Targeting Cyclic AMP Signalling in Hepatocellular Carcinoma
Abstract
1. Introduction
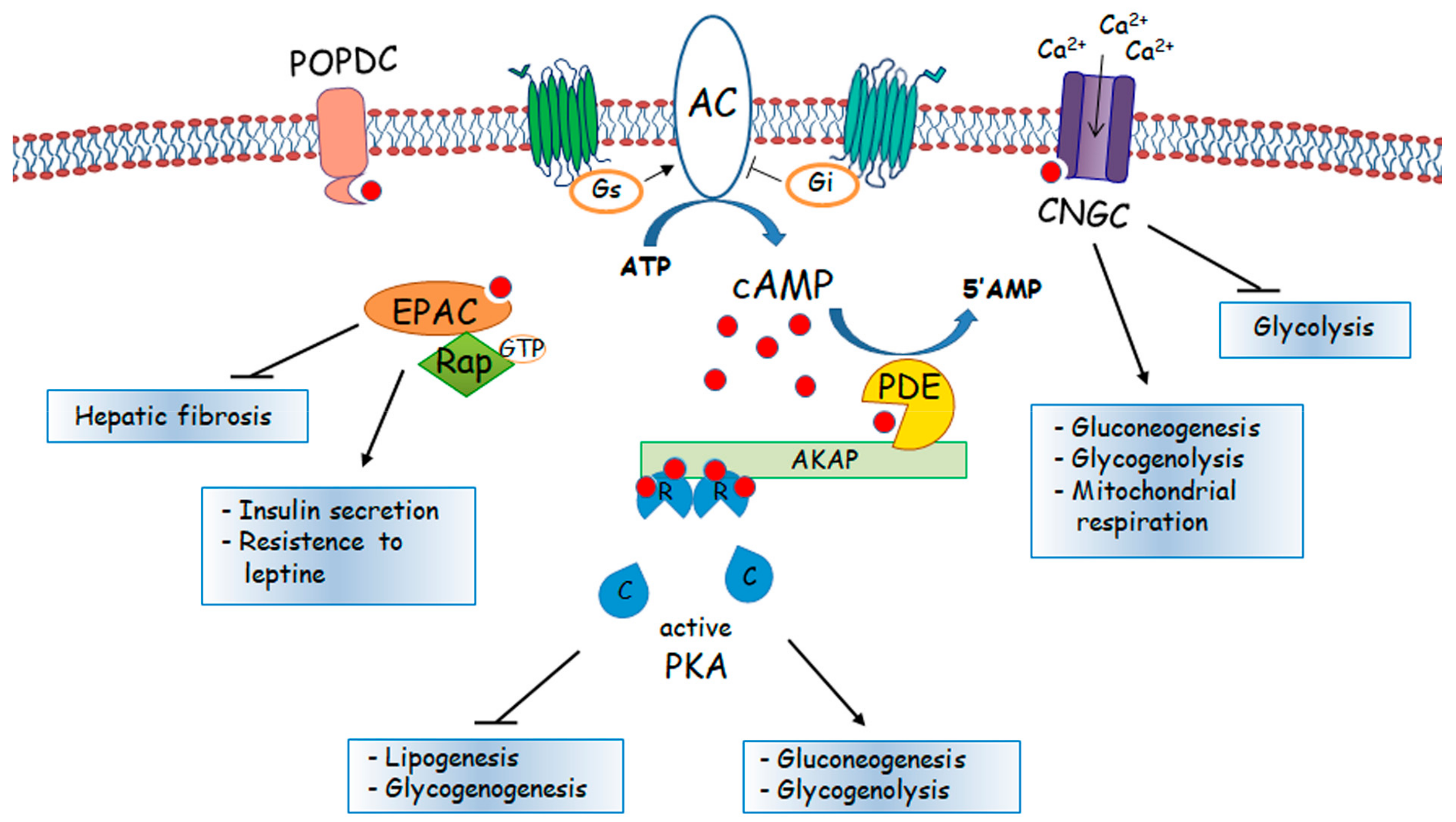
2. Physiologic Effectors of cAMP in Hepatocytes
3. Pharmacological Modulators of the cAMP Pathway
4. Role of cAMP Signalling in Non-Hepatic Tumours
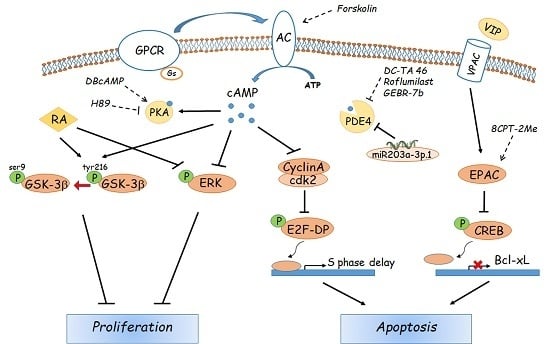
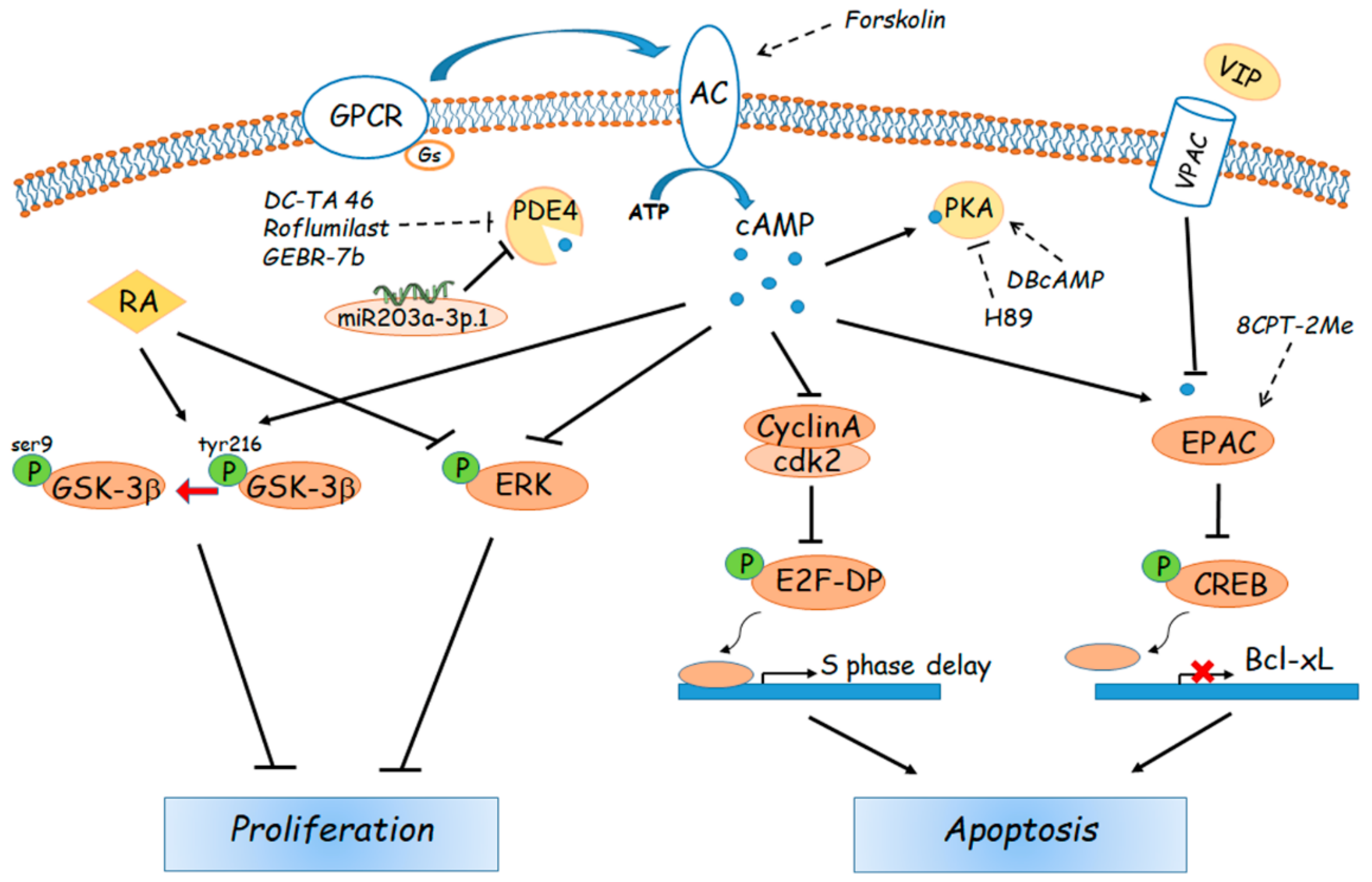
5. Role of Cyclic AMP in Hepatocellular Carcinoma
6. Concluding Remarks
Funding
Conflicts of Interest
References
- Jemal, A.; Bray, F.; Center, M.M.; Ferlay, J.; Ward, E.; Forman, D. Global cancer statistics. CA Cancer J. Clin. 2011, 61, 69–90. [Google Scholar] [CrossRef] [PubMed]
- Forner, A.; Llovet, J.M.; Bruix, J. Hepatocellular carcinoma. Lancet 2012, 379, 1245–1255. [Google Scholar] [CrossRef]
- Couri, T.; Pillai, A. Goals and targets for personalized therapy for HCC. Hepatol. Int. 2019, 13, 125–137. [Google Scholar] [CrossRef] [PubMed]
- Tang, Z.Y.; Ye, S.L.; Liu, Y.K.; Qin, L.X.; Sun, H.C.; Ye, Q.H.; Wang, L.; Zhou, J.; Qiu, S.J.; Li, Y.; et al. A decade’s studies on metastasis of hepatocellular carcinoma. J. Cancer Res. Clin. Oncol. 2004, 130, 187–196. [Google Scholar] [CrossRef] [PubMed]
- Blume, H.E. Molecular therapy and prevention of hepatocellular carcinoma. Hepatobiliary Pancreat. Dis. Int. 2003, 2, 11–22. [Google Scholar]
- Mittal, S.; El-Serag, H.B. Epidemiology of hepatocellular carcinoma: Consider the population. J. Clin. Gastroenterol. 2013, 47, S2–S6. [Google Scholar] [CrossRef]
- Zhu, A.X.; Duda, D.G.; Sahani, D.V.; Jain, R.K. HCC and angiogenesis: Possible targets and future directions. Nat. Rev. Clin. Oncol. 2011, 8, 292–301. [Google Scholar] [CrossRef]
- Huang, X.Y.; Huang, Z.L.; Zhang, P.B.; Huang, X.Y.; Huang, J.; Wang, H.C.; Xu, B.; Zhou, J.; Tang, Z.Y. CircRNA-100338 Is Associated With mTOR Signaling Pathway and Poor Prognosis in Hepatocellular Carcinoma. Front. Oncol. 2019, 9, 392. [Google Scholar] [CrossRef]
- Kim, D.W.; Talati, C.; Kim, R. Hepatocellular carcinoma (HCC): Beyond sorafenib-chemotherapy. J. Gastrointest. Oncol. 2017, 8, 256–265. [Google Scholar] [CrossRef]
- Riggle, K.M.; Turnham, R.; Scott, J.D.; Yeung, R.S.; Riehl, K.J. Fibrolamellar Hepatocellular Carcinoma: Mechanistic Distinction From Adult Hepatocellular Carcinoma. Pediatr. Blood Cancer 2016, 63, 1163–1167. [Google Scholar] [CrossRef]
- Turnham, R.E.; Smith, F.D.; Kenerson, H.L.; Omar, M.H.; Golkowski, M.; Garcia, I.; Bauer, R.; Lau, H.T.; Sullivan, K.M.; Langeberg, L.K.; et al. An acquired scaffolding function of the DNAJ-PKAc fusion contributes to oncogenic signaling in fibrolamellar carcinoma. eLife 2019, 8, e44187. [Google Scholar] [CrossRef] [PubMed]
- Savai, R.; Pullamsetti, S.S.; Banat, G.A.; Weissmann, N.; Ghofrani, H.A.; Grimminger, F.; Schermuly, R.T. Targeting cancer with phosphodiesterase inhibitors. Expert. Opin. Investig. Drugs 2010, 19, 117–131. [Google Scholar] [CrossRef] [PubMed]
- Massimi, M.; Cardarelli, S.; Galli, F.; Giardi, M.F.; Ragusa, F.; Panera, N.; Cinque, B.; Cifone, M.G.; Biagioni, S.; Giorgi, M. Increase of intracellular cyclic AMP by PDE4 inhibitors affects HepG2 cell cycle progression and survival. J. Cell. Biochem. 2017, 118, 1401–1411. [Google Scholar] [CrossRef] [PubMed]
- Peng, T.; Gong, J.; Jin, Y.; Zhou, Y.; Tong, R.; Wei, X.; Bai, L.; Shi, J. Inhibitors of phosphodiesterase as cancer therapeutics. Eur. J. Med. Chem. 2018, 150, 742–756. [Google Scholar] [CrossRef]
- Ragusa, F.; Panera, N.; Cardarelli, S.; Caioni, G.; Alisi, A.; Giorgi, M.; Massimi, M. Type 4 phosphodiesterases: A possible role in hepatocyte transformation. Eur. J. Histochem. 2018, 62, 7. [Google Scholar]
- Lugnier, C. Cyclic nucleotide phosphodiesterase (PDE) superfamily: A new target for the development of specific therapeutic agents. Pharmacol. Ther. 2006, 109, 366–398. [Google Scholar] [CrossRef]
- Conti, M.; Beavo, J. Biochemistry and physiology of cyclic nucleotide phosphodiesterases: Essential components in cyclic nucleotide signaling. Annu. Rev. Biochem. 2007, 76, 481–511. [Google Scholar] [CrossRef]
- Zippin, J.H.; Chen, Y.; Straub, S.G.; Hess, K.C.; Diaz, A.; Lee, D.; Tso, P.; Holz, G.G.; Sharp, G.W.; Levin, L.R.; et al. CO2/HCO3(-)- and calcium-regulated soluble adenylyl cyclase as a physiological ATP sensor. J. Biol. Chem. 2013, 288, 33283–33291. [Google Scholar] [CrossRef]
- Reggi, E.; Diviani, D. The role of A-kinase anchoring proteins in cancer development. Cell Signal. 2017, 40, 143–155. [Google Scholar] [CrossRef]
- Pilkis, S.J.; Claus, T.H.; el-Maghrabi, M.R. The role of cyclic AMP in rapid and long-term regulation of gluconeogenesis and glycolysis. Adv. Second Messenger Phosphoprot. Res. 1988, 22, 175–191. [Google Scholar]
- Jitrapakdee, S. Transcription factors and coactivators controlling nutrient and hormonal regulation of hepatic gluconeogenesis. Int. J. Biochem. Cell Biol. 2012, 44, 33–45. [Google Scholar] [CrossRef] [PubMed]
- Lent, B.A.; Kim, K.H. Phosphorylation and activation of acetyl-coenzyme A carboxylase kinase by the catalytic subunit of cyclic AMP-dependent protein kinase. Arch. Biochem. Biophys. 1983, 225, 972–978. [Google Scholar] [CrossRef]
- De Rooij, J.; Zwartkruis, F.J.; Verheijen, M.H.; Cool, R.H.; Nijman, S.M.; Wittinghofer, A.; Bos, J.L. Epac is a Rap1 guanine-nucleotide-exchange factor directly activated by cyclic AMP. Nature 1998, 396, 474–477. [Google Scholar] [CrossRef] [PubMed]
- Kawasaki, H.; Springett, G.M.; Mochizuki, N.; Toki, S.; Nakaya, M.; Matsuda, M.; Housman, D.E.; Graybiel, A.M. A family of cAMP-binding proteins that directly activate Rap1. Science 1998, 282, 2275–2279. [Google Scholar] [CrossRef] [PubMed]
- Fukuda, M.; Williams, K.W.; Gautron, L.; Elmquist, J.K. Induction of leptin resistance by activation of cAMP-Epac signaling. Cell Metab. 2011, 13, 331–339. [Google Scholar] [CrossRef]
- Yan, J.; Mei, F.C.; Cheng, H.; Lao, D.H.; Hu, Y.; Wei, J.; Patrikeev, I.; Hao, D.; Stutz, S.J.; Dineley, K.T.; et al. Enhanced leptin sensitivity, reduced adiposity, and improved glucose homeostasis in mice lacking exchange protein directly activated by cyclic AMP isoform 1. Mol. Cell Biol. 2013, 33, 918–926. [Google Scholar] [CrossRef]
- Almahariq, M.; Mei, F.C.; Cheng, X. Cyclic AMP sensor EPAC proteins and energy homeostasis. Trends Endocrinol. Metab. 2014, 25, 60–71. [Google Scholar] [CrossRef]
- Insel, P.A.; Murray, F.; Yokoyama, U.; Romano, S.; Yun, H.; Brown, L.; Snead, A.; Lu, D.; Aroonsakool, N. cAMP and Epac in the regulation of tissue fibrosis. Br. J. Pharm. 2012, 166, 447–456. [Google Scholar] [CrossRef]
- Schippers, M.; Beljaars, L.; Post, E.; Lotersztajn, S.; Reker-Smit, C.; Han, B.; Munoz-Llancao, P.; Schmidt, M.; Poelstra, K. Upregulation of Epac-1 in hepatic stellate cells by prostaglandin E2 in liver fibrosis is associated with reduced fibrogenesis. J. Pharm. Exp. 2017, 363, 126–135. [Google Scholar] [CrossRef]
- Rehmann, H.; Arias-Palomo, E.; Hadders, M.A.; Schwede, F.; Llorca, O.; Bos, J.L. Structure of Epac2 in complex with a cyclic AMP analogue and RAP1B. Nature 2008, 455, 124–127. [Google Scholar] [CrossRef]
- Tsalkova, T.; Blumenthal, D.K.; Mei, F.C.; White, M.A.; Cheng, X. Mechanism of Epac activation: Structural and functional analyses of Epac2 hinge mutants with constitutive and reduced activities. J. Biol. Chem. 2009, 284, 23644–23651. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Tsalkova, T.; White, M.A.; Mei, F.C.; Liu, T.; Wang, D.; Woods, V.L., Jr.; Cheng, X. Mechanism of intracellular cAMP sensor Epac2 activation: cAMP-induced conformational changes identified by amide hydrogen/deuterium exchange mass spectrometry (DXMS). J. Biol. Chem. 2011, 286, 17889–17897. [Google Scholar] [CrossRef] [PubMed]
- Consonni, S.V.; Gloerich, M.; Spanjaard, E.; Bos, J.L. cAMP regulates DEP domain-mediated binding of the guanine nucleotide exchange factor Epac1 to phosphatidic acid at the plasma membrane. Proc. Natl. Acad. Sci. USA 2012, 109, 3814–3819. [Google Scholar] [CrossRef] [PubMed]
- Fesenko, E.E.; Kolesnikov, S.S.; Lyubarsky, A.L. Induction by cyclic GMP of cationic conductance in plasma membrane of retinal rod outer segment. Nature 1985, 313, 310–313. [Google Scholar] [CrossRef]
- Kaupp, U.B.; Seifert, R. Cyclic nucleotide-gated ion channels. Physiol. Rev. 2002, 82, 769–824. [Google Scholar] [CrossRef]
- Schindler, R.F.; Brand, T. The Popeye domain containing protein family—A novel class of cAMP effectors with important functions in multiple tissues. Prog. Biophys. Mol. Biol. 2016, 120, 28–36. [Google Scholar] [CrossRef]
- Feng, L.; Subbaraya, I.; Yamamoto, N.S.; Baehr, W.; Kraus-Friedmann, N. Expression of photoreceptor cyclic nucleotide-gated cation channel alpha subunit (CNGC) in the liver and skeletal muscle. FEBS Lett. 1996, 395, 77–81. [Google Scholar] [CrossRef]
- Cook, N.J.; Molday, L.; Reid, D.; Kaupp, U.B.; Molday, R.S. The cGMP-gated channel of bovine rod photoreceptors is localized exclusively in the plasma membrane. J. Biol. Chem. 1989, 264, 6996–6999. [Google Scholar]
- Gevers, W.; Krebs, H.A. The effects of adenine nucleotides on carbohydrate metabolism in pigeon-liver homogenates. Biochem. J. 1996, 98, 720–735. [Google Scholar]
- Khoo, J.C.; Steinberg, D. Stimulation of rat liver phosphorylase kinase by micromolar concentration of Ca2+. FEBS Lett. 1975, 57, 68–72. [Google Scholar] [CrossRef]
- Ozawa, E.; Ebashi, S. Requirement of Ca ion for the stimulating effect of cyclic 3’,5’-AMP on muscle phosphorylase b kinase. J. Biochem. 1967, 62, 285–286. [Google Scholar] [CrossRef] [PubMed]
- Robb-Gaspers, L.D.; Burnett, P.; Rutter, G.A.; Denton, R.M.; Rizzuto, R.; Thomas, A.P. Integrating cytosolic calcium signals into mitochondrial metabolic responses. EMBO J. 1998, 17, 4987–5000. [Google Scholar] [CrossRef] [PubMed]
- Friedmann, N. Activation of protein kinase(s) by glucagon and cyclic-AMP in the rat liver: Relationship to metabolic effects. Biochim. Biophys. Acta 1976, 428, 495–508. [Google Scholar] [CrossRef]
- Matsui, S.; Murakami, E.; Takekoshi, N.; Emoto, J.; Matoba, M. Efficacy of dibutyryl cyclic AMP in heart failure unresponsive to catecholamines. Clin. Ther. 1987, 9, 488–499. [Google Scholar]
- Asai, J.; Takenaka, H.; Katoh, N.; Kishimoto, S. Dibutyryl cAMP influences endothelial progenitor cell recruitment during wound neovascularization. J. Invest. Derm. 2006, 126, 1159–1167. [Google Scholar] [CrossRef]
- Rundfeldt, C.; Steckel, H.; Sörensen, T.; Wlaź, P. The stable cyclic adenosine monophosphate analogue, dibutyryl cyclo-adenosine monophosphate (bucladesine), is active in a model of acute skin inflammation. Arch. Derm. Res. 2012, 304, 313–317. [Google Scholar] [CrossRef][Green Version]
- Lu, D.; Aroonsakool, N.; Yokoyama, U.; Patel, H.H.; Insel, P.A. Increase in cellular cyclic AMP concentrations reverses the profibrogenic phenotype of cardiac myofibroblasts: A novel therapeutic approach for cardiac fibrosis. Mol. Pharm. 2013, 84, 787–793. [Google Scholar] [CrossRef]
- El-Agroudy, N.N.; El-Naga, R.N.; El-Razeq, R.A.; El-Demerdash, E. Forskolin, a hedgehog signalling inhibitor, attenuates carbon tetrachloride-induced liver fibrosis in rats. Br. J. Pharm. 2016, 173, 3248–3260. [Google Scholar] [CrossRef]
- Vetrugno, M.; Uva, M.G.; Russo, V.; Iester, M.; Ciancaglini, M.; Brusini, P.; Centofanti, M.; Rossetti, L.M. Oral administration of forskolin and rutin contributes to intraocular pressure control in primary open angle glaucoma patients under maximum tolerated medical therapy. J. Ocul Pharm. 2012, 28, 536–541. [Google Scholar] [CrossRef]
- Rios-Hoyo, A.; Gutierrez-Salmean, G. New dietary supplements for obesity: What we currently know. Curr. Obes. Rep. 2016, 5, 262–270. [Google Scholar] [CrossRef]
- Sapio, L.; Gallo, M.; Illiano, M.; Chiosi, E.; Naviglio, D.; Spina, A.; Naviglio, S. The natural cAMP elevating compound Forskolin in Cancer therapy: Is it time? J. Cell Physiol 2017, 232, 922–927. [Google Scholar] [CrossRef] [PubMed]
- Yamanaka, H.; Oue, T.; Uehara, S.; Fukuzawa, M. Forskolin, a Hedgehog signal inhibitor, inhibits cell proliferation and induces apoptosis in pediatric tumor cell lines. Mol. Med. Rep. 2010, 3, 133–139. [Google Scholar] [PubMed]
- Makinodan, E.; Marneros, A.G. Protein kinase A activation inhibits oncogenic Sonic hedgehog signalling and suppresses basal cell carcinoma of the skin. Exp. Derm. 2012, 21, 847–852. [Google Scholar] [CrossRef] [PubMed]
- Cho, I.J.; Woo, N.R.; Shin, I.C.; Kim, S.G. H89, an inhibitor of PKA and MSK, inhibits cyclic-AMP response element binding protein-mediated MAPK phosphatase-1 induction by lipopolysaccharide. Inflamm. Res. 2009, 58, 863–872. [Google Scholar] [CrossRef] [PubMed]
- Diaz-Muñoz, M.D.; Osma-García, I.C.; Fresno, M.; Iñiguez, M.A. Involvement of PGE2 and the cAMP signalling pathway in the up-regulation of COX-2 and mPGES-1 expression in LPS-activated macrophages. Biochem. J. 2012, 443, 451–461. [Google Scholar] [CrossRef] [PubMed]
- Yu, M.; Liu, T.; Chen, Y.; Li, Y.; Li, W. Combination therapy with protein kinase inhibitor H89 and Tetrandrine elicits enhanced synergistic antitumor efficacy. J. Exp. Clin. Cancer Res. 2018, 37, 114. [Google Scholar] [CrossRef]
- Ma, J.; Chen, M.; Xia, S.K.; Shu, W.; Guo, Y.; Wang, Y.H.; Xu, Y.; Bai, X.M.; Zhang, L.; Zhang, H.; et al. Prostaglandin E2 promotes liver cancer cell growth by the upregulation of FUSE-binding protein 1 expression. Int. J. Oncol. 2013, 42, 1093–1104. [Google Scholar] [CrossRef]
- Enserink, J.M.; Christensen, A.E.; de Rooij, J.; van Triest, M.; Schwede, F.; Genieser, H.G.; Døskeland, S.O.; Blank, J.L.; Bos, J.L. A novel Epac-specific cAMP analogue demonstrates independent regulation of Rap1 and ERK. Nat. Cell Biol. 2002, 4, 901–906. [Google Scholar] [CrossRef]
- Shariati, B.; Thompson, E.L.; Nicol, G.D.; Vasko, M.R. Epac activation sensitizes rat sensory neurons through activation of Ras. Mol. Cell Neurosci. 2016, 70, 54–67. [Google Scholar] [CrossRef]
- Tsalkova, T.; Mei, F.C.; Li, S.; Chepurny, O.G.; Leech, C.A.; Liu, T.; Holz, G.G.; Woods, V.L., Jr.; Cheng, X. Isoform-specific antagonists of exchange proteins directly activated by cAMP. Proc. Natl. Acad. Sci. USA 2012, 109, 18613–18618. [Google Scholar] [CrossRef]
- Almahariq, M.; Chao, C.; Mei, F.C.; Hellmich, M.R.; Patrikeev, I.; Motamedi, M.; Cheng, X. Pharmacological inhibition and genetic knockdown of exchange protein directly activated by cAMP 1 reduce pancreatic cancer metastasis in vivo. Mol. Pharm. 2015, 87, 142–149. [Google Scholar] [CrossRef] [PubMed]
- Fujishige, K.; Kotera, J.; Michibata, H.; Yuasa, K.; Takebayashi, S.; Okumura, K.; Omori, K. Cloning and characterization of a novel human phosphodiesterase that hydrolyzes both cAMP and cGMP (PDE10A). J. Biol. Chem. 1999, 274, 18438–18445. [Google Scholar] [CrossRef] [PubMed]
- Gross-Langenhoff, M.; Hofbauer, K.; Weber, J.; Schultz, A.; Schultz, J.E. cAMP is a ligand for the tandem GAF domain of human phosphodiesterase 10 and cGMP for the tandem GAF domain of phosphodiesterase 11. J. Biol. Chem. 2006, 281, 2841–2846. [Google Scholar] [CrossRef] [PubMed]
- Keravis, T.; Lugnier, C. Cyclic nucleotide phosphodiesterases (PDE) and peptide motifs. Curr. Pharm. Des. 2010, 16, 1114–1125. [Google Scholar] [CrossRef] [PubMed]
- Houslay, M.D.; Baillie, G.S.; Maurice, D.H. cAMP-Specific phosphodiesterase-4 enzymes in the cardiovascular system: A molecular toolbox for generating compartmentalized cAMP signaling. Circ. Res. 2007, 100, 950–966. [Google Scholar] [CrossRef]
- Keravis, T.; Lugnier, C. Cyclic nucleotide phosphodiesterase (PDE) isozymes as target of the intracellular signlling network: Benefits of PDE inhibitors in various diseases and perspectives for future therapeutic developments. Br. J. Pharm. 2012, 165, 1288–1305. [Google Scholar] [CrossRef]
- Marivet, M.C.; Bourguignon, J.J.; Lugnier, C.; Mann, A.; Stoclet, J.C.; Wermuth, C.G. Inhibition of cyclic adenosine-3’,5’-monophosphate phosphodiesterase from vascular smooth muscle by rolipram analogues. J. Med. Chem 1989, 32, 1450–1457. [Google Scholar] [CrossRef]
- Drees, M.; Zimmermann, R.; Eisembrand, G. 3′,5′-cyclic nucleotide phosphodiesterase in tumor cells as potential target for tumor growth inhibition. Cancer Res. 1993, 53, 3058–3061. [Google Scholar]
- Spina, D. PDE4 inhibitors: Current status. Br. J. Pharm. 2008, 155, 308–315. [Google Scholar] [CrossRef]
- Brullo, C.; Massa, M.; Rocca, M.; Rotolo, C.; Guariento, S.; Rivera, D.; Ricciarelli, R.; Fedele, E.; Fossa, P.; Bruno, O. Synthesis, biological evaluation, and molecular modeling of new 3-(cyclopentyloxy)-4-methoxybenzaldehyde O-(2-(2,6-dimethylmorpholino)-2-oxoethyl) Oxime (GEBR-7b) related phosphodiesterase 4D (PDE4D) inhibitors. J. Med. Chem. 2014, 57, 7061–7072. [Google Scholar] [CrossRef]
- Bruno, O.; Romussi, A.; Spallarossa, A.; Brullo, C.; Schenone, S.; Bondavalli, F.; Vanthuyne, N.; Roussel, C. New selective phosphodiesterase 4D inhibitors differently acting on long, short, and supershort isoforms. J. Med. Chem. 2009, 52, 6546–6557. [Google Scholar] [CrossRef] [PubMed]
- Bruno, O.; Fedele, E.; Prickaerts, J.; Parker, L.A.; Canepa, E.; Brullo, C.; Cavallero, A.; Gardella, E.; Balbi, A.; Domenicotti, C.; et al. GEBR-7b, a novel PDE4D selective inhibitor that improves memory in rodents at non-emetic doses. Br. J. Pharm. 2011, 164, 2054–2063. [Google Scholar] [CrossRef] [PubMed]
- Paterniti, I.; Mazzon, E.; Gil, C.; Impellizzeri, D.; Palomo, V.; Redondo, M.; Perez, D.I.; Esposito, E.; Martinez, A.; Cuzzocrea, S. PDE 7 inhibitors: New potential drugs for the therapy of spinal cord injury. PLoS ONE 2011, 6, e15937. [Google Scholar] [CrossRef] [PubMed]
- Murata, T.; Shimizu, K.; Narita, M.; Manganiello, V.C.; Tagawa, T. Characterization of phosphodiesterase 3 in human malignant melanoma cell line. Anticancer Res. 2002, 22, 3171–3174. [Google Scholar] [PubMed]
- Yamanaka, Y.; Mammoto, T.; Kirita, T.; Mukai, M.; Mashimo, T.; Sugimura, M.; Kishi, Y.; Nakamura, H. Epinephrine inhibits invasion of oral squamous carcinoma cells by modulating intracellular cAMP. Cancer Lett. 2002, 176, 143–148. [Google Scholar] [CrossRef]
- Baljinnyam, E.; Iwatsubo, K.; Kurotani, R.; Wang, X.; Ulucan, C.; Iwatsubo, M.; Lagunoff, D.; Ishikawa, Y. Epac increases melanoma cell migration by a heparan sulfate-related mechanism. Am. J. Physiol. Cell Physiol. 2009, 297, 802–813. [Google Scholar] [CrossRef]
- Baljinnyam, E.; De Lorenzo, M.S.; Xie, L.H.; Iwatsubo, M.; Chen, S.; Goydos, J.S.; Nowycky, M.C.; Iwatsubo, K. Exchange protein directly activated by cyclic AMP increases melanoma cell migration by a Ca2+-dependent mechanism. Cancer Res. 2010, 70, 5607–5617. [Google Scholar] [CrossRef]
- Robichaux, W.G.; Cheng, X. Intracellular cAMP Sensor EPAC: Physiology, Pathophysiology, and Therapeutics Development. Physiol. Rev. 2018, 98, 919–1053. [Google Scholar] [CrossRef]
- Misra, U.K.; Pizzo, S.V. Epac1-induced cellular proliferation in prostate cancer cells is mediated by B-Raf/ERK and mTOR signaling cascades. J. Cell. Biochem. 2009, 108, 998–1011. [Google Scholar] [CrossRef]
- Grandoch, M.; Rose, A.; ter Braak, M.; Jendrossek, V.; Rübben, H.; Fischer, J.W.; Schmidt, M.; Weber, A.A. Epac inhibits migration and proliferation of human prostate carcinoma cells. Br. J. Cancer 2009, 101, 2038–2042. [Google Scholar] [CrossRef]
- Lim, J.A.; Juhnn, Y.S. Isoproterenol increases histone deacetylase 6 expression and cell migration by inhibiting ERK signaling via PKA and Epac pathways in human lung cancer cells. Exp. Mol. Med. 2016, 48, e204. [Google Scholar] [CrossRef] [PubMed]
- Park, J.Y.; Juhnn, Y.S. cAMP signaling increases histone deacetylase 8 expression via the Epac2-Rap1A-Akt pathway in H1299 lung cancer cells. Exp. Mol. Med. 2017, 49, e297. [Google Scholar] [CrossRef] [PubMed]
- Vitali, E.; Cambiaghi, V.; Spada, A.; Tresoldi, A.; Zerbi, A.; Peverelli, E.; Carnaghi, C.; Mantovani, G.; Lania, A.G. cAMP effects in neuroendocrine tumors: The role of Epac and PKA in cell proliferation and adhesion. Exp. Cell Res. 2015, 339, 241–251. [Google Scholar] [CrossRef] [PubMed]
- Moon, E.Y.; Lee, G.H.; Lee, M.S.; Kim, H.M.; Lee, J.W. Phosphodiesterase inhibitors control A172 human glioblastoma cell death through cAMP-mediated activation of protein kinase A and Epac1/Rap1 pathways. Life Sci. 2012, 90, 373–380. [Google Scholar] [CrossRef] [PubMed]
- Sapio, L.; Di Maiolo, F.; Illiano, M.; Esposito, A.; Chiosi, E.; Spina, A.; Naviglio, S. Targeting protein kinase A in cancer therapy: An update. EXCLI J. 2014, 13, 843–855. [Google Scholar]
- Cho-Chung, Y.S.; Nesterova, M.V. Tumor reversion: Protein kinase A isozyme switching. Ann. N. Y. Acad. Sci. 2005, 1058, 76–86. [Google Scholar] [CrossRef]
- Nadella, K.S.; Kirschner, L.S. Disruption of protein kinase a regulation causes immortalization and dysregulation of D-type cyclins. Cancer Res. 2005, 65, 10307–10315. [Google Scholar] [CrossRef]
- Nesterova, M.; Yokozaki, H.; McDuffie, E.; Cho-Chung, Y.S. Overexpression of RII beta regulatory subunit of protein kinase A in human colon carcinoma cell induces growth arrest and phenotypic changes that are abolished by site-directed mutation of RII beta. Eur. J. Biochem. 1996, 235, 486–494. [Google Scholar] [CrossRef]
- Marko, D.; Pahlke, G.; Merz, K.H.; Eisenbrand, G. Cyclic 3’,5’-nucleotide phosphodiesterases: Potential targets for anticancer therapy. Chem. Res. Toxicol. 2000, 13, 944–948. [Google Scholar] [CrossRef]
- Lin, D.C.; Xu, L.; Ding, L.W.; Sharma, A.; Liu, L.Z.; Yang, H.; Tan, P.; Vadgama, J.; Karlan, B.Y.; Lester, J.; et al. Genomic and functional characterizations of phosphodiesterase subtype 4D in human cancers. Proc. Natl. Acad. Sci. USA 2013, 110, 6109–6114. [Google Scholar] [CrossRef]
- Mouratidis, P.X.; Colston, K.W.; Bartlett, J.B.; Muller, G.W.; Man, H.W.; Stirling, D.; Dalgleish, A.G. Antiproliferative effects of CC-8062 and CC-8075 in pancreatic cancer cells. Pancreas 2009, 38, 78–84. [Google Scholar] [CrossRef] [PubMed]
- McEwan, D.G.; Brunton, V.G.; Baillie, G.S.; Leslie, N.R.; Houslay, M.D.; Frame, M.C. Chemoresistant KM12C colon cancer cells are addicted to low cyclic AMP levels in phosphodiesterase 4-regualted compartment via effects on phosphoinositide 3-kinase. Cancer Res. 2007, 67, 5248–5257. [Google Scholar] [CrossRef] [PubMed]
- Campagnolo, L.; Giorgi, M.; Augusti-Tocco, G. Phosphodiesterase specific inhibitors control cell growth of a human neuroepithelioma cell line. J. Neurooncol. 1997, 31, 123–127. [Google Scholar] [CrossRef] [PubMed]
- Marko, D.; Romanakis, K.; Zankl, H.; Fürstenberger, G.; Steinbauer, B.; Eisenbrand, G. Induction of apoptosis by an inhibitor of cAMP-specific PDE in malignant murine carcinoma cells overexpressing PDE activity in comparison to their nonmalignant counterparts. Cell Biochem. Biophys. 1998, 28, 75–101. [Google Scholar] [CrossRef]
- Wagner, B.; Jakobs, S.; Habermeyer, M.; Hippe, F.; Cho-Chung, Y.S.; Eisenbrand, G.; Marko, D. 7-Benzylamino-6-chloro-2-piperazino-4-pyrrolidino-pteridine, a potent inhibitor of cAMP-specific phosphodiesterase, enhancing nuclear protein binding to the CRE consensus sequence in human tumour cells. Biochem. Pharm. 2002, 63, 659–668. [Google Scholar] [CrossRef]
- Powers, G.L.; Hammer, K.D.; Domenech, M.; Frantskevich, K.; Malinowski, R.L.; Bushman, W.; Beebe, D.J.; Marker, P.C. Phosphodiesterase 4D inhibitors limit prostate cancer growth potential. Mol. Cancer Res. 2015, 13, 149–160. [Google Scholar] [CrossRef]
- Maurice, D.H.; Ke, H.; Ahmad, F.; Wang, Y.; Chung, J.; Manganiello, V.C. Advances in targeting cyclic nucleotide phosphodiesterases. Nat. Rev. Drug Discov. 2014, 13, 290–314. [Google Scholar] [CrossRef]
- Azevedo, M.F.; Faucz, F.R.; Bimpaki, E.; Horvath, A.; Levy, I.; de Alexandre, R.B.; Ahmad, F.; Manganiello, V.; Stratakis, C.A. Clinical and molecular genetics of the phosphodiesterases (PDEs). Endocr Rev. 2014, 35, 195–233. [Google Scholar] [CrossRef]
- Reneerkens, O.A.; Rutten, K.; Steinbusch, H.W.; Blokland, A.; Prickaerts, J. Selective phosphodiesterase inhibitors: A promising target for cognition enhancement. Psychopharmacology 2009, 202, 419–443. [Google Scholar] [CrossRef]
- Giorgi, M.; Modica, A.; Pompili, A.; Pacitti, C.; Gasbarri, A. The induction of cyclic nucleotide phosphodiesterase 4 gene (PDE4D) impairs memory in a water maze task. Behav. Brain Res. 2004, 154, 99–106. [Google Scholar] [CrossRef]
- Soares, L.M.; De Vry, J.; Steinbusch, H.W.M.; Milani, H.; Prickaerts, J.; Weffort de Oliveira, R.M. Rolipram improves cognition, reduces anxiety- and despair-like behaviors and impacts hippocampal neuroplasticity after transient global cerebral ischemia. Neuroscience 2016, 326, 69–83. [Google Scholar] [CrossRef] [PubMed]
- Goldhoff, P.; Warrington, N.M.; Limbrick, D.D., Jr.; Hope, A.; Woerner, B.M.; Jackson, E.; Perry, A.; Piwnica-Worms, D.; Rubin, J.B. Targeted inhibition of cyclic AMP phosphodiesterase-4 promotes brain tumor regression. Clin. Cancer Res. 2008, 14, 7717–7725. [Google Scholar] [CrossRef] [PubMed]
- Sengupta, R.; Sun, T.; Warrington, N.M.; Rubin, J.B. Treating brain tumors with PDE4 inhibitors. Trends Pharm. Sci. 2011, 32, 337–344. [Google Scholar] [CrossRef] [PubMed]
- Giorgi, M.; Caniglia, C.; Scarsella, G.; Augusti-Tocco, G. Characterization of 3′,5′ cyclic nucleotide phosphodiesterase activities of mouse neuroblastoma N18TG2 cells. FEBS Lett. 1993, 324, 76–80. [Google Scholar] [CrossRef][Green Version]
- Ramezani, S.; Vousooghi, N.; Kapourchali, F.R.; Hadjighasem, M.; Hayat, P.; Amini, N.; Joghataei, M.T. Rolipram potentiates bevacizumab-induced cell death in human glioblastoma stem-like cells. Life Sci. 2017, 173, 11–19. [Google Scholar] [CrossRef]
- Brooks, M.D.; Jackson, E.; Warrington, N.M.; Luo, J.; Forys, J.T.; Taylor, S.; Mao, D.D.; Leonard, J.R.; Kim, A.H.; Piwnica-Worms, D.; et al. PDE7B is a novel, prognostically significant mediator of glioblastoma growth whose expression is regulated by endothelial cells. PLoS ONE 2014, 9, e107397. [Google Scholar] [CrossRef]
- Lee, J.; Choi, Y.H.; Nguyen, P.M.; Kim, J.S.; Lee, S.J.; Trepel, J.B. Cyclic AMP induces inhibition of cyclin A expression and growth arrest in human hepatoma cells. Biochi. Biophis. Acta 1999, 1449, 261–268. [Google Scholar] [CrossRef]
- Tarn, W.Y.; Lai, M.C. Translational control of cyclins. Cell Div. 2011, 6, 5. [Google Scholar] [CrossRef]
- Paterlini, P.; Flejou, J.F.; De Mitri, M.S.; Pisi, E.; Franco, D.; Bréchot, C. Structure and expression of the cyclin A gene in human primary liver cancer. Correlation with flow cytometric parameters. J. Hepatol. 1995, 23, 47–52. [Google Scholar] [CrossRef]
- Vigneron, S.; Sundermann, L.; Labbé, J.C.; Pintard, L.; Radulescu, O.; Castro, A.; Lorca, T. Cyclin A-cdk1-Dependent phosphorylation of Bora Is the Triggering Factor Promoting Mitotic Entry. Dev. Cell 2018, 45, 637–650. [Google Scholar] [CrossRef]
- Chao, Y.; Shih, Y.L.; Chiu, J.H.; Chau, G.Y.; Lui, W.Y.; Yang, W.K.; Lee, S.D.; Huang, T.S. Overexpression of cyclin A but not Skp 2 correlates with the tumor relapse of human hepatocellular carcinoma. Cancer Res. 1998, 58, 985–990. [Google Scholar] [PubMed]
- Giorgi, M.; Leonetti, C.; Citro, G.; Augusti-Tocco, G. In vitro and in vivo inhibition of SK-N-MC neuroblastoma growth using cyclic nucleotide phosphodiesterase inhibitors. J. Neurooncol. 2001, 51, 25–31. [Google Scholar] [CrossRef]
- Fleischhacker, W.W.; Hinterhuber, H.; Bauer, H.; Pflug, B.; Berner, P.; Simhandl, C.; WolfGerlach, W.; Jaklitsch, H.; Sastre-y-Hernández, M.; Schmeding-Wiegel, H.; et al. A multicenter double-blind study of three different doses of the new cAMP-phosphodiesterase inhibitor rolipram in patients with major depressive disorder. Neuropsychobiology 1992, 26, 59–64. [Google Scholar] [CrossRef] [PubMed]
- Pearse, D.D.; Hughes, Z.A. PDE4B as a microglia target to reduce neuroinflammation. Glia 2016, 64, 1698–1709. [Google Scholar] [CrossRef] [PubMed]
- Ionta, M.; Rosa, M.C.; Almeida, R.B.; Freitas, V.M.; Rezende-Teixeira, P.; Machado-Santelli, G.M. Retinoic acid and cAMP inhibit rat hepatocellular carcinoma cell proliferation and enhance cell differentiation. Braz J. Med. Biol. Res. 2012, 45, 721–729. [Google Scholar] [CrossRef] [PubMed]
- Noy, N. Between death and survival: Retinoic acid in regulation of apoptosis. Ann. Rev. Nutr. 2010, 30, 201–217. [Google Scholar] [CrossRef] [PubMed]
- Ara, C.; Massimi, M.; Conti Devirgiliis, L. Retinoic acid modulates gap junctional intercellular communication in hepatocytes and hepatoma cells. Cell. Mol. Life Sci. 2002, 59, 1758–1765. [Google Scholar] [CrossRef] [PubMed]
- Ara, C.; Conti DeVirgiliis, L.; Massimi, M. Influence of retinoic acid on adhesion complexes in human hepatoma cells: A clue to its antiproliferative effects. Cell Commun. Adhes. 2004, 11, 13–23. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Jung, H.Y.; Park, S.H.; Yoo, Y.D.; Kim, J.S.; Kim, Y.H. CDK2/4 regulate retinoic acid-induced G1 arrest in hepatocellular carcinoma cells. Hepatol. Res. 2005, 31, 143–152. [Google Scholar] [CrossRef]
- Massimi, M.; Conti Devirgiliis, L. Adhesion to the extracellular matrix is positively regulated by retinoic acid in HepG2 cells. Liver Int. 2007, 27, 128–136. [Google Scholar] [CrossRef]
- Yang, J.; Shi, Q.D.; Song, T.B.; Feng, G.F.; Zang, W.J.; Zong, C.H.; Chang, L. Vasoactive intestinal peptide increases VEGF expression to promote proliferation of brain vascular endothelial cells via the cAMP/PKA pathway after ischemic insult in vitro. Peptides 2013, 42, 105–111. [Google Scholar] [CrossRef] [PubMed]
- Hara, M.; Takeba, Y.; Iiri, T.; Ohta, Y.; Ootaki, M.; Watanabe, M.; Watanabe, D.; Koizumi, S.; Otsubo, T.; Matsumoto, N. Vasoactive intestinal peptide increases apoptosis of hepatocellular carcinoma by inhibiting the cAMP/Bcl-xL pathway. Cancer Sci. 2019, 110, 235–244. [Google Scholar] [CrossRef] [PubMed]
- Vacas, E.; Fernández-Martínez, A.B.; Bajo, A.M.; Sánchez-Chapado, M.; Schally, A.V.; Prieto, J.C.; Carmena, M.J. Vasoactive intestinal peptide (VIP) inhibits human renal cell carcinoma proliferation. Biochim. Biophys. Acta 2012, 1823, 1676–1685. [Google Scholar] [CrossRef] [PubMed]
- Sun, L.; Quan, H.; Xie, C.; Wang, L.; Hu, Y.; Lou, L. Phosphodiesterase 3/4 inhibitor zardaverine exhibits potent and selective antitumor activity against hepatocellular carcinoma both in vitro and in vivo independently of phosphodiesterase inhibition. PLoS ONE 2014, 9, e90627. [Google Scholar] [CrossRef] [PubMed]
- MacKenzie, S.J.; Houslay, M.D. Action of rolipram on specific PDE4 cAMP phosphodiesterase isoforms and on the phosphorylation of cAMP-response-element-binding protein (CREB) and p38 mitogen-activated protein (MAP) kinase in U937 monocytic cells. Biochem J. 2000, 347, 571–578. [Google Scholar] [CrossRef]
- Inui, M.; Martello, G.; Piccolo, S. MicroRNA control of signal transduction. Nat. Rev. Mol. Cell Biol. 2010, 11, 252–263. [Google Scholar] [CrossRef]
- Hwang, H.W.; Mendell, J.T. MicroRNAs in cell proliferation, cell death, and tumorigenesis. Br. J. Cancer 2006, 94, 776–780. [Google Scholar] [CrossRef]
- Khella, H.W.; White, N.M.; Faragalla, H.; Gabril, M.; Boazak, M.; Dorian, D.; Khalil, B.; Antonios, H.; Bao, T.T.; Pasic, M.D.; et al. Exploring the role of miRNAs in renal cell carcinoma progression and metastasis through bioinformatic and experimental analyses. Tumour Biol. 2012, 33, 131–140. [Google Scholar] [CrossRef]
- Huo, W.; Du, M.; Pan, X.; Zhu, X.; Gao, Y.; Li, Z. miR-203a-3p.1 targets IL-24 to modulate hepatocellular carcinoma cell growth and metastasis. FEBS Open Bio. 2017, 7, 1085–1091. [Google Scholar] [CrossRef]
- Menezes, M.E.; Bhatia, S.; Bhoopathi, P.; Das, S.K.; Emdad, L.; Dasgupta, S.; Dent, P.; Wang, X.Y.; Sarkar, D.; Fisher, P.B. MDA-7/IL-24: Multifunctional cancer killing cytokine. Adv. Exp. Med. Biol. 2014, 818, 127–153. [Google Scholar]
- Chen, L.; Gao, H.; Liang, J.; Qiao, J.; Duan, J.; Shi, H.; Zhen, T.; Li, H.; Zhang, F.; Zhu, Z.; et al. miR-203a-3p promotes colorectal cancer proliferation and migration by targeting PDE4D. Am. J. Cancer Res. 2018, 8, 2387–2401. [Google Scholar] [PubMed]
- Kim, D.U.; Nam, J.; Cha, M.D.; Kim, S.W. Inhibition of phosphodiesterase 4D decreases the malignant properties of DLD 1 colorectal cancer cells by repressing the AKT/mTOR/Myc signaling pathway. Oncol Lett. 2019, 17, 3589–3598. [Google Scholar] [CrossRef] [PubMed]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Massimi, M.; Ragusa, F.; Cardarelli, S.; Giorgi, M. Targeting Cyclic AMP Signalling in Hepatocellular Carcinoma. Cells 2019, 8, 1511. https://doi.org/10.3390/cells8121511
Massimi M, Ragusa F, Cardarelli S, Giorgi M. Targeting Cyclic AMP Signalling in Hepatocellular Carcinoma. Cells. 2019; 8(12):1511. https://doi.org/10.3390/cells8121511
Chicago/Turabian StyleMassimi, Mara, Federica Ragusa, Silvia Cardarelli, and Mauro Giorgi. 2019. "Targeting Cyclic AMP Signalling in Hepatocellular Carcinoma" Cells 8, no. 12: 1511. https://doi.org/10.3390/cells8121511
APA StyleMassimi, M., Ragusa, F., Cardarelli, S., & Giorgi, M. (2019). Targeting Cyclic AMP Signalling in Hepatocellular Carcinoma. Cells, 8(12), 1511. https://doi.org/10.3390/cells8121511