Synergistic Effects of N-Acetylcysteine and Mesenchymal Stem Cell in a Lipopolysaccharide-Induced Interstitial Cystitis Rat Model
Abstract
1. Introduction
2. Materials and Methods
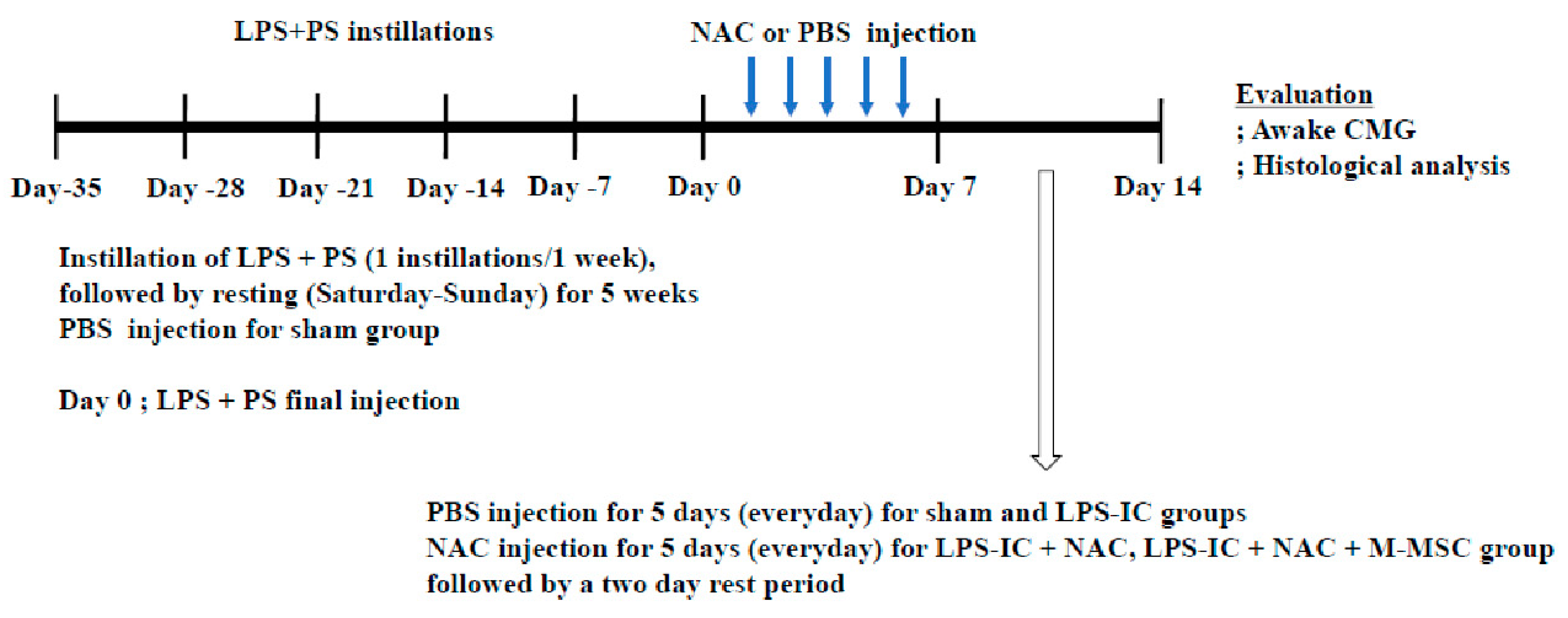
2.1. Animal Modelling
2.2. Stem Cell Preparation
2.3. Administration of NAC and/or M-MSCs
2.4. Unanesthetized and Unrestrained Cystometry
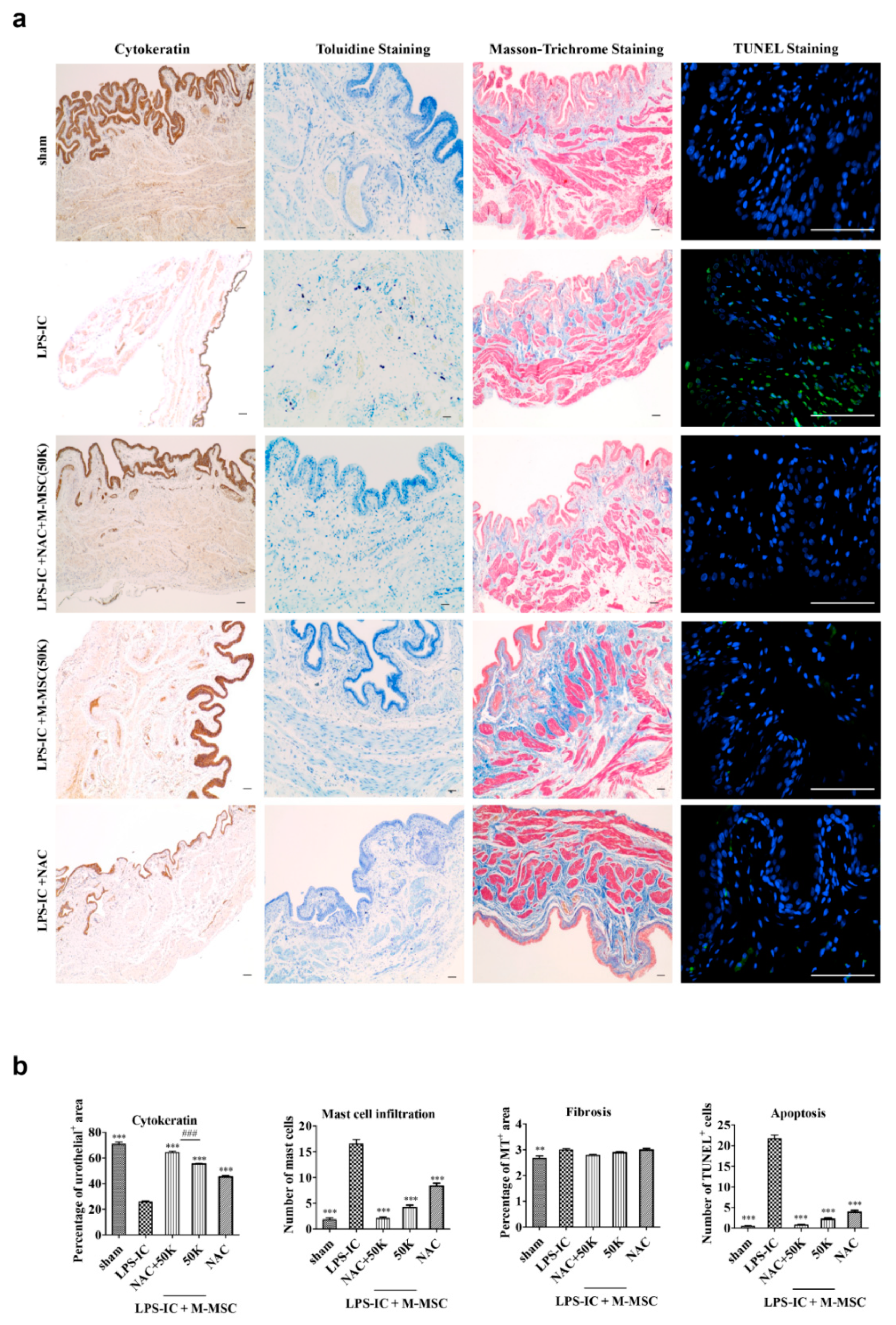
2.5. Histological Examinations
2.6. Statistical Analysis
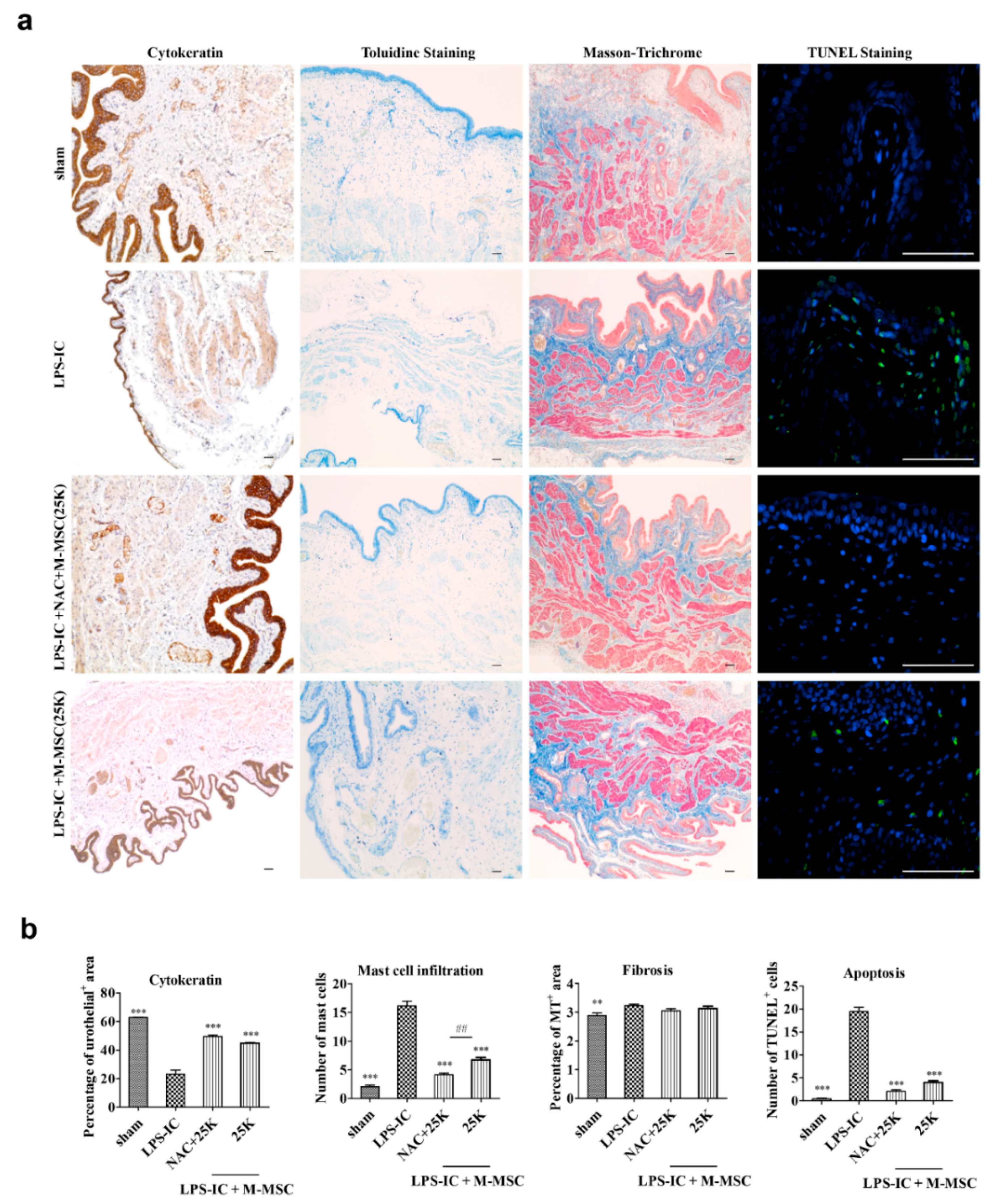
3. Results
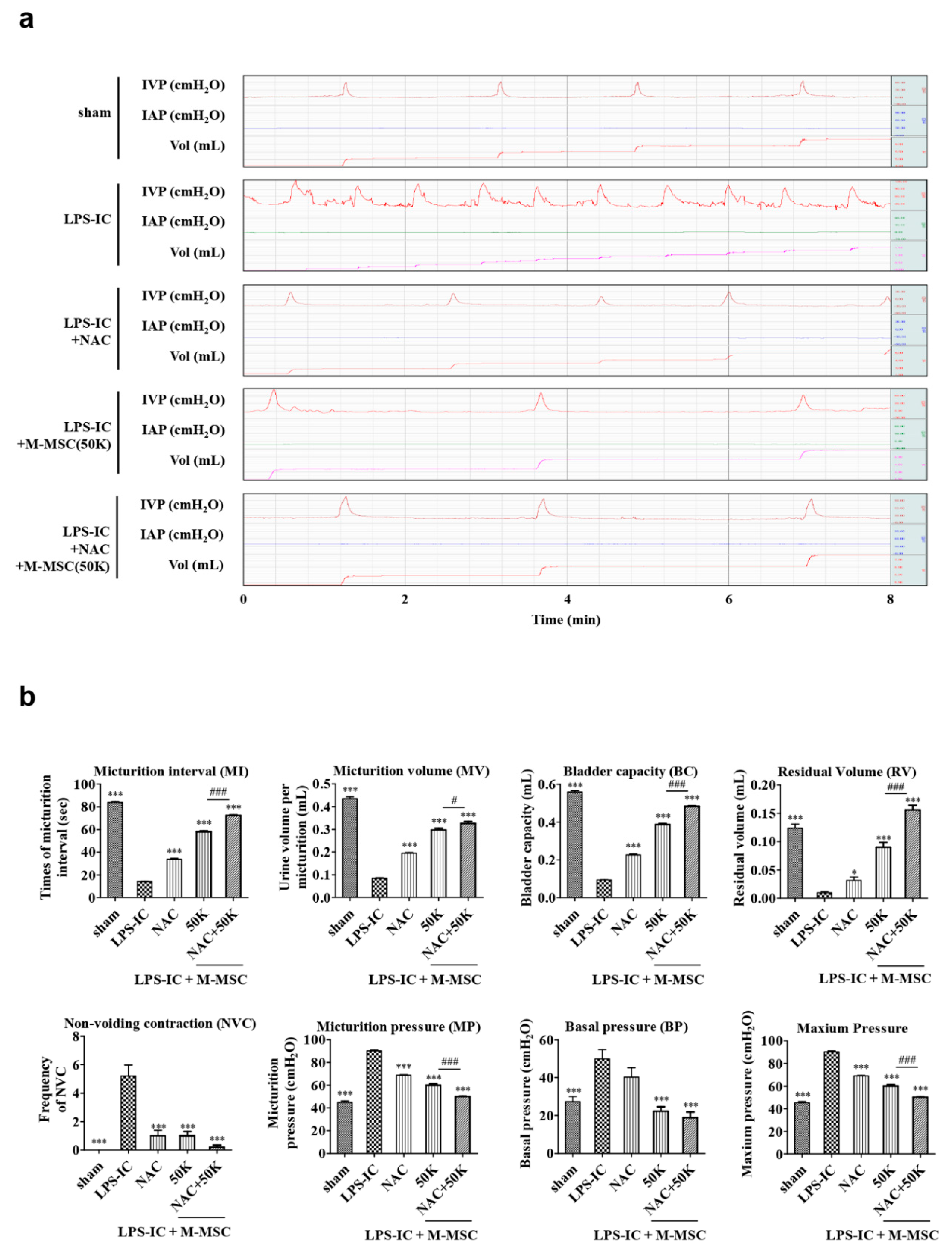
3.1. Therapeutic Efficacy of NAC or M-MSC Monotherapy
3.2. Improved Therapeutic Efficacy of NAC and M-MSC Combination Therapy
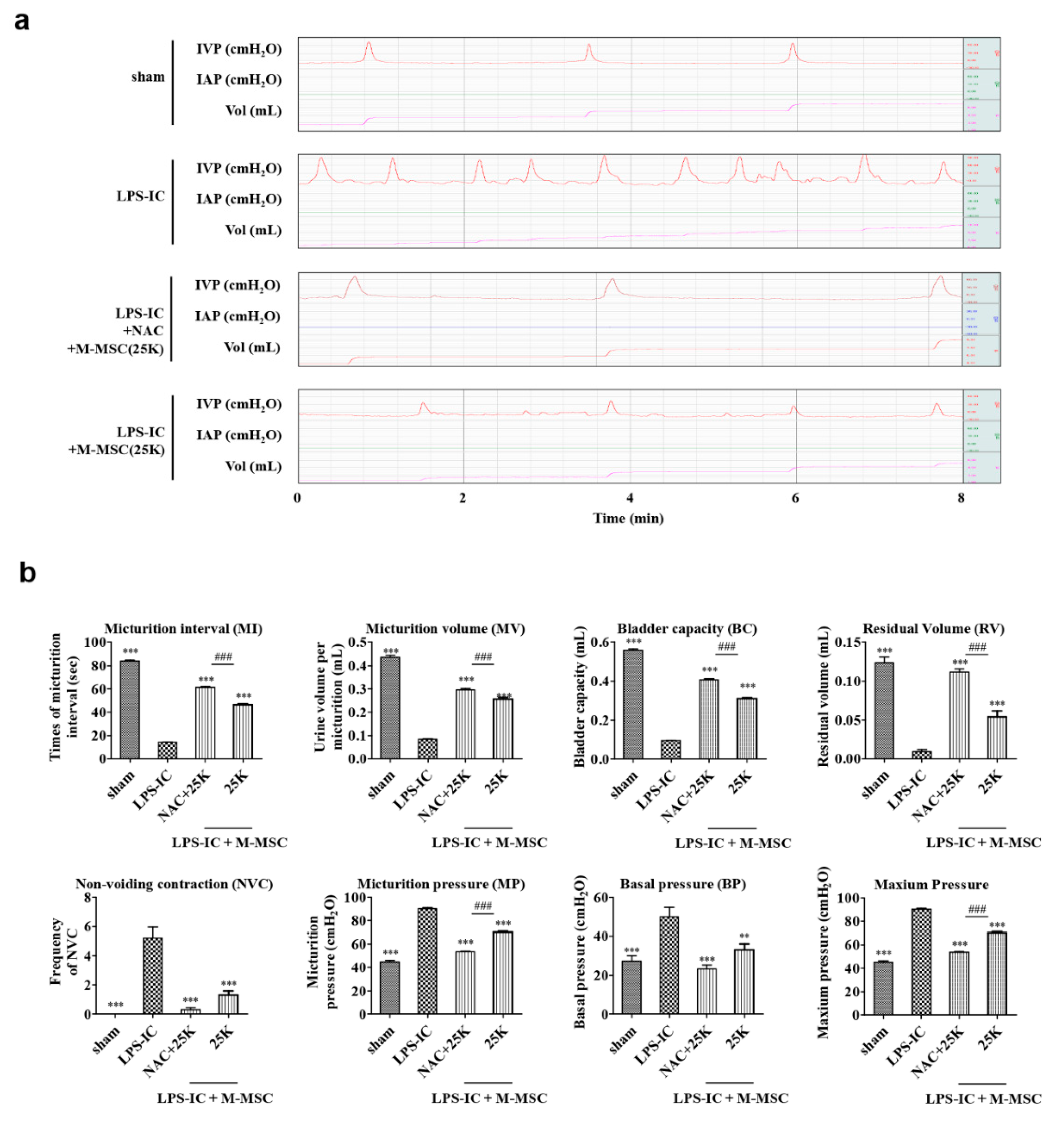
3.3. Optimization of the M-MSC and NAC Combination Therapy
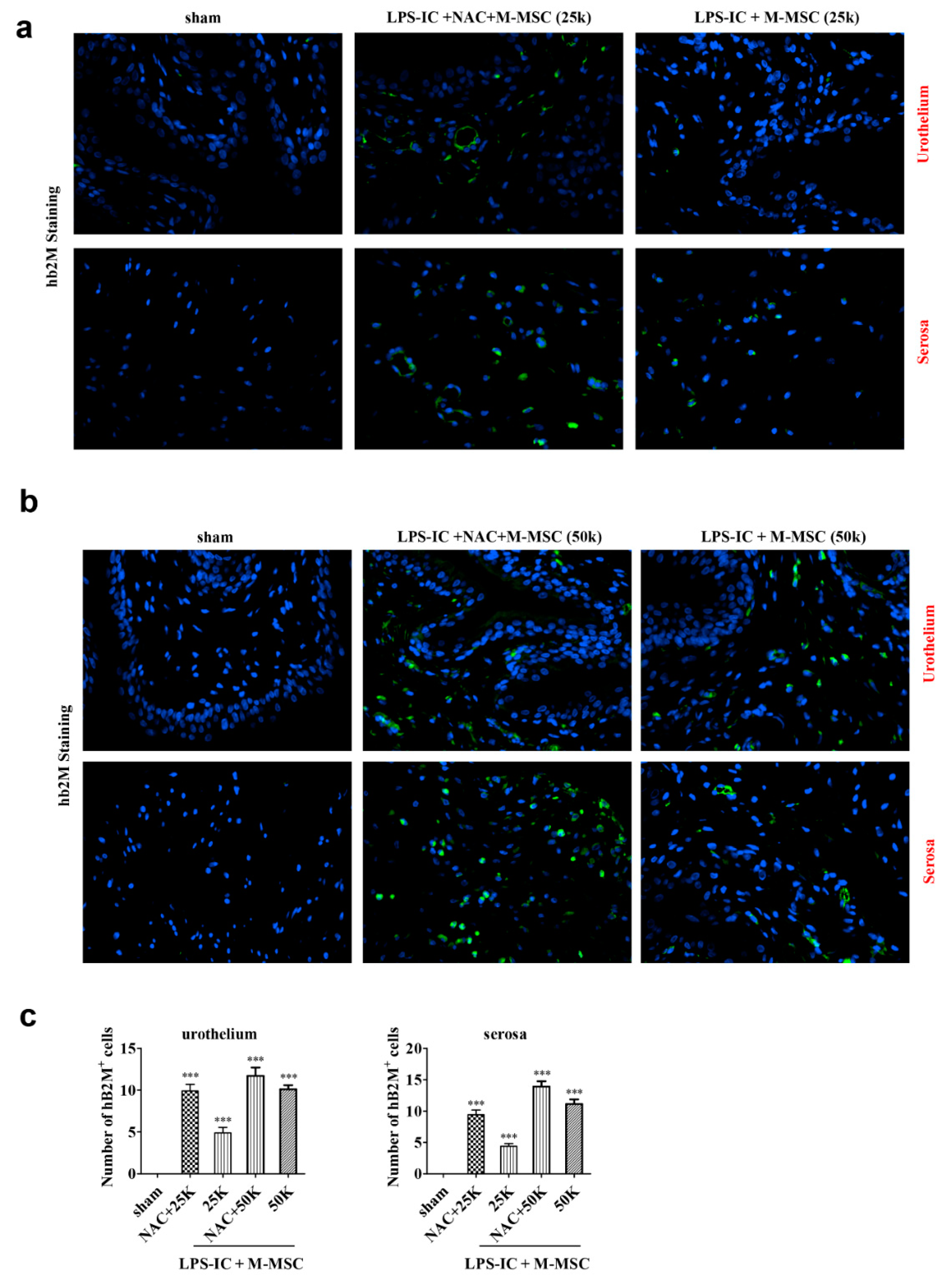
3.4. Distribution of Transplanted M-MSCs
4. Discussion
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Kim, A.; Shin, N.-M.; Choo, M.-S. Stem Cell Therapy for Interstitial Cystitis/Bladder Pain Syndrome. Curr. Urol. Rep. 2015, 17. [Google Scholar] [CrossRef]
- Kim, Y.; Jin, H.J.; Heo, J.; Ju, H.; Lee, H.-Y.; Kim, S.; Lee, S.; Lim, J.; Jeong, S.Y.; Kwon, J.; et al. Small hypoxia-primed mesenchymal stem cells attenuate graft-versus-host disease. Leuk. 2018, 32, 2672–2684. [Google Scholar] [CrossRef] [PubMed]
- Shin, J.H.; Ryu, C.-M.; Yu, H.Y.; Shin, D.-M.; Choo, M.-S. Current and Future Directions of Stem Cell Therapy for Bladder Dysfunction. Stem Cell Rev. Rep. 2019, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Song, M.; Park, J.; Choo, M.-S. MP19-06 MESENCHYMAL STEM-CELL THERAPY ALLEVIATES INTERSTITIAL CYSTITIS BY ACTIVATING WNT SIGNALING PATHWAY. J. Urol. 2015, 193, e217. [Google Scholar] [CrossRef]
- Kim, A.; Yu, H.Y.; Lim, J.; Ryu, C.-M.; Kim, Y.H.; Heo, J.; Han, J.-Y.; Lee, S.; Bae, Y.S.; Kim, J.Y.; et al. Improved efficacy and in vivo cellular properties of human embryonic stem cell derivative in a preclinical model of bladder pain syndrome. Sci. Rep. 2017, 7, 8872. [Google Scholar] [CrossRef] [PubMed]
- Ryu, C.-M.; Yu, H.Y.; Lee, H.-Y.; Shin, J.-H.; Lee, S.; Ju, H.; Paulson, B.; Lee, S.; Kim, S.; Lim, J.; et al. Longitudinal intravital imaging of transplanted mesenchymal stem cells elucidates their functional integration and therapeutic potency in an animal model of interstitial cystitis/bladder pain syndrome. Theranostics 2018, 8, 5610–5624. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.W.; Ryu, C.-M.; Shin, J.-H.; Choi, D.; Kim, A.; Yu, H.Y.; Han, J.-Y.; Lee, H.-Y.; Lim, J.; Kim, Y.H.; et al. The Therapeutic Effect of Human Embryonic Stem Cell-Derived Multipotent Mesenchymal Stem Cells on Chemical-Induced Cystitis in Rats. Int. Neurourol. J. 2018, 22, S34–S45. [Google Scholar] [CrossRef] [PubMed]
- Kim, A.; Yu, H.Y.; Heo, J.; Song, M.; Shin, J.-H.; Lim, J.; Yoon, S.-J.; Kim, Y.; Lee, S.; Kim, S.W.; et al. Mesenchymal stem cells protect against the tissue fibrosis of ketamine-induced cystitis in rat bladder. Sci. Rep. 2016, 6, 30881. [Google Scholar] [CrossRef]
- Jeong, E.M.; Yoon, J.-H.; Lim, J.; Shin, J.-W.; Cho, A.Y.; Heo, J.; Lee, K.B.; Lee, J.-H.; Lee, W.J.; Kim, H.-J.; et al. Real-Time Monitoring of Glutathione in Living Cells Reveals that High Glutathione Levels Are Required to Maintain Stem Cell Function. Stem Cell Rep. 2018, 10, 600–614. [Google Scholar] [CrossRef]
- Lee, S.; Lim, J.; Lee, J.H.; Ju, H.; Heo, J.; Kim, Y.; Kim, S.; Yeul Yu, H.; Ryu, C.M.; Lee, S.Y.; et al. Ascorbic Acid 2-Glucoside Stably Promotes the Primitiveness of Embryonic and Mesenchymal Stem Cells through Tet- and Creb1-Dependent Mechanisms. Antioxid. Redox Signal. 2019, 32. [Google Scholar] [CrossRef]
- Kim, J.M.; Hong, K.-S.; Song, W.K.; Bae, D.; Hwang, I.-K.; Kim, J.S.; Chung, H.-M. Perivascular Progenitor Cells Derived From Human Embryonic Stem Cells Exhibit Functional Characteristics of Pericytes and Improve the Retinal Vasculature in a Rodent Model of Diabetic Retinopathy. STEM CELLS Transl. Med. 2016, 5, 1268–1276. [Google Scholar] [CrossRef] [PubMed]
- Hong, K.-S.; Bae, D.; Choi, Y.; Kang, S.-W.; Moon, S.-H.; Lee, H.T.; Chung, H.-M. A Porous Membrane-Mediated Isolation of Mesenchymal Stem Cells from Human Embryonic Stem Cells. Tissue Eng. Part C: Methods 2014, 21, 322–329. [Google Scholar] [CrossRef] [PubMed]
- Desgres, M.; Menasché, P. Clinical Translation of Pluripotent Stem Cell Therapies: Challenges and Considerations. Cell Stem Cell 2019, 25, 594–606. [Google Scholar] [CrossRef] [PubMed]
- Heo, J.; Lim, J.; Lee, S.; Jeong, J.; Kang, H.; Kim, Y.; Kang, J.W.; Yu, H.Y.; Jeong, E.M.; Kim, K.; et al. Sirt1 Regulates DNA Methylation and Differentiation Potential of Embryonic Stem Cells by Antagonizing Dnmt3l. Cell Rep. 2017, 18, 1930–1945. [Google Scholar] [CrossRef]
- Ryu, C.-M.; Shin, J.H.; Yu, H.Y.; Ju, H.; Kim, S.; Lim, J.; Heo, J.; Lee, S.; Shin, D.-M.; Choo, M.-S. N-acetylcysteine prevents bladder tissue fibrosis in a lipopolysaccharide-induced cystitis rat model. Sci. Rep. 2019, 9, 8134. [Google Scholar] [CrossRef]
- Heo, J.; Noh, B.; Lee, S.; Lee, H.; Kim, Y.; Lim, J.; Ju, H.; Yu, H.Y.; Ryu, C.; Lee, P.C.; et al. Phosphorylation of TFCP2L1 by CDK1 is required for stem cell pluripotency and bladder carcinogenesis. EMBO Mol. Med. 2019, 10880. [Google Scholar] [CrossRef]
- Stein, P.C.; Pham, H.; Ito, T.; Parsons, C. Bladder Injury Model Induced in Rats by Exposure to Protamine Sulfate Followed by Bacterial Endotoxin. J. Urol. 1996, 155, 1133–1138. [Google Scholar] [CrossRef]
- Chen, Y.-T.; Chiang, H.-J.; Chen, C.-H.; Sung, P.-H.; Lee, F.-Y.; Tsai, T.-H.; Chang, C.-L.; Chen, H.-H.; Sun, C.-K.; Leu, S.; et al. Melatonin treatment further improves adipose-derived mesenchymal stem cell therapy for acute interstitial cystitis in rat. J. Pineal Res. 2014, 57, 248–261. [Google Scholar] [CrossRef]
- Zhou, H.; Ma, Q.; Zhu, P.; Ren, J.; Reiter, R.J.; Chen, Y. Protective role of melatonin in cardiac ischemia-reperfusion injury: From pathogenesis to targeted therapy. J. Pineal Res. 2018, 64, e12471. [Google Scholar] [CrossRef]
- Banaei, S.; Ahmadiasl, N.; Alihemmati, A. Comparison of the Protective Effects of Erythropoietin and Melatonin on Renal Ischemia-Reperfusion Injury. Trauma Mon. 2016, 21, 23005. [Google Scholar] [CrossRef]
- Takahashi, N.; Yoshida, T.; Ohnuma, A.; Horiuchi, H.; Ishitsuka, K.; Kashimoto, Y.; Kuwahara, M.; Nakashima, N.; Harada, T. The Enhancing Effect of the Antioxidant N-Acetylcysteine on Urinary Bladder Injury Induced by Dimethylarsinic Acid. Toxicol. Pathol. 2011, 39, 1107–1114. [Google Scholar] [CrossRef] [PubMed]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shin, J.H.; Ryu, C.-M.; Ju, H.; Yu, H.Y.; Song, S.; Shin, D.-M.; Choo, M.-S. Synergistic Effects of N-Acetylcysteine and Mesenchymal Stem Cell in a Lipopolysaccharide-Induced Interstitial Cystitis Rat Model. Cells 2020, 9, 86. https://doi.org/10.3390/cells9010086
Shin JH, Ryu C-M, Ju H, Yu HY, Song S, Shin D-M, Choo M-S. Synergistic Effects of N-Acetylcysteine and Mesenchymal Stem Cell in a Lipopolysaccharide-Induced Interstitial Cystitis Rat Model. Cells. 2020; 9(1):86. https://doi.org/10.3390/cells9010086
Chicago/Turabian StyleShin, Jung Hyun, Chae-Min Ryu, Hyein Ju, Hwan Yeul Yu, Sujin Song, Dong-Myung Shin, and Myung-Soo Choo. 2020. "Synergistic Effects of N-Acetylcysteine and Mesenchymal Stem Cell in a Lipopolysaccharide-Induced Interstitial Cystitis Rat Model" Cells 9, no. 1: 86. https://doi.org/10.3390/cells9010086
APA StyleShin, J. H., Ryu, C.-M., Ju, H., Yu, H. Y., Song, S., Shin, D.-M., & Choo, M.-S. (2020). Synergistic Effects of N-Acetylcysteine and Mesenchymal Stem Cell in a Lipopolysaccharide-Induced Interstitial Cystitis Rat Model. Cells, 9(1), 86. https://doi.org/10.3390/cells9010086




