The Impact of Mutations in the HvCPD and HvBRI1 Genes on the Physicochemical Properties of the Membranes from Barley Acclimated to Low/High Temperatures
Abstract
1. Introduction
2. Plant Material and Experimental Design
2.1. Lipid Extraction and Fatty Acid Composition Measurement
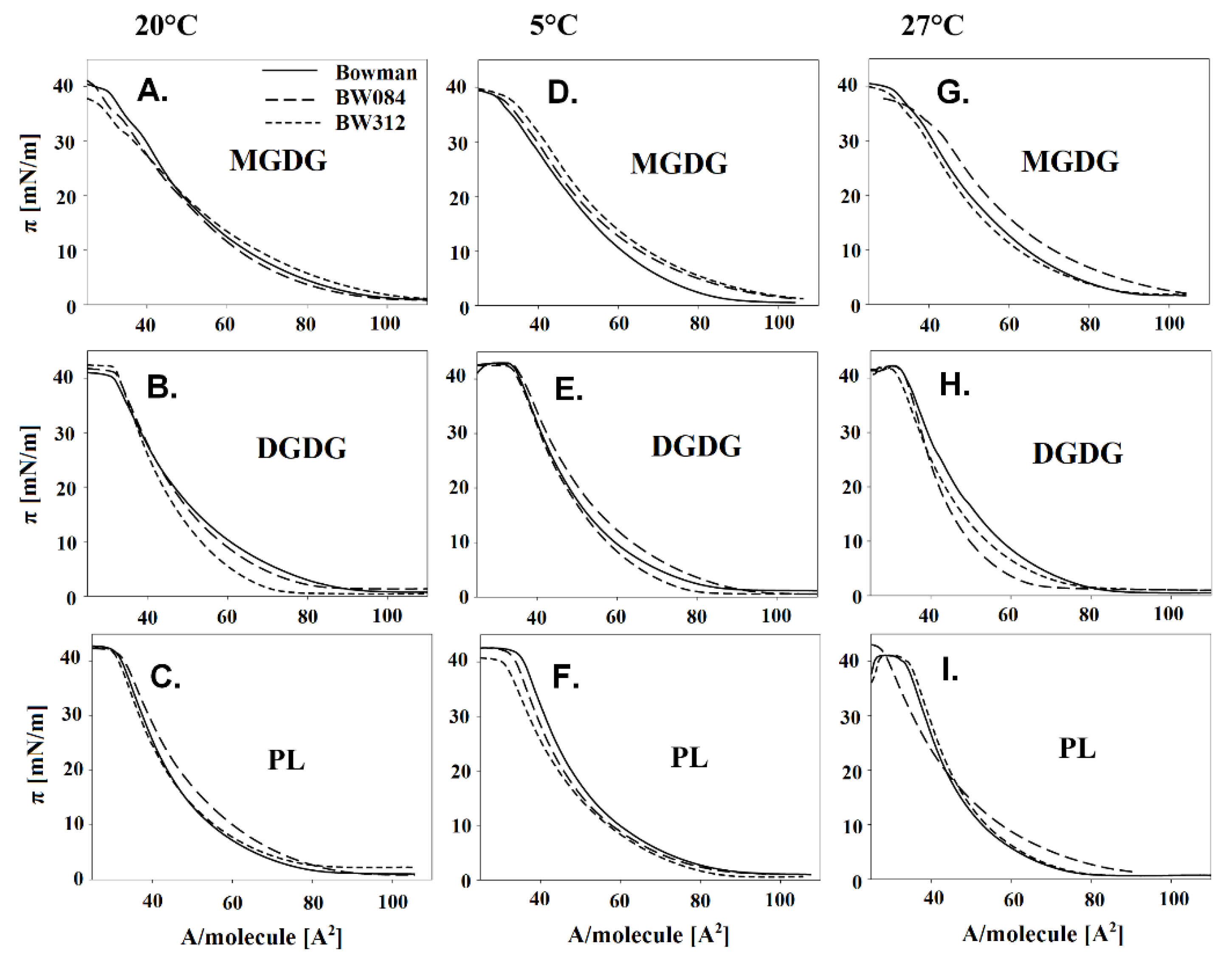
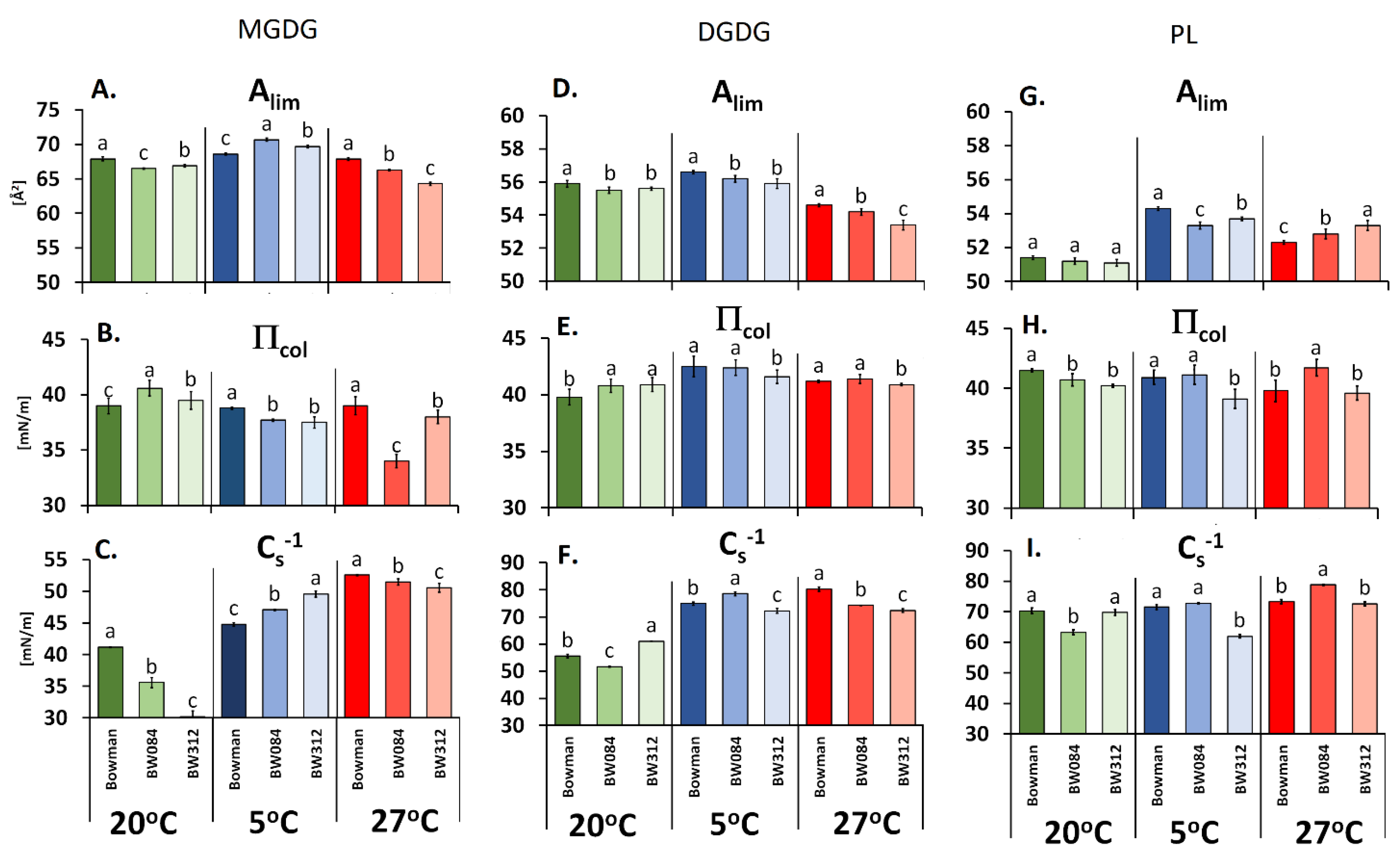
2.2. The Langmuir Monolayers and Their Physicochemical Parameters
2.3. Statistical Analysis
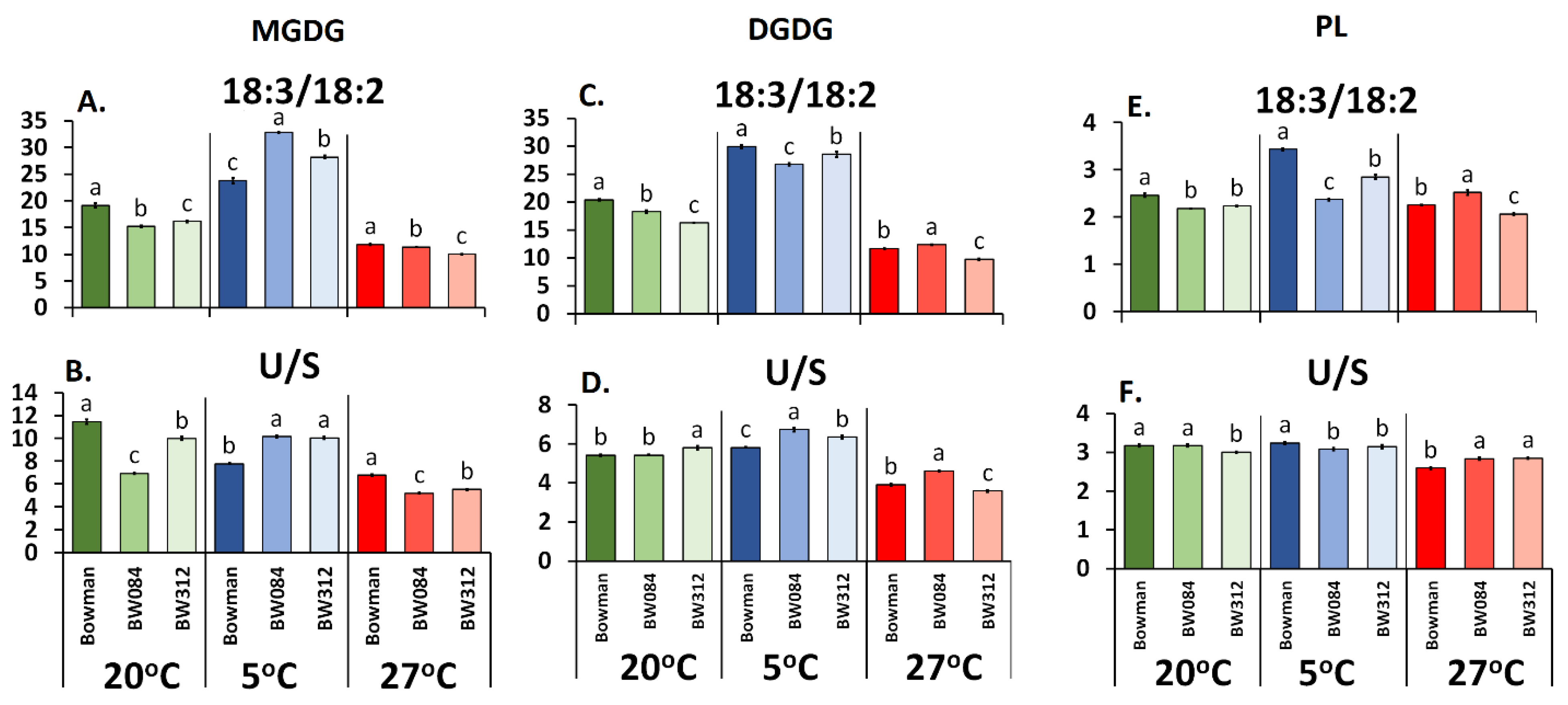
3. Results
3.1. Fatty Acid Composition of the Membrane Lipids from Barley Growing at 20 °C and the Physicochemical Properties of the Lipid Monolayers
3.2. Fatty Acid Composition of the Membrane Lipids from the Barley That Had Been Acclimated at 5 °C and the Physicochemical Properties of the Lipid Monolayers
3.3. Fatty Acid Composition of the Membrane Lipids from the Barley Acclimated at 27 °C and the Physicochemical Properties of the Lipid Monolayers
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Horváth, I.; Glatz, A.; Nakamoto, H.; Mishkind, M.L.; Munnik, T.; Saidi, Y.; Goloubinoff, P.; Harwood, J.L.; Vigh, L. Heat shock response in photosynthetic organisms: Membrane and lipid connections. Progr. Lipid Res. 2012, 51, 208–220. [Google Scholar] [CrossRef] [PubMed]
- Pearcy, R.W. Effect of growth temperature on the fatty acid composition of the leaf lipids in Atriplex lentiformis (Torr.) Wats. Plant Physiol. 1978, 61, 484–486. [Google Scholar] [CrossRef] [PubMed]
- Skoczowski, A.; Filek, M. Changes in fatty acids composition in the subcellular fraction from hypocotyls of winter rape growing at 2°C and 20°C. Plant Sci. 1994, 98, 127–133. [Google Scholar] [CrossRef]
- De la Roche, I.A.; Pomeroy, M.K.; Andrews, C.J. Changes in fatty acid composition in wheat cultivars of contrasting hardiness. Cryobiology 1975, 12, 506–512. [Google Scholar] [CrossRef]
- Yaeno, T.; Matsuda, O.; Iba, K. Role of chloroplast trienoic fatty acids in plant disease defense responses. Plant J. 2004, 40, 931–941. [Google Scholar] [CrossRef] [PubMed]
- Vicente, J.; Cascón, T.; Vicedo, B.; García-Agustín, P.; Hamberg, M.; Castresana, C. Role of 9-lipoxygenase and α-dioxygenase oxylipin pathways as modulators of local and systemic defense. Mol. Plant 2012, 5, 914–928. [Google Scholar] [CrossRef]
- Walley, J.W.; Kliebenstein, D.J.; Bostock, R.M.; Dehesh, K. Fatty acids and early detection of pathogens. Curr. Opin. Plant Biol. 2013, 16, 520–526. [Google Scholar] [CrossRef]
- Sadura, I.; Janeczko, A. Physiological and molecular mechanisms of brassinosteroid-induced tolerance to high and low temperature in plants. Biol. Plant. 2018, 62, 601–616. [Google Scholar] [CrossRef]
- Hayat, S.; Yusuf, M.; Bhardwaj, R.; Bajguz, A. Brassinosteroids: Plant Growth and Development; Springer: Singapore, 2019. [Google Scholar]
- Janeczko, A.; Hura, K.; Skoczowski, A.; Idzik, I.; Biesaga-Kościelniak, J.; Niemczyk, E. Temperature-dependent impact of 24-epibrassinolide on the fatty acid composition and sugar content in winter oilseed rape callus. Acta Physiol. Plant. 2009, 31, 71–79. [Google Scholar] [CrossRef]
- Janeczko, A.; Biesaga-Kościelniak, J.; Dziurka, M. 24-Epibrassinolide modifies seed composition in soybean, oilseed rape and wheat. Seed Sci. Technol. 2009, 37, 625–637. [Google Scholar] [CrossRef]
- Li, B.; Zhang, C.; Cao, B.; Qin, G.; Wang, W.; Tian, S. Brassinolide enhances cold stress tolerance of fruit by regulating plasma membrane proteins and lipids. Amino Acids 2012, 43, 2469–2480. [Google Scholar] [CrossRef] [PubMed]
- Fedina, E.; Yarin, A.; Mukhitova, F.; Blufard, A.; Chechetkin, I. Brassinosteroid-induced changes of lipid composition in leaves of Pisum sativum L. during senescence. Steroids 2017, 117, 25–28. [Google Scholar] [CrossRef] [PubMed]
- Filek, M.; Rudolphi-Skórska, E.; Sieprawska, A.; Kvasnica, M.; Janeczko, A. Regulation of the membrane structure by brassinosteroids and progesterone in winter wheat seedlings exposed to low temperature. Steroids 2017, 128, 37–45. [Google Scholar] [CrossRef] [PubMed]
- Janeczko, A.; Pociecha, E.; Dziurka, M.; Jurczyk, B.; Libik-Konieczny, M.; Oklestkova, J.; Pilarska, M.; Novak, O.; Filek, M.; Rudolphi-Skórska, E.; et al. Changes in content of steroid regulators during cold hardening of winter wheat—steroid physiological/biochemical activity and impact on frost tolerance. Plant Physiol. Biochem. 2019, 139, 215–228. [Google Scholar] [CrossRef]
- Sadura, I.; Pociecha, E.; Dziurka, M.; Oklestkova, J.; Novak, O.; Gruszka, D.; Janeczko, A. Mutations in the HvDWARF, HvCPD and HvBRI1 genes-involved in brassinosteroid biosynthesis/signalling: Altered photosynthetic efficiency, hormonal homeostasis and tolerance to high/low temperatures in barley. J. Plant Growth Regul. 2019, 38, 1062–1081. [Google Scholar] [CrossRef]
- Sadura, I.; Libik-Konieczny, M.; Jurczyk, B.; Gruszka, D.; Janeczko, A. Plasma membrane ATPase and the aquaporin HvPIP1 in barley brassinosteroid mutants acclimated to high and low temperature. J. Plant Physiol. 2020, 244, 153090. [Google Scholar] [CrossRef]
- Sadura, I.; Libik-Konieczny, M.; Jurczyk, B.; Gruszka, D.; Janeczko, A. The HSP transcript and protein accumulation in brassinosteroid barley mutants acclimated to low and high temperature. Int. J. Mol. Sci. 2020, 21, 1889. [Google Scholar] [CrossRef]
- Rudolphi-Skórska, E.; Sieprawska, A. Physicochemical techniques in description of interactions in model and native plant membranes under stressful conditions and in physiological processes. Acta Physiol. Plant. 2016, 38, 1–17. [Google Scholar] [CrossRef]
- Salvi, S.; Druka, A.; Milner, S.; Gruszka, D. Induced genetic variation, TILLING and NGS-based cloning. In Biotechnological Approaches to Barley Improvement; Biotechnology in Agriculture and Forestry; Kumlehn, J., Stein, N., Eds.; Springer: Berlin/Heidelberg, Germany, 2014; Volume 69, pp. 287–310. [Google Scholar]
- Gruszka, D.; Janeczko, A.; Dziurka, M.; Pociecha, E.; Oklestkova, J.; Szarejko, I. Barley brassinosteroid mutants provide an insight into phytohormonal homeostasis in plant reaction to drought stress. Front. Plant Sci. 2016, 7, 1824. [Google Scholar] [CrossRef]
- Dockter, C.; Gruszka, D.; Braumann, I.; Druka, A.; Druka, I.; Franckowiak, J.; Gough, S.P.; Janeczko, A.; Kurowska, M.; Lundqvist, J.; et al. Induced variations in brassinosteroid genes define barley height and sturdiness, and expand the green revolution genetic toolkit. Plant Physiol. 2014, 166, 1912–1927. [Google Scholar] [CrossRef]
- Bligh, E.G.; Dyer, W.J. A rapid method of total lipid extraction and purification. Can. J. Biochem. Physiol. 1959, 37, 911–917. [Google Scholar] [CrossRef] [PubMed]
- Gzyl-Malcher, B.; Filek, M.; Brezesinski, G.; Fischer, A. The influence of plant hormones on phospholipid monolayer stability. Z. Nat. C J. Biosci. 2007, 62, 55–60. [Google Scholar] [CrossRef] [PubMed]
- Cunniff, P.A. International Official Methods of Analysis, 16th ed.; AOAC Int.: Gaithersburg, MD, USA, 1999. [Google Scholar]
- Falcone, D.L.; Ogas, J.P.; Somerville, C.R. Regulation of membrane fatty acid composition by temperature in mutants of Arabidopsis with alterations in membrane lipid composition. BMC Plant Biol. 2004, 4, 17. [Google Scholar] [CrossRef]
- Boudière, L.; Michaud, M.; Petroutsos, D.; Rébeillé, F.; Falconet, D.; Bastien, O.; Roy, S.; Finazzi, G.; Rolland, N.; Jouhet, J.; et al. Glycerolipids in photosynthesis: Composition, synthesis and trafficking. Biochim. Biophys. Acta 2014, 1837, 470–480. [Google Scholar] [CrossRef] [PubMed]
- Jarvis, P.; Dormann, P.; Peto, C.A.; Lutes, J.; Benning, C.; Chory, J. Galactolipid deficiency and abnormal chloroplast development in the Arabidopsis MGD synthase 1 mutant. Proc. Natl. Acad. Sci. USA 2000, 97, 8175–8179. [Google Scholar] [CrossRef]
- Rudolphi-Skórska, E.; Filek, M.; Zembala, M. The Effects of the Structure and Composition of the Hydrophobic Parts of phosphatidylcholine-Containing Systems on Phosphatidylcholine Oxidation by Ozone. J. Membr. Biol. 2017, 250, 493–505. [Google Scholar] [CrossRef]
- Rudolphi-Skórska, E.; Filek, M.; Zembala, M. Mechanical and electrokinetic effect of polyamines/phospholipid interaction in model membranes. J. Membr. Biol. 2014, 247, 81–92. [Google Scholar] [CrossRef]
- Escribá, P.V.; González-Ros, J.M.; Gońi, F.M.; Kinnunen, P.K.; Vigh, L.; Sánchez-Magraner, L.; Fernandez, A.M.; Busquets, X.; Horvath, I.; Barcelo-Coblijn, G. Membranes: A meeting point for lipids, proteins and therapies. J. Cell Mol. Med. 2008, 12, 829–875. [Google Scholar] [CrossRef]
- Niu, Y.; Xiang, Y. An Overview of Biomembrane Functions in Plant Responses to High-Temperature Stress. Front. Plant Sci. 2018, 9, 91. [Google Scholar] [CrossRef]
- Sanchez, J.; Mangat, P.K.; Angeles-Shim, R.B. Weathering the Cold: Modifying Membrane and Storage Fatty Acid Composition of Seeds to Improve Cold Germination Ability in Upland Cotton (Gossypium hirsutum L.). Agronomy 2019, 9, 684. [Google Scholar] [CrossRef]
- Qu, T.; Liu, R.; Wang, W.; An, L.; Chen, T.; Liu, G.; Zhao, Z. Brassinosteroids regulate pectin methylesterase activity and AtPME41 expression in Arabidopsis under chilling stress. Cryobiology 2011, 63, 111–117. [Google Scholar] [CrossRef] [PubMed]
- Eremina, M.; Unterholzner, S.J.; Rathnayake, A.I.; Castellanos, M.; Khan, M.; Kugler, K.G.; May, S.T.; Mayer, K.F.X.; Rozhon, W.; Poppenberger, B. Brassinosteroids participate in the control of basal and acquired freezing tolerance of plants. Proc. Nat. Acad. Sci. USA 2016, 113, 5982–5991. [Google Scholar] [CrossRef] [PubMed]
| Temperature | Plants | Fatty Acids [mol %] | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 14:0 | 16:0 | 16:1n-9 | 16:1n-7 | 18:0 | 18:1n-9 | 18:1n-7 | 18:2n-6 | 18:3n-3 | 20:0 | 20:1 | ||
| MGDG | ||||||||||||
| 20 °C | Bowman | 0.321 b | 5.270 c | 0.049 b | 0.069 a | 2.695 c | 1.208 b | 0.190 b | 4.458 b | 85.335 a | 0.075 c | 0.046 a |
| 20 °C | BW084 | 0.584 a | 8.271 a | 0.094 a | 0.071 a | 4.201 a | 1.340 a | 0.252 a | 5.216 a | 79.399 c | 0.121 a | 0.049 a |
| 20 °C | BW312 | 0.298 b | 6.011 b | 0.035 c | 0.061 a | 3.040 b | 1.005 c | 0.207 b | 5.180 a | 83.800 b | 0.088 b | 0.031 b |
| 5 °C | Bowman | 0.257 b | 7.380 a | 0.041 b | 0.046 a | 3.900 a | 1.157 a | 0.191 a | 3.483 a | 82.928 c | 0.152 a | 0.078 a |
| 5 °C | BW084 | 0.233 b | 5.128 c | 0.032 b | 0.052 a | 3.767 b | 0.770 c | 0.173 a | 2.648 c | 86.709 a | 0.141 a | 0.034 c |
| 5 °C | BW312 | 0.293 a | 5.702 b | 0.057 a | 0.056 a | 3.288 c | 1.066 b | 0.195 a | 3.043 b | 85.840 b | 0.093 b | 0.056 b |
| 27 °C | Bowman | 0.387 c | 7.858 b | 0.031 c | 0.067 a | 4.874 c | 1.017 b | 0.211 a | 6.640 b | 78.427 a | 0.147 b | 0.030 a |
| 27 °C | BW084 | 0.484 b | 9.282 a | 0.074 b | 0.069 a | 6.646 a | 1.046 b | 0.222 a | 6.600 b | 74.951 b | 0.196 a | 0.026 a |
| 27 °C | BW312 | 0.582 a | 9.369 a | 0.222 a | 0.054 b | 5.767 b | 1.172 a | 0.205 a | 7.425 a | 74.527 c | 0.159 b | 0.030 a |
| DGDG | ||||||||||||
| 20 °C | Bowman | 0.228 c | 13.474 a | 0.103 b | 0.085 ab | 1.998 a | 1.606 b | 0.354 ab | 3.821 c | 77.904 a | 0.028 b | 0.061 a |
| 20 °C | BW084 | 0.326 a | 13.532 a | 0.106 b | 0.091 a | 1.899 b | 1.733 a | 0.370 a | 4.228 b | 77.151 b | 0.040 a | 0.061 a |
| 20 °C | BW312 | 0.246 b | 12.920 b | 0.189 a | 0.072 b | 1.689 c | 1.524 c | 0.336 b | 4.783 a | 77.803 a | 0.030 b | 0.057 a |
| 5 °C | Bowman | 0.178 a | 12.986 a | 0.124 a | 0.084 a | 1.582 b | 1.364 a | 0.341 a | 2.678 c | 80.131 c | 0.051 b | 0.075 a |
| 5 °C | BW084 | 0.140 b | 11.694 b | 0.037 c | 0.075 b | 1.167 c | 1.022 c | 0.314 a | 3.070 a | 82.039 a | 0.046 b | 0.070 a |
| 5 °C | BW312 | 0.132 b | 11.728 b | 0.054 b | 0.064 c | 1.809 a | 1.171 b | 0.320 a | 2.852 b | 81.413 b | 0.083 a | 0.047 b |
| 27 °C | Bowman | 0.320 b | 17.561 b | 0.058 c | 0.082 a | 2.630 a | 1.446 a | 0.344 a | 6.093 b | 70.943 b | 0.038 a | 0.056 a |
| 27 °C | BW084 | 0.298 b | 15.433 c | 0.065 b | 0.085 a | 2.217 b | 1.077 b | 0.328 a | 5.988 b | 73.967 a | 0.041 a | 0.042 b |
| 27 °C | BW312 | 0.562 a | 18.933 a | 0.220 a | 0.071 b | 2.595 a | 1.457 a | 0.331 a | 6.987 a | 68.074 c | 0.057 a | 0.026 c |
| PL | ||||||||||||
| 20 °C | Bowman | 0.165 b | 22.483 b | 0.045 b | 0.070 a | 1.200 b | 3.839 a | 0.427 b | 20.456 b | 50.346 a | 0.117 b | 0.220 a |
| 20 °C | BW084 | 0.169 b | 22.393 b | 0.054 a | 0.070 a | 1.264 ab | 3.654 b | 0.506 a | 22.228 a | 48.504 c | 0.174 a | 0.221 a |
| 20 °C | BW312 | 0.198 a | 23.345 a | 0.040 c | 0.069 a | 1.305 a | 2.661 c | 0.404 b | 22.005 a | 49.017 b | 0.124 b | 0.168 b |
| 5 °C | Bowman | 0.136 a | 22.258 b | 0.028 a | 0.052 b | 1.064 a | 2.409 a | 0.329 a | 16.411 c | 56.357 a | 0.087 c | 0.231 a |
| 5 °C | BW084 | 0.119 b | 23.162 a | 0.034 a | 0.061 a | 1.071 a | 1.992 b | 0.329 a | 21.462 a | 50.779 c | 0.151 a | 0.160 c |
| 5 °C | BW312 | 0.108 b | 22.730 a | 0.019 b | 0.048 b | 1.113 a | 2.393 a | 0.348 a | 18.826 b | 53.552 b | 0.112 b | 0.196 b |
| 27 °C | Bowman | 0.246 a | 25.844 a | 0.070 a | 0.072 b | 1.640 a | 3.585 a | 0.481 a | 20.618 b | 46.404 c | 0.106 c | 0.139 a |
| 27 °C | BW084 | 0.246 a | 24.314 b | 0.073 a | 0.083 a | 1.409 c | 2.493 c | 0.435 b | 19.856 c | 49.991 a | 0.143 b | 0.096 b |
| 27 °C | BW312 | 0.227 b | 24.122 b | 0.072 a | 0.069 b | 1.523 b | 2.691 b | 0.397 c | 22.808 a | 46.957 b | 0.156 a | 0.101 b |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rudolphi-Szydło, E.; Sadura, I.; Filek, M.; Gruszka, D.; Janeczko, A. The Impact of Mutations in the HvCPD and HvBRI1 Genes on the Physicochemical Properties of the Membranes from Barley Acclimated to Low/High Temperatures. Cells 2020, 9, 1125. https://doi.org/10.3390/cells9051125
Rudolphi-Szydło E, Sadura I, Filek M, Gruszka D, Janeczko A. The Impact of Mutations in the HvCPD and HvBRI1 Genes on the Physicochemical Properties of the Membranes from Barley Acclimated to Low/High Temperatures. Cells. 2020; 9(5):1125. https://doi.org/10.3390/cells9051125
Chicago/Turabian StyleRudolphi-Szydło, Elżbieta, Iwona Sadura, Maria Filek, Damian Gruszka, and Anna Janeczko. 2020. "The Impact of Mutations in the HvCPD and HvBRI1 Genes on the Physicochemical Properties of the Membranes from Barley Acclimated to Low/High Temperatures" Cells 9, no. 5: 1125. https://doi.org/10.3390/cells9051125
APA StyleRudolphi-Szydło, E., Sadura, I., Filek, M., Gruszka, D., & Janeczko, A. (2020). The Impact of Mutations in the HvCPD and HvBRI1 Genes on the Physicochemical Properties of the Membranes from Barley Acclimated to Low/High Temperatures. Cells, 9(5), 1125. https://doi.org/10.3390/cells9051125







