Conditioned Medium from Cells Overexpressing TDP-43 Alters the Metabolome of Recipient Cells
Abstract
1. Introduction
2. Materials and Methods
2.1. Plasmids
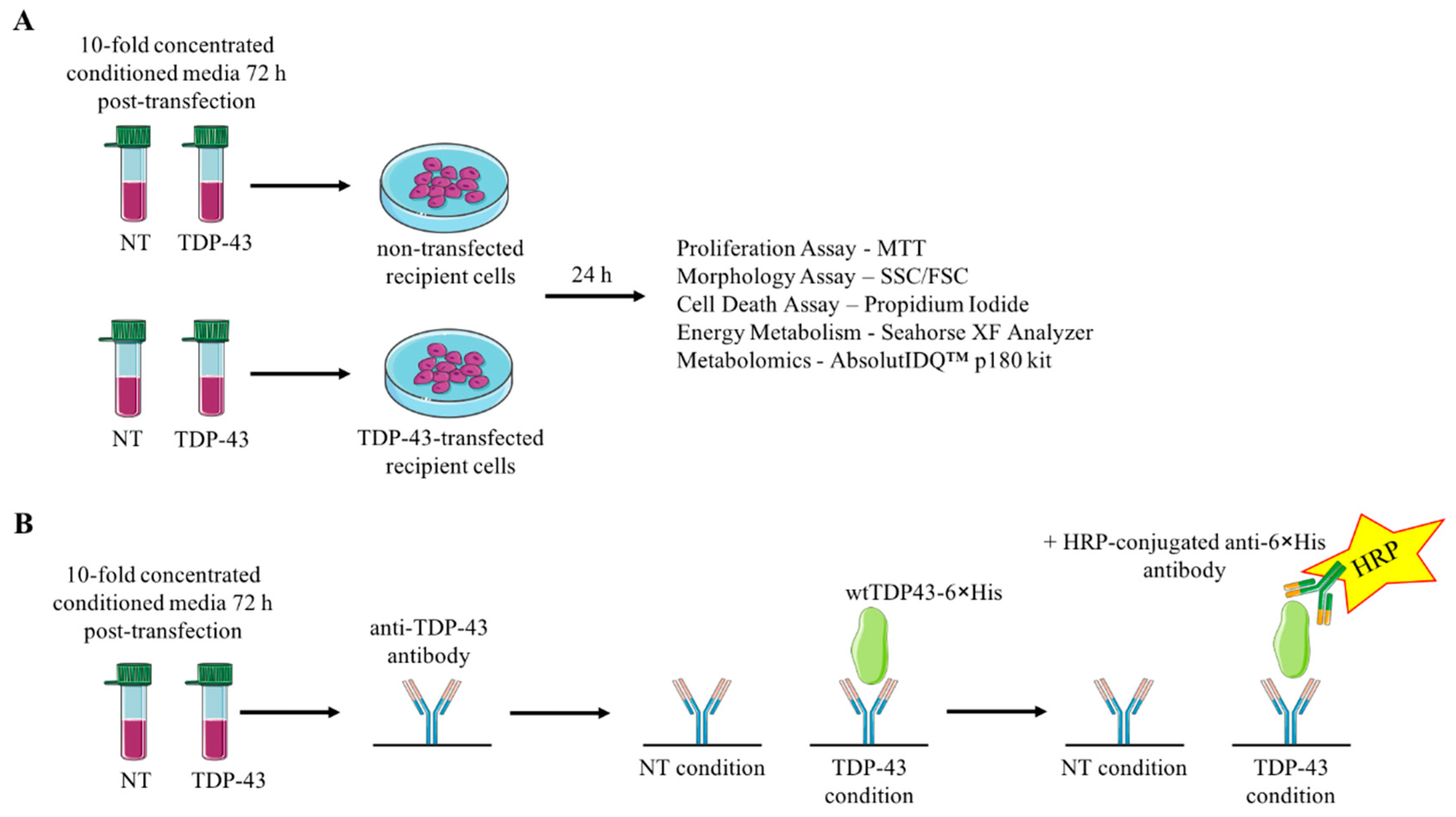
2.2. Cell Culture and Generation of Conditioned Medium
2.3. Enzyme-Linked Immunosorbent Assay (ELISA) on Conditioned Media
2.4. Recipient Cell Culture
2.5. Cell Death, Morphology, and Proliferation
2.6. Immuno-Detection of TDP-43 in Recipient Cells
2.7. Glycolysis and Cellular Respiration of Recipient Cells
2.8. Metabolomics Analysis of Recipient Cells
2.9. Statistical Analyses
3. Results
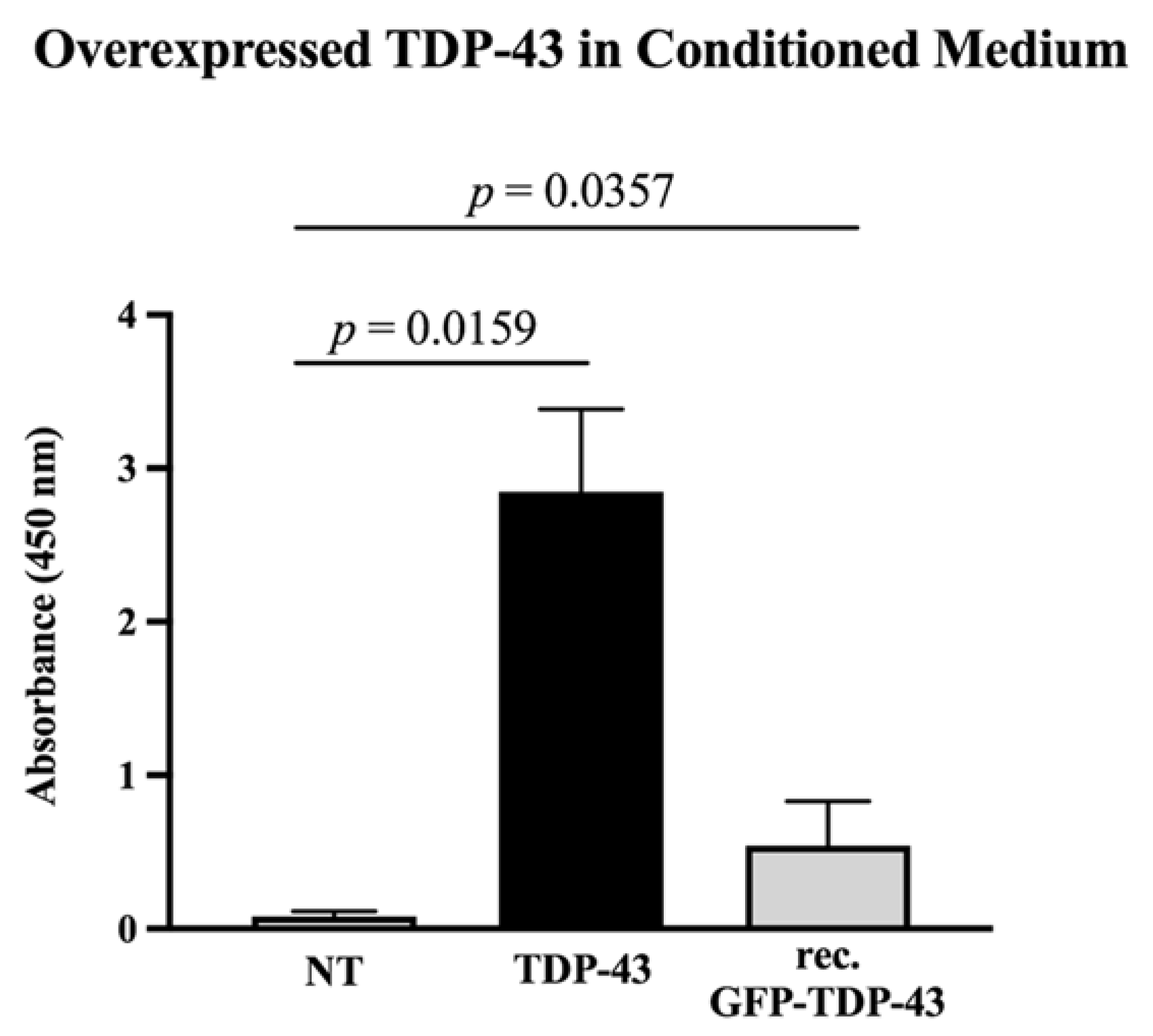
3.1. Overexpressed TDP-43 Is Released into the Culture Medium
3.2. No Propagation of Overexpressed TDP-43
3.3. Increased Proliferation, Increased Propidium Iodide Uptake, and Decreased Membrane Integrity
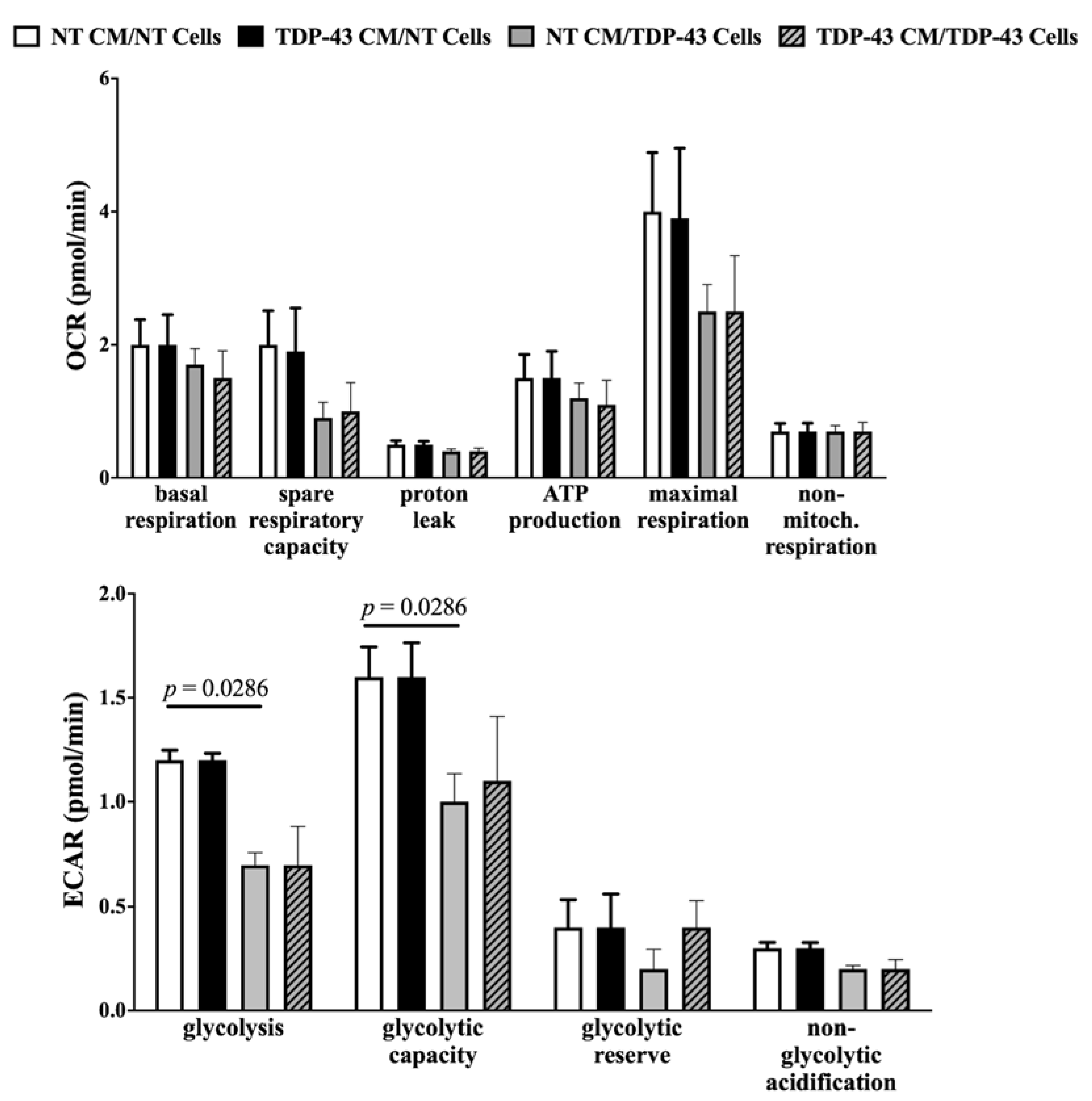
3.4. No Change in Oxidative Phosphorylation or Glycolysis
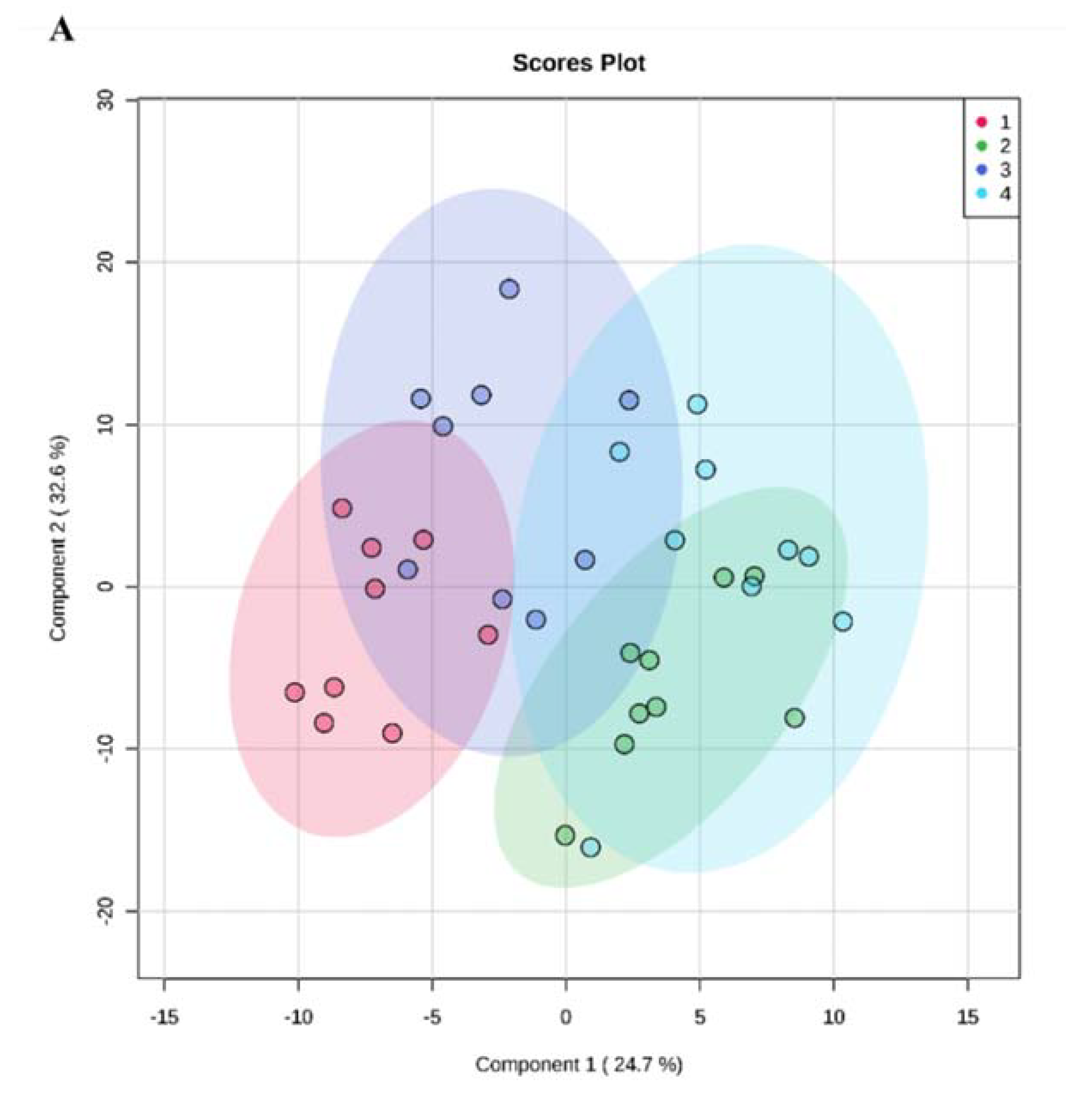
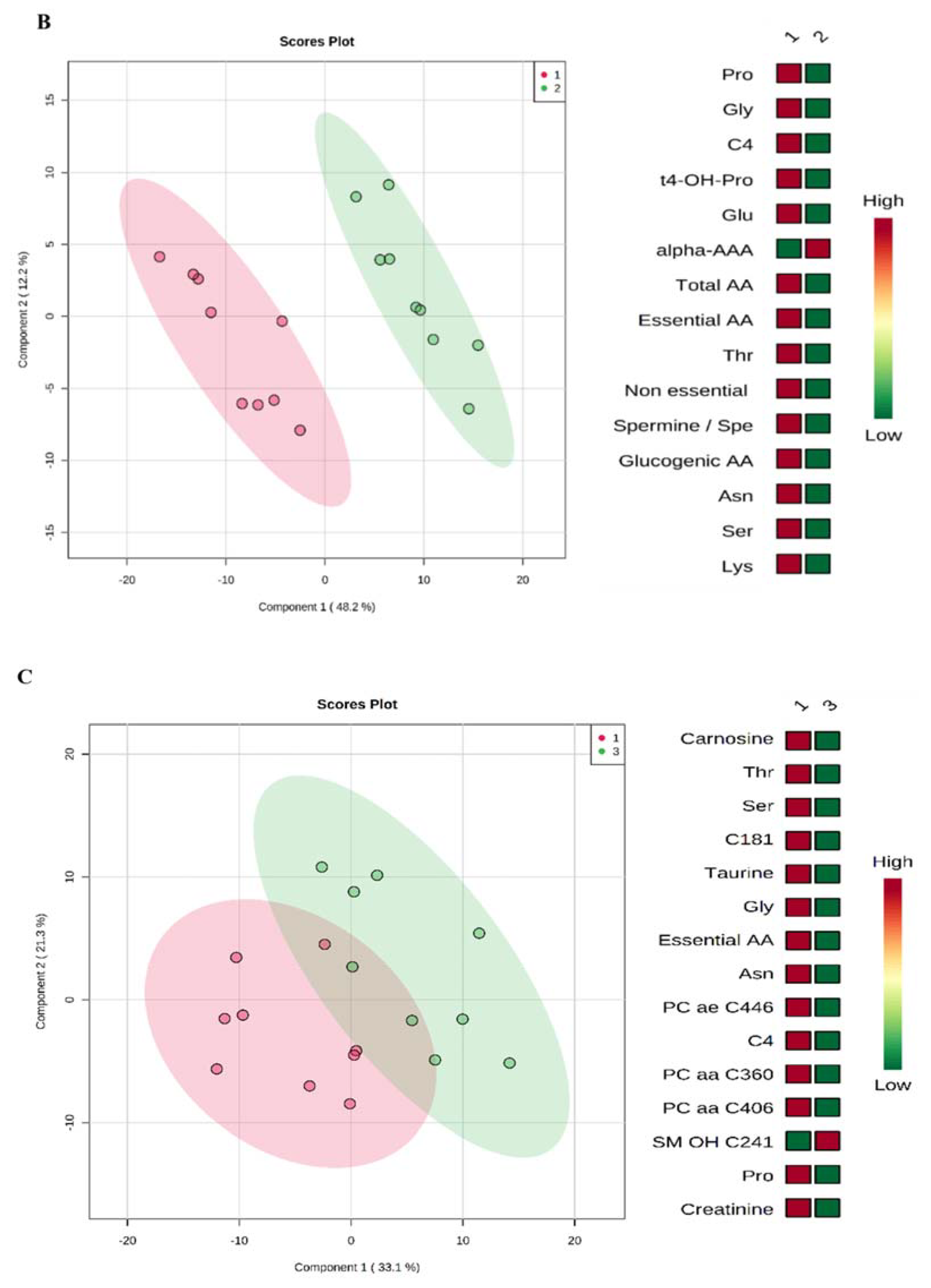
3.5. TDP-43-Conditioned Medium Caused ALS-Related Perturbations in the Cellular Metabolome
4. Discussion
4.1. Debatable Findings of Previous Studies on TDP-43 in Conditioned Medium
4.2. A Fraction of Overexpressed TDP-43 Is Released into the Medium
4.3. TDP-43 Propagation Did Not Occur by Conditioned Medium
4.4. TDP-43-Conditioned Medium Decreased Structural Integrity, Increased Proliferation Rate and Uptake of Propidium Iodide
4.5. TDP-43-Conditioned Medium and TDP-43 Overexpression Induced Different Metabolome Profiles
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Gil-Bea, F.J.; Aldanondo, G.; Fernandez, H.L.; Vallejo-Illarramendi, A.; De Munain, A.L. Insights into the mechanisms of copper dyshomeostasis in amyotrophic lateral sclerosis. Expert Rev. Mol. Med. 2017, 19, 17085. [Google Scholar] [CrossRef]
- Arai, T.; Hasegawa, M.; Akiyama, H.; Ikeda, K.; Nonaka, T.; Mori, H.; Mann, D.; Tsuchiya, K.; Yoshida, M.; Hashizume, Y.; et al. TDP-43 is a component of ubiquitin-positive tau-negative inclusions in frontotemporal lobar degeneration and amyotrophic lateral sclerosis. Biochem. Biophys. Res. Commun. 2006, 351, 602–611. [Google Scholar] [CrossRef] [PubMed]
- Neumann, M.; Sampathu, D.M.; Kwong, L.K.; Truax, A.C.; Micsenyi, M.C.; Chou, T.T.; Bruce, J.; Schuck, T.; Grossman, M.; Clark, C.M.; et al. Ubiquitinated TDP-43 in Frontotemporal Lobar Degeneration and Amyotrophic Lateral Sclerosis. Science 2006, 314, 130–133. [Google Scholar] [CrossRef] [PubMed]
- Hergesheimer, R.C.; Chami, A.A.; De Assis, D.R.; Vourc’H, P.; Andres, C.R.; Corcia, P.; Lanznaster, D.; Blasco, H. The debated toxic role of aggregated TDP-43 in amyotrophic lateral sclerosis: A resolution in sight? Brain 2019, 142, 1176–1194. [Google Scholar] [CrossRef] [PubMed]
- King, O.D.; Gitler, A.D.; Shorter, J. The tip of the iceberg: RNA-binding proteins with prion-like domains in neurodegenerative disease. Brain Res. 2012, 1462, 61–80. [Google Scholar] [CrossRef]
- Lazarev, V.F.; Benken, K.A.; Semenyuk, P.I.; Sarantseva, S.V.; Bolshakova, O.I.; Mikhaylova, E.R.; Muronetz, V.I.; Guzhova, I.V.; Margulis, B.A. GAPDH binders as potential drugs for the therapy of polyglutamine diseases: Design of a new screening assay. FEBS Lett. 2015, 589, 581–587. [Google Scholar] [CrossRef]
- Yerbury, J.J.; McAlary, L. Strategies to promote the maturation of ALS-associated SOD1 mutants: Small molecules return to the fold. Neural Regen. Res. 2019, 14, 1511–1512. [Google Scholar] [CrossRef]
- Surgucheva, I.; Sharov, V.S.; Surguchov, A. gamma-Synuclein: Seeding of alpha-synuclein aggregation and transmission between cells. Biochemistry 2012, 51, 4743–4754. [Google Scholar] [CrossRef]
- Laferriere, F.; Maniecka, Z.; Pérez-Berlanga, M.; Hruska-Plochan, M.; Gilhespy, L.; Hock, E.-M.; Wagner, U.; Afroz, T.; Boersema, P.J.; Barmettler, G.; et al. TDP-43 extracted from frontotemporal lobar degeneration subject brains displays distinct aggregate assemblies and neurotoxic effects reflecting disease progression rates. Nat. Neurosci. 2018, 22, 65–77. [Google Scholar] [CrossRef]
- Nonaka, T.; Masuda-Suzukake, M.; Arai, T.; Hasegawa, Y.; Akatsu, H.; Obi, T.; Yoshida, M.; Murayama, S.; Mann, D.M.; Akiyama, H.; et al. Prion-like Properties of Pathological TDP-43 Aggregates from Diseased Brains. Cell Rep. 2013, 4, 124–134. [Google Scholar] [CrossRef]
- Smethurst, P.; Newcombe, J.; Troakes, C.; Simone, R.; Chen, Y.-R.; Patani, R.; Sidle, K. In vitro prion-like behaviour of TDP-43 in ALS. Neurobiol. Dis. 2016, 96, 236–247. [Google Scholar] [CrossRef] [PubMed]
- Zeineddine, R.; Whiten, D.R.; Farrawell, N.E.; McAlary, L.; Hanspal, M.A.; Kumita, J.R.; Wilson, M.R.; Yerbury, J.J. Flow cytometric measurement of the cellular propagation of TDP-43 aggregation. Prion 2017, 11, 195–204. [Google Scholar] [CrossRef]
- Feiler, M.S.; Strobel, B.; Freischmidt, A.; Helferich, A.M.; Kappel, J.; Brewer, B.M.; Li, D.; Thal, D.R.; Walther, P.; Ludolph, A.C.; et al. TDP-43 is intercellularly transmitted across axon terminals. J. Cell Boil. 2015, 211, 897–911. [Google Scholar] [CrossRef]
- Blasco, H.; Lanznaster, D.; Veyrat-Durebex, C.; Hergesheimer, R.; Vourch, P.; Maillot, F.; Andres, C.R.; Pradat, P.-F.; Corcia, P. Understanding and managing metabolic dysfunction in Amyotrophic Lateral Sclerosis. Expert Rev. Neurother. 2020, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Manzo, E.; O’Conner, A.G.; Barrows, J.M.; Shreiner, D.D.; Birchak, G.J.; Zarnescu, D.C. Medium-Chain Fatty Acids, Beta-Hydroxybutyric Acid and Genetic Modulation of the Carnitine Shuttle Are Protective in a Drosophila Model of ALS Based on TDP-43. Front. Mol. Neurosci. 2018, 11, 182. [Google Scholar] [CrossRef] [PubMed]
- Lanznaster, D.; Bourgeais, J.; Bruno, C.; Hergesheimer, R.C.; Thepault, R.-A.; Vourc’H, P.; Corcia, P.; Andres, C.R.; Herault, O.; Blasco, H.; et al. TDP-43-Mediated Toxicity in HEK293T Cells: A Fast and Reproducible Protocol To Be Employed in the Search of New Therapeutic Options against Amyotrophic Lateral Sclerosis. Cells 2019, 9, 68. [Google Scholar] [CrossRef]
- Da Cruz, S.; Cleveland, D.W. Understanding the role of TDP-43 and FUS/TLS in ALS and beyond. Curr. Opin. Neurobiol. 2011, 21, 904–919. [Google Scholar] [CrossRef]
- Pokrishevsky, E.; Grad, L.I.; Cashman, N.R. TDP-43 or FUS-induced misfolded human wild-type SOD1 can propagate intercellularly in a prion-like fashion. Sci. Rep. 2016, 6, 22155. [Google Scholar] [CrossRef]
- Patin, F.; Baranek, T.; Vourc’H, P.; Nadal-Desbarats, L.; Goossens, J.-F.; Marouillat, S.; Dessein, A.-F.; Descat, A.; Hounoum, B.M.; Bruno, C.; et al. Combined Metabolomics and Transcriptomics Approaches to Assess the IL-6 Blockade as a Therapeutic of ALS: Deleterious Alteration of Lipid Metabolism. Neurotherapeutics 2016, 13, 905–917. [Google Scholar] [CrossRef]
- Eskelinen, M.H.; Kivipelto, M. Caffeine as a protective factor in dementia and Alzheimer’s disease. J. Alzheimers Dis. 2010, 20, 167–174. [Google Scholar] [CrossRef]
- Hounoum, B.M.; Blasco, H.; Coque, E.; Vourc’H, P.; Emond, P.; Corcia, P.; Andres, C.R.; Raoul, C.; Mavel, S. The Metabolic Disturbances of Motoneurons Exposed to Glutamate. Mol. Neurobiol. 2018, 55, 7669–7676. [Google Scholar] [CrossRef] [PubMed]
- Xia, J.; Wishart, D.S. Using MetaboAnalyst 3.0 for Comprehensive Metabolomics Data Analysis. Curr. Protoc. Bioinform. 2016, 55, 14.10.1–14.10.91. [Google Scholar] [CrossRef]
- Iguchi, Y.; Eid, L.; Parent, M.; Soucy, G.; Bareil, C.; Riku, Y.; Kawai, K.; Takagi, S.; Yoshida, M.; Katsuno, M.; et al. Exosome secretion is a key pathway for clearance of pathological TDP-43. Brain 2016, 139, 3187–3201. [Google Scholar] [CrossRef] [PubMed]
- Henriques, A.; De Aguilar, J.-L.G. Can Transcriptomics Cut the Gordian Knot of Amyotrophic Lateral Sclerosis? Curr. Genom. 2011, 12, 506–515. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Valbuena, G.N.; Rizzardini, M.; Cimini, S.; Siskos, A.P.; Bendotti, C.; Cantoni, L.; Keun, H.C. Metabolomic Analysis Reveals Increased Aerobic Glycolysis and Amino Acid Deficit in a Cellular Model of Amyotrophic Lateral Sclerosis. Mol. Neurobiol. 2015, 53, 2222–2240. [Google Scholar] [CrossRef] [PubMed]
- Kawahara, M.; Tanaka, K.-I.; Kato-Negishi, M. Zinc, Carnosine, and Neurodegenerative Diseases. Nutrition 2018, 10, 147. [Google Scholar] [CrossRef]
- Menzie, J.; Pan, C.; Prentice, H.; Wu, J.-Y. Taurine and central nervous system disorders. Amino Acids 2012, 46, 31–46. [Google Scholar] [CrossRef]
- Lanznaster, D.; Bejan-Angoulvant, T.; Patin, F.; Andres, C.R.; Vourc’H, P.; Corcia, P.; Blasco, H. Plasma creatinine and amyotrophic lateral sclerosis prognosis: A systematic review and meta-analysis. Amyotroph. Lat. Scler. Front. Degener. 2019, 20, 199–206. [Google Scholar] [CrossRef]
- Veyrat-Durebex, C.; Bris, C.; Codron, P.; Bocca, C.; Chupin, S.; Corcia, P.; Vourc’H, P.; Hergesheimer, R.; Cassereau, J.; Funalot, B.; et al. Metabo-lipidomics of Fibroblasts and Mitochondrial-Endoplasmic Reticulum Extracts from ALS Patients Shows Alterations in Purine, Pyrimidine, Energetic, and Phospholipid Metabolisms. Mol. Neurobiol. 2019, 56, 5780–5791. [Google Scholar] [CrossRef]
- Fernández-Eulate, G.; Ruiz-Sanz, J.I.; Riancho, J.; Zufiría, M.; Gereñu, G.; Fernández-Torrón, R.; Poza-Aldea, J.J.; Ondaro, J.; Espinal, J.B.; González-Chinchón, G.; et al. A comprehensive serum lipidome profiling of amyotrophic lateral sclerosis. Amyotroph. Lat. Scler. Front. Degener. 2020, 21, 252–262. [Google Scholar] [CrossRef]
- Guidetti, P.; Schwarcz, R. Determination of α-aminoadipic acid in brain, peripheral tissues, and body fluids using GC/MS with negative chemical ionization. Mol. Brain Res. 2003, 118, 132–139. [Google Scholar] [CrossRef] [PubMed]
- Wu, H.-Q.; Ungerstedt, U.; Schwarcz, R. l-α-Aminoadipic acid as a regulator of kynurenic acid production in the hippocampus: A microdialysis study in freely moving rats. Eur. J. Pharmacol. 1995, 281, 55–61. [Google Scholar] [CrossRef] [PubMed]

© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hergesheimer, R.; Lanznaster, D.; Bourgeais, J.; Hérault, O.; Vourc’h, P.; Andres, C.R.; Corcia, P.; Blasco, H. Conditioned Medium from Cells Overexpressing TDP-43 Alters the Metabolome of Recipient Cells. Cells 2020, 9, 2198. https://doi.org/10.3390/cells9102198
Hergesheimer R, Lanznaster D, Bourgeais J, Hérault O, Vourc’h P, Andres CR, Corcia P, Blasco H. Conditioned Medium from Cells Overexpressing TDP-43 Alters the Metabolome of Recipient Cells. Cells. 2020; 9(10):2198. https://doi.org/10.3390/cells9102198
Chicago/Turabian StyleHergesheimer, Rudolf, Débora Lanznaster, Jérôme Bourgeais, Olivier Hérault, Patrick Vourc’h, Christian R. Andres, Philippe Corcia, and Hélène Blasco. 2020. "Conditioned Medium from Cells Overexpressing TDP-43 Alters the Metabolome of Recipient Cells" Cells 9, no. 10: 2198. https://doi.org/10.3390/cells9102198
APA StyleHergesheimer, R., Lanznaster, D., Bourgeais, J., Hérault, O., Vourc’h, P., Andres, C. R., Corcia, P., & Blasco, H. (2020). Conditioned Medium from Cells Overexpressing TDP-43 Alters the Metabolome of Recipient Cells. Cells, 9(10), 2198. https://doi.org/10.3390/cells9102198







