Ginsenoside Rb1 as an Anti-Diabetic Agent and Its Underlying Mechanism Analysis
Abstract
1. Introduction
2. Source and Chemistry
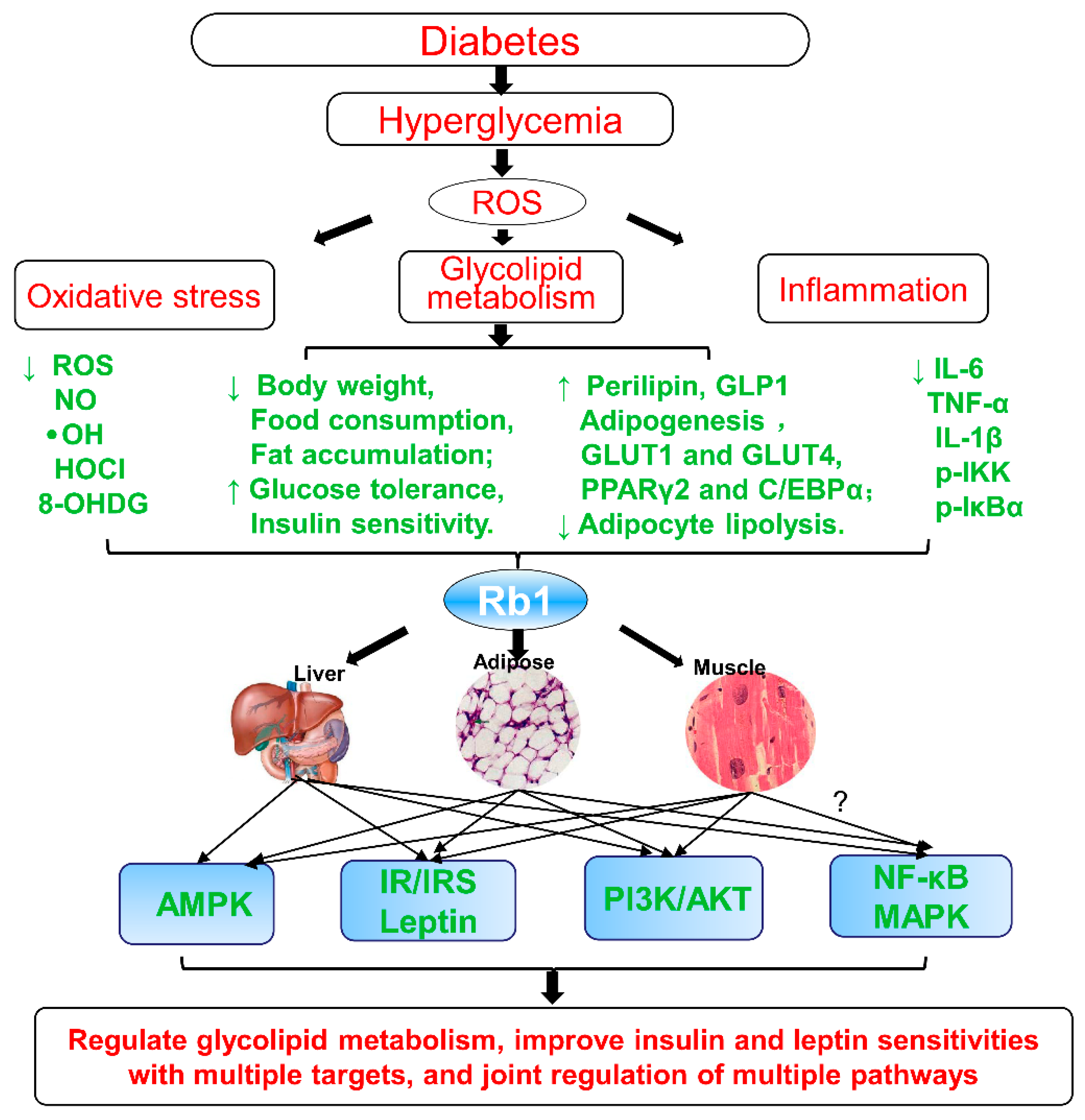
3. Anti-Diabetic Effects and Mechanisms of Ginsenoside Rb1
3.1. In Vivo
3.2. In Vitro
- Inhibit lipolysis and reduce free fats release and ectopic triglycerides deposition;
- Reduce hepatic fat accumulation and suppress adipocyte lipolysis;
- Decrease inflammatory stress in adipose and liver tissues;
- Promote adipogenesis and the 3T3-L1 adipocytes differentiation;
- Upregulate PPARγ and enhance GLUTs expression and translocation of GLUT1/ GLUT4;
- Upregulate perilipin in adipocytes and in fat cells;
- Decrease 11β-HSD1 and its mRNA in liver and adipose tissue;
- Decrease the expression of inflammatory markers (IL-6, IL-1β, and p-IKK);
- Negatively regulate leptin signaling (SOCS3 and PTP1B);
- Inhibit HG-induced caspase-3 activation and apoptosis;
- Increase adiponectin receptor gene expression, upregulate perilipin adipocytes, upregulate PPARγ2, C/EBPα, and activate the adiponectin signaling pathway;
- Regulate PI3K and Akt activity;
- Activate AMPK, regulate energy metabolism, and promote glucose uptake, at least partially, by activating the insulin signaling pathway;
- Regulate NF-κB pathway (p-IKK and p-IκBα) and leptin–STAT3 signaling.
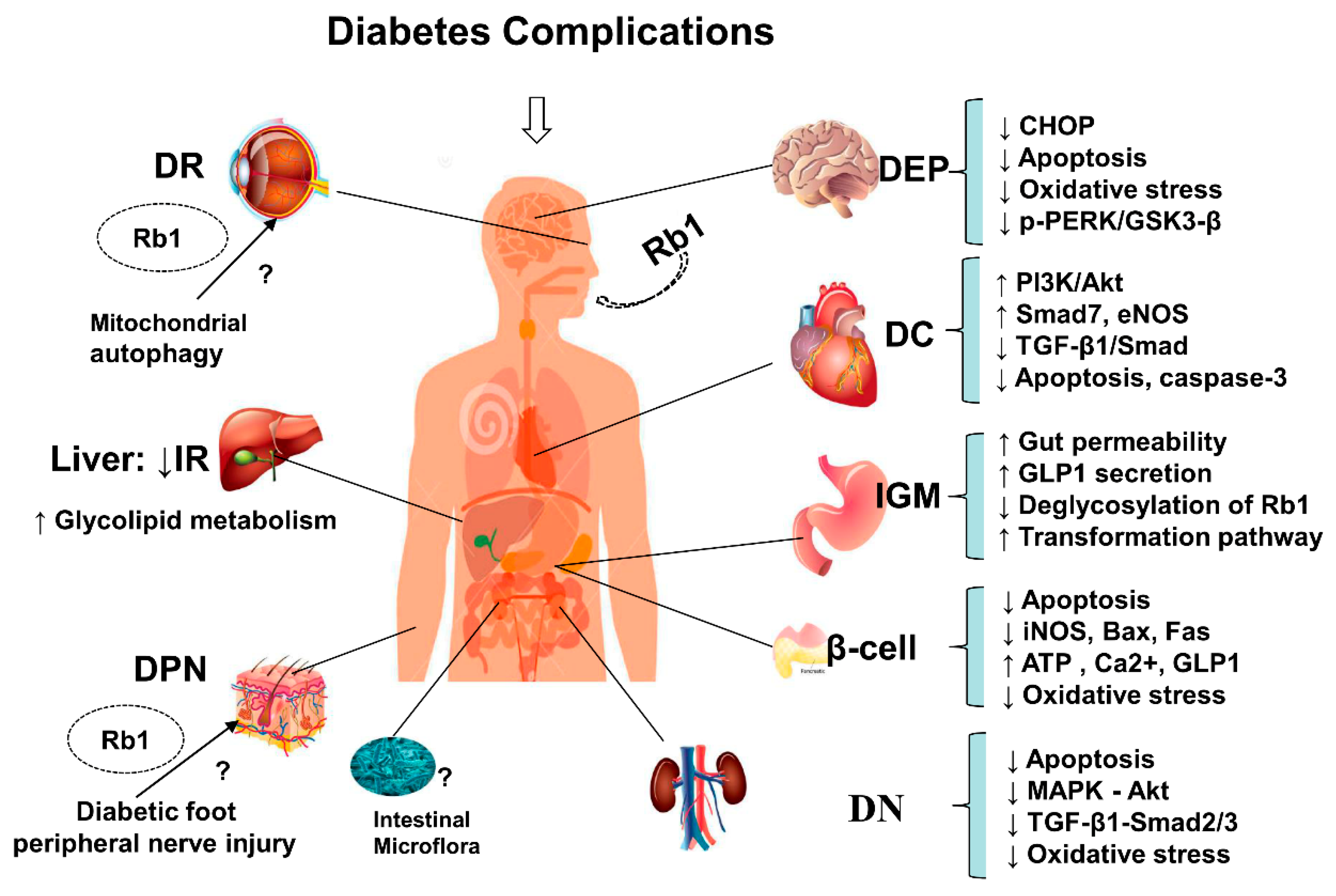
4. Overview of Anti-Diabetic Complications of Ginsenoside Rb1
4.1. Protective Effects on the Islets
4.2. Protective Effects on Diabetic Nephropathy
4.3. Protective Effects on Diabetic Encephalopathy
4.4. Protective Effects on Diabetic Cardiovascular Complications
4.5. Intestine and Gut Microbiota Effects
- Rb1 protects neurons from HG-induced cell damage and exerts neuroprotective effects against oxidative damage and apoptosis by inhibiting GSK-3β-mediated CHOP induction and reducing ROS production.
- Rb1 has protective effects against high glucose podocyte injury, kidney damage, and DN by inhibiting oxidative stress. It also decreases ROS overproduction, protects against mitochondrial dysfunction, restores cellular redox enzymes imbalances, and reduces fibronectin expression under diabetic conditions by regulating p44/4, p38-MAPK, JNK/SAPK, and Akt phosphorylation levels, reducing TGF-β1 protein expression, and blocking the TGF-β1-Smad2/3 signaling pathway.
- Rb1 inhibits fibrosis and improves cardiac function by suppressing TGF-β1/Smad signaling. It also exerts cardioprotective effects on both myocardial ischemia/reperfusion under diabetic conditions and diabetic damage from myocardial ischemia/reperfusion. Rb1 attenuates myocardial ischemia/reperfusion injury and reduces infarct size, cardiomyocyte apoptosis, and caspase-3 activity, partly by inhibiting oxidative stress and apoptosis, enhancing eNOS expression, and increasing NO concentrations, as well as activating the PI3K/Akt pathway.
5. Conclusion and Recommendation
Funding
Conflicts of Interest
References
- Muoio, D.M.; Newgard, C.B. Mechanisms of disease: Molecular and metabolic mechanisms of insulin resistance and beta-cell failure in type 2 diabetes. Nat. Rev. Mol. Cell Biol. 2008, 9, 193–205. [Google Scholar] [CrossRef] [PubMed]
- Kahn, S.E.; Hull, R.L.; Utzschneider, K.M. Mechanisms linking obesity to insulin resistance and type 2 diabetes. Nature 2006, 444, 840. [Google Scholar] [CrossRef] [PubMed]
- Venables, M.C.; Jeukendrup, A.E. Physical inactivity and obesity: Links with insulin resistance and type 2 diabetes mellitus. Diabetes Metab. Res. Rev. 2010, 25, S18–S23. [Google Scholar] [CrossRef] [PubMed]
- Romero-Aroca, P.; Mendez-Marin, I.; Baget-Bernaldiz, M.; Fernéndez-Ballart, J.; Santos-Blanco, E. Review of the relationship between renal and retinal microangiopathy in diabetes mellitus patients. Curr. Diabetes Rev. 2010, 6, 88–101. [Google Scholar] [CrossRef] [PubMed]
- Ferrannini, E.; Natali, A.; Camastra, S.; Nannipieri, M.; Mari, A.; Adam, K.P.; Milburn, M.V.; Kastenmüller, G.; Adamski, J.; Tuomi, T. Early metabolic markers of the development of dysglycemia and type 2 diabetes and their physiological significance. Diabetes 2013, 62, 1730–1737. [Google Scholar] [CrossRef] [PubMed]
- Brownlee, M. Biochemistry and molecular cell biology of diabetic complications. Nature 2001, 414, 813–820. [Google Scholar] [CrossRef] [PubMed]
- Setter, S.M.; Iltz, J.L.; Thams, J.; Campbell, R.K. Metformin hydrochloride in the treatment of type 2 diabetes mellitus: A clinical review with a focus on dual therapy. Clin. Ther. 2003, 25, 2991–3026. [Google Scholar] [CrossRef]
- Nanjan, M.J.; Mohammed, M.; Prashantha Kumar, B.R.; Chandrasekar, M.J.N. Thiazolidinediones as antidiabetic agents: A critical review. Bioorganic Chem. 2018, 77, 548–567. [Google Scholar] [CrossRef] [PubMed]
- Oikonomou, E.; Mourouzis, K.; Fountoulakis, P.; Papamikroulis, G.A.; Siasos, G.; Antonopoulos, A.; Vogiatzi, G.; Tsalamadris, S.; Vavuranakis, M.; Tousoulis, D. Interrelationship between diabetes mellitus and heart failure: The role of peroxisome proliferator-activated receptors in left ventricle performance. Heart Fail. Rev. 2018, 23, 389–408. [Google Scholar] [CrossRef] [PubMed]
- Thangavel, N.; Al Bratty, M.; Akhtar Javed, S.; Ahsan, W.; Alhazmi, H.A. Targeting peroxisome proliferator-activated receptors using thiazolidinediones: Strategy for design of novel antidiabetic drugs. Int. J. Med. Chem. 2017, 2017, 1069718. [Google Scholar] [CrossRef] [PubMed]
- Glosse, P.; Foller, M. AMP-Activated protein kinase (AMPK)-dependent regulation of renal transport. Int. J. Mol. Sci. 2018, 19, 3481. [Google Scholar] [CrossRef] [PubMed]
- Rozanska, D.; Regulska-Ilow, B. The significance of anthocyanins in the prevention and treatment of type 2 diabetes. Adv. Clin. Exp. Med. 2018, 27, 135–142. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Liu, S.; Zhai, A.; Zhang, B.; Tian, G. AMPK-mediated regulation of lipid metabolism by phosphorylation. Biol. Pharm. Bull. 2018, 41, 985–993. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Galiano, D.; Borges, B.C.; Allen, S.J.; Elias, C.F. PI3K signaling in leptin receptor cells: Role in growth and reproduction. J. Neuroendocrinol. 2019. [Google Scholar] [CrossRef] [PubMed]
- John, C.M.; Mohamed Yusof, N.I.S.; Abdul Aziz, S.H.; Mohd Fauzi, F. Maternal cognitive impairment associated with gestational diabetes mellitus—A review of potential contributing mechanisms. Int. J. Mol. Sci. 2018, 19, 3894. [Google Scholar] [CrossRef] [PubMed]
- Mabhida, S.E.; Dludla, P.V.; Johnson, R.; Ndlovu, M.; Louw, J.; Opoku, A.R.; Mosa, R.A. Protective effect of triterpenes against diabetes-induced beta-cell damage: An overview of in vitro and in vivo studies. Pharm. Res 2018, 137, 179–192. [Google Scholar] [CrossRef] [PubMed]
- Ferguson, R.D.; Gallagher, E.J.; Scheinman, E.J.; Damouni, R.; LeRoith, D. The Epidemiology and Molecular Mechanisms Linking Obesity, Diabetes, and Cancer. Vitam. Horm. 2013, 93, 51–98. [Google Scholar] [PubMed]
- Hirosumi, J.; Tuncman, G.; Chang, L.; Görgün, C.Z.; Uysal, K.T.; Maeda, K.; Karin, M.; Hotamisligil, G.S. A central role for JNK in obesity and insulin resistance. Nature 2002, 420, 333–336. [Google Scholar] [CrossRef] [PubMed]
- Sheetz, M.J.; King, G.L. Molecular understanding of hyperglycemia’s adverse effects for diabetic complications. JAMA 2002, 288, 2579–2588. [Google Scholar] [CrossRef] [PubMed]
- Hunt, J.V.; Wolff, S.P. Oxidative glycation and free radical production: A causal mechanism of diabetic complications. Free Radic. Res. Commun. 1990, 12, 115–123. [Google Scholar] [CrossRef]
- Ishii, H.; Hayashino, Y.; Akai, Y.; Yabuta, M.; Tsujii, S. Dipeptidyl peptidase—4 inhibitors as preferable oral hypoglycemic agents in terms of treatment satisfaction: Results from a multicenter, 12-week, open label, randomized controlled study in Japan (PREFERENCE 4 study). J. Diabetes Investig. 2018, 9, 137–145. [Google Scholar] [CrossRef] [PubMed]
- Choi, K.; Kim, Y.B. Molecular mechanism of insulin resistance in obesity and type 2 diabetes. Korean J. Intern. Med. 2010, 25, 119–129. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.; Huang, K.; Lei, X.G. Selenium and diabetes-evidence from animal studies. Free Radic. Biol. Med. 2013, 65, 1548–1556. [Google Scholar] [CrossRef] [PubMed]
- Weyer, C.; Funahashi, T.; Tanaka, S.; Hotta, K.; Matsuzawa, Y.; Pratley, R.E.; Tataranni, P.A. Hypoadiponectinemia in obesity and type 2 diabetes: Close association with insulin resistance and hyperinsulinemia. J. Clin. Endocrinol. Metab. 2001, 86, 1930–1935. [Google Scholar] [CrossRef] [PubMed]
- Dios, S.T.D.; Frontanilla, K.V.; Nigro, J.; Ballinger, M.L.; Ivey, M.E.; Cawson, E.A.; Little, P.J. Regulation of the atherogenic properties of vascular smooth muscle proteoglycans by oral anti-hyperglycemic agents. J. Diabetes Its Complicat. 2007, 21, 108–117. [Google Scholar] [CrossRef] [PubMed]
- Bekele, B.B. The prevalence of macro and microvascular complications of DM among patients in Ethiopia 1990-2017: Systematic review. Diabetes Metab. Syndr. 2019, 13, 672–677. [Google Scholar] [CrossRef] [PubMed]
- Feng, Y.; Fang, Y.; Wang, Y.; Hao, Y. Acupoint therapy on diabetes mellitus and its common chronic complications: A review of its mechanisms. Biomed. Res. Int. 2018, 2018, 3128378. [Google Scholar] [CrossRef] [PubMed]
- Lao, Y.; Wang, X.; Xu, N.; Zhang, H.; Xu, H. Application of proteomics to determine the mechanism of action of traditional Chinese medicine remedies. J. Ethnopharmacol. 2014, 155, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Sharma, B.R.; Gautam, L.N.; Adhikari, D.; Karki, R. A comprehensive review on chemical profiling of nelumbo nucifera: potential for drug development. Phytother. Res. 2017, 31, 3–26. [Google Scholar] [CrossRef] [PubMed]
- Sun, G.D.; Li, C.Y.; Cui, W.P.; Guo, Q.Y.; Dong, C.Q.; Zou, H.B.; Liu, S.J.; Dong, W.P.; Miao, L.N. Review of herbal traditional chinese medicine for the treatment of diabetic nephropathy. J. Diabetes Res. 2016, 2016, 5749857. [Google Scholar] [CrossRef] [PubMed]
- Zhang, T.T.; Jiang, J.G. Active ingredients of traditional Chinese medicine in the treatment of diabetes and diabetic complications. Expert Opin. Investig. Drugs 2012, 21, 1625–1642. [Google Scholar] [CrossRef] [PubMed]
- Zhou, X.; Chen, L.-L.; Xie, R.F.; Lam, W.; Zhang, Z.-J.; Jiang, Z.-L.; Cheng, Y.-C. Chemosynthesis pathway and bioactivities comparison of saponins in radix and flower of Panax notoginseng (Burk.) F.H. Chen. J. Ethnopharmacol. 2017, 201, 56–72. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.H. Pharmacological and medical applications of Panax ginseng and ginsenosides: A review for use in cardiovascular diseases. J. Ginseng Res. 2018, 42, 264–269. [Google Scholar] [CrossRef] [PubMed]
- Su, P.; Wang, L.; Du, S.J.; Xin, W.F.; Zhang, W.S. Advance in studies of Panax notoginseng saponins on pharmacological mechanism of nervous system disease. China J. Chin. Mater. Med. 2014, 39, 4516–4521. (In Chinese) [Google Scholar]
- Mancuso, C.; Santangelo, R. Panax ginseng and Panax quinquefolius: From pharmacology to toxicology. Food Chem. Toxicol. 2017, 107, 362–372. [Google Scholar] [CrossRef] [PubMed]
- Behl, T.; Kotwani, A. Chinese herbal drugs for the treatment of diabetic retinopathy. J. Pharm. Pharmacol. 2017, 69, 223–235. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.H.; Li, J.; Liu, J.; Zhao, Y.; Zhang, P.; Zhang, M.X.; Zhang, L. Saponins isolated from the root of Panax notoginseng showed significant anti-diabetic effects in KK-Ay mice. Am. J. Chin. Med. 2008, 36, 939–951. [Google Scholar] [CrossRef] [PubMed]
- Du, Y.G.; Wang, L.P.; Qian, J.W.; Zhang, K.N.; Chai, K.F. Panax notoginseng saponins protect kidney from diabetes by up-regulating silent information regulator 1 and activating antioxidant proteins in rats. Chin. J. Integr. Med. 2016, 22, 910–917. [Google Scholar] [CrossRef] [PubMed]
- Allison, D.J.; Ditor, D.S. The common inflammatory etiology of depression and cognitive impairment: A therapeutic target. J. Neuroinflamm. 2014, 11, 151. [Google Scholar] [CrossRef] [PubMed]
- Jeon, S.W.; Kim, Y.K. Neuroinflammation and cytokine abnormality in major depression: Cause or consequence in that illness? World J. Psychiatry 2016, 6, 283–293. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Ding, L.; Wang, B.; Ren, G.; Sun, A.; Deng, C.; Wei, X.; Mani, S.; Wang, Z.; Dou, W. Notoginsenoside R1 attenuates experimental inflammatory bowel disease via pregnane X receptor activation. J. Pharmacol. Exp. Ther. 2015, 352, 315–324. [Google Scholar] [CrossRef] [PubMed]
- Zheng, X.; Liang, Y.; Kang, A.; Ma, S.J.; Xing, L.; Zhou, Y.Y.; Dai, C.; Xie, H.; Xie, L.; Wang, G.J.; et al. Peripheral immunomodulation with ginsenoside Rg1 ameliorates neuroinflammation-induced behavioral deficits in rats. Neuroscience 2014, 256, 210–222. [Google Scholar] [CrossRef] [PubMed]
- Xie, T.; Zhou, X.P.; Lin, L.L.; Xu, J.Y.; Shen, C.S.; Feng, Z.; Zhou, L.L.; Shan, J.J. Metabolomics analysis of Tripterygium wilfordii formulation based on theory of detoxicity compatibility. China J. Chin. Mater. Med. 2016, 41, 1124–1129. (In Chinese) [Google Scholar] [CrossRef]
- Zhang, H.; Li, Z.; Zhou, Z.; Yang, H.; Zhong, Z.; Lou, C. Antidepressant-like effects of ginsenosides: A comparison of ginsenoside Rb3 and its four deglycosylated derivatives, Rg3, Rh2, compound K, and 20(S)-protopanaxadiol in mice models of despair. Pharmacol. Biochem. Behav. 2016, 140, 17–26. [Google Scholar] [CrossRef] [PubMed]
- Pang, H.H.; Li, M.Y.; Wang, Y.; Tang, M.K.; Ma, C.H.; Huang, J.M. Effect of compatible herbs on the pharmacokinetics of effective components of Panax notoginseng in Fufang Xueshuantong Capsule. J. Zhejiang Univ. Sci. B 2017, 18, 343–352. [Google Scholar] [CrossRef] [PubMed]
- Tian, Z.; Pang, H.; Du, S.; Lu, Y.; Zhang, L.; Wu, H.; Guo, S.; Wang, M.; Zhang, Q. Effect of Panax notoginseng saponins on the pharmacokinetics of aspirin in rats. J. Chromatogr. B 2017, 1040, 136–143. [Google Scholar] [CrossRef] [PubMed]
- Zhao, S.; Zheng, M.X.; Chen, H.E.; Wu, C.Y.; Wang, W.T. Effect of panax notoginseng saponins injection on the p38MAPK pathway in lung tissue in a rat model of hypoxic pulmonary hypertension. Chin. J. Integr. Med. 2015, 21, 147–151. [Google Scholar] [CrossRef] [PubMed]
- Zhai, Y.; Meng, X.; Luo, Y.; Wu, Y.; Ye, T.; Zhou, P.; Ding, S.; Wang, M.; Lu, S.; Zhu, L. Notoginsenoside R1 ameliorates diabetic encephalopathy by activating the Nrf2 pathway and inhibiting NLRP3 inflammasome activation. Oncotarget 2018, 9, 9344–9363. [Google Scholar] [CrossRef] [PubMed]
- Hou, Q.L.; Wang, Y.; Li, Y.B.; Hu, X.L.; Wang, S.L. Protective effect of notoginsenoside R1 on neuron injury induced by OGD/R through ATF6/Akt signaling pathway. China J. Chin. Mater. Med. 2017, 42, 1167–1174. (In Chinese) [Google Scholar]
- Li, W.; Ling, S.; Yang, Y.; Hu, Z.; Davies, H.; Fang, M. Systematic hypothesis for post-stroke depression caused inflammation and neurotransmission and resultant on possible treatments. Neuro Endocrinol. Lett. 2014, 35, 104–109. [Google Scholar] [PubMed]
- Yang, X.; Yang, S.; Hong, C.; Yu, W.; Guonian, W. Panax Notoginseng Saponins attenuates sevofluraneinduced nerve cell injury by modulating AKT signaling pathway. Mol. Med. Rep. 2017, 16, 7829–7834. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.; Guo, R.; Zhou, G.; Zhou, X.; Kou, Z.; Sui, F.; Li, C.; Tang, L.; Wang, Z. Traditional uses, botany, phytochemistry, pharmacology and toxicology of Panax notoginseng (Burk.) F.H. Chen: A review. J. Ethnopharmacol. 2016, 188, 234–258. [Google Scholar] [CrossRef] [PubMed]
- Berge, L.I.; Riise, T. Comorbidity between Type 2 diabetes and depression in the adult population: Directions of the association and its possible pathophysiological mechanisms. Int. J. Endocrinol. 2015, 2015, 164760. [Google Scholar] [CrossRef] [PubMed]
- Xie, W.; Meng, X.; Zhai, Y.; Zhou, P.; Ye, T.; Wang, Z.; Sun, G.; Sun, X. Panax notoginseng saponins: A review of its mechanisms of antidepressant or anxiolytic effects and network analysis on phytochemistry and pharmacology. Molecules 2018, 23, 940. [Google Scholar] [CrossRef] [PubMed]
- Xie, W.; Zhou, P.; Sun, Y.; Meng, X.; Dai, Z.; Sun, G.; Sun, X. Protective effects and target network analysis of ginsenoside Rg1 in cerebral ischemia and reperfusion injury: A comprehensive overview of experimental studies. Cells 2018, 7, 270. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.F.; Chiou, W.F.; Zhang, J.T. Comparison of the pharmacological effects of Panax ginseng and Panax quinquefolium. Acta Pharmacol. Sin. 2008, 29, 1103–1108. [Google Scholar] [CrossRef] [PubMed]
- Chen, F.; Wen, Q.; Jiang, J.; Li, H.L.; Tan, Y.F.; Li, Y.H.; Zeng, N.K. Could the gut microbiota reconcile the oral bioavailability conundrum of traditional herbs? J. Ethnopharmacol. 2016, 179, 253–264. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Tang, Y.; Liu, C.; Zhang, Y. Development of a method to screen and isolate potential alpha-glucosidase inhibitors from Panax japonicus CA Meyer by ultrafiltration, liquid chromatography, and counter-current chromatography. J. Sep. Sci. 2015, 38, 2014–2023. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Wu, L.; Fan, X.; Zhang, B.; Gao, X.; Wang, Y.; Cheng, Y. Network pharmacology study on major active compounds of Fufang Danshen formula. China J. Chin. Mater. Med. 2011, 36, 2911–2915. (In Chinese) [Google Scholar]
- Park, M.J.; Bae, C.S.; Lim, S.K.; Kim, D.I.; Lim, J.C.; Kim, J.C.; Han, H.J.; Moon, J.H.; Kim, K.Y.; Yoon, K.C.; et al. Effect of protopanaxadiol derivatives in high glucose-induced fibronectin expression in primary cultured rat mesangial cells: Role of mitogen-activated protein kinases and Akt. Arch. Pharmacal. Res. 2010, 33, 151–157. [Google Scholar] [CrossRef] [PubMed]
- Tabandeh, M.R.; Jafari, H.; Hosseini, S.A.; Hashemitabar, M. Ginsenoside Rb1 stimulates adiponectin signaling in C2C12 muscle cells through up-regulation of AdipoR1 and AdipoR2 proteins. Pharm. Biol. 2015, 53, 125–132. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.; Yu, Y.; Szabo, A.; Han, M.; Huang, X.F. Central inflammation and leptin resistance are attenuated by ginsenoside Rb1 treatment in obese mice fed a high-fat diet. PLoS ONE 2014, 9, e92618. [Google Scholar] [CrossRef] [PubMed]
- Xue, B.; Sun, L.; Li, X.; Wang, X.; Zhang, Y.; Mu, Y.; Liang, L. Ginsenoside Rb1 relieves glucose fluctuation-induced oxidative stress and apoptosis in Schwann cells. Neural Regen. Res. 2012, 7, 2340–2346. [Google Scholar] [CrossRef] [PubMed]
- Yu, X.; Ye, L.; Zhang, H.; Zhao, J.; Wang, G.; Guo, C.; Shang, W. Ginsenoside Rb1 ameliorates liver fat accumulation by upregulating perilipin expression in adipose tissue of db/db obese mice. J. Ginseng Res. 2015, 39, 199–205. [Google Scholar] [CrossRef] [PubMed]
- Sanada, S.; Kondo, N.; Shoji, J.; Tanaka, O.; Shibata, S. Studies on the saponins of ginseng. II. Structures of ginsenoside-Re, -Rf and -Rg2. Chem. Pharm. Bull. 2008, 22, 2407–2412. [Google Scholar] [CrossRef]
- Chen, Q.S. Pharmacological studies on notoginseng saponins isolated from the fibrous root of Panax notoginseng. Zhong Yao Tong Bao 1987, 12, 45–47. [Google Scholar] [PubMed]
- Washida, D.; Kitanaka, S. Determination of polyacetylenes and ginsenosides in Panax species using high performance liquid chromatography. Chem. Pharm. Bull. 2003, 51, 1314–1317. [Google Scholar] [CrossRef] [PubMed]
- Nah, S.Y.; Park, H.J.; Mccleskey, E.W. A trace component of ginseng that inhibits Ca2+ channels through a pertussis toxin-sensitive G protein. Proc. Natl. Acad. Sci. USA 1995, 92, 8739–8743. [Google Scholar] [CrossRef] [PubMed]
- Wang, B.; Cheng, K.K. Hypothalamic AMPK as a Mediator of Hormonal Regulation of Energy Balance. Int. J. Mol. Sci. 2018, 19, 3552. [Google Scholar] [CrossRef] [PubMed]
- Lyons, C.L.; Roche, H.M. Nutritional Modulation of AMPK-Impact upon Metabolic-Inflammation. Int. J. Mol. Sci. 2018, 19, 3092. [Google Scholar] [CrossRef] [PubMed]
- Xiong, Y.; Shen, L.; Liu, K.J.; Tso, P.; Xiong, Y.; Wang, G.; Woods, S.C.; Liu, M. Antiobesity and antihyperglycemic effects of ginsenoside Rb1 in rats. Diabetes 2010, 59, 2505–2512. [Google Scholar] [CrossRef] [PubMed]
- Waki, I.; Kyo, H.; Yasuda, M.; Kimura, M. Effects of a hypoglycemic component of ginseng radix on insulin biosynthesis in normal and diabetic animals. J. Pharm. Dyn. 1982, 5, 547–554. [Google Scholar] [CrossRef]
- Zhong, Z.D.; Wang, C.M.; Wang, W.; Shen, L.; Chen, Z.H. Major hypoglycemic ingredients of Panax notoginseng saponins for treating diabetes. J. Sichuan Univ. 2014, 45, 235–239. (In Chinese) [Google Scholar]
- Shang, W.; Yang, Y.; Jiang, B.; Jin, H.; Zhou, L.; Liu, S.; Chen, M. Ginsenoside Rb1 promotes adipogenesis in 3T3-L1 cells by enhancing PPARgamma2 and C/EBPalpha gene expression. Life Sci. 2007, 80, 618–625. [Google Scholar] [CrossRef] [PubMed]
- Shang, W.; Yang, Y.; Zhou, L.; Jiang, B.; Jin, H.; Chen, M. Ginsenoside Rb1 stimulates glucose uptake through insulin-like signaling pathway in 3T3-L1 adipocytes. J. Endocrinol. 2008, 198, 561–569. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Li, R.; Li, N.; Zheng, F.; Dai, Y.; Ge, Y.; Yue, H.; Yu, S. Mechanism of antidiabetic and synergistic effects of ginseng polysaccharide and ginsenoside Rb1 on diabetic rat model. J. Pharm. Biomed. Anal. 2018, 158, 451–460. [Google Scholar] [CrossRef] [PubMed]
- Xie, Z.; Loi Truong, T.; Zhang, P.; Xu, F.; Xu, X.; Li, P. Dan-Qi prescription ameliorates insulin resistance through overall corrective regulation of glucose and fat metabolism. J. Ethnopharmacol. 2015, 172, 70–79. [Google Scholar] [CrossRef] [PubMed]
- Shang, W.B.; Guo, C.; Zhao, J.; Yu, X.Z.; Zhang, H. Ginsenoside Rb1 upregulates expressions of GLUTs to promote glucose consumption in adiopcytes. China J. Chin. Mater. Med. 2014, 39, 4448–4452. [Google Scholar]
- Song, B.; Ding, L.; Zhang, H.; Chu, Y.; Chang, Z.; Yu, Y.; Guo, D.; Zhang, S.; Liu, X. Ginsenoside Rb1 increases insulin sensitivity through suppressing 11beta-hydroxysteroid dehydrogenase type I. Am. J. Transl. Res. 2017, 9, 1049–1057. [Google Scholar] [PubMed]
- Sadie-Van Gijsen, H. Adipocyte biology: It is time to upgrade to a new model. J. Cell. Physiol. 2018, 234, 2399–2425. [Google Scholar] [CrossRef] [PubMed]
- Smith, C.J.; Wejksnora, P.J.; Warner, J.R.; Rubin, C.S.; Rosen, O.M. Insulin-stimulated protein phosphorylation in 3T3-L1 preadipocytes. Proc. Natl. Acad. Sci. USA 1979, 76, 2725–2729. [Google Scholar] [CrossRef] [PubMed]
- Shen, L.; Haas, M.; Wang, D.Q.; May, A.; Lo, C.C.; Obici, S.; Tso, P.; Woods, S.C.; Liu, M. Ginsenoside Rb1 increases insulin sensitivity by activating AMP-activated protein kinase in male rats. Physiol. Rep. 2015, 3, e12543. [Google Scholar] [CrossRef] [PubMed]
- Shen, L.; Xiong, Y.; Wang, D.Q.; Howles, P.; Basford, J.E.; Wang, J.; Xiong, Y.Q.; Hui, D.Y.; Woods, S.C.; Liu, M. Ginsenoside Rb1 reduces fatty liver by activating AMP-activated protein kinase in obese rats. J. Lipid Res. 2013, 54, 1430–1438. [Google Scholar] [CrossRef] [PubMed]
- Chen, F.; Chen, Y.; Kang, X.; Zhou, Z.; Zhang, Z.; Liu, D. Anti-apoptotic function and mechanism of ginseng saponins in Rattus pancreatic beta-cells. Biol. Pharm. Bull. 2012, 35, 1568–1573. [Google Scholar] [CrossRef] [PubMed]
- Shen, N.; Li, X.; Zhou, T.; Bilal, M.U.; Du, N.; Hu, Y.; Qin, W.; Xie, Y.; Wang, H.; Wu, J.; et al. Shensong Yangxin Capsule prevents diabetic myocardial fibrosis by inhibiting TGF-beta1/Smad signaling. J. Ethnopharmacol. 2014, 157, 161–170. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; Zhang, M.; Hu, M.Y.; Guo, H.F.; Li, J.; Yu, Y.L.; Jin, S.; Wang, X.T.; Liu, L.; Liu, X.D. Increased glucagon-like peptide-1 secretion may be involved in antidiabetic effects of ginsenosides. J. Endocrinol. 2013, 217, 185–196. [Google Scholar] [CrossRef] [PubMed]
- DeFronzo, R.A. Pharmacologic therapy for type 2 diabetes mellitus. Ann. Intern. Med. 1999, 131, 281–303. [Google Scholar] [CrossRef] [PubMed]
- Turner, R.C.; Cull, C.A.; Frighi, V.; Holman, R.R. Glycemic control with diet, sulfonylurea, metformin, or insulin in patients with type 2 diabetes mellitus: Progressive requirement for multiple therapies (UKPDS 49). UK Prospective Diabetes Study (UKPDS) Group. JAMA 1999, 281, 2005–2012. [Google Scholar] [CrossRef] [PubMed]
- Cushman, W.C.; Evans, G.W.; Byington, R.P.; Goff, D.C.; Grimm, R.H.; Cutler, J.A.; Simons-Morton, D.G.; Basile, J.N.; Corson, M.A.; Probstfield, J.L. Effects of intensive blood-pressure control in type 2 diabetes mellitus. N. Engl. J. Med. 2010, 362, 1575–1585. [Google Scholar] [PubMed]
- Uzayisenga, R.; Ayeka, P.A.; Wang, Y. Anti-diabetic potential of Panax notoginseng saponins (PNS): A review. Phytother. Res. 2014, 28, 510–516. [Google Scholar] [CrossRef] [PubMed]
- Bloomgarden, Z.T. Diabetes Complications. Diabetes Care 2004, 27, 1506. [Google Scholar] [CrossRef] [PubMed]
- Jiping, L.; Shuyuan, W.; Liang, F.; Dongying, M.; Qiang, F.; Yu, S.; Xiaobin, J.; Shiping, M. Hypoglycemic and antioxidant activities of paeonol and its beneficial effect on diabetic encephalopathy in streptozotocin-induced diabetic rats. J. Med. Food 2013, 16, 577–586. [Google Scholar]
- Sima, A.A.; Zhang, W.; Kreipke, C.W.; Rafols, J.A.; Hoffman, W.H. Inflammation in Diabetic Encephalopathy is Prevented by C-Peptide. Rev. Diabet. Stud. 2009, 6, 37–42. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, F.K.; Zhiheng, H.; King, G.L. Molecular targets of diabetic cardiovascular complications. Curr. Drug Targets 2005, 6, 487–494. [Google Scholar] [CrossRef] [PubMed]
- Desmond, J.; Hirofumi, H.; Griendling, K.K. Oxidative stress and diabetic cardiovascular complications. Free Radic. Biol. Med. 2006, 40, 183–192. [Google Scholar]
- Lu, J.M.; Weakley, S.M.; Yang, Z.; Hu, M.; Yao, Q.; Chen, C. Ginsenoside Rb1 directly scavenges hydroxyl radical and hypochlorous acid. Curr. Pharm. Des. 2012, 18, 6339–6347. [Google Scholar] [CrossRef] [PubMed]
- Zhu, J.; Wang, K.Z.; Chu, C.T. After the banquet: Mitochondrial biogenesis, mitophagy, and cell survival. Autophagy 2013, 9, 1663–1676. [Google Scholar] [CrossRef] [PubMed]
- Marini, M.G.; Sonnino, C.; Previtero, M.; Biasucci, L.M. Targeting inflammation: impact on atherothrombosis. J. Cardiovasc. Transl. Res. 2014, 7, 9–18. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Shao, Z.H.; Xie, J.T.; Wang, C.Z.; Ramachandran, S.; Yin, J.J.; Aung, H.; Li, C.Q.; Qin, G.; Vanden Hoek, T.; et al. The effects of ginsenoside Rb1 on JNK in oxidative injury in cardiomyocytes. Arch. Pharmacal Res. 2012, 35, 1259–1267. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.; Jiang, J.; Lu, J.M.; Chai, H.; Wang, X.; Lin, P.H.; Yao, Q. Resistin decreases expression of endothelial nitric oxide synthase through oxidative stress in human coronary artery endothelial cells. Am. J. Physiol. Heart Circ. Physiol. 2010, 299, H193–H201. [Google Scholar] [CrossRef] [PubMed]
- Hui, C.; Qiuyan, W.; Lifeng, H.; Tian, X.; Yan, F. Ginsenoside Rb1 inhibits tumor necrosis factor-alpha-induced vascular cell adhesion molecule-1 expression in human endothelial cells. Biol. Pharm. Bull. 2008, 31, 2050. [Google Scholar]
- Wang, Z.; Huang, Y.; Cheng, Y.; Tan, Y.; Wu, F.; Wu, J.; Shi, H.; Zhang, H.; Yu, X.; Gao, H. Endoplasmic reticulum stress-induced neuronal inflammatory response and apoptosis likely plays a key role in the development of diabetic encephalopathy. Oncotarget 2016, 7, 78455–78472. [Google Scholar] [CrossRef] [PubMed]
- Carlos Alberto, Y.W.; Helena Maria, T.B.; Carmen Regla, V. GABAergic modulation in diabetic encephalopathy-related depression. Curr. Pharm. Des. 2015, 21, 4980–4988. [Google Scholar]
- Sang, H.L.; Jung, B.H.; Sun, Y.K.; Lee, E.H.; Chung, B.C. The antistress effect of ginseng total saponin and ginsenoside Rg3 and Rb1 evaluated by brain polyamine level under immobilization stress. Pharmacol. Res. 2006, 54, 46–49. [Google Scholar]
- Liu, D.; Zhang, H.; Gu, W.; Liu, Y.; Zhang, M. Ginsenoside Rb1 protects hippocampal neurons from high glucose-induced neurotoxicity by inhibiting GSK3beta-mediated CHOP induction. Mol. Med. Rep. 2014, 9, 1434–1438. [Google Scholar] [CrossRef] [PubMed]
- Xia, R.; Zhao, B.; Wu, Y.; Hou, J.B.; Zhang, L.; Xu, J.J.; Xia, Z.Y. Ginsenoside Rb1 preconditioning enhances eNOS expression and attenuates myocardial ischemia/reperfusion injury in diabetic rats. J. Biomed. Biotechnol. 2011, 2011, 767930. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.; Xia, Z.Y.; Dou, J.; Zhang, L.; Xu, J.J.; Zhao, B.; Lei, S.; Liu, H.M. Protective effect of ginsenoside Rb1 against myocardial ischemia/reperfusion injury in streptozotocin-induced diabetic rats. Mol. Biol. Rep. 2011, 38, 4327–4335. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; Hu, M.; Guo, H.; Zhang, M.; Zhang, J.; Li, F.; Zhong, Z.; Chen, Y.; Li, Y.; Xu, P.; et al. Combined contribution of increased intestinal permeability and inhibited deglycosylation of ginsenoside Rb1 in the intestinal tract to the enhancement of ginsenoside Rb1 exposure in diabetic rats after oral administration. Drug Metab. Dispos. 2015, 43, 1702–1710. [Google Scholar] [CrossRef] [PubMed]
- Holst, J.J. The physiology of glucagon-like peptide 1. Physiol. Rev. 2007, 87, 1409–1439. [Google Scholar] [CrossRef] [PubMed]
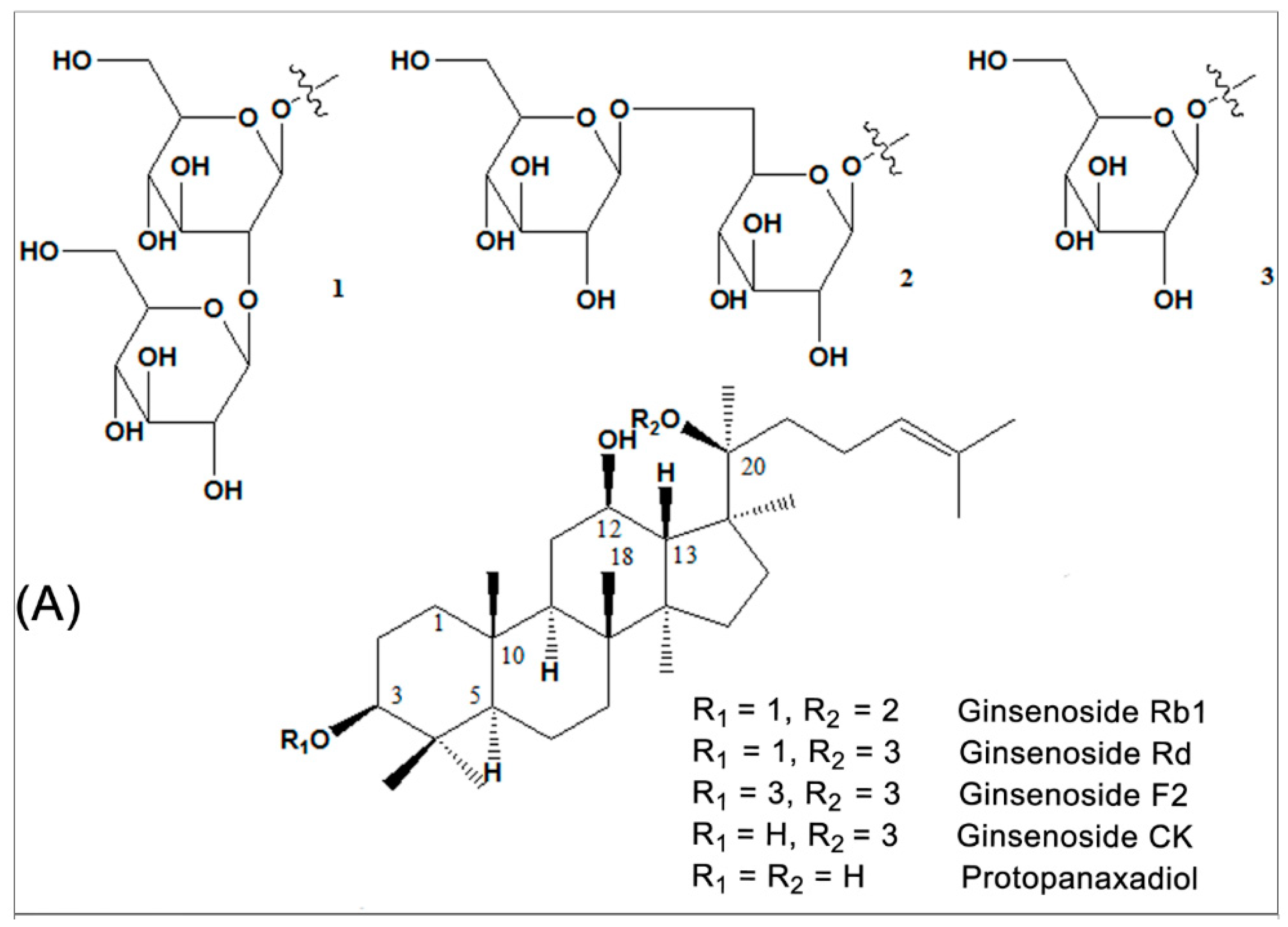
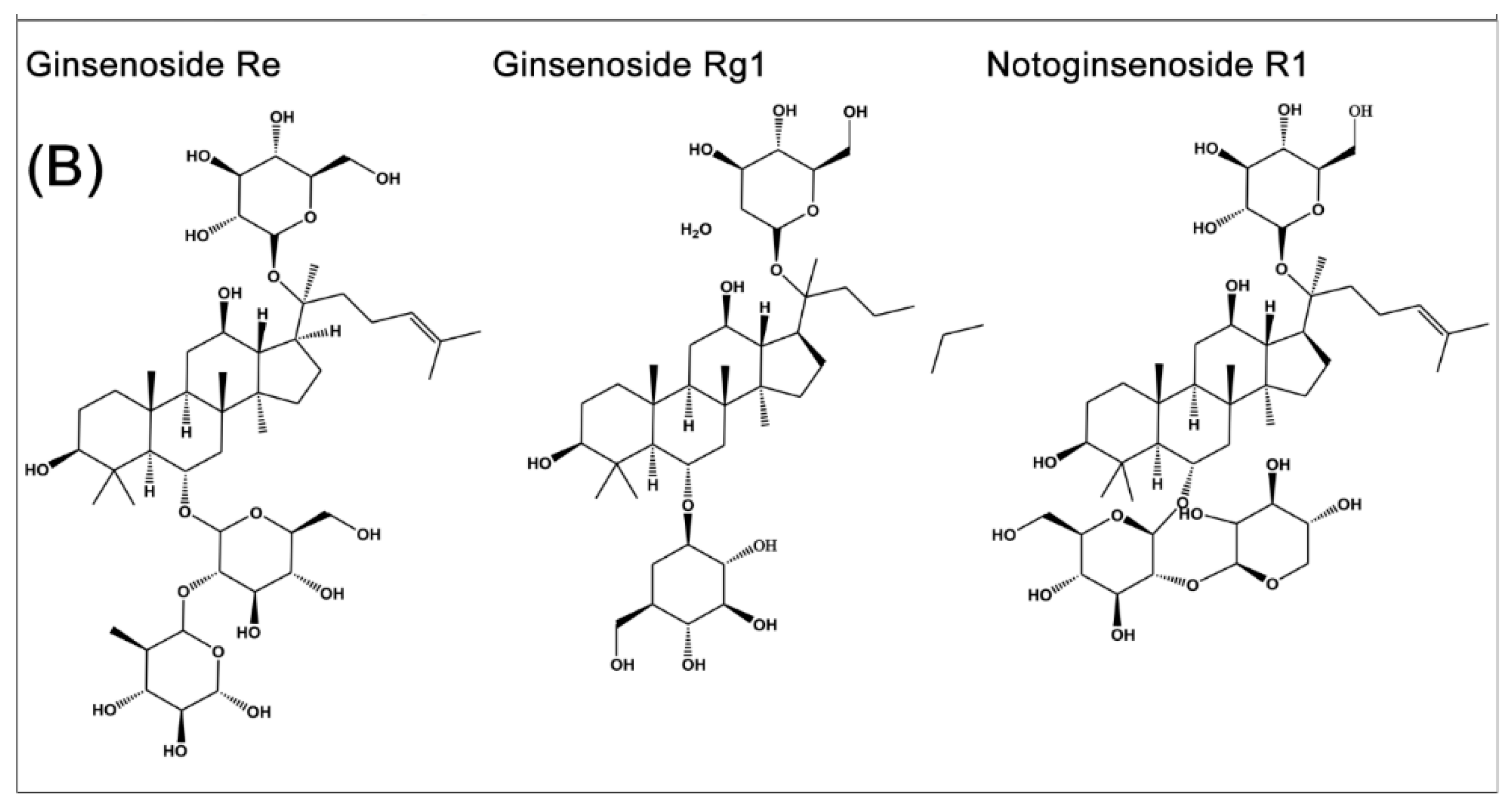
| Model | Type | Inducer | Animal/Cell | Effects | Mechanisms | Ref. |
|---|---|---|---|---|---|---|
| DM | In vitro | AdipoR1 sense siRNA | C2C12 myotubes | ↑Basal AdipoR levels ↑GLUT4 translocations | ↑Translocations of GLUT4; Adiponectin signaling pathway | [61] |
| T2DM; Obesity | In vivo | HFD | Obese mouse | ↓Body weight gain ↓Fat accumulation ↑Glucose tolerance | Modulate inflammation ↑Central leptin sensitivity ↓Pro-inflammatory cytokines | [62] |
| T2DM | In vivo | db/db | C57; db/db mice | ↑Insulin sensitivity ↓Adipocyte lipolysis ↓Levels of free fatty acids ↑Hepatic fat accumulation ↓Liver weight, hepatic triglyceride | ↓TNF-α ↑Perilipin ↑Insulin sensitivity ↑Level of adiponectin | [64] |
| T2DM; Obesity | In vivo | HFD | Rat | ↓Food intake ↓Body weight gain ↓Body fat content ↑Energy expenditure | ↑c-Fos ↓NPY ↓PI3k/Akt signaling pathway | [71] |
| Obesity | In vivo | KK-Ay | C57 KK-Ay mice | ↓Body weight gain ↓FBG and food consumption | ↑Insulin/leptin sensitivities ↓Insulin resistance index | [73] |
| T2DM; Obesity | In vitro | Differentiation inducer | 3T3-L1 cells | ↑Glucose uptake ↑Lipid accumulation ↑Proliferation of 3T3-L1 preadipocytes | ↑ap2, GLUT4 ↑Adipogenesis ↑PPARγ2 and C/EBPα | [74] |
| T2DM; Obesity | In vitro | Differentiation inducer | 3T3-L1 cells; C2C12 myotubes | ↑PI3K activity ↑Glucose uptake ↑IRS1 and PKB phosphorylation | Activating insulin signaling pathway | [75] |
| T2DM; Obesity | In vivo | db/db | db/db mice | ↑GLUT1 and GLUT4 ↑Akt Phosphorylation ↓HOMA-IR and FBG and FINS | ↑Glucose metabolism ↑Insulin sensitizing activity | [78] |
| T2DM | In vivo | HFD | C57BL/C mice | ↑Glucose tolerance ↓11β-HSD1 levels ↑Fasting blood glucose | ↓11β-HSD1 ↑Insulin sensitivity | [79] |
| DM | In vitro | HG; Cytokine | Rin-m5F | ↓iNOS expression and NO ↓Pancreatic β-cell apoptosis | ↓Caspase-3 ↓Apoptosis-related genes | [84] |
| T2DM | In vivo; In vitro | HFD; STZ | Male SD rats; NCI-H716 cells | ↑GLP-1 secretion ↑ATP:ADP ratio | ↑GLP-1 ↑Proglucagon | [86] |
| Model | Type | Inducer | Animal/Cell | Effects | Mechanisms | Ref. |
|---|---|---|---|---|---|---|
| T2DM | In vivo | HFD; HG STZ | Male Wistar rats; Intestinal microflora | ↓Blood sugar levels ↑Biotransformation pathway ↑Fecal-d-glucosidase activity | ↑Pathway (Rb1→Rd→F2→CK); gut microbiota-mediated bioconversion | [57] |
| DM DN | In vitro | HG | Mesangial cells | ↓Phosphorylation of p38, JNK, and p44/42 MAPK ↓Fibronectin expression | ↓Phosphorylation of p44/42 MAPK, p38 MAPK, JNK/SAPK, and Akt | [60] |
| DPN | In vitro | HG; | Schwann cells | ↓Bax ↓ROS and 8-OHDG | ↑Bcl-2 ↓Apoptosis ↓Oxidative stress | [63] |
| T2DM | In vitro | Inducers | 3T3-L1 adipocytes C2C12 myotubes | ↑PI3K activity ↑Glucose uptake ↑IRS1 and PKB | ↑GLUT1 and GLUT4 ↑Insulin signaling pathway | [75] |
| DC DM | In vivo | STZ | Diabetic rats | ↓Heart weight/body weight ↑Impaired cardiac function ↓Col-1, col-3, MMP-2, MMP-9 and α-SMA | ↑Smad7 ↓Cardiac fibrosis ↓TGF-β1 and p-Smad2/3, TGF-β1/Smad signaling | [85] |
| DM | In vitro | HG; cytokine | Rin-m5F | ↓NO production ↓Pancreatic β-cell apoptosis | ↓iNOS ↓Fas and caspase-3 | [84] |
| T2DM | In vivo; In vitro | HFD; STZ | Male SD rats NCI-H716 cells | ↑Ratio of the ATP:ADP ↑GLP-1 secretion | ↑GLP-1 ↑Proglucagon | [86] |
| DM | In vitro | ROS | Cell-free system | ↓·OH and HOCl | Unique anti-oxidant mechanisms | [96]. |
| DM | In vitro | HG | Hippocampal neurons | ↓Neuronal loss ↑Cell viability | ↓CHOP protein ↓p-PERK and p-GSK-3β | [105] |
| DM MI/R | In vivo | STZ; MI/R | Male SD rat | ↓Plasma CK and LDH ↓Myocardial infarct size ↓Myocardial oxidative stress | ↑eNOS and NO; ↓Oxidative stress | [106] |
| DM MI/R | In vivo | STZ; MI/R | Male SD rat | ↓Infarct size ↓Caspase-3 activity ↓Cardiomyocyte apoptosis | ↑Phosphorylated Akt; activation of PI3K/Akt pathway | [107] |
| T2DM | In vivo; In vitro | HFD; STZ | Male SD rat Caco-2 cells | ↑Rb1 absorption ↑Rb1 systemic exposures ↑Portal Rb1 concentration | ↓Rb1 deglycosylation ↑Rb1 intestinal absorption | [108] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhou, P.; Xie, W.; He, S.; Sun, Y.; Meng, X.; Sun, G.; Sun, X. Ginsenoside Rb1 as an Anti-Diabetic Agent and Its Underlying Mechanism Analysis. Cells 2019, 8, 204. https://doi.org/10.3390/cells8030204
Zhou P, Xie W, He S, Sun Y, Meng X, Sun G, Sun X. Ginsenoside Rb1 as an Anti-Diabetic Agent and Its Underlying Mechanism Analysis. Cells. 2019; 8(3):204. https://doi.org/10.3390/cells8030204
Chicago/Turabian StyleZhou, Ping, Weijie Xie, Shuaibing He, Yifan Sun, Xiangbao Meng, Guibo Sun, and Xiaobo Sun. 2019. "Ginsenoside Rb1 as an Anti-Diabetic Agent and Its Underlying Mechanism Analysis" Cells 8, no. 3: 204. https://doi.org/10.3390/cells8030204
APA StyleZhou, P., Xie, W., He, S., Sun, Y., Meng, X., Sun, G., & Sun, X. (2019). Ginsenoside Rb1 as an Anti-Diabetic Agent and Its Underlying Mechanism Analysis. Cells, 8(3), 204. https://doi.org/10.3390/cells8030204





