High Glucose Concentrations Negatively Regulate the IGF1R/Src/ERK Axis through the MicroRNA-9 in Colorectal Cancer
Abstract
1. Introduction
2. Materials and Methods
2.1. Cell Culture
2.2. Drug
2.3. Transient Transfection
2.4. Western Blotting
2.5. RNA Extraction and qRT-PCR Using Vector Constructs and Transfection
2.6. Cell Cycle Analysis
2.7. Cell Migration and Invasion Assays
2.8. Wound Healing
2.9. Colorectal Tissue Preparation
2.10. Statistical Analysis
3. Results
3.1. d-glucose Promoted Cell Proliferation and Increased Cell-Cycle-Regulated Protein Expression in CRC Cells
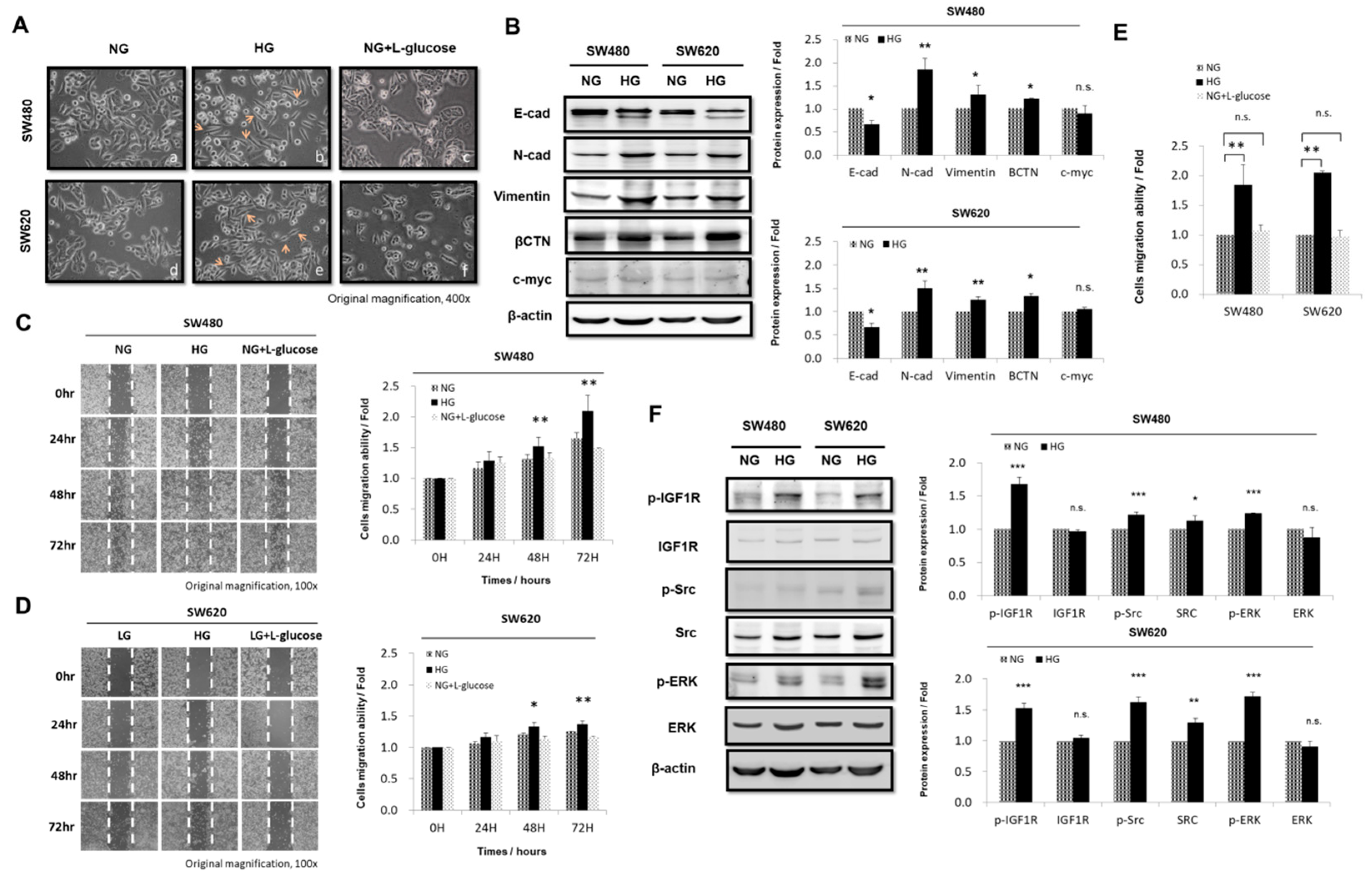
3.2. HG Concentration Induced Epithelial-to-Mesenchymal Transition Protein Expression and Enhanced Migration Activity in CRC Cells
3.3. HG Concentration Regulated IGF1R and Src and Promoted Downstream Signaling Pathways in CRC Cells
3.4. Expression and Regulation of miR-9 in CRC Cell Lines by HG Concentration
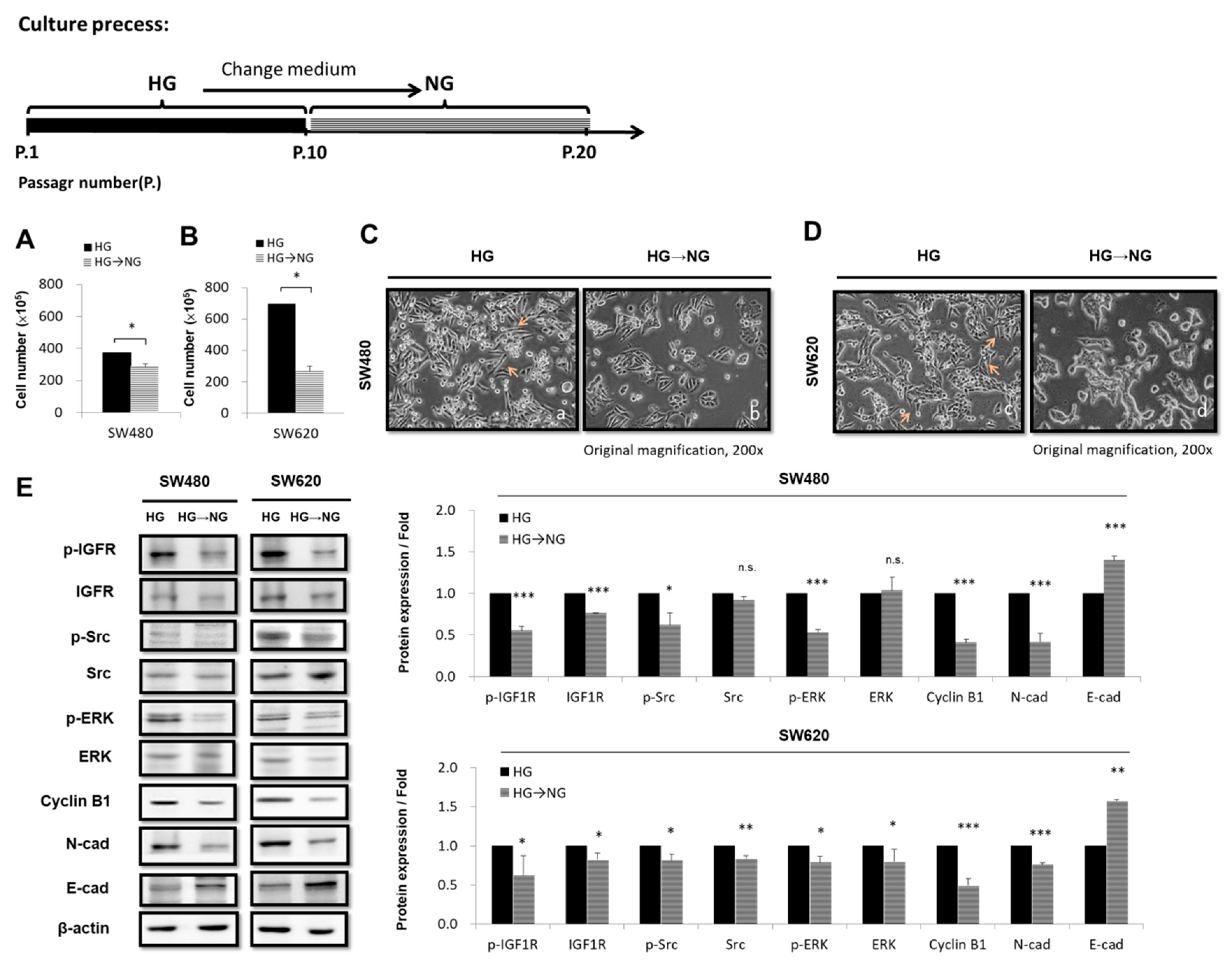
3.5. Transferring CRC Cell Lines from HG-Concentration Medium to NG-Concentration Medium Affects Cell Proliferation and Morphology
3.6. E-cadherin Is a Regulator Targeting miR-9 that Was Negatively Correlated with Carcinoembryonic Antigen CEA in Patients with CRC and Hyperglycemia
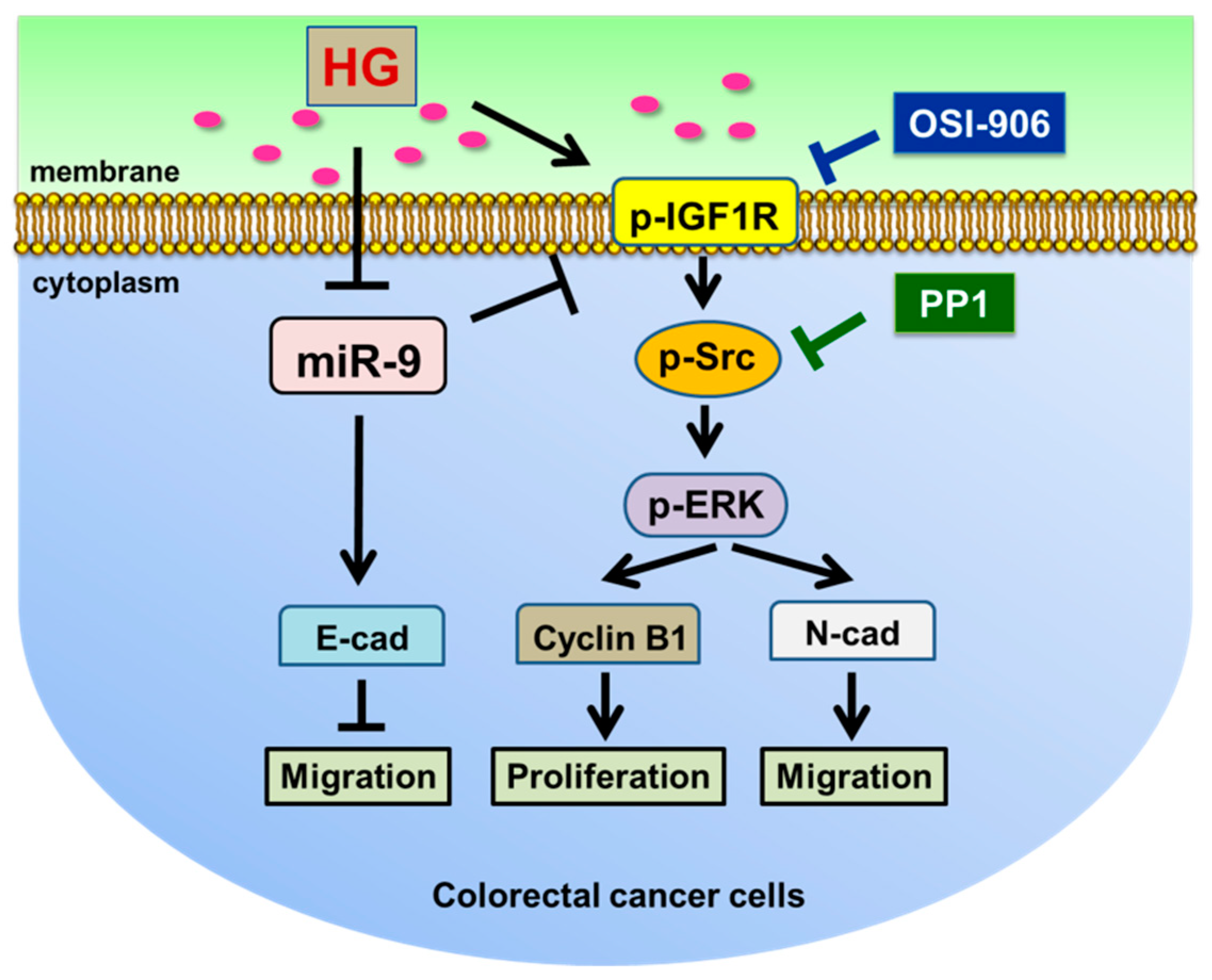
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Siegel, R.L.; Miller, K.D.; Fedewa, S.A.; Ahnen, D.J.; Meester, R.G.S.; Barzi, A.; Jemal, A. Colorectal cancer statistics, 2017. CA Cancer J. Clin. 2017, 67, 177–193. [Google Scholar] [CrossRef]
- Lin, C.Y.; Lee, C.H.; Huang, C.C.; Lee, S.T.; Guo, H.R.; Su, S.B. Impact of high glucose on metastasis of colon cancer cells. World J. Gastroenterol. 2015, 21, 2047–2057. [Google Scholar] [CrossRef]
- Lee, M.Y.; Lin, K.D.; Hsiao, P.J.; Shin, S.J. Thiamine corrects delayed replication and decreases production of lactate and advanced glycation end-products in bovine retinal and human umbilical vein endothelial cells cultured under high glucose conditions. Metabolism 2012, 61, 242–249. [Google Scholar] [CrossRef] [PubMed]
- Zhu, B.; Wu, X.; Wu, B.; Pei, D.; Zhang, L.; Wei, L. The relationship between diabetes and colorectal cancer prognosis: A meta-analysis based on the cohort studies. PLoS ONE 2017, 12, e0176068. [Google Scholar] [CrossRef] [PubMed]
- Duan, W.; Shen, X.; Lei, J.; Xu, Q.; Yu, Y.; Li, R.; Wu, E.; Ma, Q. Hyperglycemia, a neglected factor during cancer progression. Biomed. Res. Int 2014, 2014, 461917. [Google Scholar] [CrossRef]
- Vulcan, A.; Manjer, J.; Ohlsson, B. High blood glucose levels are associated with higher risk of colon cancer in men: A cohort study. BMC Cancer 2017, 17, 842. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Ma, Q.; Li, J. High glucose promotes cell proliferation and enhances gdnf and ret expression in pancreatic cancer cells. Mol. Cell. Biochem. 2011, 347, 95–101. [Google Scholar] [CrossRef] [PubMed]
- Hou, Y.; Zhou, M.; Xie, J.; Chao, P.; Feng, Q.; Wu, J. High glucose levels promote the proliferation of breast cancer cells through gtpases. Breast Cancer (Dove Medical Press) 2017, 9, 429–436. [Google Scholar] [CrossRef]
- Ding, C.Z.; Guo, X.F.; Wang, G.L.; Wang, H.T.; Xu, G.H.; Liu, Y.Y.; Wu, Z.J.; Chen, Y.H.; Wang, J.; Wang, W.G. High glucose contributes to the proliferation and migration of non-small cell lung cancer cells via GAS5-TRIB3 axis. Biosci. Rep. 2018, 38, BSR20171014. [Google Scholar] [CrossRef]
- Cheng, C.J.; Bahal, R.; Babar, I.A.; Pincus, Z.; Barrera, F.; Liu, C.; Svoronos, A.; Braddock, D.T.; Glazer, P.M.; Engelman, D.M.; et al. MicroRNA silencing for cancer therapy targeted to the tumour microenvironment. Nature 2015, 518, 107–110. [Google Scholar] [CrossRef] [PubMed]
- Tan, W.; Liu, B.; Qu, S.; Liang, G.; Luo, W.; Gong, C. MicroRNAs and cancer: Key paradigms in molecular therapy. Oncol. Lett. 2018, 15, 2735–2742. [Google Scholar] [CrossRef]
- Gorospe, M.; Abdelmohsen, K. Microregulators come of age in senescence. Trends Genet. 2011, 27, 233–241. [Google Scholar] [CrossRef]
- Lujambio, A.; Lowe, S.W. The microcosmos of cancer. Nature 2012, 482, 347–355. [Google Scholar] [CrossRef] [PubMed]
- Hamam, R.; Hamam, D.; Alsaleh, K.A.; Kassem, M.; Zaher, W.; Alfayez, M.; Aldahmash, A.; Alajez, N.M. Circulating microRNAs in breast cancer: Novel diagnostic and prognostic biomarkers. Cell Death Dis. 2017, 8, e3045. [Google Scholar] [CrossRef] [PubMed]
- Selcuklu, S.D.; Donoghue, M.T.; Rehmet, K.; de Souza Gomes, M.; Fort, A.; Kovvuru, P.; Muniyappa, M.K.; Kerin, M.J.; Enright, A.J.; Spillane, C. MicroRNA-9 inhibition of cell proliferation and identification of novel miR-9 targets by transcriptome profiling in breast cancer cells. J. Biol. Chem. 2012, 287, 29516–29528. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.; Lee, E.; Jung, J.; Lee, J.W.; Kim, H.J.; Kim, J.; Yoo, H.J.; Lee, H.J.; Chae, S.Y.; Jeon, S.M.; et al. MicroRNA-155 positively regulates glucose metabolism via PIK3R1-FOXO3a-cMYC axis in breast cancer. Oncogene 2018, 37, 2982–2991. [Google Scholar] [CrossRef] [PubMed]
- Zaleski, M.; Kobilay, M.; Schroeder, L.; Debald, M.; Semaan, A.; Hettwer, K.; Uhlig, S.; Kuhn, W.; Hartmann, G.; Holdenrieder, S. Improved sensitivity for detection of breast cancer by combination of miR-34a and tumor markers CA 15-3 or CEA. Oncotarget 2018, 9, 22523–22536. [Google Scholar] [CrossRef]
- Zhu, M.; Xu, Y.; Ge, M.; Gui, Z.; Yan, F. Regulation of uhrf1 by microRNA-9 modulates colorectal cancer cell proliferation and apoptosis. Cancer Sci. 2015, 106, 833–839. [Google Scholar] [CrossRef] [PubMed]
- Cekaite, L.; Rantala, J.K.; Bruun, J.; Guriby, M.; Agesen, T.H.; Danielsen, S.A.; Lind, G.E.; Nesbakken, A.; Kallioniemi, O.; Lothe, R.A.; et al. MiR-9, -31, and -182 deregulation promote proliferation and tumor cell survival in colon cancer. Neoplasia 2012, 14, 868–879. [Google Scholar] [CrossRef]
- Bandres, E.; Agirre, X.; Bitarte, N.; Ramirez, N.; Zarate, R.; Roman-Gomez, J.; Prosper, F.; Garcia-Foncillas, J. Epigenetic regulation of microRNA expression in colorectal cancer. Int. J. Cancer 2009, 125, 2737–2743. [Google Scholar] [CrossRef]
- Roberts, D.L.; Dive, C.; Renehan, A.G. Biological mechanisms linking obesity and cancer risk: New perspectives. Annu. Rev. Med. 2010, 61, 301–316. [Google Scholar] [CrossRef]
- Handelsman, Y.; Leroith, D.; Bloomgarden, Z.T.; Dagogo-Jack, S.; Einhorn, D.; Garber, A.J.; Grunberger, G.; Harrell, R.M.; Gagel, R.F.; Lebovitz, H.E.; et al. Diabetes and cancer—An aace/ace consensus statement. Endocr. Pract. 2013, 19, 675–693. [Google Scholar] [CrossRef]
- Gristina, V.; Cupri, M.G.; Torchio, M.; Mezzogori, C.; Cacciabue, L.; Danova, M. Diabetes and cancer: A critical appraisal of the pathogenetic and therapeutic links. Biomed. Rep. 2015, 3, 131–136. [Google Scholar] [CrossRef]
- Zha, J.; Lackner, M.R. Targeting the insulin-like growth factor receptor-1R pathway for cancer therapy. Clin. Cancer Res. 2010, 16, 2512–2517. [Google Scholar] [CrossRef]
- Lieu, C.; Kopetz, S. The src family of protein tyrosine kinases: A new and promising target for colorectal cancer therapy. Clin. Colorectal Cancer 2010, 9, 89–94. [Google Scholar] [CrossRef]
- Chen, J.; Elfiky, A.; Han, M.; Chen, C.; Saif, M.W. The role of src in colon cancer and its therapeutic implications. Clin. Colorectal Cancer 2014, 13, 5–13. [Google Scholar] [CrossRef]
- Leontieva, O.V.; Demidenko, Z.N.; Blagosklonny, M.V. Rapamycin reverses insulin resistance (IR) in high-glucose medium without causing IR in normoglycemic medium. Cell Death Dis. 2014, 5, e1214. [Google Scholar] [CrossRef][Green Version]
- Han, J.; Zhang, L.; Guo, H.; Wysham, W.Z.; Roque, D.R.; Willson, A.K.; Sheng, X.; Zhou, C.; Bae-Jump, V.L. Glucose promotes cell proliferation, glucose uptake and invasion in endometrial cancer cells via AMPK/mTOR/S6 and MAPK signaling. Gynecol. Oncol. 2015, 138, 668–675. [Google Scholar] [CrossRef]
- Han, L.; Ma, Q.; Li, J.; Liu, H.; Li, W.; Ma, G.; Xu, Q.; Zhou, S.; Wu, E. High glucose promotes pancreatic cancer cell proliferation via the induction of EGF expression and transactivation of EGFR. PLoS ONE 2011, 6, e27074. [Google Scholar] [CrossRef]
- Sikander, M.; Malik, S.; Chauhan, N.; Khan, P.; Kumari, S.; Kashyap, V.K.; Khan, S.; Ganju, A.; Halaweish, F.T.; Yallapu, M.M.; et al. Cucurbitacin d reprograms glucose metabolic network in prostate cancer. Cancers 2019, 11, 364. [Google Scholar] [CrossRef]
- Shen, J.; Liu, M.; Xu, J.; Sun, B.; Xu, H.; Zhang, W. Arl15 overexpression attenuates high glucose-induced impairment of insulin signaling and oxidative stress in human umbilical vein endothelial cells. Life Sci. 2019, 220, 127–135. [Google Scholar] [CrossRef]
- Chen, S.; Ma, J.; Zhu, H.; Deng, S.; Gu, M.; Qu, S. Hydroxysafflor yellow a attenuates high glucose-induced human umbilical vein endothelial cell dysfunction. Hum. Exp. Toxicol. 2019, 960327119831065. [Google Scholar] [CrossRef]
- Hay, N. Reprogramming glucose metabolism in cancer: Can it be exploited for cancer therapy? Nat. Rev. Cancer 2016, 16, 635–649. [Google Scholar] [CrossRef]
- Saengboonmee, C.; Seubwai, W.; Pairojkul, C.; Wongkham, S. High glucose enhances progression of cholangiocarcinoma cells via STAT3 activation. Sci. Rep. 2016, 6, 18995. [Google Scholar] [CrossRef]
- Wallbillich, J.J.; Josyula, S.; Saini, U.; Zingarelli, R.A.; Dorayappan, K.D.; Riley, M.K.; Wanner, R.A.; Cohn, D.E.; Selvendiran, K. High glucose-mediated STAT3 activation in endometrial cancer is inhibited by metformin: Therapeutic implications for endometrial cancer. PLoS ONE 2017, 12, e0170318. [Google Scholar] [CrossRef]
- Zheng, J. Energy metabolism of cancer: Glycolysis versus oxidative phosphorylation (review). Oncol. Lett. 2012, 4, 1151–1157. [Google Scholar] [CrossRef]
- Ganapathy-Kanniappan, S.; Geschwind, J.F. Tumor glycolysis as a target for cancer therapy: Progress and prospects. Mol. Cancer 2013, 12, 152. [Google Scholar] [CrossRef]
- Phoomak, C.; Vaeteewoottacharn, K.; Silsirivanit, A.; Saengboonmee, C.; Seubwai, W.; Sawanyawisuth, K.; Wongkham, C.; Wongkham, S. High glucose levels boost the aggressiveness of highly metastatic cholangiocarcinoma cells via o-glcnacylation. Sci. Rep. 2017, 7, 43842. [Google Scholar] [CrossRef]
- Petrova, V.; Annicchiarico-Petruzzelli, M.; Melino, G.; Amelio, I. The hypoxic tumour microenvironment. Oncogenesis 2018, 7, 10. [Google Scholar] [CrossRef]
- Masur, K.; Vetter, C.; Hinz, A.; Tomas, N.; Henrich, H.; Niggemann, B.; Zanker, K.S. Diabetogenic glucose and insulin concentrations modulate transcriptome and protein levels involved in tumour cell migration, adhesion and proliferation. Br. J. Cancer 2011, 104, 345–352. [Google Scholar] [CrossRef]
- Ma, Y.S.; Yang, I.P.; Tsai, H.L.; Huang, C.W.; Juo, S.H.; Wang, J.Y. High glucose modulates antiproliferative effect and cytotoxicity of 5-fluorouracil in human colon cancer cells. DNA Cell Biol. 2014, 33, 64–72. [Google Scholar] [CrossRef]
- Chae, S.W.; Sohn, J.H.; Kim, D.H.; Choi, Y.J.; Park, Y.L.; Kim, K.; Cho, Y.H.; Pyo, J.S.; Kim, J.H. Overexpressions of Cyclin B1, cdc2, p16 and p53 in human breast cancer: The clinicopathologic correlations and prognostic implications. Yonsei Med. J. 2011, 52, 445–453. [Google Scholar] [CrossRef]
- Shen, M.; Feng, Y.; Gao, C.; Tao, D.; Hu, J.; Reed, E.; Li, Q.Q.; Gong, J. Detection of Cyclin B1 expression in G(1)-phase cancer cell lines and cancer tissues by postsorting western blot analysis. Cancer Res. 2004, 64, 1607–1610. [Google Scholar] [CrossRef]
- Dai, C.Y.; Furth, E.E.; Mick, R.; Koh, J.; Takayama, T.; Niitsu, Y.; Enders, G.H. p16(INK4a) expression begins early in human colon neoplasia and correlates inversely with markers of cell proliferation. Gastroenterology 2000, 119, 929–942. [Google Scholar] [CrossRef]
- Zhou, N.; Gu, Q. Prognostic and clinicopathological value of p16 protein aberrant expression in colorectal cancer: A prisma-compliant meta-analysis. Medicine (Baltimore) 2018, 97, e0195. [Google Scholar] [CrossRef]
- Zhang, Y.Y.; Guo, Q.Y.; Wu, M.Y.; Zang, C.S.; Ma, F.Z.; Sun, T.; Wang, W.N.; Miao, L.N.; Xu, Z.G. p16INK4a expression is increased through 12-lipoxygenase in high glucose-stimulated glomerular mesangial cells and type 2 diabetic glomeruli. Nephron 2015, 130, 141–150. [Google Scholar] [CrossRef]
- Dongre, A.; Weinberg, R.A. New insights into the mechanisms of epithelial–mesenchymal transition and implications for cancer. Nat. Rev. Mol. Cell Biol. 2019, 20, 69–84. [Google Scholar] [CrossRef]
- Clemmons, D.R. Metabolic actions of insulin-like growth factor-i in normal physiology and diabetes. Endocrinol. Metab. Clin. North. Am. 2012, 41, 425–443. [Google Scholar] [CrossRef]
- Zinn, R.L.; Gardner, E.E.; Marchionni, L.; Murphy, S.C.; Dobromilskaya, I.; Hann, C.L.; Rudin, C.M. Erk phosphorylation is predictive of resistance to igf-1r inhibition in small cell lung cancer. Mol. Cancer Ther. 2013, 12, 1131–1139. [Google Scholar] [CrossRef]
- Salisbury, T.B.; Tomblin, J.K. Insulin/insulin-like growth factors in cancer: New roles for the aryl hydrocarbon receptor, tumor resistance mechanisms, and new blocking strategies. Front. Endocrinol. (Lausanne) 2015, 6, 12. [Google Scholar] [CrossRef][Green Version]
- Flanigan, S.A.; Pitts, T.M.; Newton, T.P.; Kulikowski, G.N.; Tan, A.C.; McManus, M.C.; Spreafico, A.; Kachaeva, M.I.; Selby, H.M.; Tentler, J.J.; et al. Overcoming IGF1R/IR resistance through inhibition of MEK signaling in colorectal cancer models. Clin. Cancer Res. 2013, 19, 6219–6229. [Google Scholar] [CrossRef]
- Hurwitz, H.; Fehrenbacher, L.; Novotny, W.; Cartwright, T.; Hainsworth, J.; Heim, W.; Berlin, J.; Baron, A.; Griffing, S.; Holmgren, E.; et al. Bevacizumab plus irinotecan, fluorouracil, and leucovorin for metastatic colorectal cancer. N. Engl. J. Med. 2004, 350, 2335–2342. [Google Scholar] [CrossRef]
- Bartscht, T.; Lehnert, H.; Gieseler, F.; Ungefroren, H. The src family kinase inhibitors PP2 and PP1 effectively block TGF-beta1-induced cell migration and invasion in both established and primary carcinoma cells. Cancer Chemother. Pharmacol. 2012, 70, 221–230. [Google Scholar] [CrossRef]
- Han, L.; Peng, B.; Ma, Q.; Ma, J.; Li, J.; Li, W.; Duan, W.; Chen, C.; Liu, J.; Xu, Q.; et al. Indometacin ameliorates high glucose-induced proliferation and invasion via modulation of e-cadherin in pancreatic cancer cells. Curr. Med. Chem. 2013, 20, 4142–4152. [Google Scholar] [CrossRef]
- Wang, L.; Li, J.J.; Guo, L.Y.; Li, P.; Zhao, Z.; Zhou, H.; Di, L.J. Molecular link between glucose and glutamine consumption in cancer cells mediated by CtBP and SIRT4. Oncogenesis 2018, 7, 26. [Google Scholar] [CrossRef]
- Oberg, A.L.; French, A.J.; Sarver, A.L.; Subramanian, S.; Morlan, B.W.; Riska, S.M.; Borralho, P.M.; Cunningham, J.M.; Boardman, L.A.; Wang, L.; et al. Mirna expression in colon polyps provides evidence for a multihit model of colon cancer. PLoS ONE 2011, 6, e20465. [Google Scholar] [CrossRef]
- Ramachandran, D.; Roy, U.; Garg, S.; Ghosh, S.; Pathak, S.; Kolthur-Seetharam, U. Sirt1 and miR-9 expression is regulated during glucose-stimulated insulin secretion in pancreatic beta-islets. FEBS J. 2011, 278, 1167–1174. [Google Scholar] [CrossRef]
- Hu, D.; Wang, Y.; Zhang, H.; Kong, D. Identification of miR-9 as a negative factor of insulin secretion from beta cells. J. Physiol. Biochem. 2018, 74, 291–299. [Google Scholar] [CrossRef]
- Suh, Y.S.; Bhat, S.; Hong, S.H.; Shin, M.; Bahk, S.; Cho, K.S.; Kim, S.W.; Lee, K.S.; Kim, Y.J.; Jones, W.D.; et al. Genome-wide microRNA screening reveals that the evolutionary conserved miR-9a regulates body growth by targeting sNPFR1/NPYR. Nat. Commun. 2015, 6, 7693. [Google Scholar] [CrossRef]
- Park, Y.R.; Lee, S.T.; Kim, S.L.; Liu, Y.C.; Lee, M.R.; Shin, J.H.; Seo, S.Y.; Kim, S.H.; Kim, I.H.; Lee, S.O.; et al. MicroRNA-9 suppresses cell migration and invasion through downregulation of TM4SF1 in colorectal cancer. Int. J. Oncol. 2016, 48, 2135–2143. [Google Scholar] [CrossRef]
- Guo, F.; Hou, X.; Sun, Q. MicroRNA-9-5p functions as a tumor suppressor in papillary thyroid cancer via targeting braf. Oncol. Lett. 2018, 16, 6815–6821. [Google Scholar] [CrossRef]
- Liu, S.; Kumar, S.M.; Lu, H.; Liu, A.; Yang, R.; Pushparajan, A.; Guo, W.; Xu, X. MicroRNA-9 up-regulates e-cadherin through inhibition of NF-kappaB1-snail1 pathway in melanoma. J. Pathol. 2012, 226, 61–72. [Google Scholar] [CrossRef]
- Duffy, M.J. Carcinoembryonic antigen as a marker for colorectal cancer: Is it clinically useful? Clin. Chem. 2001, 47, 624–630. [Google Scholar]
- McNally, G.E.; Lloyd, D.M.; Grondona, J.P. Carcinoembryonic antigen as a prognostic factor in colorectal cancer with liver metastases. JCT 2015, 6, 1035–1044. [Google Scholar] [CrossRef][Green Version]




© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, Y.-C.; Ou, M.-C.; Fang, C.-W.; Lee, T.-H.; Tzeng, S.-L. High Glucose Concentrations Negatively Regulate the IGF1R/Src/ERK Axis through the MicroRNA-9 in Colorectal Cancer. Cells 2019, 8, 326. https://doi.org/10.3390/cells8040326
Chen Y-C, Ou M-C, Fang C-W, Lee T-H, Tzeng S-L. High Glucose Concentrations Negatively Regulate the IGF1R/Src/ERK Axis through the MicroRNA-9 in Colorectal Cancer. Cells. 2019; 8(4):326. https://doi.org/10.3390/cells8040326
Chicago/Turabian StyleChen, Ya-Chun, Ming-Che Ou, Chia-Wei Fang, Tsung-Hsien Lee, and Shu-Ling Tzeng. 2019. "High Glucose Concentrations Negatively Regulate the IGF1R/Src/ERK Axis through the MicroRNA-9 in Colorectal Cancer" Cells 8, no. 4: 326. https://doi.org/10.3390/cells8040326
APA StyleChen, Y.-C., Ou, M.-C., Fang, C.-W., Lee, T.-H., & Tzeng, S.-L. (2019). High Glucose Concentrations Negatively Regulate the IGF1R/Src/ERK Axis through the MicroRNA-9 in Colorectal Cancer. Cells, 8(4), 326. https://doi.org/10.3390/cells8040326





