Shedding Light on a Secretive Tertiary Urodelean Relict: Hynobiid Salamanders (Paradactylodon persicus s.l.) from Iran, Illuminated by Phylogeographic, Developmental, and Transcriptomic Data
Abstract
1. Introduction
2. Materials and Methods
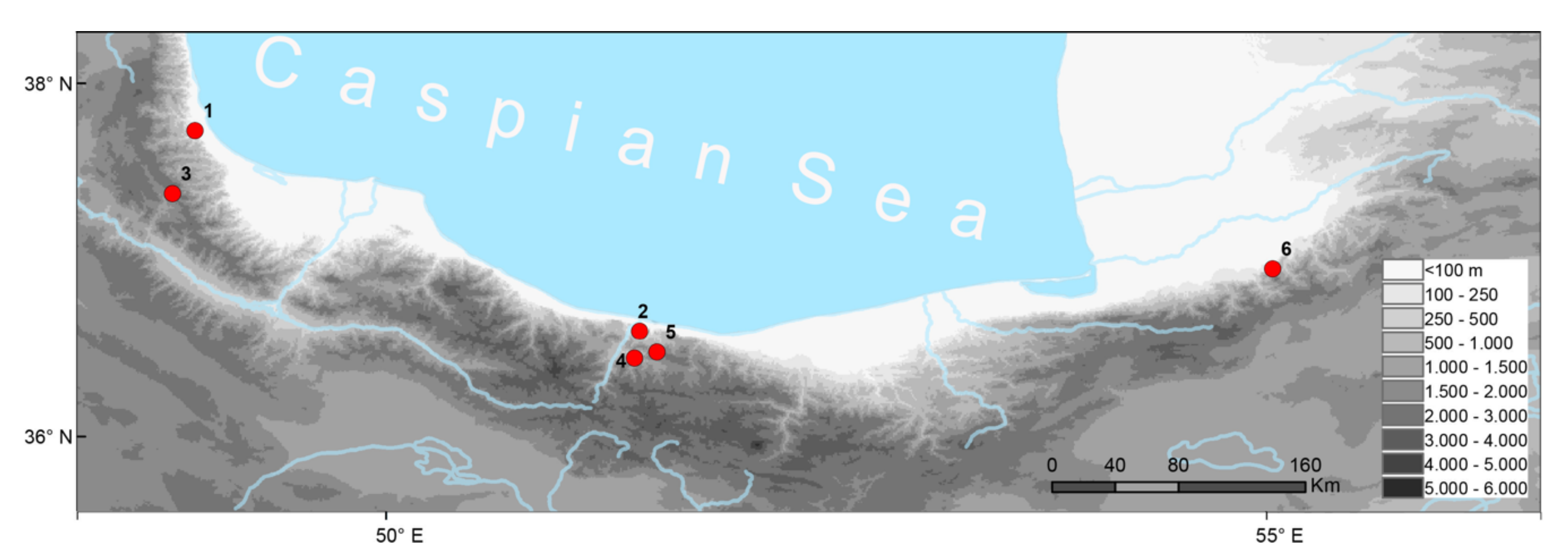
2.1. Animals and Samples
2.2. Gonadal Gross Morphology and Histology
2.3. DNA Extraction of Topotypic Samples
2.4. Primers Developed for PCR-Amplification of mtDNA-Fragments from Archival Samples
2.5. Sequencing and Reconstruction of a Complete mtDNA-Genome of Topotypic Paradactylodon persicus
2.6. Phylogenetic Analyses
2.7. Sequencing of Two Transcriptomes, Functional Annotation, and Classification
2.8. Genes Involved in Sex Determination or Sex Differentiation
3. Results
3.1. Transcriptomes
3.2. Genes Involved in Sex Determination or Sex Differentiation
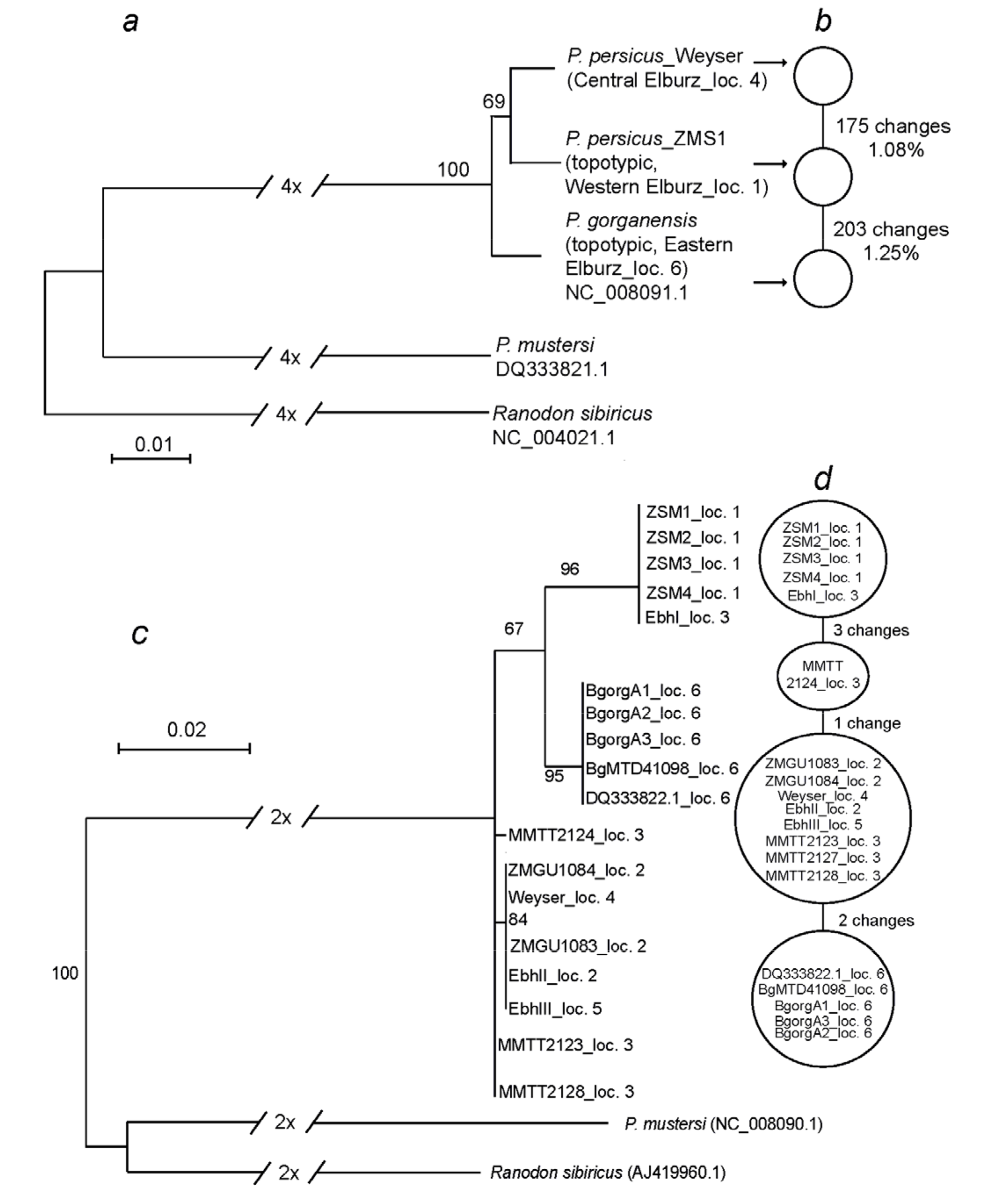
3.3. Mitochondrial Genomes
3.4. Phylogenetic Analyses
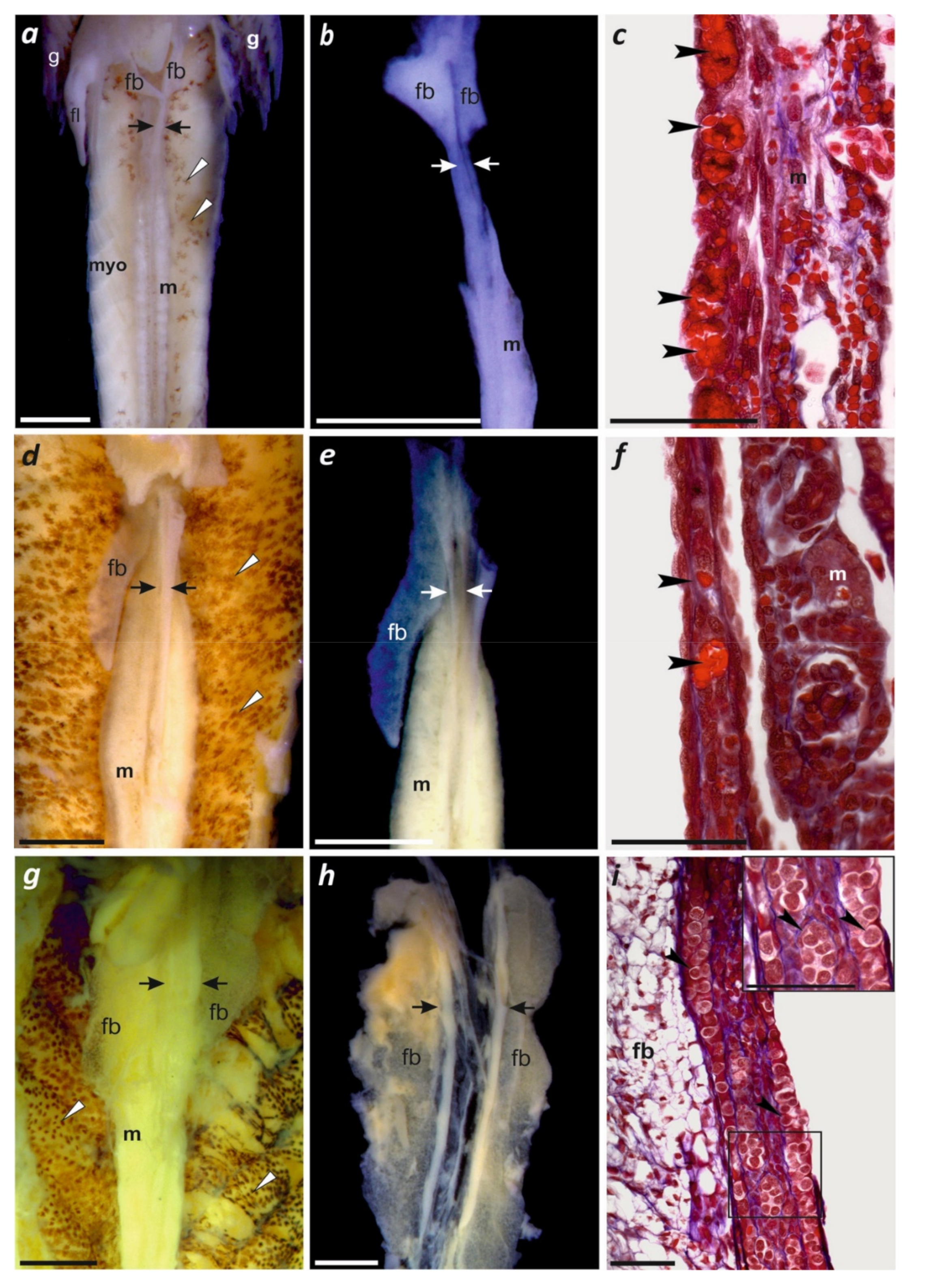
3.5. Larval Ontogeny with First Observations of Gonadal Development
4. Discussion
4.1. Transcriptomes of Embryo and Larva of Paradactylodon Among the First in Hynobiidae
4.2. Gonadal Differentiation in Paradactylodon and Other Hynobiid Salamanders
4.3. Phylogeography, Taxonomy, and Conservation
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Bobek, H. Die natürlichen Wälder und Gehölzfluren Irans. [The natural forests and shrubby terrains of Iran.]. Bonn. Geogr. Abhand. 1951, 8. [Google Scholar]
- Leestmans, R. Le refuge caspiens et son importance en biogéographie. Linn. Belg. 2005, 20, 97–102. [Google Scholar]
- Ramezani, E.; Marvie-Mohadjer, M.R.; Knapp, H.D.; Ahmadi, H.; Joosten, H. The late-Holocene vegetation history of the Central Caspian (Hyrcanian) forests of northern Iran. Holocene 2008, 18, 307–321. [Google Scholar] [CrossRef]
- Sosson, M.; Rolland, Y.; Müller, C.; Danelian, T.; Melkonyan, R.; Kekelia, S.; Mosar, J. Subductions, obduction and collision in the Lesser Caucasus (Armenia, Azerbaijan, Georgia), new insights. In Sedimentary Basin Tectonics from the Black Sea and Caucasus to the Arabian Platform; Sosson, M., Kaymakci, N., Stephanson, R., Bergarat, F., Storatchenoko, V., Eds.; Geological Society of London: London, UK, 2010; pp. 329–352. [Google Scholar]
- Maharramova, E.; Huseynova, I.; Kolbaia, S.; Gruenstaeudl, M.; Borsch, T.; Muller, L.A.H. Phylogeography and population genetics of the riparian relict tree Pterocarya fraxinifolia (Juglandaceae) in the South Caucasus. Syst. Biodiv. 2018, 16, 14–27. [Google Scholar] [CrossRef]
- Katouzian, A.R.; Sari, A.; Macher, J.N.; Weiss, M.; Saboori, A.; Leese, F.; Weigand, A.M. Drastic underestimation of amphipod biodiversity in the endangered Irano-Anatolian and Caucasus biodiversity hotspots. Sci. Rep. 2016, 6, 22507. [Google Scholar] [CrossRef][Green Version]
- Risch, J.-P. Breve diagnose de Paradactylodon, genre nouveau d’urodele de l’Iran (Amphibia, Caudata, Hynobiidae). Alytes 1984, 3, 44–46. [Google Scholar]
- Eiselt, J.; Steiner, H.M. Erstfund eines hynobiiden Molches in Iran. Ann. Nat. Hist. Mus. Vienna 1970, 74, 77–90. [Google Scholar]
- Schmidtler, J.J.; Schmidtler, J.F. Eine Salamander-Novität aus Persien, Batrachuperus persicus. Aquar. Mag. 1971, 5, 443–445. [Google Scholar]
- Clergue-Gazeau, M.; Farcy, J.P. Un Batrachuperus adulte dans une grotte d’Iran. Espèce nouvelle? Int. J. Speleol. 1978, 10, 185–193. [Google Scholar] [CrossRef]
- Clergue-Gazeau, M.; Thorn, R. Une nouvelle espèce de salamandre du genre Batrachuperus en provenence de l’Iran septentrional (Amphibia, Caudata, Hynobiidae). Bull. Soc. Hist. Nat. 1979, 114, 455–460. [Google Scholar]
- Stöck, M. On the biology and the taxonomic status of Batrachuperus gorganensis Clergue-Gazeau et Thorn, 1979 based on topotypic specimens (Amphibia: Caudata: Hynobiidae). Abh. Staatl. Mus. Tierkd. Dresd. 1999, 50, 217–241. [Google Scholar]
- Litvinchuk, S.N.; Borkin, L.J.; Rozanov, J.M. Intraspecific and interspecific genome size variation in hynobiid salamanders of Russia and Kazakhstan: Determination by flow cytometry. Asian Herpetol. Res. 2004, 10, 279–291. [Google Scholar]
- Liedtke, C.; Gower, D.J.; Wilkinson, M.; Gomez-Mestre, I. Macroevolutionary shift in the size of amphibian genomes and the role of life history and climate. Nat. Ecol. Evol. 2018, 2, 1792–1799. [Google Scholar] [CrossRef] [PubMed]
- Nowoshilow, S.; Schloissnig, S.; Ji-Feng, F.; Dahl, A.; Pang, A.W.C.; Pippel, M.; Winkler, S.; Hastie, A.R.; Young, G.; Roscito, J.G.; et al. The axolotl genome and the evolution of key tissue formation regulators. Nature 2018, 554, 50–55. [Google Scholar] [CrossRef]
- Ebrahimi, M.; Kami, H.G.; Stöck, M. First description of egg sacs and early larval development in Hynobiid Salamanders (Urodela, Hynobiidae, Batrachuperus) from north-eastern Iran. Asian Herpetol. Res. 2004, 10, 168–175. [Google Scholar]
- Paraskiv, K.P. Semirechensky triton (lyagushkozub) (Semirechensk newt), Izvestiya Akad. nauk KazSSR, Ser. Biol. News Acad. Sci. KazSSR. Biol. Series 1953, 8, 47–56. [Google Scholar]
- Brushko, Z.K.; Narbayeva, S.P. Razmnozhenie semirechenskogo lyagushkozuba v doline r. Borokhudzir (Yugo-Vostochnyu Kazakhstan) (Breeding of Siberian salamander in the Borokhudzir River valley (southeastern Kazakhstan)). Ecology 1988, 2, 45–48. [Google Scholar]
- Baloutch, M.; Kami, H.G. Amphibians of Iran; University of Tehran Press: Tehran, Iran, 1995; p. 177. [Google Scholar]
- Kami, H.G.; Vakilpoure, E. Geographic distribution of Batrachuperus persicus. Herpetol. Rev. 1996, 27, 147. [Google Scholar]
- Ahmadzadeh, F.; Kami, H.G. Distribution and conservation status of the Persian brook salamander, Batrachuperus (Paradactylodon) persicus (Amphibia: Caudata: Hynobiidae) in north-western Iran. Iran. J. Anim. Biosyst. 2009, 5, 9–15. [Google Scholar]
- Kami, H.G. Additional specimens of the Persian Mountain Salamander, Batrachuperus persicus, from Iran (Amphibia: Hynobiidae). Zool. Middle East 1999, 19, 37–42. [Google Scholar] [CrossRef]
- Kami, H.G. On the biology of Persian Mountain Salamander, Batrachuperus persicus (Amphibia, Caudata, Hynobiidae) in Golestan Province, Iran. Asian Herpetol. Res. 2004, 10, 182–190. [Google Scholar]
- Zivari, S.; Kami, H.G. Skeletochronological assessment of age in the Persian mountain salamander, Paradactylodon gorganensis (Clergue-Gazeau and Thorn, 1979) (Caudata: Hynobiidae) from Golestan Province, Iran. Caspian J. Environ. Sci. 2017, 15, 75–84. [Google Scholar]
- Zhang, P.; Chen, Y.Q.; Zhou, H.; Liu, Y.F.; Wang, X.L.; Papenfuss, T.J.; Wake, D.B.; Qu, L.H. Phylogeny, evolution, and biogeography of Asiatic salamanders (Hynobiidae). Proc. Natl. Acad. Sci. USA 2006, 103, 7360–7365. [Google Scholar] [CrossRef] [PubMed]
- Weissrock, D.W.; Macey, J.R.; Matsui, M.; Mulcahy, D.G.; Papenfuss, T.J. Molecular phylogenetic reconstruction of the endemic Asian salamander family Hynobiidae (Amphibia, Caudata). Zootaxa 2013, 3626, 77–93. [Google Scholar] [CrossRef]
- Chen, M.Y.; Mao, R.L.; Liang, D.; Kuro-o, M.; Zeng, X.M.; Zhang, P. A reinvestigation of phylogeny and divergence times of Hynobiidae (Amphibia, Caudata) based on 29 nuclear genes. Mol. Phyl. Evol. 2015, 83, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Iwasawa, H.; Kera, Y. Normal stages of development of Japanese lungless salamander, Onychodactylus japonicus (Houttuyn). Jap. J. Herpetol. 1980, 8, 73–89. [Google Scholar] [CrossRef]
- Iwasawa, H.; Yamashita, K. Normal stages of development of a hynobiid salamander Hynobius nigrescens Stejneger. Jap. J. Herpetol. 1991, 14, 39–62. [Google Scholar] [CrossRef][Green Version]
- Vassilieva, A.B.; Smirnov, S.V. Development and morphology of the dentition in the Asian salamander Ranodon sibiricus (Urodela: Hynobiidae). Russ. J. Herpetol. 2001, 8, 105–116. [Google Scholar]
- Ogielska, M.; Kotusz, A. Pattern and rate of ovary differentiation with reference to somatic development in anuran amphibians. J. Morphol. 2004, 259, 41–54. [Google Scholar] [CrossRef] [PubMed]
- Haczkiewicz, K.; Ogielska, M. Gonadal sex differentiation in frogs: How testes become shorter than ovaries. Zool. Sci. 2013, 30, 125–134. [Google Scholar] [CrossRef]
- Wilting, A.; Patel, R.; Pfestorf, H.; Kern, C.; Sultan, K.; Ario, A.; Peñaloza, F.; Kramer-Schadt, S.; Radchuk, V.; Förster, D.W.; Fickel, J. Evolutionary history and conservation significance of the Javan leopard Panthera pardus melas. J. Zool. 2016, 299, 239–250. [Google Scholar] [CrossRef]
- Fortes, G.G.; Paijmans, J.L.A. Analysis of whole mitogenomes from ancient samples. Methods Mol. Biol. 2015, 1347, 179–195. [Google Scholar]
- Martin, M. Cutadapt removes adapter sequences from high throughput sequencing reads. EMBnet 2011, 17, 10–12. [Google Scholar] [CrossRef]
- Bolger, A.M.; Lohse, M.; Usadel, B. Trimmomatic: A flexible trimmer for Illumina sequence data. Bioinformatics 2014, 30, 2114–2120. [Google Scholar] [CrossRef]
- Magoč, T.; Salzberg, S.L. FLASH: Fast length adjustment of short reads to improve genome assemblies. Bioinformatics 2011, 27, 2957–2963. [Google Scholar] [CrossRef]
- Li, H.; Durbin, R. Fast and accurate short read alignment with Burrows-Wheeler transform. Bioinformatics 2009, 25, 1754–1760. [Google Scholar] [CrossRef]
- Lefort, V.; Longueville, J.E.; Gascuel, O. SMS: Smart Model Selection in PhyML. Mol. Biol. Evol. 2017, 34, 2422–2424. [Google Scholar] [CrossRef]
- Guindon, S.; Dufayard, J.F.; Lefort, V.; Anisimova, M.; Hordijk, W.; Gascuel, O. New algorithms and methods to estimate maximum-likelihood phylogenies: Assessing the performance of PhyML 3.0. Syst. Biol. 2010, 59, 307–321. [Google Scholar] [CrossRef]
- Grabherr, M.G.; Haas, B.J.; Yassour, M.; Levin, J.Z.; Thompson, D.A.; Amit, I.; Adiconis, X.; Fan, L.; Raychowdhury, R.; Zeng, Q.; et al. Full-length transcriptome assembly from RNA-Seq data without a reference genome. Nat. Biotechnol. 2011, 15, 644–652. [Google Scholar] [CrossRef]
- Pertea, G.; Huang, X.; Liang, F.; Antonescu, V.; Sultana, R.; Karamycheva, S.; Lee, Y.; White, J.; Cheung, F.; Parvizi, B.; Tsai, J.; Quackenbush, J. TIGR Gene indices clustering tools (TGICL): A software system for fast clustering of large EST datasets. Bioinformatics 2003, 19, 651–652. [Google Scholar] [CrossRef] [PubMed]
- Altschul, S.F.; Madden, T.L.; Schäffer, A.A.; Zhang, J.; Zhang, Z.; Miller, W.; Lipman, D.J. Gapped BLAST and PSI- BLAST: A new generation of protein database search programs. Nucleic Acids Res. 1997, 25, 3389–3402. [Google Scholar] [CrossRef]
- Conesa, A.; Götz, S.; García-Gómez, J.M.; Terol, J.; Talón, M.; Robles, M. Blast2GO: A universal tool for annotation, visualization and analysis in functional genomics research. Bioinformatics 2005, 21, 3674–3676. [Google Scholar] [CrossRef]
- James-Zorn, C.; Ponferrada, V.G.; Burns, K.A.; Fortriede, J.D.; Lotay, V.S.; Liu, Y. Xenbase: Core features, data acquisition and data processing. Genesis 2015, 53, 486–497. [Google Scholar] [CrossRef] [PubMed]
- Kent, W.J. BLAT-the BLAST-like alignment tool. Genome Res. 2002, 12, 656–664. [Google Scholar] [CrossRef]
- Peng, Y.; Leung, H.C.; Yiu, S.M.; Chin, F.Y. IDBA-UD: A de novo assembler for single-cell and metagenomic sequencing data with highly uneven depth. Bioinformatics 2012, 28, 1420–1428. [Google Scholar] [CrossRef] [PubMed]
- Iseli, C.; Jongeneel, C.V.; Buche, P. ESTScan: A program for detecting, evaluating, and reconstructing potential coding regions in EST sequences. Proc. Int. Conf. Intell. Syst. Mol. Biol. 1999, 138–148. [Google Scholar]
- Che, R.; Sun, Y.; Wang, R.; Xu, T. Transcriptomic analysis of endangered Chinese salamander: identification of immune, sex and reproduction-related genes and genetic Markers. PLoS ONE 2014, 9, e87940. [Google Scholar] [CrossRef]
- Green, D.M.; Sessions, S.K. Amphibian Cytogenetics and Evolution. J. Evol. Biol. 1991, 6, 300–302. [Google Scholar]
- Sessions, S.K.; Bizjak Mali, L.; Green, D.M.; Trifonov, V.; Ferguson-Smith, M. Evidence for sex chromosome turnover in proteid salamanders. Cytogenet. Genome. Res 2016, 148, 305–313. [Google Scholar] [CrossRef] [PubMed]
- Keinath, M.C.; Timoshevskaya, N.; Timoshevskiy, V.A.; Voss, R.; Smith, J.J. Miniscule differences between sex chromosomes in the giant genome of a salamander. Sci. Rep. 2018, 8, 17882. [Google Scholar] [CrossRef]
- Sakata, N.; Tamori, Y.; Wakahara, M. P450 aromatase expression in the temperature-sensitive sexual differentiation of salamander (Hynobius retardatus) gonads. Int. J. Dev. Biol. 2005, 49, 417–425. [Google Scholar] [CrossRef] [PubMed]
- Bulakhova, N.A.; Berman, D.I. Reproductive System of the Schrenckii Salamander (Salamandrella schrenckii, Amphibia, Caudata, Hynobiidae) in spring and fall. Biol. Bull. 2013, 40, 664–677. [Google Scholar] [CrossRef]
- Bulakhova, N.A.; Berman, D.I. Male reproductive cycle of the Siberian salamander Salamandrella keyserlingii (Caudata: Hynobiidae) in coastal tundra of the Sea of Okhotsk. Polar Biol. 2014, 37, 123–133. [Google Scholar] [CrossRef]
- Bulakhova, N.A.; Berman, D.I. Reproductive Cycle of Females and Reproduction of the Siberian Salamander (Salamandrella keyserlingii, Caudata, Hynobiidae) on the Coast of the Sea of Okhotsk. Biol. Bull. 2017, 44, 688–699. [Google Scholar] [CrossRef]
- Yartsev, V.V.; Kuranova, V.N. Seasonal dynamics of male and female reproductive systems in the Siberian salamander, Salamandrella keyserlingii (Caudata, Hynobiidae). Asian Herpetol. Res. 2015, 6, 169–183. [Google Scholar]
- Yartsev, V.V.; Kuranova, V.N.; Martynova, G.S. The male urogenital system of the Siberian salamander Salamandrella keyserlingii (Caudata: Hynobiidae) with special reference to the microstructure of the testes and sperm transport complex. Russ. J. Herpetol. 2016, 23, 1–6. [Google Scholar]
- Ogielska, M. The undifferentiated amphibian gonad. In Reproduction of Amphibians; Ogielska, M., Ed.; Science Publishers: Enfield, NH, USA, 2009; pp. 1–33. [Google Scholar]
- Kanki, K.; Wakahara, M. The possible contribution of pituitary hormones to the heterochronic development of gonads and external morphology in overwintered larvae of Hynobius retardatus. Int. J. Dev. Biol. 2001, 45, 725–732. [Google Scholar]
- Moritz, C.; Schneider, C.J.; Wake, D.B. Evolutionary relationships within the Ensantina eschscholtzii complex confirm the ring species interpretation. Syst. Biol. 1992, 41, 273–291. [Google Scholar] [CrossRef]
- Pereira, R.J.; Monahan, W.B.; Wake, D.B. Predictors for reproductive isolation in a ring species complex following genetic and ecological divergence. BMC Evol. Biol. 2011, 11, 194. [Google Scholar] [CrossRef]
- Hendrix, R.; Hauswaldt, J.S.; Veith, M.; Steinfartz, S. Strong correlation between cross-amplification success and genetic distance across all members of ‘True Salamanders’ (Amphibia: Salamandridae) revealed by Salamandra salamandra-specific microsatellite loci. Mol. Ecol. Res. 2010, 10, 1038–1047. [Google Scholar] [CrossRef]
- Vences, V.; Sanchez, E.; Hauswaldt, S.; Eikelmann, D.; Rodríguez, A.; Carranza, S.; Donaire, D.; Gehara, M.; Helfer, V.; Lötters, S.; et al. Nuclear and mitochondrial multilocus phylogeny and survey of alkaloid content in true salamanders of the genus Salamandra (Salamandridae). Mol. Phylo. Evol. 2014, 73, 208–216. [Google Scholar] [CrossRef] [PubMed]
- Xia, Y.; Gu, H.-F.; Peng, R.; Chen, Q.; Zheng, Y.-C.; Murphy, R.; Zeng, X.-M. COI is better than 16S rRNA for DNA barcoding Asiatic salamanders (Amphibia: Caudata: Hynobiidae). Mol. Ecol. Res. 2012, 12, 48–56. [Google Scholar] [CrossRef] [PubMed]
- Fu, J.; Zeng, X. How many species are in the genus Batrachuperus? A phylogeographical analysis of the stream salamanders (family Hynobiidae) from southwestern China. Mol. Ecol. 2008, 17, 1469–1488. [Google Scholar] [CrossRef] [PubMed]
- Sparreboom, M. Salamanders of the Old World; KNNV Publishing: Zeist, The Netherlands, 2014. [Google Scholar]
- Rafaelli, J. Les Urodèles du Monde, 2nd ed.; Penclen Edition: London, UK, 2013; p. 472. [Google Scholar]
- Dubois, A.; Raffaëlli, J. A new ergotaxonomy of the order Urodela Duméril, 1805 (Amphibia, Batrachia). Alytes 2012, 28, 77–161. [Google Scholar]
- Frost, D.R. Amphibian Species of the World: An Online Reference.Version 6.0. American Museum of Natural History, New York, USA, April 2019. Available online: http://research.amnh.org/herpetology/amphibia/index.html (accessed on 19 April 2019).
- Akhani, H.; Djamali, M.; Ghorbanalizadeh, A.; Ramezani, E. Plant biodiversity of Hyrcanian relict forests in Iran: An overview of flora, vegetation, paleoecology and conservation. Pak. J. Bot. 2010, 42, 231–258. [Google Scholar]

| Developmental Stages in | |||||
|---|---|---|---|---|---|
| Paradactylodon persicus | Onychodactylus japonicus (Iwasawa & Kera 1980) [28] | Hynobius nigrescens (Iwasawa & Yamashita 1991) [29] | Ranodon sibiricus (Vassilieva & Smirnov 2001 [30] | ||
| Photographs Figure 3 | Days of Development | Samples Examined | |||
| a | 1 day after spawning embryos in the sac, cleavage | – | 3–4 | 3 | – |
| b | 12 days after spawning, embryos in the sac, gastrulation | – | 22 | 12B | – |
| c | 18 days after spawning, embryos in the sac, neural plates | – | 29 | 18 | – |
| e | single embryo, egg sac removed | Transcriptome 1 | 46 | 36–37 | – |
| f | larvae before hatching, external gills | – | 48 | 39 | – |
| g, h | larva at day 4 after hatching | Gonadal histology (Figure 4a–c) | 49 | 42–43 | II |
| i | 24 days | – | 55 | 53 | VI |
| k | 29 days | Gonadal histology(Figure 4d–f) | 58 | 55 | VII–IX |
| l | 38 days | Transcriptome 2 | 60 | 57 | XII |
| m | 41 days | Gonadal histology(Figure 4g–i) | 66 | 63 | XV |
| n | 58 days | – | 69 | 65 | – |
| o | 58 days | – | 69 | 65 | – |
| p | 60 days | – | 69 | 66 | – |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Stöck, M.; Fakharzadeh, F.; Kuhl, H.; Rozenblut-Kościsty, B.; Leinweber, S.; Patel, R.; Ebrahimi, M.; Voitel, S.; Schmidtler, J.F.; Kami, H.G.; et al. Shedding Light on a Secretive Tertiary Urodelean Relict: Hynobiid Salamanders (Paradactylodon persicus s.l.) from Iran, Illuminated by Phylogeographic, Developmental, and Transcriptomic Data. Genes 2019, 10, 306. https://doi.org/10.3390/genes10040306
Stöck M, Fakharzadeh F, Kuhl H, Rozenblut-Kościsty B, Leinweber S, Patel R, Ebrahimi M, Voitel S, Schmidtler JF, Kami HG, et al. Shedding Light on a Secretive Tertiary Urodelean Relict: Hynobiid Salamanders (Paradactylodon persicus s.l.) from Iran, Illuminated by Phylogeographic, Developmental, and Transcriptomic Data. Genes. 2019; 10(4):306. https://doi.org/10.3390/genes10040306
Chicago/Turabian StyleStöck, Matthias, Fatemeh Fakharzadeh, Heiner Kuhl, Beata Rozenblut-Kościsty, Sophie Leinweber, Riddhi Patel, Mehregan Ebrahimi, Sebastian Voitel, Josef Friedrich Schmidtler, Haji Gholi Kami, and et al. 2019. "Shedding Light on a Secretive Tertiary Urodelean Relict: Hynobiid Salamanders (Paradactylodon persicus s.l.) from Iran, Illuminated by Phylogeographic, Developmental, and Transcriptomic Data" Genes 10, no. 4: 306. https://doi.org/10.3390/genes10040306
APA StyleStöck, M., Fakharzadeh, F., Kuhl, H., Rozenblut-Kościsty, B., Leinweber, S., Patel, R., Ebrahimi, M., Voitel, S., Schmidtler, J. F., Kami, H. G., Ogielska, M., & Förster, D. W. (2019). Shedding Light on a Secretive Tertiary Urodelean Relict: Hynobiid Salamanders (Paradactylodon persicus s.l.) from Iran, Illuminated by Phylogeographic, Developmental, and Transcriptomic Data. Genes, 10(4), 306. https://doi.org/10.3390/genes10040306







