Several One-Domain Zinc Finger µ-Proteins of Haloferax Volcanii Are Important for Stress Adaptation, Biofilm Formation, and Swarming
Abstract
:1. Introduction
2. Materials and Methods
2.1. Strains and Media
2.2. Generation of in Frame Deletion Mutants
2.3. Growth Analyses
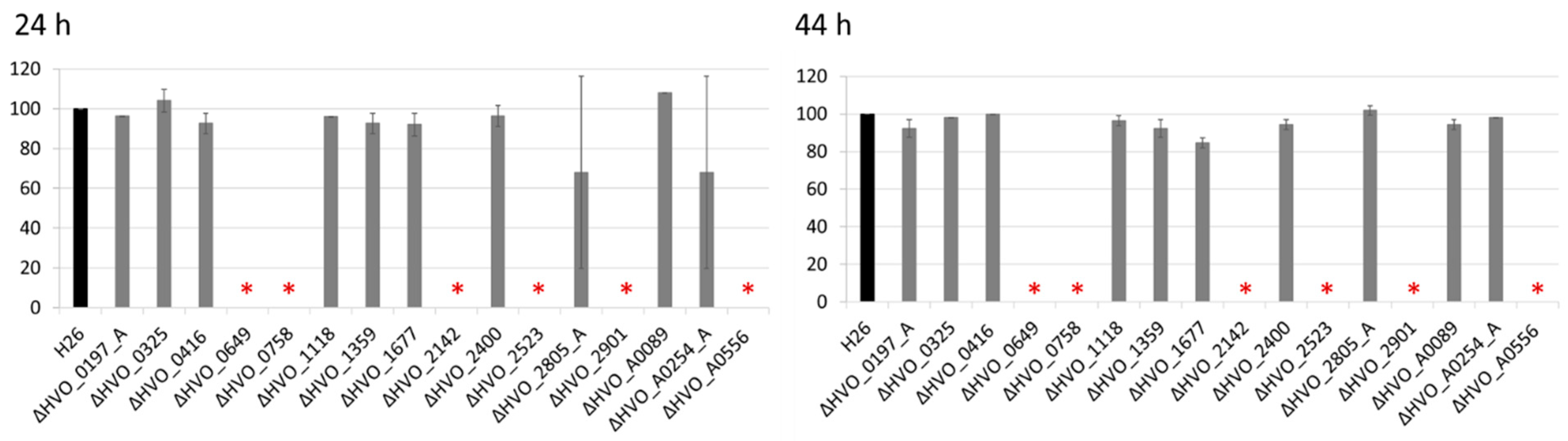
2.4. Analysis of the Sensitivity to Bile Acids
2.5. Swarm Plate Assays
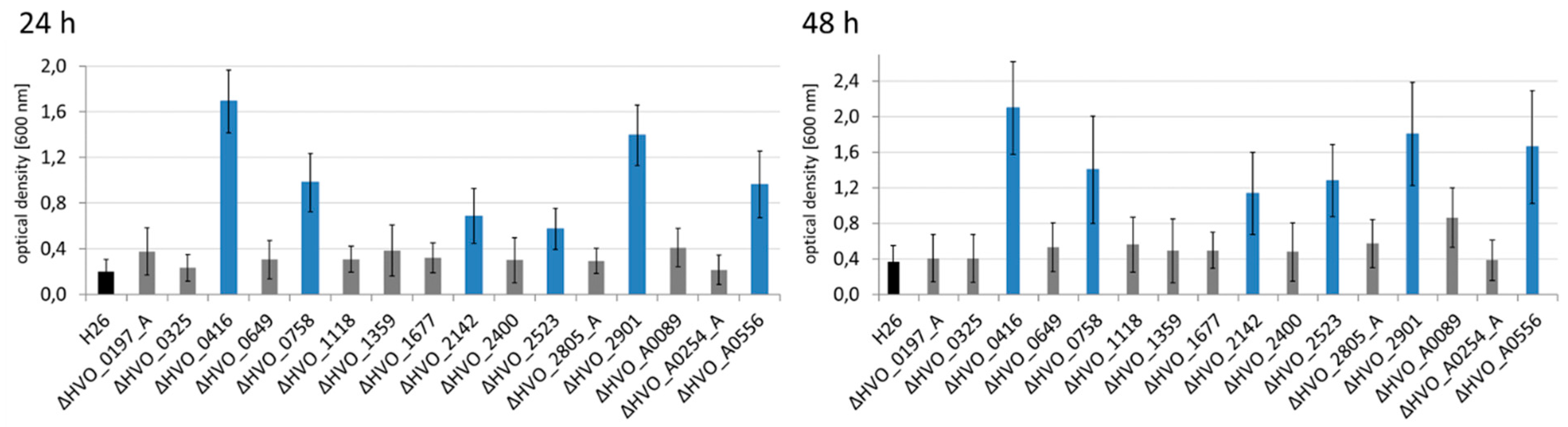
2.6. Analysis of Biofilm Formation
2.7. Databases and Bioinformatics Analyses
3. Results
3.1. Selection of Genes and Generation of In Frame Deletion Mutants
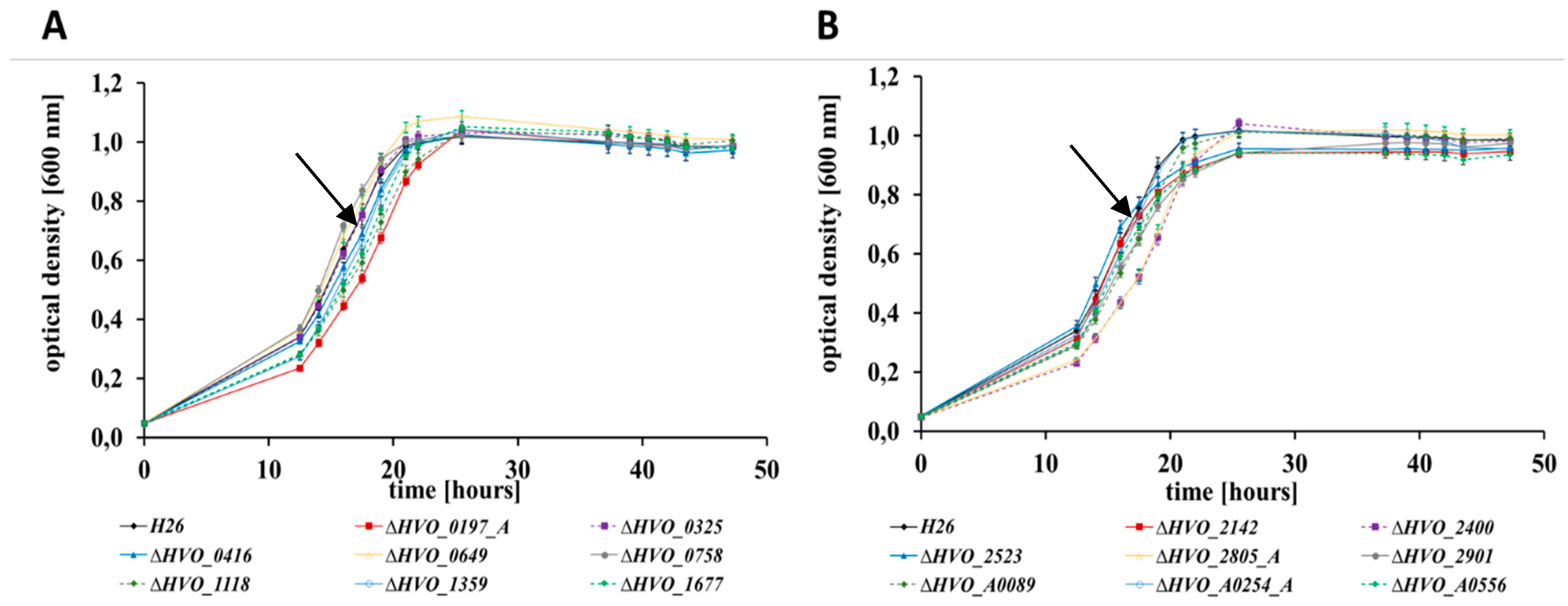
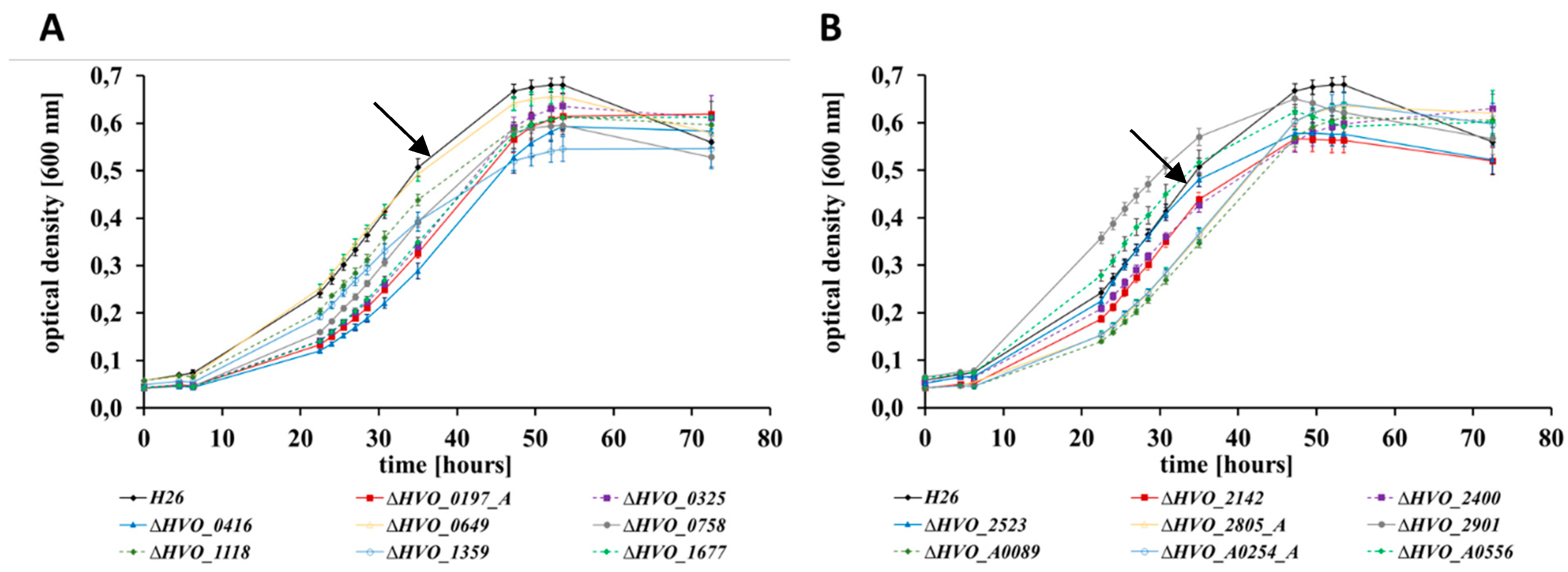
3.2. Growth Analyses in Media with Different Carbon and Phosphate Sources
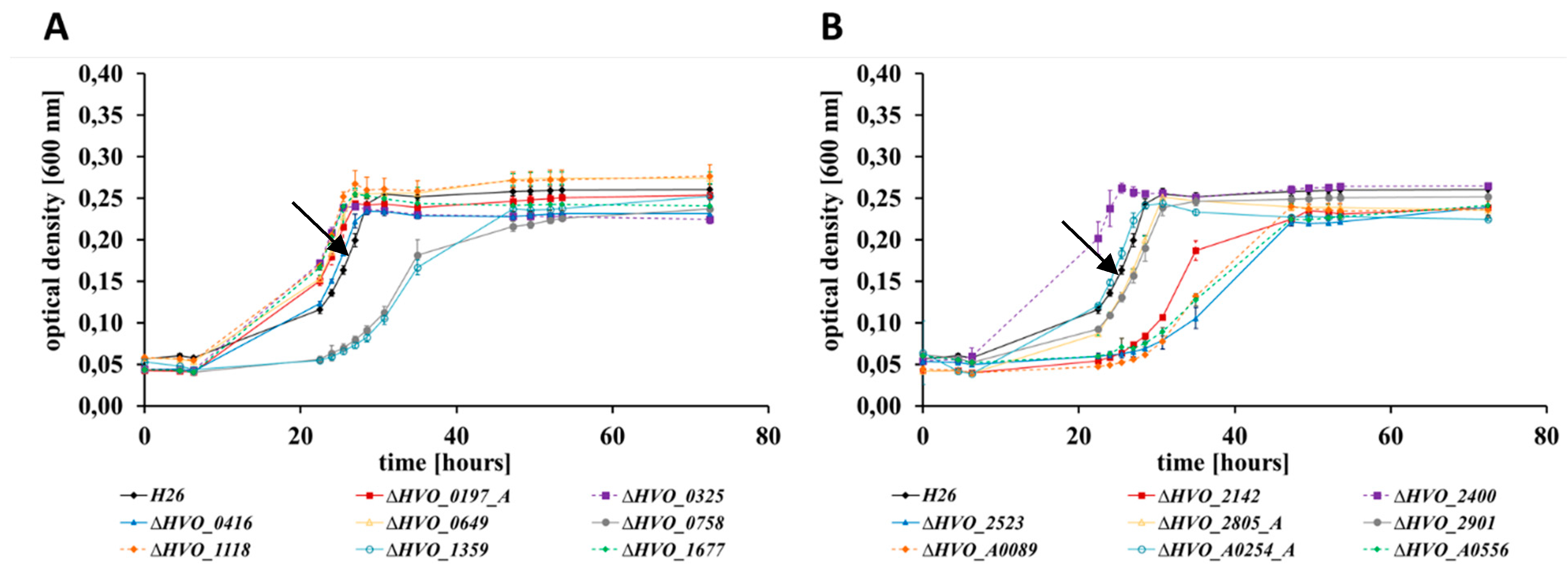
3.3. Sensitivities to Bile Acids
3.4. The Different Life Styles of H. volcanii: Swarming and Biofilm Formation
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Duval, M.; Cossart, P. Small bacterial and phagic proteins: An updated view on a rapidly moving field. Curr. Opin. Microbiol. 2017, 39, 81–88. [Google Scholar] [CrossRef] [PubMed]
- Baumgartner, D.; Kopf, M.; Klähn, S.; Steglich, C.; Hess, W.R. Small proteins in cyanobacteria provide a paradigm for the functional analysis of the bacterial micro-proteome. BMC Microbiol. 2016, 16, 285. [Google Scholar] [CrossRef] [PubMed]
- Storz, G.; Wolf, Y.I.; Ramamurthi, K.S. Small proteins can no longer be ignored. Annu. Rev. Biochem. 2014, 83, 753–777. [Google Scholar] [CrossRef] [PubMed]
- Delcourt, V.; Staskevicius, A.; Salzet, M.; Fournier, I.; Roucou, X. Small proteins encoded by unannotated ORFs are rising stars of the proteome, confirming shortcomings in genome annotations and current vision of an mRNA. Proteomics 2018, 18, e1700058. [Google Scholar] [CrossRef] [PubMed]
- Plaza, S.; Menschaert, G.; Payre, F. In Search of lost small peptides. Annu. Rev. Cell Dev. Biol. 2017, 33, 391–416. [Google Scholar] [CrossRef]
- Cabrera-Quio, L.E.; Herberg, S.; Pauli, A. Decoding sORF translation—From small proteins to gene regulation. RNA Biol. 2016, 13, 1051–1059. [Google Scholar] [CrossRef]
- Hao, Y.; Zhang, L.; Niu, Y.; Cai, T.; Luo, J.; He, S.; Zhang, B.; Zhang, D.; Qin, Y.; Yang, F.; et al. SmProt: A database of small proteins encoded by annotated coding and non-coding RNA loci. Brief. Bioinform. 2018, 19, 636–643. [Google Scholar] [CrossRef]
- Olexiouk, V.; van Criekinge, W.; Menschaert, G. An update on sORFs.org: A repository of small ORFs identified by ribosome profiling. Nucleic Acids Res. 2018, 46, D497–D502. [Google Scholar] [CrossRef]
- Mumtaz, M.A.S.; Couso, J.P. Ribosomal profiling adds new coding sequences to the proteome. Biochem. Soc. Trans. 2015, 43, 1271–1276. [Google Scholar] [CrossRef]
- Schrader, M. Origins, technological development, and applications of peptidomics. Methods Mol. Biol. 2018, 1719, 3–39. [Google Scholar] [CrossRef]
- Klein, C.; Aivaliotis, M.; Olsen, J.V.; Falb, M.; Besir, H.; Scheffer, B.; Bisle, B.; Tebbe, A.; Konstantinidis, K.; Siedler, F.; et al. The low molecular weight proteome of Halobacterium salinarum. J. Proteome Res. 2007, 6, 1510–1518. [Google Scholar] [CrossRef] [PubMed]
- Maret, W. Zinc biochemistry: From a single zinc enzyme to a key element of life. Adv. Nutr. 2013, 4, 82–91. [Google Scholar] [CrossRef]
- Cassandri, M.; Smirnov, A.; Novelli, F.; Pitolli, C.; Agostini, M.; Malewicz, M.; Melino, G.; Raschellà, G. Zinc-finger proteins in health and disease. Cell Death Discov. 2017, 3, 17071. [Google Scholar] [CrossRef] [PubMed]
- Eom, K.S.; Cheong, J.S.; Lee, S.J. Structural analyses of zinc finger domains for specific interactions with DNA. J. Microbiol. Biotechnol. 2016, 26, 2019–2029. [Google Scholar] [CrossRef] [PubMed]
- Matthews, J.M.; Sunde, M. Zinc fingers—Folds for many occasions. IUBMB Life 2002, 54, 351–355. [Google Scholar] [CrossRef] [PubMed]
- Krishna, S.S.; Majumdar, I.; Grishin, N.V. Structural classification of zinc fingers: Survey and summary. Nucleic Acids Res. 2003, 31, 532–550. [Google Scholar] [CrossRef]
- Chou, A.Y.; Archdeacon, J.; Kado, C.I. Agrobacterium transcriptional regulator Ros is a prokaryotic zinc finger protein that regulates the plant oncogene IPT. Proc. Natl. Acad. Sci. USA 1998, 95, 5293–5298. [Google Scholar] [CrossRef] [PubMed]
- Bouhouche, N.; Syvanen, M.; Kado, C.I. The origin of prokaryotic C2H2 zinc finger regulators. Trends Microbiol. 2000, 8, 77–81. [Google Scholar] [CrossRef]
- Malgieri, G.; Palmieri, M.; Russo, L.; Fattorusso, R.; Pedone, P.V.; Isernia, C. The prokaryotic zinc-finger: Structure, function and comparison with the eukaryotic counterpart. FEBS J. 2015, 282, 4480–4496. [Google Scholar] [CrossRef] [PubMed]
- Pereira, L.E.; Tsang, J.; Mrázek, J.; Hoover, T.R. The zinc-ribbon domain of Helicobacter pylori HP0958: Requirement for RpoN accumulation and possible roles of homologs in other bacteria. Microb. Inform. Exp. 2011, 1, 1–10. [Google Scholar] [CrossRef]
- Weidenbach, K.; Ehlers, C.; Schmitz, R.A. The transcriptional activator NrpA is crucial for inducing nitrogen fixation in Methanosarcina mazei Gö1 under nitrogen-limited conditions. FEBS J. 2014, 281, 3507–3522. [Google Scholar] [CrossRef]
- Tarasov, V.Y.; Besir, H.; Schwaiger, R.; Klee, K.; Furtwängler, K.; Pfeiffer, F.; Oesterhelt, D. A small protein from the bop-brp intergenic region of Halobacterium salinarum contains a zinc finger motif and regulates bop and crtB1 transcription. Mol. Microbiol. 2008, 67, 772–780. [Google Scholar] [CrossRef] [PubMed]
- Soppa, J. From genomes to function: Haloarchaea as model organisms. Microbiology 2006, 152, 585–590. [Google Scholar] [CrossRef] [PubMed]
- Pohlschroder, M.; Schulze, S. Haloferax volcanii. Trends Microbiol. 2019, 27, 86–87. [Google Scholar] [CrossRef]
- Pfeiffer, F.; Oesterhelt, D. A manual curation strategy to improve genome annotation: Application to a set of haloarchael genomes. Life 2015, 5, 1427–1444. [Google Scholar] [CrossRef]
- Allers, T.; Ngo, H.-P.; Mevarech, M.; Lloyd, R.G. Development of additional selectable markers for the halophilic archaeon Haloferax volcanii based on the leuB and trpA genes. Appl. Environ. Microbiol. 2004, 70, 943–953. [Google Scholar] [CrossRef] [PubMed]
- Dambeck, M.; Soppa, J. Characterization of a Haloferax volcanii member of the enolase superfamily: Deletion mutant construction, expression analysis, and transcriptome comparison. Arch. Microbiol. 2008, 190, 341–353. [Google Scholar] [CrossRef]
- Green, M.R.; Sambrook, K. Molecular Cloning: A Laboratory Manual; Cold Spring Harbor Laboratory Press: Cold Spring Harbor, NY, USA, 2012. [Google Scholar]
- Jaschinski, K.; Babski, J.; Lehr, M.; Burmester, A.; Benz, J.; Heyer, R.; Dörr, M.; Marchfelder, A.; Soppa, J. Generation and phenotyping of a collection of sRNA gene deletion mutants of the haloarchaeon Haloferax volcanii. PLoS ONE 2014, 9, e90763. [Google Scholar] [CrossRef] [PubMed]
- Hammelmann, M.; Soppa, J. Optimized generation of vectors for the construction of Haloferax volcanii deletion mutants. J. Microbiol. Methods 2008, 75, 201–204. [Google Scholar] [CrossRef]
- Breuert, S.; Allers, T.; Spohn, G.; Soppa, J. Regulated polyploidy in halophilic archaea. PLoS ONE 2006, 1, e92. [Google Scholar] [CrossRef]
- Jantzer, K.; Zerulla, K.; Soppa, J. Phenotyping in the archaea: Optimization of growth parameters and analysis of mutants of Haloferax volcanii. FEMS Microbiol. Lett. 2011, 322, 123–130. [Google Scholar] [CrossRef] [PubMed]
- Zerulla, K.; Chimileski, S.; Näther, D.; Gophna, U.; Papke, R.T.; Soppa, J. DNA as a phosphate storage polymer and the alternative advantages of polyploidy for growth or survival. PLoS ONE 2014, 9, e94819. [Google Scholar] [CrossRef]
- Kamekura, M.; Oesterhelt, D.; Wallace, R.; Anderson, P.; Kushner, D.J. Lysis of halobacteria in bacto-peptone by bile acids. Appl. Environ. Microbiol. 1988, 54, 990–995. [Google Scholar] [PubMed]
- Elevi Bardavid, R.; Oren, A. Sensitivity of Haloquadratum and Salinibacter to antibiotics and other inhibitors: Implications for the assessment of the contribution of Archaea and Bacteria to heterotrophic activities in hypersaline environments. FEMS Microbiol. Ecol. 2008, 63, 309–315. [Google Scholar] [CrossRef]
- Kumar, V.; Saxena, J.; Tiwari, S.K. Description of a halocin-producing Haloferax larsenii HA1 isolated from Pachpadra salt lake in Rajasthan. Arch. Microbiol. 2016, 198, 181–192. [Google Scholar] [CrossRef]
- Legerme, G.; Yang, E.; Esquivel, R.N.; Kiljunen, S.; Savilahti, H.; Pohlschroder, M. Screening of a Haloferax volcanii transposon library reveals novel motility and adhesion mutants. Life 2016, 6, 41. [Google Scholar] [CrossRef] [PubMed]
- Pfeiffer, F.; Broicher, A.; Gillich, T.; Klee, K.; Mejía, J.; Rampp, M.; Oesterhelt, D. Genome information management and integrated data analysis with HaloLex. Arch. Microbiol. 2008, 190, 281–299. [Google Scholar] [CrossRef] [PubMed]
- Freese, N.H.; Norris, D.C.; Loraine, A.E. Integrated genome browser: Visual analytics platform for genomics. Bioinformatics 2016, 32, 2089–2095. [Google Scholar] [CrossRef] [PubMed]
- Babski, J.; Haas, K.A.; Näther-Schindler, D.; Pfeiffer, F.; Förstner, K.U.; Hammelmann, M.; Hilker, R.; Becker, A.; Sharma, C.M.; Marchfelder, A.; et al. Genome-wide identification of transcriptional start sites in the haloarchaeon Haloferax volcanii based on differential RNA-Seq (dRNA-Seq). BMC Genom. 2016, 17, 629. [Google Scholar] [CrossRef] [PubMed]
- Laass, S.; Monzon, V.A.; Kliemt, J.; Hammelmann, M.; Pfeiffer, F.; Förstner, K.U.; Soppa, J. Characterization of the transcriptome of Haloferax volcanii, grown under four different conditions, with mixed RNA-Seq. PLoS ONE 2019, e0215986. [Google Scholar] [CrossRef]
- Bitan-Banin, G.; Ortenberg, R.; Mevarech, M. Development of a gene knockout system for the halophilic archaeon Haloferax volcanii by use of the pyrE gene. J. Bacteriol. 2003, 185, 772–778. [Google Scholar] [CrossRef]
- Sherwood, K.E.; Cano, D.J.; Maupin-Furlow, J.A. Glycerol-mediated repression of glucose metabolism and glycerol kinase as the sole route of glycerol catabolism in the haloarchaeon Haloferax volcanii. J. Bacteriol. 2009, 191, 4307–4315. [Google Scholar] [CrossRef] [PubMed]
- Fröls, S.; Dyall-Smith, M.; Pfeifer, F. Biofilm formation by haloarchaea. Environ. Microbiol. 2012, 14, 3159–3174. [Google Scholar] [CrossRef] [PubMed]
- Mullakhanbhai, M.F.; Larsen, H. Halobacterium volcanii spec. nov., a Dead Sea halobacterium with a moderate salt requirement. Arch. Microbiol. 1975, 104, 207–214. [Google Scholar] [CrossRef]
- Tripepi, M.; Imam, S.; Pohlschröder, M. Haloferax volcanii flagella are required for motility but are not involved in PibD-dependent surface adhesion. J. Bacteriol. 2010, 192, 3093–3102. [Google Scholar] [CrossRef]
- Kimura, M.; Kimura, J.; Davie, P.; Reinhardt, R.; Dijk, J. The amino acid sequence of a small DNA binding protein from the archaebacterium Sulfolobus solfataricus. FEBS Lett. 1984, 176, 176–178. [Google Scholar] [CrossRef]
- Prasse, D.; Thomsen, J.; de Santis, R.; Muntel, J.; Becher, D.; Schmitz, R.A. First description of small proteins encoded by spRNAs in Methanosarcina mazei strain Gö1. Biochimie 2015, 117, 138–148. [Google Scholar] [CrossRef]
- Humbard, M.A.; Miranda, H.V.; Lim, J.-M.; Krause, D.J.; Pritz, J.R.; Zhou, G.; Chen, S.; Wells, L.; Maupin-Furlow, J.A. Ubiquitin-like small archaeal modifier proteins (SAMPs) in Haloferax volcanii. Nature 2010, 463, 54–60. [Google Scholar] [CrossRef] [PubMed]
- Dantuluri, S.; Wu, Y.; Hepowit, N.L.; Chen, H.; Chen, S.; Maupin-Furlow, J.A. Proteome targets of ubiquitin-like samp1ylation are associated with sulfur metabolism and oxidative stress in Haloferax volcanii. Proteomics 2016, 16, 1100–1110. [Google Scholar] [CrossRef] [PubMed]
- Hepowit, N.L.; de Vera, I.M.S.; Cao, S.; Fu, X.; Wu, Y.; Uthandi, S.; Chavarria, N.E.; Englert, M.; Su, D.; Söll, D.; et al. Mechanistic insight into protein modification and sulfur mobilization activities of noncanonical E1 and associated ubiquitin-like proteins of Archaea. FEBS J. 2016, 283, 3567–3586. [Google Scholar] [CrossRef]
- Guttenplan, S.B.; Kearns, D.B. Regulation of flagellar motility during biofilm formation. FEMS Microbiol. Rev. 2013, 37, 849–871. [Google Scholar] [CrossRef]
- Verstraeten, N.; Braeken, K.; Debkumari, B.; Fauvart, M.; Fransaer, J.; Vermant, J.; Michiels, J. Living on a surface: Swarming and biofilm formation. Trends Microbiol. 2008, 16, 496–506. [Google Scholar] [CrossRef] [PubMed]
- Bak, G.; Lee, J.; Suk, S.; Kim, D.; Young Lee, J.; Kim, K.-S.; Choi, B.-S.; Lee, Y. Identification of novel sRNAs involved in biofilm formation, motility, and fimbriae formation in Escherichia coli. Sci. Rep. 2015, 5, 15287. [Google Scholar] [CrossRef]
- Esquivel, R.N.; Schulze, S.; Xu, R.; Hippler, M.; Pohlschroder, M. Identification of Haloferax volcanii Pilin N-glycans with diverse roles in pilus biosynthesis, adhesion, and microcolony formation. J. Biol. Chem. 2016, 291, 10602–10614. [Google Scholar] [CrossRef] [PubMed]
- Pohlschroder, M.; Esquivel, R.N. Archaeal type IV pili and their involvement in biofilm formation. Front. Microbiol. 2015, 6, 190. [Google Scholar] [CrossRef] [PubMed]
- Chimileski, S.; Franklin, M.J.; Papke, R.T. Biofilms formed by the archaeon Haloferax volcanii exhibit cellular differentiation and social motility, and facilitate horizontal gene transfer. BMC Biol. 2014, 12, 65. [Google Scholar] [CrossRef] [PubMed]
| Length up to aa | All Proteins | C(P)XCG Motif Proteins | ||||
|---|---|---|---|---|---|---|
| No. | Annotated Function | (%) | No. | (%) | Annotated Function | |
| 40 | 27 | 0 | 0 | 3 | 11 | 0 |
| 50 | 80 | 3 | 4 | 17 | 21 | 0 |
| 60 | 167 | 10 | 6 | 36 | 22 | 0 |
| 70 | 282 | 24 | 9 | 43 | 15 | 0 |
| 80 | 373 | 30 | 8 | 56 | 15 | 0 |
| 90 | 468 | 49 | 10 | 61 | 13 | 0 |
| 100 | 575 | 72 | 13 | 69 | 12 | 0 |
| Deletion Mutant | Growth in Four Media *1 | Growth on Glycerol | Bile Acid *2 | Bio-Film | Swarming | Pattern | No. of Phenotypes |
|---|---|---|---|---|---|---|---|
| HVO_0197_A | - | - | (loss) | - | - | 1 | |
| HVO_0325 | - | - | - | - | - | 0 | |
| HVO_0416 | - | - | - | gain | - | 1 | |
| HVO_0649 | - | - | (loss) | - | loss | 2 | |
| HVO_0758 | - | loss | - | gain | loss | (Yes) | 3 |
| HVO_1118 | - | - | (loss) | - | - | 1 | |
| HVO_1359 | - | loss | (loss) | - | - | 2 | |
| HVO_1677 | - | - | - | - | - | 0 | |
| HVO_2142 | - | loss | (gain) | gain | loss | Yes | 4 |
| HVO_2400 | - | gain | - | - | - | 1 | |
| HVO_2523 | - | loss | (gain) | gain | loss | Yes | 4 |
| HVO_2805_A | - | - | - | - | - | 0 | |
| HVO_2901 | - | - | (gain) | gain | loss | (Yes) | 3 |
| HVO_A0089 | - | loss | - | - | - | 1 | |
| HVO_A0254_A | - | - | - | - | - | 0 | |
| HVO_A0556 | - | loss | (gain) | gain | loss | Yes | 4 |
| sum | 0 | 7 | 8 | 6 | 6 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nagel, C.; Machulla, A.; Zahn, S.; Soppa, J. Several One-Domain Zinc Finger µ-Proteins of Haloferax Volcanii Are Important for Stress Adaptation, Biofilm Formation, and Swarming. Genes 2019, 10, 361. https://doi.org/10.3390/genes10050361
Nagel C, Machulla A, Zahn S, Soppa J. Several One-Domain Zinc Finger µ-Proteins of Haloferax Volcanii Are Important for Stress Adaptation, Biofilm Formation, and Swarming. Genes. 2019; 10(5):361. https://doi.org/10.3390/genes10050361
Chicago/Turabian StyleNagel, Chantal, Anja Machulla, Sebastian Zahn, and Jörg Soppa. 2019. "Several One-Domain Zinc Finger µ-Proteins of Haloferax Volcanii Are Important for Stress Adaptation, Biofilm Formation, and Swarming" Genes 10, no. 5: 361. https://doi.org/10.3390/genes10050361
APA StyleNagel, C., Machulla, A., Zahn, S., & Soppa, J. (2019). Several One-Domain Zinc Finger µ-Proteins of Haloferax Volcanii Are Important for Stress Adaptation, Biofilm Formation, and Swarming. Genes, 10(5), 361. https://doi.org/10.3390/genes10050361





