Ets Family Transcription Factor Fli-1 Promotes Leukocyte Recruitment and Production of IL-17A in the MRL/Lpr Mouse Model of Lupus Nephritis
Abstract
1. Introduction
2. Materials and Methods
2.1. Mice
2.2. Genotyping by Polymerase Chain Reaction (PCR)
2.3. Histopathology, Immunohistochemistry, and Immunofluorescence Staining
2.4. Quantitative Evaluation of Cytokines, Chemokines and Related Cell Signaling Molecules by Real-Time PCR
2.5. Measurement of Serum Cytokine IL-6 and IL-17A
2.6. Measurement of Serum Double-Strand DNA Levels and Urine Protein Levels
2.7. Statistics
3. Results
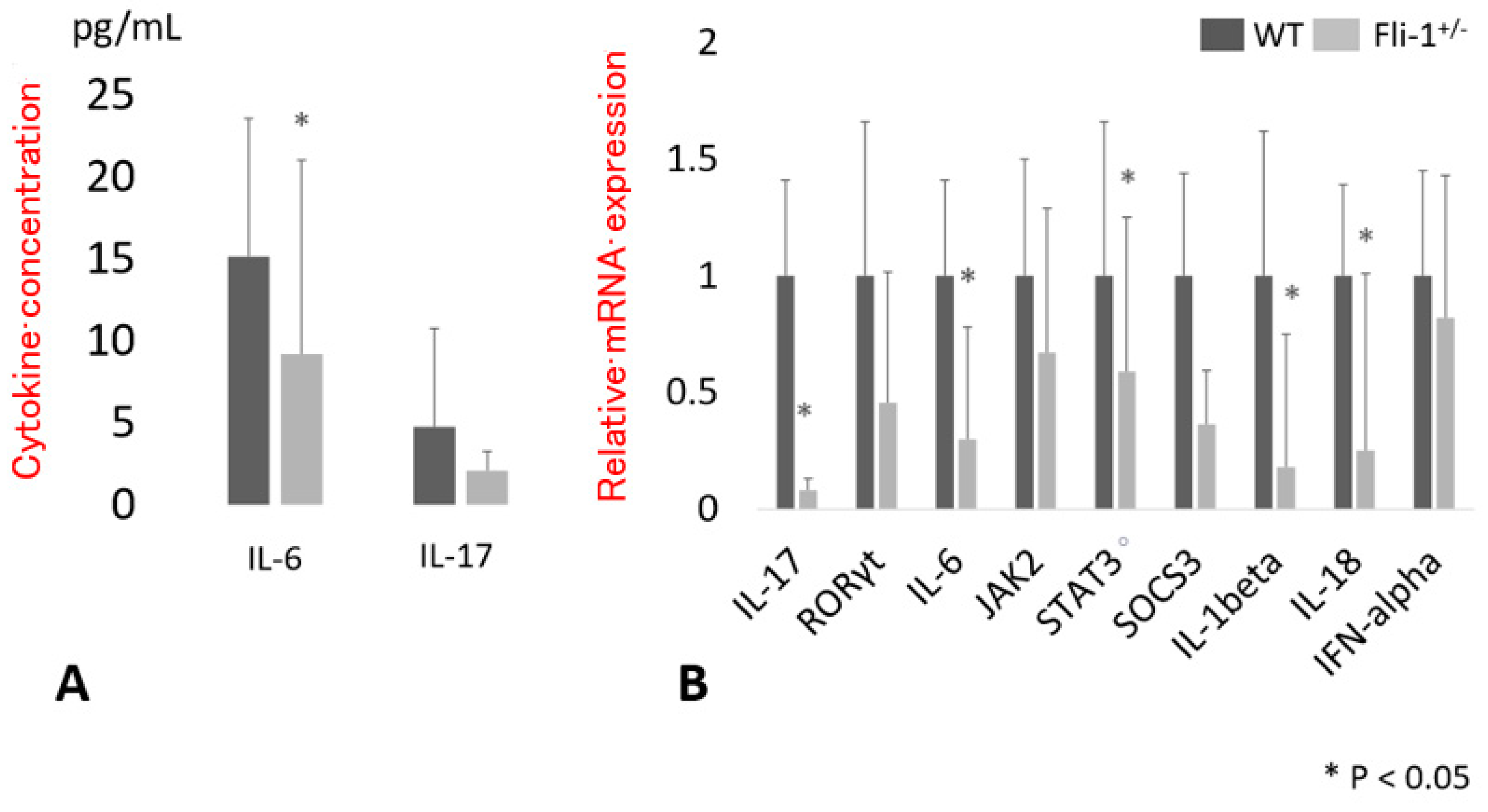
3.1. Serum Cytokine Levels in MRL/lpr Fli-1+/− Mice
3.2. Expression of Transcript Encoding IL-17A in the Kidney of MRL/lpr Fli-1+/− Mice
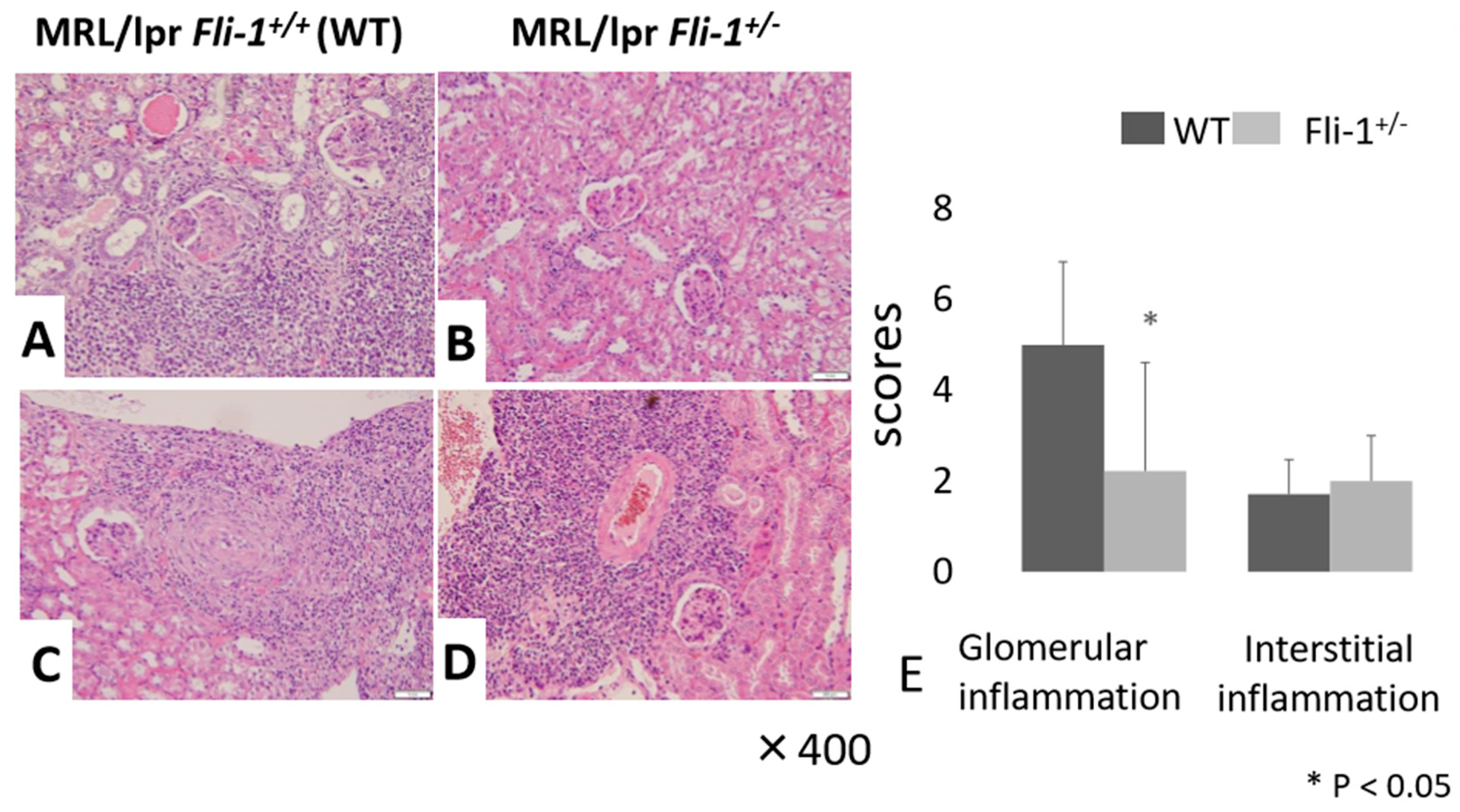
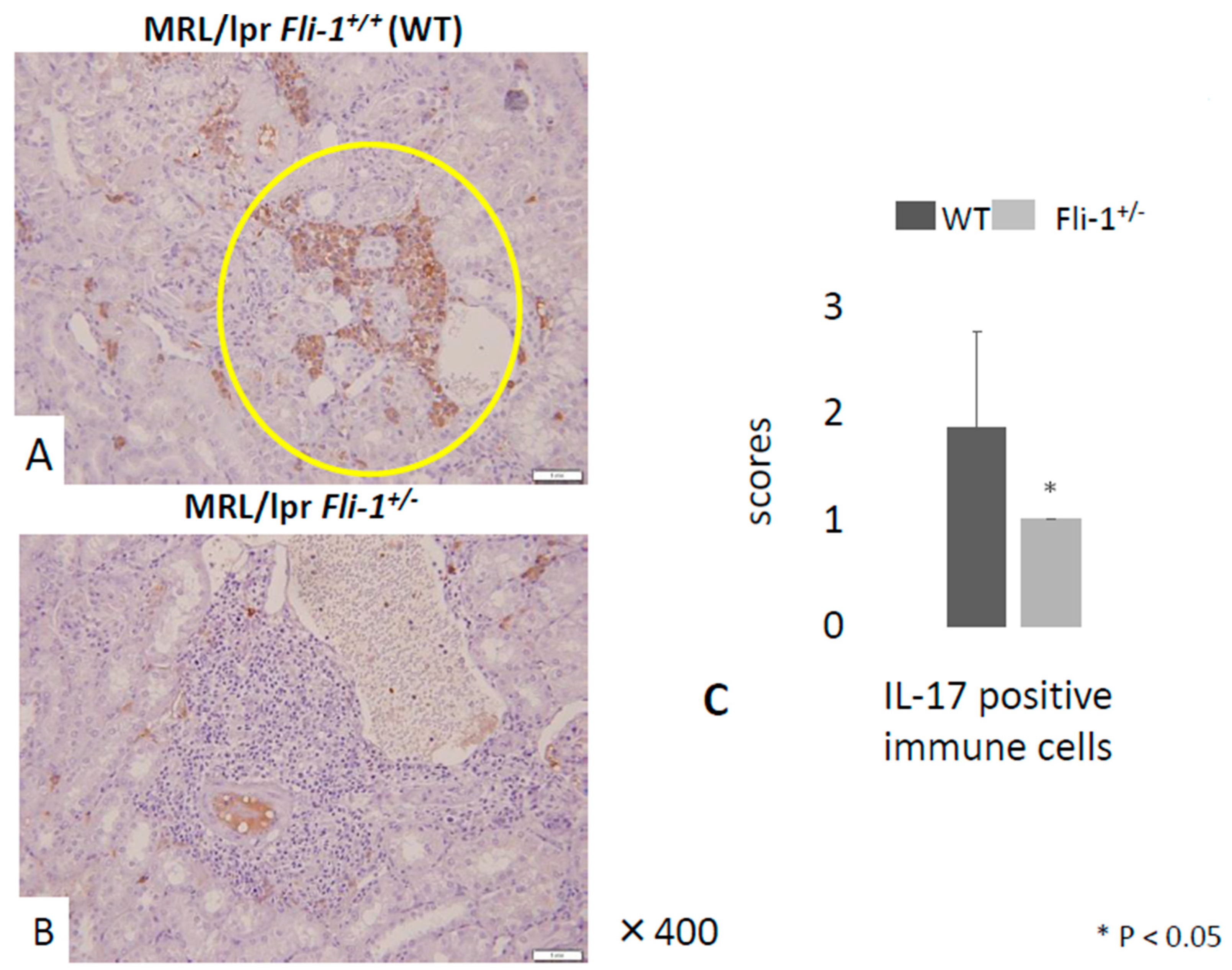
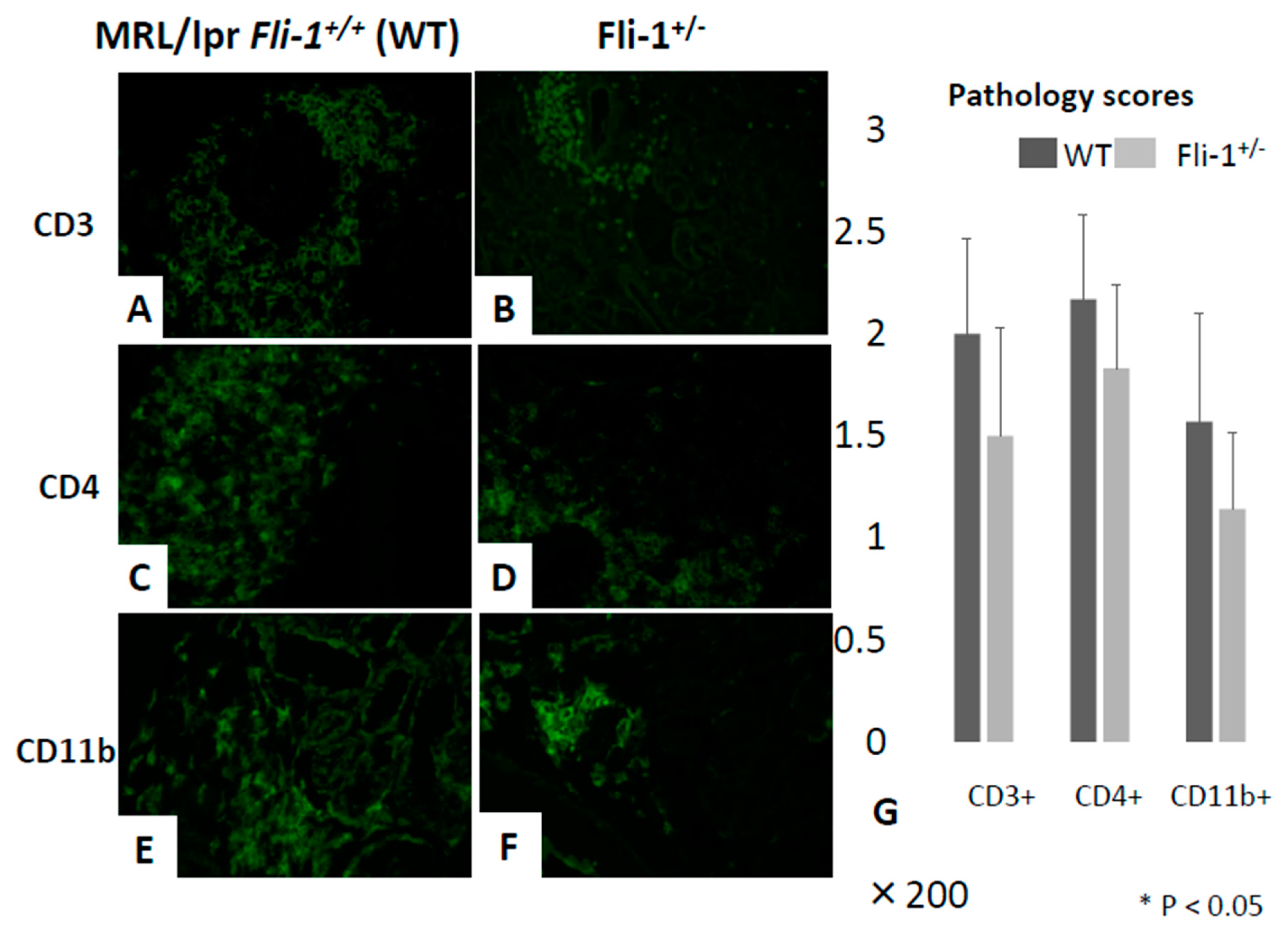
3.3. Reduced IL-17+ Cells in Renal Infiltrates from MRL/lpr Fli-1+/− Mice
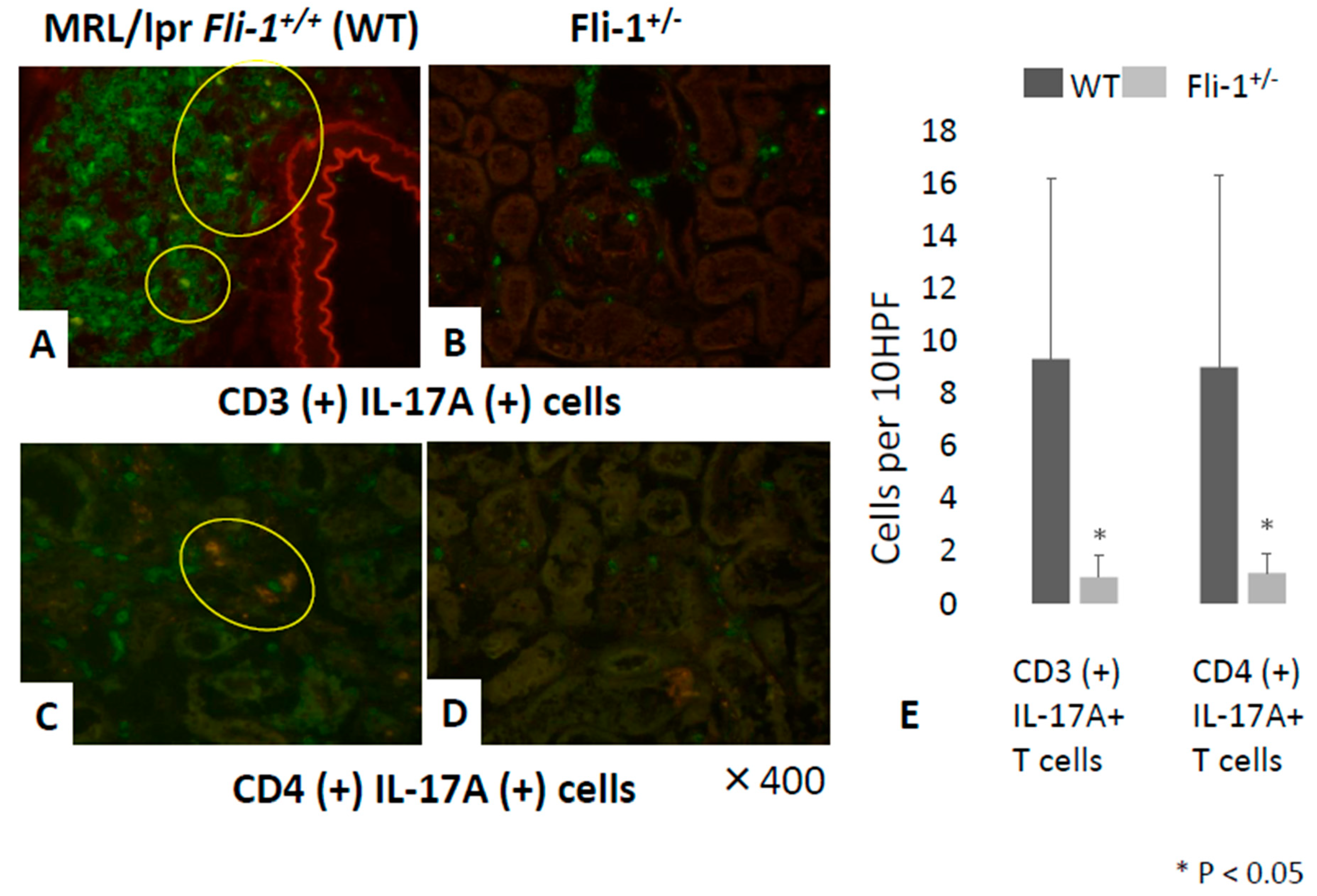
3.4. Reduced Numbers of CD3+ IL-17+ and CD4+ IL-17+ Cells in the Renal Interstitium of MRL/lpr Fli-1+/− Mice
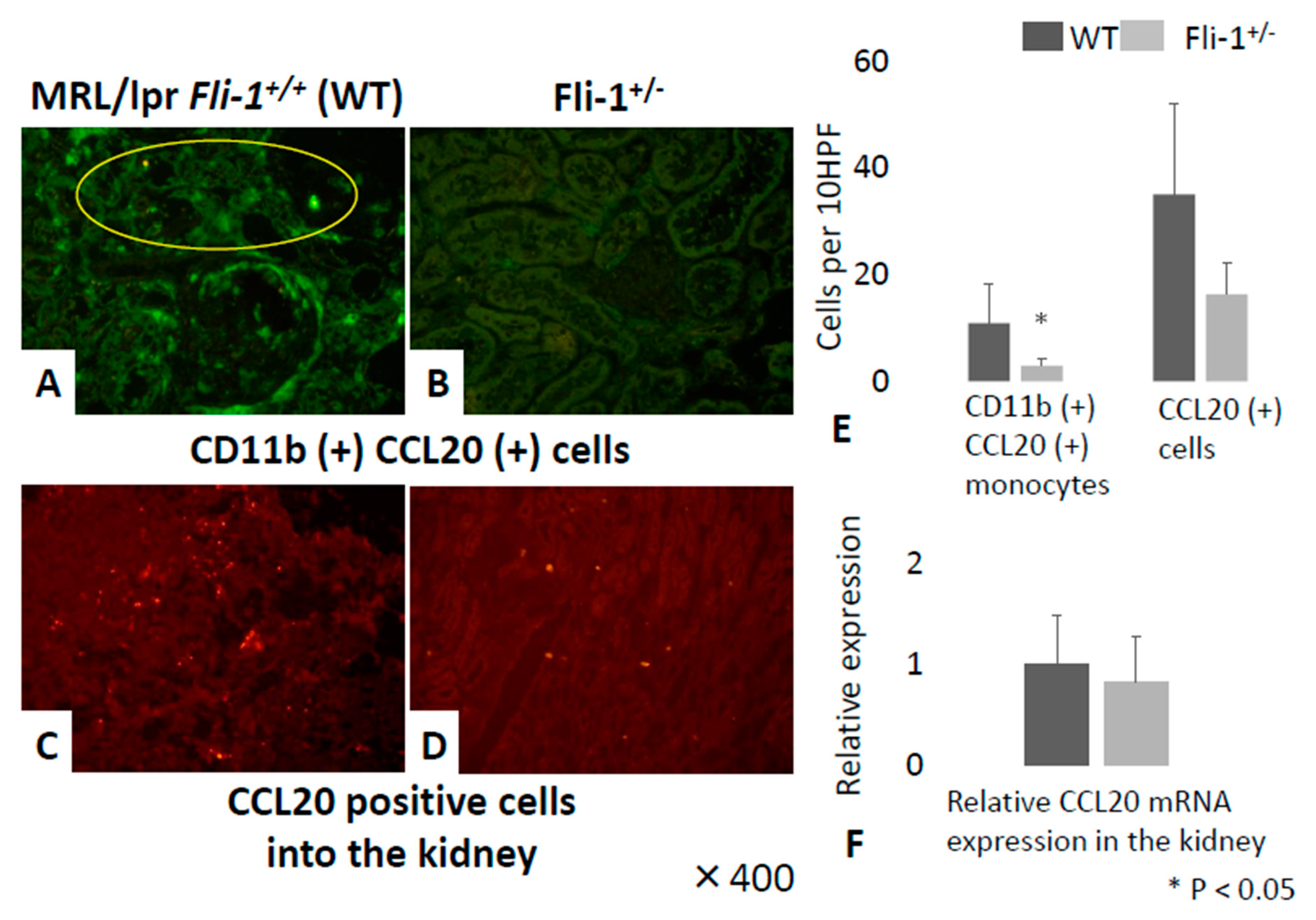
3.5. Detection of CCL20+CD11b+ Cells in Renal Tissue from MRL/lpr Fli-1+/− Mice
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| CCL20 | CC chemokine ligand 20 |
| CCR6 | CC chemokine receptor 6 |
| CXCL | chemokine (C-X-C) motif ligand |
| CXCR 3 | chemokine (C-X-C) motif receptor 3 |
| Fli-1 | Friend leukaemia integration 1 |
| G-CSF | granulocyte colony stimulating factor |
| IFN | interferon |
| IL | interleukin |
| JAK | Janus kinase |
| MCP-1 | monocyte chemoattractant protein-1 |
| RANTES | Regulated on Activation, Normal T cell Expressed and Secreted |
| RORγt | Retinoic Acid Related Orphan Nuclear Receptor gamma T |
| SLE | systemic lupus erythematosus |
| SOCS | suppressor of cytokine signaling |
| STAT | signal transducer and activator of transcription |
| Th 17 | T-helper 17 |
References
- Tsokos, G.C. Systemic lupus erythematosus. N. Engl. J. Med. 2011, 365, 2110–2121. [Google Scholar] [CrossRef] [PubMed]
- Wilson, P.C.; Kashgarian, M.; Moeckel, G. Interstitial inflammation and interstitial fibrosis and tubular atrophy predict renal survival in lupus nephritis. Clin. Kidney J. 2018, 11, 207–218. [Google Scholar] [CrossRef] [PubMed]
- Hsu, T.; Trojanowska, M.; Watson, D.K. Ets proteins in biological control and cancer. J. Cell Biochem. 2004, 91, 896–903. [Google Scholar] [CrossRef] [PubMed]
- Spyropoulos, D.D.; Pharr, P.N.; Lavenburg, K.R.; Jackers, P.; Papas, T.S.; Ogawa, M.; Watson, D.K. Hemorrhage, impaired hematopoiesis, and lethality in mouse embryos carrying a targeted disruption of the Fli1 transcription factor. Mol. Cell Biol. 2000, 20, 5643–5652. [Google Scholar] [CrossRef]
- Papas, T.S.; Bhat, N.K.; Spyropoulos, D.D.; Mjaatvedt, A.E.; Vournakis, J.; Seth, A.; Watson, D.K. Functional relationships among ETS gene family members. Leukemia 1997, 11, 557–566. [Google Scholar]
- Pusztaszeri, M.P.; Seelentag, W.; Bosman, F.T. Immunohistochemical expression of endothelial markers CD31, CD34, von Willebrand factor, and Fli-1 in normal human tissues. J. Histochem. Cytochem. 2006, 54, 385–395. [Google Scholar] [CrossRef]
- Zhang, X.K.; Gallant, S.; Molano, I.; Moussa, O.M.; Ruiz, P.; Spyropoulos, D.D.; Watson, D.K.; Gilkeson, G. Decreased expression of the Ets family transcription factor Fli-1 markedly prolongs survival and significantly reduces renal disease in MRL/lpr mice. J. Immunol. 2004, 173, 6481–6489. [Google Scholar] [CrossRef]
- Mathenia, J.; Reyes-Cortes, E.; Williams, S.; Molano, I.; Ruiz, P.; Watson, D.K.; Gilkeson, G.S.; Zhang, X.K. Impact of Fli-1 transcription factor on autoantibody and lupus nephritis in NZM2410 mice. Clin. Exp. Immunol. 2010, 162, 362–371. [Google Scholar] [CrossRef]
- Zhang, L.; Eddy, A.; Teng, Y.T.; Fritzler, M.; Kluppel, M.; Melet, F.; Bernstein, A. An immunological renal disease in transgenic mice that overexpress Fli-1, a member of the ets family of transcription factor genes. Mol. Cell Biol. 1995, 15, 6961–6970. [Google Scholar] [CrossRef]
- Suzuki, E.; Karam, E.; Williams, S.; Watson, D.K.; Gilkeson, G.; Zhang, X.K. Fli-1 transcription factor affects glomerulonephritis development by regulating expression of monocyte chemoattractant protein-1 in endothelial cells in the kidney. Clin. Immunol. 2012, 145, 201–208. [Google Scholar] [CrossRef]
- Lennard Richard, M.L.; Sato, S.; Suzuki, E.; Williams, S.; Nowling, T.K.; Zhang, X.K. The Fli-1 transcription factor regulates the expression of CCL5/RANTES. J. Immunol. 2014, 193, 2661–2668. [Google Scholar] [CrossRef] [PubMed]
- Lennard Richard, M.L.; Brandon, D.; Lou, N.; Sato, S.; Caldwell, T.; Nowling, T.K.; Gilkeson, G.; Zhang, X.K. Acetylation impacts Fli-1-driven regulation of granulocyte colony stimulating factor. Eur. J. Immunol. 2016, 46, 2322–2332. [Google Scholar] [CrossRef] [PubMed]
- Sato, S.; Lennard Richard, M.; Brandon, D.; Jones Buie, J.N.; Oates, J.C.; Gilkeson, G.S.; Zhang, X.K. A critical role of the transcription factor Fli-1 in murine lupus development by regulation of interleukin-6 expression. Arthritis Rheumatol. 2014, 66, 3436–3444. [Google Scholar] [CrossRef] [PubMed]
- Sato, S.; Zhang, X.K. The Friend leukaemia virus integration 1 (Fli-1) transcription factor affects lupus nephritis development by regulating inflammatory cell infiltration into the kidney. Clin. Exp. Immunol. 2014, 177, 102–109. [Google Scholar] [CrossRef]
- Lou, N.; Lennard Richard, M.L.; Yu, J.; Kindy, M.; Zhang, X.K. The Fli-1 transcription factor is a critical regulator for controlling the expression of chemokine C-X-C motif ligand 2 (CXCL2). Mol. Immunol. 2017, 81, 59–66. [Google Scholar] [CrossRef]
- Georgiou, P.; Maroulakou, I.; Green, J.; Dantis, P.; Romanospica, V.; Kottaridis, S.; Lautenberger, J.; Watson, D.; Papas, T.; Fischinger, P.; et al. Expression of ets family of genes in systemic lupus erythematosus and Sjogren’s syndrome. Int. J. Oncol. 1996, 9, 9–18. [Google Scholar] [CrossRef]
- Mackay, M.; Oswald, M.; Sanchez-Guerrero, J.; Lichauco, J.; Aranow, C.; Kotkin, S.; Korsunsky, I.; Gregersen, P.K.; Diamond, B. Molecular signatures in systemic lupus erythematosus: Distinction between disease flare and infection. Lupus. Sci. Med. 2016, 3, e000159. [Google Scholar] [CrossRef]
- Yu, S.L.; Kuan, W.P.; Wong, C.K.; Li, E.K.; Tam, L.S. Immunopathological roles of cytokines, chemokines, signaling molecules, and pattern-recognition receptors in systemic lupus erythematosus. Clin. Dev. Immunol. 2012, 2012, 715190. [Google Scholar] [CrossRef]
- Park, H.; Li, Z.; Yang, X.O.; Chang, S.H.; Nurieva, R.; Wang, Y.H.; Wang, Y.; Hood, L.; Zhu, Z.; Tian, Q.; et al. A distinct lineage of CD4 T cells regulates tissue inflammation by producing interleukin 17. Nat. Immunol. 2005, 6, 1133–1141. [Google Scholar] [CrossRef]
- Chen, S.Y.; Liu, M.F.; Kuo, P.Y.; Wang, C.R. Upregulated expression of STAT3/IL-17 in patients with systemic lupus erythematosus. Clin. Rheumatol. 2019, 38, 1361–1366. [Google Scholar] [CrossRef]
- Zickert, A.; Amoudruz, P.; Sundström, Y.; Rönnelid, J.; Malmström, V.; Gunnarsson, I. IL-17 and IL-23 in lupus nephritis - association to histopathology and response to treatment. BMC Immunol. 2015, 16, 7. [Google Scholar] [CrossRef] [PubMed]
- Raymond, W.; Ostli-Eilertsen, G.; Griffiths, S.; Nossent, J. IL-17A levels in systemic lupus erythematosus associated with inflammatory markers and lower rates of malignancy and heart damage: Evidence for a dual role. Eur. J. Rheumatol. 2017, 4, 29–35. [Google Scholar] [CrossRef] [PubMed]
- Koga, T.; Ichinose, K.; Tsokos, G.C. T cells and IL-17 in lupus nephritis. Clin. Immunol. 2017, 185, 95–99. [Google Scholar] [CrossRef] [PubMed]
- Crispín, J.C.; Oukka, M.; Bayliss, G.; Cohen, R.A.; Van Beek, C.A.; Stillman, I.E.; Kyttaris, V.C.; Juang, Y.T.; Tsokos, G.C. Expanded double negative T cells in patients with systemic lupus erythematosus produce IL-17 and infiltrate the kidneys. J. Immunol. 2008, 181, 8761–8766. [Google Scholar] [CrossRef]
- Amarilyo, G.; Lourenço, E.V.; Shi, F.D.; La Cava, A. IL-17 promotes murine lupus. J. Immunol. 2014, 193, 540–543. [Google Scholar] [CrossRef]
- Turner, J.E.; Paust, H.J.; Steinmetz, O.M.; Peters, A.; Riedel, J.H.; Erhardt, A.; Wegscheid, C.; Velden, J.; Fehr, S.; Mittrücker, H.W.; et al. CCR6 recruits regulatory T cells and Th17 cells to the kidney in glomerulonephritis. J. Am. Soc. Nephrol. 2010, 21, 974–985. [Google Scholar] [CrossRef]
- Chen, Z.; Laurence, A.; Kanno, Y.; Pacher-Zavisin, M.; Zhu, B.M.; Tato, C.; Yoshimura, A.; Hennighausen, L.; O’Shea, J.J. Selective regulatory function of Socs3 in the formation of IL-17-secreting T cells. Proc. Natl. Acad. Sci. USA 2006, 103, 8137–8142. [Google Scholar] [CrossRef]
- Hodge, D.R.; Li, D.; Qi, S.M.; Farrar, W.L. IL-6 induces expression of the Fli-1 proto-oncogene via STAT3. Biochem. Biophys. Res. Commun. 2002, 292, 287–291. [Google Scholar] [CrossRef]
- Sundararaj, K.P.; Thiyagarajan, T.; Molano, I.; Basher, F.; Powers, T.W.; Drake, R.R.; Nowling, T.K. FLI1 Levels Impact CXCR3 Expression and Renal Infiltration of T Cells and Renal Glycosphingolipid Metabolism in the MRL/lpr Lupus Mouse Strain. J. Immunol. 2015, 195, 5551–5560. [Google Scholar] [CrossRef]
- Enghard, P.; Humrich, J.Y.; Rudolph, B.; Rosenberger, S.; Biesen, R.; Kuhn, A.; Manz, R.; Hiepe, F.; Radbruch, A.; Burmester, G.R.; et al. CXCR3+CD4+ T cells are enriched in inflamed kidneys and urine and provide a new biomarker for acute nephritis flares in systemic lupus erythematosus patients. Arthritis Rheum. 2009, 60, 199–206. [Google Scholar] [CrossRef]
- Steinmetz, O.M.; Turner, J.E.; Paust, H.J.; Lindner, M.; Peters, A.; Heiss, K.; Velden, J.; Hopfer, H.; Fehr, S.; Krieger, T.; et al. CXCR3 Mediates Renal Th1 and Th17 Immune Response in Murine Lupus Nephritis. J. Immunol. 2009, 183, 4693–4704. [Google Scholar] [CrossRef]
- Li, Q.; Laumonnier, Y.; Syrovets, T.; Simmet, T. Recruitment of CCR6-expressing Th17 cells by CCL20 secreted from plasmin-stimulated macrophages. Acta Biochim. Biophys. Sin. (Shanghai). 2013, 45, 593–600. [Google Scholar] [CrossRef]
- Hirota, K.; Yoshitomi, H.; Hashimoto, M.; Maeda, S.; Teradaira, S.; Sugimoto, N.; Yamaguchi, T.; Nomura, T.; Ito, H.; Nakamura, T.; et al. Preferential recruitment of CCR6-expressing Th17 cells to inflamed joints via CCL20 in rheumatoid arthritis and its animal model. J. Exp. Med. 2007, 204, 2803–2812. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, T.; Paust, H.J.; Krebs, C.F.; Turner, J.E.; Kaffke, A.; Bennstein, S.B.; Koyro, T.; Peters, A.; Velden, J.; Hünemörder, S.; et al. Function of the Th17/interleukin-17A immune response in murine lupus nephritis. Arthritis Rheumatol. 2015, 67, 475–487. [Google Scholar] [CrossRef] [PubMed]
- Kwan, B.C.; Tam, L.S.; Lai, K.B.; Lai, F.M.; Li, E.K.; Wang, G.; Chow, K.M.; Li, P.K.; Szeto, C.C. The gene expression of type 17 T-helper cell-related cytokines in the urinary sediment of patients with systemic lupus erythematosus. Rheumatology (Oxford) 2009, 48, 1491–1497. [Google Scholar] [CrossRef] [PubMed]
| Primer Name | Forward Primer | Reverse Primer |
|---|---|---|
| IL-17 | 5′ACTTTCAGGGTCGAGAAGA-3′ | 5′TTCTGAATCTGCCTCTGAAT-3′ |
| RORγt | 5′AGCTTTGTGCAGATCTAAGG-3′ | 5′TGTCCTCCTCAGTAGGGTAG-3′ |
| SOCS3 | 5′ACCTTCAGCTCCAAAAGCGAGTAC-3′ | 5′CGCTCCAGTAGAATCCGCTCTC-3′ |
| IL-1β | 5′CTTCAGGCAGGCAGTATCACTCAT-3′ | 5′TCTAATGGGAACGTCACACACCAG-3′ |
| IL-18 | 5′GCTGTGACCCTCTCTGTGAA-3′ | 5′GGCAAGCAAGAAAGTGTCCT-3′ |
| IFN-α | 5′CATTCTGCAATGACCTCCAC-3′ | 5′TCAGGGGAAATTCCTGCAC-3′ |
| CCL20 | 5′ATGGCCTGCGGTGGCAAGCGTCTG-3′ | 5′TAGGCTGAGGAGGTTCACAGCCCT-3′ |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sato, S.; Zhang, X.K.; Temmoku, J.; Fujita, Y.; Matsuoka, N.; Yashiro-Furuya, M.; Asano, T.; Kobayashi, H.; Watanabe, H.; Migita, K. Ets Family Transcription Factor Fli-1 Promotes Leukocyte Recruitment and Production of IL-17A in the MRL/Lpr Mouse Model of Lupus Nephritis. Cells 2020, 9, 714. https://doi.org/10.3390/cells9030714
Sato S, Zhang XK, Temmoku J, Fujita Y, Matsuoka N, Yashiro-Furuya M, Asano T, Kobayashi H, Watanabe H, Migita K. Ets Family Transcription Factor Fli-1 Promotes Leukocyte Recruitment and Production of IL-17A in the MRL/Lpr Mouse Model of Lupus Nephritis. Cells. 2020; 9(3):714. https://doi.org/10.3390/cells9030714
Chicago/Turabian StyleSato, Shuzo, Xian K. Zhang, Jumpei Temmoku, Yuya Fujita, Naoki Matsuoka, Makiko Yashiro-Furuya, Tomoyuki Asano, Hiroko Kobayashi, Hiroshi Watanabe, and Kiyoshi Migita. 2020. "Ets Family Transcription Factor Fli-1 Promotes Leukocyte Recruitment and Production of IL-17A in the MRL/Lpr Mouse Model of Lupus Nephritis" Cells 9, no. 3: 714. https://doi.org/10.3390/cells9030714
APA StyleSato, S., Zhang, X. K., Temmoku, J., Fujita, Y., Matsuoka, N., Yashiro-Furuya, M., Asano, T., Kobayashi, H., Watanabe, H., & Migita, K. (2020). Ets Family Transcription Factor Fli-1 Promotes Leukocyte Recruitment and Production of IL-17A in the MRL/Lpr Mouse Model of Lupus Nephritis. Cells, 9(3), 714. https://doi.org/10.3390/cells9030714





