Exposure of Human Skin Organoids to Low Genotoxic Stress Can Promote Epithelial-to-Mesenchymal Transition in Regenerating Keratinocyte Precursor Cells
Abstract
1. Introduction
2. Materials and Methods
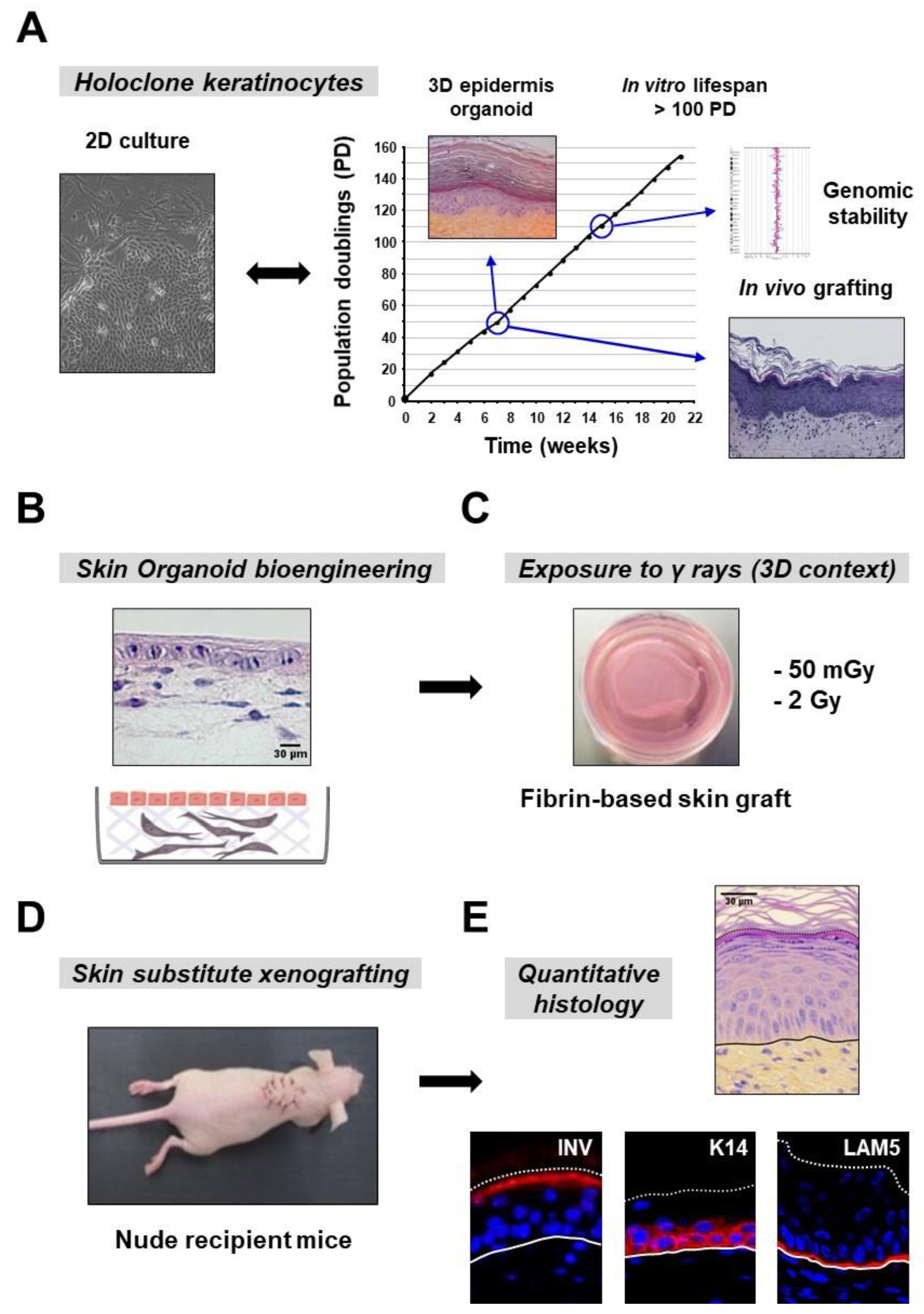
2.1. Human Tissue and Cell Materials
2.2. Bi-Dimensional Culture of Keratinocytes
2.3. Three-Dimensional Skin Substitute Bioengineering
2.4. Skin Substitute Irradiation
2.5. Skin Substitute Xenografting
2.6. Preparation of Skin Sections for Histological Characterization
2.7. Quantitative Histology
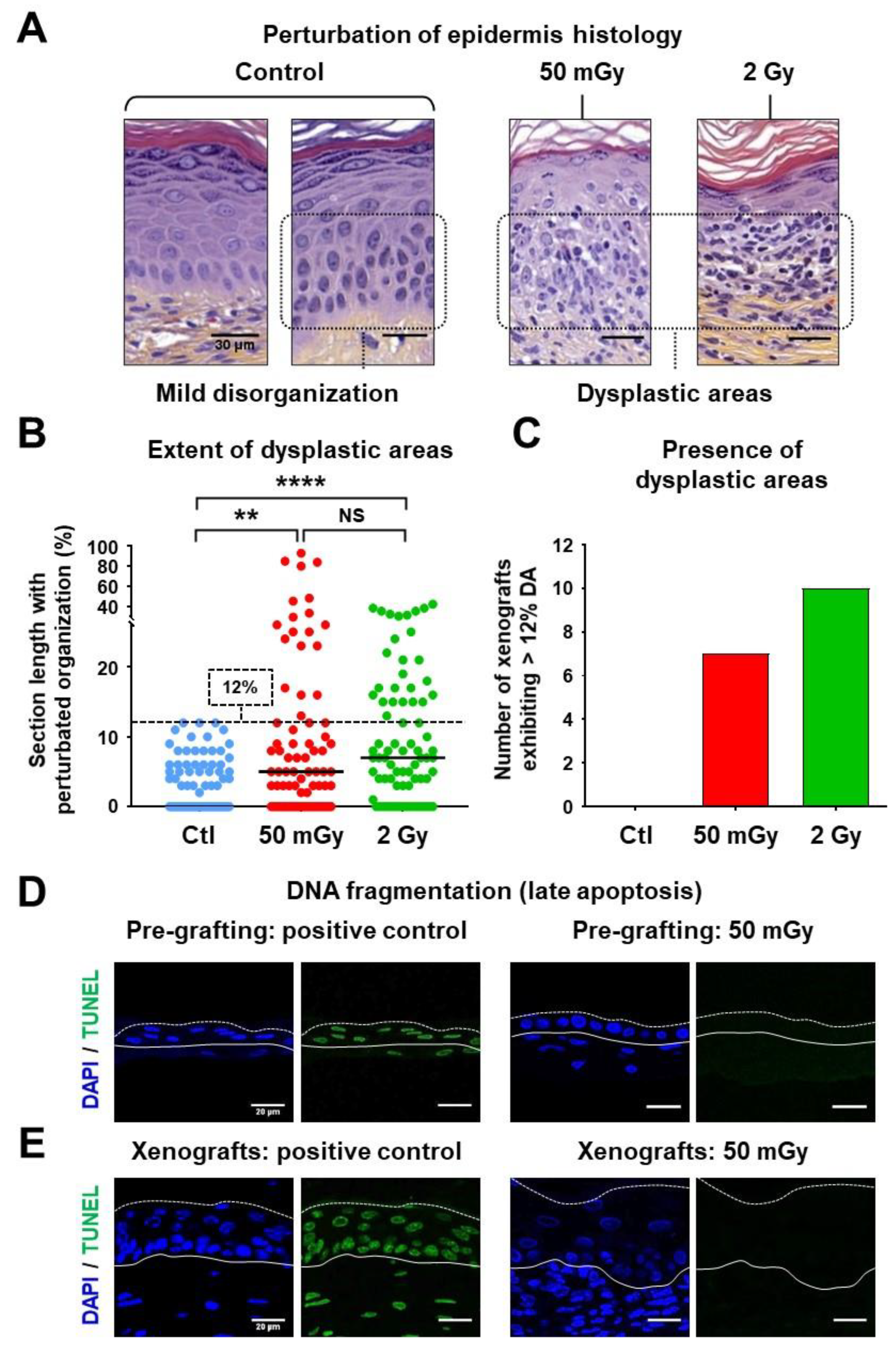
2.7.1. Percentage of Section Length Displaying Abnormal Epidermis Histology
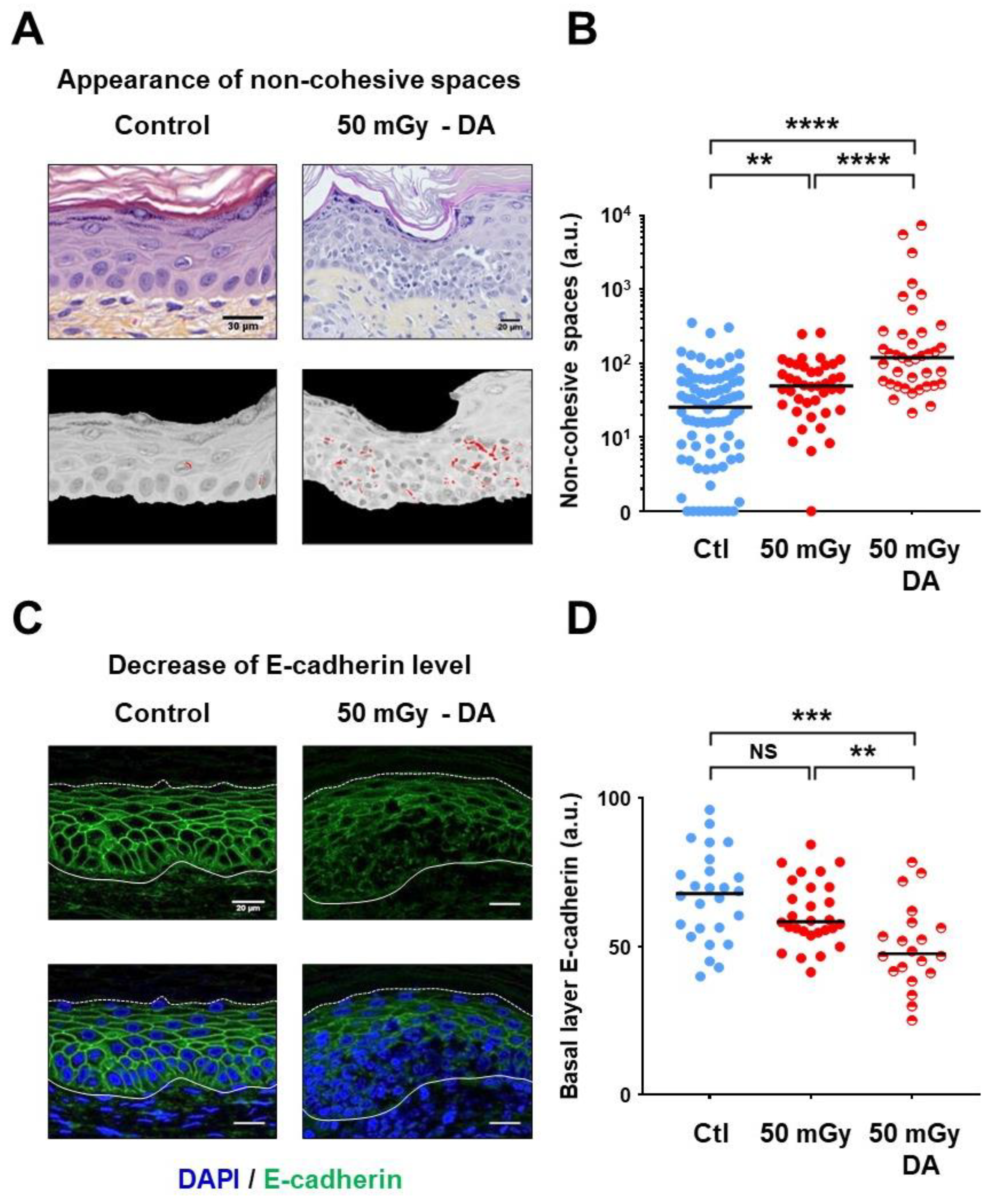
2.7.2. Detection of Non-Cohesive Spaces within Regenerated Epidermises
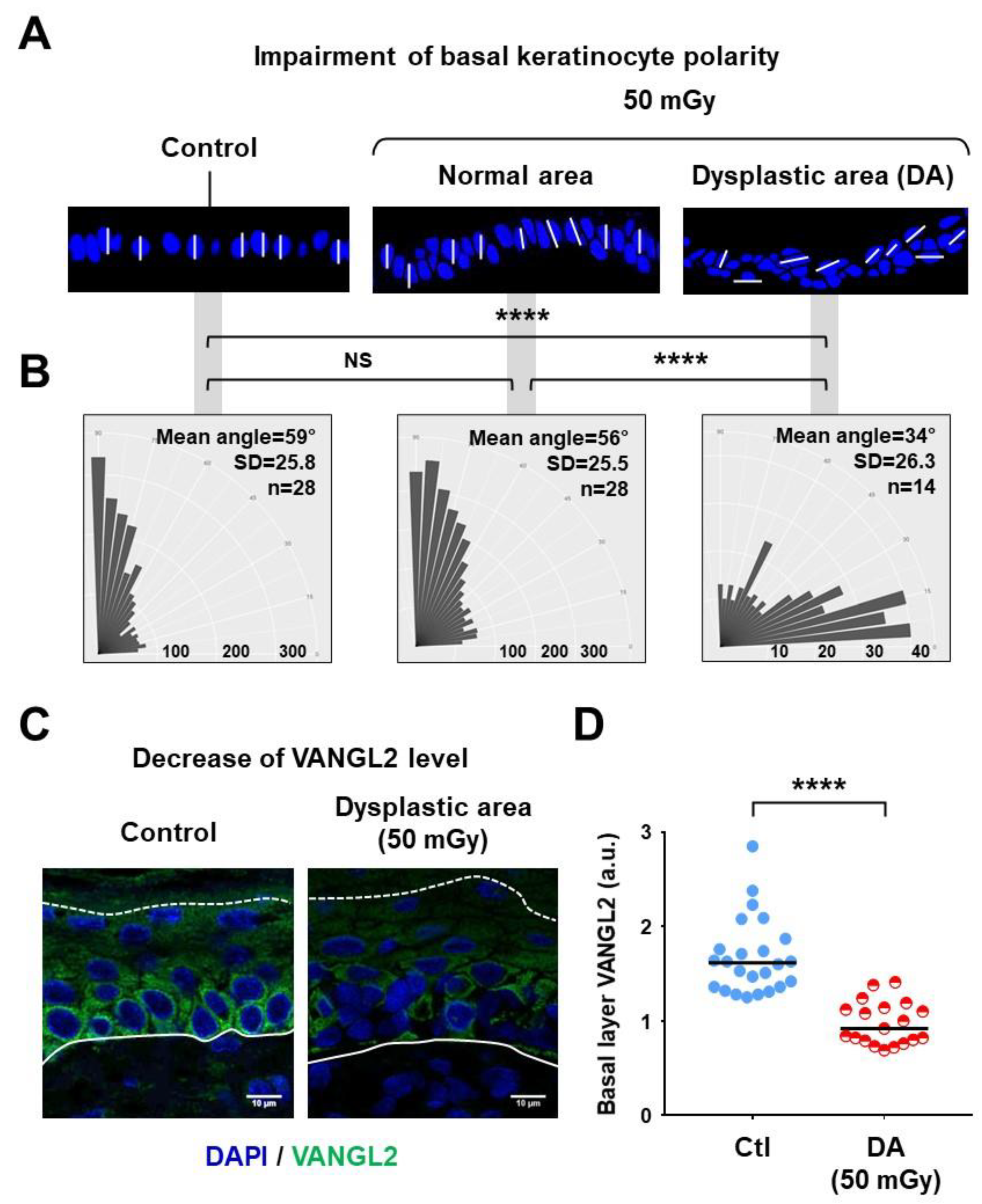
2.7.3. Assessment of Keratinocyte Polarity
2.8. Section Processing for Immunofluorescence Analyses
2.9. Semi-Quantitative Immunofluorescence Analyses
2.10. Apoptosis Assay
2.11. Statistics
3. Results
3.1. Experimental Design
3.2. Irradiation Did Not Compromise Epidermis Regeneration but Induced Local Dysplasia
3.3. Dysplastic Areas Exhibited Impaired Polarity of Basal Keratinocytes
3.4. Defective Cell–Cell Interactions Were Observed in Dysplastic Areas
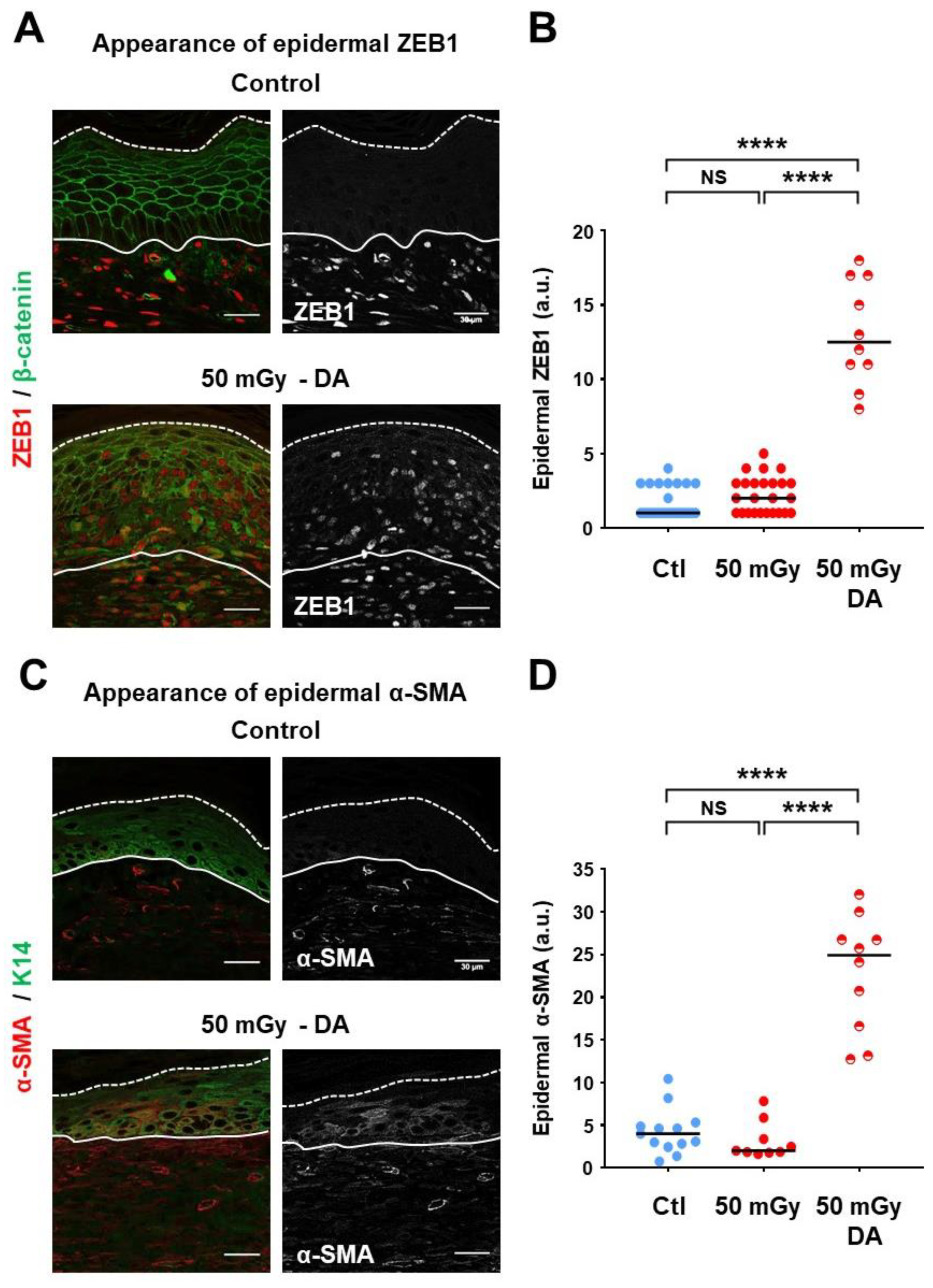
3.5. Epithelial-to-Mesenchymal Transition Markers Were Detected in Epidermal Dysplastic Areas
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Richards, D.J.; Li, Y.; Kerr, C.M.; Yao, J.; Beeson, G.C.; Coyle, R.C.; Chen, X.; Jia, J.; Damon, B.; Wilson, R.; et al. Human cardiac organoids for the modelling of myocardial infarction and drug cardiotoxicity. Nat. Biomed. Eng. 2020, 4, 446–462. [Google Scholar] [CrossRef] [PubMed]
- Pașca, A.M.; Park, J.Y.; Shin, H.W.; Qi, Q.; Revah, O.; Krasnoff, R.; O’Hara, R.; Willsey, A.J.; Palmer, T.D.; Pașca, S.P. Human 3D cellular model of hypoxic brain injury of prematurity. Nat. Med. 2019, 25, 784–791. [Google Scholar] [CrossRef] [PubMed]
- Ballabio, C.; Anderle, M.; Gianesello, M.; Lago, C.; Miele, E.; Cardano, M.; Aiello, G.; Piazza, S.; Caron, D.; Gianno, F.; et al. Modeling medulloblastoma in vivo and with human cerebellar organoids. Nat. Commun. 2020, 11, 583. [Google Scholar] [CrossRef] [PubMed]
- Huch, M.; Gehart, H.; van Boxtel, R.; Hamer, K.; Blokzijl, F.; Verstegen, M.M.; Ellis, E.; van Wenum, M.; Fuchs, S.A.; de Ligt, J.; et al. Long-term culture of genome-stable bipotent stem cells from adult human liver. Cell 2015, 160, 299–312. [Google Scholar] [CrossRef]
- Broutier, L.; Mastrogiovanni, G.; Verstegen, M.M.; Francies, H.E.; Gavarró, L.M.; Bradshaw, C.R.; Allen, G.E.; Arnes-Benito, R.; Sidorova, O.; Gaspersz, M.P.; et al. Human primary liver cancer-derived organoid cultures for disease modeling and drug screening. Nat. Med. 2017, 23, 1424–1435. [Google Scholar] [CrossRef]
- Clevers, H. Modeling Development and Disease with Organoids. Cell 2016, 165, 1586–1597. [Google Scholar] [CrossRef]
- Drost, J.; van Boxtel, R.; Blokzijl, F.; Mizutani, T.; Sasaki, N.; Sasselli, V.; de Ligt, J.; Behjati, S.; Grolleman, J.E.; van Wezel, T.; et al. Use of CRISPR-modified human stem cell organoids to study the origin of mutational signatures in cancer. Science 2017, 358, 234–238. [Google Scholar] [CrossRef]
- Pendaries, V.; Malaisse, J.; Pellerin, L.; Le Lamer, M.; Nachat, R.; Kezic, S.; Schmitt, A.M.; Paul, C.; Poumay, Y.; Serre, G.; et al. Knockdown of filaggrin in a three-dimensional reconstructed human epidermis impairs keratinocyte differentiation. J. Investig. Dermatol. 2014, 134, 2938–2946. [Google Scholar] [CrossRef]
- Bernerd, F.; Asselineau, D.; Vioux, C.; Chevallier-Lagente, O.; Bouadjar, B.; Sarasin, A.; Magnaldo, T. Clues to epidermal cancer proneness revealed by reconstruction of DNA repair-deficient xeroderma pigmentosum skin in vitro. Proc. Natl. Acad. Sci. USA 2001, 98, 7817–7822. [Google Scholar] [CrossRef]
- Fortunel, N.O.; Hatzfeld, J.A.; Rosemary, P.-A.; Ferraris, C.; Monier, M.-N.; Haydont, V.; Longuet, J.; Brethon, B.; Lim, B.; Castiel, I.; et al. Long-term expansion of human functional epidermal precursor cells: Promotion of extensive amplification by low TGF-beta1 concentrations. J. Cell Sci. 2003, 116, 4043–4052. [Google Scholar] [CrossRef]
- Fortunel, N.O.; Cadio, E.; Vaigot, P.; Chadli, L.; Moratille, S.; Bouet, S.; Roméo, P.-H.; Martin, M.T. Exploration of the functional hierarchy of the basal layer of human epidermis at the single-cell level using parallel clonal microcultures of keratinocytes. Exp. Dermatol. 2010, 19, 387–392. [Google Scholar] [CrossRef] [PubMed]
- Fortunel, N.O.; Chadli, L.; Coutier, J.; Lemaître, G.; Auvré, F.; Domingues, S.; Bouissou-Cadio, E.; Vaigot, P.; Cavallero, S.; Deleuze, J.-F.; et al. KLF4 inhibition promotes the expansion of keratinocyte precursors from adult human skin and of embryonic-stem-cell-derived keratinocytes. Nat. Biomed. Eng. 2019, 3, 985–997. [Google Scholar] [CrossRef] [PubMed]
- Larderet, G.; Fortunel, N.O.; Vaigot, P.; Cegalerba, M.; Maltère, P.; Zobiri, O.; Gidrol, X.; Waksman, G.; Martin, M.T. Human side population keratinocytes exhibit long-term proliferative potential and a specific gene expression profile and can form a pluristratified epidermis. Stem Cells 2006, 24, 965–974. [Google Scholar] [CrossRef] [PubMed]
- Kopp, M.; Loewe, T.; Wuest, W.; Brand, M.; Wetzl, M.; Nitsch, W.; Schmidt, D.; Beck, M.; Schmidt, B.; Uder, M.; et al. Individual calculation of effective dose and risk of malignancy based on Monte Carlo simulations after whole body computed tomography. Sci. Rep. 2020, 10, 9475. [Google Scholar] [CrossRef] [PubMed]
- Bernier, M.O.; Baysson, H.; Pearce, M.S.; Moissonnier, M.; Cardis, E.; Hauptmann, M.; Struelens, L.; Dabin, J.; Johansen, C.; Journy, N.; et al. Cohort profile: The EPI-CT study: A european pooled epidemiological study to quantify the risk of radiation-induced cancer from paediatric CT. Int. J. Epidemiol. 2019, 48, 379–381. [Google Scholar] [CrossRef]
- Martin, M.T.; Vulin, A.; Hendry, J.H. Human epidermal stem cells: Role in adverse skin reactions and carcinogenesis from radiation. Mutat. Res. 2016, 770, 349–368. [Google Scholar] [CrossRef]
- Sivan, V.; Vozenin-Brotons, M.-C.; Tricaud, Y.; Lefaix, J.-L.; Cosset, J.-M.; Dubray, B.; Martin, M.T. Altered proliferation and differentiation of human epidermis in cases of skin fibrosis after radiotherapy. Int. J. Radiat. Oncol. Biol. Phys. 2002, 53, 385–393. [Google Scholar] [CrossRef]
- Turesson, I.; Nyman, J.; Qvarnström, F.; Simonsson, M.; Book, M.; Hermansson, I.; Sigurdardottir, S.; Johansson, K.A. A low-dose hypersensitive keratinocyte loss in response to fractionated radiotherapy is associated with growth arrest and apoptosis. Radiother. Oncol. 2010, 94, 90–101. [Google Scholar] [CrossRef]
- Qvarnström, F.; Simonsson, M.; Nyman, J.; Hermansson, I.; Book, M.; Johansson, K.A.; Turesson, I. Double strand break induction and kinetics indicate preserved hypersensitivity in keratinocytes to subtherapeutic doses for 7weeks of radiotherapy. Radiother. Oncol. 2017, 122, 163–169. [Google Scholar]
- Alexaline, M.M.; Trouillas, M.; Nivet, M.; Bourreau, E.; Leclerc, T.; Duhamel, P.; Martin, M.T.; Doucet, C.; Fortunel, N.O.; Lataillade, J.-J. Bioengineering a human plasma-based epidermal substitute with efficient grafting capacity and high content in clonogenic cells. Stem Cells Transl. Med. 2015, 4, 643–654. [Google Scholar] [CrossRef]
- Fortunel, N.O.; Bouissou-Cadio, E.; Coutier, J.; Martin, M.T. Iterative three-dimensional epidermis bioengineering and xenografting to assess long-term regenerative potential in human keratinocyte precursor cells. Methods Mol. Biol. 2020, 2109, 155–167. [Google Scholar] [PubMed]
- Sasaki, M.; Kajiya, H.; Ozeki, S.; Okabe, K.; Ikebe, T. Reactive oxygen species promotes cellular senescence in normal human epidermal keratinocytes through epigenetic regulation of p16(INK4a.). Biochem. Biophys. Res. Commun. 2014, 452, 622–628. [Google Scholar] [CrossRef] [PubMed]
- Williams, B.B.; Cantrell, V.A.; Mundell, N.A.; Bennett, A.C.; Quick, R.E.; Jessen, J.R. VANGL2 regulates membrane trafficking of MMP14 to control cell polarity and migration. J. Cell Sci. 2012, 125, 2141–2147. [Google Scholar] [CrossRef] [PubMed]
- Nagaoka, T.; Inutsuka, A.; Begum, K.; Bin Hafiz, K.M.; Kishi, M. Vangl2 regulates E-cadherin in epithelial cells. Sci. Rep. 2014, 4, 6940. [Google Scholar] [CrossRef] [PubMed]
- Zhitnyak, I.Y.; Rubtsova, S.N.; Litovka, N.I.; Gloushankova, N.A. Early events in actin cytoskeleton dynamics and E-cadherin-mediated cell-cell adhesion during epithelial-mesenchymal transition. Cells 2020, 9, 578. [Google Scholar] [CrossRef]
- Martin, M.; Lefaix, J.; Delanian, S. TGF-beta1 and radiation fibrosis: A master switch and a specific therapeutic target? Int. J. Radiat. Oncol. Biol. Phys. 2000, 47, 277–290. [Google Scholar] [CrossRef]
- Reisdorf, P.; Lawrence, D.A.; Sivan, V.; Klising, E.; Martin, M.T. Alteration of transforming growth factor-beta1 response involves down-regulation of Smad3 signaling in myofibroblasts from skin fibrosis. Am. J. Pathol. 2001, 159, 263–272. [Google Scholar] [CrossRef]
- Mezentsev, A.; Amundson, S.A. Global gene expression responses to low- or high-dose radiation in a human three-dimensional tissue model. Radiat. Res. 2011, 175, 677–688. [Google Scholar] [CrossRef]
- Sugiyama, H.; Misumi, M.; Kishikawa, M.; Iseki, M.; Yonehara, S.; Hayashi, T.; Soda, M.; Tokuoka, S.; Shimizu, Y.; Sakata, R.; et al. Skin cancer incidence among atomic bomb survivors from 1958 to 1996. Radiat. Res. 2014, 181, 531–539. [Google Scholar] [CrossRef]
- Hirsch, T.; Rothoeft, T.; Teig, N.; Bauer, J.W.; Pellegrini, G.; De Rosa, L.; Scaglione, D.; Reichelt, J.; Klausegger, A.; Kneisz, D.; et al. Regeneration of the entire human epidermis using transgenic stem cells. Nature 2017, 551, 327–332. [Google Scholar] [CrossRef]
- Ronfard, V.; Rives, J.-M.; Neveux, Y.; Carsin, H.; Barrandon, Y. Long-term regeneration of human epidermis on third degree burns transplanted with autologous cultured epithelium grown on a fibrin matrix. Transplantation 2000, 70, 1588–1598. [Google Scholar] [CrossRef] [PubMed]
- Box, K.; Joyce, B.W.; Devenport, D. Epithelial geometry regulates spindle orientation and progenitor fate during formation of the mammalian epidermis. Elife 2019, 8, e47102. [Google Scholar] [CrossRef] [PubMed]
- Kimura-Yoshida, C.; Mochida, K.; Nakaya, M.A.; Mizutani, T.; Matsuo, I. Cytoplasmic localization of GRHL3 upon epidermal differentiation triggers cell shape change for epithelial morphogenesis. Nat. Commun. 2018, 9, 4059. [Google Scholar] [CrossRef] [PubMed]
- Caddy, J.; Wilanowski, T.; Darido, C.; Dworkin, S.; Ting, S.B.; Zhao, Q.; Rank, G.; Auden, A.; Srivastava, S.; Papenfuss, T.A.; et al. Epidermal wound repair is regulated by the planar cell polarity signaling pathway. Dev. Cell 2010, 19, 138–147. [Google Scholar] [CrossRef] [PubMed]
- Fontana, F.; Raimondi, M.; Marzagalli, M.; Sommariva, M.; Limonta, P.; Gagliano, N. Epithelial-to-mesenchymal transition markers and CD44 isoforms are differently expressed in 2D and 3D cell cultures of prostate cancer cells. Cells 2019, 8, 143. [Google Scholar] [CrossRef]
- Miro, C.; Di Cicco, E.; Ambrosio, R.; Mancino, G.; Di Girolamo, D.; Cicatiello, A.G.; Sagliocchi, S.; Nappi, A.; De Stefano, M.A.; Luongo, C.; et al. Thyroid hormone induces progression and invasiveness of squamous cell carcinomas by promoting a ZEB-1/E-cadherin switch. Nat. Commun. 2019, 10, 5410. [Google Scholar] [CrossRef]
- Svoboda, E. When skin’s defence against pollution fails. Nature 2018, 563, S89–S90. [Google Scholar] [CrossRef]
| Primary Antibodies | Blocking Reagents |
|---|---|
| Rabbit polyclonal anti-ZEB1 [H-102] (sc-25388, Santa Cruz, Heidelberg, Germany) | BSA |
| Rabbit polyclonal anti-involucrin (ab53112, Abcam, Paris, France) | BSA |
| Rabbit polyclonal anti-αSMA (ab5694, Abcam, Paris, France) | Serum |
| Mouse monoclonal anti-lamin 5 (ab78286, Abcam, Paris, France) | Diagomics, Blagnac, France |
| Mouse monoclonal anti-cytokeratin 14 [LL002] (Leica Biosystems, Nanterre, France) | BSA |
| Mouse monoclonal anti-βcatenin [15B8] (ab6301, Abcam, Paris, France) | Serum |
| Mouse monoclonal anti-E-cadherin [M168] (ab76055, Abcam, Paris, France) | Serum |
| Secondary Antibodies | |
| Goat anti-Mouse, Alexa Fluor®594 conjugate (A-11032, ThermoFisher scientific, Les Ulis, France) | |
| Goat anti-Mouse, Alexa Fluor®488 conjugate (A-11001, ThermoFisher scientific, Les Ulis, France) | |
| Goat anti-Rabbit, Alexa Fluor®594 conjugate (A-11037, ThermoFisher scientific, Les Ulis, France) | |
| Donkey anti-Rabbit, FITC conjugate (ab97063, Abcam, Paris, France) | |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cavallero, S.; Neves Granito, R.; Stockholm, D.; Azzolin, P.; Martin, M.T.; Fortunel, N.O. Exposure of Human Skin Organoids to Low Genotoxic Stress Can Promote Epithelial-to-Mesenchymal Transition in Regenerating Keratinocyte Precursor Cells. Cells 2020, 9, 1912. https://doi.org/10.3390/cells9081912
Cavallero S, Neves Granito R, Stockholm D, Azzolin P, Martin MT, Fortunel NO. Exposure of Human Skin Organoids to Low Genotoxic Stress Can Promote Epithelial-to-Mesenchymal Transition in Regenerating Keratinocyte Precursor Cells. Cells. 2020; 9(8):1912. https://doi.org/10.3390/cells9081912
Chicago/Turabian StyleCavallero, Sophie, Renata Neves Granito, Daniel Stockholm, Peggy Azzolin, Michèle T. Martin, and Nicolas O. Fortunel. 2020. "Exposure of Human Skin Organoids to Low Genotoxic Stress Can Promote Epithelial-to-Mesenchymal Transition in Regenerating Keratinocyte Precursor Cells" Cells 9, no. 8: 1912. https://doi.org/10.3390/cells9081912
APA StyleCavallero, S., Neves Granito, R., Stockholm, D., Azzolin, P., Martin, M. T., & Fortunel, N. O. (2020). Exposure of Human Skin Organoids to Low Genotoxic Stress Can Promote Epithelial-to-Mesenchymal Transition in Regenerating Keratinocyte Precursor Cells. Cells, 9(8), 1912. https://doi.org/10.3390/cells9081912







