A Caspase-3 Reporter for Fluorescence Lifetime Imaging of Single-Cell Apoptosis
Abstract
1. Introduction
2. Protocol 1: Generating and Maintaining Stable Cell Lines
2.1. Materials
- HEK 293T human embryonic kidney cells (293T, ATCC® CRL-3216™)
- MDA-MB-231 metastatic breast cancer cells (231, ATCC® HTB-26™)
- HS-5 immortalized human bone marrow cell line (HS-5, ATCC® CRL-11882™)
- HS-27a immortalized human bone marrow cell line (HS-27a, ATCC® CRL-2496™)
- Cell culture media supplies
- Dulbecco’s Modified Eagle Medium containing high glucose and pyruvate (DMEM, Gibco® cat. # 11995-065)
- Standard Fetal Bovine Serum (FBS, HyClone™ cat. # SH300088.03)
- Penicillin-Streptomycin, 100× (P/S, Gibco® cat. # 15140-122)
- GlutaMAX Supplement™, 100× (GMx, Gibco® cat. # 35050-061)
- 0.25% Trypsin-EDTA, 1× (Gibco® cat. #25200-056)
- Sterile Phosphate Buffered Saline pH 7.4, 1× (PBS, Gibco® cat. # 10010-049)
- Various cell culture supplies such as incubators, plasticware, and sterile pipettes
- pcDNA™6/V5-His A, B, & C Mammalian Expression Vectors (pcDNA, Invitrogen™ cat. # V220-20)
- PCR primers for blasticidin reading frame amplification (IDT® or similar vendor)
- 5′-GTGGTTTTCCTTTGAAAAACACGATGATAATATGGCCAAGCCTTTGTCTC-3′
- 5′-CCAGACGCGTTCAATTAATTAGCCCTCCCACACATAACCAG-3′
- Lentiviral vector pLVX IRES puromycin (Clontech cat. # 632183)
- Fluorescent protein LSS-mOrange (gift of V. Verkhusha, Albert Einstein College of Medicine) [8]
- PCR primers for LSS-mOrange amplification (IDT® or similar vendor)
- 5′-ATGCGCTAGCGCCACCATGGTGAGCAAGGGCGAGGAG-3′
- 5′-GCATGCGGCCGCTTACTTGTACAGCTCGTCCATGCCGC-3′
- Blasticidin S HCl, powder (ThermoFisher Scientific cat. # R21001)
- Sterile water (sterilized by institutional autoclave standards)
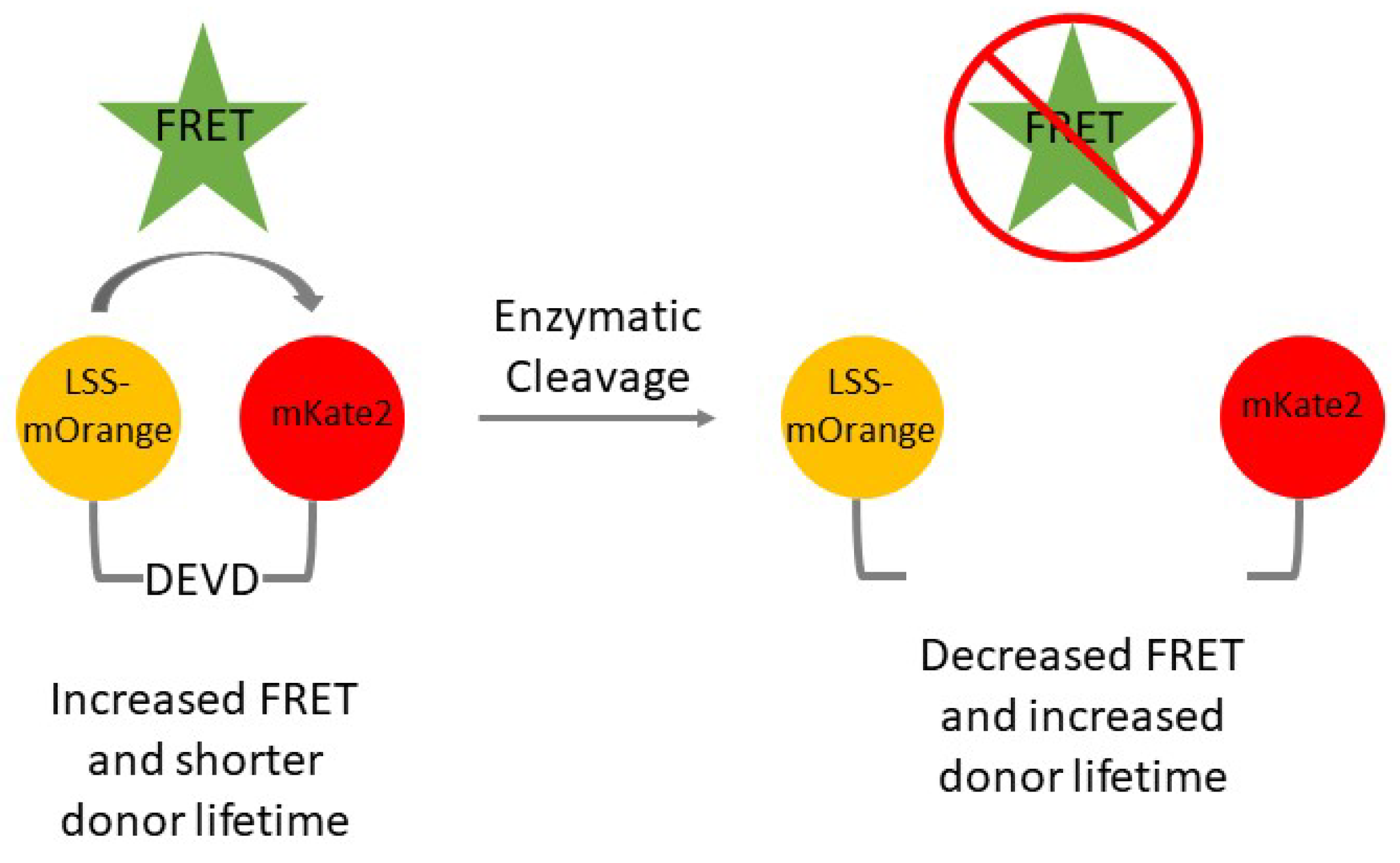
- Fluorescent protein LSS-mOrange-DEVD-mKate2 (gift of V. Verkhusha, Albert Einstein College of Medicine) [8]
- PB-CMV-MCS-EF1-Puro cDNA Cloning and Expression Vector (Systems Bioscience cat. # PB510B-1)
- Super PiggyBac Transposase expression vector (Systems Bioscience cat. # PB210PA-1)
- α-tri-Calcium phosphate (Sigma-Aldrich® cat. # 50553)
- FuGENE® 6 Transfection Reagent (Promega cat. # E2691)
- Enzymes, buffers, and equipment for PCR
- Enzymes for DNA restriction digests and ligations
2.2. Methods
- Culture cell lines as recommended by the supplier. We maintained the discussed cell lines in DMEM supplemented with 10% FBS, 1% P/S, and 1% GMx in a 5% CO2 incubator at 37 °C. We passaged these cells as necessary by trypsinization and resuspension approximately every three days.
- For details and general protocols on transferring reporters of interest to lentiviral vectors, transfecting and transducing cell lines, and selecting for a homogenous population of stably-expressing cells, please refer to standard molecular biology texts
- LSS-mOrange lentiviral vector generation
- Using the PCR primers for blasticidin reading frame amplification listed in the materials, amplify the blasticidin reading frame within the pcDNA™6/V5-His A expression vector to create a selection marker for blasticidin resistance.
- Ligate the generated PCR product (blasticidin resistance) into the BmgB1 and MluI sites on the lentiviral pLVX-IRES-puro vector to remove the gene cassette for puromycin and create the pLVX-IRES-blasticidin vector.
- Amplify the LSS-mOrange cassette using the primers for PCR amplification of LSS-mOrange (materials section #13). Digest the resultant PCR product and ligate into the pLVX-IRES-blasticidin vector, generated in the previous step, at the NheI and NotI sites.
- Produce lentivirus of the LSS-mOrange pLVX-IRES-blasticidin vector by transient transfection of HEK 293T cells.
- Generate stably-expressing cell lines with the above-generated LSS-mOrange pLVX-IRES-blasticidin lentivirus and use blasticidin treatment to select for stably-expressing cells.
- LSS-mOrange-DEVD-mKate2 vector generation
- Excise the LSS-mOrange-DEVD-mKate2 FRET reporter cassette at the NheI and NotI sites. Ligate the cassette into the PiggyBac transposon vector at the corresponding NheI and NotI sites. A transposon must be used while generating cells expressing the LSS-mOrange-DEVD-mKate2 fluorescent reporter to prevent homologous recombination of LSS-mOrange and mKate2. These two fluorescent proteins have similar DNA sequences and that may lead to homologous recombination and result in a lack of a functional reporter.
- Based on protocols optimized for each cell type, use calcium phosphate or FuGENE® 6 to transfect 293T and 231 cells with the PiggyBac LSS-mOrange-DEVD-mKate2 transposon vector and corresponding transposase plasmid, respectively.
- Sort for the top 30% of cells with positive expression of the LSS-mOrange-DEVD-mKate2 reporter using fluorescence-activated cell sorting (FACS). We sorted on mKate2 using a 561 nm excitation wavelength. We sorted for the top 30% to standardize the reporter expression levels.
3. Protocol 2: Apoptosis Imaging Experimental Setup
3.1. Materials
- Reporter verification studies
- Plasmid encoding apoptosis regulator bcl-2-like protein 4 (BAX, gift of S. Galbán, University of Michigan)
- pLenti-GIII-EF1a empty plasmid vector for vector control (gift of S. Galbán, University of Michigan)
- Two-dimensional cancer models and FLIM studies
- Trametinib (GSK112021, SelleckChem © cat. # S2673)
- Staurosporine (Cell Signaling Technology® cat. # 9953S)
- Dimethyl Sulfoxide (DMSO, Corning® cat. # 25-950-CQC)
- Dulbecco’s Modified Eagle Medium without glucose, glutamine, or phenol red (Gibco® cat. #A1443001)
- l-Glutamine, 200 nM (Gibco® cat. #25030081)
- Glucose Solution (Gibco® cat. #A2494001)
- Culture media supplies
- Dulbecco’s Modified Eagle Medium with high glucose and no phenol red (PRF DMEM, Gibco® cat. # 31053-028)
- Standard Fetal Bovine Serum (FBS, HyClone™ cat. # SH300088.03)
- Penicillin-Streptomycin-Glutamine, 100× (P/S/G, Gibco® cat. # 10378-016)
- Sodium pyruvate, 100× (Gibco® cat. # 11360-070)
- Sodium dichloroacetate (DCA, Sigma-Aldrich® cat. # 347795)
- Spheroid model treatment and imaging studies
- 384-well low volume black round bottom polystyrene NBS™ microplate, nonsterile (Corning® cat. # 3676)
- Transfer, imaging, and analysis (TRIM) plate. Note: We previously designed and described the TRIM plate to transfer and stabilize spheroids for fluorescence microscopy [13].
- Animal models and intravital microscopy
- 12–14-week-old female NOD.Cg-Prkdcscid Il2rgtm1Wjl/SzJ mice (NSG, The Jackson Laboratory).
- Small animal shaver (Wahl compact cordless trimmer or similar instrument)
- A depilatory solution such as Nair™
- Isoflurane
- Various desired surgical supplies
- Sterile 0.9% w/v NaCl solution
- Trametinib (GSK112021, SelleckChem © cat. # S2673)
- Dimethyl Sulfoxide (DMSO, Corning® cat. # 25-950-CQC)
- Carboxymethylcellulose, Sodium Salt, Low Viscosity (Calbiochem cat. # 217277)
- Tween® 80 (Sigma-Aldrich® cat. # P4780-100ML)
- Histology
- Cleaved Caspase-3 (Asp175) Antibody (Cell Signaling Technology® cat. #9661)
3.2. Methods
- Reporter verification studies
- For BAX apoptosis studies, transiently transfect stably-expressing 293T LSS-mOrange-DEVD-mKate2 or stably-expressing 293T LSS-mOrange cells using increasing concentrations of either the BAX-encoding plasmid or empty vector (control) and calcium phosphate. Amounts of 500, 1000, and 2000 ng plasmid proved a useful range for our study.
- Wait 24 h after transfection and seed 1.4e5 cells of BAX-293T LSS-mOrange-DEVD-mKate2 or BAX-293T LSS-mOrange cells into the 6 wells of an entire 6-well plate using phenol red free (PRF) DMEM supplemented with 10% FBS and 1% P/S/G. Using phenol red-free media for fluorescence imaging assays minimizes background noise.
- 24 h after re-seeding the BAX-transfected cells, image cells for caspase-3 LSS-mOrange-DEVD-mKate2 reporter activation and compare to LSS-mOrange control cell lifetimes for reporter verification using FLIM.
- Two-dimensional cancer models and FLIM studies
- For studying effects of nutrient deprivation and various compounds (DCA, trametinib, and staurosporine) on apoptosis and fluorescence lifetime, seed 1.4e5 231 LSS-mOrange-DEVD-mKate2 cells into each well of an entire 6-well plate using PRF DMEM supplemented with 10% FBS and 1% P/S/G.
- Treat cells 24 h before performing imaging studies.
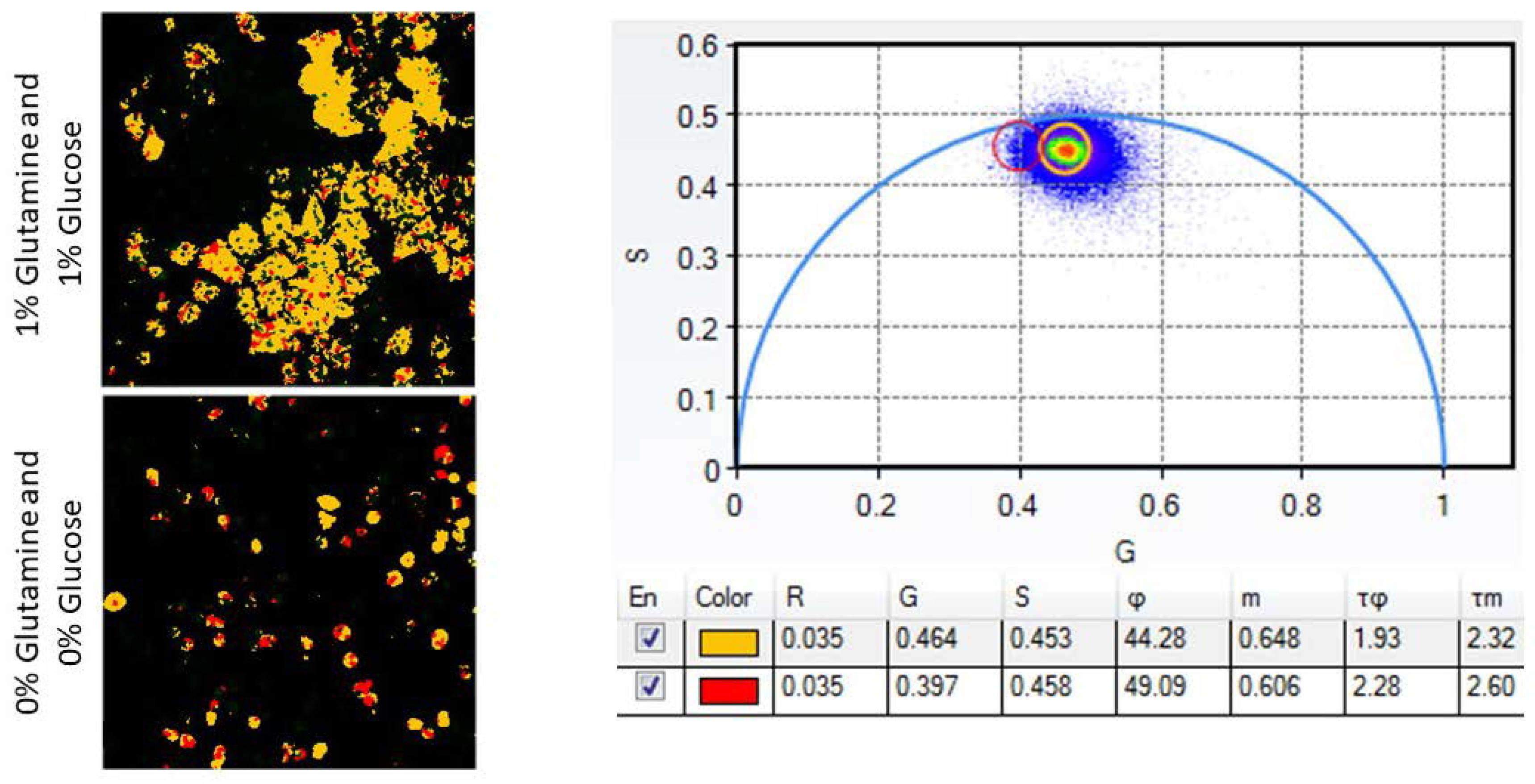
- Nutrient deprivation: Treat cells with phenol red, glutamine, and glucose-free DMEM supplemented with 10% FBS and either 1% glucose, 1% glutamine, or neither.
- DCA: Treat cells with 40mM DCA dissolved in phenol red, glutamine, and glucose-free DMEM supplemented with 10% FBS and 1% sodium pyruvate and either 1% glucose or 1% glutamine.
- Compound studies: Treat cells with 100 nM trametinib, 1 μM staurosporine, or DMSO vehicle control in PRF DMEM supplemented with 10% FBS and 1% P/S/G.
- Image cells for caspase-3 LSS-mOrange-DEVD-mKate2 reporter activation and changes in LSS-mOrange fluorescence lifetime with nutrient deprivation and compound treatment.
- Cancer spheroid model and FLIM studies
- Prepare for seeding cells for spheroid formation by sterilizing 384-well plates by radiating with UV light for 90 s. Formulate media with base PRF DMEM supplemented with 10% FBS and 1% P/S/G.
- Place 3000 total cells in 25 μL of media per well with 20% cancer cells (LSS-mOrange-DEVD-mKate2 expressing 231 cells) mixed with 80% bone marrow stromal cells (either HS-5 or HS-27A cells). Seeding cancer cells with stromal cells allow for the cells to form viable spheroids that some cancer cell lines may not be able to form alone.
- After waiting 24 h for cells to form compact spheroids, gently remove 18 μL of media from each spheroid-containing well and add 20 μL fresh media containing either 100 nM trametinib or DMSO as its vehicle control.
- 24 h after treatment, transfer spheroids to a TRIM plate and image for 3D caspase-3 reporter activation using FLIM imaging.
- Murine models and intravital tumor microscopy
- Verify that animal procedures have been approved by the local IACUC.
- Create a concentrated suspension of 5e5 LSS-mOrange-DEVD-mKate2 expressing 231 cells/50 μL in sterile NaCl solution. Keep the solution cold by leaving cell suspension on ice until use.
- Prepare 12- to 14-week-old female NSG mice for surgery and inject 50 μL of the above-generated cell suspension into both fourth inguinal mammary fat pads as we have previously described [14]. We choose to use NSG mice as they are highly immunocompromised and allow for effective growth of injected human breast cancer cells.
- Once 3 to 4 mm diameter tumors have formed, treat mice daily with of either 1 mg/kg trametinib or appropriate vehicle control by 50 μL of oral gavage as previously detailed [15]. Using the aforementioned parameters, tumor formation requires approximately 20 days. This length of time varies with mouse strain, cancer cell type, and concentration of injected cells.
- Using the FLIM imaging techniques described in Protocol 4, perform intravital microscopy for caspase-3 activation and LSS-mOrange lifetime after both 8 and 14 days of consecutive treatment.
- Histological Verification of in-vivo FLIM studies
- Following 14 days of either treatment or vehicle control, remove the formed mammary fat pad tumors and fix using standard techniques and 10% formalin. Post-fixation, follow standard protocols and embed, slice, and process tissue.
- Perform histology with hematoxylin and eosin staining and immunohistochemistry for caspase-3 reporter cleavage with the Cleaved Caspase-3 (Asp175) Antibody. Please refer to standard texts for details on immunohistochemistry and staining protocols.
- Image slides and determine the fraction of cells positive for caspase-3 reporter cleavage. Use these data to verify results from the acquired FLIM data.
4. Protocol 3: Glycolysis Stress Tests
4.1. Materials
- Culture media supplies
- Dulbecco’s Modified Eagle Medium with high glucose and pyruvate (DMEM, Gibco® cat. # 11995-065)
- Standard Fetal Bovine Serum (FBS, HyClone™ cat. # SH300088.03)
- Penicillin-Streptomycin, 100× (P/S, Gibco® cat. # 15140-122)
- Glutamine, 100× (Gibco® cat. # 25030-081
- Pyruvate, 100× (Gibco® cat. # 11360-070)
- Glucose, dissolved in sterile water, 1 M (Sigma Aldrich, cat. # G5767)
- Miscellaneous desired cell culture supplies such as plasticware, incubators, and sterile pipettes
- Non-CO2, 37 °C incubator
- Seahorse Bioscience XF96 96-well cell culture microplate (Agilent, Santa Clara, CA, USA, part # 101085-004)
- Seahorse Bioscience XFe96 extracellular flux assay kit (Agilent, Santa Clara, CA, USA, part # W26316) for the XFe96 analyzer
- Seahorse Bioscience XF Base Medium (part # 102353-100)
- Seahorse Bioscience XF Calibrant (Agilent, Santa Clara, CA, USA, part # 100840-000)
- Seahorse Bioscience Glycolysis Stress Test kit (part # 103017-100)
4.2. Methods
- 24 h before performing the metabolic flux assay, seed 5e3 231 cells in 80 μL DMEM supplemented with 10% FBS, 1% P/S, and 1% glutamine per well in a Seahorse Bioscience XF96 96-well plate. Add 200 μL XF calibrant to each well of the utility plate. Incubate cells in a 37 °C, 5% CO2 incubator and utility plate in a 37 °C, non-CO2 incubator overnight.
- One day after seeding cells, prepare Seahorse assay medium. Mix the below-listed ingredients, adjust the solution’s pH to 7.4, and sterile filter it. Store the medium at 37 °C until use.
- 100 mL Seahorse Bioscience XF Base medium pre-warmed to 37 °C
- 1 mL of 100 mM pyruvate
- 1 mL of 200 mM glutamine
- 1 mL of 1 M glucose solution
- Change the medium in the cell culture plate by gently washing each well twice with 180 μL of the Seahorse assay medium. In each well, gently add the final volume of 180 μL of the Seahorse assay medium. Before inserting the cell culture plate into the XF analyzer, place the plate into a non-CO2 incubator at 37 °C for 1 h.
- Following the instructions found in the Seahorse manual, suspend the components from the glycolysis stress test (glucose, oligomycin, and 2-deoxyglucose) in the Seahorse assay medium.
- Using a multichannel pipette, inject 10 mM glucose, 2 mM oligomycin, and 100 mM 2-deoxyglucose to Ports A, B, and C of the utility plate, respectively, immediately before beginning the experiment.
- Following the instructions displayed on the analyzer, place the utility plate inside the XF analyzer. Subsequently, insert the prepared cell culture plate.
- Setup the XF analyzer by indicating the injection and measurement strategies. Follow subsequent instructions from the software to perform the experiment.
- Using the Wave program provided by Seahorse Bioscience, analyze the acquired data.
5. Protocol 4: FLIM Techniques and Analysis
5.1. Materials
- 2-photon upright imaging system with variable laser power and compatible 25× objective (Olympus FVE1000 MPE microscope and 25× NIR corrected objective, XLPLN25XWMP, NA = 1.05, Olympus, Tokyo, Japan or similar product)
- 80 MHz pulsed scanning laser (Spectra-Physics Mai Tai-Deep Sea or comparable product)
- 572/15 nm bandpass emission filter
- FLIM instrument with frequency domain analysis capabilities (FastFLIM, ISS, Champaign, IL, USA or comparable product)
- Vista Vision Software or similar product for data analysis (ISS, Champaign, IL, USA)
5.2. Methods
- Allow laser to stabilize at 820 nm by turning it on for at least 45 min before calibration. Perform FLIM system lifetime calibration as per the manufacturer’s instructions. We used a 4.1 ns fluorescein slide for lifetime calibration.
- Immerse objective in the sample (6-well plate, TRIM plate, or tumor) and focus on a region of interest. Follow protocols to image orthotopic mammary tumors as we have previously discussed [16].
- Using an 820 nm wavelength, excite the LSS-mOrange fluorescent protein. We found a 256 × 256-pixel image with a 100 μs dwell time over 15–30 frames allow for sufficient counts and accurate data.
- LSS-mOrange is a long stokes shift fluorescent protein and emits in the orange channel (572 nm). Acquire and measure fluorescence lifetime of molecules in the orange channel. Literature reports the known LSS-mOrange lifetime as 2.75 ± 0.07 ns [17].
- After imaging the samples of interest, open and analyze images with ISS VistaVision or similar processing software. Using the Multi-Image Phasor Analysis function, set the minimum threshold count to eliminate background data and the Gaussian smoothing operation to 3 to smooth acquired data while maintaining spatial accuracy.
- Use the phasor plot and ROI function to define the portion of pixels with an increased LSS-mOrange lifetime. Pseudocolor this population as red and the shorter lifetime pixels as yellow with another ROI. Cells displaying increased amounts of yellow or red indicate increased or decreased amounts of apoptosis, respectively. Detailed reports on fluorescence lifetime analysis using phasor plots have been previously published [18].
6. Results
Author Contributions
Funding
Conflicts of Interest
References
- Elmore, S. Apoptosis: A review of programmed cell death. Toxicol. Pathol. 2007, 35, 495–516. [Google Scholar] [CrossRef] [PubMed]
- Cohen, G.M. Caspases: The executioners of apoptosis. Biochem. J. 1997, 326, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Suhling, K.; Hirvonen, L.M.; Levitt, J.A.; Chung, P.-H.; Tregidgo, C.; Le Marois, A.; Rusakov, D.A.; Zheng, K.; Ameer-Beg, S.; Poland, S.; et al. Fluorescence lifetime imaging (FLIM): Basic concepts and some recent developments. Med. Photonics 2015, 27, 3–40. [Google Scholar] [CrossRef]
- Sekar, R.B.; Periasamy, A. Fluorescence resonance energy transfer (FRET) microscopy imaging of live cell protein localizations. J. Cell Biol. 2003, 160, 629–633. [Google Scholar] [CrossRef] [PubMed]
- Hoppe, A.; Christensen, K.; Swanson, J.A. Fluorescence resonance energy transfer-based stoichiometry in living cells. Biophys. J. 2002, 83, 3652–3664. [Google Scholar] [CrossRef]
- Shrestha, D.; Jenei, A.; Nagy, P.; Vereb, G.; Szollosi, J. Understanding fret as a research tool for cellular studies. Int. J. Mol. Sci. 2015, 16, 6718–6756. [Google Scholar] [CrossRef] [PubMed]
- Yellen, G.; Mongeon, R. Quantitative two-photon imaging of fluorescent biosensors. Curr. Opin. Chem. Biol. 2015, 27, 24–30. [Google Scholar] [CrossRef] [PubMed]
- Stringari, C.; Nourse, J.L.; Flanagan, L.A.; Gratton, E. Phasor fluorescence lifetime microscopy of free and protein-bound nadh reveals neural stem cell differentiation potential. PLoS ONE 2012, 7, e48014. [Google Scholar] [CrossRef] [PubMed]
- McGlynn, S.P. Fluorescence and phosphorescence analysis. Principle and applications. J. Am. Chem. Soc. 1966, 88, 5688. [Google Scholar] [CrossRef]
- Shcherbakova, D.; Hink, M.; Joosen, L.; Gadella, T.; Verkhusha, V. An orange fluorescent protein with a large stokes shift for single-excitation multicolor FCCS and FRET imaging. J. Am. Chem. Soc. 2012, 134, 7913–7923. [Google Scholar] [CrossRef] [PubMed]
- Xiao, A.; Gibbons, A.E.; Luker, K.E.; Luker, G.D. Fluorescence lifetime imaging of apoptosis. Tomography 2015, 1, 115–124. [Google Scholar] [PubMed]
- Buschhaus, J.M.; Gibbons, A.E.; Luker, K.E.; Luker, G.D. Fluorescence lifetime imaging of a caspase-3 apoptosis reporter. Curr. Protoc. Cell Biol. 2017, 21, 1–21. [Google Scholar]
- Cavnar, S.; Salomonsson, E.; Luker, K.; Luker, G.; Takayama, S. Transfer, imaging, and analysis plate for facile handling of 384 hanging drop 3d tissue spheroids. J. Lab. Autom. 2014, 19, 208–214. [Google Scholar] [CrossRef] [PubMed]
- Luker, K.E.; Mihalko, L.A.; Schmidt, B.T.; Lewin, S.A.; Ray, P.; Shcherbo, D.; Chudakov, D.M.; Luker, G.D. In vivo imaging of ligand receptor binding with Gaussia luciferase complementation. Nat. Med. 2012, 18, 172–177. [Google Scholar] [CrossRef] [PubMed]
- Kwong, L.N.; Costello, J.C.; Liu, H.; Jiang, S.; Helms, T.L.; Langsdorf, A.E.; Jakubosky, D.; Genovese, G.; Muller, F.L.; Jeong, J.H.; et al. Oncogenic NRAS signaling differentially regulates survival and proliferation in melanoma. Nat. Med. 2012, 18, 1503–1510. [Google Scholar] [CrossRef] [PubMed]
- Salomonnson, E.; Mihalko, L.A.; Verkhusha, V.V.; Luker, K.E.; Luker, G.D. Cell-based and in vivo spectral analysis of fluorescent proteins for multiphoton microscopy. J. Biomed. Opt. 2012, 17, 096001. [Google Scholar] [CrossRef] [PubMed]
- Demeautis, C.; Sipieter, F.; Roul, J.; Chapuis, C.; Padilla-Parra, S.; Riquet, F.B.; Tramier, M. Multiplexing PKA and ERK1&2 kinases fret biosensors in living cells using single excitation wavelength dual colour flim. Sci. Rep. 2017, 7, 41026. [Google Scholar] [PubMed]
- Digman, M.A.; Caiolfa, V.R.; Zamai, M.; Gratton, E. The phasor approach to fluorescence lifetime imaging analysis. Biophys. J. 2008, 94, L14–L16. [Google Scholar] [CrossRef] [PubMed]
- Zherdeva, V.V.; Kazachkina, N.I.; Shcheslavskiy, V.I.; Savitsky, A.P. Long-term fluorescence lifetime imaging of a genetically encoded sensor for caspase-3 activity in mouse tumor xenografts. J. Biomed. Opt. 2018, 23, 035002. [Google Scholar]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Buschhaus, J.M.; Humphries, B.; Luker, K.E.; Luker, G.D. A Caspase-3 Reporter for Fluorescence Lifetime Imaging of Single-Cell Apoptosis. Cells 2018, 7, 57. https://doi.org/10.3390/cells7060057
Buschhaus JM, Humphries B, Luker KE, Luker GD. A Caspase-3 Reporter for Fluorescence Lifetime Imaging of Single-Cell Apoptosis. Cells. 2018; 7(6):57. https://doi.org/10.3390/cells7060057
Chicago/Turabian StyleBuschhaus, Johanna M., Brock Humphries, Kathryn E. Luker, and Gary D. Luker. 2018. "A Caspase-3 Reporter for Fluorescence Lifetime Imaging of Single-Cell Apoptosis" Cells 7, no. 6: 57. https://doi.org/10.3390/cells7060057
APA StyleBuschhaus, J. M., Humphries, B., Luker, K. E., & Luker, G. D. (2018). A Caspase-3 Reporter for Fluorescence Lifetime Imaging of Single-Cell Apoptosis. Cells, 7(6), 57. https://doi.org/10.3390/cells7060057



