The Ins and Outs of Cathepsins: Physiological Function and Role in Disease Management
Abstract
1. Introduction
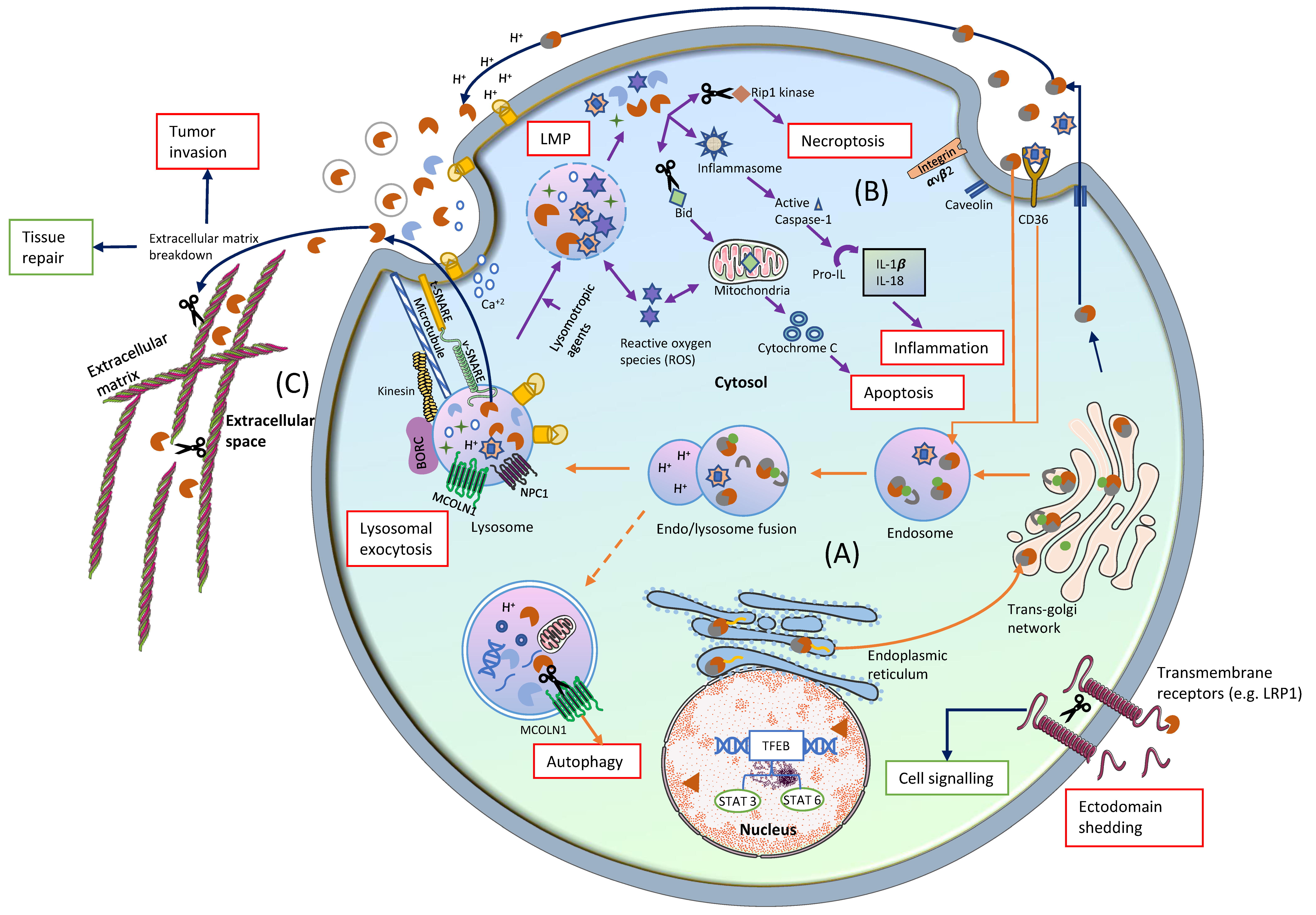
2. Cathepsins in Lysosomes
2.1. Regulation
2.2. Synthesis
M6P-Independent Sorting
2.3. Physiological Functions of Cathepsins in the Endo/Lysosomes
2.3.1. Immune Responses
2.3.2. Autophagy
2.3.3. Growth and Development Related Functions
2.4. Pathological Role of Cathepsins in the Endo/Lysosomes
3. Cathepsins in Cytosol
3.1. Mechanism of Translocation
3.1.1. Oxidative Stress-Induced LMP
3.1.2. Lysosomotropic Agents
3.2. Transport of Cathepsins to Different Regions of Cytosol
3.3. Physiological Functions of Cathepsins in the Cytosol
3.3.1. Apoptosis and Necroptosis
3.3.2. Inflammation
3.3.3. Functions of Nuclear Cathepsins
3.4. Pathological Functions of Cathepsins in the Cytosol
4. Cathepsins in the Extracellular Space
4.1. Mechanism of Translocation
4.1.1. Lysosomal Exocytosis of Cathepsins
4.1.2. Alternative Sorting of Cathepsins into Extracellular Space
4.2. Physiological Function of Extracellular Cathepsins
4.2.1. ECM Degradation
4.2.2. Functions of Cathepsins on the Plasma Membrane
4.2.3. Functions of Cathepsins in the Secretory Vesicles
4.3. Pathological Functions of Extracellular Cathepsins
4.3.1. Cancer
4.3.2. Metabolic Disorders
5. Targeting Cathepsins in Disease Management
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| AIF | Apoptosis-inducing factor |
| ATP | Adenosine triphosphate |
| Bcl-2 | B-cell lymphoma-2 |
| BORC | BLOC-one-related complex |
| CLN | Ceroid lipofuscinosis, neuronal |
| CNS | Central nervous system |
| CX3CL1 | C-X3-C Motif chemokine ligand 1 |
| ECM | Extracellular matrix |
| EGFR | Epidermal growth factor receptor |
| ER | Endoplasmic reticulum |
| IGF-I | Insulin-like growth factor-1 |
| IRES | Internal ribosomal entry site |
| LDL | Low-density lipoprotein |
| LDLR | Low-density lipoprotein receptor |
| LFA-1 | Lymphocyte function associated antigen-1 |
| LM | Lysosomal membrane |
| LMP | Lysosomal membrane permeabilization |
| LRP1 | Lipoprotein receptor-related protein 1 |
| LSD | Lysosomal storage disorders |
| MCOLN1 | Mucolipin 1 |
| MHC | Major histocompatibility complex |
| MSDH | O-methyl-serine dodecylamide hydrochloride |
| NASH | Non-alcoholic steatohepatitis |
| NAFLD | Non-alcoholic fatty liver disease |
| NCL | Neuronal ceroid lipofuscinosis |
| NF-Y | Nuclear factor |
| NPC | Niemann–Pick disease type C |
| OMIM | Online mendelian inheritance in man |
| oxLDL | oxidized low-density lipoprotein |
| PM | Plasma membrane |
| ROS | Reactive oxygen species |
| TAM | Tumor-associated macrophages |
| TFEB | Transcription factor EB |
| TGF | Transforming growth factor |
| TGN | Trans-Golgi network |
| TLR | Toll-like receptor |
| V-ATPase | vacuolar H+ ATPase |
| XIAP | X-linked inhibitor of apoptosis |
References
- Perera, R.M.; Zoncu, R. The Lysosome as a Regulatory Hub. Annu. Rev. Cell Dev. Biol. 2016, 32, 223–253. [Google Scholar] [CrossRef] [PubMed]
- De Duve, C. The lysosome turns fifty. Nat. Cell Biol. 2005, 7, 847–849. [Google Scholar] [CrossRef] [PubMed]
- Xu, H.; Ren, D. Lysosomal physiology. Annu. Rev. Physiol. 2015, 77, 57–80. [Google Scholar] [CrossRef]
- Lawrence, R.E.; Zoncu, R. The lysosome as a cellular centre for signalling, metabolism and quality control. Nat. Cell Biol. 2019, 21, 133–142. [Google Scholar] [CrossRef]
- Lim, C.Y.; Zoncu, R. The lysosome as a command-and-control center for cellular metabolism. J. Cell Biol. 2016, 214, 653–664. [Google Scholar] [CrossRef] [PubMed]
- Turk, V.; Stoka, V.; Vasiljeva, O.; Renko, M.; Sun, T.; Turk, B.; Turk, D. Cysteine cathepsins: From structure, function and regulation to new frontiers. Biochim. Biophys. Acta 2012, 1824, 68–88. [Google Scholar] [CrossRef]
- Vasiljeva, O.; Reinheckel, T.; Peters, C.; Turk, D.; Turk, V.; Turk, B. Emerging roles of cysteine cathepsins in disease and their potential as drug targets. Curr. Pharm. Des. 2007, 13, 387–403. [Google Scholar] [CrossRef]
- Brix, K.; Dunkhorst, A.; Mayer, K.; Jordans, S. Cysteine cathepsins: Cellular roadmap to different functions. Biochimie 2008, 90, 194–207. [Google Scholar] [CrossRef]
- Patel, S.; Homaei, A.; El-Seedi, H.R.; Akhtar, N. Cathepsins: Proteases that are vital for survival but can also be fatal. Biomed. Pharm. 2018, 105, 526–532. [Google Scholar] [CrossRef]
- Rossi, A.; Deveraux, Q.; Turk, B.; Sali, A. Comprehensive search for cysteine cathepsins in the human genome. Biol. Chem. 2004, 385, 363–372. [Google Scholar] [CrossRef]
- Sanman, L.E.; van der Linden, W.A.; Verdoes, M.; Bogyo, M. Bifunctional Probes of Cathepsin Protease Activity and pH Reveal Alterations in Endolysosomal pH during Bacterial Infection. Cell Chem. Biol. 2016, 23, 793–804. [Google Scholar] [CrossRef]
- Bromme, D.; Bonneau, P.R.; Lachance, P.; Wiederanders, B.; Kirschke, H.; Peters, C.; Thomas, D.Y.; Storer, A.C.; Vernet, T. Functional expression of human cathepsin S in Saccharomyces cerevisiae. Purification and characterization of the recombinant enzyme. J. Biol. Chem. 1993, 268, 4832–4838. [Google Scholar] [PubMed]
- Kirschke, H.; Schmidt, I.; Wiederanders, B.; Cathepsin, S. The cysteine proteinase from bovine lymphoid tissue is distinct from cathepsin L (EC 3.4.22.15). Biochem. J. 1986, 240, 455–459. [Google Scholar] [CrossRef]
- Sapolsky, A.I.; Howell, D.S.; Woessner, J.F., Jr. Neutral proteases and cathepsin D in human articular cartilage. J. Clin. Investig. 1974, 53, 1044–1053. [Google Scholar] [CrossRef] [PubMed]
- Naseem, R.H.; Hedegard, W.; Henry, T.D.; Lessard, J.; Sutter, K.; Katz, S.A. Plasma cathepsin D isoforms and their active metabolites increase after myocardial infarction and contribute to plasma renin activity. Basic Res. Cardiol. 2005, 100, 139–146. [Google Scholar] [CrossRef] [PubMed]
- Vidak, E.; Javorsek, U.; Vizovisek, M.; Turk, B. Cysteine Cathepsins and their Extracellular Roles: Shaping the Microenvironment. Cells 2019, 8, 264. [Google Scholar] [CrossRef] [PubMed]
- Vizovisek, M.; Fonovic, M.; Turk, B. Cysteine cathepsins in extracellular matrix remodeling: Extracellular matrix degradation and beyond. Matrix Biol. 2019, 75–76, 141–159. [Google Scholar] [CrossRef]
- Reiser, J.; Adair, B.; Reinheckel, T. Specialized roles for cysteine cathepsins in health and disease. J. Clin. Investig. 2010, 120, 3421–3431. [Google Scholar] [CrossRef]
- Stoka, V.; Turk, V.; Turk, B. Lysosomal cathepsins and their regulation in aging and neurodegeneration. Ageing Res. Rev. 2016, 32, 22–37. [Google Scholar] [CrossRef]
- Vidoni, C.; Follo, C.; Savino, M.; Melone, M.A.; Isidoro, C. The Role of Cathepsin D in the Pathogenesis of Human Neurodegenerative Disorders. Med. Res. Rev. 2016, 36, 845–870. [Google Scholar] [CrossRef]
- Cermak, S.; Kosicek, M.; Mladenovic-Djordjevic, A.; Smiljanic, K.; Kanazir, S.; Hecimovic, S. Loss of Cathepsin B and L Leads to Lysosomal Dysfunction, NPC-Like Cholesterol Sequestration and Accumulation of the Key Alzheimer’s Proteins. PLoS ONE 2016, 11, e0167428. [Google Scholar] [CrossRef]
- Pislar, A.; Kos, J. Cysteine Cathepsins in Neurological Disorders. Mol. Neurobiol. 2014, 49, 1017–1030. [Google Scholar] [CrossRef]
- Nagai, A.; Murakawa, Y.; Terashima, M.; Shimode, K.; Umegae, N.; Takeuchi, H.; Kobayashi, S. Cystatin C and cathepsin B in CSF from patients with inflammatory neurologic diseases. Neurology 2000, 55, 1828–1832. [Google Scholar] [CrossRef] [PubMed]
- Lutgens, S.P.M.; Cleutjens, K.B.J.M.; Daemen, M.J.A.P.; Heeneman, S. Cathepsin cysteine proteases in cardiovascular disease. FASEB J. 2007, 21, 3029–3041. [Google Scholar] [CrossRef] [PubMed]
- Mohamed, M.M.; Sloane, B.F. Cysteine cathepsins: Multifunctional enzymes in cancer. Nat. Rev. Cancer 2006, 6, 764–775. [Google Scholar] [CrossRef] [PubMed]
- Naour, N.; Rouault, C.; Fellahi, S.; Lavoie, M.E.; Poitou, C.; Keophiphath, M.; Eberle, D.; Shoelson, S.; Rizkalla, S.; Bastard, J.P.; et al. Cathepsins in Human Obesity: Changes in Energy Balance Predominantly Affect Cathepsin S in Adipose Tissue and in Circulation. J. Clin. Endocrinol. Metab. 2010, 95, 1861–1868. [Google Scholar] [CrossRef] [PubMed]
- Turk, B.; Turk, D.; Turk, V. Lysosomal cysteine proteases: More than scavengers. Biochim. Biophys. Acta 2000, 1477, 98–111. [Google Scholar] [CrossRef]
- Jean, D.; Guillaume, N.; Frade, R. Characterization of human cathepsin L promoter and identification of binding sites for NF-Y, Sp1 and Sp3 that are essential for its activity. Biochem. J. 2002, 361, 173–184. [Google Scholar] [CrossRef]
- Yan, S.; Berquin, I.M.; Troen, B.R.; Sloane, B.F. Transcription of human cathepsin B is mediated by Sp1 and Ets family factors in glioma. DNA Cell Biol. 2000, 19, 79–91. [Google Scholar] [CrossRef]
- Nakken, B.; Varga, T.; Szatmari, I.; Szeles, L.; Gyongyosi, A.; Illarionov, P.A.; Dezso, B.; Gogolak, P.; Rajnavolgyi, E.; Nagy, L. Peroxisome Proliferator-Activated Receptor gamma-Regulated Cathepsin D Is Required for Lipid Antigen Presentation by Dendritic Cells. J. Immunol. 2011, 187, 240–247. [Google Scholar] [CrossRef]
- Cavailles, V.; Augereau, P.; Rochefort, H. Cathepsin D gene is controlled by a mixed promoter, and estrogens stimulate only TATA-dependent transcription in breast cancer cells. Proc. Natl. Acad. Sci. USA 1993, 90, 203–207. [Google Scholar] [CrossRef]
- Berquin, I.M.; Sloane, B.F. Cathepsin B expression in human tumors. Adv. Exp. Med. Biol. 1996, 389, 281–294. [Google Scholar] [CrossRef]
- Mittal, S.; Mir, R.A.; Chauhan, S.S. Post-transcriptional regulation of human cathepsin L expression. Biol. Chem. 2011, 392, 405–413. [Google Scholar] [CrossRef] [PubMed]
- Braulke, T.; Bonifacino, J.S. Sorting of lysosomal proteins. BBA-Mol. Cell Res. 2009, 1793, 605–614. [Google Scholar] [CrossRef]
- Coutinho, M.F.; Prata, M.J.; Alves, S. Mannose-6-phosphate pathway: A review on its role in lysosomal function and dysfunction. Mol. Genet. Metab. 2012, 105, 542–550. [Google Scholar] [CrossRef]
- Verma, S.; Dixit, R.; Pandey, K.C. Cysteine Proteases: Modes of Activation and Future Prospects as Pharmacological Targets. Front. Pharm. 2016, 7, 107. [Google Scholar] [CrossRef] [PubMed]
- Roberts, R. Lysosomal cysteine proteases: Structure, function and inhibition of cathepsins. Drug News Perspect. 2005, 10, 605–614. [Google Scholar] [CrossRef]
- Laurent-Matha, V.; Derocq, D.; Prebois, C.; Katunuma, N.; Liaudet-Coopman, E. Processing of human cathepsin D is independent of its catalytic function and auto-activation: Involvement of cathepsins L and B. J. Biochem. 2006, 139, 363–371. [Google Scholar] [CrossRef] [PubMed]
- Follo, C.; Castino, R.; Nicotra, G.; Trincheri, N.F.; Isidoro, C. Folding, activity and targeting of mutated human cathepsin D that cannot be processed into the double-chain form. Int. J. Biochem. Cell Biol. 2007, 39, 638–649. [Google Scholar] [CrossRef]
- Canuel, M.; Korkidakis, A.; Konnyu, K.; Morales, C.R. Sortilin mediates the lysosomal targeting of cathepsins D and H. Biochem. Biophys. Res. Commun. 2008, 373, 292–297. [Google Scholar] [CrossRef] [PubMed]
- Derocq, D.; Prebois, C.; Beaujouin, M.; Laurent-Matha, V.; Pattingre, S.; Smith, G.K.; Liaudet-Coopman, E. Cathepsin D is partly endocytosed by the LRP1 receptor and inhibits LRP1-regulated intramembrane proteolysis. Oncogene 2012, 31, 3202–3212. [Google Scholar] [CrossRef] [PubMed]
- Markmann, S.; Thelen, M.; Cornils, K.; Schweizer, M.; Brocke-Ahmadinejad, N.; Willnow, T.; Heeren, J.; Gieselmann, V.; Braulke, T.; Kollmann, K. Lrp1/LDL Receptor Play Critical Roles in Mannose 6-Phosphate-Independent Lysosomal Enzyme Targeting. Traffic 2015, 16, 743–759. [Google Scholar] [CrossRef]
- Castino, R.; Isidoro, C. The transport of soluble lysosomal hydrolases from the Golgi complex to lysosomes. In The Golgi Apparatus; Springer: Vienna, Austria, 2008; pp. 402–413. [Google Scholar] [CrossRef]
- Gopalakrishnan, M.M.; Grosch, H.W.; Locatelli-Hoops, S.; Werth, N.; Smolenova, E.; Nettersheim, M.; Sandhoff, K.; Hasilik, A. Purified recombinant human prosaposin forms oligomers that bind procathepsin D and affect its autoactivation. Biochem. J. 2004, 383, 507–515. [Google Scholar] [CrossRef]
- Tanaka, Y.; Tanaka, R.; Kawabata, T.; Noguchi, Y.; Himeno, M. Lysosomal cysteine protease, cathepsin B, is targeted to lysosomes by the mannose 6-phosphate-independent pathway in rat hepatocytes: Site-specific phosphorylation in oligosaccharides of the proregion. J. Biochem. 2000, 128, 39–48. [Google Scholar] [CrossRef]
- Boonen, M.; Staudt, C.; Gilis, F.; Oorschot, V.; Klumperman, J.; Jadot, M. Cathepsin D and its newly identified transport receptor SEZ6L2 can modulate neurite outgrowth. J. Cell Sci. 2016, 129, 557–568. [Google Scholar] [CrossRef]
- Bright, N.A.; Davis, L.J.; Luzio, J.P. Endolysosomes Are the Principal Intracellular Sites of Acid Hydrolase Activity. Curr. Biol. 2016, 26, 2233–2245. [Google Scholar] [CrossRef] [PubMed]
- Creasy, B.M.; McCoy, K.L. Cytokines regulate cysteine cathepsins during TLR responses. Cell. Immunol. 2011, 267, 56–66. [Google Scholar] [CrossRef] [PubMed]
- Bird, P.I.; Trapani, J.A.; Villadangos, J.A. Endolysosomal proteases and their inhibitors in immunity. Nat. Rev. Immunol. 2009, 9, 871–882. [Google Scholar] [CrossRef] [PubMed]
- Guncar, G.; Pungercic, G.; Klemencic, I.; Turk, V.; Turk, D. Crystal structure of MHC class II-associated p41 Ii fragment bound to cathepsin L reveals the structural basis for differentiation between cathepsins L and S. EMBO J. 1999, 18, 793–803. [Google Scholar] [CrossRef]
- Deussing, J.; Roth, W.; Saftig, P.; Peters, C.; Ploegh, H.L.; Villadangos, J.A. Cathepsins B and D are dispensable for major histocompatibility complex class II-mediated antigen presentation. Proc. Natl. Acad. Sci. USA 1998, 95, 4516–4521. [Google Scholar] [CrossRef]
- Ward, C.; Martinez-Lopez, N.; Otten, E.G.; Carroll, B.; Maetzel, D.; Singh, R.; Sarkar, S.; Korolchuk, V.I. Autophagy, lipophagy and lysosomal lipid storage disorders. Biochim. Biophys. Acta 2016, 1861, 269–284. [Google Scholar] [CrossRef]
- Dennemarker, J.; Lohmuller, T.; Muller, S.; Aguilar, S.V.; Tobin, D.J.; Peters, C.; Reinheckel, T. Impaired turnover of autophagolysosomes in cathepsin L deficiency. Biol. Chem. 2010, 391, 913–922. [Google Scholar] [CrossRef]
- Man, S.M.; Kanneganti, T.D. Regulation of lysosomal dynamics and autophagy by CTSB/cathepsin B. Autophagy 2016, 12, 2504–2505. [Google Scholar] [CrossRef]
- Yang, M.; Liu, J.; Shao, J.; Qin, Y.; Ji, Q.; Zhang, X.; Du, J. Cathepsin S-mediated autophagic flux in tumor-associated macrophages accelerate tumor development by promoting M2 polarization. Mol. Cancer 2014, 13, 43. [Google Scholar] [CrossRef] [PubMed]
- Authier, F.; Kouach, M.; Briand, G. Endosomal proteolysis of insulin-like growth factor-I at its C-terminal D-domain by cathepsin B. FEBS Lett. 2005, 579, 4309–4316. [Google Scholar] [CrossRef]
- Guha, S.; Padh, H. Cathepsins: Fundamental effectors of endolysosomal proteolysis. Indian J. Biochem. Biophys. 2008, 45, 75–90. [Google Scholar]
- Kovsan, J.; Ben-Romano, R.; Souza, S.C.; Greenberg, A.S.; Rudich, A. Regulation of adipocyte lipolysis by degradation of the perilipin protein: Nelfinavir enhances lysosome-mediated perilipin proteolysis. J. Biol. Chem. 2007, 282, 21704–21711. [Google Scholar] [CrossRef] [PubMed]
- Robker, R.L.; Russell, D.L.; Espey, L.L.; Lydon, J.P.; O’Malley, B.W.; Richards, J.S. Progesterone-regulated genes in the ovulation process: ADAMTS-1 and cathepsin L proteases. Proc. Natl. Acad. Sci. USA 2000, 97, 4689–4694. [Google Scholar] [CrossRef]
- Follo, C.; Ozzano, M.; Mugoni, V.; Castino, R.; Santoro, M.; Isidoro, C. Knock-down of cathepsin D affects the retinal pigment epithelium, impairs swim-bladder ontogenesis and causes premature death in zebrafish. PLoS ONE 2011, 6, e21908. [Google Scholar] [CrossRef] [PubMed]
- Saftig, P.; Hetman, M.; Schmahl, W.; Weber, K.; Heine, L.; Mossmann, H.; Koster, A.; Hess, B.; Evers, M.; von Figura, K.; et al. Mice deficient for the lysosomal proteinase cathepsin D exhibit progressive atrophy of the intestinal mucosa and profound destruction of lymphoid cells. EMBO J. 1995, 14, 3599–3608. [Google Scholar] [CrossRef]
- Felbor, U.; Kessler, B.; Mothes, W.; Goebel, H.H.; Ploegh, H.L.; Bronson, R.T.; Olsen, B.R. Neuronal loss and brain atrophy in mice lacking cathepsins B and L. Proc. Natl. Acad. Sci. USA 2002, 99, 7883–7888. [Google Scholar] [CrossRef]
- Ketterer, S.; Gomez-Auli, A.; Hillebrand, L.E.; Petrera, A.; Ketscher, A.; Reinheckel, T. Inherited diseases caused by mutations in cathepsin protease genes. FEBS J. 2017, 284, 1437–1454. [Google Scholar] [CrossRef]
- Siintola, E.; Partanen, S.; Stromme, P.; Haapanen, A.; Haltia, M.; Maehlen, J.; Lehesjoki, A.E.; Tyynela, J. Cathepsin D deficiency underlies congenital human neuronal ceroid-lipofuscinosis. Brain 2006, 129, 1438–1445. [Google Scholar] [CrossRef] [PubMed]
- Hart, T.C.; Hart, P.S.; Bowden, D.W.; Michalec, M.D.; Callison, S.A.; Walker, S.J.; Zhang, Y.Z.; Firatli, E. Mutations of the cathepsin C gene are responsible for Papillon-Lefevre syndrome. J. Med. Genet. 1999, 36, 881–887. [Google Scholar]
- Ho, N.; Punturieri, A.; Wilkin, D.; Szabo, J.; Johnson, M.; Whaley, J.; Davis, J.; Clark, A.; Weiss, S.; Francomano, C. Mutations of CTSK result in pycnodysostosis via a reduction in cathepsin K protein. J. Bone Min. Res. 1999, 14, 1649–1653. [Google Scholar] [CrossRef] [PubMed]
- Mizunoe, Y.; Sudo, Y.; Okita, N.; Hiraoka, H.; Mikami, K.; Narahara, T.; Negishi, A.; Yoshida, M.; Higashibata, R.; Watanabe, S.; et al. Involvement of lysosomal dysfunction in autophagosome accumulation and early pathologies in adipose tissue of obese mice. Autophagy 2017, 13, 642–653. [Google Scholar] [CrossRef] [PubMed]
- Tatti, M.; Motta, M.; Di Bartolomeo, S.; Scarpa, S.; Cianfanelli, V.; Cecconi, F.; Salvioli, R. Reduced cathepsins B and D cause impaired autophagic degradation that can be almost completely restored by overexpression of these two proteases in Sap C-deficient fibroblasts. Hum. Mol. Genet. 2012, 21, 5159–5173. [Google Scholar] [CrossRef] [PubMed]
- McGlinchey, R.P.; Lee, J.C. Cysteine cathepsins are essential in lysosomal degradation of alpha-synuclein. Proc. Natl. Acad. Sci. USA 2015, 112, 9322–9327. [Google Scholar] [CrossRef] [PubMed]
- Carnevali, O.; Cionna, C.; Tosti, L.; Lubzens, E.; Maradonna, F. Role of cathepsins in ovarian follicle growth and maturation. Gen. Comp. Endocrinol. 2006, 146, 195–203. [Google Scholar] [CrossRef]
- Tsukuba, T.; Yamamoto, K. [Atopic dermatitis and cathepsin E]. Nihon Yakurigaku Zasshi 2003, 122, 15–20. [Google Scholar] [CrossRef][Green Version]
- Adkison, A.M.; Raptis, S.Z.; Kelley, D.G.; Pham, C.T. Dipeptidyl peptidase I activates neutrophil-derived serine proteases and regulates the development of acute experimental arthritis. J. Clin. Investig. 2002, 109, 363–371. [Google Scholar] [CrossRef]
- Hao, L.; Zhu, G.; Lu, Y.; Wang, M.; Jules, J.; Zhou, X.; Chen, W. Deficiency of cathepsin K prevents inflammation and bone erosion in rheumatoid arthritis and periodontitis and reveals its shared osteoimmune role. FEBS Lett. 2015, 589, 1331–1339. [Google Scholar] [CrossRef]
- Ho, C.M.; Ho, S.L.; Jeng, Y.M.; Lai, Y.S.; Chen, Y.H.; Lu, S.C.; Chen, H.L.; Chang, P.Y.; Hu, R.H.; Lee, P.H. Accumulation of free cholesterol and oxidized low-density lipoprotein is associated with portal inflammation and fibrosis in nonalcoholic fatty liver disease. J. Inflamm. (Lond.) 2019, 16, 7. [Google Scholar] [CrossRef]
- Kos, J.; Jevnikar, Z.; Obermajer, N. The role of cathepsin X in cell signaling. Cell Adhes. Migr. 2009, 3, 164–166. [Google Scholar] [CrossRef] [PubMed]
- Kiuchi, S.; Tomaru, U.; Ishizu, A.; Imagawa, M.; Kiuchi, T.; Iwasaki, S.; Suzuki, A.; Otsuka, N.; Deguchi, T.; Shimizu, T.; et al. Expression of cathepsins V and S in thymic epithelial tumors. Hum. Pathol. 2017, 60, 66–74. [Google Scholar] [CrossRef] [PubMed]
- Wex, T.; Buhling, F.; Wex, H.; Gunther, D.; Malfertheiner, P.; Weber, E.; Bromme, D. Human cathepsin W, a cysteine protease predominantly expressed in NK cells, is mainly localized in the endoplasmic reticulum. J. Immunol. 2001, 167, 2172–2178. [Google Scholar] [CrossRef] [PubMed]
- Santamaria, I.; Velasco, G.; Pendas, A.M.; Fueyo, A.; Lopez-Otin, C. Cathepsin Z, a novel human cysteine proteinase with a short propeptide domain and a unique chromosomal location. J. Biol. Chem. 1998, 273, 16816–16823. [Google Scholar] [CrossRef] [PubMed]
- Carlsson, S.R.; Roth, J.; Piller, F.; Fukuda, M. Isolation and Characterization of Human Lysosomal Membrane-Glycoproteins, H-Lamp-1 and H-Lamp-2-Major Sialoglycoproteins Carrying Polylactosaminoglycan. J. Biol. Chem. 1988, 263, 18911–18919. [Google Scholar] [PubMed]
- Boya, P.; Kroemer, G. Lysosomal membrane permeabilization in cell death. Oncogene 2008, 27, 6434–6451. [Google Scholar] [CrossRef] [PubMed]
- Kurz, T.; Terman, A.; Gustafsson, B.; Brunk, U.T. Lysosomes in iron metabolism, ageing and apoptosis. Histochem. Cell Biol. 2008, 129, 389–406. [Google Scholar] [CrossRef]
- Kowanko, I.C.; Ferrante, A. Stimulation of neutrophil respiratory burst and lysosomal enzyme release by human interferon-gamma. Immunology 1987, 62, 149–151. [Google Scholar]
- Hoppe, G.; O’Neil, J.; Hoff, H.F.; Sears, J. Products of lipid peroxidation induce missorting of the principal lysosomal protease in retinal pigment epithelium. Biochim. Biophys. Acta 2004, 1689, 33–41. [Google Scholar] [CrossRef]
- Yuan, X.M.; Li, W.; Olsson, A.G.; Brunk, U.T. The toxicity to macrophages of oxidized low-density lipoprotein is mediated through lysosomal damage. Atherosclerosis 1997, 133, 153–161. [Google Scholar] [CrossRef]
- Turrens, J.F. Mitochondrial formation of reactive oxygen species. J. Physiol. 2003, 552, 335–344. [Google Scholar] [CrossRef] [PubMed]
- Aits, S.; Jaattela, M. Lysosomal cell death at a glance. J. Cell Sci. 2013, 126, 1905–1912. [Google Scholar] [CrossRef] [PubMed]
- Miller, D.K.; Griffiths, E.; Lenard, J.; Firestone, R.A. Cell killing by lysosomotropic detergents. J. Cell Biol. 1983, 97, 1841–1851. [Google Scholar] [CrossRef]
- Deamera, D.W.; Bramhall, J. Permeability of lipid bilayers to water and ionic solutes. In Chemistry and Physics of Lipids; Elsevier Scientific Publishers Ireland Ltd.: Shannon, Ireland, 1986; Volume 40, pp. 167–188. [Google Scholar]
- Deng, D.; Jiang, N.; Hao, S.J.; Sun, H.; Zhang, G.J. Loss of membrane cholesterol influences lysosomal permeability to potassium ions and protons. Biochim. Biophys. Acta 2009, 1788, 470–476. [Google Scholar] [CrossRef]
- Yi, Y.P.; Wang, X.; Zhang, G.; Fu, T.S.; Zhang, G.J. Phosphatidic acid osmotically destabilizes lysosomes through increased permeability to K+ and H+. Gen. Physiol. Biophys. 2006, 25, 149–160. [Google Scholar] [PubMed]
- Goulet, B.; Baruch, A.; Moon, N.S.; Poirier, M.; Sansregret, L.L.; Erickson, A.; Bogyo, M.; Nepveu, A. A cathepsin L isoform that is devoid of a signal peptide localizes to the nucleus in S phase and processes the CDP/Cux transcription factor. Mol. Cell 2004, 14, 207–219. [Google Scholar] [CrossRef]
- Muntener, K.; Zwicky, R.; Csucs, G.; Rohrer, J.; Baici, A. Exon skipping of cathepsin B: Mitochondrial targeting of a lysosomal peptidase provokes cell death. J. Biol. Chem. 2004, 279, 41012–41017. [Google Scholar] [CrossRef]
- Bestvater, F.; Dallner, C.; Spiess, E. The C-terminal subunit of artificially truncated human cathepsin B mediates its nuclear targeting and contributes to cell viability. BMC Cell Biol. 2005, 6, 16. [Google Scholar] [CrossRef]
- Elmore, S. Apoptosis: A review of programmed cell death. Toxicol. Pathol. 2007, 35, 495–516. [Google Scholar] [CrossRef]
- Gupta, S. Molecular steps of death receptor and mitochondrial pathways of apoptosis. Life Sci. 2001, 69, 2957–2964. [Google Scholar] [CrossRef]
- Chwieralski, C.E.; Welte, T.; Buhling, F. Cathepsin-regulated apoptosis. Apoptosis 2006, 11, 143–149. [Google Scholar] [CrossRef] [PubMed]
- Droga-Mazovec, G.; Bojic, L.; Petelin, A.; Ivanova, S.; Romih, R.; Repnik, U.; Salvesen, G.S.; Stoka, V.; Turk, V.; Turk, B. Cysteine cathepsins trigger caspase-dependent cell death through cleavage of Bid and antiapoptotic Bcl-2 homologues. J. Biol. Chem. 2008, 283, 19140–19150. [Google Scholar] [CrossRef] [PubMed]
- Conus, S.; Perozzo, R.; Reinheckel, T.; Peters, C.; Scapozza, L.; Yousefi, S.; Simon, H.U. Caspase-8 is activated by cathepsin D initiating neutrophil apoptosis during the resolution of inflammation. J. Exp. Med. 2008, 205, 685–698. [Google Scholar] [CrossRef] [PubMed]
- Bidere, N.; Lorenzo, H.K.; Carmona, S.; Laforge, M.; Harper, F.; Dumont, C.; Senik, A. Cathepsin D triggers Bax activation, resulting in selective apoptosis-inducing factor (AIF) relocation in T lymphocytes entering the early commitment phase to apoptosis. J. Biol. Chem. 2003, 278, 31401–31411. [Google Scholar] [CrossRef]
- Johansson, A.C.; Steen, H.; Ollinger, K.; Roberg, K. Cathepsin D mediates cytochrome c release and caspase activation in human fibroblast apoptosis induced by staurosporine. Cell Death Differ. 2003, 10, 1253–1259. [Google Scholar] [CrossRef]
- McComb, S.; Shutinoski, B.; Thurston, S.; Cessford, E.; Kumar, K.; Sad, S. Cathepsins Limit Macrophage Necroptosis through Cleavage of Rip1 Kinase. J. Immunol. 2014, 192, 5671–5678. [Google Scholar] [CrossRef]
- Kelley, N.; Jeltema, D.; Duan, Y.; He, Y. The NLRP3 Inflammasome: An Overview of Mechanisms of Activation and Regulation. Int. J. Mol. Sci. 2019, 20, 3328. [Google Scholar] [CrossRef]
- Campden, R.I.; Zhang, Y. The role of lysosomal cysteine cathepsins in NLRP3 inflammasome activation. Arch. Biochem. Biophys. 2019, 670, 32–42. [Google Scholar] [CrossRef]
- Li, S.L.; Du, L.L.; Zhang, L.; Hu, Y.; Xia, W.C.; Wu, J.; Zhu, J.; Chen, L.L.; Zhu, F.Q.; Li, C.X.; et al. Cathepsin B Contributes to Autophagy-related 7 (Atg7)-induced Nod-like Receptor 3 (NLRP3)-dependent Proinflammatory Response and Aggravates Lipotoxicity in Rat Insulinoma Cell Line. J. Biol. Chem. 2013, 288, 30094–30104. [Google Scholar] [CrossRef]
- Niemi, K.; Teirila, L.; Lappalainen, J.; Rajamaki, K.; Baumann, M.H.; Oorni, K.; Wolff, H.; Kovanen, P.T.; Matikainen, S.; Eklund, K.K. Serum amyloid A activates the NLRP3 inflammasome via P2X7 receptor and a cathepsin B-sensitive pathway. J. Immunol. 2011, 186, 6119–6128. [Google Scholar] [CrossRef]
- Orlowski, G.M.; Colbert, J.D.; Sharma, S.; Bogyo, M.; Robertson, S.A.; Rock, K.L. Multiple Cathepsins Promote Pro-IL-1beta Synthesis and NLRP3-Mediated IL-1beta Activation. J. Immunol. 2015, 195, 1685–1697. [Google Scholar] [CrossRef] [PubMed]
- Soond, S.M.; Kozhevnikova, M.V.; Frolova, A.S.; Savvateeva, L.V.; Plotnikov, E.Y.; Townsend, P.A.; Han, Y.P.; Zamyatnin, A.A., Jr. Lost or Forgotten: The nuclear cathepsin protein isoforms in cancer. Cancer Lett. 2019, 462, 43–50. [Google Scholar] [CrossRef]
- Tamhane, T.; Illukkumbura, R.; Lu, S.Y.; Maelandsmo, G.M.; Haugen, M.H.; Brix, K. Nuclear cathepsin L activity is required for cell cycle progression of colorectal carcinoma cells. Biochimie 2016, 122, 208–218. [Google Scholar] [CrossRef] [PubMed]
- Bach, A.S.; Derocq, D.; Laurent-Matha, V.; Montcourrier, P.; Sebti, S.; Orsetti, B.; Theillet, C.; Gongora, C.; Pattingre, S.; Ibing, E.; et al. Nuclear cathepsin D enhances TRPS1 transcriptional repressor function to regulate cell cycle progression and transformation in human breast cancer cells. Oncotarget 2015, 6, 28084–28103. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Zhou, Y.; Yu, X.; Huang, Q.; Fang, W.; Li, J.; Bonventre, J.V.; Sukhova, G.K.; Libby, P.; Shi, G.P. Differential Roles of Cysteinyl Cathepsins in TGF-beta Signaling and Tissue Fibrosis. iScience 2019, 19, 607–622. [Google Scholar] [CrossRef]
- Leist, M.; Jaattela, M. Triggering of apoptosis by cathepsins. Cell Death Differ. 2001, 8, 324–326. [Google Scholar] [CrossRef]
- Thibeaux, S.; Siddiqi, S.; Zhelyabovska, O.; Moinuddin, F.; Masternak, M.M.; Siddiqi, S.A. Cathepsin B regulates hepatic lipid metabolism by cleaving liver fatty acid-binding protein. J. Biol. Chem. 2018, 293, 1910–1923. [Google Scholar] [CrossRef]
- Roberts, L.R.; Kurosawa, H.; Bronk, S.F.; Fesmier, P.J.; Agellon, L.B.; Leung, W.Y.; Mao, F.; Gores, G.J. Cathepsin B contributes to bile salt-induced apoptosis of rat hepatocytes. Gastroenterology 1997, 113, 1714–1726. [Google Scholar] [CrossRef]
- Agostini, M.; Tucci, P.; Melino, G. Cell death pathology: Perspective for human diseases. Biochem. Biophys. Res. Commun. 2011, 414, 451–455. [Google Scholar] [CrossRef] [PubMed]
- Mattson, M.P. Neuronal life-and-death signaling, apoptosis, and neurodegenerative disorders. Antioxid. Redox Signal. 2006, 8, 1997–2006. [Google Scholar] [CrossRef]
- Lin, Y.; Epstein, D.L.; Liton, P.B. Intralysosomal iron induces lysosomal membrane permeabilization and cathepsin D-mediated cell death in trabecular meshwork cells exposed to oxidative stress. Investig. Ophthalmol. Vis. Sci. 2010, 51, 6483–6495. [Google Scholar] [CrossRef] [PubMed]
- Sato, Y.; Suzuki, Y.; Ito, E.; Shimazaki, S.; Ishida, M.; Yamamoto, T.; Yamamoto, H.; Toda, T.; Suzuki, M.; Suzuki, A.; et al. Identification and characterization of an increased glycoprotein in aging: Age-associated translocation of cathepsin D. Mech. Ageing Dev. 2006, 127, 771–778. [Google Scholar] [CrossRef] [PubMed]
- Jung, H.; Lee, E.Y.; Lee, S.I. Age-related changes in ultrastructural features of cathepsin B- and D-containing neurons in rat cerebral cortex. Brain Res. 1999, 844, 43–54. [Google Scholar] [CrossRef]
- Yu, J.W.; Lee, M.S. Mitochondria and the NLRP3 inflammasome: Physiological and pathological relevance. Arch. Pharm. Res. 2016, 39, 1503–1518. [Google Scholar] [CrossRef]
- Cocchiaro, P.; De Pasquale, V.; Della Morte, R.; Tafuri, S.; Avallone, L.; Pizard, A.; Moles, A.; Pavone, L.M. The Multifaceted Role of the Lysosomal Protease Cathepsins in Kidney Disease. Front. Cell Dev. Biol. 2017, 5, 114. [Google Scholar] [CrossRef]
- Sever, S.; Altintas, M.M.; Nankoe, S.R.; Moller, C.C.; Ko, D.; Wei, C.L.; Henderson, J.; del Re, E.C.; Hsing, L.; Erickson, A.; et al. Proteolytic processing of dynamin by cytoplasmic cathepsin L is a mechanism for proteinuric kidney disease. J. Clin. Investig. 2007, 117, 2095–2104. [Google Scholar] [CrossRef]
- Maubach, G.; Lim, M.C.; Zhuo, L. Nuclear cathepsin F regulates activation markers in rat hepatic stellate cells. Mol. Biol. Cell 2008, 19, 4238–4248. [Google Scholar] [CrossRef]
- Baici, A.; Muntener, K.; Willimann, A.; Zwicky, R. Regulation of human cathepsin B by alternative mRNA splicing: Homeostasis, fatal errors and cell death. Biol. Chem. 2006, 387, 1017–1021. [Google Scholar] [CrossRef]
- Olson, O.C.; Joyce, J.A. Cysteine cathepsin proteases: Regulators of cancer progression and therapeutic response. Nat. Rev. Cancer 2015, 15, 712–729. [Google Scholar] [CrossRef]
- Samie, M.A.; Xu, H. Lysosomal exocytosis and lipid storage disorders. J. Lipid Res. 2014, 55, 995–1009. [Google Scholar] [CrossRef]
- Guardia, C.M.; Farias, G.G.; Jia, R.; Pu, J.; Bonifacino, J.S. BORC Functions Upstream of Kinesins 1 and 3 to Coordinate Regional Movement of Lysosomes along Different Microtubule Tracks. Cell Rep. 2016, 17, 1950–1961. [Google Scholar] [CrossRef] [PubMed]
- Rao, S.K.; Huynh, C.; Proux-Gillardeaux, V.; Galli, T.; Andrews, N.W. Identification of SNAREs involved in synaptotagmin VII-regulated lysosomal exocytosis. J. Biol. Chem. 2004, 279, 20471–20479. [Google Scholar] [CrossRef] [PubMed]
- Medina, D.L.; Fraldi, A.; Bouche, V.; Annunziata, F.; Mansueto, G.; Spampanato, C.; Puri, C.; Pignata, A.; Martina, J.A.; Sardiello, M.; et al. Transcriptional activation of lysosomal exocytosis promotes cellular clearance. Dev. Cell 2011, 21, 421–430. [Google Scholar] [CrossRef] [PubMed]
- Castro-Gomes, T.; Corrotte, M.; Tam, C.; Andrews, N.W. Plasma Membrane Repair Is Regulated Extracellularly by Proteases Released from Lysosomes. PLoS ONE 2016, 11, e0152583. [Google Scholar] [CrossRef]
- Olson, L.J.; Hindsgaul, O.; Dahms, N.M.; Kim, J.J. Structural insights into the mechanism of pH-dependent ligand binding and release by the cation-dependent mannose 6-phosphate receptor. J. Biol. Chem. 2008, 283, 10124–10134. [Google Scholar] [CrossRef]
- Jane, D.T.; Morvay, L.; DaSilva, L.; Cavallo-Medved, D.; Sloane, B.F.; Dufresne, M.J. Cathepsin B localizes to plasma membrane caveolae of differentiating myoblasts and is secreted in an active form at physiological pH. Biol. Chem. 2006, 387, 223–234. [Google Scholar] [CrossRef]
- Jordans, S.; Jenko-Kokalj, S.; Kuhl, N.M.; Tedelind, S.; Sendt, W.; Bromme, D.; Turk, D.; Brix, K. Monitoring compartment-specific substrate cleavage by cathepsins B, K, L, and S at physiological pH and redox conditions. BMC Biochem. 2009, 10. [Google Scholar] [CrossRef]
- Uhlman, A.; Folkers, K.; Liston, J.; Pancholi, H.; Hinton, A. Effects of Vacuolar H(+)-ATPase Inhibition on Activation of Cathepsin B and Cathepsin L Secreted from MDA-MB231 Breast Cancer Cells. Cancer Microenviron. 2017, 10, 49–56. [Google Scholar] [CrossRef]
- Yang, D.Q.; Feng, S.; Chen, W.; Zhao, H.; Paulson, C.; Li, Y.P. V-ATPase subunit ATP6AP1 (Ac45) regulates osteoclast differentiation, extracellular acidification, lysosomal trafficking, and protease exocytosis in osteoclast-mediated bone resorption. J. Bone Min. Res. 2012, 27, 1695–1707. [Google Scholar] [CrossRef]
- Runger, T.M.; Quintanilla-Dieck, M.J.; Bhawan, J. Role of cathepsin K in the turnover of the dermal extracellular matrix during scar formation. J. Investig. Derm. 2007, 127, 293–297. [Google Scholar] [CrossRef]
- Goto, T.; Yamaza, T.; Tanaka, T. Cathepsins in the osteoclast. J. Electron. Microsc. (Tokyo) 2003, 52, 551–558. [Google Scholar] [CrossRef]
- Saftig, P.; Hunziker, E.; Everts, V.; Jones, S.; Boyde, A.; Wehmeyer, O.; Suter, A.; von Figura, K. Functions of cathepsin K in bone resorption. Lessons from cathepsin K deficient mice. Adv. Exp. Med. Biol. 2000, 477, 293–303. [Google Scholar] [CrossRef]
- Bonnans, C.; Chou, J.; Werb, Z. Remodelling the extracellular matrix in development and disease. Nat. Rev. Mol. Cell Biol. 2014, 15, 786–801. [Google Scholar] [CrossRef]
- Fonovic, M.; Turk, B. Cysteine cathepsins and extracellular matrix degradation. Biochim. Biophys. Acta 2014, 1840, 2560–2570. [Google Scholar] [CrossRef] [PubMed]
- Raggatt, L.J.; Partridge, N.C. Cellular and molecular mechanisms of bone remodeling. J. Biol. Chem. 2010, 285, 25103–25108. [Google Scholar] [CrossRef]
- Troen, B.R. The role of cathepsin K in normal bone resorption. Drug News Perspect. 2004, 17, 19–28. [Google Scholar] [CrossRef] [PubMed]
- Page, A.E.; Hayman, A.R.; Andersson, L.M.B.; Chambers, T.J.; Warburton, M.J. Degradation of Bone-Matrix Proteins by Osteoclast Cathepsins. Int. J. Biochem. 1993, 25, 545–550. [Google Scholar] [CrossRef]
- Saini, M.G.; Bix, G.J. Oxygen-glucose deprivation (OGD) and interleukin-1 (IL-1) differentially modulate cathepsin B/L mediated generation of neuroprotective perlecan LG3 by neurons. Brain Res. 2012, 1438, 65–74. [Google Scholar] [CrossRef] [PubMed]
- Roberts, J.; Kahle, M.P.; Bix, G.J. Perlecan and the blood-brain barrier: Beneficial proteolysis? Front. Pharm. 2012, 3, 155. [Google Scholar] [CrossRef]
- Buth, H.; Luigi Buttigieg, P.; Ostafe, R.; Rehders, M.; Dannenmann, S.R.; Schaschke, N.; Stark, H.J.; Boukamp, P.; Brix, K. Cathepsin B is essential for regeneration of scratch-wounded normal human epidermal keratinocytes. Eur. J. Cell Biol. 2007, 86, 747–761. [Google Scholar] [CrossRef]
- Obermajer, N.; Repnik, U.; Jevnikar, Z.; Turk, B.; Kreft, M.; Kos, J. Cysteine protease cathepsin X modulates immune response via activation of beta2 integrins. Immunology 2008, 124, 76–88. [Google Scholar] [CrossRef] [PubMed]
- Hook, V.; Yasothornsrikul, S.; Greenbaum, D.; Medzihradszky, K.F.; Troutner, K.; Toneff, T.; Bundey, R.; Logrinova, A.; Reinheckel, T.; Peters, C.; et al. Cathepsin L and Arg/Lys aminopeptidase: A distinct prohormone processing pathway for the biosynthesis of peptide neurotransmitters and hormones. Biol. Chem. 2004, 385, 473–480. [Google Scholar] [CrossRef] [PubMed]
- Funkelstein, L.; Lu, W.D.; Koch, B.; Mosier, C.; Toneff, T.; Taupenot, L.; O’Connor, D.T.; Reinheckel, T.; Peters, C.; Hook, V. Human cathepsin V protease participates in production of enkephalin and NPY neuropeptide neurotransmitters. J. Biol. Chem. 2012, 287, 15232–15241. [Google Scholar] [CrossRef] [PubMed]
- Hwang, S.R.; Garza, C.; Mosier, C.; Toneff, T.; Wunderlich, E.; Goldsmith, P.; Hook, V. Cathepsin L expression is directed to secretory vesicles for enkephalin neuropeptide biosynthesis and secretion. J. Biol. Chem. 2007, 282, 9556–9563. [Google Scholar] [CrossRef]
- Yasothornsrikul, S.; Greenbaum, D.; Medzihradszky, K.F.; Toneff, T.; Bundey, R.; Miller, R.; Schilling, B.; Petermann, I.; Dehnert, J.; Logvinova, A.; et al. Cathepsin L in secretory vesicles functions as a prohormone-processing enzyme for production of the enkephalin peptide neurotransmitter. Proc. Natl. Acad. Sci. USA 2003, 100, 9590–9595. [Google Scholar] [CrossRef]
- Yan, D.; Wang, H.W.; Bowman, R.L.; Joyce, J.A. STAT3 and STAT6 Signaling Pathways Synergize to Promote Cathepsin Secretion from Macrophages via IRE1alpha Activation. Cell Rep. 2016, 16, 2914–2927. [Google Scholar] [CrossRef]
- Sloane, B.F.; Yan, S.; Podgorski, I.; Linebaugh, B.E.; Cher, M.L.; Mai, J.; Cavallo-Medved, D.; Sameni, M.; Dosescu, J.; Moin, K. Cathepsin B and tumor proteolysis: Contribution of the tumor microenvironment. Semin. Cancer Biol. 2005, 15, 149–157. [Google Scholar] [CrossRef]
- Hashimoto, Y.; Kondo, C.; Katunuma, N. An Active 32-kDa Cathepsin L Is Secreted Directly from HT 1080 Fibrosarcoma Cells and Not via Lysosomal Exocytosis. PLoS ONE 2015, 10, e0145067. [Google Scholar] [CrossRef]
- Nicotra, G.; Castino, R.; Follo, C.; Peracchio, C.; Valente, G.; Isidoro, C. The dilemma: Does tissue expression of cathepsin D reflect tumor malignancy? The question: Does the assay truly mirror cathepsin D mis-function in the tumor? Cancer Biomark. 2010, 7, 47–64. [Google Scholar] [CrossRef]
- Gocheva, V.; Zeng, W.; Ke, D.; Klimstra, D.; Reinheckel, T.; Peters, C.; Hanahan, D.; Joyce, J.A. Distinct roles for cysteine cathepsin genes in multistage tumorigenesis. Genes Dev. 2006, 20, 543–556. [Google Scholar] [CrossRef]
- Lkhider, M.; Castino, R.; Bouguyon, E.; Isidoro, C.; Ollivier-Bousquet, M. Cathepsin D released by lactating rat mammary epithelial cells is involved in prolactin cleavage under physiological conditions. J. Cell Sci. 2004, 117, 5155–5164. [Google Scholar] [CrossRef][Green Version]
- Erdmann, S.; Ricken, A.; Merkwitz, C.; Struman, I.; Castino, R.; Hummitzsch, K.; Gaunitz, F.; Isidoro, C.; Martial, J.; Spanel-Borowski, K. The expression of prolactin and its cathepsin D-mediated cleavage in the bovine corpus luteum vary with the estrous cycle. Am. J. Physiol. Endocrinol. Metab. 2007, 293, E1365–E1377. [Google Scholar] [CrossRef]
- Sobotic, B.; Vizovisek, M.; Vidmar, R.; Van Damme, P.; Gocheva, V.; Joyce, J.A.; Gevaert, K.; Turk, V.; Turk, B.; Fonovic, M. Proteomic Identification of Cysteine Cathepsin Substrates Shed from the Surface of Cancer Cells. Mol. Cell Proteom. 2015, 14, 2213–2228. [Google Scholar] [CrossRef] [PubMed]
- Mizunoe, Y.; Kobayashi, M.; Tagawa, R.; Nakagawa, Y.; Shimano, H.; Higami, Y. Association between Lysosomal Dysfunction and Obesity-Related Pathology: A Key Knowledge to Prevent Metabolic Syndrome. Int. J. Mol. Sci. 2019, 20, 3688. [Google Scholar] [CrossRef] [PubMed]
- Bourne, L.C.; Lamb, D.J.; Collis, C.S.; O’Brien, M.; Leake, D.S.; Rice-Evans, C. Non-oxidative modification of low density lipoprotein by ruptured myocytes. FEBS Lett. 1997, 414, 576–580. [Google Scholar] [CrossRef]
- Benes, P.; Vetvicka, V.; Fusek, M. Cathepsin D—Many functions of one aspartic protease. Crit. Rev. Oncol. Hematol. 2008, 68, 12–28. [Google Scholar] [CrossRef] [PubMed]
- Khurana, P.; Yadati, T.; Goyal, S.; Dolas, A.; Houben, T.; Oligschlaeger, Y.; Agarwal, A.K.; Kulkarni, A.; Shiri-Sverdlov, R. Inhibiting Extracellular Cathepsin D Reduces Hepatic Steatosis in Sprague(-)Dawley Rats (dagger). Biomolecules 2019, 9, 171. [Google Scholar] [CrossRef]
- Zhao, C.F.; Herrington, D.M. The function of cathepsins B, D, and X in atherosclerosis. Am. J. Cardiovasc. Dis. 2016, 6, 163–170. [Google Scholar]
- Wiener, J.J.; Sun, S.; Thurmond, R.L. Recent advances in the design of cathepsin S inhibitors. Curr. Top. Med. Chem. 2010, 10, 717–732. [Google Scholar] [CrossRef]
- Maciewicz, R.A.; Wotton, S.F. Degradation of cartilage matrix components by the cysteine proteinases, cathepsins B and L. Biomed. Biochim. Acta 1991, 50, 561–564. [Google Scholar]
- Buck, M.R.; Karustis, D.G.; Day, N.A.; Honn, K.V.; Sloane, B.F. Degradation of extracellular-matrix proteins by human cathepsin B from normal and tumour tissues. Biochem. J. 1992, 282 Pt 1, 273–278. [Google Scholar] [CrossRef]
- Ishidoh, K.; Kominami, E. Procathepsin L degrades extracellular matrix proteins in the presence of glycosaminoglycans in vitro. Biochem. Biophys. Res. Commun. 1995, 217, 624–631. [Google Scholar] [CrossRef] [PubMed]
- Wang, B.; Sun, J.; Kitamoto, S.; Yang, M.; Grubb, A.; Chapman, H.A.; Kalluri, R.; Shi, G.P. Cathepsin S controls angiogenesis and tumor growth via matrix-derived angiogenic factors. J. Biol. Chem. 2006, 281, 6020–6029. [Google Scholar] [CrossRef] [PubMed]
- Son, E.D.; Kim, H.; Choi, H.; Lee, S.H.; Lee, J.Y.; Kim, S.; Closs, B.; Lee, S.; Chung, J.H.; Hwang, J.S. Cathepsin G increases MMP expression in normal human fibroblasts through fibronectin fragmentation, and induces the conversion of proMMP-1 to active MMP-1. J. Derm. Sci. 2009, 53, 150–152. [Google Scholar] [CrossRef] [PubMed]
- Mai, J.; Sameni, M.; Mikkelsen, T.; Sloane, B.F. Degradation of extracellular matrix protein tenascin-C by cathepsin B: An interaction involved in the progression of gliomas. Biol. Chem. 2002, 383, 1407–1413. [Google Scholar] [CrossRef] [PubMed]
- Mort, J.S.; Magny, M.C.; Lee, E.R. Cathepsin B: An alternative protease for the generation of an aggrecan ‘metalloproteinase’ cleavage neoepitope. Biochem. J. 1998, 335 Pt 3, 491–494. [Google Scholar] [CrossRef]
- Garcia, M.; Platet, N.; Liaudet, E.; Laurent, V.; Derocq, D.; Brouillet, J.P.; Rochefort, H. Biological and clinical significance of cathepsin D in breast cancer metastasis. Stem Cells 1996, 14, 642–650. [Google Scholar] [CrossRef]
- Du, X.; Chen, N.L.; Wong, A.; Craik, C.S.; Bromme, D. Elastin degradation by cathepsin V requires two exosites. J. Biol. Chem. 2013, 288, 34871–34881. [Google Scholar] [CrossRef]
- Staudt, N.D.; Aicher, W.K.; Kalbacher, H.; Stevanovic, S.; Carmona, A.K.; Bogyo, M.; Klein, G. Cathepsin X is secreted by human osteoblasts, digests CXCL-12 and impairs adhesion of hematopoietic stem and progenitor cells to osteoblasts. Haematologica 2010, 95, 1452–1460. [Google Scholar] [CrossRef]
- Ondr, J.K.; Pham, C.T. Characterization of murine cathepsin W and its role in cell-mediated cytotoxicity. J. Biol. Chem. 2004, 279, 27525–27533. [Google Scholar] [CrossRef]
- Fonovic, M.; Turk, B. Cysteine cathepsins and their potential in clinical therapy and biomarker discovery. Proteom. Clin. Appl. 2014, 8, 416–426. [Google Scholar] [CrossRef]
- Joyce, J.A.; Baruch, A.; Chehade, K.; Meyer-Morse, N.; Giraudo, E.; Tsai, F.Y.; Greenbaum, D.C.; Hager, J.H.; Bogyo, M.; Hanahan, D. Cathepsin cysteine proteases are effectors of invasive growth and angiogenesis during multistage tumorigenesis. Cancer Cell 2004, 5, 443–453. [Google Scholar] [CrossRef]
- Gabrijelcic, D.; Svetic, B.; Spaic, D.; Skrk, J.; Budihna, M.; Dolenc, I.; Popovic, T.; Cotic, V.; Turk, V. Cathepsins B, H and L in human breast carcinoma. Eur. J. Clin. Chem. Clin. Biochem. 1992, 30, 69–74. [Google Scholar]
- Hashimoto, Y.; Kondo, C.; Kojima, T.; Nagata, H.; Moriyama, A.; Hayakawa, T.; Katunuma, N. Significance of 32-kDa cathepsin L secreted from cancer cells. Cancer Biother. Radiopharm. 2006, 21, 217–224. [Google Scholar] [CrossRef]
- Singh, N.; Das, P.; Gupta, S.; Sachdev, V.; Srivasatava, S.; Datta Gupta, S.; Pandey, R.M.; Sahni, P.; Chauhan, S.S.; Saraya, A. Plasma cathepsin L: A prognostic marker for pancreatic cancer. World J. Gastroenterol. 2014, 20, 17532–17540. [Google Scholar] [CrossRef] [PubMed]
- Ruan, J.; Zheng, H.; Fu, W.; Zhao, P.; Su, N.; Luo, R. Increased expression of cathepsin L: A novel independent prognostic marker of worse outcome in hepatocellular carcinoma patients. PLoS ONE 2014, 9, e112136. [Google Scholar] [CrossRef]
- Schweiger, A.; Staib, A.; Werle, B.; Krasovec, M.; Lah, T.T.; Ebert, W.; Turk, V.; Kos, J. Cysteine proteinase cathepsin H in tumours and sera of lung cancer patients: Relation to prognosis and cigarette smoking. Br. J. Cancer 2000, 82, 782–788. [Google Scholar] [CrossRef] [PubMed]
- Kos, J.; Stabuc, B.; Schweiger, A.; Krasovec, M.; Cimerman, N.; KopitarJerala, N.; Vrhovec, I. Cathepsins B, H, and L and their inhibitors stefin A and cystatin C in sera of melanoma patients. Clin. Cancer Res. 1997, 3, 1815–1822. [Google Scholar] [PubMed]
- Schweiger, A.; Christensen, I.J.; Nielsen, H.J.; Sorensen, S.; Brunner, N.; Kos, J. Serum cathepsin H as a potential prognostic marker in patients with colorectal cancer. Int. J. Biol. Markers 2004, 19, 289–294. [Google Scholar] [CrossRef]
- Kos, J.; Werle, B.; Lah, T.; Brunner, N. Cysteine proteinases and their inhibitors in extracellular fluids: Markers for diagnosis and prognosis in cancer. Int. J. Biol. Markers 2000, 15, 84–89. [Google Scholar] [CrossRef] [PubMed]
- Wu, H.; Du, Q.; Dai, Q.; Ge, J.; Cheng, X. Cysteine Protease Cathepsins in Atherosclerotic Cardiovascular Diseases. J. Atheroscler. Thromb. 2018, 25, 111–123. [Google Scholar] [CrossRef] [PubMed]
- Walenbergh, S.M.; Houben, T.; Rensen, S.S.; Bieghs, V.; Hendrikx, T.; van Gorp, P.J.; Oligschlaeger, Y.; Jeurissen, M.L.; Gijbels, M.J.; Buurman, W.A.; et al. Plasma cathepsin D correlates with histological classifications of fatty liver disease in adults and responds to intervention. Sci. Rep. 2016, 6, 38278. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Chen, B.; Zhang, X.; Tan, L.; Wang, D.W. Increased Cathepsin D Correlates with Clinical Parameters in Newly Diagnosed Type 2 Diabetes. Dis. Markers 2017, 2017, 5286408. [Google Scholar] [CrossRef] [PubMed]
- Ding, L.L.; Goossens, G.H.; Oligschlaeger, Y.; Houben, T.; Blaak, E.E.; Shiri-Sverdlov, R. Plasma cathepsin D activity is negatively associated with hepatic insulin sensitivity in overweight and obese humans. Diabetologia 2019. [Google Scholar] [CrossRef]
- Dera, A.A.; Ranganath, L.; Barraclough, R.; Vinjamuri, S.; Hamill, S.; Barraclough, D.L. Cathepsin Z as a novel potential biomarker for osteoporosis. Sci. Rep. 2019, 9. [Google Scholar] [CrossRef]
- Wang, L.; Hu, Y.Q.; Zhao, Z.J.; Zhang, H.Y.; Gao, B.; Lu, W.G.; Xu, X.L.; Lin, X.S.; Wang, J.P.; Jie, Q.; et al. Screening and validation of serum protein biomarkers for early postmenopausal osteoporosis diagnosis. Mol. Med. Rep. 2017, 16, 8427–8433. [Google Scholar] [CrossRef] [PubMed]
- Kramer, L.; Turk, D.; Turk, B. The Future of Cysteine Cathepsins in Disease Management. Trends Pharm. Sci. 2017, 38, 873–898. [Google Scholar] [CrossRef] [PubMed]
- Marques, A.R.A.; Di Spiezio, A.; Thiessen, N.; Schmidt, L.; Grotzinger, J.; Lullmann-Rauch, R.; Damme, M.; Storck, S.E.; Pietrzik, C.U.; Fogh, J.; et al. Enzyme replacement therapy with recombinant pro-CTSD (cathepsin D) corrects defective proteolysis and autophagy in neuronal ceroid lipofuscinosis. Autophagy 2020, 16, 811–825. [Google Scholar] [CrossRef]
- Desnick, R.J.; Schuchman, E.H. Enzyme replacement therapy for lysosomal diseases: Lessons from 20 years of experience and remaining challenges. Annu. Rev. Genom. Hum. Genet. 2012, 13, 307–335. [Google Scholar] [CrossRef]
- Khaket, T.P.; Kwon, T.K.; Kang, S.C. Cathepsins: Potent regulators in carcinogenesis. Pharm. Ther. 2019, 198, 1–19. [Google Scholar] [CrossRef]
- Verbovsek, U.; Van Noorden, C.J.; Lah, T.T. Complexity of cancer protease biology: Cathepsin K expression and function in cancer progression. Semin. Cancer Biol. 2015, 35, 71–84. [Google Scholar] [CrossRef]
- Mullard, A. Merck & Co. drops osteoporosis drug odanacatib. Nat. Rev. Drug Discov. 2016, 15, 669. [Google Scholar] [CrossRef]
- Panwar, P.; Soe, K.; Guido, R.V.; Bueno, R.V.; Delaisse, J.M.; Bromme, D. A novel approach to inhibit bone resorption: Exosite inhibitors against cathepsin K. Br. J. Pharm. 2016, 173, 396–410. [Google Scholar] [CrossRef] [PubMed]
- Panwar, P.; Xue, L.M.; Soe, K.; Srivastava, K.; Law, S.; Delaisse, J.M.; Bromme, D. An Ectosteric Inhibitor of Cathepsin K Inhibits Bone Resorption in Ovariectomized Mice (vol 32, pg 2415, 2017). J. Bone Miner. Res. 2019, 34, 777–778. [Google Scholar] [CrossRef] [PubMed]
- Burden, R.E.; Gormley, J.A.; Jaquin, T.J.; Small, D.M.; Quinn, D.J.; Hegarty, S.M.; Ward, C.; Walker, B.; Johnston, J.A.; Olwill, S.A.; et al. Antibody-mediated inhibition of cathepsin S blocks colorectal tumor invasion and angiogenesis. Clin. Cancer Res. 2009, 15, 6042–6051. [Google Scholar] [CrossRef]
- Burden, R.E.; Gormley, J.A.; Kuehn, D.; Ward, C.; Kwok, H.F.; Gazdoiu, M.; McClurg, A.; Jaquin, T.J.; Johnston, J.A.; Scott, C.J.; et al. Inhibition of Cathepsin S by Fsn0503 enhances the efficacy of chemotherapy in colorectal carcinomas. Biochimie 2012, 94, 487–493. [Google Scholar] [CrossRef] [PubMed]
- Mirkovic, B.; Markelc, B.; Butinar, M.; Mitrovic, A.; Sosic, I.; Gobec, S.; Vasiljeva, O.; Turk, B.; Cemazar, M.; Sersa, G.; et al. Nitroxoline impairs tumor progression in vitro and in vivo by regulating cathepsin B activity. Oncotarget 2015, 6, 19027–19042. [Google Scholar] [CrossRef] [PubMed]
- Mikhaylov, G.; Klimpel, D.; Schaschke, N.; Mikac, U.; Vizovisek, M.; Fonovic, M.; Turk, V.; Turk, B.; Vasiljeva, O. Selective targeting of tumor and stromal cells by a nanocarrier system displaying lipidated cathepsin B inhibitor. Angew. Chem. Int. Ed. Engl. 2014, 53, 10077–10081. [Google Scholar] [CrossRef] [PubMed]

| Cathepsin | Enzyme Commission Number | Catalytic Type | Function | Pathology | OMIM ID | Reference |
|---|---|---|---|---|---|---|
| Cathepsin A | 3.4.16.5 | serine | dual function: a. protective:β-galactosidase and neuraminidase b. degradative: bioactive peptides like bradykinin, angiotensin, oxytocin, endothelin 1 | hypertension Galactosialidosis | 256540 | [63] |
| Cathepsin B | 3.4.22.1 | cysteine | degrades amyloid-β; activation of pro-hormones and pro-enzymes; trypsin activation; promotes viral entry into cells | Alzheimer’s; gaucher disease acute pancreatitis | [21,68] | |
| Cathepsin C | 3.4.14.1 | cysteine | inflammatory responses and activation of serine proteases including neutrophil elastase and cathepsin G | Papillon–Lefèvre syndrome Periodontitis | 245000 | [65] |
| Cathepsin D | 3.4.23.5 | aspartic | embryo and neuronal development brain antigen processing of α-Synuclein; tau, amyloid β, apoE; degradation of hormones, proenzymes and growth factors | Alzheimer’s disease; CLN 10 Parkinson’s; Huntington’s | 610127 | [20,61,64] |
| Cathepsin E | 3.4.23.34 | aspartic | carboxypeptidase A and IgE processing | atopic dermatitis | [71] | |
| Cathepsin F | 3.4.22.41 | cysteine | li chain processing and MHC-II class responses | CLN 13 | 615362 | [63] |
| Cathepsin G | 3.4.21.20 | serine | auto antigen processing | auto-immune diseases | [72] | |
| Cathepsin H | 3.4.22.16 | cysteine | prohormone processing | type 1 diabetes | [9] | |
| Cathepsin K | 3.4.22.38 | cysteine | TLR signaling; processing of β-endorphin in brain | periodontitis; pycnodysostosis | 265800 | [73,74] |
| Cathepsin L | 3.4.22.15 | cysteine | antigen and li chain processing; prohormone processing; degradation of α-Synuclein, tau; promotes viral entry into cells | Parkinson’s disease; frontotemporal dementia | [19,49] | |
| Cathepsin S | 3.4.22.27 | cysteine | antigen processing and presentation; li chain processing | auto-immune diseases | [50] | |
| Cathepsin X | 3.4.18.1 | cysteine | T-cell migration and invasion | - | [75] | |
| Cathepsin O | 3.4.22.42 | cysteine | - | - | ||
| Cathepsin V | 3.4.22.43 | cysteine | natural killer cell and CD8+ cytotoxic cell production | thymic pathology | [76] | |
| Cathepsin W | 3.4.22.- | cysteine | component of endoplasmic reticulum proteolytic machinery | - | [77] | |
| Cathepsin Z | 3.4.18.1 | cysteine | intracellular protein turnover | - | [78] |
| Cathepsin | Extra Lysosomal Location | Function | Reference |
|---|---|---|---|
| Cathepsin B, D and L | cytosol | proteolytic processing of Bid during apoptosis | [97,111] |
| Cathepsin B, C, L, S and Z | cytosol | NLRP3 inflammasome activation | [103,104,105,106] |
| Cathepsin B | cytosol | regulation of hepatic lipid metabolism by degrading liver fatty acid binding protein | [112] |
| Cathepsin L and H | nucleus | cell cycle regulation | [91] |
| Cathepsin B, K, L and S | nucleus | TGF-β signaling | [110] |
| Cathepsin B | nucleus | bile-salt induced apoptosis | [113] |
| Cathepsin A, E, G, S, X, O, V, W, Z * | - | - | - |
| Cathepsin | Substrate | Pathological State | Reference |
|---|---|---|---|
| Cathepsin B, K, and L | proteoglycan | osteoarthritis | [137,165] |
| Cathepsin B, L, G, and S | fibronectin | cancer and adipogenesis | [166,167,168,169] |
| Cathepsin B, L, and S | laminin | cancer neovascularization, intestinal trauma | [166,167,168] |
| Cathepsin K | collagen type I | osteoporosis, rheumatized arthritis, osteoarthritis | [137] |
| Cathepsin B, K, L, and S | collagen type 2 | lung fibrosis, cardiovascular diseases and cancer | [138,139] |
| Cathepsin B | tenascin | cancer | [170] |
| Cathepsin B, K, L, and S | aggrecan | osteoarthritis | [136,171] |
| Cathepsin L, S, and B | plexin | tumorigenesis | [158] |
| Cathepsin S | fractalkine | neuropathic pain | [164] |
| Cathepsin D | fibroblast growth factor | breast cancer | [172] |
| Cathepsin V | elastin | cancer | [173] |
| Cathepsin X | CXCL-12 | - | [174] |
| Cathepsin W | - | cell-mediated cytotoxicity | [175] |
| Cathepsin A, C, E, F, O, and Z * | - | - |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yadati, T.; Houben, T.; Bitorina, A.; Shiri-Sverdlov, R. The Ins and Outs of Cathepsins: Physiological Function and Role in Disease Management. Cells 2020, 9, 1679. https://doi.org/10.3390/cells9071679
Yadati T, Houben T, Bitorina A, Shiri-Sverdlov R. The Ins and Outs of Cathepsins: Physiological Function and Role in Disease Management. Cells. 2020; 9(7):1679. https://doi.org/10.3390/cells9071679
Chicago/Turabian StyleYadati, Tulasi, Tom Houben, Albert Bitorina, and Ronit Shiri-Sverdlov. 2020. "The Ins and Outs of Cathepsins: Physiological Function and Role in Disease Management" Cells 9, no. 7: 1679. https://doi.org/10.3390/cells9071679
APA StyleYadati, T., Houben, T., Bitorina, A., & Shiri-Sverdlov, R. (2020). The Ins and Outs of Cathepsins: Physiological Function and Role in Disease Management. Cells, 9(7), 1679. https://doi.org/10.3390/cells9071679





