TUG1 Is a Regulator of AFP and Serves as Prognostic Marker in Non-Hepatitis B Non-Hepatitis C Hepatocellular Carcinoma †
Abstract
1. Introduction
2. Materials and Methods
2.1. Cell Culture
2.2. Human Hepatoma Specimens
2.3. Microarray Analysis
2.4. Quantitative Reverse Transcription-PCR (qRT-PCR)
2.5. Immunoblot Analysis
2.6. Establishment of TUG1 Knockdown and TUG1-Activating Cell Lines
2.7. Detection of Cell Cycle Progression
2.8. Cell Proliferation Assay
2.9. Soft Agar Assay
2.10. TUNEL Assay
2.11. Public Datasets
2.12. Statistical Analysis
3. Results
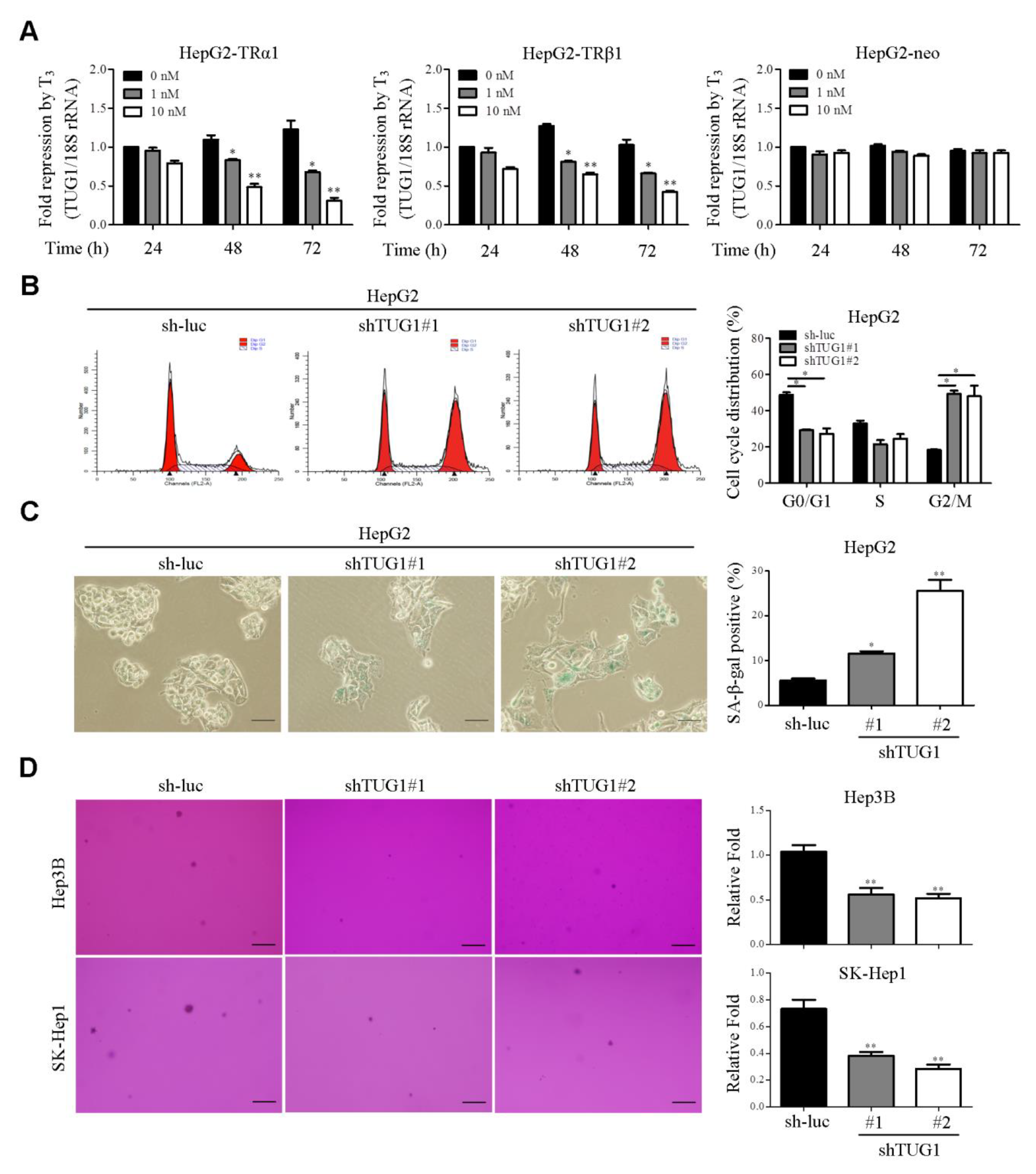
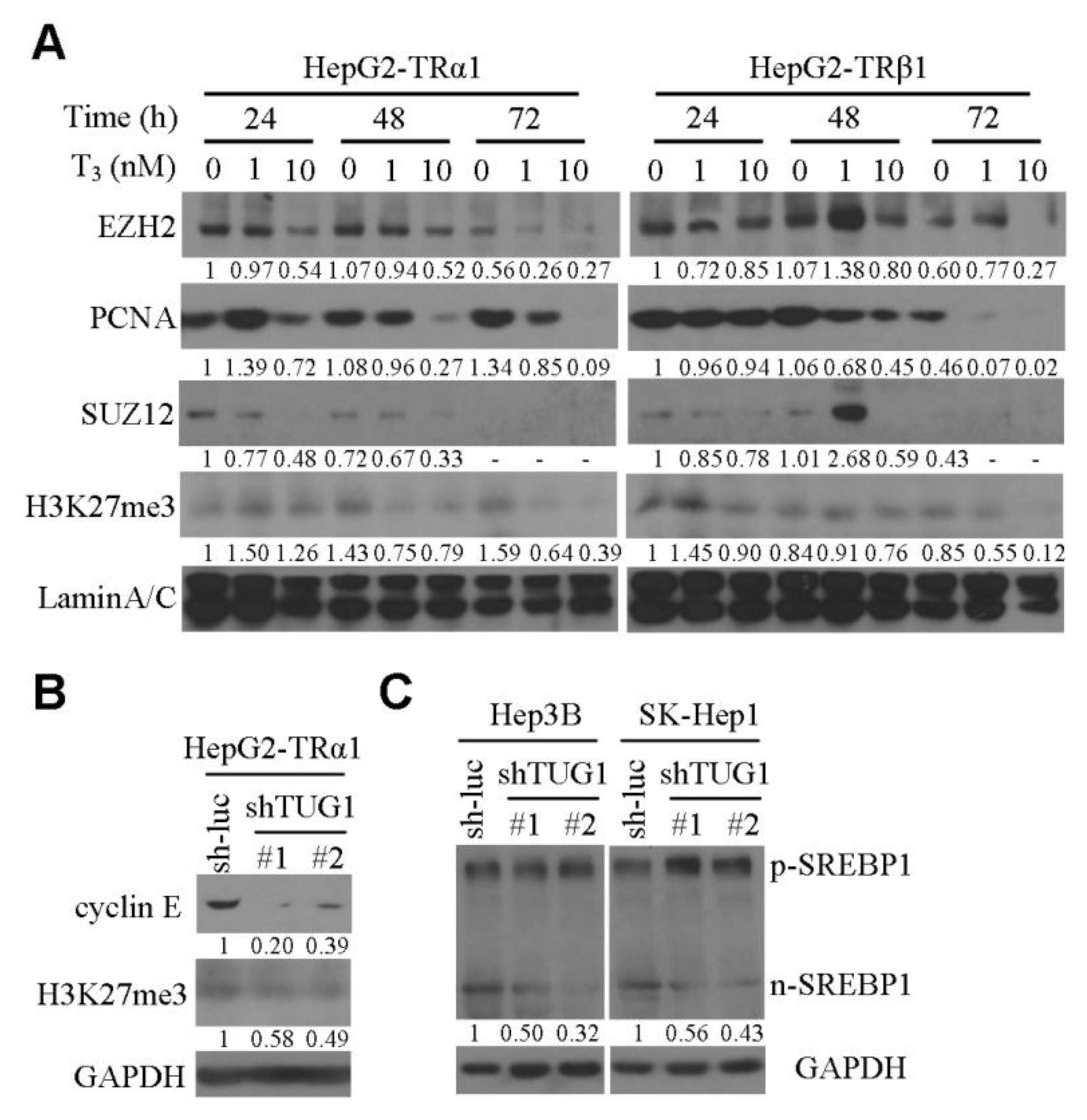
3.1. TUG1 is Significantly Downregulated by T3/TR In Vitro and Regulates Cell Cycle Progression, Cellular Senescence and Soft Agar Colony Formation
3.2. AFP is Significantly Downregulated by the T3/TR/TUG1 Axis
3.3. TUG1 is Involved in T3-Mediated Functions In Vitro
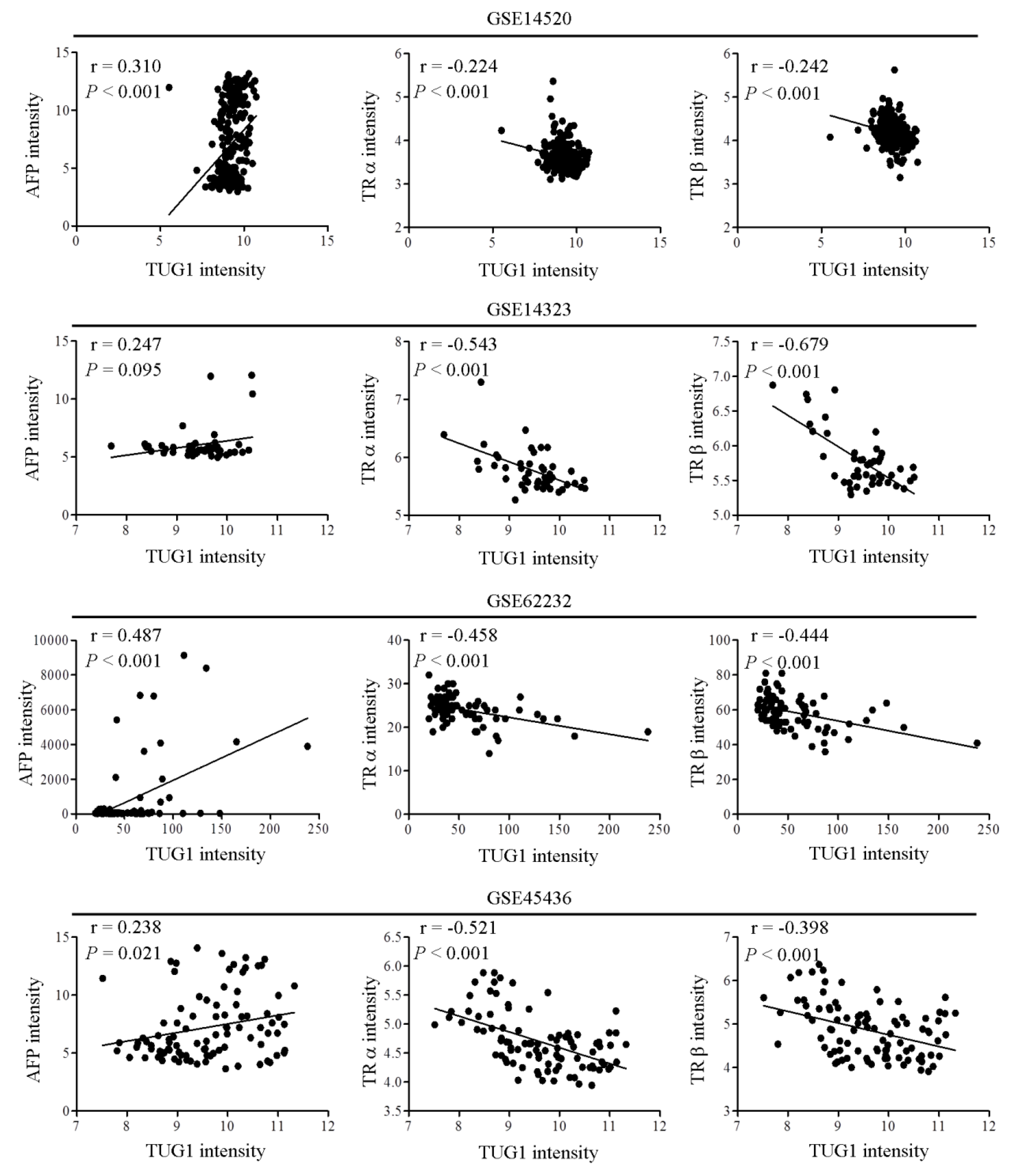
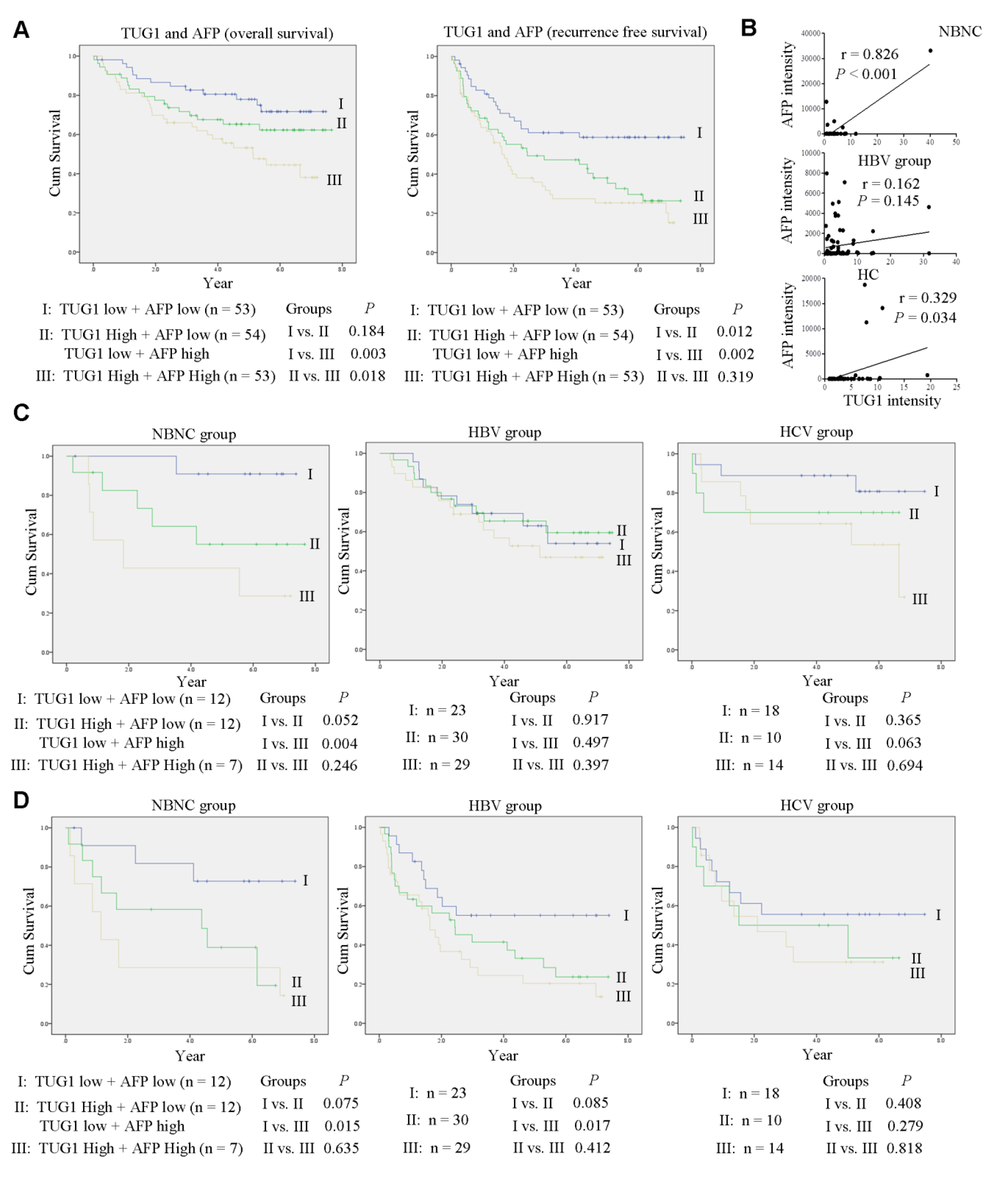
3.4. Clinical Correlation between TUG1 and AFP
3.5. Combined Expression of TUG1 and AFP is a Stronger Predictor of OS and RFS in Patients with NBNC-HCC
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| T3 | thyroid hormone |
| TR | thyroid hormone receptor |
| HCC | hepatocellular carcinoma |
| AFP | alpha-fetoprotein |
| lncRNA | long non-coding RNA |
| TUG1 | taurine upregulated gene 1 |
| qRT-PCR | quantitative reverse transcription-PCR |
| OS | overall survival |
| RFS | recurrence-free survival |
References
- Chi, H.C.; Chen, S.L.; Tsai, C.Y.; Chuang, W.Y.; Huang, Y.H.; Tsai, M.M.; Wu, S.M.; Sun, C.P.; Yeh, C.T.; Lin, K.H. Thyroid hormone suppresses hepatocarcinogenesis via DAPK2 and SQSTM1-dependent selective autophagy. Autophagy 2016, 12, 2271–2285. [Google Scholar] [CrossRef] [PubMed]
- Wu, S.M.; Cheng, W.L.; Lin, C.D.; Lin, K.H. Thyroid hormone actions in liver cancer. Cell Mol. Life Sci. 2013, 70, 1915–1936. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.J.; Tsai, N.M.; Liu, Y.C.; Ho, L.I.; Hsieh, H.F.; Yen, C.Y.; Harn, H.J. Telomerase activity in human hepatocellular carcinoma: Parallel correlation with human telomerase reverse transcriptase (hTERT) mRNA isoform expression but not with cell cycle modulators or c-Myc expression. Eur. J. Surg Oncol. 2002, 28, 225–234. [Google Scholar] [CrossRef] [PubMed]
- Ando, S.; Sarlis, N.J.; Oldfield, E.H.; Yen, P.M. Somatic mutation of TRbeta can cause a defect in negative regulation of TSH in a TSH-secreting pituitary tumor. J. Clin. Endocrinol. Metab. 2001, 86, 5572–5576. [Google Scholar] [CrossRef][Green Version]
- Perra, A.; Plateroti, M.; Columbano, A. T3/TRs axis in hepatocellular carcinoma: New concepts for an old pair. Endocr. Relat. Cancer 2016, 23, R353–R369. [Google Scholar] [CrossRef][Green Version]
- Puzianowska-Kuznicka, M.; Krystyniak, A.; Madej, A.; Cheng, S.Y.; Nauman, J. Functionally impaired TR mutants are present in thyroid papillary cancer. J. Clin. Endocrinol. Metab. 2002, 87, 1120–1128. [Google Scholar] [CrossRef]
- Hassan, M.M.; Kaseb, A.; Li, D.; Patt, Y.Z.; Vauthey, J.N.; Thomas, M.B.; Curley, S.A.; Spitz, M.R.; Sherman, S.I.; Abdalla, E.K.; et al. Association between hypothyroidism and hepatocellular carcinoma: A case-control study in the United States. Hepatology 2009, 49, 1563–1570. [Google Scholar] [CrossRef]
- Liangpunsakul, S.; Chalasani, N. Is hypothyroidism a risk factor for non-alcoholic steatohepatitis? J. Clin. Gastroenterol. 2003, 37, 340–343. [Google Scholar] [CrossRef]
- El-Serag, H.B. Epidemiology of viral hepatitis and hepatocellular carcinoma. Gastroenterology 2012, 142, 1264–1273 e1261. [Google Scholar] [CrossRef]
- Chang, M.H.; Chen, C.J.; Lai, M.S.; Hsu, H.M.; Wu, T.C.; Kong, M.S.; Liang, D.C.; Shau, W.Y.; Chen, D.S. Universal hepatitis B vaccination in Taiwan and the incidence of hepatocellular carcinoma in children. Taiwan Childhood Hepatoma Study Group. N. Engl. J. Med. 1997, 336, 1855–1859. [Google Scholar] [CrossRef]
- Testino, G.; Leone, S.; Borro, P. Alcohol and hepatocellular carcinoma: A review and a point of view. World J. Gastroenterol. 2014, 20, 15943–15954. [Google Scholar] [CrossRef] [PubMed]
- Regimbeau, J.M.; Colombat, M.; Mognol, P.; Durand, F.; Abdalla, E.; Degott, C.; Degos, F.; Farges, O.; Belghiti, J. Obesity and diabetes as a risk factor for hepatocellular carcinoma. Liver Transpl. 2004, 10, S69–S73. [Google Scholar] [CrossRef]
- Benedict, M.; Zhang, X. Non-alcoholic fatty liver disease: An expanded review. World J. Hepatol. 2017, 9, 715–732. [Google Scholar] [CrossRef]
- Di Bisceglie, A.M. Issues in screening and surveillance for hepatocellular carcinoma. Gastroenterology 2004, 127, S104–S107. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.C.; Crowe, A.J.; Barton, M.C. p53-mediated repression of alpha-fetoprotein gene expression by specific DNA binding. Mol. Cell Biol. 1999, 19, 1279–1288. [Google Scholar] [CrossRef] [PubMed]
- Kojima, K.; Takata, A.; Vadnais, C.; Otsuka, M.; Yoshikawa, T.; Akanuma, M.; Kondo, Y.; Kang, Y.J.; Kishikawa, T.; Kato, N.; et al. MicroRNA122 is a key regulator of alpha-fetoprotein expression and influences the aggressiveness of hepatocellular carcinoma. Nat. Commun. 2011, 2, 338. [Google Scholar] [CrossRef]
- Schmitt, A.M.; Chang, H.Y. Long Noncoding RNAs in Cancer Pathways. Cancer Cell 2016, 29, 452–463. [Google Scholar] [CrossRef]
- Shao, H.; Dong, D.; Shao, F. Long non-coding RNA TUG1-mediated down-regulation of KLF4 contributes to metastasis and the epithelial-to-mesenchymal transition of colorectal cancer by miR-153-1. Cancer Manag. Res. 2019, 11, 8699–8710. [Google Scholar] [CrossRef]
- Hui, B.; Xu, Y.; Zhao, B.; Ji, H.; Ma, Z.; Xu, S.; He, Z.; Wang, K.; Lu, J. Overexpressed long noncoding RNA TUG1 affects the cell cycle, proliferation, and apoptosis of pancreatic cancer partly through suppressing RND3 and MT2A. Onco Targets 2019, 12, 1043–1057. [Google Scholar] [CrossRef]
- Zhu, J.; Shi, H.; Liu, H.; Wang, X.; Li, F. Long non-coding RNA TUG1 promotes cervical cancer progression by regulating the miR-138-5p-SIRT1 axis. Oncotarget 2017, 8, 65253–65264. [Google Scholar] [CrossRef]
- Huang, M.D.; Chen, W.M.; Qi, F.Z.; Sun, M.; Xu, T.P.; Ma, P.; Shu, Y.Q. Long non-coding RNA TUG1 is up-regulated in hepatocellular carcinoma and promotes cell growth and apoptosis by epigenetically silencing of KLF2. Mol. Cancer 2015, 14, 165. [Google Scholar] [CrossRef]
- Lin, Y.H.; Wu, M.H.; Huang, Y.H.; Yeh, C.T.; Cheng, M.L.; Chi, H.C.; Tsai, C.Y.; Chung, I.H.; Chen, C.Y.; Lin, K.H. Taurine up-regulated gene 1 functions as a master regulator to coordinate glycolysis and metastasis in hepatocellular carcinoma. Hepatology 2018, 67, 188–203. [Google Scholar] [CrossRef]
- He, C.; Liu, Z.; Jin, L.; Zhang, F.; Peng, X.; Xiao, Y.; Wang, X.; Lyu, Q.; Cai, X. lncRNA TUG1-Mediated Mir-142-3p Downregulation Contributes to Metastasis and the Epithelial-to-Mesenchymal Transition of Hepatocellular Carcinoma by Targeting ZEB1. Cell Physiol. Biochem. 2018, 48, 1928–1941. [Google Scholar] [CrossRef]
- He, H.; Chen, D.; Cui, S.; Piao, H.; Tang, H.; Wang, X.; Ye, P.; Jin, S. Identification of a long noncoding RNAmediated competitive endogenous RNA network in hepatocellular carcinoma. Oncol. Rep. 2019, 42, 745–752. [Google Scholar] [CrossRef]
- Lv, J.; Kong, Y.; Gao, Z.; Liu, Y.; Zhu, P.; Yu, Z. LncRNA TUG1 interacting with miR-144 contributes to proliferation, migration and tumorigenesis through activating the JAK2/STAT3 pathway in hepatocellular carcinoma. Int. J. Biochem. Cell Biol. 2018, 101, 19–28. [Google Scholar] [CrossRef]
- Li, J.; Zhang, Q.; Fan, X.; Mo, W.; Dai, W.; Feng, J.; Wu, L.; Liu, T.; Li, S.; Xu, S.; et al. The long noncoding RNA TUG1 acts as a competing endogenous RNA to regulate the Hedgehog pathway by targeting miR-132 in hepatocellular carcinoma. Oncotarget 2017, 8, 65932–65945. [Google Scholar] [CrossRef]
- Chen, R.N.; Huang, Y.H.; Yeh, C.T.; Liao, C.H.; Lin, K.H. Thyroid hormone receptors suppress pituitary tumor transforming gene 1 activity in hepatoma. Cancer Res. 2008, 68, 1697–1706. [Google Scholar] [CrossRef]
- Wu, S.M.; Huang, Y.H.; Yeh, C.T.; Tsai, M.M.; Liao, C.H.; Cheng, W.L.; Chen, W.J.; Lin, K.H. Cathepsin H regulated by the thyroid hormone receptors associate with tumor invasion in human hepatoma cells. Oncogene 2011, 30, 2057–2069. [Google Scholar] [CrossRef]
- Huang, Y.H.; Tseng, Y.H.; Lin, W.R.; Hung, G.; Chen, T.C.; Wang, T.H.; Lee, W.C.; Yeh, C.T. HBV polymerase overexpression due to large core gene deletion enhances hepatoma cell growth by binding inhibition of microRNA-100. Oncotarget 2016, 7, 9448–9461. [Google Scholar] [CrossRef]
- Roessler, S.; Jia, H.L.; Budhu, A.; Forgues, M.; Ye, Q.H.; Lee, J.S.; Thorgeirsson, S.S.; Sun, Z.; Tang, Z.Y.; Qin, L.X.; et al. A unique metastasis gene signature enables prediction of tumor relapse in early-stage hepatocellular carcinoma patients. Cancer Res. 2010, 70, 10202–10212. [Google Scholar] [CrossRef]
- Mas, V.R.; Maluf, D.G.; Archer, K.J.; Yanek, K.; Kong, X.; Kulik, L.; Freise, C.E.; Olthoff, K.M.; Ghobrial, R.M.; McIver, P.; et al. Genes involved in viral carcinogenesis and tumor initiation in hepatitis C virus-induced hepatocellular carcinoma. Mol. Med. 2009, 15, 85–94. [Google Scholar] [CrossRef]
- Schulze, K.; Imbeaud, S.; Letouze, E.; Alexandrov, L.B.; Calderaro, J.; Rebouissou, S.; Couchy, G.; Meiller, C.; Shinde, J.; Soysouvanh, F.; et al. Exome sequencing of hepatocellular carcinomas identifies new mutational signatures and potential therapeutic targets. Nat. Genet. 2015, 47, 505–511. [Google Scholar] [CrossRef] [PubMed]
- Shao, W.; Espenshade, P.J. Expanding roles for SREBP in metabolism. Cell Metab. 2012, 16, 414–419. [Google Scholar] [CrossRef] [PubMed]
- Yamamoto, K.; Imamura, H.; Matsuyama, Y.; Kume, Y.; Ikeda, H.; Norman, G.L.; Shums, Z.; Aoki, T.; Hasegawa, K.; Beck, Y.; et al. AFP, AFP-L3, DCP, and GP73 as markers for monitoring treatment response and recurrence and as surrogate markers of clinicopathological variables of HCC. J. Gastroenterol. 2010, 45, 1272–1282. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Zhu, M.; Wang, Q.; Hou, Y.; Li, L.; Weng, H.; Zhao, Y.; Chen, D.; Ding, H.; Guo, J.; et al. Alpha-fetoprotein inhibits autophagy to promote malignant behaviour in hepatocellular carcinoma cells by activating PI3K/AKT/mTOR signalling. Cell Death Dis. 2018, 9, 1027. [Google Scholar] [CrossRef] [PubMed]
- Hanahan, D.; Weinberg, R.A. Hallmarks of cancer: The next generation. Cell 2011, 144, 646–674. [Google Scholar] [CrossRef]
- Wu, S.M.; Cheng, W.L.; Liao, C.J.; Chi, H.C.; Lin, Y.H.; Tseng, Y.H.; Tsai, C.Y.; Chen, C.Y.; Lin, S.L.; Chen, W.J.; et al. Negative modulation of the epigenetic regulator, UHRF1, by thyroid hormone receptors suppresses liver cancer cell growth. Int. J. Cancer 2015, 137, 37–49. [Google Scholar] [CrossRef]
- Li, Y.; Zhang, T.; Zhang, Y.; Zhao, X.; Wang, W. Targeting the FOXM1-regulated long noncoding RNA TUG1 in osteosarcoma. Cancer Sci. 2018, 109, 3093–3104. [Google Scholar] [CrossRef]
- Lin, Y.H.; Huang, Y.H.; Wu, M.H.; Wu, S.M.; Chi, H.C.; Liao, C.J.; Chen, C.Y.; Tseng, Y.H.; Tsai, C.Y.; Tsai, M.M.; et al. Thyroid hormone suppresses cell proliferation through endoglin-mediated promotion of p21 stability. Oncogene 2013, 32, 3904–3914. [Google Scholar] [CrossRef]
- Liao, C.H.; Yeh, C.T.; Huang, Y.H.; Wu, S.M.; Chi, H.C.; Tsai, M.M.; Tsai, C.Y.; Liao, C.J.; Tseng, Y.H.; Lin, Y.H.; et al. Dickkopf 4 positively regulated by the thyroid hormone receptor suppresses cell invasion in human hepatoma cells. Hepatology 2012, 55, 910–920. [Google Scholar] [CrossRef]
- Huang, P.S.; Lin, Y.H.; Chi, H.C.; Chen, P.Y.; Huang, Y.H.; Yeh, C.T.; Wang, C.S.; Lin, K.H. Thyroid hormone inhibits growth of hepatoma cells through induction of miR-214. Sci. Rep. 2017, 7, 14868. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.H.; Wu, M.H.; Huang, Y.H.; Yeh, C.T.; Chi, H.C.; Tsai, C.Y.; Chuang, W.Y.; Yu, C.J.; Chung, I.H.; Chen, C.Y.; et al. Thyroid hormone negatively regulates tumorigenesis through suppression of BC200. Endocr. Relat. Cancer 2018, 25, 967–979. [Google Scholar] [CrossRef] [PubMed]
- Chi, H.C.; Chen, S.L.; Liao, C.J.; Liao, C.H.; Tsai, M.M.; Lin, Y.H.; Huang, Y.H.; Yeh, C.T.; Wu, S.M.; Tseng, Y.H.; et al. Thyroid hormone receptors promote metastasis of human hepatoma cells via regulation of TRAIL. Cell Death Differ. 2012, 19, 1802–1814. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Chi, H.C.; Chen, S.L.; Cheng, Y.H.; Lin, T.K.; Tsai, C.Y.; Tsai, M.M.; Lin, Y.H.; Huang, Y.H.; Lin, K.H. Chemotherapy resistance and metastasis-promoting effects of thyroid hormone in hepatocarcinoma cells are mediated by suppression of FoxO1 and Bim pathway. Cell Death Dis. 2016, 7, e2324. [Google Scholar] [CrossRef] [PubMed]
- Yan, K.S.; Lin, C.Y.; Liao, T.W.; Peng, C.M.; Lee, S.C.; Liu, Y.J.; Chan, W.P.; Chou, R.H. EZH2 in Cancer Progression and Potential Application in Cancer Therapy: A Friend or Foe? Int. J. Mol. Sci. 2017, 18, 1172. [Google Scholar] [CrossRef] [PubMed]
- Laugesen, A.; Hojfeldt, J.W.; Helin, K. Molecular Mechanisms Directing PRC2 Recruitment and H3K27 Methylation. Mol. Cell 2019, 74, 8–18. [Google Scholar] [CrossRef]
- Zhang, E.; He, X.; Yin, D.; Han, L.; Qiu, M.; Xu, T.; Xia, R.; Xu, L.; Yin, R.; De, W. Increased expression of long noncoding RNA TUG1 predicts a poor prognosis of gastric cancer and regulates cell proliferation by epigenetically silencing of p57. Cell Death Dis. 2016, 7, e2109. [Google Scholar] [CrossRef]
- Nie, L.; Wei, Y.; Zhang, F.; Hsu, Y.H.; Chan, L.C.; Xia, W.; Ke, B.; Zhu, C.; Deng, R.; Tang, J.; et al. CDK2-mediated site-specific phosphorylation of EZH2 drives and maintains triple-negative breast cancer. Nat. Commun. 2019, 10, 5114. [Google Scholar] [CrossRef]
- Yen, C.C.; Huang, Y.H.; Liao, C.Y.; Liao, C.J.; Cheng, W.L.; Chen, W.J.; Lin, K.H. Mediation of the inhibitory effect of thyroid hormone on proliferation of hepatoma cells by transforming growth factor-beta. J. Mol. Endocrinol. 2006, 36, 9–21. [Google Scholar] [CrossRef]
- Zhang, H.; Cao, D.; Zhou, L.; Zhang, Y.; Guo, X.; Li, H.; Chen, Y.; Spear, B.T.; Wu, J.W.; Xie, Z.; et al. ZBTB20 is a sequence-specific transcriptional repressor of alpha-fetoprotein gene. Sci. Rep. 2015, 5, 11979. [Google Scholar] [CrossRef]
- Kajiyama, Y.; Tian, J.; Locker, J. Regulation of alpha-fetoprotein expression by Nkx2.8. Mol. Cell Biol. 2002, 22, 6122–6130. [Google Scholar] [CrossRef] [PubMed]
- Paul, S.B.; Gulati, M.S.; Sreenivas, V.; Madan, K.; Gupta, A.K.; Mukhopadhyay, S.; Acharya, S.K. Evaluating patients with cirrhosis for hepatocellular carcinoma: Value of clinical symptomatology, imaging and alpha-fetoprotein. Oncology 2007, 72, 117–123. [Google Scholar] [CrossRef] [PubMed]
- Song, P.P.; Xia, J.F.; Inagaki, Y.; Hasegawa, K.; Sakamoto, Y.; Kokudo, N.; Tang, W. Controversies regarding and perspectives on clinical utility of biomarkers in hepatocellular carcinoma. World J. Gastroenterol. 2016, 22, 262–274. [Google Scholar] [CrossRef] [PubMed]
- Shen, Q.; Fan, J.; Yang, X.R.; Tan, Y.; Zhao, W.; Xu, Y.; Wang, N.; Niu, Y.; Wu, Z.; Zhou, J.; et al. Serum DKK1 as a protein biomarker for the diagnosis of hepatocellular carcinoma: A large-scale, multicentre study. Lancet Oncol. 2012, 13, 817–826. [Google Scholar] [CrossRef]
- Tomimaru, Y.; Eguchi, H.; Nagano, H.; Wada, H.; Kobayashi, S.; Marubashi, S.; Tanemura, M.; Tomokuni, A.; Takemasa, I.; Umeshita, K.; et al. Circulating microRNA-21 as a novel biomarker for hepatocellular carcinoma. J. Hepatol. 2012, 56, 167–175. [Google Scholar] [CrossRef] [PubMed]
- Tangkijvanich, P.; Chanmee, T.; Komtong, S.; Mahachai, V.; Wisedopas, N.; Pothacharoen, P.; Kongtawelert, P. Diagnostic role of serum glypican-3 in differentiating hepatocellular carcinoma from non-malignant chronic liver disease and other liver cancers. J. Gastroenterol. Hepatol. 2010, 25, 129–137. [Google Scholar] [CrossRef]
- Shang, S.; Plymoth, A.; Ge, S.; Feng, Z.; Rosen, H.R.; Sangrajrang, S.; Hainaut, P.; Marrero, J.A.; Beretta, L. Identification of osteopontin as a novel marker for early hepatocellular carcinoma. Hepatology 2012, 55, 483–490. [Google Scholar] [CrossRef]
- Yuan, W.; Sun, Y.; Liu, L.; Zhou, B.; Wang, S.; Gu, D. Circulating LncRNAs Serve as Diagnostic Markers for Hepatocellular Carcinoma. Cell Physiol. Biochem. 2017, 44, 125–132. [Google Scholar] [CrossRef]
- Li, J.; Wang, X.; Tang, J.; Jiang, R.; Zhang, W.; Ji, J.; Sun, B. HULC and Linc00152 Act as Novel Biomarkers in Predicting Diagnosis of Hepatocellular Carcinoma. Cell Physiol. Biochem. 2015, 37, 687–696. [Google Scholar] [CrossRef]

| Parameters | n = 160 | AFP Mean a ± SE | p b |
|---|---|---|---|
| Age (years) | |||
| <65 | 101 | 1006 ± 366.9 | 0.8646 |
| ≥65 | 59 | 1108 ± 456.2 | |
| Gender | |||
| Male | 80 | 1167 ± 514.8 | 0.0106 |
| Female | 80 | 920.5 ± 250 | |
| Cirrhosis | |||
| No | 97 | 573.8 ± 171.9 | 0.0407 |
| Yes | 63 | 1767 ± 667.9 | |
| Viral status | |||
| NBNC | 31 | 1852 ± 1134 | |
| HBV | 82 | 772.6 ± 181.3 | 0.6999 |
| HCV | 42 | 1100 ± 603 | |
| Tumor type | |||
| Solitary | 127 | 848 ± 298.9 | 0.1794 |
| Multiple | 33 | 1794 ± 765.5 | |
| Tumor size | |||
| <5 cm | 93 | 917.9 ± 296.8 | 0.6052 |
| ≥5 cm | 67 | 1218 ± 545.3 | |
| Vascular invasion | |||
| No | 81 | 555.1 ± 214.5 | 0.083 |
| Yes | 79 | 1545 ± 530.6 | |
| Pathological stage | |||
| I | 76 | 640.3 ± 235.2 | 0.4044 |
| II | 52 | 1448 ± 728.5 | |
| III | 32 | 1345 ± 572.9 | |
| Grading | |||
| 1 | 4 | 688.3 ± 687.1 | 0.0648 |
| 2 | 112 | 699.2 ± 227.2 | |
| 3 | 44 | 1953 ± 852.4 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lin, Y.-H.; Wu, M.-H.; Huang, Y.-H.; Yeh, C.-T.; Lin, K.-H. TUG1 Is a Regulator of AFP and Serves as Prognostic Marker in Non-Hepatitis B Non-Hepatitis C Hepatocellular Carcinoma. Cells 2020, 9, 262. https://doi.org/10.3390/cells9020262
Lin Y-H, Wu M-H, Huang Y-H, Yeh C-T, Lin K-H. TUG1 Is a Regulator of AFP and Serves as Prognostic Marker in Non-Hepatitis B Non-Hepatitis C Hepatocellular Carcinoma. Cells. 2020; 9(2):262. https://doi.org/10.3390/cells9020262
Chicago/Turabian StyleLin, Yang-Hsiang, Meng-Han Wu, Ya-Hui Huang, Chau-Ting Yeh, and Kwang-Huei Lin. 2020. "TUG1 Is a Regulator of AFP and Serves as Prognostic Marker in Non-Hepatitis B Non-Hepatitis C Hepatocellular Carcinoma" Cells 9, no. 2: 262. https://doi.org/10.3390/cells9020262
APA StyleLin, Y.-H., Wu, M.-H., Huang, Y.-H., Yeh, C.-T., & Lin, K.-H. (2020). TUG1 Is a Regulator of AFP and Serves as Prognostic Marker in Non-Hepatitis B Non-Hepatitis C Hepatocellular Carcinoma. Cells, 9(2), 262. https://doi.org/10.3390/cells9020262





