CD38 Expression by Myeloma Cells and Its Role in the Context of Bone Marrow Microenvironment: Modulation by Therapeutic Agents
Abstract
1. Introduction
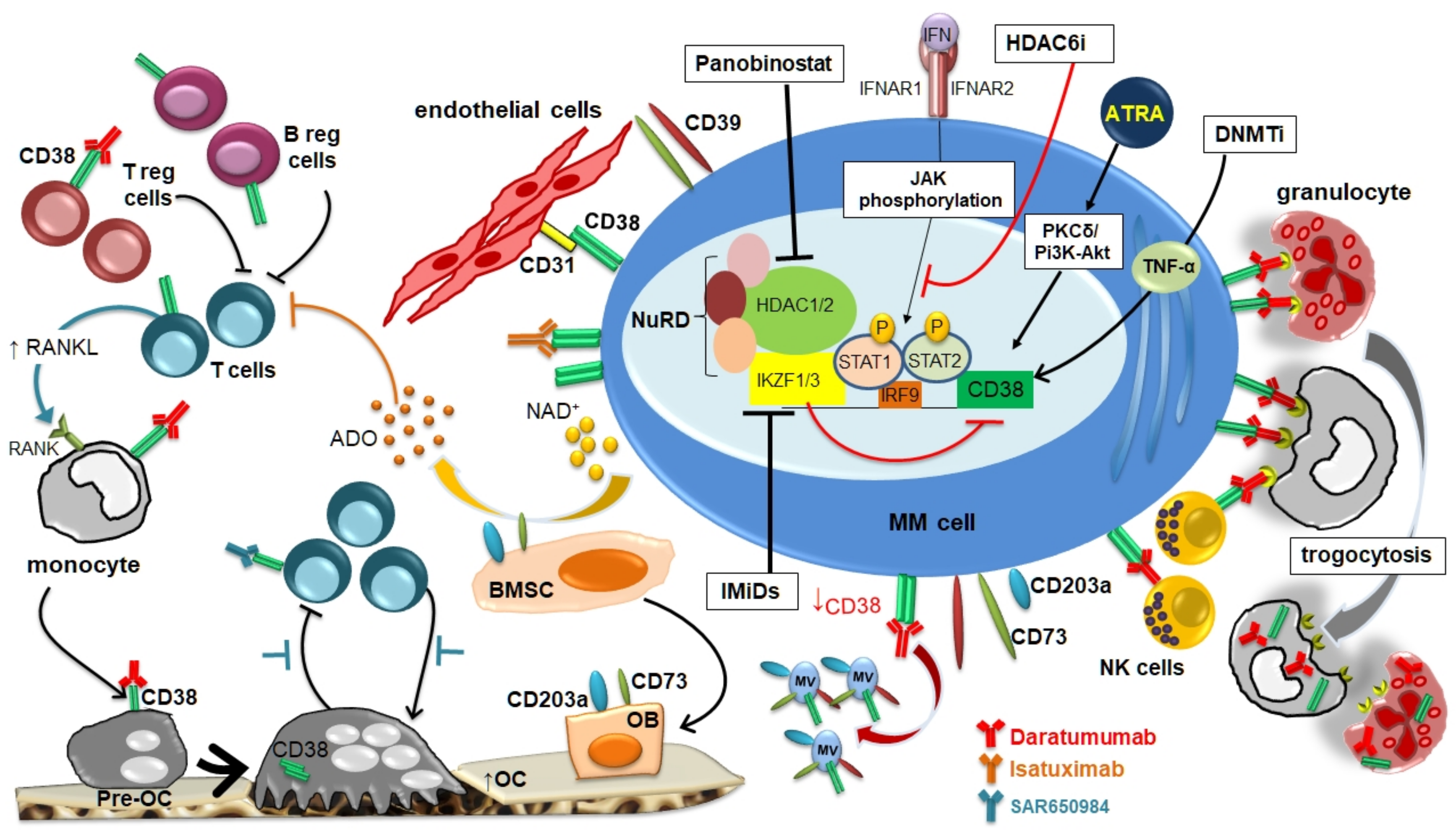
2. CD38 Expression by MM Cells and BM Microenvironment
3. CD38 Modulation by Anti-CD38 Monoclonal Antibodies in MM Cells
4. Drug-Mediated Modulation of CD38 Expression by MM Cells
5. Clinical Results of Anti-CD38 mAbs in Combination with Agents Able to Upregulate CD38 Expression in MM Patients
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Anderson, K.C. Multiple myeloma. Hematol. Oncol. Clin. North. Am. 2014, 28, xi–xii. [Google Scholar] [CrossRef] [PubMed]
- Chillemi, A.; Zaccarello, G.; Quarona, V.; Ferracin, M.; Ghimenti, C.; Massaia, M.; Horenstein, A.L.; Malavasi, F. Anti-CD38 antibody therapy: Windows of opportunity yielded by the functional characteristics of the target molecule. Mol. Med. 2013, 19, 99–108. [Google Scholar] [CrossRef] [PubMed]
- Lonial, S.; Weiss, B.M.; Usmani, S.Z.; Singhal, S.; Chari, A.; Bahlis, N.J.; Belch, A.; Krishnan, A.; Vescio, R.A.; Mateos, M.V.; et al. Daratumumab monotherapy in patients with treatment-refractory multiple myeloma (SIRIUS): An open-label, randomised, phase 2 trial. Lancet 2016, 387, 1551–1560. [Google Scholar] [CrossRef]
- Frerichs, K.A.; Nagy, N.A.; Lindenbergh, P.L.; Bosman, P.; Marin Soto, J.; Broekmans, M.; Groen, R.W.J.; Themeli, M.; Nieuwenhuis, L.; Stege, C.; et al. CD38-targeting antibodies in multiple myeloma: Mechanisms of action and clinical experience. Expert Rev. Clin. Immunol. 2018, 14, 197–206. [Google Scholar] [CrossRef]
- Oostvogels, R.; Jak, M.; Raymakers, R.; Mous, R.; Minnema, M.C. Efficacy of retreatment with immunomodulatory drugs and proteasome inhibitors following daratumumab monotherapy in relapsed and refractory multiple myeloma patients. Br. J. Haematol. 2018, 183, 60–67. [Google Scholar] [CrossRef]
- Usmani, S.Z.; Weiss, B.M.; Plesner, T.; Bahlis, N.J.; Belch, A.; Lonial, S.; Lokhorst, H.M.; Voorhees, P.M.; Richardson, P.G.; Chari, A.; et al. Clinical efficacy of daratumumab monotherapy in patients with heavily pretreated relapsed or refractory multiple myeloma. Blood 2016, 128, 37–44. [Google Scholar] [CrossRef]
- Dimopoulos, M.A.; Oriol, A.; Nahi, H.; San-Miguel, J.; Bahlis, N.J.; Usmani, S.Z.; Rabin, N.; Orlowski, R.Z.; Komarnicki, M.; Suzuki, K.; et al. Daratumumab, Lenalidomide, and Dexamethasone for Multiple Myeloma. New Engl. J. Med. 2016, 375, 1319–1331. [Google Scholar] [CrossRef]
- Nijhof, I.S.; Casneuf, T.; van Velzen, J.; van Kessel, B.; Axel, A.E.; Syed, K.; Groen, R.W.; van Duin, M.; Sonneveld, P.; Minnema, M.C.; et al. CD38 expression and complement inhibitors affect response and resistance to daratumumab therapy in myeloma. Blood 2016, 128, 959–970. [Google Scholar] [CrossRef]
- Kyle, R.A.; Rajkumar, S.V. Multiple myeloma. New Engl. J. Med. 2004, 351, 1860–1873. [Google Scholar] [CrossRef]
- Palumbo, A.; Anderson, K. Multiple myeloma. New Engl. J. Med. 2011, 364, 1046–1060. [Google Scholar] [CrossRef]
- Santonocito, A.M.; Consoli, U.; Bagnato, S.; Milone, G.; Palumbo, G.A.; Di Raimondo, F.; Stagno, F.; Guglielmo, P.; Giustolisi, R. Flow cytometric detection of aneuploid CD38(++) plasmacells and CD19(+) B-lymphocytes in bone marrow, peripheral blood and PBSC harvest in multiple myeloma patients. Leuk. Res. 2004, 28, 469–477. [Google Scholar] [CrossRef] [PubMed]
- Atanackovic, D.; Steinbach, M.; Radhakrishnan, S.V.; Luetkens, T. Immunotherapies targeting CD38 in Multiple Myeloma. Oncoimmunology 2016, 5, e1217374. [Google Scholar] [CrossRef] [PubMed]
- Lokhorst, H.M.; Plesner, T.; Laubach, J.P.; Nahi, H.; Gimsing, P.; Hansson, M.; Minnema, M.C.; Lassen, U.; Krejcik, J.; Palumbo, A.; et al. Targeting CD38 with Daratumumab Monotherapy in Multiple Myeloma. New Engl. J. Med. 2015, 373, 1207–1219. [Google Scholar] [CrossRef] [PubMed]
- Harada, H.; Kawano, M.M.; Huang, N.; Harada, Y.; Iwato, K.; Tanabe, O.; Tanaka, H.; Sakai, A.; Asaoku, H.; Kuramoto, A. Phenotypic difference of normal plasma cells from mature myeloma cells. Blood 1993, 81, 2658–2663. [Google Scholar] [CrossRef] [PubMed]
- Bataille, R.; Jego, G.; Robillard, N.; Barille-Nion, S.; Harousseau, J.L.; Moreau, P.; Amiot, M.; Pellat-Deceunynck, C. The phenotype of normal, reactive and malignant plasma cells. Identification of “many and multiple myelomas” and of new targets for myeloma therapy. Haematologica 2006, 91, 1234–1240. [Google Scholar]
- Krejcik, J.; Casneuf, T.; Nijhof, I.S.; Verbist, B.; Bald, J.; Plesner, T.; Syed, K.; Liu, K.; van de Donk, N.W.; Weiss, B.M.; et al. Daratumumab depletes CD38+ immune regulatory cells, promotes T-cell expansion, and skews T-cell repertoire in multiple myeloma. Blood 2016, 128, 384–394. [Google Scholar] [CrossRef]
- Malavasi, F.; Deaglio, S.; Funaro, A.; Ferrero, E.; Horenstein, A.L.; Ortolan, E.; Vaisitti, T.; Aydin, S. Evolution and function of the ADP ribosyl cyclase/CD38 gene family in physiology and pathology. Physiol. Rev. 2008, 88, 841–886. [Google Scholar] [CrossRef]
- Deaglio, S.; Aydin, S.; Vaisitti, T.; Bergui, L.; Malavasi, F. CD38 at the junction between prognostic marker and therapeutic target. Trends Mol. Med. 2008, 14, 210–218. [Google Scholar] [CrossRef]
- Deaglio, S.; Mallone, R.; Baj, G.; Arnulfo, A.; Surico, N.; Dianzani, U.; Mehta, K.; Malavasi, F. CD38/CD31, a receptor/ligand system ruling adhesion and signaling in human leukocytes. Chem. Immunol. 2000, 75, 99–120. [Google Scholar]
- Deaglio, S.; Morra, M.; Mallone, R.; Ausiello, C.M.; Prager, E.; Garbarino, G.; Dianzani, U.; Stockinger, H.; Malavasi, F. Human CD38 (ADP-ribosyl cyclase) is a counter-receptor of CD31, an Ig superfamily member. J. Immunol. 1998, 160, 395–402. [Google Scholar]
- Watt, S.M.; Gschmeissner, S.E.; Bates, P.A. PECAM-1: Its expression and function as a cell adhesion molecule on hemopoietic and endothelial cells. Leuk. Lymphoma 1995, 17, 229–244. [Google Scholar] [CrossRef] [PubMed]
- Vallario, A.; Chilosi, M.; Adami, F.; Montagna, L.; Deaglio, S.; Malavasi, F.; Caligaris-Cappio, F. Human myeloma cells express the CD38 ligand CD31. Br. J. Haematol. 1999, 105, 441–444. [Google Scholar] [CrossRef] [PubMed]
- Accardi, F.; Notarfranchi, L.; Palma, B.D.; Manfra, I.; De Luca, F.; Mancini, C.; Martella, E.; Marchica, V.; Storti, P.; Bolzoni, M.; et al. The Loss of Cd38 Expression by Myeloma Plasma Cells May Occur in the Extramedullary Disease. Haematologica 2018, 103, S54. [Google Scholar]
- Aarhus, R.; Graeff, R.M.; Dickey, D.M.; Walseth, T.F.; Lee, H.C. ADP-ribosyl cyclase and CD38 catalyze the synthesis of a calcium-mobilizing metabolite from NADP. J. Biol. Chem. 1995, 270, 30327–30333. [Google Scholar] [CrossRef] [PubMed]
- Horenstein, A.L.; Chillemi, A.; Zaccarello, G.; Bruzzone, S.; Quarona, V.; Zito, A.; Serra, S.; Malavasi, F. A CD38/CD203a/CD73 ectoenzymatic pathway independent of CD39 drives a novel adenosinergic loop in human T lymphocytes. Oncoimmunology 2013, 2, e26246. [Google Scholar] [CrossRef]
- Horenstein, A.L.; Quarona, V.; Toscani, D.; Costa, F.; Chillemi, A.; Pistoia, V.; Giuliani, N.; Malavasi, F. Adenosine Generated in the Bone Marrow Niche Through a CD38-Mediated Pathway Correlates with Progression of Human Myeloma. Mol. Med. 2016, 22. [Google Scholar] [CrossRef]
- Morandi, F.; Marimpietri, D.; Horenstein, A.L.; Bolzoni, M.; Toscani, D.; Costa, F.; Castella, B.; Faini, A.C.; Massaia, M.; Pistoia, V.; et al. Microvesicles released from multiple myeloma cells are equipped with ectoenzymes belonging to canonical and non-canonical adenosinergic pathways and produce adenosine from ATP and NAD+. Oncoimmunology 2018, 7, e1458809. [Google Scholar] [CrossRef]
- Horenstein, A.L.; Chillemi, A.; Quarona, V.; Zito, A.; Roato, I.; Morandi, F.; Marimpietri, D.; Bolzoni, M.; Toscani, D.; Oldham, R.J.; et al. NAD+-Metabolizing Ectoenzymes in Remodeling Tumor-Host Interactions: The Human Myeloma Model. Cells 2015, 4, 520–537. [Google Scholar] [CrossRef]
- Abe, M.; Hiura, K.; Wilde, J.; Shioyasono, A.; Moriyama, K.; Hashimoto, T.; Kido, S.; Oshima, T.; Shibata, H.; Ozaki, S.; et al. Osteoclasts enhance myeloma cell growth and survival via cell-cell contact: A vicious cycle between bone destruction and myeloma expansion. Blood 2004, 104, 2484–2491. [Google Scholar] [CrossRef]
- Quarona, V.; Ferri, V.; Chillemi, A.; Bolzoni, M.; Mancini, C.; Zaccarello, G.; Roato, I.; Morandi, F.; Marimpietri, D.; Faccani, G.; et al. Unraveling the contribution of ectoenzymes to myeloma life and survival in the bone marrow niche. Ann. New York Acad. Sci. 2015, 1335, 10–22. [Google Scholar] [CrossRef]
- Costa, F.; Toscani, D.; Chillemi, A.; Quarona, V.; Bolzoni, M.; Marchica, V.; Vescovini, R.; Mancini, C.; Martella, E.; Campanini, N.; et al. Expression of CD38 in myeloma bone niche: A rational basis for the use of anti-CD38 immunotherapy to inhibit osteoclast formation. Oncotarget 2017, 8, 56598–56611. [Google Scholar] [CrossRef] [PubMed]
- Nijhof, I.S.; Groen, R.W.; Lokhorst, H.M.; van Kessel, B.; Bloem, A.C.; van Velzen, J.; de Jong-Korlaar, R.; Yuan, H.; Noort, W.A.; Klein, S.K.; et al. Upregulation of CD38 expression on multiple myeloma cells by all-trans retinoic acid improves the efficacy of daratumumab. Leukemia 2015, 29, 2039–2049. [Google Scholar] [CrossRef] [PubMed]
- Sun, L.; Adebanjo, O.A.; Moonga, B.S.; Corisdeo, S.; Anandatheerthavarada, H.K.; Biswas, G.; Arakawa, T.; Hakeda, Y.; Koval, A.; Sodam, B.; et al. CD38/ADP-ribosyl cyclase: A new role in the regulation of osteoclastic bone resorption. J. Cell Biol. 1999, 146, 1161–1172. [Google Scholar] [CrossRef] [PubMed]
- Sun, L.; Iqbal, J.; Dolgilevich, S.; Yuen, T.; Wu, X.B.; Moonga, B.S.; Adebanjo, O.A.; Bevis, P.J.; Lund, F.; Huang, C.L.; et al. Disordered osteoclast formation and function in a CD38 (ADP-ribosyl cyclase)-deficient mouse establishes an essential role for CD38 in bone resorption. Faseb J. Off. Publ. Fed. Am. Soc. Exp. Biol. 2003, 17, 369–375. [Google Scholar] [CrossRef]
- Sun, L.; Adebanjo, O.A.; Koval, A.; Anandatheerthavarada, H.K.; Iqbal, J.; Wu, X.Y.; Moonga, B.S.; Wu, X.B.; Biswas, G.; Bevis, P.J.; et al. A novel mechanism for coupling cellular intermediary metabolism to cytosolic Ca2+ signaling via CD38/ADP-ribosyl cyclase, a putative intracellular NAD+ sensor. Faseb J. Off. Publ. Fed. Am. Soc. Exp. Biol. 2002, 16, 302–314. [Google Scholar] [CrossRef]
- Grassi, F.; Manferdini, C.; Cattini, L.; Piacentini, A.; Gabusi, E.; Facchini, A.; Lisignoli, G. T cell suppression by osteoclasts in vitro. J. Cell. Physiol. 2011, 226, 982–990. [Google Scholar] [CrossRef]
- Takayanagi, H.; Ogasawara, K.; Hida, S.; Chiba, T.; Murata, S.; Sato, K.; Takaoka, A.; Yokochi, T.; Oda, H.; Tanaka, K.; et al. T-cell-mediated regulation of osteoclastogenesis by signalling cross-talk between RANKL and IFN-gamma. Nature 2000, 408, 600–605. [Google Scholar] [CrossRef]
- An, G.; Acharya, C.; Feng, X.; Wen, K.; Zhong, M.; Zhang, L.; Munshi, N.C.; Qiu, L.; Tai, Y.T.; Anderson, K.C. Osteoclasts promote immune suppressive microenvironment in multiple myeloma: Therapeutic implication. Blood 2016, 128, 1590–1603. [Google Scholar] [CrossRef]
- Feng, X.; Zhang, L.; Acharya, C.; An, G.; Wen, K.; Qiu, L.; Munshi, N.C.; Tai, Y.T.; Anderson, K.C. Targeting CD38 Suppresses Induction and Function of T Regulatory Cells to Mitigate Immunosuppression in Multiple Myeloma. Clin. Cancer Res. Off. J. Am. Assoc. Cancer Res. 2017, 23, 4290–4300. [Google Scholar] [CrossRef]
- Van de Donk, N.W.; Janmaat, M.L.; Mutis, T.; Lammerts van Bueren, J.J.; Ahmadi, T.; Sasser, A.K.; Lokhorst, H.M.; Parren, P.W. Monoclonal antibodies targeting CD38 in hematological malignancies and beyond. Immunol. Rev. 2016, 270, 95–112. [Google Scholar] [CrossRef]
- Jiang, H.; Acharya, C.; An, G.; Zhong, M.; Feng, X.; Wang, L.; Dasilva, N.; Song, Z.; Yang, G.; Adrian, F.; et al. SAR650984 directly induces multiple myeloma cell death via lysosomal-associated and apoptotic pathways, which is further enhanced by pomalidomide. Leukemia 2016, 30, 399–408. [Google Scholar] [CrossRef]
- Van de Donk, N. Immunomodulatory effects of CD38-targeting antibodies. Immunol. Lett. 2018, 199, 16–22. [Google Scholar] [CrossRef]
- Alici, E.; Chrobok, M.; Lund, J.; Ahmadi, T.; Khan, I.; Duru, A.D.; Nahi, H. Re-challenging with anti-CD38 monotherapy in triple-refractory multiple myeloma patients is a feasible and safe approach. Br. J. Haematol. 2016, 174, 473–477. [Google Scholar] [CrossRef]
- Krejcik, J.; Frerichs, K.A.; Nijhof, I.S.; van Kessel, B.; van Velzen, J.F.; Bloem, A.C.; Broekmans, M.E.C.; Zweegman, S.; van Meerloo, J.; Musters, R.J.P.; et al. Monocytes and Granulocytes Reduce CD38 Expression Levels on Myeloma Cells in Patients Treated with Daratumumab. Clin. Cancer Res. Off. J. Am. Assoc. Cancer Res. 2017, 23, 7498–7511. [Google Scholar] [CrossRef]
- Garcia-Guerrero, E.; Gogishvili, T.; Danhof, S.; Schreder, M.; Pallaud, C.; Perez-Simon, J.A.; Einsele, H.; Hudecek, M. Panobinostat induces CD38 upregulation and augments the antimyeloma efficacy of daratumumab. Blood 2017, 129, 3386–3388. [Google Scholar] [CrossRef]
- Moreno, L.; Perez, C.; Zabaleta, A.; Manrique, I.; Alignani, D.; Ajona, D.; Blanco, L.; Lasa, M.; Maiso, P.; Rodriguez, I.; et al. The Mechanism of Action of the Anti-CD38 Monoclonal Antibody Isatuximab in Multiple Myeloma. Clin. Cancer Res. Off. J. Am. Assoc. Cancer Res. 2019, 25, 3176–3187. [Google Scholar] [CrossRef]
- Kishimoto, H.; Hoshino, S.; Ohori, M.; Kontani, K.; Nishina, H.; Suzawa, M.; Kato, S.; Katada, T. Molecular mechanism of human CD38 gene expression by retinoic acid. Identification of retinoic acid response element in the first intron. J. Biol. Chem. 1998, 273, 15429–15434. [Google Scholar] [CrossRef]
- Uruno, A.; Noguchi, N.; Matsuda, K.; Nata, K.; Yoshikawa, T.; Chikamatsu, Y.; Kagechika, H.; Harigae, H.; Ito, S.; Okamoto, H.; et al. All-trans retinoic acid and a novel synthetic retinoid tamibarotene (Am80) differentially regulate CD38 expression in human leukemia HL-60 cells: Possible involvement of protein kinase C-delta. J. Leukoc. Biol. 2011, 90, 235–247. [Google Scholar] [CrossRef]
- Lewandowski, D.; Linassier, C.; Iochmann, S.; Degenne, M.; Domenech, J.; Colombat, P.; Binet, C.; Herault, O. Phosphatidylinositol 3-kinases are involved in the all-trans retinoic acid-induced upregulation of CD38 antigen on human haematopoietic cells. Br. J. Haematol. 2002, 118, 535–544. [Google Scholar] [CrossRef]
- Quach, H.; Ritchie, D.; Stewart, A.K.; Neeson, P.; Harrison, S.; Smyth, M.J.; Prince, H.M. Mechanism of action of immunomodulatory drugs (IMiDS) in multiple myeloma. Leukemia 2010, 24, 22–32. [Google Scholar] [CrossRef]
- Bolzoni, M.; Storti, P.; Bonomini, S.; Todoerti, K.; Guasco, D.; Toscani, D.; Agnelli, L.; Neri, A.; Rizzoli, V.; Giuliani, N. Immunomodulatory drugs lenalidomide and pomalidomide inhibit multiple myeloma-induced osteoclast formation and the RANKL/OPG ratio in the myeloma microenvironment targeting the expression of adhesion molecules. Exp. Hematol. 2013, 41, 387–397 e381. [Google Scholar] [CrossRef] [PubMed]
- Fedele, P.L.; Willis, S.N.; Liao, Y.; Low, M.S.; Rautela, J.; Segal, D.H.; Gong, J.N.; Huntington, N.D.; Shi, W.; Huang, D.C.S.; et al. IMiDs prime myeloma cells for daratumumab-mediated cytotoxicity through loss of Ikaros and Aiolos. Blood 2018, 132, 2166–2178. [Google Scholar] [CrossRef] [PubMed]
- Pratt, G. Panobinostat plus bortezomib and dexamethasone for relapsed myeloma. Lancet Haematol. 2016, 3, e498–e499. [Google Scholar] [CrossRef]
- Lai, A.Y.; Wade, P.A. Cancer biology and NuRD: A multifaceted chromatin remodelling complex. Nat. Rev. Cancer 2011, 11, 588–596. [Google Scholar] [CrossRef]
- Bat-Erdene, A.; Nakamura, S.; Oda, A.; Iwasa, M.; Teramachi, J.; Ashtar, M.; Harada, T.; Miki, H.; Tenshin, H.; Hiasa, M.; et al. Class 1 HDAC and HDAC6 inhibition inversely regulates CD38 induction in myeloma cells via interferon-alpha and ATRA. Br. J. Haematol. 2019, 185, 969–974. [Google Scholar] [CrossRef]
- Ferrero, E.; Malavasi, F. Human CD38, a leukocyte receptor and ectoenzyme, is a member of a novel eukaryotic gene family of nicotinamide adenine dinucleotide+-converting enzymes: Extensive structural homology with the genes for murine bone marrow stromal cell antigen 1 and aplysian ADP-ribosyl cyclase. J. Immunol. 1997, 159, 3858–3865. [Google Scholar]
- Bauvois, B.; Durant, L.; Laboureau, J.; Barthelemy, E.; Rouillard, D.; Boulla, G.; Deterre, P. Upregulation of CD38 gene expression in leukemic B cells by interferon types I and II. J. Interferon Cytokine Res. Off. J. Int. Soc. Interferon Cytokine Res. 1999, 19, 1059–1066. [Google Scholar] [CrossRef]
- Choudhry, P.; Mariano, M.C.; Geng, H.; Martin, T.G., 3rd; Wolf, J.L.; Wong, S.W.; Shah, N.; Wiita, A.P. DNA methyltransferase inhibitors upregulate CD38 protein expression and enhance daratumumab efficacy in multiple myeloma. Leukemia 2019. [Google Scholar] [CrossRef]
- Facon, T.; Kumar, S.; Plesner, T.; Orlowski, R.Z.; Moreau, P.; Bahlis, N.; Basu, S.; Nahi, H.; Hulin, C.; Quach, H.; et al. Daratumumab plus Lenalidomide and Dexamethasone for Untreated Myeloma. New Engl. J. Med. 2019, 380, 2104–2115. [Google Scholar] [CrossRef]
- Chari, A.; Suvannasankha, A.; Fay, J.W.; Arnulf, B.; Kaufman, J.L.; Ifthikharuddin, J.J.; Weiss, B.M.; Krishnan, A.; Lentzsch, S.; Comenzo, R.; et al. Daratumumab plus pomalidomide and dexamethasone in relapsed and/or refractory multiple myeloma. Blood 2017, 130, 974–981. [Google Scholar] [CrossRef]
- Martin, T.; Baz, R.; Benson, D.M.; Lendvai, N.; Wolf, J.; Munster, P.; Lesokhin, A.M.; Wack, C.; Charpentier, E.; Campana, F.; et al. A phase 1b study of isatuximab plus lenalidomide and dexamethasone for relapsed/refractory multiple myeloma. Blood 2017, 129, 3294–3303. [Google Scholar] [CrossRef] [PubMed]
- Mikhael, J.; Richardson, P.; Usmani, S.Z.; Raje, N.; Bensinger, W.; Karanes, C.; Campana, F.; Kanagavel, D.; Dubin, F.; Liu, Q.; et al. A phase 1b study of isatuximab plus pomalidomide/dexamethasone in relapsed/refractory multiple myeloma. Blood 2019, 134, 123–133. [Google Scholar] [CrossRef] [PubMed]
| Drug Combination | Trial Name (NCT number) [Ref.] | Phase | Number of Patients | Primary Outcome | Results Reported | AEs (Grade 3–4) |
|---|---|---|---|---|---|---|
| DARA/ Lenalidomide | 54767414MMY3008 (NCT02252172) [59] | III | 737 | PFS | PFS 70.6% at 30 mos | Neutropenia 50%, lymphopenia 15% |
| DARA/ Lenalidomide | Pollux Study (NCT02076009) [7] | III | 569 | PFS | ORR 92.9% vs 76.4% Median PFS nr vs 17.5 months (HR 0.44) | Neutropenia (54%), anemia (15.5%), pneumonia (12%) |
| Isatuximab/ Lenalidomide | A Phase 1b Study of SAR650984 (Anti-CD38 mAb) in Combination with Len and Dex for the Treatment of RRMM (NCT01749969) [61] | I | 57 | MTD of the combination | ORR 56% Median PFS 8.5 months | Neutropenia (60%), lymphopenia (58%) |
| DARA/Pomalidomide | 54767414MMY1001 (NCT01998971) [60] | Ib | 103 | MTD of the combination | ORR 60% Median PFS 8.8 months | Neutropenia (77%), anemia (28%), thrombocytopenia (19%) |
| Isatuximab/ Pomalidomide | TCD14079 (NCT02283775) [62] | Ib | 45 | MTD of the combination | ORR 62% Median PFS 17.6 months | (AEs all grade) Fatigue (62%), upper respiratory tract infection (42%) |
| DARA/ ATRA | A Phase 1 and Phase 2 Study of DARA in Combination with ATRA in RRMM (NCT02751255) | I/II | 60 | 1) MTD 2) ORR 3) RDL | No result posted | No result posted |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Costa, F.; Dalla Palma, B.; Giuliani, N. CD38 Expression by Myeloma Cells and Its Role in the Context of Bone Marrow Microenvironment: Modulation by Therapeutic Agents. Cells 2019, 8, 1632. https://doi.org/10.3390/cells8121632
Costa F, Dalla Palma B, Giuliani N. CD38 Expression by Myeloma Cells and Its Role in the Context of Bone Marrow Microenvironment: Modulation by Therapeutic Agents. Cells. 2019; 8(12):1632. https://doi.org/10.3390/cells8121632
Chicago/Turabian StyleCosta, Federica, Benedetta Dalla Palma, and Nicola Giuliani. 2019. "CD38 Expression by Myeloma Cells and Its Role in the Context of Bone Marrow Microenvironment: Modulation by Therapeutic Agents" Cells 8, no. 12: 1632. https://doi.org/10.3390/cells8121632
APA StyleCosta, F., Dalla Palma, B., & Giuliani, N. (2019). CD38 Expression by Myeloma Cells and Its Role in the Context of Bone Marrow Microenvironment: Modulation by Therapeutic Agents. Cells, 8(12), 1632. https://doi.org/10.3390/cells8121632





