Genomic Features, Comparative Genomic Analysis, and Antimicrobial Susceptibility Patterns of Chryseobacterium arthrosphaerae Strain ED882-96 Isolated in Taiwan
Abstract
1. Introduction
2. Materials and Methods
2.1. Strain ED882-96
2.2. Species Identification Using 16S Ribosomal RNA Gene Sequencing
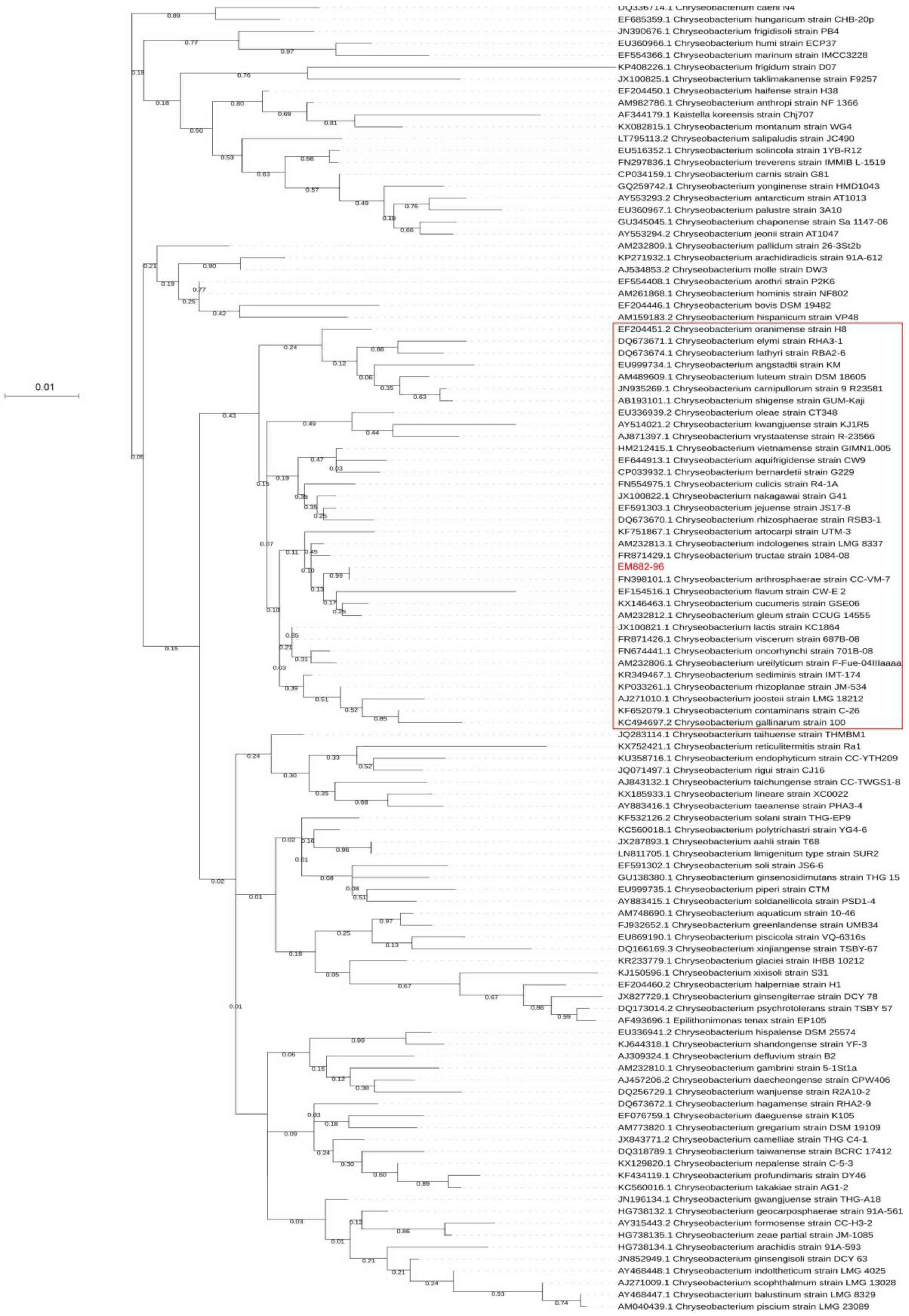
2.3. Phylogenetic Tree Based on 16S rRNA Gene Sequences
2.4. Whole-Genome Sequencing
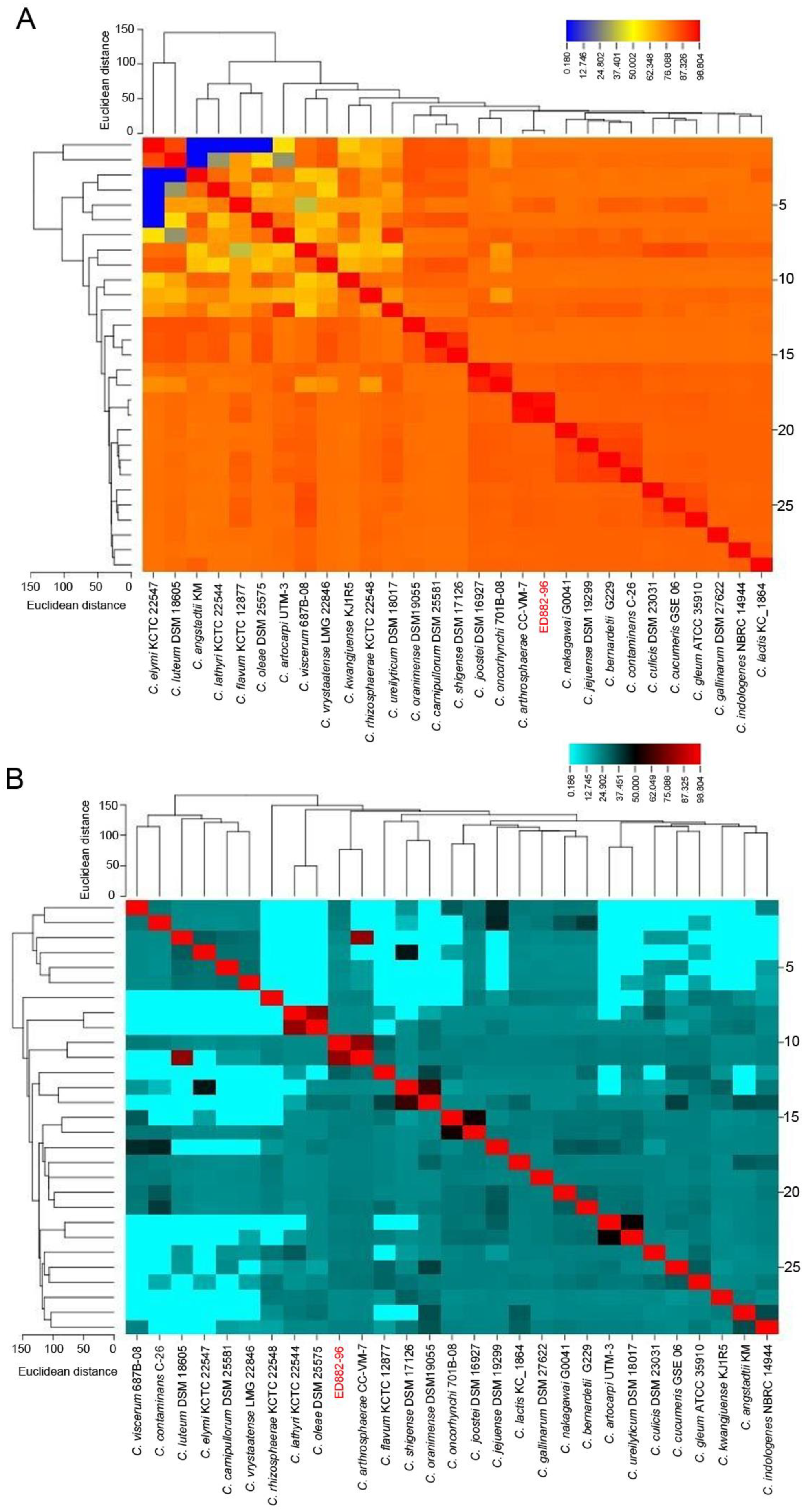
2.5. Analysis of Genome Similarity
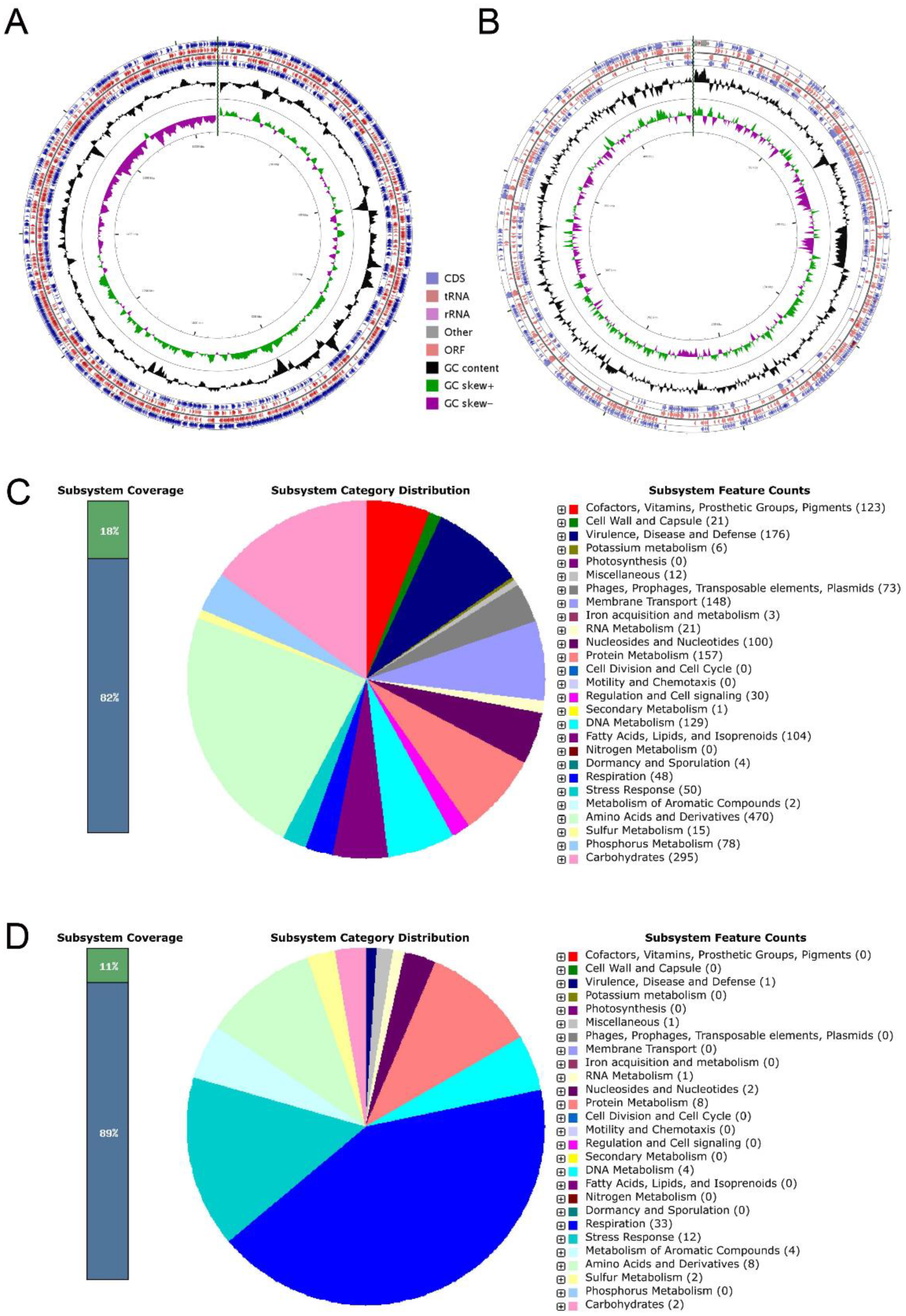
2.6. Genome Annotation and Function Analysis of Strain ED882-96
2.7. Antimicrobial Susceptibility and Antibiotic Resistance Genes
2.8. Type Strains of the Genus Chryseobacterium and Whole-Genome Sequences of C. arthrosphaerae
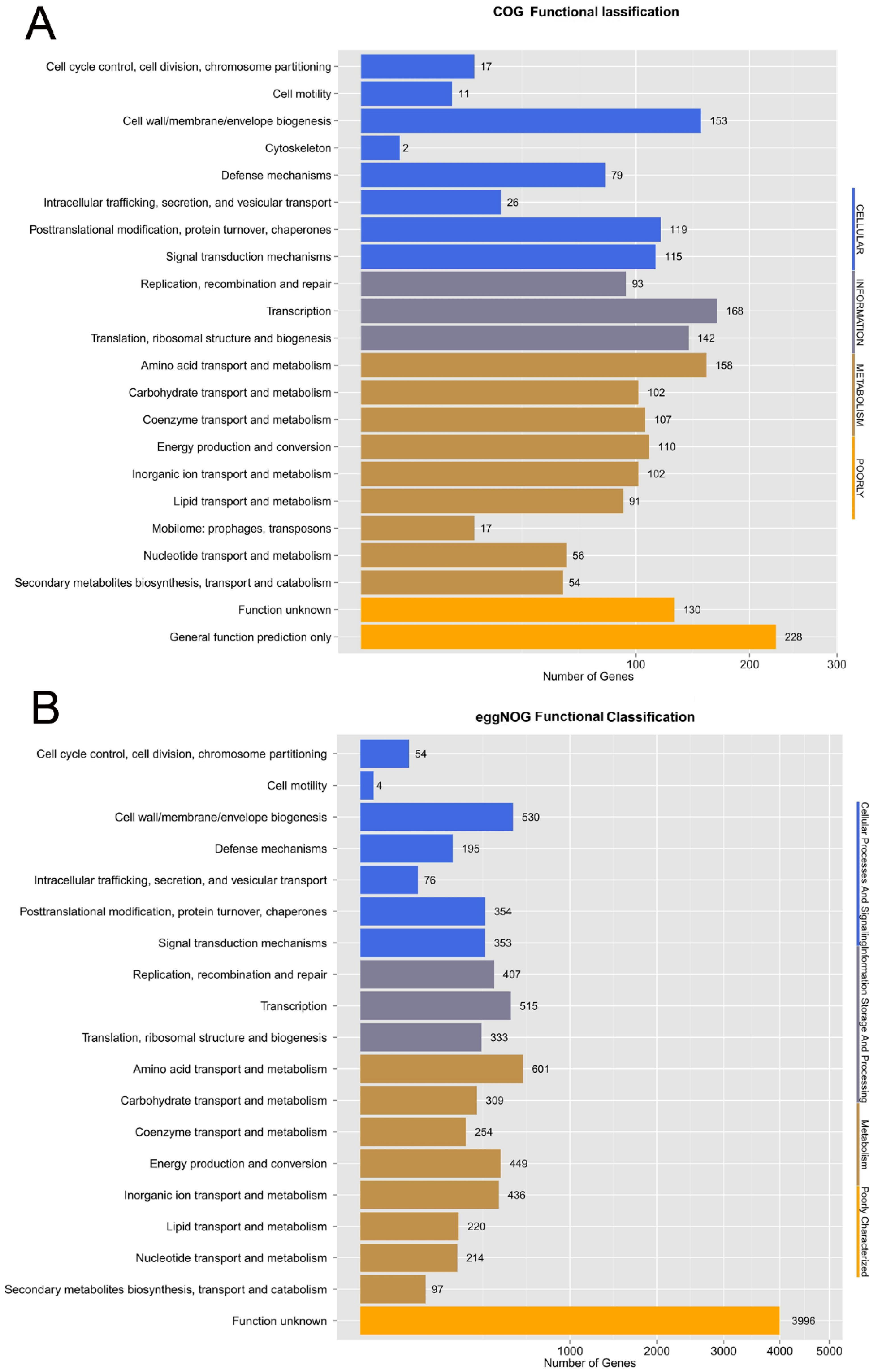
2.9. Comparative Analysis of COGs and KEGG
3. Results and Discussion
3.1. 16S rRNA-based Phylogenetic Relationship
3.2. Basic Data of Whole-Genome Sequencing of Strain ED882-96
3.3. Similarity of Whole Genomes
3.4. General Genomic Annotation
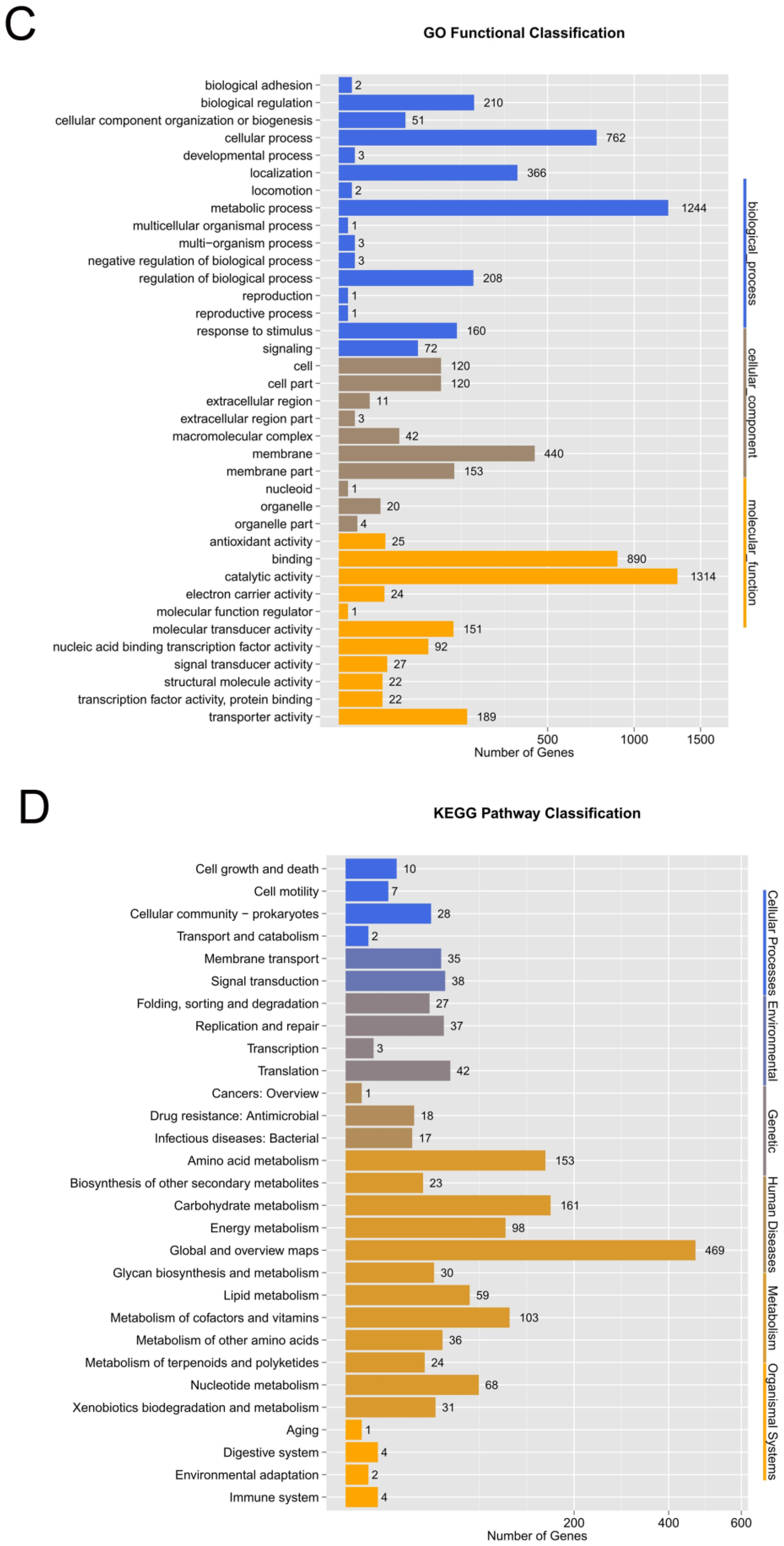
3.5. Orthologous Genes, GO, and KEGG of Strain ED882-96
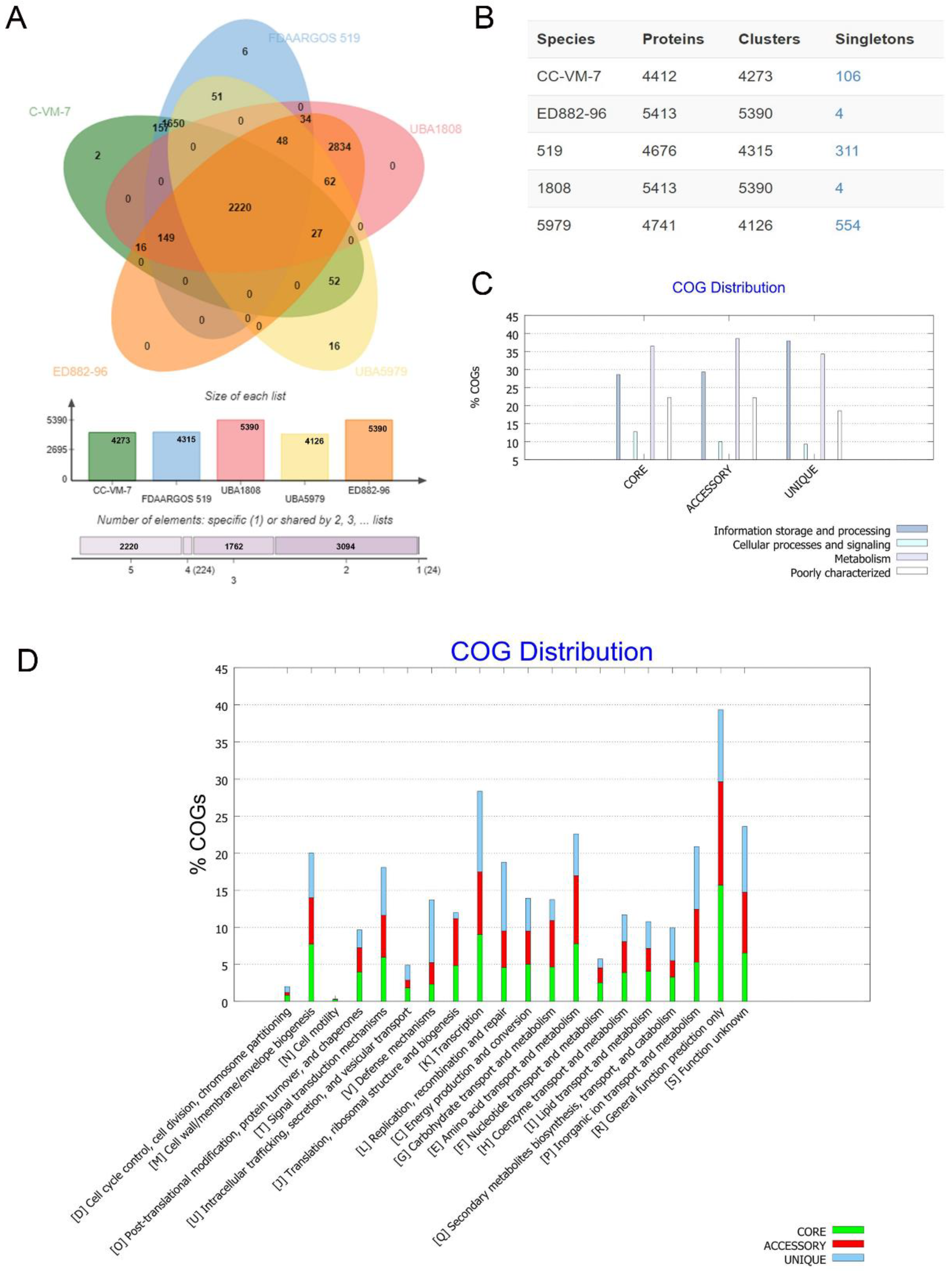
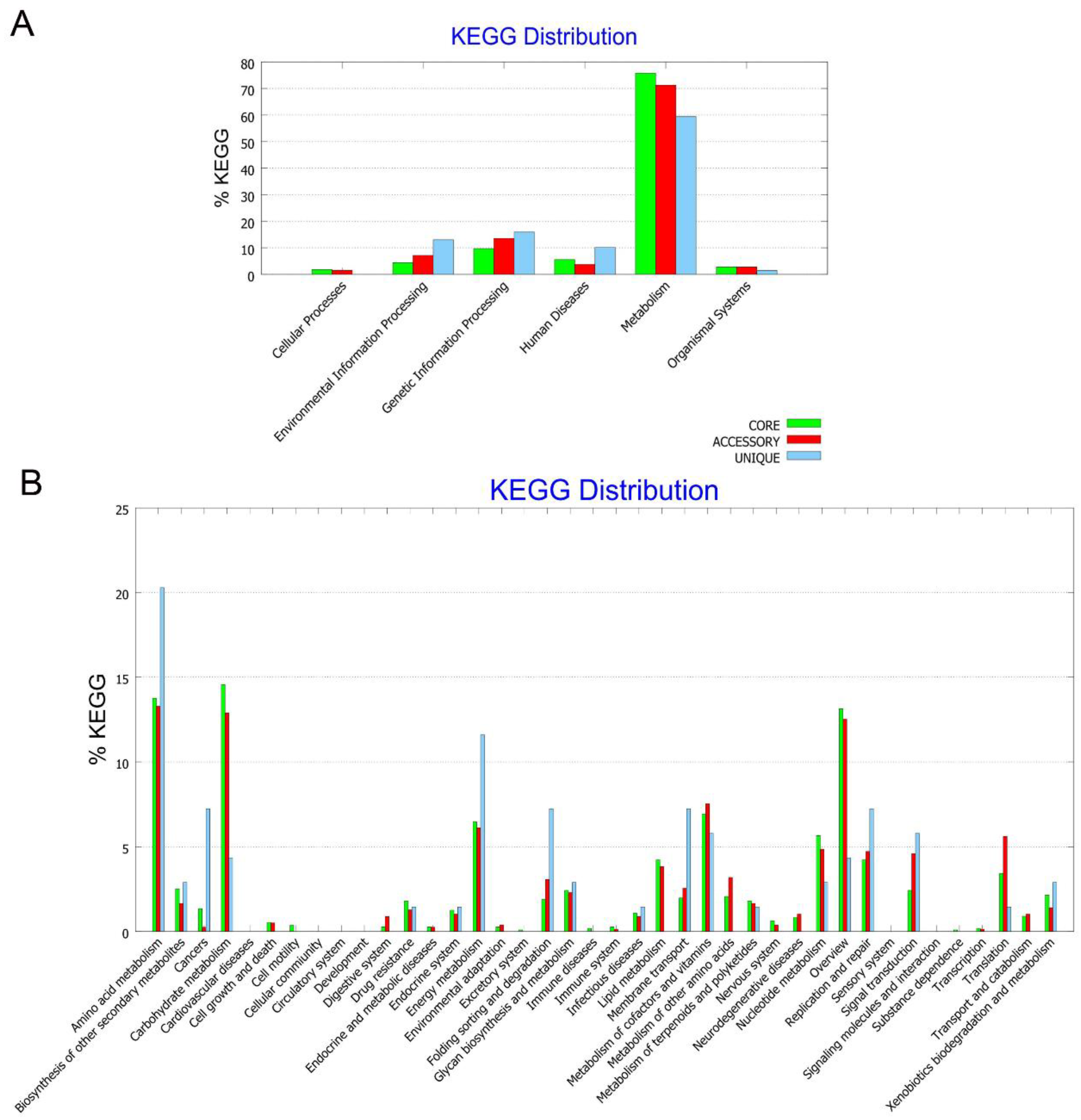
3.6. Comparative Genomic Analysis of COGs and KEGG in Five C. arthrosphaerae Strains
3.7. Virulence Factors
3.8. Antimicrobial Resistance and Associated Genes
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Vandamme, P.; Bernardet, J.F.; Segers, P.; Kersters, K.; Holmes, B. New perspectives in the classification of the Flavobacteria: Description of Chryseobacterium gen. nov., Bergeyella gen. nov., and Empedobacter nom. rev. Int. J. Syst. Evol. Microbiol. 1994, 44, 827–831. [Google Scholar] [CrossRef]
- Kämpfer, P.; Arun, A.B.; Young, C.C.; Chen, W.M.; Sridhar, K.R.; Rekha, P.D. Chryseobacterium arthrosphaerae sp. nov., isolated from the faeces of the pill millipede Arthrosphaera magna Attems. Int. J. Syst. Evol. Microbiol. 2010, 60, 1765–1769. [Google Scholar] [CrossRef]
- Jeong, J.J.; Lee, D.W.; Park, B.; Sang, M.K.; Choi, I.G.; Kim, K.D. Chryseobacterium cucumeris sp. nov., an endophyte isolated from cucumber (Cucumis sativus L.) root, and emended description of Chryseobacterium arthrosphaerae. Int. J. Syst. Evol. Microbiol. 2017, 67, 610–616. [Google Scholar] [CrossRef] [PubMed]
- Chryseobacterium. Available online: http://www.bacterio.net/chryseobacterium.html (accessed on 30 November 2017).
- Lin, J.N.; Lai, C.H.; Yang, C.H.; Huang, Y.H.; Lin, H.F.; Lin, H.H. Comparison of four automated microbiology systems with 16S rRNA gene sequencing for identification of Chryseobacterium and Elizabethkingia species. Sci. Rep. 2017, 7, 13824. [Google Scholar] [CrossRef] [PubMed]
- Lin, J.N.; Teng, S.H.; Lai, C.H.; Yang, C.H.; Huang, Y.H.; Lin, H.F.; Lin, H.H. Comparison of the Vitek MS and Bruker matrix-assisted laser desorption ionization-time of flight mass spectrometry systems for identification of Chryseobacterium isolated from clinical specimens and report of uncommon Chryseobacterium infections in humans. J. Clin. Microbiol. 2018, 56, e00712-18. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Stecher, G.; Tamura, K. MEGA7: Molecular evolutionary genetics analysis version 7.0 for bigger datasets. Mol. Biol. Evol. 2016, 33, 1870–1874. [Google Scholar] [CrossRef]
- Letunic, I.; Bork, P. Interactive Tree Of Life (iTOL) v4: Recent updates and new developments. Nucleic Acids Res. 2019. [Google Scholar] [CrossRef]
- Hurgobin, B. Short read alignment using SOAP2. Methods Mol. Biol. Clifton NJ 2016, 1374, 241–252. [Google Scholar]
- Chin, C.S.; Alexander, D.H.; Marks, P.; Klammer, A.A.; Drake, J.; Heiner, C.; Clum, A.; Copeland, A.; Huddleston, J.; Eichler, E.E.; et al. Non-hybrid, finished microbial genome assemblies from long-read SMRT sequencing data. Nat. Methods 2013, 10, 563–569. [Google Scholar] [CrossRef]
- Li, S.; Li, R.; Li, H.; Lu, J.; Li, Y.; Bolund, L.; Schierup, M.H.; Wang, J. SOAPindel: Efficient identification of indels from short paired reads. Genome Res. 2013, 23, 195–200. [Google Scholar] [CrossRef]
- Lee, I.; Kim, Y.O.; Park, S.C.; Chun, J. OrthoANI: An improved algorithm and software for calculating average nucleotide identity. Int. J. Syst. Evol. Microbiol. 2015, 66, 1100–1103. [Google Scholar] [CrossRef] [PubMed]
- Richter, M.; Rosselló-Móra, R. Shifting the genomic gold standard for the prokaryotic species definition. Proc. Natl. Acad. Sci. USA 2009, 106, 19126–19131. [Google Scholar] [CrossRef] [PubMed]
- Meier-Kolthoff, J.P.; Auch, A.F.; Klenk, H.P.; Göker, M. Genome sequence-based species delimitation with confidence intervals and improved distance functions. BMC Bioinform. 2013, 14, 60. [Google Scholar] [CrossRef]
- Tatusova, T.; DiCuccio, M.; Badretdin, A.; Chetvernin, V.; Nawrocki, E.P.; Zaslavsky, L.; Lomsadze, A.; Pruitt, K.D.; Borodovsky, M.; Ostell, J. NCBI prokaryotic genome annotation pipeline. Nucleic Acids Res. 2016, 44, 6614–6624. [Google Scholar] [CrossRef]
- Aziz, R.K.; Bartels, D.; Best, A.A.; DeJongh, M.; Disz, T.; Edwards, R.A.; Formsma, K.; Gerdes, S.; Glass, E.M.; Kubal, M.; et al. The RAST Server: Rapid annotations using subsystems technology. BMC Genom. 2008, 9, 75. [Google Scholar] [CrossRef] [PubMed]
- Overbeek, R.; Olson, R.; Pusch, G.D.; Olsen, G.J.; Davis, J.J.; Disz, T.; Edwards, R.A.; Gerdes, S.; Parrello, B.; Shukla, M.; et al. The SEED and the rapid annotation of microbial genomes using subsystems technology (RAST). Nucleic Acids Res. 2014, 42, D20–D214. [Google Scholar] [CrossRef]
- Galperin, M.Y.; Makarova, K.S.; Wolf, Y.I.; Koonin, E.V. Expanded microbial genome coverage and improved protein family annotation in the COG database. Nucleic Acids Res. 2015, 43, D261–D269. [Google Scholar] [CrossRef]
- Huerta-Cepas, J.; Szklarczyk, D.; Forslund, K.; Cook, H.; Heller, D.; Walter, M.C.; Rattei, T.; Mende, D.R.; Sunagawa, S.; Kuhn, M.; et al. eggNOG 4.5: A hierarchical orthology framework with improved functional annotations for eukaryotic, prokaryotic and viral sequences. Nucleic Acids Res. 2016, 44, D286–D293. [Google Scholar] [CrossRef]
- Gene Ontology Consortium. The gene ontology project in 2008. Nucleic Acids Res. 2008, 36, D440–D444. [Google Scholar] [CrossRef]
- Jones, P.; Binns, D.; Chang, H.-Y.; Fraser, M.; Li, W.; McAnulla, C.; McWilliam, H.; Maslen, J.; Mitchell, A.; Nuka, G.; et al. InterProScan 5: Genome-scale protein function classification. Bioinformatics 2014, 30, 1236–1240. [Google Scholar] [CrossRef]
- Kanehisa, M.; Sato, Y.; Furumichi, M.; Morishima, K.; Tanabe, M. New approach for understanding genome variations in KEGG. Nucleic Acids Res. 2018, 47, D590–D595. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Zheng, D.; Liu, B.; Yang, J.; Jin, Q. VFDB 2016: Hierarchical and refined dataset for big data analysis—10 years on. Nucleic Acids Res. 2016, 44, D694–D697. [Google Scholar] [CrossRef]
- Grant, J.R.; Stothard, P. The CGView Server: A comparative genomics tool for circular genomes. Nucleic Acids Res. 2008, 36, W181–W184. [Google Scholar] [CrossRef] [PubMed]
- Clinical and Laboratory Standards Institute. Performance Standards for Antimicrobial Susceptibility Testing; CLSI: Wayne, PA, USA, 2017. [Google Scholar]
- Kelesidis, T.; Karageorgopoulos, D.E.; Kelesidis, I.; Falagas, M.E. Tigecycline for the treatment of multidrug-resistant Enterobacteriaceae: A systematic review of the evidence from microbiological and clinical studies. J. Antimicrob. Chemother. 2008, 62, 895–904. [Google Scholar] [CrossRef]
- Lin, J.N.; Lai, C.H.; Yang, C.H.; Huang, Y.H. Differences in clinical manifestations, antimicrobial susceptibility patterns and mutations of fluoroquinolone target genes between Chryseobacterium gleum and Chryseobacterium indologenes. Antimicrob. Agents Chemother. 2019, 18, AAC-02256. [Google Scholar] [CrossRef]
- Liu, B.; Pop, M. ARDB—antibiotic resistance genes database. Nucleic Acids Res. 2009, 37, D443–D447. [Google Scholar] [CrossRef]
- Jia, B.; Raphenya, A.R.; Alcock, B.; Waglechner, N.; Guo, P.; Tsang, K.K.; Lago, B.A.; Dave, B.M.; Pereira, S.; Sharma, A.N.; et al. CARD 2017: Expansion and model-centric curation of the comprehensive antibiotic resistance database. Nucleic Acids Res. 2017, 45, D566–D573. [Google Scholar] [CrossRef]
- Wang, Y.; Coleman-Derr, D.; Chen, G.; Gu, Y.Q. OrthoVenn: A web server for genome wide comparison and annotation of orthologous clusters across multiple species. Nucleic Acids Res. 2015, 43, W78–W84. [Google Scholar] [CrossRef]
- Chaudhari, N.M.; Gupta, V.K.; Dutta, C. BPGA—An ultra-fast pan-genome analysis pipeline. Sci. Rep. 2016, 6, 24373. [Google Scholar] [CrossRef]
- How, K.Y.; Hong, K.W.; Sam, C.K.; Koh, C.L.; Yin, W.F.; Chan, K.G. Unravelling the genome of long chain N-acylhomoserine lactone-producing Acinetobacter sp. strain GG2 and identification of its quorum sensing synthase gene. Front. Microbiol. 2015, 6, 240. [Google Scholar] [CrossRef] [PubMed]
- Tatusov, R.L.; Fedorova, N.D.; Jackson, J.D.; Jacobs, A.R.; Kiryutin, B.; Koonin, E.V.; Krylov, D.M.; Mazumder, R.; Mekhedov, S.L.; Nikolskaya, A.N.; et al. The COG database: An updated version includes eukaryotes. BMC Bioinform. 2003, 4, 41. [Google Scholar] [CrossRef] [PubMed]
- Hsueh, P.R.; Teng, L.J.; Yang, P.C.; Ho, S.W.; Hsieh, W.C.; Luh, K.T. Increasing incidence of nosocomial Chryseobacterium indologenes infections in Taiwan. Eur. J. Clin. Microbiol. 1997, 16, 568–574. [Google Scholar] [CrossRef]
- Kirby, J.T.; Sader, H.S.; Walsh, T.R.; Jones, R.N. Antimicrobial susceptibility and epidemiology of a worldwide collection of Chryseobacterium spp: Report from the SENTRY antimicrobial surveillance program (1997–2001). J. Clin. Microbiol. 2004, 42, 445–448. [Google Scholar] [CrossRef] [PubMed]
- Chen, F.L.; Wang, G.C.; Teng, S.O.; Ou, T.Y.; Yu, F.L.; Lee, W.S. Clinical and epidemiological features of Chryseobacterium indologenes infections: Analysis of 215 cases. J. Microbiol. Immunol. Infect. 2013, 46, 425–432. [Google Scholar] [CrossRef] [PubMed]
- Jacoby, G.A. Mechanisms of resistance to quinolones. Clin. Infect. Dis. 2005, 41, S120–S126. [Google Scholar] [CrossRef] [PubMed]
| Assembly and annotation | Chromosome | Plasmid |
|---|---|---|
| Total sequence length (bp) | 4,249,864 | 435,667 |
| Number of contigs | 2 | 1 |
| Contig N50 (bp) | 2,386,100 | 435,667 |
| Contig L50 | 1 | 1 |
| GC content (%) | 38.3 | 38.1 |
| Number of subsystems | 191 | 29 |
| Number of coding sequences | 5413 | 512 |
| Number of rRNA | 6 | 3 |
| Number of tRNA | 37 | 22 |
| Antibiotic Group | Antibiotics | MIC | Interpretation |
|---|---|---|---|
| β-lactams, β-lactam/β-lactamase inhibitors | Piperacillin | >64 | R |
| Piperacillin-tazobactam | >128/4 | R | |
| Ticarcillin-clavulanic acid | >64/2 | R | |
| Ceftazidime | >16 | R | |
| Cefepime | >32 | R | |
| Ceftriaxone | >32 | R | |
| Aztreonam | >16 | R | |
| Imipenem | >8 | R | |
| Meropenem | >8 | R | |
| Aminoglycosides | Gentamicin | >8 | R |
| Tobramycin | >8 | R | |
| Amikacin | >32 | R | |
| Tetracyclines/glycylcycline | Tetracycline | >8 | R |
| Minocycline | >8 | R | |
| Tigecycline | >8 | R | |
| Fluoroquinolones | Ciprofloxacin | >2 | R |
| Levofloxacin | >8 | R | |
| Folate pathway inhibitors | Trimethoprim-sulfamethoxazole | >4/76 | R |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liang, C.-Y.; Yang, C.-H.; Lai, C.-H.; Huang, Y.-H.; Lin, J.-N. Genomic Features, Comparative Genomic Analysis, and Antimicrobial Susceptibility Patterns of Chryseobacterium arthrosphaerae Strain ED882-96 Isolated in Taiwan. Genes 2019, 10, 309. https://doi.org/10.3390/genes10040309
Liang C-Y, Yang C-H, Lai C-H, Huang Y-H, Lin J-N. Genomic Features, Comparative Genomic Analysis, and Antimicrobial Susceptibility Patterns of Chryseobacterium arthrosphaerae Strain ED882-96 Isolated in Taiwan. Genes. 2019; 10(4):309. https://doi.org/10.3390/genes10040309
Chicago/Turabian StyleLiang, Chih-Yu, Chih-Hui Yang, Chung-Hsu Lai, Yi-Han Huang, and Jiun-Nong Lin. 2019. "Genomic Features, Comparative Genomic Analysis, and Antimicrobial Susceptibility Patterns of Chryseobacterium arthrosphaerae Strain ED882-96 Isolated in Taiwan" Genes 10, no. 4: 309. https://doi.org/10.3390/genes10040309
APA StyleLiang, C.-Y., Yang, C.-H., Lai, C.-H., Huang, Y.-H., & Lin, J.-N. (2019). Genomic Features, Comparative Genomic Analysis, and Antimicrobial Susceptibility Patterns of Chryseobacterium arthrosphaerae Strain ED882-96 Isolated in Taiwan. Genes, 10(4), 309. https://doi.org/10.3390/genes10040309





