Role of microRNAs as Clinical Cancer Biomarkers for Ovarian Cancer: A Short Overview
Abstract
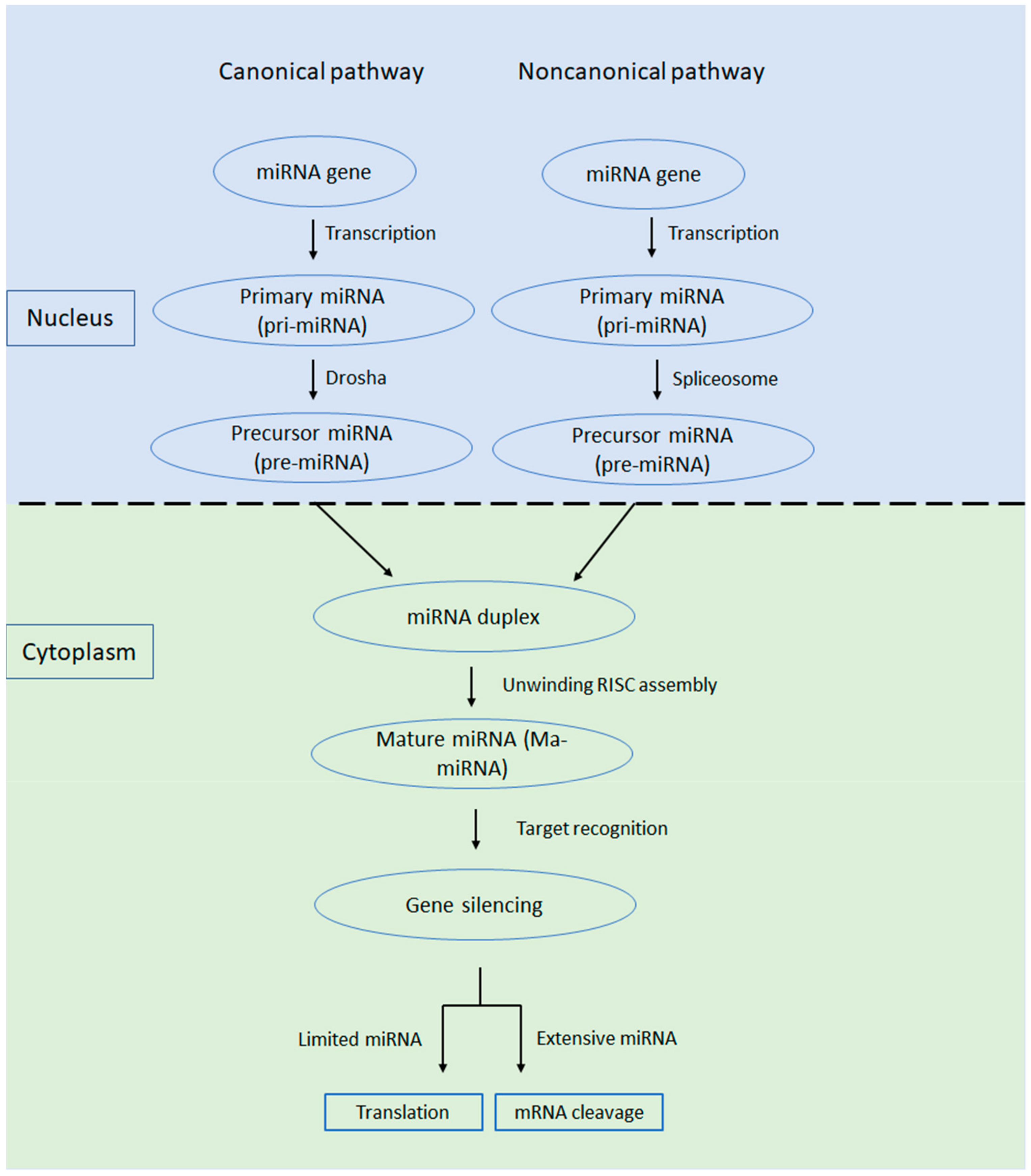
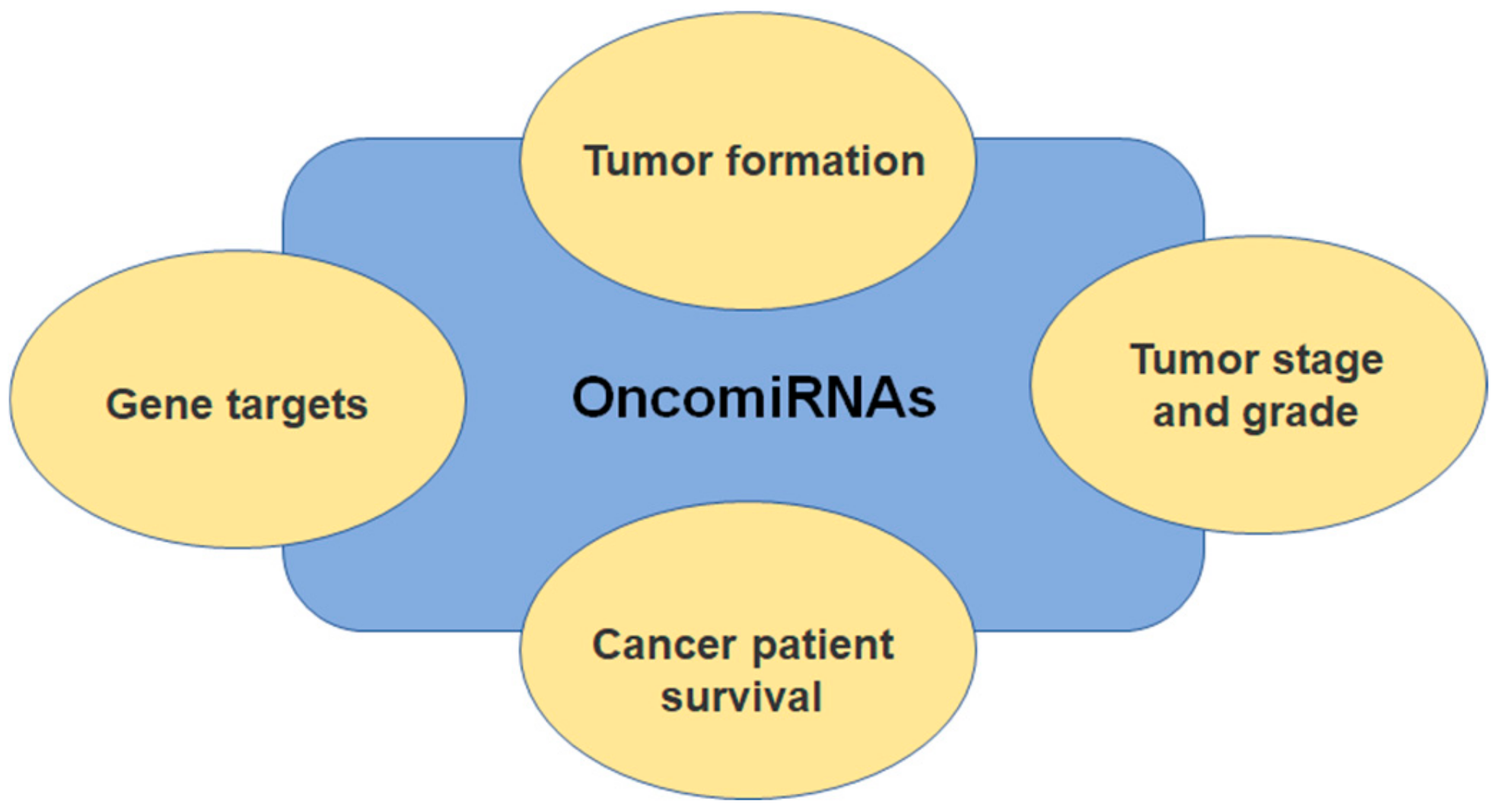
1. Introduction
2. Types of miRNAs in Ovarian Cancer (Circulating Cell-Free and Exosomal miRNAs)
3. Ovarian Cancer and miRNA Expression Profiles
4. miRNAs in Ovarian Cancer Invasion and Metastasis
5. miRNAs and Chemotherapy Sensitivity
6. miRNAs and Their Therapeutic Potential
- The inhibition of tumor proliferation, by the downregulation of genes and pathways involved in this process. In detail, miR-218 is targeting Runt-related transcription factor 2 (RUNX2) [102], miR-199a overexpression produces a downregulation of CD44 [103], miR-532-5p functions as a tumor suppressor by downregulation of casein kinase II subunit alpha 2 (CSNK2A2), SH3 and PX domain-containing protein 2A (SH3PXD2A), and Chromodomain-helicase-DNA-binding protein 4 (CHD4) [104], miR-222-3p and miR-221 reduce tumor cell proliferation by inhibiting AKT phosphorylation [105] and miR-135a-3p mediates the expression of Baculoviral IAP repeat-containing protein 3 (BIRC3), GAGRA3, and Sperm protein associated with the nucleus, X chromosome (SPANX) B1/SPANX B2 [106];
- Apoptosis of cancer cells, which occurs by miR-29b inducing PTEN hypomethylation (tumor suppressor), and, in turn, increases the expression of PTEN in cancer cells by directly binding to the 3′-UTR of PTEN [107]. miR-491-5p induces tumor cell apoptosis by inhibiting BCL-XL and EGFR signaling [108], and miR-744-5p induces tumor cell apoptosis in ovarian cancer by targeting Heterogeneous nuclear ribonucleoproteins C1/C2 (HNRNPC) and Nuclear factor 1 X-type (NFIX) [109];
- Reduction in the rate of migration and formation of tumor metastases: miR-200 regulates IL-8 and CXCL1 activity [110], miR-199a is targeting Hypoxia-inducible factor (HIF)-1α and HIF-2α [111], miR-92α mediates the suppression of peritoneal metastasis by inhibiting α5 integrin [112] and miR-143-3p while using TGF-beta activated kinase 1 (TAK1) signaling [113];
- It affects tumorigenesis and chemosensitivity by modulating oxidative stress through the miR-141 and miR-200a regulation at the level of p38α protein [114];
- The use of therapeutic approaches based on miRNAs in cancer therapy can have multiple benefits. Among these benefits, the most important is the ease of miRNA administration and delivery. In comparison with other molecules used in gene therapy (e.g., large viral vectors or plasmids), miRNAs can be manipulated easier and delivered due to their low molecular weight and small size [94].
7. Molecular Clustering Analysis in Ovarian Cancer
8. Conclusions and Future Perspectives
Author Contributions
Funding
Conflicts of Interest
References
- NCI Surveillance, Epidemiology, and End Results Program. Cancer Stat Facts: Ovarian Cancer. Available online: https://seer.cancer.gov/statfacts/html/ovary.html (accessed on 14 October 2019).
- Ehdaivand, S. WHO Classification of Ovarian Neoplasms. Available online: https://www.pathologyoutlines.com/topic/ovarytumorwhoclassif.html (accessed on 14 October 2019).
- Lisio, M.A.; Fu, L.; Goyeneche, A.; Gao, Z.H.; Telleria, C. High-Grade Serous Ovarian Cancer: Basic Sciences, Clinical and Therapeutic Standpoints. Int. J. Mol. Sci. 2019, 20, 952. [Google Scholar] [CrossRef]
- Elias, K.M.; Guo, J.; Bast, R.C., Jr. Early Detection of Ovarian Cancer. Hematol. Oncol. Clin. N. Am. 2018, 32, 903–914. [Google Scholar] [CrossRef] [PubMed]
- NCI Dictionary of Cancer Terms: Biomarker. Available online: https://www.cancer.gov/publications/dictionaries/cancer-terms/def/biomarker (accessed on 14 October 2019).
- WHO International Programme on Chemical Safety Biomarkers in Risk Assessment: Validity and Validations. Available online: http://www.inchem.org/documents/ehc/ehc/ehc222.htm (accessed on 14 October 2019).
- Lee, R.C.; Feinbaum, R.L.; Ambros, V. The C. elegans heterochronic gene lin-4 encodes small RNAs with antisense complementarity to lin-14. Cell 1993, 75, 843–854. [Google Scholar] [CrossRef]
- He, L.; Hannon, G.J. MicroRNAs: Small RNAs with a big role in gene regulation. Nat. Rev. Genet. 2004, 5, 522–531. [Google Scholar] [CrossRef] [PubMed]
- Cretoiu, S.M. Circulating microRNAs in cardiovascular diseases: Recent progress and challenges. J. Hypertens. Res. 2016, 1, 15–18. [Google Scholar]
- Gregory, R.I.; Chendrimada, T.P.; Cooch, N.; Shiekhattar, R. Human RISC couples microRNA biogenesis and posttranscriptional gene silencing. Cell 2005, 123, 631–640. [Google Scholar] [CrossRef]
- Roden, C.; Gaillard, J.; Kanoria, S.; Rennie, W.; Barish, S.; Cheng, J.; Pan, W.; Liu, J.; Cotsapas, C.; Ding, Y.; et al. Novel determinants of mammalian primary microRNA processing revealed by systematic evaluation of hairpin-containing transcripts and human genetic variation. Genome Res. 2017, 27, 374–384. [Google Scholar] [CrossRef]
- Tafrihi, M.; Hasheminasab, E. MiRNAs: Biology, Biogenesis, their Web-based Tools, and Databases. Microrna 2019, 8, 4–27. [Google Scholar] [CrossRef]
- Svoronos, A.A.; Engelman, D.M.; Slack, F.J. OncomiR or Tumor Suppressor? The Duplicity of MicroRNAs in Cancer. Cancer Res. 2016, 76, 3666–3670. [Google Scholar] [CrossRef]
- Frixa, T.; Donzelli, S.; Blandino, G. Oncogenic MicroRNAs: Key Players in Malignant Transformation. Cancers 2015, 7, 2466–2485. [Google Scholar] [CrossRef]
- Di Leva, G.; Croce, C.M. The Role of microRNAs in the Tumorigenesis of Ovarian Cancer. Front. Oncol. 2013, 3, 153. [Google Scholar] [CrossRef] [PubMed]
- Londin, E.; Loher, P.; Telonis, A.G.; Quann, K.; Clark, P.; Jing, Y.; Hatzimichael, E.; Kirino, Y.; Honda, S.; Lally, M.; et al. Analysis of 13 cell types reveals evidence for the expression of numerous novel primate- and tissue-specific microRNAs. Proc. Natl. Acad. Sci. USA 2015, 112, E1106–E1115. [Google Scholar] [CrossRef] [PubMed]
- Kamanu, T.K.; Radovanovic, A.; Archer, J.A.; Bajic, V.B. Exploration of miRNA families for hypotheses generation. Sci. Rep. 2013, 3, 2940. [Google Scholar] [CrossRef] [PubMed]
- Gambari, R.; Brognara, E.; Spandidos, D.A.; Fabbri, E. Targeting oncomiRNAs and mimicking tumor suppressor miRNAs: Nuew trends in the development of miRNA therapeutic strategies in oncology (Review). Int. J. Oncol. 2016, 49, 5–32. [Google Scholar] [CrossRef] [PubMed]
- Wong, N.W.; Chen, Y.; Chen, S.; Wang, X. OncomiR: An online resource for exploring pan-cancer microRNA dysregulation. Bioinformatics 2018, 34, 713–715. [Google Scholar] [CrossRef] [PubMed]
- Almeida, M.I.; Reis, R.M.; Calin, G.A. MicroRNA history: Discovery, recent applications, and next frontiers. Mutat. Res. 2011, 717, 1–8. [Google Scholar] [CrossRef]
- Cretoiu, D.; Xu, J.; Xiao, J.; Suciu, N.; Cretoiu, S.M. Circulating MicroRNAs as Potential Molecular Biomarkers in Pathophysiological Evolution of Pregnancy. Dis. Mark. 2016, 2016, 3851054. [Google Scholar] [CrossRef]
- Erbes, T.; Hirschfeld, M.; Rucker, G.; Jaeger, M.; Boas, J.; Iborra, S.; Mayer, S.; Gitsch, G.; Stickeler, E. Feasibility of urinary microRNA detection in breast cancer patients and its potential as an innovative non-invasive biomarker. BMC Cancer 2015, 15, 193. [Google Scholar] [CrossRef]
- Vaksman, O.; Trope, C.; Davidson, B.; Reich, R. Exosome-derived miRNAs and ovarian carcinoma progression. Carcinogenesis 2014, 35, 2113–2120. [Google Scholar] [CrossRef]
- Tokuhisa, M.; Ichikawa, Y.; Kosaka, N.; Ochiya, T.; Yashiro, M.; Hirakawa, K.; Kosaka, T.; Makino, H.; Akiyama, H.; Kunisaki, C.; et al. Exosomal miRNAs from Peritoneum Lavage Fluid as Potential Prognostic Biomarkers of Peritoneal Metastasis in Gastric Cancer. PLoS ONE 2015, 10, e0130472. [Google Scholar] [CrossRef]
- Weber, J.A.; Baxter, D.H.; Zhang, S.; Huang, D.Y.; Huang, K.H.; Lee, M.J.; Galas, D.J.; Wang, K. The microRNA spectrum in 12 body fluids. Clin. Chem. 2010, 56, 1733–1741. [Google Scholar] [CrossRef] [PubMed]
- Xie, Z.; Yin, X.; Gong, B.; Nie, W.; Wu, B.; Zhang, X.; Huang, J.; Zhang, P.; Zhou, Z.; Li, Z. Salivary microRNAs show potential as a noninvasive biomarker for detecting resectable pancreatic cancer. Cancer Prev. Res. (Phila.) 2015, 8, 165–173. [Google Scholar] [CrossRef] [PubMed]
- Akers, J.C.; Ramakrishnan, V.; Kim, R.; Skog, J.; Nakano, I.; Pingle, S.; Kalinina, J.; Hua, W.; Kesari, S.; Mao, Y.; et al. MiR-21 in the extracellular vesicles (EVs) of cerebrospinal fluid (CSF): A platform for glioblastoma biomarker development. PLoS ONE 2013, 8, e78115. [Google Scholar] [CrossRef] [PubMed]
- Shin, Y.M.; Yun, J.; Lee, O.J.; Han, H.S.; Lim, S.N.; An, J.Y.; Lee, K.H.; Lee, K.M.; Choe, K.H. Diagnostic Value of Circulating Extracellular miR-134, miR-185, and miR-22 Levels in Lung Adenocarcinoma-Associated Malignant Pleural Effusion. Cancer Res. Treat. 2014, 46, 178–185. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Sun, H.; Wang, X.; Yu, Q.; Li, S.; Yu, X.; Gong, W. Increased exosomal microRNA-21 and microRNA-146a levels in the cervicovaginal lavage specimens of patients with cervical cancer. Int. J. Mol. Sci. 2014, 15, 758–773. [Google Scholar] [CrossRef] [PubMed]
- Schwarzenbach, H.; Nishida, N.; Calin, G.A.; Pantel, K. Clinical relevance of circulating cell-free microRNAs in cancer. Nat. Rev. Clin. Oncol. 2014, 11, 145–156. [Google Scholar] [CrossRef]
- Anfossi, S.; Babayan, A.; Pantel, K.; Calin, G.A. Clinical utility of circulating non-coding RNAs—An update. Nat. Rev. Clin. Oncol. 2018, 15, 541–563. [Google Scholar] [CrossRef]
- Fabbri, M. MicroRNAs and miRceptors: A new mechanism of action for intercellular communication. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2018, 373. [Google Scholar] [CrossRef]
- Chevillet, J.R.; Kang, Q.; Ruf, I.K.; Briggs, H.A.; Vojtech, L.N.; Hughes, S.M.; Cheng, H.H.; Arroyo, J.D.; Meredith, E.K.; Gallichotte, E.N.; et al. Quantitative and stoichiometric analysis of the microRNA content of exosomes. Proc. Natl. Acad. Sci. USA 2014, 111, 14888–14893. [Google Scholar] [CrossRef]
- Morello, M.; Minciacchi, V.R.; de Candia, P.; Yang, J.; Posadas, E.; Kim, H.; Griffiths, D.; Bhowmick, N.; Chung, L.W.; Gandellini, P.; et al. Large oncosomes mediate intercellular transfer of functional microRNA. Cell Cycle 2013, 12, 3526–3536. [Google Scholar] [CrossRef]
- Nakamura, K.; Sawada, K.; Yoshimura, A.; Kinose, Y.; Nakatsuka, E.; Kimura, T. Clinical relevance of circulating cell-free microRNAs in ovarian cancer. Mol. Cancer 2016, 15, 48. [Google Scholar] [CrossRef] [PubMed]
- Faraldi, M.; Gomarasca, M.; Banfi, G.; Lombardi, G. Free Circulating miRNAs Measurement in Clinical Settings: The Still Unsolved Issue of the Normalization. Adv. Clin. Chem. 2018, 87, 113–139. [Google Scholar] [CrossRef] [PubMed]
- Meng, X.; Muller, V.; Milde-Langosch, K.; Trillsch, F.; Pantel, K.; Schwarzenbach, H. Diagnostic and prognostic relevance of circulating exosomal miR-373, miR-200a, miR-200b and miR-200c in patients with epithelial ovarian cancer. Oncotarget 2016, 7, 16923–16935. [Google Scholar] [CrossRef] [PubMed]
- Pan, C.; Stevic, I.; Muller, V.; Ni, Q.; Oliveira-Ferrer, L.; Pantel, K.; Schwarzenbach, H. Exosomal microRNAs as tumor markers in epithelial ovarian cancer. Mol. Oncol. 2018, 12, 1935–1948. [Google Scholar] [CrossRef]
- Kobayashi, M.; Sawada, K.; Nakamura, K.; Yoshimura, A.; Miyamoto, M.; Shimizu, A.; Ishida, K.; Nakatsuka, E.; Kodama, M.; Hashimoto, K.; et al. Exosomal miR-1290 is a potential biomarker of high-grade serous ovarian carcinoma and can discriminate patients from those with malignancies of other histological types. J. Ovarian Res. 2018, 11, 81. [Google Scholar] [CrossRef]
- Deftu, A.T.; Radu, B.M.; Cretoiu, D.; Deftu, A.F.; Cretoiu, S.M.; Xiao, J. Exosomes as intercellular communication messengers for cardiovascular and cerebrovascular diseases. In Exosomes; Edelstein, L., Smythies, J., Quesenberry, P., Noble, D., Eds.; Academic Press, Elsevier Inc.: Amsterdam, The Netherlands, 2020; pp. 199–238. [Google Scholar]
- Taylor, D.D.; Gercel-Taylor, C. MicroRNA signatures of tumor-derived exosomes as diagnostic biomarkers of ovarian cancer. Gynecol. Oncol. 2008, 110, 13–21. [Google Scholar] [CrossRef]
- Zhou, J.; Gong, G.; Tan, H.; Dai, F.; Zhu, X.; Chen, Y.; Wang, J.; Liu, Y.; Chen, P.; Wu, X.; et al. Urinary microRNA-30a-5p is a potential biomarker for ovarian serous adenocarcinoma. Oncol. Rep. 2015, 33, 2915–2923. [Google Scholar] [CrossRef]
- Zhang, H.; Xu, S.; Liu, X. MicroRNA profiling of plasma exosomes from patients with ovarian cancer using high-throughput sequencing. Oncol. Lett. 2019, 17, 5601–5607. [Google Scholar] [CrossRef]
- Penyige, A.; Marton, E.; Soltesz, B.; Szilagyi-Bonizs, M.; Poka, R.; Lukacs, J.; Szeles, L.; Nagy, B. Circulating miRNA Profiling in Plasma Samples of Ovarian Cancer Patients. Int. J. Mol. Sci. 2019, 20, 4533. [Google Scholar] [CrossRef]
- Vilming Elgaaen, B.; Olstad, O.K.; Haug, K.B.; Brusletto, B.; Sandvik, L.; Staff, A.C.; Gautvik, K.M.; Davidson, B. Global miRNA expression analysis of serous and clear cell ovarian carcinomas identifies differentially expressed miRNAs including miR-200c-3p as a prognostic marker. BMC Cancer 2014, 14, 80. [Google Scholar] [CrossRef]
- Yan, J.; Jiang, J.Y.; Meng, X.N.; Xiu, Y.L.; Zong, Z.H. MiR-23b targets cyclin G1 and suppresses ovarian cancer tumorigenesis and progression. J. Exp. Clin. Cancer Res. 2016, 35, 31. [Google Scholar] [CrossRef] [PubMed]
- Shen, W.; Song, M.; Liu, J.; Qiu, G.; Li, T.; Hu, Y.; Liu, H. MiR-26a promotes ovarian cancer proliferation and tumorigenesis. PLoS ONE 2014, 9, e86871. [Google Scholar] [CrossRef] [PubMed]
- Ying, X.; Wei, K.; Lin, Z.; Cui, Y.; Ding, J.; Chen, Y.; Xu, B. MicroRNA-125b Suppresses Ovarian Cancer Progression via Suppression of the Epithelial-Mesenchymal Transition Pathway by Targeting the SET Protein. Cell Physiol. Biochem. 2016, 39, 501–510. [Google Scholar] [CrossRef] [PubMed]
- Guan, Y.; Yao, H.; Zheng, Z.; Qiu, G.; Sun, K. MiR-125b targets BCL3 and suppresses ovarian cancer proliferation. Int. J. Cancer 2011, 128, 2274–2283. [Google Scholar] [CrossRef] [PubMed]
- Mak, C.S.; Yung, M.M.; Hui, L.M.; Leung, L.L.; Liang, R.; Chen, K.; Liu, S.S.; Qin, Y.; Leung, T.H.; Lee, K.F.; et al. MicroRNA-141 enhances anoikis resistance in metastatic progression of ovarian cancer through targeting KLF12/Sp1/survivin axis. Mol. Cancer 2017, 16, 11. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Zhang, S.; Zou, Y.; Wu, L.; Pei, M.; Jiang, Y. miR-145 promotes miR-133b expression through c-myc and DNMT3A-mediated methylation in ovarian cancer cells. J. Cell Physiol. 2019. [Google Scholar] [CrossRef]
- Cui, Y.; She, K.; Tian, D.; Zhang, P.; Xin, X. miR-146a Inhibits Proliferation and Enhances Chemosensitivity in Epithelial Ovarian Cancer via Reduction of SOD2. Oncol. Res. 2016, 23, 275–282. [Google Scholar] [CrossRef]
- Yan, M.; Yang, X.; Shen, R.; Wu, C.; Wang, H.; Ye, Q.; Yang, P.; Zhang, L.; Chen, M.; Wan, B.; et al. miR-146b promotes cell proliferation and increases chemosensitivity, but attenuates cell migration and invasion via FBXL10 in ovarian cancer. Cell Death Dis. 2018, 9, 1123. [Google Scholar] [CrossRef]
- Wen, Z.; Zhao, S.; Liu, S.; Liu, Y.; Li, X.; Li, S. MicroRNA-148a inhibits migration and invasion of ovarian cancer cells via targeting sphingosine-1-phosphate receptor 1. Mol. Med. Rep. 2015, 12, 3775–3780. [Google Scholar] [CrossRef]
- Shi, C.; Yang, Y.; Zhang, L.; Yu, J.; Qin, S.; Xu, H.; Gao, Y. MiR-200a-3p promoted the malignant behaviors of ovarian cancer cells through regulating PCDH9. Oncol. Targets Ther. 2019, 12, 8329–8338. [Google Scholar] [CrossRef]
- Zhang, Z.; Zhang, L.; Wang, B.; Wei, R.; Wang, Y.; Wan, J.; Zhang, C.; Zhao, L.; Zhu, X.; Zhang, Y.; et al. MiR-337-3p suppresses proliferation of epithelial ovarian cancer by targeting PIK3CA and PIK3CB. Cancer Lett. 2019, 469, 54–67. [Google Scholar] [CrossRef] [PubMed]
- Liang, T.; Guo, Q.; Li, L.; Cheng, Y.; Ren, C.; Zhang, G. MicroRNA-433 inhibits migration and invasion of ovarian cancer cells via targeting Notch1. Neoplasma 2016, 63, 696–704. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Zhang, J.Y.; Lu, L.J.; Wang, C.H.; Wang, L.H. MiR-630 promotes epithelial ovarian cancer proliferation and invasion via targeting KLF6. Eur. Rev. Med. Pharmacol. Sci. 2017, 21, 4542–4547. [Google Scholar] [PubMed]
- Yang, B.; Sun, L.; Liang, L. MiRNA-802 suppresses proliferation and migration of epithelial ovarian cancer cells by targeting YWHAZ. J. Ovarian Res. 2019, 12, 100. [Google Scholar] [CrossRef] [PubMed]
- Jiao, Y.; Zhu, G.; Yu, J.; Li, Y.; Wu, M.; Zhao, J.; Tian, X. miR-1271 inhibits growth, invasion and epithelial-mesenchymal transition by targeting ZEB1 in ovarian cancer cells. Oncol. Targets Ther. 2019, 12, 6973–6980. [Google Scholar] [CrossRef] [PubMed]
- Clarke-Pearson, D.L. Clinical practice. Screening for ovarian cancer. N. Engl. J. Med. 2009, 361, 170–177. [Google Scholar] [CrossRef]
- Resnick, K.E.; Alder, H.; Hagan, J.P.; Richardson, D.L.; Croce, C.M.; Cohn, D.E. The detection of differentially expressed microRNAs from the serum of ovarian cancer patients using a novel real-time PCR platform. Gynecol. Oncol. 2009, 112, 55–59. [Google Scholar] [CrossRef]
- Gao, Y.C.; Wu, J. MicroRNA-200c and microRNA-141 as potential diagnostic and prognostic biomarkers for ovarian cancer. Tumour Biol. 2015, 36, 4843–4850. [Google Scholar] [CrossRef]
- Zuberi, M.; Mir, R.; Das, J.; Ahmad, I.; Javid, J.; Yadav, P.; Masroor, M.; Ahmad, S.; Ray, P.C.; Saxena, A. Expression of serum miR-200a, miR-200b, and miR-200c as candidate biomarkers in epithelial ovarian cancer and their association with clinicopathological features. Clin. Transl. Oncol. 2015, 17, 779–787. [Google Scholar] [CrossRef]
- Yokoi, A.; Matsuzaki, J.; Yamamoto, Y.; Yoneoka, Y.; Takahashi, K.; Shimizu, H.; Uehara, T.; Ishikawa, M.; Ikeda, S.I.; Sonoda, T.; et al. Integrated extracellular microRNA profiling for ovarian cancer screening. Nat. Commun. 2018, 9, 4319. [Google Scholar] [CrossRef]
- Ren, X.; Zhang, H.; Cong, H.; Wang, X.; Ni, H.; Shen, X.; Ju, S. Diagnostic Model of Serum miR-193a-5p, HE4 and CA125 Improves the Diagnostic Efficacy of Epithelium Ovarian Cancer. Pathol. Oncol. Res. 2018, 24, 739–744. [Google Scholar] [CrossRef] [PubMed]
- Ayaz, L.; Cayan, F.; Balci, S.; Gorur, A.; Akbayir, S.; Yildirim Yaroglu, H.; Dogruer Unal, N.; Tamer, L. Circulating microRNA expression profiles in ovarian cancer. J. Obstet. Gynaecol. 2014, 34, 620–624. [Google Scholar] [CrossRef] [PubMed]
- Palma Flores, C.; Garcia-Vazquez, R.; Gallardo Rincon, D.; Ruiz-Garcia, E.; Astudillo de la Vega, H.; Marchat, L.A.; Salinas Vera, Y.M.; Lopez-Camarillo, C. MicroRNAs driving invasion and metastasis in ovarian cancer: Opportunities for translational medicine (Review). Int. J. Oncol. 2017, 50, 1461–1476. [Google Scholar] [CrossRef] [PubMed]
- Loganathan, S.; Kandala, P.K.; Gupta, P.; Srivastava, S.K. Inhibition of EGFR-AKT axis results in the suppression of ovarian tumors in vitro and in preclinical mouse model. PLoS ONE 2012, 7, e43577. [Google Scholar] [CrossRef]
- Zhou, X.; Hu, Y.; Dai, L.; Wang, Y.; Zhou, J.; Wang, W.; Di, W.; Qiu, L. MicroRNA-7 inhibits tumor metastasis and reverses epithelial-mesenchymal transition through AKT/ERK1/2 inactivation by targeting EGFR in epithelial ovarian cancer. PLoS ONE 2014, 9, e96718. [Google Scholar] [CrossRef]
- Vang, S.; Wu, H.T.; Fischer, A.; Miller, D.H.; MacLaughlan, S.; Douglass, E.; Comisar, L.; Steinhoff, M.; Collins, C.; Smith, P.J.; et al. Identification of ovarian cancer metastatic miRNAs. PLoS ONE 2013, 8, e58226. [Google Scholar] [CrossRef]
- Jin, M.; Yang, Z.; Ye, W.; Xu, H.; Hua, X. MicroRNA-150 predicts a favorable prognosis in patients with epithelial ovarian cancer, and inhibits cell invasion and metastasis by suppressing transcriptional repressor ZEB1. PLoS ONE 2014, 9, e103965. [Google Scholar] [CrossRef]
- Bhaumik, D.; Scott, G.K.; Schokrpur, S.; Patil, C.K.; Campisi, J.; Benz, C.C. Expression of microRNA-146 suppresses NF-kappaB activity with reduction of metastatic potential in breast cancer cells. Oncogene 2008, 27, 5643–5647. [Google Scholar] [CrossRef]
- Wilczynski, M.; Zytko, E.; Szymanska, B.; Dzieniecka, M.; Nowak, M.; Danielska, J.; Stachowiak, G.; Wilczynski, J.R. Expression of miR-146a in patients with ovarian cancer and its clinical significance. Oncol. Lett. 2017, 14, 3207–3214. [Google Scholar] [CrossRef]
- Li, J.; Liang, S.; Jin, H.; Xu, C.; Ma, D.; Lu, X. Tiam1, negatively regulated by miR-22, miR-183 and miR-31, is involved in migration, invasion and viability of ovarian cancer cells. Oncol. Rep. 2012, 27, 1835–1842. [Google Scholar] [CrossRef]
- Fang, Y.; Xu, C.; Fu, Y. MicroRNA-17-5p induces drug resistance and invasion of ovarian carcinoma cells by targeting PTEN signaling. J. Biol. Res. (Thessalon) 2015, 22, 12. [Google Scholar] [CrossRef] [PubMed]
- Turchinovich, A.; Samatov, T.R.; Tonevitsky, A.G.; Burwinkel, B. Circulating miRNAs: Cell-cell communication function? Front. Genet. 2013, 4, 119. [Google Scholar] [CrossRef] [PubMed]
- Hannafon, B.N.; Ding, W.Q. Intercellular communication by exosome-derived microRNAs in cancer. Int. J. Mol. Sci. 2013, 14, 14240–14269. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, K.; Sawada, K.; Kobayashi, M.; Miyamoto, M.; Shimizu, A.; Yamamoto, M.; Kinose, Y.; Kimura, T. Role of the Exosome in Ovarian Cancer Progression and Its Potential as a Therapeutic Target. Cancers 2019, 11, 1147. [Google Scholar] [CrossRef]
- Mihanfar, A.; Fattahi, A.; Nejabati, H.R. MicroRNA-mediated drug resistance in ovarian cancer. J. Cell Physiol. 2019, 234, 3180–3191. [Google Scholar] [CrossRef]
- Zou, Y.T.; Gao, J.Y.; Wang, H.L.; Wang, Y.; Wang, H.; Li, P.L. Downregulation of microRNA-630 inhibits cell proliferation and invasion and enhances chemosensitivity in human ovarian carcinoma. Genet. Mol. Res. 2015, 14, 8766–8777. [Google Scholar] [CrossRef]
- Yang, H.; Kong, W.; He, L.; Zhao, J.J.; O’Donnell, J.D.; Wang, J.; Wenham, R.M.; Coppola, D.; Kruk, P.A.; Nicosia, S.V.; et al. MicroRNA expression profiling in human ovarian cancer: MiR-214 induces cell survival and cisplatin resistance by targeting PTEN. Cancer Res. 2008, 68, 425–433. [Google Scholar] [CrossRef]
- Pink, R.C.; Samuel, P.; Massa, D.; Caley, D.P.; Brooks, S.A.; Carter, D.R. The passenger strand, miR-21-3p, plays a role in mediating cisplatin resistance in ovarian cancer cells. Gynecol. Oncol. 2015, 137, 143–151. [Google Scholar] [CrossRef]
- Li, X.; Chen, W.; Zeng, W.; Wan, C.; Duan, S.; Jiang, S. microRNA-137 promotes apoptosis in ovarian cancer cells via the regulation of XIAP. Br. J. Cancer 2017, 116, 66–76. [Google Scholar] [CrossRef]
- Tian, S.; Zhang, M.; Chen, X.; Liu, Y.; Lou, G. MicroRNA-595 sensitizes ovarian cancer cells to cisplatin by targeting ABCB1. Oncotarget 2016, 7, 87091–87099. [Google Scholar] [CrossRef]
- Huh, J.H.; Kim, T.H.; Kim, K.; Song, J.A.; Jung, Y.J.; Jeong, J.Y.; Lee, M.J.; Kim, Y.K.; Lee, D.H.; An, H.J. Dysregulation of miR-106a and miR-591 confers paclitaxel resistance to ovarian cancer. Br. J. Cancer 2013, 109, 452–461. [Google Scholar] [CrossRef] [PubMed]
- Zamfir-Chiru-Anton, A.; Gheorghe, D.; Nitu, L.; Bertesteanu, S. Relaţia dintre metaloproteinaze şi răspunsul imun în cancer. Rom. J. Med. Pract. 2014, 9, 211–213. [Google Scholar]
- Helleman, J.; Jansen, M.P.; Burger, C.; van der Burg, M.E.; Berns, E.M. Integrated genomics of chemotherapy resistant ovarian cancer: A role for extracellular matrix, TGFbeta and regulating microRNAs. Int. J. Biochem. Cell Biol. 2010, 42, 25–30. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Jiao, J.W.; Sun, K.X.; Zong, Z.H.; Zhao, Y. MicroRNA-133b targets glutathione S-transferase pi expression to increase ovarian cancer cell sensitivity to chemotherapy drugs. Drug Des. Dev. Ther. 2015, 9, 5225–5235. [Google Scholar] [CrossRef] [PubMed]
- Sun, K.X.; Jiao, J.W.; Chen, S.; Liu, B.L.; Zhao, Y. MicroRNA-186 induces sensitivity of ovarian cancer cells to paclitaxel and cisplatin by targeting ABCB1. J. Ovarian Res. 2015, 8, 80. [Google Scholar] [CrossRef]
- Wu, D.D.; Li, X.S.; Meng, X.N.; Yan, J.; Zong, Z.H. MicroRNA-873 mediates multidrug resistance in ovarian cancer cells by targeting ABCB1. Tumour Biol. 2016, 37, 10499–10506. [Google Scholar] [CrossRef]
- Chen, Y.; Gao, D.Y.; Huang, L. In vivo delivery of miRNAs for cancer therapy: Challenges and strategies. Adv. Drug Deliv. Rev. 2015, 81, 128–141. [Google Scholar] [CrossRef]
- Zong, C.; Wang, J.; Shi, T.M. MicroRNA 130b enhances drug resistance in human ovarian cancer cells. Tumour Biol. 2014, 35, 12151–12156. [Google Scholar] [CrossRef]
- Chen, S.; Chen, X.; Xiu, Y.L.; Sun, K.X.; Zong, Z.H.; Zhao, Y. microRNA 490-3P enhances the drug-resistance of human ovarian cancer cells. J. Ovarian Res. 2014, 7, 84. [Google Scholar] [CrossRef]
- Petrillo, M.; Zannoni, G.F.; Beltrame, L.; Martinelli, E.; DiFeo, A.; Paracchini, L.; Craparotta, I.; Mannarino, L.; Vizzielli, G.; Scambia, G.; et al. Identification of high-grade serous ovarian cancer miRNA species associated with survival and drug response in patients receiving neoadjuvant chemotherapy: A retrospective longitudinal analysis using matched tumor biopsies. Ann. Oncol. 2016, 27, 625–634. [Google Scholar] [CrossRef]
- Meijer, L.L.; Puik, J.R.; Vivaldi, C.; Capula, M.; Vasile, E.; Kazemier, G.; Giovannetti, E. Modification of Drug Response in Cancer by MicroRNAs. In MicroRNAs in Diseases and Disorders; Peplow, P.V., Martinez, B., Calin, G.A., Esquela-Kerscher, A., Eds.; Royal Society of Chemistry: Cambridge, UK, 2019; pp. 416–451. [Google Scholar]
- Tanaka, H.; Matsushima, H.; Mizumoto, N.; Takashima, A. Classification of chemotherapeutic agents based on their differential in vitro effects on dendritic cells. Cancer Res. 2009, 69, 6978–6986. [Google Scholar] [CrossRef]
- Mollaei, H.; Safaralizadeh, R.; Rostami, Z. MicroRNA replacement therapy in cancer. J. Cell Physiol. 2019, 234, 12369–12384. [Google Scholar] [CrossRef] [PubMed]
- Hutvagner, G.; Zamore, P.D. A microRNA in a multiple-turnover RNAi enzyme complex. Science 2002, 297, 2056–2060. [Google Scholar] [CrossRef] [PubMed]
- Li, S.D.; Zhang, J.R.; Wang, Y.Q.; Wan, X.P. The role of microRNAs in ovarian cancer initiation and progression. J. Cell Mol. Med. 2010, 14, 2240–2249. [Google Scholar] [CrossRef] [PubMed]
- Banno, K.; Yanokura, M.; Iida, M.; Adachi, M.; Nakamura, K.; Nogami, Y.; Umene, K.; Masuda, K.; Kisu, I.; Nomura, H.; et al. Application of microRNA in diagnosis and treatment of ovarian cancer. Biomed. Res. Int. 2014, 2014, 232817. [Google Scholar] [CrossRef] [PubMed]
- Li, N.; Wang, L.; Tan, G.; Guo, Z.; Liu, L.; Yang, M.; He, J. MicroRNA-218 inhibits proliferation and invasion in ovarian cancer by targeting Runx2. Oncotarget 2017, 8, 91530–91541. [Google Scholar] [CrossRef] [PubMed]
- Cheng, W.; Liu, T.; Wan, X.; Gao, Y.; Wang, H. MicroRNA-199a targets CD44 to suppress the tumorigenicity and multidrug resistance of ovarian cancer-initiating cells. FEBS J. 2012, 279, 2047–2059. [Google Scholar] [CrossRef]
- Wang, F.; Chang, J.T.; Kao, C.J.; Huang, R.S. High Expression of miR-532-5p, a Tumor Suppressor, Leads to Better Prognosis in Ovarian Cancer Both In Vivo and In Vitro. Mol. Cancer Ther. 2016, 15, 1123–1131. [Google Scholar] [CrossRef]
- Fu, X.; Li, Y.; Alvero, A.; Li, J.; Wu, Q.; Xiao, Q.; Peng, Y.; Hu, Y.; Li, X.; Yan, W.; et al. MicroRNA-222-3p/GNAI2/AKT axis inhibits epithelial ovarian cancer cell growth and associates with good overall survival. Oncotarget 2016, 7, 80633–80654. [Google Scholar] [CrossRef]
- Fukagawa, S.; Miyata, K.; Yotsumoto, F.; Kiyoshima, C.; Nam, S.O.; Anan, H.; Katsuda, T.; Miyahara, D.; Murata, M.; Yagi, H.; et al. MicroRNA-135a-3p as a promising biomarker and nucleic acid therapeutic agent for ovarian cancer. Cancer Sci. 2017, 108, 886–896. [Google Scholar] [CrossRef]
- Dai, F.; Zhang, Y.; Zhu, X.; Shan, N.; Chen, Y. Anticancer role of MUC1 aptamer-miR-29b chimera in epithelial ovarian carcinoma cells through regulation of PTEN methylation. Target Oncol. 2012, 7, 217–225. [Google Scholar] [CrossRef] [PubMed]
- Denoyelle, C.; Lambert, B.; Meryet-Figuiere, M.; Vigneron, N.; Brotin, E.; Lecerf, C.; Abeilard, E.; Giffard, F.; Louis, M.H.; Gauduchon, P.; et al. miR-491-5p-induced apoptosis in ovarian carcinoma depends on the direct inhibition of both BCL-XL and EGFR leading to BIM activation. Cell Death Dis. 2014, 5, e1445. [Google Scholar] [CrossRef] [PubMed]
- Kleemann, M.; Schneider, H.; Unger, K.; Sander, P.; Schneider, E.M.; Fischer-Posovszky, P.; Handrick, R.; Otte, K. MiR-744-5p inducing cell death by directly targeting HNRNPC and NFIX in ovarian cancer cells. Sci. Rep. 2018, 8, 9020. [Google Scholar] [CrossRef] [PubMed]
- Pecot, C.V.; Rupaimoole, R.; Yang, D.; Akbani, R.; Ivan, C.; Lu, C.; Wu, S.; Han, H.D.; Shah, M.Y.; Rodriguez-Aguayo, C.; et al. Tumour angiogenesis regulation by the miR-200 family. Nat. Commun. 2013, 4, 2427. [Google Scholar] [CrossRef]
- Joshi, H.P.; Subramanian, I.V.; Schnettler, E.K.; Ghosh, G.; Rupaimoole, R.; Evans, C.; Saluja, M.; Jing, Y.; Cristina, I.; Roy, S.; et al. Dynamin 2 along with microRNA-199a reciprocally regulate hypoxia-inducible factors and ovarian cancer metastasis. Proc. Natl. Acad. Sci. USA 2014, 111, 5331–5336. [Google Scholar] [CrossRef]
- Ohyagi-Hara, C.; Sawada, K.; Kamiura, S.; Tomita, Y.; Isobe, A.; Hashimoto, K.; Kinose, Y.; Mabuchi, S.; Hisamatsu, T.; Takahashi, T.; et al. miR-92a inhibits peritoneal dissemination of ovarian cancer cells by inhibiting integrin alpha5 expression. Am. J. Pathol. 2013, 182, 1876–1889. [Google Scholar] [CrossRef]
- Shi, H.; Shen, H.; Xu, J.; Zhao, S.; Yao, S.; Jiang, N. MiR-143-3p suppresses the progression of ovarian cancer. Am. J. Transl. Res. 2018, 10, 866–874. [Google Scholar]
- Mateescu, B.; Batista, L.; Cardon, M.; Gruosso, T.; de Feraudy, Y.; Mariani, O.; Nicolas, A.; Meyniel, J.P.; Cottu, P.; Sastre-Garau, X.; et al. miR-141 and miR-200a act on ovarian tumorigenesis by controlling oxidative stress response. Nat. Med. 2011, 17, 1627–1635. [Google Scholar] [CrossRef]
- Garzon, R.; Marcucci, G.; Croce, C.M. Targeting microRNAs in cancer: Rationale, strategies and challenges. Nat. Rev. Drug Discov. 2010, 9, 775–789. [Google Scholar] [CrossRef]
- Raza, K. Clustering analysis of cancerous microarray data. J. Chem. Pharm. Res. 2014, 6, 488–493. [Google Scholar]
- Ducie, J.; Dao, F.; Considine, M.; Olvera, N.; Shaw, P.A.; Kurman, R.J.; Shih, I.M.; Soslow, R.A.; Cope, L.; Levine, D.A. Molecular analysis of high-grade serous ovarian carcinoma with and without associated serous tubal intra-epithelial carcinoma. Nat. Commun. 2017, 8, 990. [Google Scholar] [CrossRef] [PubMed]
- McDonald, M.E.; Salinas, E.A.; Devor, E.J.; Newtson, A.M.; Thiel, K.W.; Goodheart, M.J.; Bender, D.P.; Smith, B.J.; Leslie, K.K.; Gonzalez-Bosquet, J. Molecular Characterization of Non-responders to Chemotherapy in Serous Ovarian Cancer. Int. J. Mol. Sci. 2019, 20, 1175. [Google Scholar] [CrossRef] [PubMed]
- Dahiya, N.; Sherman-Baust, C.A.; Wang, T.L.; Davidson, B.; Shih Ie, M.; Zhang, Y.; Wood, W., 3rd; Becker, K.G.; Morin, P.J. MicroRNA expression and identification of putative miRNA targets in ovarian cancer. PLoS ONE 2008, 3, e2436. [Google Scholar] [CrossRef] [PubMed]
- Bodelon, C.; Killian, J.K.; Sampson, J.N.; Anderson, W.F.; Matsuno, R.; Brinton, L.A.; Lissowska, J.; Anglesio, M.S.; Bowtell, D.D.L.; Doherty, J.A.; et al. Molecular Classification of Epithelial Ovarian Cancer Based on Methylation Profiling: Evidence for Survival Heterogeneity. Clin. Cancer Res. 2019, 25, 5937–5946. [Google Scholar] [CrossRef]
- Wang, C.; Armasu, S.M.; Kalli, K.R.; Maurer, M.J.; Heinzen, E.P.; Keeney, G.L.; Cliby, W.A.; Oberg, A.L.; Kaufmann, S.H.; Goode, E.L. Pooled Clustering of High-Grade Serous Ovarian Cancer Gene Expression Leads to Novel Consensus Subtypes Associated with Survival and Surgical Outcomes. Clin. Cancer Res. 2017, 23, 4077–4085. [Google Scholar] [CrossRef]
- Li, L.; Cai, S.; Liu, S.; Feng, H.; Zhang, J. Bioinformatics analysis to screen the key prognostic genes in ovarian cancer. J. Ovarian Res. 2017, 10, 27. [Google Scholar] [CrossRef]
- Luo, J.; Pan, C.; Xiang, G.; Yin, Y. A Novel Cluster-Based Computational Method to Identify miRNA Regulatory Modules. IEEE/ACM Trans. Comput. Biol. Bioinform. 2019, 16, 681–687. [Google Scholar] [CrossRef]
- Kuhn, H.W. The Hungarian Method for The Assignment Problem. Nav. Res. Logist. Q. 1955, 2, 83–97. [Google Scholar] [CrossRef]
- Berge, C. Paths, trees, and flowers. Can. J. Math. 1965, 449–467. [Google Scholar]
- Ding, L.; Feng, Z.; Bai, Y. Clustering analysis of microRNA and mRNA expression data from TCGA using maximum edge-weighted matching algorithms. BMC Med. Genom. 2019, 12, 117. [Google Scholar] [CrossRef]
- Elias, K.M.; Fendler, W.; Stawiski, K.; Fiascone, S.J.; Vitonis, A.F.; Berkowitz, R.S.; Frendl, G.; Konstantinopoulos, P.; Crum, C.P.; Kedzierska, M.; et al. Diagnostic potential for a serum miRNA neural network for detection of ovarian cancer. Elife 2017, 6. [Google Scholar] [CrossRef] [PubMed]
- Hu, X.; Macdonald, D.M.; Huettner, P.C.; Feng, Z.; El Naqa, I.M.; Schwarz, J.K.; Mutch, D.G.; Grigsby, P.W.; Powell, S.N.; Wang, X. A miR-200 microRNA cluster as prognostic marker in advanced ovarian cancer. Gynecol. Oncol. 2009, 114, 457–464. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.; Zhang, L.; Wang, B.; Li, F.; Zhang, Z. Feature clustering based support vector machine recursive feature elimination for gene selection. Appl. Intell. 2018, 48, 594–607. [Google Scholar] [CrossRef]
- Dong, J.; Xu, M. A 19miRNA Support Vector Machine classifier and a 6miRNA risk score system designed for ovarian cancer patients. Oncol. Rep. 2019, 41, 3233–3243. [Google Scholar] [CrossRef]
| miRNA | Type of Ovarian Cells | Changes in Ovarian Cancer |
|---|---|---|
| miR-23b | OVCAR3, HO8910-PM, SKOV3/DDP cells | miR-23b transfection slows down cell growth, blocks cell cycle in G1, increases the number of apoptotic cells, and reduces the rate of cell migration [46] |
| miR-26a | SKOV-3, ES2 cells | Overexpression of miR-26b in ovarian cancer cells [47] miR26-b is involved in tumor progression by targeting estrogen receptor alpha (ERα) [47] |
| miR-125b | SKOV3 cells | Overexpression or downregulation of miR-125b did not affect the in vivo cancer cells proliferation and apoptosis [48] |
| miR-125b | SKOV3, ES2 cells | Low expression of miR-125b in ovarian tumor cells Through overexpression of miR-125b, tumor proliferation is prevented by controlling the G2 phase of the cell cycle and by targeting the BCL3 gene [49] |
| miR-141 | Ovarian cancer cells (SKOV3, OVCA433, and A2780cp) Cervical cancer cells (OV2008 and C13*) | miR-141 increases anchorage-independent growth and survival of ovarian cancer cells in vitro [50] miR-141 enhances anoikis resistance in metastatic tumor progression by targeting the Kruppel-related zinc finger protein AP-2rep (KLF12)/specificity protein 1 (Sp1)/survivin axis |
| miR-145 miR-133b | Ovarian cancer cells (3AO, SKOV3) | miR-145 promotes miR-133b expression through c-myc and DNMT3A-mediated methylation in ovarian cancer cells [51] miR-133b upregulation mediates the inhibitory activity of miR-145, in aerobic glycolysis conditions (i.e., Warburg effect), by targeting hexokinase-2 (HK2) in ovarian cancer cells [51] |
| miR-146a | Epithelial ovarian cancer cells (OVCAR3, CAOV3, HEY) Normal ovarian cells (HOSE) | Through overexpression, it acts as a tumor suppressor, but through its downregulation, it inhibits apoptosis, increases proliferation and chemoresistance [52] |
| miR-146b | Epithelial ovarian cancer cells (SKOV3, OVCAR3, HO8910, A2780) | miR-146b overexpression upregulates VIM and ZO-1 and consequently inhibits tumor dissemination [53] |
| miR-148a | Ovarian cancer cells (SKOV3, OVCAR, and A2780) Normal ovarian epithelial cells (HUM-CELL-0088) | Downregulation of miR-148a in ovarian cancer cells [54] miR-148a inhibits migration and invasion of ovarian cancer cells via targeting sphingosine-1-phosphate receptor 1 (S1PR1) [54] |
| miR-200a-3p | Ovarian cancer cells (ES2, HO8919PM, SKOV3, HO8910) Ovarian surface epithelial cells (HOSEpiC) | Overexpression of miR-200a-3p strongly promotes the proliferation, colony formation and invasion of ovarian cancer cells [55] Binds the 3′-UTR of PCDH9 and decreased the expression of PCDH9 [55] |
| miR-337-3p | Ovarian cancer cells (ES2, A2780, SKOV-3, OVCAR-3) | miR-337-3p inhibits cell proliferation and decreases the PI3K/AKT signaling pathway activation (its targets are PIK3CA and PIK3C) [56] Its ectopic expression inhibits proliferation and induces apoptosis and cell cycle arrest in G0/G1 phase [56] |
| miR-433 | Ovarian cancer cells (SKOV3 and OVCAR3) | miR-433 inhibits migration and invasion of ovarian cancer cells via targeting Notch1 [57] |
| miR-630 | Ovarian cancer cells (SKOV3) | miR-630 overexpression stimulates in vitro cell proliferation and migration [58] miR-630 targets directly Krüppel-like factor 6 (KLF6) in ovarian cancer cells [58] |
| miR-802 | Epithelial ovarian cancer cells (OVCAR3, SKOV3, A2780, and CAOV3) Normal ovarian surface epithelial cells (HOSEpiC) | miR-802 is downregulated in epithelial ovarian cancer cell lines [59] Overexpression of miR-802 suppresses migration, proliferation, invasion and induces apoptosis in epithelial ovarian cancer cell lines [59] miR-802 directly targets Monooxygenase/Tryptophan 5-Monooxygenase Activation Protein Zeta (YWHAZ) gene in epithelial ovarian cancer cells [59] |
| miR-1271 | Ovarian cancer cells (SKOV3 and CAOV3) Normal ovarian cells (IOSE80) | Suppresses cell viability and invasion in ovarian cancer cells [60] Directly binds the 3ʹ-UTR of ZEB1 mRNA and regulates the expression of ZEB1 [60] |
| miRNA | Type of Ovarian Tissue | Changes in Ovarian Cancer |
|---|---|---|
| miR-23b | Nude mice injected subcutaneously with mock or hsa-miR-23b–transfected OVCAR3 cells | miR-23b induces downregulation of cyclin G1 (CCNG1) in tumor xenografts and reduction of tumor size in mice [46] |
| miR-26a | Nude mice injected subcutaneously with SKOV3 cells transfected with miR-26a or anti-miR-26a | miR-26a is involved in cell proliferation and tumor development in epithelial ovarian cancer induced in animal models [47] |
| miR-125b | Nude mice inoculated with SKOV3 cells that were transfected with the vector control, miR-125b mimic or inhibitor | miR-125b inhibits the in vivo cancer cell migration and invasion [48] |
| miR-125b | Nude mice injected subcutaneously with SKOV3 cells transfected with miR-125b or anti-miR-125b | miR-125b suppresses the development of ovarian cancer [49] |
| miR-141 | BALB/cAnN nude mice injected intraperitoneal with stable SKOV3 miR-141-expressing clones, or A2780cp shSu knockdown clone and the scrambled controls | miR-141 increases tumor growth in vivo and induces the appearance of a great number of macroscopic tumor nodules, especially in the omentum and the peritoneal cavity [50] |
| miR-146b | Nude mice injected with control cells or cells overexpressing miR-146b | Overexpression of miR-146b reduces cell migration and decreases the level of F-box and leucine-rich repeat protein 10 (FBXL10) protein [53] |
| miR-337-3p | Xenograft models of ovarian cancer induced by inoculation of A2780 and OVCAR-3 cells in female BALB/c athymic nude mice | miR-337-3p is a tumor suppressor that controls the expression of p110α and p110β (i.e., PIK3CA and PIK3CB encoded proteins) [56] Tumors injected with miR-337–3p agomiR grew more slowly than those injected with agomiR-NC for both xenograft models [56] |
| miR-630 | Balb/c mice injected subcutaneously with SKOV3 cells transfected with inhibitors, mimics or negative control of miR-630 | miR-630 overexpression stimulates in vivo ovarian cancer proliferation [58] |
| miRNA | Type of Ovarian Tissue | Changes in Ovarian Cancer |
|---|---|---|
| miR-125b | Surgical resection of tumor tissues and the corresponding adjacent normal tissues in epithelial ovarian cancer patients | miR-125b is downregulated in ovarian cancer [48] miR-125b suppresses ovarian cancer progression via suppression of the epithelial-mesenchymal transition pathway by targeting the SET protein (binds directly to the SET 3′-UTR) [48] |
| miR-141 | Surgical specimens of ovarian cancer and normal ovarian tissues | miR-141 is upregulated in clinical ovarian cancer samples having a ~10-fold higher expression than in normal ovary tissues [50] |
| miR-133b miR-145 | Human normal ovarian tissue samples and ovarian carcinomas (e.g., serous cancer, mucinous, endometrioid cancer, clear cell cancer) | miR-145 and miR-133b were downregulated in endothelial ovarian cancer, their expression being positively correlated [51] |
| miR-148a | Surgical resection of ovarian cancer tissue and their matched normal adjacent tissues | Downregulation of miR-148a in ovarian cancer tissue [54] |
| miR-200a-3p | Surgically excised tissue from ovarian cancer patients | miR-200a-3p expression was negatively correlated with the PCDH9 expression in ovarian cancer [55] |
| miR-337-3p | Surgically excised epithelial ovarian cancer specimens | Downregulation of miR-337-3p in epithelial ovarian cancer tissues and correlated with the pathological grade of patients [56] |
| miR-433 | Surgical resections of ovarian cancer tissues and matched normal ovary tissues | Downregulation of miR-433 expression and upregulation of Notch1 expression in ovarian cancer tissues compared with normal ovarian tissues [57] |
| miR-630 | Surgically excised ovarian cancer and normal ovarian tissue samples | miR-630 is upregulated in ovarian cancer [58] |
| miRNA-802 | Surgical specimens of epithelial ovarian cancer and adjacent normal tissues | Down-regulation in epithelial ovarian cancer specimens [59] |
| miR-1271 | Surgical specimens of ovarian cancer tissues and peritumoral normal tissues | Inverse correlation between miR-1271 expression and ZEB1 in ovarian cancer tissues [60] |
| miRNA | Serum/Plasma | Changes in Ovarian Cancer |
|---|---|---|
| miR-26a | Plasma | Upregulation of miR-26a in human epithelial ovarian cancer [47] |
| miRNA-21 miRNA-29a miRNA-92 miRNA-93 miRNA-99b miRNA-126 miRNA-127 miRNA-155 | Serum | Upregulation of miRNA-21, miRNA-29a, miRNA-92, miRNA-93, miRNA-126 in ovarian cancer [62] Downregulation of miRNA-99, miRNA-127, miRNA-155 in ovarian cancer [62] |
| miR-145 miR-133b | Serum | Expression of miR-145 and miR-133b is significantly decreased in the serum of patients with ovarian cancer [51] |
| miR-193a-5p | Serum | Combined detection of miR-193-5p, HE4 and CA125 improves the diagnostic efficacy of epithelium ovarian cancer [66] |
| miR-19a-3p miR-30a-5p miR-645 miR-150-5p miR-191-5p miR-206 miR-34c-5p miR-548a-3p miR-320a miR-574-3p miR-590-5p miR-106b-5p | Plasma | Downregulation of miR-19a-3p, miR-30a-5p, miR-645, miR-150-5p in ovarian cancer [67] Upregulation of miR-191-5p, miR-206, miR-34c-5p, miR-548a-3p, miR-320a, miR-574-3p, miR-590-5p, miR-106b-5p in ovarian cancer [67] |
| Hierarchical Clustering | k-Means Clustering |
|---|---|
| Can’t handle large miRNA expression data—quadratic complexity | Can handle large miRNA expression datasets—linear complexity |
| Reproducible as every miRNA expressed is assigned a cluster, and the clustering occurs based on the closeness of previously generated clusters. | Unreproducible clustering due to the prerequisite of a random number of clusters. |
| Produces more intuitive results in the form of a dendrogram. | Produces less intuitive results if data does not group into hyper spherical clusters. |
| Poor performance and higher time of execution as the number of generated clusters increases. | Higher time of execution associated with large miRNA expression datasets. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Staicu, C.E.; Predescu, D.-V.; Rusu, C.M.; Radu, B.M.; Cretoiu, D.; Suciu, N.; Crețoiu, S.M.; Voinea, S.-C. Role of microRNAs as Clinical Cancer Biomarkers for Ovarian Cancer: A Short Overview. Cells 2020, 9, 169. https://doi.org/10.3390/cells9010169
Staicu CE, Predescu D-V, Rusu CM, Radu BM, Cretoiu D, Suciu N, Crețoiu SM, Voinea S-C. Role of microRNAs as Clinical Cancer Biomarkers for Ovarian Cancer: A Short Overview. Cells. 2020; 9(1):169. https://doi.org/10.3390/cells9010169
Chicago/Turabian StyleStaicu, Cristina Elena, Dragoș-Valentin Predescu, Călin Mircea Rusu, Beatrice Mihaela Radu, Dragos Cretoiu, Nicolae Suciu, Sanda Maria Crețoiu, and Silviu-Cristian Voinea. 2020. "Role of microRNAs as Clinical Cancer Biomarkers for Ovarian Cancer: A Short Overview" Cells 9, no. 1: 169. https://doi.org/10.3390/cells9010169
APA StyleStaicu, C. E., Predescu, D.-V., Rusu, C. M., Radu, B. M., Cretoiu, D., Suciu, N., Crețoiu, S. M., & Voinea, S.-C. (2020). Role of microRNAs as Clinical Cancer Biomarkers for Ovarian Cancer: A Short Overview. Cells, 9(1), 169. https://doi.org/10.3390/cells9010169








