholar] [CrossRef] [PubMed]
- Ji, N.; Zhang, T.; Zheng, M.; Wang, A.; Wang, H.; Wang, X.; Shu, Y.; Stottlemyer, A.L.; Chen, J.G. Catalytic conversion of cellulose into ethylene glycol over supported carbide catalysts. Catal. Today 2009, 147, 77–85. [Google Scholar] [CrossRef]
- Sun, J.; Liu, H. Selective hydrogenolysis of biomass-derived xylitol to ethylene glycol and propylene glycol on supported Ru catalysts. Green Chem. 2011, 13, 135–142. [Google Scholar] [CrossRef]
- Pang, J.; Zheng, M.; Wang, A.; Zhang, T. Catalytic hydrogenation of corn stalk to ethylene glycol and 1,2-propylene glycol. Ind. Eng. Chem. Res. 2011, 50, 6601–6608. [Google Scholar] [CrossRef]
- Song, T.; Xu, Y.; Ye, Y.; Chen, Y.; Shen, S. Electricity generation from terephthalic acid using a microbial fuel cell. J. Chem. Technol. Biotechnol. 2009, 84, 356–360. [Google Scholar] [CrossRef]
- The Coca Cola Company Coca-Cola Produces World’s First PET Bottle Made Entirely From Plants. Available online: https://www.coca-colacompany.com/press-center/press-releases/coca-cola-produces-worlds-first-pet-bottle-made-entirely-from-plants (accessed on 5 March 2019).
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).