The Enigma of Centriole Loss in the 1182-4 Cell Line
Abstract
1. Introduction
2. Material and Methods
2.1. Genomic DNA Preparation
2.2. mh Expression Construct
3. Historical Perspective: The Origins of the Acentriolar 1182-4 Drosophila Cell Line
4. Rebound: Identification of the mh Gene
5. Genomic Analyses of 1182-4 Cells
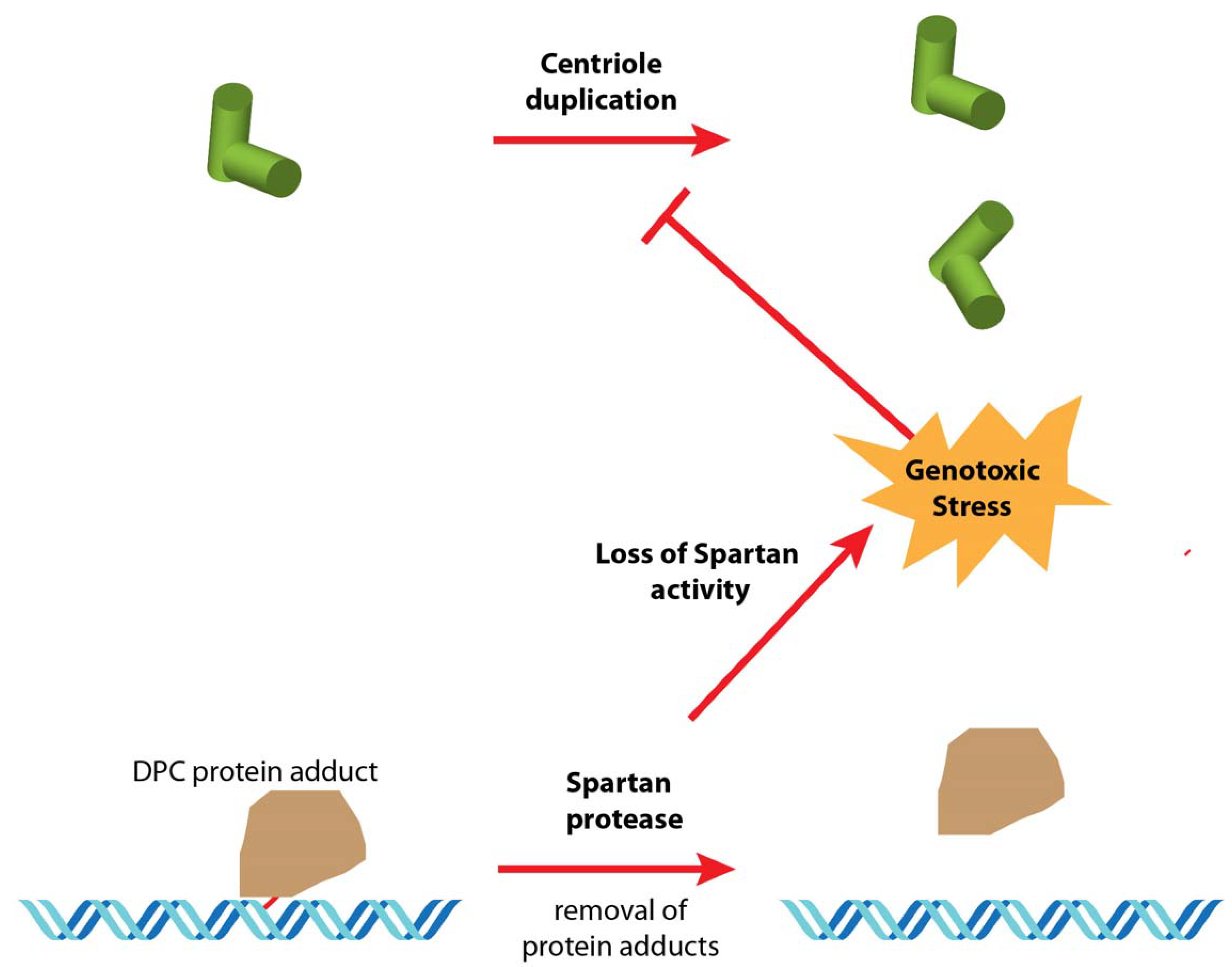
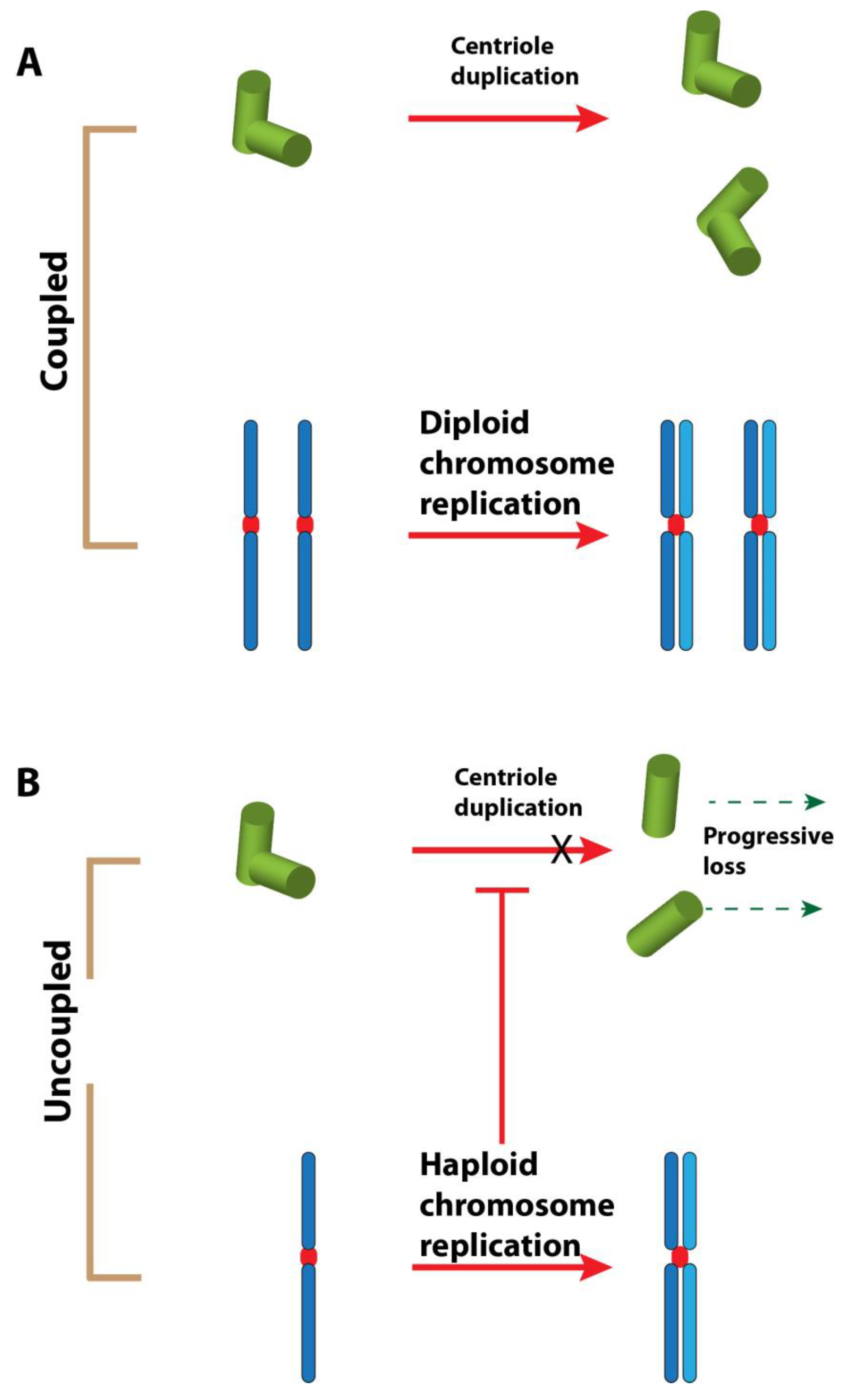
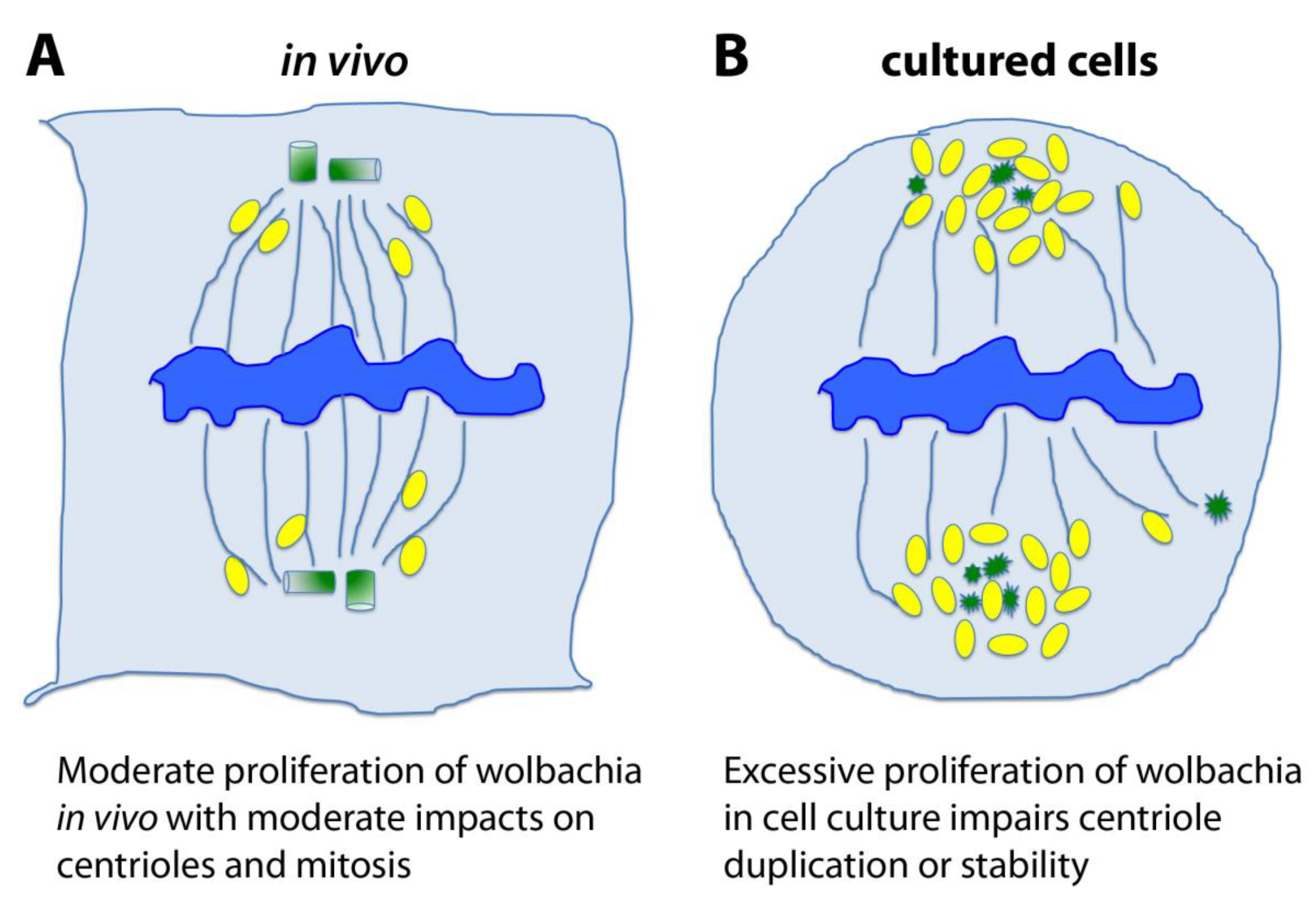
6. Hypotheses and Perspectives
7. Conclusions: Something Has Been Lost?
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Debec, A.; Sullivan, W.; Bettencourt-Dias, M. Centrioles: Active players or passengers during mitosis? Cell. Mol. Life Sci. 2010, 67, 2173–2194. [Google Scholar] [CrossRef]
- Azimzadeh, J.; Wong, M.L.; Downhour, D.M.; Sanchez Alvarado, A.; Marshall, W.F. Centrosome loss in the evolution of planarians. Science 2012, 335, 461–463. [Google Scholar] [CrossRef] [PubMed]
- Lipka, E.; Herrmann, A.; Mueller, S. Mechanisms of plant cell division. Wiley Interdiscip. Rev. Dev. Biol. 2015, 4, 391–405. [Google Scholar] [CrossRef] [PubMed]
- Meunier, S.; Vernos, I. Acentrosomal Microtubule Assembly in Mitosis: The Where, When, and How. Trends Cell Biol. 2016, 26, 80–87. [Google Scholar] [CrossRef] [PubMed]
- Chavali, P.L.; Peset, I.; Gergely, F. Centrosomes and mitotic spindle poles: A recent liaison? Biochem. Soc. Trans. 2015, 43, 13–18. [Google Scholar] [CrossRef]
- Yi, P.; Goshima, G. Microtubule nucleation and organization without centrosomes. Curr. Opin. Plant. Biol. 2018, 46, 1–7. [Google Scholar] [CrossRef]
- Nigg, E.A.; Holland, A.J. Once and only once: Mechanisms of centriole duplication and their deregulation in disease. Nat. Rev. Mol. Cell. Biol. 2018, 19, 297–312. [Google Scholar] [CrossRef]
- Loncarek, J.; Bettencourt-Dias, M. Building the right centriole for each cell type. J. Cell. Biol. 2018, 217, 823–835. [Google Scholar] [CrossRef]
- Guichard, P.; Hamel, V.; Gonczy, P. The Rise of the Cartwheel: Seeding the Centriole Organelle. Bioessays 2018, 40, e1700241. [Google Scholar] [CrossRef]
- Gonczy, P.; Hatzopoulos, G.N. Centriole assembly at a glance. J. Cell. Sci. 2019, 132, jcs:228833. [Google Scholar] [CrossRef]
- Banterle, N.; Gonczy, P. Centriole Biogenesis: From Identifying the Characters to Understanding the Plot. Annu. Rev. Cell. Dev. Biol. 2017, 33, 23–49. [Google Scholar] [CrossRef] [PubMed]
- Tillery, M.M.L.; Blake-Hedges, C.; Zheng, Y.; Buchwalter, R.A.; Megraw, T.L. Centrosomal and Non-Centrosomal Microtubule-Organizing Centers (MTOCs) in Drosophila melanogaster. Cells 2018, 7, 121. [Google Scholar] [CrossRef] [PubMed]
- Nigg, E.A.; Stearns, T. The centrosome cycle: Centriole biogenesis, duplication and inherent asymmetries. Nat. Cell. Biol. 2011, 13, 1154–1160. [Google Scholar] [CrossRef] [PubMed]
- Avidor-Reiss, T.; Gopalakrishnan, J. Building a centriole. Curr. Opin. Cell. Biol. 2013, 25, 72–77. [Google Scholar] [CrossRef]
- Debec, A.; Szollosi, A.; Szollosi, D. A Drosophila melanogaster cell line lacking centriole. Biol. Cell. 1982, 44, 133–138. [Google Scholar]
- Szollosi, A.; Ris, H.; Szollosi, D.; Debec, A. A centriole-free Drosophila cell line. A high voltage EM study. Eur. J. Cell. Biol. 1986, 40, 100–104. [Google Scholar]
- Delabaere, L.; Orsi, G.A.; Sapey-Triomphe, L.; Horard, B.; Couble, P.; Loppin, B. The Spartan ortholog maternal haploid is required for paternal chromosome integrity in the Drosophila zygote. Curr. Biol. 2014, 24, 2281–2287. [Google Scholar] [CrossRef]
- Tang, X.; Cao, J.; Zhang, L.; Huang, Y.; Zhang, Q.; Rong, Y.S. Maternal Haploid, a Metalloprotease Enriched at the Largest Satellite Repeat and Essential for Genome Integrity in Drosophila Embryos. Genetics 2017, 206, 1829–1839. [Google Scholar] [CrossRef]
- Kao, L.R.; Megraw, T.L. RNAi in cultured Drosophila cells. Methods Mol. Biol. 2004, 247, 443–457. [Google Scholar]
- Gans, M.; Audit, C.; Masson, M. Isolation and characterization of sex-linked female-sterile mutants in Drosophila melanogaster. Genetics 1975, 81, 683–704. [Google Scholar]
- Zalokar, M.; Audit, C.; Erk, I. Developmental defects of female-sterile mutants of Drosophila melanogaster. Dev. Biol. 1975, 47, 419–432. [Google Scholar] [CrossRef]
- Santamaria, P.; Gans, M. Chimaeras of Drosophila melanogaster obtained by injection of haploid nuclei. Nature 1980, 287, 143–144. [Google Scholar] [CrossRef] [PubMed]
- Loppin, B.; Berger, F.; Couble, P. Paternal chromosome incorporation into the zygote nucleus is controlled by maternal haploid in Drosophila. Dev. Biol. 2001, 231, 383–396. [Google Scholar] [CrossRef] [PubMed]
- Debec, A. Haploid cell cultures of Drosophila melanogaster. Nature 1978, 274, 255–256. [Google Scholar] [CrossRef]
- Debec, A. Evolution of karyotype in haploid cell lines of Drosophila melanogaster. Exp. Cell. Res. 1984, 151, 236–246. [Google Scholar] [CrossRef]
- Piel, M.; Nordberg, J.; Euteneuer, U.; Bornens, M. Centrosome-dependent exit of cytokinesis in animal cells. Science 2001, 291, 1550–1553. [Google Scholar] [CrossRef]
- Dolfini, S.F. Spontaneous chromosome rearrangements in an established cell line of Drosophila melanogaster. Chromosoma 1974, 47, 253–261. [Google Scholar] [CrossRef]
- Szollosi, D.; Calarco, P.; Donahue, R.P. Absence of centrioles in the first and second meiotic spindles of mouse oocytes. J. Cell. Sci. 1972, 11, 521–541. [Google Scholar]
- Basto, R.; Lau, J.; Vinogradova, T.; Gardiol, A.; Woods, C.G.; Khodjakov, A.; Raff, J.W. Flies without centrioles. Cell 2006, 125, 1375–1386. [Google Scholar] [CrossRef]
- Megraw, T.L.; Kao, L.R.; Kaufman, T.C. Zygotic development without functional mitotic centrosomes. Curr. Biol. 2001, 11, 116–120. [Google Scholar] [CrossRef]
- Edgar, B.A.; Kiehle, C.P.; Schubiger, G. Cell cycle control by the nucleo-cytoplasmic ratio in early Drosophila development. Cell 1986, 44, 365–372. [Google Scholar] [CrossRef]
- Sullivan, W. Independence of fushi tarazu expression with respect to cellular density in Drosophila embryos. Nature 1987, 327, 164–167. [Google Scholar] [CrossRef] [PubMed]
- Debec, A.; Detraves, C.; Montmory, C.; Geraud, G.; Wright, M. Polar organization of gamma-tubulin in acentriolar mitotic spindles of Drosophila melanogaster cells. J. Cell. Sci. 1995, 108, 2645–2653. [Google Scholar] [PubMed]
- Moutinho-Pereira, S.; Debec, A.; Maiato, H. Microtubule cytoskeleton remodeling by acentriolar microtubule-organizing centers at the entry and exit from mitosis in Drosophila somatic cells. Mol. Biol. Cell 2009, 20, 2796–2808. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Hinchcliffe, E.H.; Miller, F.J.; Cham, M.; Khodjakov, A.; Sluder, G. Requirement of a centrosomal activity for cell cycle progression through G1 into S phase. Science 2001, 291, 1547–1550. [Google Scholar] [CrossRef] [PubMed]
- Khodjakov, A.; Rieder, C.L. Centrosomes enhance the fidelity of cytokinesis in vertebrates and are required for cell cycle progression. J. Cell. Biol. 2001, 153, 237–242. [Google Scholar] [CrossRef] [PubMed]
- Khodjakov, A.; Cole, R.W.; Oakley, B.R.; Rieder, C.L. Centrosome-independent mitotic spindle formation in vertebrates. Curr. Biol. 2000, 10, 59–67. [Google Scholar] [CrossRef]
- Loppin, B.; Docquier, M.; Bonneton, F.; Couble, P. The maternal effect mutation sesame affects the formation of the male pronucleus in Drosophila melanogaster. Dev. Biol. 2000, 222, 392–404. [Google Scholar] [CrossRef]
- Loppin, B.; Berger, F.; Couble, P. The Drosophila maternal gene sesame is required for sperm chromatin remodeling at fertilization. Chromosoma 2001, 110, 430–440. [Google Scholar] [CrossRef]
- Stingele, J.; Bellelli, R.; Boulton, S.J. Mechanisms of DNA-protein crosslink repair. Nat Rev. Mol. Cell Biol 2017, 18, 563–573. [Google Scholar] [CrossRef]
- Fielden, J.; Ruggiano, A.; Popovic, M.; Ramadan, K. DNA protein crosslink proteolysis repair: From yeast to premature ageing and cancer in humans. DNA Repair (Amst) 2018, 71, 198–204. [Google Scholar] [CrossRef]
- Puah, W.C.; Chinta, R.; Wasser, M. Quantitative microscopy uncovers ploidy changes during mitosis in live Drosophila embryos and their effect on nuclear size. Biol. Open 2017, 6, 390–401. [Google Scholar] [CrossRef] [PubMed]
- Svetec, N.; Cridland, J.M.; Zhao, L.; Begun, D.J. The Adaptive Significance of Natural Genetic Variation in the DNA Damage Response of Drosophila melanogaster. PLoS Genet. 2016, 12, e1005869. [Google Scholar] [CrossRef] [PubMed]
- Blake-Hedges, C.; Megraw, T.L. Coordination of Embryogenesis by the Centrosome in Drosophila melanogaster. Results Probl. Cell Differ. 2019, 67, 277–321. [Google Scholar] [PubMed]
- Megraw, T.L.; Li, K.; Kao, L.R.; Kaufman, T.C. The centrosomin protein is required for centrosome assembly and function during cleavage in Drosophila. Development 1999, 126, 2829–2839. [Google Scholar] [PubMed]
- Sibon, O.C.; Kelkar, A.; Lemstra, W.; Theurkauf, W.E. DNA-replication/DNA-damage-dependent centrosome inactivation in Drosophila embryos. Nat. Cell Biol. 2000, 2, 90–95. [Google Scholar] [CrossRef] [PubMed]
- Takada, S.; Kelkar, A.; Theurkauf, W.E. Drosophila checkpoint kinase 2 couples centrosome function and spindle assembly to genomic integrity. Cell 2003, 113, 87–99. [Google Scholar] [CrossRef]
- Wilhelm, T.; Olziersky, A.M.; Harry, D.; De Sousa, F.; Vassal, H.; Eskat, A.; Meraldi, P. Mild replication stress causes chromosome mis-segregation via premature centriole disengagement. Nat. Commun. 2019, 10, 3585. [Google Scholar] [CrossRef]
- Cherbas, L.; Willingham, A.; Zhang, D.; Yang, L.; Zou, Y.; Eads, B.D.; Carlson, J.W.; Landolin, J.M.; Kapranov, P.; Dumais, J.; et al. The transcriptional diversity of 25 Drosophila cell lines. Genome Res. 2011, 21, 301–314. [Google Scholar] [CrossRef]
- Fu, J.; Lipinszki, Z.; Rangone, H.; Min, M.; Mykura, C.; Chao-Chu, J.; Schneider, S.; Dzhindzhev, N.S.; Gottardo, M.; Riparbelli, M.G.; et al. Conserved molecular interactions in centriole-to-centrosome conversion. Nat. Cell Biol. 2016, 18, 87–99. [Google Scholar] [CrossRef]
- Rodrigues-Martins, A.; Riparbelli, M.; Callaini, G.; Glover, D.M.; Bettencourt-Dias, M. Revisiting the role of the mother centriole in centriole biogenesis. Science 2007, 316, 1046–1050. [Google Scholar] [CrossRef] [PubMed]
- Stevens, N.R.; Raposo, A.A.; Basto, R.; St Johnston, D.; Raff, J.W. From stem cell to embryo without centrioles. Curr. Biol. 2007, 17, 1498–1503. [Google Scholar] [CrossRef] [PubMed]
- Lecland, N.; Debec, A.; Delmas, A.; Moutinho-Pereira, S.; Malmanche, N.; Bouissou, A.; Dupre, C.; Jourdan, A.; Raynaud-Messina, B.; Maiato, H.; et al. Establishment and mitotic characterization of new Drosophila acentriolar cell lines from DSas-4 mutant. Biol. Open 2013, 2, 314–323. [Google Scholar] [CrossRef] [PubMed]
- Oegema, K.; Marshall, W.F.; Sedat, J.W.; Alberts, B.M. Two proteins that cycle asynchronously between centrosomes and nuclear structures: Drosophila CP60 and CP190. J. Cell Sci 1997, 110, 1573–1583. [Google Scholar]
- Debec, A.; Abbadie, C. The acentriolar state of the Drosophila cell lines 1182. Biol. Cell 1989, 67, 307–311. [Google Scholar] [CrossRef]
- Yaguchi, K.; Yamamoto, T.; Matsui, R.; Tsukada, Y.; Shibanuma, A.; Kamimura, K.; Koda, T.; Uehara, R. Uncoordinated centrosome cycle underlies the instability of non-diploid somatic cells in mammals. J. Cell Biol. 2018, 217, 2463–2483. [Google Scholar] [CrossRef]
- Uzbekov, R.; Garanina, A.; Bressac, C. Centrioles without microtubules: A new morphological type of centriole. Biol. Open 2018, 7. [Google Scholar] [CrossRef]
- Tirmarche, S.; Kimura, S.; Dubruille, R.; Horard, B.; Loppin, B. Unlocking sperm chromatin at fertilization requires a dedicated egg thioredoxin in Drosophila. Nat. Commun. 2016, 7, 13539. [Google Scholar] [CrossRef]
- Fuyama, Y. Genetics of Parthenogenesis in DROSOPHILA MELANOGASTER. I. the Modes of Diploidization in the Gynogenesis Induced by a Male-Sterile Mutant, ms(3)K81. Genetics 1986, 112, 237–248. [Google Scholar]
- Debec, A.; Szollosi, A. Presence of Rickettsias in haploid Drosophila melanogaster cell lines. Biol. Cell 1980, 38, 129–134. [Google Scholar]
- Tram, U.; Sullivan, W. Role of delayed nuclear envelope breakdown and mitosis in Wolbachia-induced cytoplasmic incompatibility. Science 2002, 296, 1124–1126. [Google Scholar] [CrossRef] [PubMed]
- Charlat, S.; Mercot, H. Cytoplasmic incompatibility and maternal-haploid. Trends Genet. 2001, 17, 440–441. [Google Scholar] [CrossRef]
- Loncarek, J.; Khodjakov, A. Ab ovo or de novo? Mechanisms of centriole duplication. Mol. Cells 2009, 27, 135–142. [Google Scholar] [CrossRef] [PubMed]
- Bobinnec, Y.; Khodjakov, A.; Mir, L.M.; Rieder, C.L.; Edde, B.; Bornens, M. Centriole disassembly in vivo and its effect on centrosome structure and function in vertebrate cells. J. Cell Biol. 1998, 143, 1575–1589. [Google Scholar] [CrossRef]
- Khodjakov, A.; Rieder, C.L.; Sluder, G.; Cassels, G.; Sibon, O.; Wang, C.L. De novo formation of centrosomes in vertebrate cells arrested during S phase. J. Cell Biol. 2002, 158, 1171–1181. [Google Scholar] [CrossRef]
- La Terra, S.; English, C.N.; Hergert, P.; McEwen, B.F.; Sluder, G.; Khodjakov, A. The de novo centriole assembly pathway in HeLa cells: Cell cycle progression and centriole assembly/maturation. J. Cell Biol. 2005, 168, 713–722. [Google Scholar] [CrossRef]
- Marshall, W.F.; Vucica, Y.; Rosenbaum, J.L. Kinetics and regulation of de novo centriole assembly. Implications for the mechanism of centriole duplication. Curr. Biol. 2001, 11, 308–317. [Google Scholar] [CrossRef]
- Uetake, Y.; Loncarek, J.; Nordberg, J.J.; English, C.N.; La Terra, S.; Khodjakov, A.; Sluder, G. Cell cycle progression and de novo centriole assembly after centrosomal removal in untransformed human cells. J. Cell Biol. 2007, 176, 173–182. [Google Scholar] [CrossRef]
| Gene | Aberrations * | Location of Aberration in Gene |
|---|---|---|
| SAK | 1bp Del at 21509098 | 110 bp 5′ from transcription start. |
| Sas-6 | None | |
| Sas-4 | None | |
| Asl | None | |
| Cep135/bld10 | 1bp Ins at 15109935 | 5′ UTR |
| 2bp Del at 15112702 | intron | |
| 1bp Ins at 15112720 | intron | |
| 2bp Del at 15112725 | intron | |
| 1bp Ins at 15112962 | intron | |
| 20bp Del at 15115633 | intron | |
| 1bp Ins at 15119269 | intron | |
| ana1 | 1bp Del at 20356251 | 5′ UTR |
| 1bp Del at 20356417 | 5′ UTR | |
| 3bp Del at 20359488 | middle of ORF | |
| 18bp Ins at 20359480 | middle of ORF | |
| 3bp Ins at 20361873 (CAG) | near 3′ end of ORF | |
| ana2 | 24bp Del at 4786186 | About 40 bp 3′ of transc stop. |
| ana3 | None | |
| Cp110 | 1bp Ins at 21884283 | 5′UTR |
| 4bp Del at 21884285 | 5′UTR | |
| 30bp Del before 21884288? | 5′UTR | |
| 2bp Ins at 21886886 | intron | |
| 2bp Del at 21886889 | intron | |
| 5bp Ins at 21886895 | intron | |
| 1bp Del at 21886898 | intron | |
| 4bp Del at 21888272 | intron | |
| 7bp Del at 21888275 | intron | |
| 36bp Del before 21888280? | intron | |
| 3bp Ins at 21888375 | intron | |
| 14bp Del at 21888813 | intron | |
| 1bp Del at 21888879 | intron |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Debec, A.; Loppin, B.; Zheng, C.; Liu, X.; Megraw, T.L. The Enigma of Centriole Loss in the 1182-4 Cell Line. Cells 2020, 9, 1300. https://doi.org/10.3390/cells9051300
Debec A, Loppin B, Zheng C, Liu X, Megraw TL. The Enigma of Centriole Loss in the 1182-4 Cell Line. Cells. 2020; 9(5):1300. https://doi.org/10.3390/cells9051300
Chicago/Turabian StyleDebec, Alain, Benjamin Loppin, Chunfeng Zheng, Xiuwen Liu, and Timothy L. Megraw. 2020. "The Enigma of Centriole Loss in the 1182-4 Cell Line" Cells 9, no. 5: 1300. https://doi.org/10.3390/cells9051300
APA StyleDebec, A., Loppin, B., Zheng, C., Liu, X., & Megraw, T. L. (2020). The Enigma of Centriole Loss in the 1182-4 Cell Line. Cells, 9(5), 1300. https://doi.org/10.3390/cells9051300






