Evolutionary Insights of the ZW Sex Chromosomes in Snakes: A New Chapter Added by the Amazonian Puffing Snakes of the Genus Spilotes
Abstract
1. Introduction
2. Materials and Methods
2.1. Sampling, Mitotic Chromosomes Preparation, C-Banding, and Ag-NORs
2.2. Probes for Chromosome Hybridization
2.3. Fluorescence in Situ Hybridization (FISH) for Repetitive DNA Mapping
2.4. Preparation of Probes for Comparative Genome Hybridization (CGH)
2.5. Comparative Genomic Hybridization (CGH)
2.6. Microscopic Analyzes
3. Results
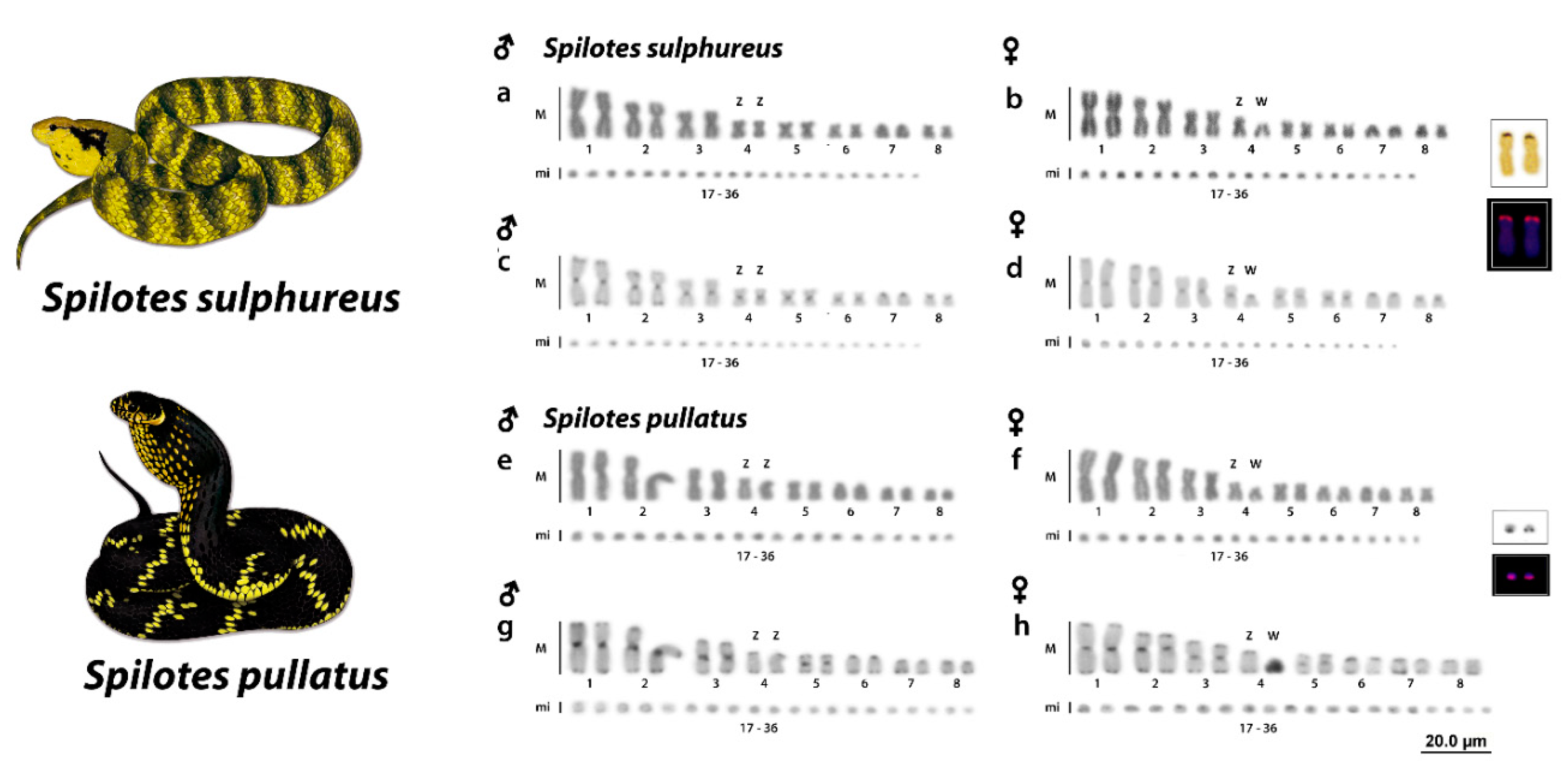
3.1. Karyotype
3.2. Ag-NORs, 18S rDNA, and C-Positive Heterochromatin
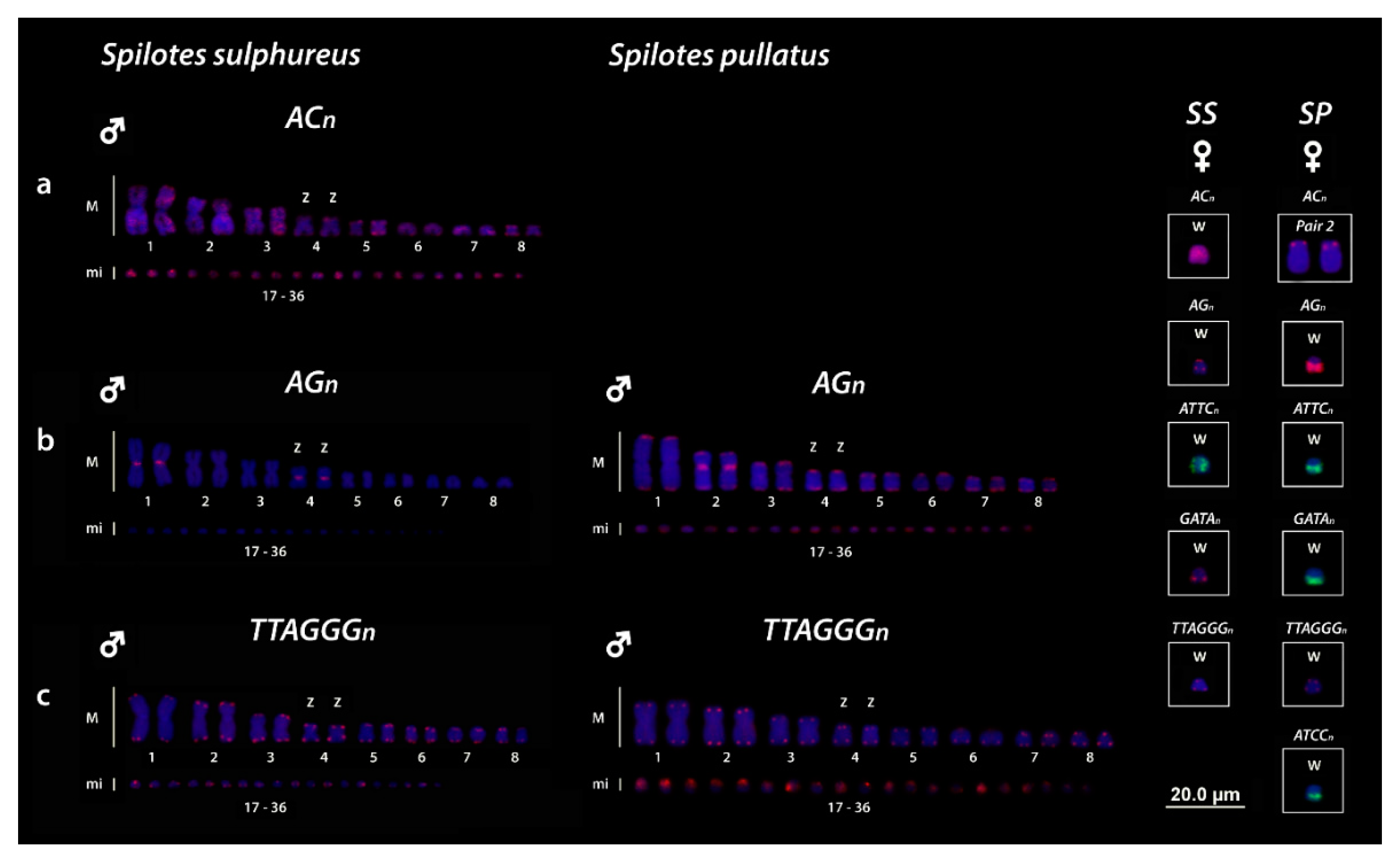
3.3. Chromosomal Mapping of Microsatellite Motifs and (TTAGGG)n Sequences
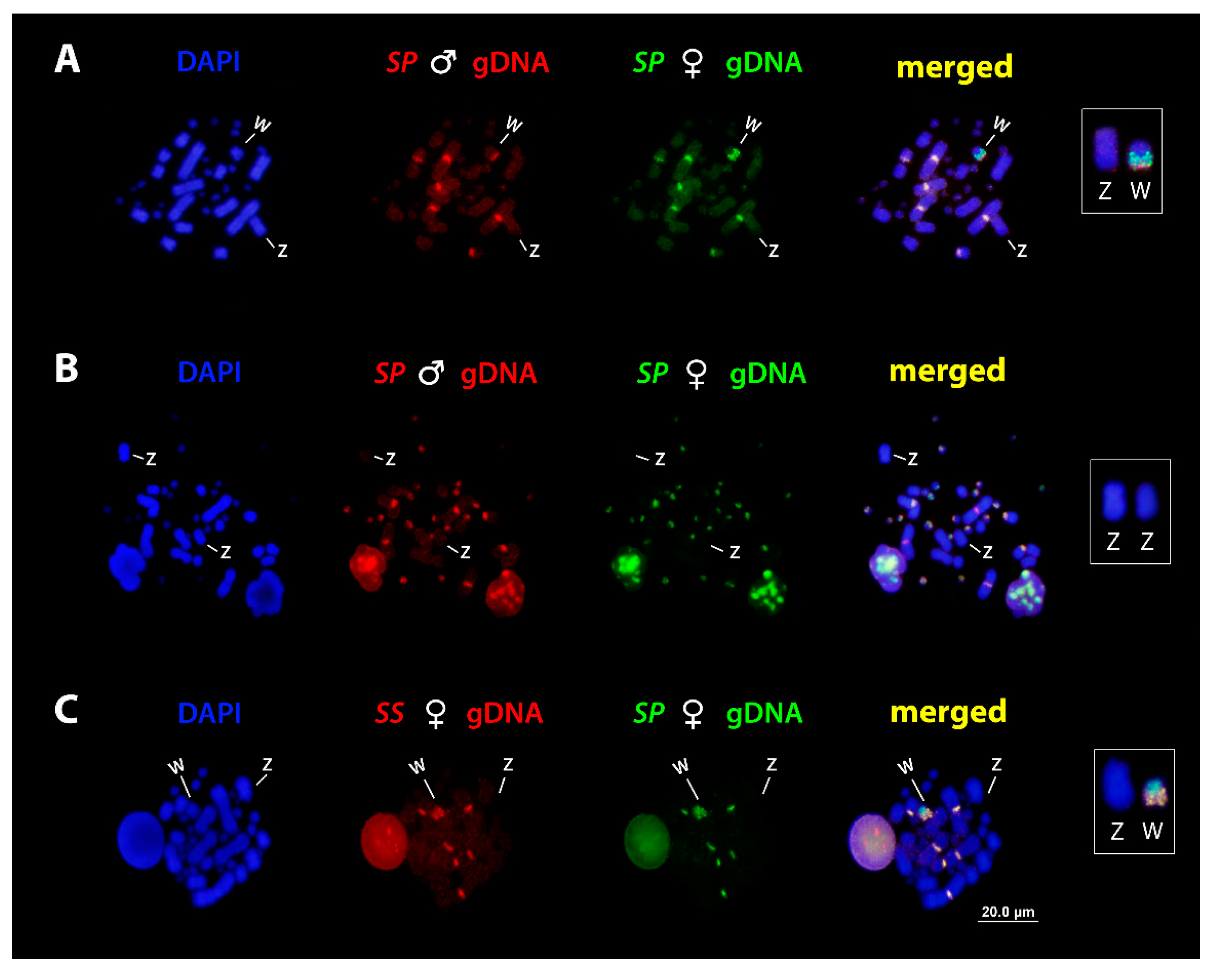
3.4. Comparative Genomic Hybridization (CGH)
4. Discussion
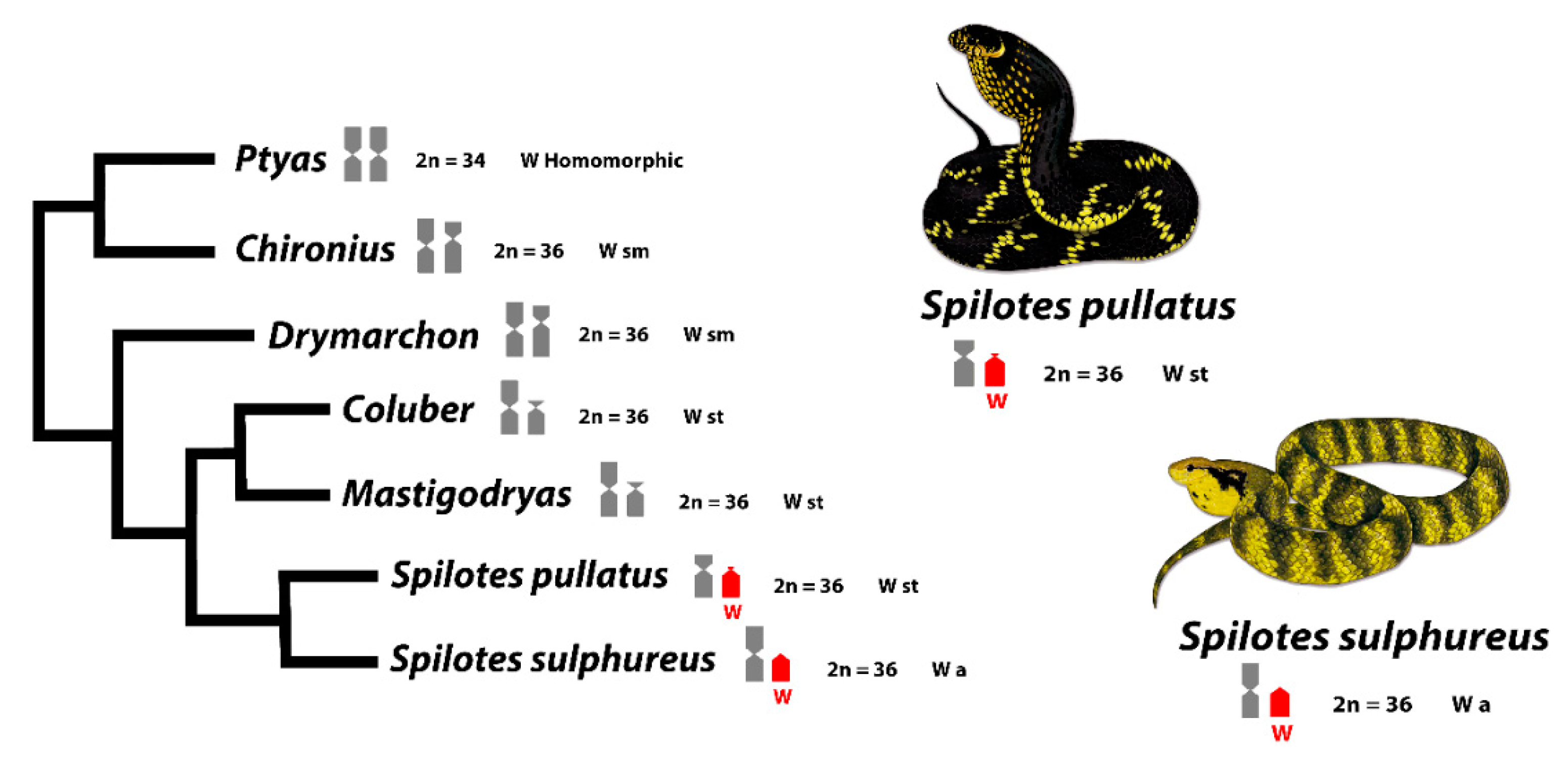
4.1. Cytotaxonomy in Spilotes Clade
4.2. Ribosomal Sequences in the Evolution of Snakes
4.3. Trends in the Role of Repetitive Sequences in the W Evolution of Caenophidian Snakes
4.4. Stability of Gene Content in Snakes Sex Chromosomes
4.5. Origin and Evolution of Sex Chromosomes in Caenophidian Snakes
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Ethics Statement
References
- Uetz, P.; Freed, P.; Hošek, J. (Eds.) The Reptile Database. Available online: http://www.reptile-database.org (accessed on 26 November 2018).
- Dowling, H.G.; Duellman, W.E. Systematic Herpetology: A Synopsis of Families and Higher Categories; Herpetological Information Search System Publications: New York, NY, USA, 1978. [Google Scholar]
- Vidal, N.; Dewynter, M.; Gower, D.J. Dissecting the major American snake radiation: A molecular phylogeny of the Dipsadidae Bonaparte (Serpentes, Caenophidia). C. R. Biol. 2010, 333, 48–55. [Google Scholar] [CrossRef]
- Pyron, R.A.; Burbrink, F.T.; Colli, G.R.; Montes de Oca, A.N.; Vitt, L.J.; Kuczynski, C.A.; Wiens, J.J. The phylogeny of advanced snakes (Colubroidea), with discovery of a new subfamily and comparison of support methods for likelihood trees. Mol. Phylogenet. Evol. 2011, 58, 329–342. [Google Scholar] [CrossRef]
- Pyron, R.A.; Burbrink, F.T.; John, J. A phylogeny and revised classification of Squamata, including 4161species of lizards and snakes. BMC Evol. Biol. 2013, 13, 93. [Google Scholar] [CrossRef] [PubMed]
- Pyron, R.A.; Hendry, C.R.; Chou, V.M.; Lemmon, E.M.; Lemmon, A.R.; Burbrink, F.T. Effectiveness of phylogenomic data and coalescent species tree methods for resolving difficult nodes in the phylogeny of advanced snakes (Serpentes: Caenophidia). Mol. Phylogenet. Evol. 2014, 81, 221–231. [Google Scholar] [CrossRef] [PubMed]
- Duellman, W.E. Patterns of species diversity in anuran amphibians in the American tropics. Ann. Mo. Bot. Gard. 1988, 75, 79–104. [Google Scholar] [CrossRef]
- Duellman, W.E. Hylid Frogs of Middle America, 2nd ed.; SSAR: Ithaca, NY, USA, 2001. [Google Scholar]
- Bergmann, P.J.; Russell, A.P. Systematics and biogeography of the widespread Neotropical gekkonid genus Thecadactylus (Squamata), with the description of a new cryptic species. Zool. J. Linn. Soc. 2007, 149, 339–370. [Google Scholar] [CrossRef]
- Ribas, C.C.; Aleixo, A.; Nogueira, A.C.; Miyaki, C.Y.; Cracraft, J. A palaeobiogeographic model for biotic diversification within Amazonia over the past three million years. Proc. R. Soc. Lond. B 2012, 279, 681–689. [Google Scholar] [CrossRef]
- Cunha, O.R.; Nascimento, F.P. Ofídios da Amazônia. X - As Cobras da Região Leste do Pará; Publicacoes Avulsas; Museu Paraense Emilio Goeldi: Belem, Portugal, 1978; Volume 31, pp. 1–218. [Google Scholar]
- Perez, S.; Moreno, A.G. Ofidios de Colombia; Museo Regionale di Scienze Naturali di Torino: Torino, Italy, 1988; 517p. [Google Scholar]
- De Fraga, R.; Lima, A.P.; Prudente, A.L.C.; Magnusson, W.E. Guia de Cobras da Região de Manaus-Amazônia Central; Editora INPA: Manaus, Brazil, 2013; 303p. [Google Scholar]
- Dixon, J.R.; Soini, P. The Reptiles of the Upper Amazon Basin, Iquitos Region, Peru. II. Crocodilians, Turtles and Snakes; Contributions in Biology and Geology; Milwaukee Public Museum: Milwaukee, WI, USA, 1977; pp. 1–71. [Google Scholar]
- Chippaux, J.P. Les Serpents de la Guyane Française: Faune Tropicale XXVII; IRD (Institut de recherche pour le développement): Paris, France, 1986; 167p. [Google Scholar]
- Wallach, V.; Williams, K.L.; Boundy, J. Snakes of the World: A Catalogue of Living and Extinct Species; CRC Press: Boca Raton, FL, USA, 2014. [Google Scholar] [CrossRef]
- Fitzinger, L. Systema Reptilium, Fasciculus Primus: Amblyglossae; Braumüller et Seidel: Wien, Austria, 1843; 106p. [Google Scholar]
- Jadin, R.C.; Burbrink, F.T.; Rivas, G.A.; Vitt, L.J.; Barrio-Amoros, C.L.; Guralnick, A.P. Finding arboreal snakes in an evolutionary tree: Phylogenetic placement and systematic revision of the Neotropical birdsnakes. J. Zool. Syst. Evol. Res. 2013, 52, 257–264. [Google Scholar] [CrossRef]
- Olmo, E.; Signorino, G. Chromorep: A Reptile Chromosomes Database. 2005. Available online: http://chromorep.univpm.it/ (accessed on 30 March 2018).
- Oguiura, N.; Ferrarezzi, H.; Batistic, R.F. Cytogenetics and molecular data in snakes: A phylogenetic approach. Cytogenet. Genome Res. 2009, 127, 128–142. [Google Scholar] [CrossRef]
- Beçak, W.; Beçak, M.L.; Nazareth, H.R.S.; Ohno, S. Close karyological kinship between the reptilian suborder Serpentes and the class Aves. Chromosoma 1964, 15, 606–617. [Google Scholar] [CrossRef]
- Beçak, W.; Beçak, M.L. Cytotaxonomy and chromosomal evolution in Serpentes. Cytogenetics 1969, 8, 247–262. [Google Scholar] [CrossRef] [PubMed]
- Rovatsos, M.; Johnson Pokorná, M.; Kratochvíl, L. Differentiation of sex chromosomes and karyotype characterisation in the dragonsnake Xenodermus javanicus (Squamata: Xenodermatidae). Cytogenet. Genome Res. 2015, 147, 48–54. [Google Scholar] [CrossRef]
- Falcione, C.; Hernando, A.B.; Di Pietro, D.O. Karyotypes of four species of Xenodontini snakes (Serpentes) and implications for taxonomy. Contrib. Zool. 2016, 85, 265–273. [Google Scholar] [CrossRef]
- Patawang, I.; Tanomtong, A.; Kaewmad, P.; Chuaynkern, Y.; Duengkae, P. New record on karyological analysis and first study of NOR localization of parthenogenetic brahminy blind snake, Ramphotyphlops braminus (Squamata, Typhlopidae) in Thailand. Nucleus 2016, 59, 61–66. [Google Scholar] [CrossRef]
- Ohno, S. Sex Chromosomes and Sex-Linked Genes; Springer: New York, NY, USA, 1967. [Google Scholar]
- Ezaz, T.; Sarre, S.D.; O’Meally, D.; Graves, J.A.; Georges, A. Sex chromosome evolution in lizards: Independent origins and rapid transitions. Cytogenet. Genome Res. 2009, 127, 249–260. [Google Scholar] [CrossRef] [PubMed]
- Rhen, T.; Schroeder, A. Molecular mechanisms of sex determination in reptiles. Sex Dev. 2010, 4, 16–28. [Google Scholar] [CrossRef] [PubMed]
- Ezaz, T.; Srikulnath, K.; Graves, J.A.M. Origin of amniote sex chromosomes: An ancestral super-sex chromosome, or common requirements? J. Hered. 2017, 108, 94–105. [Google Scholar] [CrossRef] [PubMed]
- Alam, S.; Sarre, S.; Gleeson, D.; Georges, A.; Ezaz, T. Did Lizards Follow Unique Pathways in Sex Chromosome Evolution? Genes 2018, 9, 239. [Google Scholar] [CrossRef]
- Pokorna, M.; Kratochvíl, L. Phylogeny of sex-determining mechanisms in squamate reptiles: Are sex chromosomes an evolutionary trap? Zool. J. Linn. Soc. 2009, 156, 168–183. [Google Scholar] [CrossRef]
- Gamble, T.; Castoe, T.A.; Nielsen, S.V.; Banks, J.L.; Card, D.C.; Schield, D.R.; Schuett, G.W.; Booth, W. The discovery of XY sex chromosomes in a boa and report the discovery of XY sex chromosomes in a boa and python. Curr. Biol. 2017, 27, 1–6. [Google Scholar] [CrossRef]
- Matsubara, K.; Tarui, H.; Toriba, M.; Yamada, K.; Nishida-Umehara, C.; Agata, K.; Matsuda, Y. Evidence for different origin of sex chromosomes in snakes, birds, and mammals and step-wise differentiation of snake sex chromosomes. PNAS 2006, 103, 18190–18195. [Google Scholar] [CrossRef] [PubMed]
- O’Meally, D.; Patel, H.R.; Stiglec, R.; Sarre, S.D.; Georges, A.; Graves, J.A.M.; Ezaz, T. Non-homologous sex chromosomes of birds and snakes share repetitive sequences. Chromosome Res. 2010, 18, 787–800. [Google Scholar] [CrossRef]
- Vicoso, B.; Emerson, J.J.; Zektser, Y.; Mahajan, S.; Bachtrog, D. Comparative sex chromosome genomics in snakes: Differentiation, evolutionary strata, and lack of global dosage compensation. PLoS Biol. 2013, 11, e1001643. [Google Scholar] [CrossRef] [PubMed]
- Viana, P.F.; Ribeiro, L.B.; Souza, G.M.; Chalkidis, H.d.M.; Gross, M.C.; Feldberg, E. Is the Karyotype of Neotropical Boid Snakes Really Conserved? Cytotaxonomy, Chromosomal Rearrangements and Karyotype Organization in the Boidae Family. PLoS ONE 2016, 11, e0160274. [Google Scholar] [CrossRef]
- Singh, L.; Purdom, I.F.; Jones, K.W. Sex chromosome associated satellite DNA: Evolution and conservation. Chromosoma 1980, 79, 137–157. [Google Scholar] [CrossRef] [PubMed]
- Augstenová, B.; Mazzoleni, S.; Kratochvíl, L.; Rovatsos, M. Evolutionary dynamics of the W chromosome in caenophidian snakes. Genes 2017, 9, 5. [Google Scholar] [CrossRef] [PubMed]
- Rovatsos, M.; Vuki´c, J.; Lymberakis, P.; Kratochvíl, L. Evolutionary stability of sex chromosomes in snakes. Proc. R. Soc. B Biol. Sci. 2015, 282, 20151992. [Google Scholar] [CrossRef]
- Matsubara, K.; Nishida, C.; Matsuda, Y.; Kumazawa, Y. Sex chromosome evolution in snakes inferred from divergence patterns of two gametologous genes and chromosome distribution of sex chromosome-linked repetitive sequences. Zool. Lett. 2016, 2, 19. [Google Scholar] [CrossRef] [PubMed]
- Deakin, J.E.; Ezaz, T. Understanding the Evolution of Reptile Chromosomes through Applications of Combined Cytogenetics and Genomics Approaches. Cytogenet. Genome Res. 2019. [Google Scholar] [CrossRef] [PubMed]
- Carvalho, P.C.; de Oliveira, E.A.; Bertollo, L.A.C.; Yano, C.F.; Oliveira, C.; Decru, E.; Jegede, O.I.; Hatanaka, T.; Liehr, T.; Al-Rikabi, A.B.H.; et al. First chromosomal analysis in Hepsetidae (Actinopterygii, Characiformes): Insights into relationship between African and Neotropical fish groups. Front. Genet. 2017, 8, 203. [Google Scholar] [CrossRef]
- Hatanaka, T.; De Oliveira, E.A.; Ráb, P.; Yano, C.F.; Bertollo, L.A.; Ezaz, T.; Jegede, O.O.I.; Liehr, T.; Olaleye, V.F.; Cioffi, M.d.B. First chromosomal analysis in Gymnarchus niloticus (Gymnarchidae: Osteoglossiformes): Insights into the karyotype evolution of this ancient fish order. Biol. J. Linn. Soc. 2018, 125, 83–92. [Google Scholar] [CrossRef]
- Singchat, W.; O’Connor, R.E.; Tawichasri, P.; Suntronpong, A.; Sillapaprayoon, S.; Suntrarachun, S.; Muangmai, N.; Baicharoen, S.; Peyachoknagul, S.; Chanhome, L.; et al. Chromosome map of the Siamese cobra: Did partial synteny of sex chromosomes in the amniote represent “a hypothetical ancestral super-sex chromosome” or random distribution? BMC Genomics 2018, 19, 939. [Google Scholar] [CrossRef] [PubMed]
- Viana, P.F.; Ribeiro, L.B.; Lima, T.; de Carvalho, V.T.; Vogt, R.C.; Gross, M.C.; Feldberg, E. An optimized protocol for obtaining mitotic chromosomes from cultured reptilian lymphocytes. Nucleus 2016, 59, 1–5. [Google Scholar] [CrossRef]
- Sumner, A.T. A simple technique for demonstrating centromeric heterochromatin. Exp. Cell Res. 1972, 75, 204–206. [Google Scholar] [CrossRef]
- Howell, W.M.; Black, D.A. Controlled silver staining of nucleolus organizer regions with a protective colloidal developer: A 1-step method. Experientia 1980, 36, 1014–1015. [Google Scholar] [CrossRef] [PubMed]
- Gross, M.C.; Schneider, C.H.; Valente, G.T.; Porto, J.I.R.; Martins, C.; Feldberg, E. Variability of 18S rDNA locus among Symphysodon fishes: Chromosomal rearrangements. J. Fish Biol. 2010, 76, 1117–1127. [Google Scholar] [CrossRef] [PubMed]
- Ijdo, J.W.; Wells, R.A.; Baldini, A.; Reeders, S.T. Improved telomere detection using a telomere repeat probe (TTAGGG)n generated by PCR. Nucleic Acids Res. 1991, 19, 4780. [Google Scholar] [CrossRef]
- Kubat, Z.; Hobza, R.; Vyskot, B.; Kejnovsky, E. Microsatellite accumulation in the Y chromosome of Silene latifolia. Genome 2008, 51, 350–356. [Google Scholar] [CrossRef]
- Pinkel, D.; Straume, T.; Gray, J. Cytogenetic analysis using quantitative, high sensitivity, fluorescence hybridization. Proc. Natl. Acad. Sci. USA 1986, 83, 2934–2938. [Google Scholar] [CrossRef] [PubMed]
- Zwick, M.S.; Hanson, R.E.; Islam-Faridi, M.H.; Stelly, D.M.; Wing, R.A.; Price, H.J.; McKnight, T.D. A rapid procedure for the isolation of C0t-1 DNA from plants. Genome 1997, 40, 138–142. [Google Scholar] [CrossRef] [PubMed]
- Symonová, R.; Sember, A.; Majtánová, Z.; Ráb, P. Characterization of fish genomes by GISH and CGH. In Fish Cytogenetic Techniques. Ray-Fin Fishes and Chondrichthyans; Ozouf-Costaz, C., Pisano, E., Foresti, F., Almeida Toledo, L.F., Eds.; CCR Press: Boca Raton, FL, USA, 2015; pp. 118–131. [Google Scholar]
- Levan, A.; Fredga, K.; Sandberg, A.A. Nomenclature for centromeric position on chromosomes. Hereditas 1964, 52, 201–220. [Google Scholar] [CrossRef]
- Figueroa, A.; McKelvy, A.D.; Grismer, L.L.; Bell, C.D.; Lailvaux, S.P. A species-level phylogeny of extant snakes with description of a new colubrid subfamily and genus. PLoS ONE 2016, 11, e0161070. [Google Scholar] [CrossRef] [PubMed]
- Abe, A.S.; Fernandes, W. Polymorphism in Spilotes pullatus anomalepis Bocourt (Reptilia, Serpentes: Colubridae). J. Herpetol. 1977, 11, 98–100. [Google Scholar] [CrossRef]
- Beçak, W. Constituição cromossômica e mecanismo de determinação do sexo em ofídios sul americanos. I. Aspectos cariotípicos. Mem Inst Butantan 1965, 32, 37–78. [Google Scholar] [PubMed]
- Porter, C.A.; Hamilton, M.J.; Sites, J.W., Jr.; Baker, R.J. Location of ribosomal DNA in chromosomes of squamate reptiles: Systematic and evolutionary implications. Herpetologica 1991, 47, 271–280. [Google Scholar]
- Camper, J.D.; Hanks, B. Variation in the nucleolus organizer region among New World snakes. J. Herpetol. 1995, 29, 468–471. [Google Scholar] [CrossRef]
- Moreno, R.; Navarro, J.; Iturra, P.; Veloso, A. The karyotype of Philodryas chamissonis (Colubridae). Identification of nucleolar organizer regions (NOR) and sex chromosomes by banding methods. Rev. Bras. Genet. 1987, 10, 497–506. [Google Scholar]
- Trajtengertz, I.; Beçak, M.L.; Ruiz, I.R.G. Ribosomal cistrons in Bothrops neuwiedi (Serpentes) subspecies from Brazil. Genome 1995, 38, 601–606. [Google Scholar] [CrossRef]
- García, J.A.R.; Hernando, A. Standard karyotype and nucleolus organizer region of Neotropical blindsnake Typhlops brongersmianus (Serpentes: Typhlopidae). Acta Herpetol. 2007, 2, 117–120. [Google Scholar]
- Abramyan, J.; Ezaz, T.; Graves, J.A.M.; Koopman, P. Z and W sex chromosomes in the cane toad (Bufo marinus). Chromosome Res. 2009, 17, 1015–1024. [Google Scholar] [CrossRef]
- Scacchetti, P.C.; Utsunomia, R.; Pansonato-Alves, J.C.; da Costa Silva, G.J.; Vicari, M.R.; Artoni, R.F.; Oliveira, C.; Foresti, F. Repetitive DNA Sequences and Evolution of ZZ/ZW Sex Chromosomes in Characidium (Teleostei: Characiformes). PLoS ONE 2015, 10, e0137231. [Google Scholar] [CrossRef] [PubMed]
- Kawagoshi, T.; Uno, Y.; Nishida, C.; Matsuda, Y. The Staurotypus turtles and aves share the same origin of sex chromosomes but evolved different types of heterogametic sex determination. PLoS ONE 2014, 9, e105315. [Google Scholar] [CrossRef]
- Viana, P.F.; Ezaz, T.; Marajó, L.; Ferreira, M.; Zuanon, J.; Cioffi, M.B.; Bertollo, L.A.C.; Feldberg, E. Genomic Organization of Repetitive DNAs and Differentiation of an XX/XY Sex Chromosome System in the Amazonian Puffer Fish, Colomesus asellus (Tetraodontiformes). Cytogenet. Genome Res. 2017, 153, 96–104. [Google Scholar] [CrossRef]
- Cioffi, M.B.; Bertollo, L.A.C. Chromosomal distribution and evolution of repetitive DNAs in fish. In Repetitive DNAs; Garrido, R., Ed.; Karger: Basel, Switzerland, 2012. [Google Scholar]
- Ezaz, T.; Azad, B.; O’Meally, D.; Young, M.J.; Matsubara, K.; Edwards, M.J.; Zhang, X.; Holleley, C.E.; Deakin, J.E.; Graves, J.A.M.; et al. Sequence and gene content of a large fragment of a lizard sex chromosome and evaluation of candidate sex differentiating gene R-spondin 1. BMC Genomics 2013, 14, 899. [Google Scholar] [CrossRef] [PubMed]
- Matsubara, K.; O’Meally, D.; Azad, B.; Georges, A.; Sarre, S.D.; Graves, J.A.M.; Matsuda, Y.; Ezaz, T. Amplification of microsatellite repeat motifs is associated with the evolutionary differentiation and heterochromatinization of sex chromosomes in Sauropsida. Chromosoma 2016, 125, 111–123. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, M.; Garcia, C.; Matoso, D.A.; de Jesus, I.S.; Feldberg, E. A new multiple sex chromosome system X1X1X2X2/X1Y1X2Y2 in Siluriformes: Cytogenetic characterization of Bunocephalus coracoideus (Aspredinidae). Genetica 2016, 144, 591–599. [Google Scholar] [CrossRef] [PubMed]
- Rovatsos, M.; Johnson Pokorná, M.; Altmanová, M.; Kratochvíl, L. Mixed-Up Sex Chromosomes: Identification of Sex Chromosomes in the X1X1X2X2/X1X2Y System of the Legless Lizards of the Genus Lialis (Squamata: Gekkota: Pygopodidae). Cytogenet Genome Res. 2016, 149, 282–289. [Google Scholar] [CrossRef] [PubMed]
- Souza e Sousa, J.F.D.; Viana, P.F.; Bertollo, L.A.; Cioffi, M.B.; Feldberg, E. Evolutionary Relationships among Boulengerella Species (Ctenoluciidae, Characiformes): Genomic Organization of Repetitive DNAs and Highly Conserved Karyotypes. Cytogenet. Genome Res. 2017, 152, 194–203. [Google Scholar] [CrossRef]
- Rovatsos, M.T.; Marchal, J.A.; Romero-Fernández, I.; Fernández, F.J.; Giagia-Athanosopoulou, E.B.; Sánchez, A. Rapid, independent, and extensive amplification of telomeric repeats in pericentromeric regions in karyotypes of arvicoline rodents. Chromosome Res. 2011, 19, 869–882. [Google Scholar] [CrossRef]
- Young, M.J.; O’Meally, D.; Sarre, S.D.; Georges, A.; Ezaz, T. Molecular cytogenetic map of the central bearded dragon, Pogona vitticeps (Squamata: Agamidae). Chromosome Res. 2013, 21, 361–374. [Google Scholar] [CrossRef]
- Matsubara, K.; Uno, Y.; Srikulnath, K.; Matsuda, Y.; Miller, E.; Olsson, M. No interstitial telomeres on autosomes but remarkable amplification of telomeric repeats on theWsex chromosome in the sand lizard (Lacerta agilis). J. Hered. 2015, 106, 753–757. [Google Scholar] [CrossRef] [PubMed]
- Rovatsos, M.; Kratochvíl, L.; Altmanová, M.; Johnson Pokorná, M. Interstitial telomeric motifs in squamate reptiles: When the exceptions outnumber the rule. PLoS ONE 2015, 10, e0134985. [Google Scholar] [CrossRef] [PubMed]
- Hardman, N. Structure and function of repetitive DNA in eukaryotes. Biochem. J. 1986, 234, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Pokorná, M.; Kratochvíl, L.; Kejnovský, E. Microsatellite distribution on sex chromosomes at different stages of heteromorphism and heterochromatinization in two lizard species (Squamata: Eublepharidae: Coleonyx elegans and lacertidae: Eremias velox). BMC Genet. 2011, 12, 90. [Google Scholar] [CrossRef] [PubMed]
- Matsubara, K.; Sarre, S.D.; Georges, A.; Matsuda, Y.; Graves, J.A.M.; Ezaz, T. Highly differentiated ZW sex microchromosomes in the Australian Varanus species evolved through rapid amplification of repetitive sequences. PLoS ONE 2014, 9, e95226. [Google Scholar] [CrossRef] [PubMed]
- Ray-Chaudhuri, S.P.; Singh, L.; Sharma, T. Evolution of sex chromosomes and formation of W chromatin in snakes. Chromosoma 1971, 33, 239–251. [Google Scholar] [CrossRef] [PubMed]
- Jones, K.W.; Singh, L. Snakes and evolution of sex chromosomes. TIG 1985, 1, 55–61. [Google Scholar] [CrossRef]
- Pucci, M.B.; Barbosa, P.; Nogaroto, V.; Almeida, M.C.; Artoni, R.F.; Scacchetti, P.C.; Pansonato-Alves, J.C.; Foresti, F.; Moreira-Filho, O.; Vicari, M.R. Chromosomal Spreading of Microsatellites and (TTAGGG) n Sequences in the Characidium zebra and C. gomesi Genomes (Characiformes: Crenuchidae). Cytogenet. Genome Res. 2016, 149, 182–190. [Google Scholar] [CrossRef]
- Pucci, M.B.; Nogaroto, V.; Bertollo, L.A.C.; Moreira-Filho, O.; Vicari, M.R. The karyotypes and evolution of ZZ/ZW sex chromosomes in the genus Characidium (Characiformes, Crenuchidae). Comp. Cytogenet. 2018, 12, 421–438. [Google Scholar] [CrossRef]
- Singh, L.; Wadhwa, R.; Naidu, S.; Nagaraj, R.; Ganesan, M. Sex- and tissue-specific Bkm(GATA)-binding protein in the germcells of heterogametic sex. J. Biol. Chem. 1994, 269, 25321–25327. [Google Scholar]
- Trainor, C.D.; Omichinski, J.G.; Vandergon, T.L.; Gronenborn, A.M.; Clore, G.M.; Felsenfeld, G. A palindromic regulatory site within vertebrate GATA-1 promoters requires both zinc fingers of the GATA-1 DNA-binding domain for high-affinity interaction. Mol. Cell Biol. 1996, 16, 2238–2247. [Google Scholar] [CrossRef] [PubMed]
- Priyadarshini, P.; Murthy, B.S.; Nagaraju, J.; Singh, L. A GATA-binding protein expressed predominantly in the pupal ovary of the silkworm, Bombyx mori. Insect Biochem. Mol. Biol. 2003, 33, 185–195. [Google Scholar] [CrossRef]
- Subramanian, S.; Mishra, R.K.; Singh, L. Genomewide analysis of Bkm sequences (GATA repeats): Predominant association with sex chromosomes and potential role in higher order chromatin organization and function. Bioinformatics 2003, 19, 681–685. [Google Scholar] [CrossRef] [PubMed]
- Rovatsos, M.; Altmanová, M.; Johnson Pokorná, M.; Augstenová, B.; Kratochvíl, L. Cytogenetics of the Javan file snake (Acrochordus javanicus) and the evolution of snake sex chromosomes. J. Zool. Syst. Evol. Res. 2018, 56, 117–125. [Google Scholar] [CrossRef]
- Adams, R.H.; Blackmon, H.; Reyes-Velasco, J.; Schield, D.R.; Card, D.C.; Andrew, A.L.; Waynewood, N.; Castoe, T.A. Microsatellite landscape evolutionary dynamics across 450 million years of vertebrate genome evolution. Genome 2016, 59, 295–310. [Google Scholar] [CrossRef] [PubMed]
- O’Meally, D.; Ezaz, T.; Georges, A.; Sarre, S.D.; Graves, J.A.M. Are some chromosomes particularly good at sex? Insights from amniotes. Chromosome Res. 2012, 20, 7–19. [Google Scholar] [CrossRef] [PubMed]
- Singh, L. Evolution of karyotypes in snakes. Chromosoma 1972, 38, 185–236. [Google Scholar] [CrossRef] [PubMed]
- Poltronieri, J.; Marquioni, V.; Bertollo, L.A.C.; Kejnovsky, E.; Molina, W.F.; Liehr, T.; Cioffi, M.B. Comparative chromosomal mapping of microsatellites in Leporinus species (Characiformes, Anostomidae): Unequal accumulation on the W chromosomes. Cytogenet. Genome Res. 2014, 142, 40–45. [Google Scholar] [CrossRef]
- Yano, C.F.; Bertollo, L.A.C.; Ezaz, T.; Trifonov, V.; Sember, A.; Liehr, T.; Cioffi, M.B. Highly conserved Z and molecularly diverged W chromosomes in the fish genus Triportheus (Characiformes, Triportheidae). Heredity 2017, 118, 276–283. [Google Scholar] [CrossRef]
- Kretschmer, R.; de Oliveira, T.D.; de Oliveira Furo, I.; Oliveira Silva, F.A.; Gunski, R.J.; del Valle Garnero, A.; de Bello Cioffi, M.; de Oliveira, E.H.C.; de Freitas, T.R.O. Repetitive DNAs and shrink genomes: A chromosomal analysis in nine Columbidae species (Aves, Columbiformes). Genet. Mol. Biol. 2018, 41, 98–106. [Google Scholar] [CrossRef]
- Mank, J.E.; Promislow, D.E.L.; Avise, J.C. Evolution of alternative sex-determining mechanisms in teleost fishes. Biol. J. Linn. Soc. 2006, 87, 83–93. [Google Scholar] [CrossRef]
- Cioffi, M.; Sánchez, A.; Marchal, J.A.; Kosyakova, N.; Liehr, T.; Trifonov, V.; Bertollo, L.A.C. Whole chromosome painting reveals independent origin of sex chromosomes in closely related forms of a fish species. Genetica 2011, 139, 1065. [Google Scholar] [CrossRef] [PubMed]
- Vicoso, B.; Charlesworth, B. Effective population size and the Faster-X Effect: An extended model. Evolution 2009, 63, 2413–2426. [Google Scholar] [CrossRef]
- Mank, J.E.; Nam, K.; Ellegren, H. Faster-Z evolution is predominantly due to genetic drift. Mol. Biol. Evol. 2010, 27, 661–670. [Google Scholar] [CrossRef] [PubMed]
- Laopichienpong, N.; Muangmai, N.; Chanhome, L.; Suntrarachun, S.; Twilprawat, P.; Peyachoknagul, S.; Srikulnath, K. Evolutionary dynamics of the gametologous CTNNB1 gene on the Z and W chromosomes of snakes. J. Hered. 2017, 108, 142–151. [Google Scholar] [PubMed]
- De Smet, W.H.O. Chromosomes of 23 species of snakes. Acta Zool. Pathol. Antverp. 1978, 70, 85–118. [Google Scholar]
- Baker, R.J.; Mengden, G.A.; Bull, J.J. Karyotypic studies of thirty-eight species of North American snakes. Copeia 1972, 1972, 257–265. [Google Scholar] [CrossRef]
- Laopichienpong, N.; Tawichasri, P.; Chanhome, L.; Phatcharakullawarawat, R.; Singchat, W.; Kantachumpoo, A.; Suntrarachun, S.; Matsubara, K.; Peyachoknagul, S.; Srikulnath, K. A novel method of caenophidian snake sex identification using molecular markers based on two gametologous genes. Ecol. Evol. 2017, 7, 4661–4669. [Google Scholar] [CrossRef]

© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
F. Viana, P.; Ezaz, T.; de Bello Cioffi, M.; Jackson Almeida, B.; Feldberg, E. Evolutionary Insights of the ZW Sex Chromosomes in Snakes: A New Chapter Added by the Amazonian Puffing Snakes of the Genus Spilotes. Genes 2019, 10, 288. https://doi.org/10.3390/genes10040288
F. Viana P, Ezaz T, de Bello Cioffi M, Jackson Almeida B, Feldberg E. Evolutionary Insights of the ZW Sex Chromosomes in Snakes: A New Chapter Added by the Amazonian Puffing Snakes of the Genus Spilotes. Genes. 2019; 10(4):288. https://doi.org/10.3390/genes10040288
Chicago/Turabian StyleF. Viana, Patrik, Tariq Ezaz, Marcelo de Bello Cioffi, Breno Jackson Almeida, and Eliana Feldberg. 2019. "Evolutionary Insights of the ZW Sex Chromosomes in Snakes: A New Chapter Added by the Amazonian Puffing Snakes of the Genus Spilotes" Genes 10, no. 4: 288. https://doi.org/10.3390/genes10040288
APA StyleF. Viana, P., Ezaz, T., de Bello Cioffi, M., Jackson Almeida, B., & Feldberg, E. (2019). Evolutionary Insights of the ZW Sex Chromosomes in Snakes: A New Chapter Added by the Amazonian Puffing Snakes of the Genus Spilotes. Genes, 10(4), 288. https://doi.org/10.3390/genes10040288




