Duplication and Nuclear Envelope Insertion of the Yeast Microtubule Organizing Centre, the Spindle Pole Body
Abstract
1. Introduction
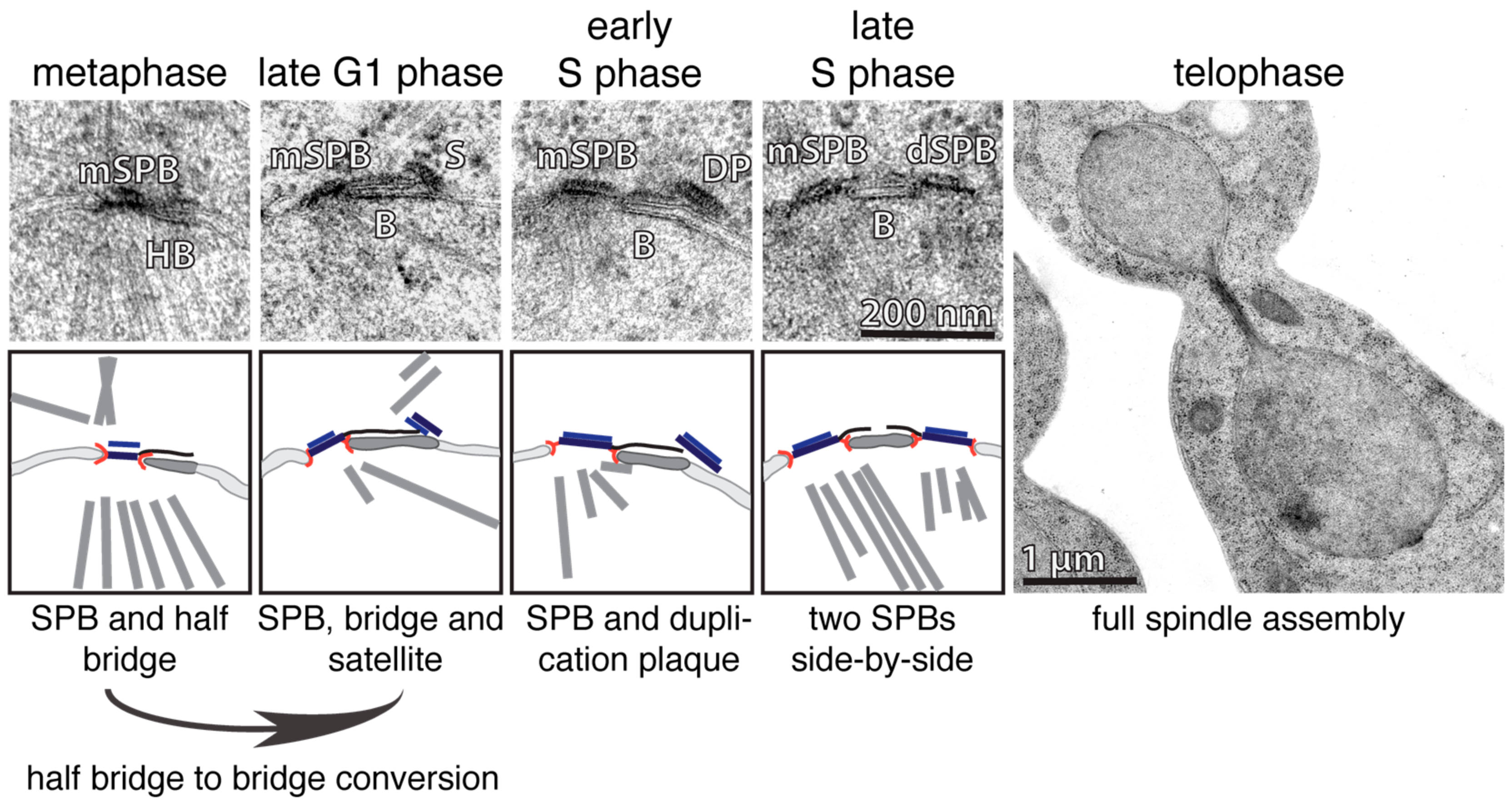
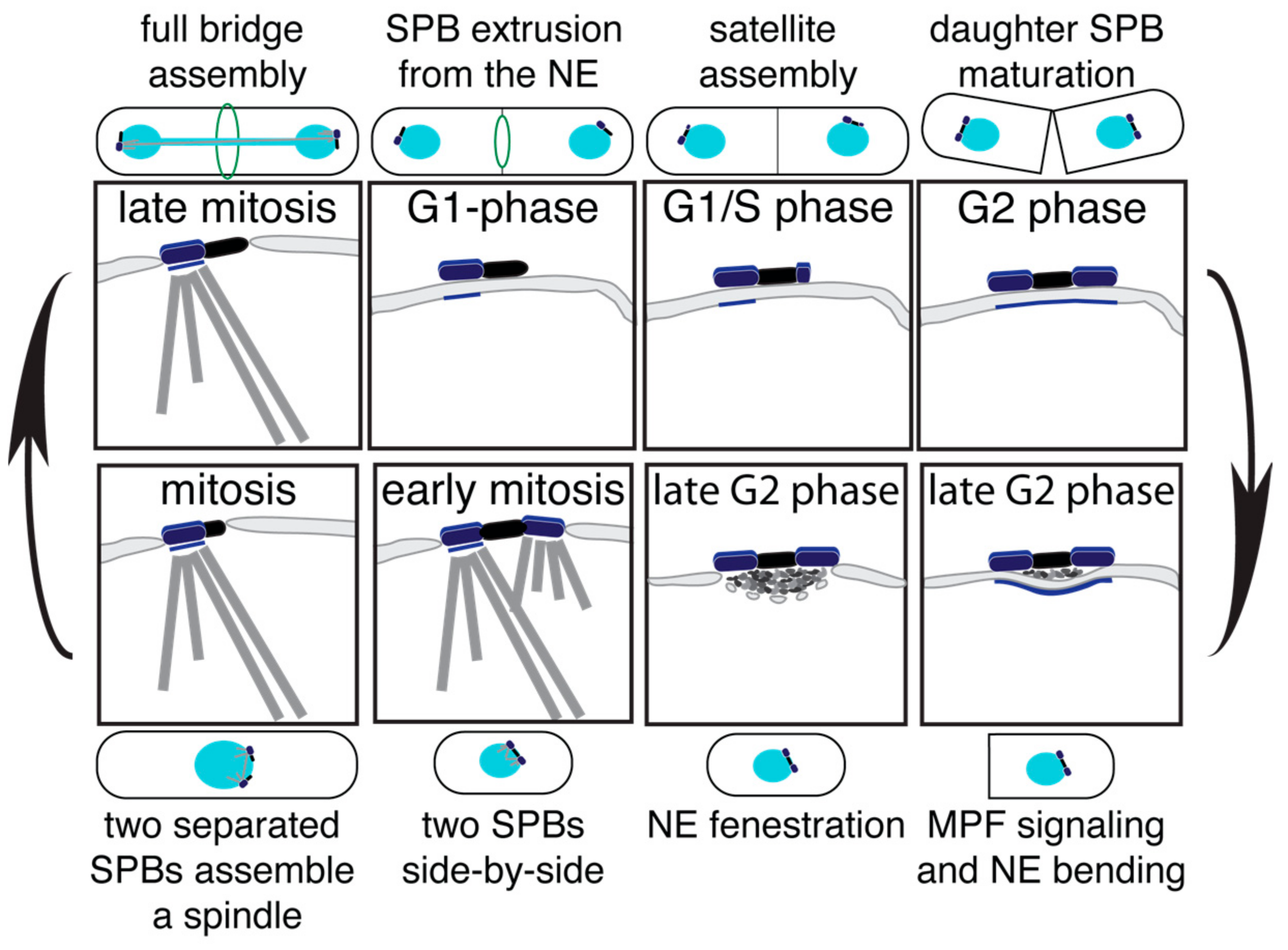
2. Duplication Cycle of the S. cerevisiae SPB
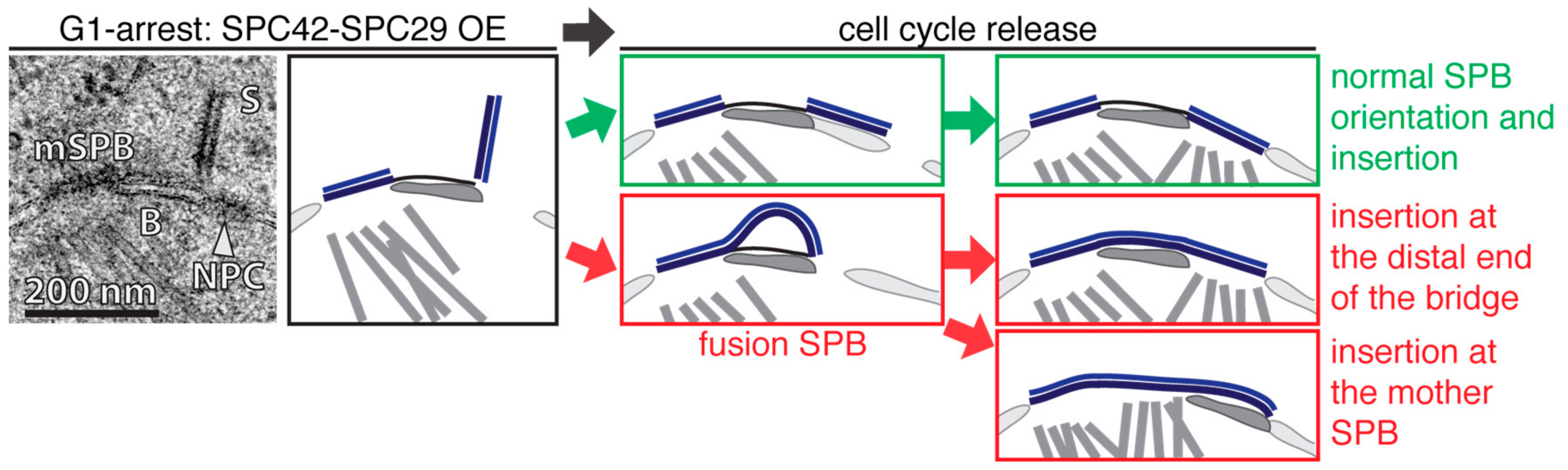
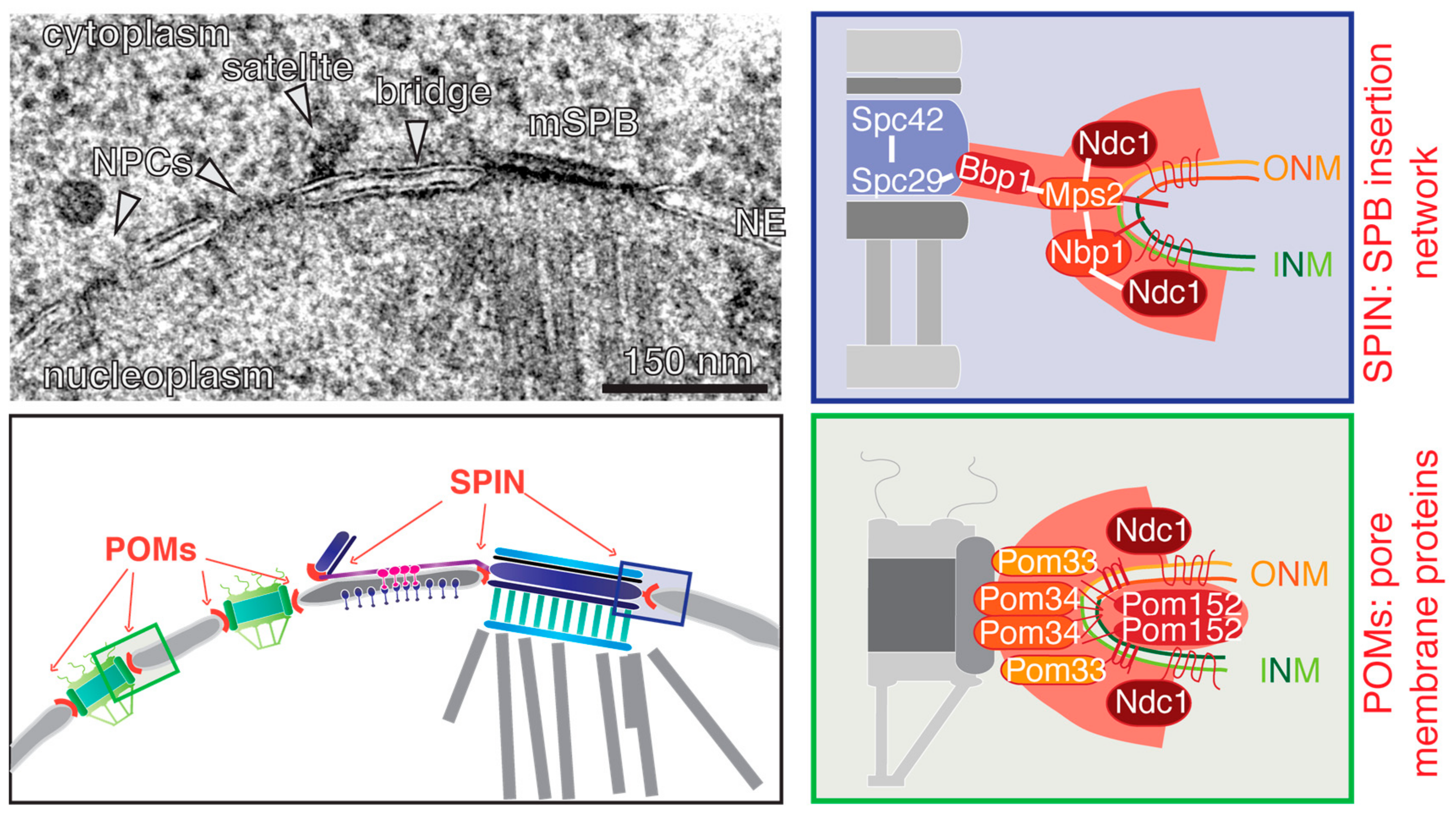
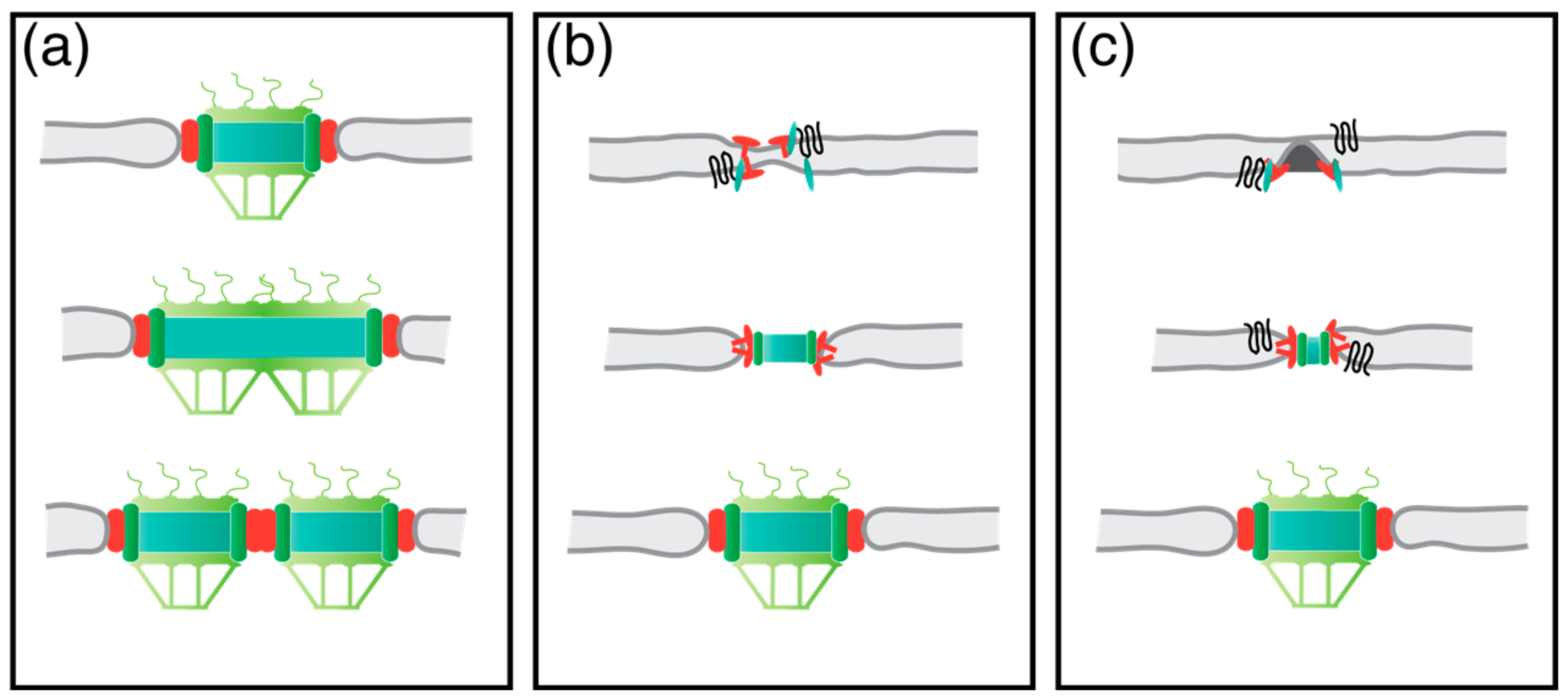
3. Insertion of the New SPB in S. cerevisiae
4. Duplication and Insertion of the S. pombe SPB
5. The Nuclear Pore Complex (NPC)
6. Closing Remarks and Future Directions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Kilmartin, J.V. Lessons from yeast: The spindle pole body and the centrosome. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2014, 369. [Google Scholar] [CrossRef] [PubMed]
- Nigg, E.A.; Holland, A.J. Once and only once: Mechanisms of centriole duplication and their deregulation in disease. Nat. Rev. Mol. Cell Biol. 2018, 19, 297–312. [Google Scholar] [CrossRef] [PubMed]
- Gönczy, P. Towards a molecular architecture of centriole assembly. Nat. Rev. Mol. Cell Biol. 2012, 13, 425–435. [Google Scholar] [CrossRef] [PubMed]
- Jaspersen, S.L.; Winey, M. The budding yeast spindle pole body: Structure, duplication, and function. Annu. Rev. Cell Dev. Biol 2004, 20, 1–28. [Google Scholar] [CrossRef] [PubMed]
- Schatten, G. The centrosome and its mode of inheritance: The reduction of the centrosome during gametogenesis and its restoration during fertilization. Dev. Biol. 1994, 165, 299–335. [Google Scholar] [CrossRef] [PubMed]
- Inoue, D.; Wittbrodt, J.; Gruss, O.J. Loss and Rebirth of the Animal Microtubule Organizing Center: How Maternal Expression of Centrosomal Proteins Cooperates with the Sperm Centriole in Zygotic Centrosome Reformation. Bioessays 2018, 40. [Google Scholar] [CrossRef] [PubMed]
- Lambrus, B.G.; Uetake, Y.; Clutario, K.M.; Daggubati, V.; Snyder, M.; Sluder, G.; Holland, A.J. p53 protects against genome instability following centriole duplication failure. J. Cell Biol. 2015, 210, 63–77. [Google Scholar] [CrossRef] [PubMed]
- Wong, Y.L.; Anzola, J.V.; Davis, R.L.; Yoon, M.; Motamedi, A.; Kroll, A.; Seo, C.P.; Hsia, J.E.; Kim, S.K.; Mitchell, J.W.; et al. Cell biology. Reversible centriole depletion with an inhibitor of Polo-like kinase 4. Science 2015, 348, 1155–1160. [Google Scholar] [CrossRef] [PubMed]
- Tarapore, P.; Fukasawa, K. Loss of p53 and centrosome hyperamplification. Oncogene 2002, 21, 6234–6240. [Google Scholar] [CrossRef] [PubMed]
- Ogden, A.; Rida, P.C.G.; Aneja, R. Centrosome amplification: A suspect in breast cancer and racial disparities. Endocr. Relat. Cancer 2017, 24, T47–T64. [Google Scholar] [CrossRef] [PubMed]
- Levine, M.S.; Bakker, B.; Boeckx, B.; Moyett, J.; Lu, J.; Vitre, B.; Spierings, D.C.; Lansdorp, P.M.; Cleveland, D.W.; Lambrechts, D.; et al. Centrosome Amplification Is Sufficient to Promote Spontaneous Tumorigenesis in Mammals. Dev. Cell 2017, 40, 313.e315–322.e315. [Google Scholar] [CrossRef] [PubMed]
- Werner, S.; Pimenta-Marques, A.; Bettencourt-Dias, M. Maintaining centrosomes and cilia. J. Cell Sci. 2017, 130, 3789–3800. [Google Scholar] [CrossRef] [PubMed]
- Loncarek, J.; Bettencourt-Dias, M. Building the right centriole for each cell type. J. Cell Biol. 2018, 217, 823–835. [Google Scholar] [CrossRef] [PubMed]
- Jakobsen, L.; Vanselow, K.; Skogs, M.; Toyoda, Y.; Lundberg, E.; Poser, I.; Falkenby, L.G.; Bennetzen, M.; Westendorf, J.; Nigg, E.A.; et al. Novel asymmetrically localizing components of human centrosomes identified by complementary proteomics methods. EMBO J. 2011, 30, 1520–1535. [Google Scholar] [CrossRef] [PubMed]
- Byers, B.; Goetsch, L. Duplication of spindle plaques and integration of the yeast cell cycle. Cold Spring Harb. Symp. Quant. Biol. 1974, 38, 123–131. [Google Scholar] [CrossRef] [PubMed]
- Moens, P.B.; Rapport, E. Spindles, spindle plaques, and meiosis in the yeast Saccharomyces cerevisiae (Hansen). J. Cell Biol. 1971, 50, 344–361. [Google Scholar] [CrossRef] [PubMed]
- Robinow, C.F.; Marak, J. A fiber apparatus in the nucleus of the yeast cell. J. Cell Biol. 1966, 29, 129–151. [Google Scholar] [CrossRef] [PubMed]
- Ding, R.; West, R.R.; Morphew, D.M.; Oakley, B.R.; McIntosh, J.R. The spindle pole body of Schizosaccharomyces pombe enters and leaves the nuclear envelope as the cell cycle proceeds. Mol. Biol. Cell 1997, 8, 1461–1479. [Google Scholar] [CrossRef] [PubMed]
- Cavanaugh, A.M.; Jaspersen, S.L. Big Lessons from Little Yeast: Budding and Fission Yeast Centrosome Structure, Duplication, and Function. Annu. Rev. Genet. 2017, 51, 361–383. [Google Scholar] [CrossRef] [PubMed]
- Hartwell, L.H.; Mortimer, R.K.; Culotti, J.; Culotti, M. Genetic Control of the Cell Division Cycle in Yeast: V. Genetic Analysis of cdc Mutants. Genetics 1973, 74, 267–286. [Google Scholar] [CrossRef]
- Schild, D.; Ananthaswamy, H.N.; Mortimer, R.K. An endomitotic effect of a cell cycle mutation of Saccharomyces cerevisiae. Genetics 1981, 97, 551–562. [Google Scholar] [PubMed]
- Spang, A.; Courtney, I.; Fackler, U.; Matzner, M.; Schiebel, E. The calcium-binding protein cell division cycle 31 of Saccharomyces cerevisiae is a component of the half bridge of the spindle pole body. J. Cell Biol. 1993, 123, 405–416. [Google Scholar] [CrossRef] [PubMed]
- Weiss, E.; Winey, M. The Saccharomyces cerevisiae spindle pole body duplication gene MPS1 is part of a mitotic checkpoint. J. Cell Biol. 1996, 132, 111–123. [Google Scholar] [CrossRef] [PubMed]
- Salisbury, J.L.; Suino, K.M.; Busby, R.; Springett, M. Centrin-2 is required for centriole duplication in mammalian cells. Curr. Biol. 2002, 12, 1287–1292. [Google Scholar] [CrossRef]
- Yang, C.H.; Kasbek, C.; Majumder, S.; Yusof, A.M.; Fisk, H.A. Mps1 phosphorylation sites regulate the function of centrin 2 in centriole assembly. Mol. Biol. Cell 2010, 21, 4361–4372. [Google Scholar] [CrossRef] [PubMed]
- Sawant, D.B.; Majumder, S.; Perkins, J.L.; Yang, C.H.; Eyers, P.A.; Fisk, H.A. Centrin 3 is an inhibitor of centrosomal Mps1 and antagonizes centrin 2 function. Mol. Biol. Cell 2015, 26, 3741–3753. [Google Scholar] [CrossRef] [PubMed]
- Kilmartin, J.V. Sfi1p has conserved centrin-binding sites and an essential function in budding yeast spindle pole body duplication. J. Cell Biol. 2003, 162, 1211–1221. [Google Scholar] [CrossRef] [PubMed]
- Lee, I.J.; Wang, N.; Hu, W.; Schott, K.; Bahler, J.; Giddings, T.H., Jr.; Pringle, J.R.; Du, L.L.; Wu, J.Q. Regulation of spindle pole body assembly and cytokinesis by the centrin-binding protein Sfi1 in fission yeast. Mol. Biol. Cell 2014, 25, 2735–2749. [Google Scholar] [CrossRef] [PubMed]
- Azimzadeh, J.; Hergert, P.; Delouvée, A.; Euteneuer, U.; Formstecher, E.; Khodjakov, A.; Bornens, M. hPOC5 is a centrin-binding protein required for assembly of full-length centrioles. J. Cell Biol. 2009, 185, 101–114. [Google Scholar] [CrossRef] [PubMed]
- Oakley, C.E.; Oakley, B.R. Identification of gamma-tubulin, a new member of the tubulin superfamily encoded by mipA gene of Aspergillus nidulans. Nature 1989, 338, 662–664. [Google Scholar] [CrossRef] [PubMed]
- Oakley, B.R.; Paolillo, V.; Zheng, Y. gamma-Tubulin complexes in microtubule nucleation and beyond. Mol. Biol. Cell 2015, 26, 2957–2962. [Google Scholar] [CrossRef] [PubMed]
- Gall, J.G. Centriole replication. A study of spermatogenesis in the snail Viviparus. J. Biophys. Biochem. Cytol. 1961, 10, 163–193. [Google Scholar] [CrossRef] [PubMed]
- McCully, E.K.; Robinow, C.F. Mitosis in the fission yeast Schizosaccharomyces pombe: A comparative study with light and electron microscopy. J. Cell Sci. 1971, 9, 475–507. [Google Scholar] [PubMed]
- Byers, B.; Goetsch, L. Behavior of spindles and spindle plaques in the cell cycle and conjugation of Saccharomyces cerevisiae. J. Bacteriol. 1975, 124, 511–523. [Google Scholar] [PubMed]
- Adams, I.R.; Kilmartin, J.V. Localization of core spindle pole body (SPB) components during SPB duplication in Saccharomyces cerevisiae. J. Cell Biol. 1999, 145, 809–823. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Sandercock, A.M.; Conduit, P.; Robinson, C.V.; Williams, R.L.; Kilmartin, J.V. Structural role of Sfi1p-centrin filaments in budding yeast spindle pole body duplication. J. Cell Biol. 2006, 173, 867–877. [Google Scholar] [CrossRef] [PubMed]
- Elserafy, M.; Šarić, M.; Neuner, A.; Lin, T.C.; Zhang, W.; Seybold, C.; Sivashanmugam, L.; Schiebel, E. Molecular mechanisms that restrict yeast centrosome duplication to one event per cell cycle. Curr. Biol. 2014, 24, 1456–1466. [Google Scholar] [CrossRef] [PubMed]
- Avena, J.S.; Burns, S.; Yu, Z.; Ebmeier, C.C.; Old, W.M.; Jaspersen, S.L.; Winey, M. Licensing of yeast centrosome duplication requires phosphoregulation of sfi1. PLoS Genet. 2014, 10, e1004666. [Google Scholar] [CrossRef] [PubMed]
- O’Toole, E.T.; Winey, M.; McIntosh, J.R. High-voltage electron tomography of spindle pole bodies and early mitotic spindles in the yeast Saccharomyces cerevisiae. Mol. Biol. Cell 1999, 10, 2017–2031. [Google Scholar] [CrossRef] [PubMed]
- Rüthnick, D.; Neuner, A.; Dietrich, F.; Kirrmaier, D.; Engel, U.; Knop, M.; Schiebel, E. Characterization of spindle pole body duplication reveals a regulatory role for nuclear pore complexes. J. Cell Biol. 2017, 216, 2425–2442. [Google Scholar] [CrossRef] [PubMed]
- Seybold, C.; Elserafy, M.; Ruthnick, D.; Ozboyaci, M.; Neuner, A.; Flottmann, B.; Heilemann, M.; Wade, R.C.; Schiebel, E. Kar1 binding to Sfi1 C-terminal regions anchors the SPB bridge to the nuclear envelope. J. Cell Biol. 2015, 209, 843–861. [Google Scholar] [CrossRef] [PubMed]
- Rüthnick, D.; Schiebel, E. Duplication of the Yeast Spindle Pole Body Once per Cell Cycle. Mol. Cell. Biol. 2016, 36, 1324–1331. [Google Scholar] [CrossRef] [PubMed]
- Castillo, A.R.; Meehl, J.B.; Morgan, G.; Schutz-Geschwender, A.; Winey, M. The yeast protein kinase Mps1p is required for assembly of the integral spindle pole body component Spc42p. J. Cell Biol. 2002, 156, 453–465. [Google Scholar] [CrossRef] [PubMed]
- Jaspersen, S.L.; Huneycutt, B.J.; Giddings, T.H.; Resing, K.A.; Ahn, N.G.; Winey, M. Cdc28/Cdk1 regulates spindle pole body duplication through phosphorylation of Spc42 and Mps1. Dev. Cell 2004, 7, 263–274. [Google Scholar] [CrossRef] [PubMed]
- Jaspersen, S.L.; Ghosh, S. Nuclear envelope insertion of spindle pole bodies and nuclear pore complexes. Nucleus 2012, 3, 226–236. [Google Scholar] [CrossRef] [PubMed]
- Rothballer, A.; Kutay, U. Poring over pores: Nuclear pore complex insertion into the nuclear envelope. Trends Biochem. Sci. 2013, 38, 292–301. [Google Scholar] [CrossRef] [PubMed]
- Han, J.; Pluhackova, K.; Bockmann, R.A. The Multifaceted Role of SNARE Proteins in Membrane Fusion. Front. Physiol. 2017, 8, 5. [Google Scholar] [CrossRef] [PubMed]
- Harrison, S.C. Viral membrane fusion. Virology 2015, 479–480, 498–507. [Google Scholar] [CrossRef] [PubMed]
- Ries, J.; Kaplan, C.; Platonova, E.; Eghlidi, H.; Ewers, H. A simple, versatile method for GFP-based super-resolution microscopy via nanobodies. Nat. Methods 2012, 9, 582–584. [Google Scholar] [CrossRef] [PubMed]
- Burns, S.; Avena, J.S.; Unruh, J.R.; Yu, Z.; Smith, S.E.; Slaughter, B.D.; Winey, M.; Jaspersen, S.L. Structured illumination with particle averaging reveals novel roles for yeast centrosome components during duplication. Elife 2015, 4. [Google Scholar] [CrossRef] [PubMed]
- Araki, Y.; Lau, C.K.; Maekawa, H.; Jaspersen, S.L.; Giddings, T.H.; Schiebel, E.; Winey, M. The Saccharomyces cerevisiae spindle pole body (SPB) component Nbp1p is required for SPB membrane insertion and interacts with the integral membrane proteins Ndc1p and Mps2p. Mol. Biol. Cell 2006, 17, 1959–1970. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Winey, M.; Hoyt, M.A.; Chan, C.; Goetsch, L.; Botstein, D.; Byers, B. NDC1: A nuclear periphery component required for yeast spindle pole body duplication. J. Cell Biol. 1993, 122, 743–751. [Google Scholar] [CrossRef] [PubMed]
- Winey, M.; Goetsch, L.; Baum, P.; Byers, B. MPS1 and MPS2: Novel yeast genes defining distinct steps of spindle pole body duplication. J. Cell Biol. 1991, 114, 745–754. [Google Scholar] [CrossRef] [PubMed]
- Schramm, C.; Elliott, S.; Shevchenko, A.; Schiebel, E. The Bbp1p-Mps2p complex connects the SPB to the nuclear envelope and is essential for SPB duplication. EMBO J. 2000, 19, 421–433. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Muñoz-Centeno, M.C.; McBratney, S.; Monterrosa, A.; Byers, B.; Mann, C.; Winey, M. Saccharomyces cerevisiae MPS2 encodes a membrane protein localized at the spindle pole body and the nuclear envelope. Mol. Biol. Cell 1999, 10, 2393–2406. [Google Scholar] [CrossRef] [PubMed]
- Kupke, T.; Malsam, J.; Schiebel, E. A ternary membrane protein complex anchors the spindle pole body in the nuclear envelope in budding yeast. J. Biol. Chem. 2017, 292, 8447–8458. [Google Scholar] [CrossRef] [PubMed]
- Kupke, T.; Di Cecco, L.; Müller, H.M.; Neuner, A.; Adolf, F.; Wieland, F.; Nickel, W.; Schiebel, E. Targeting of Nbp1 to the inner nuclear membrane is essential for spindle pole body duplication. EMBO J. 2011, 30, 3337–3352. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Drin, G.; Antonny, B. Amphipathic helices and membrane curvature. FEBS Lett. 2010, 584, 1840–1847. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, K.; Kanbe, T. Mitosis in the fission yeast Schizosaccharomyces pombe as revealed by freeze-substitution electron microscopy. J. Cell Sci. 1986, 80, 253–268. [Google Scholar] [PubMed]
- Uzawa, S.; Li, F.; Jin, Y.; McDonald, K.L.; Braunfeld, M.B.; Agard, D.A.; Cande, W.Z. Spindle pole body duplication in fission yeast occurs at the G1/S boundary but maturation is blocked until exit from S by an event downstream of cdc10+. Mol. Biol. Cell 2004, 15, 5219–5230. [Google Scholar] [CrossRef] [PubMed]
- Bouhlel, I.B.; Scheffler, K.; Tran, P.T.; Paoletti, A. Monitoring SPB biogenesis in fission yeast with high resolution and quantitative fluorescent microscopy. Methods Cell. Biol. 2015, 129, 383–392. [Google Scholar] [PubMed]
- Bestul, A.J.; Yu, Z.; Unruh, J.R.; Jaspersen, S.L. Molecular model of fission yeast centrosome assembly determined by superresolution imaging. J. Cell Biol. 2017, 216, 2409–2424. [Google Scholar] [CrossRef] [PubMed]
- Bouhlel, I.B.; Ohta, M.; Mayeux, A.; Bordes, N.; Dingli, F.; Boulanger, J.; Velve Casquillas, G.; Loew, D.; Tran, P.T.; Sato, M.; et al. Cell cycle control of spindle pole body duplication and splitting by Sfi1 and Cdc31 in fission yeast. J. Cell Sci. 2015, 128, 1481–1493. [Google Scholar] [CrossRef] [PubMed]
- Paoletti, A.; Bordes, N.; Haddad, R.; Schwartz, C.L.; Chang, F.; Bornens, M. Fission yeast cdc31p is a component of the half-bridge and controls SPB duplication. Mol. Biol. Cell 2003, 14, 2793–2808. [Google Scholar] [CrossRef] [PubMed]
- Rosenberg, J.A.; Tomlin, G.C.; McDonald, W.H.; Snydsman, B.E.; Muller, E.G.; Yates, J.R., III; Gould, K.L. Ppc89 links multiple proteins, including the septation initiation network, to the core of the fission yeast spindle-pole body. Mol. Biol. Cell 2006, 17, 3793–3805. [Google Scholar] [CrossRef] [PubMed]
- West, R.R.; Vaisberg, E.V.; Ding, R.; Nurse, P.; McIntosh, J.R. cut11(+): A gene required for cell cycle-dependent spindle pole body anchoring in the nuclear envelope and bipolar spindle formation in Schizosaccharomyces pombe. Mol. Biol. Cell 1998, 9, 2839–2855. [Google Scholar] [CrossRef] [PubMed]
- Bridge, A.J.; Morphew, M.; Bartlett, R.; Hagan, I.M. The fission yeast SPB component Cut12 links bipolar spindle formation to mitotic control. Genes Dev. 1998, 12, 927–942. [Google Scholar] [CrossRef] [PubMed]
- Hagan, I.M. The spindle pole body plays a key role in controlling mitotic commitment in the fission yeast Schizosaccharomyces pombe. Biochem. Soc. Trans. 2008, 36, 1097–1101. [Google Scholar] [CrossRef] [PubMed]
- Tallada, V.A.; Tanaka, K.; Yanagida, M.; Hagan, I.M. The S. pombe mitotic regulator Cut12 promotes spindle pole body activation and integration into the nuclear envelope. J. Cell Biol. 2009, 185, 875–888. [Google Scholar] [CrossRef] [PubMed]
- Hagan, I.; Yanagida, M. The product of the spindle formation gene sad1+ associates with the fission yeast spindle pole body and is essential for viability. J. Cell Biol. 1995, 129, 1033–1047. [Google Scholar] [CrossRef] [PubMed]
- Miki, F.; Kurabayashi, A.; Tange, Y.; Okazaki, K.; Shimanuki, M.; Niwa, O. Two-hybrid search for proteins that interact with Sad1 and Kms1, two membrane-bound components of the spindle pole body in fission yeast. Mol. Genet. Genom. 2004, 270, 449–461. [Google Scholar] [CrossRef] [PubMed]
- Walde, S.; King, M.C. The KASH protein Kms2 coordinates mitotic remodeling of the spindle pole body. J. Cell Sci. 2014, 127, 3625–3640. [Google Scholar] [CrossRef] [PubMed]
- Funabiki, H.; Hagan, I.; Uzawa, S.; Yanagida, M. Cell cycle-dependent specific positioning and clustering of centromeres and telomeres in fission yeast. J. Cell Biol. 1993, 121, 961–976. [Google Scholar] [CrossRef] [PubMed]
- Chikashige, Y.; Ding, D.Q.; Funabiki, H.; Haraguchi, T.; Mashiko, S.; Yanagida, M.; Hiraoka, Y. Telomere-led premeiotic chromosome movement in fission yeast. Science 1994, 264, 270–273. [Google Scholar] [CrossRef] [PubMed]
- Chikashige, Y.; Tsutsumi, C.; Yamane, M.; Okamasa, K.; Haraguchi, T.; Hiraoka, Y. Meiotic proteins bqt1 and bqt2 tether telomeres to form the bouquet arrangement of chromosomes. Cell. 2006, 125, 59–69. [Google Scholar] [CrossRef] [PubMed]
- Fernandez-Alvarez, A.; Bez, C.; O’Toole, E.T.; Morphew, M.; Cooper, J.P. Mitotic Nuclear Envelope Breakdown and Spindle Nucleation Are Controlled by Interphase Contacts between Centromeres and the Nuclear Envelope. Dev. Cell 2016, 39, 544–559. [Google Scholar] [CrossRef] [PubMed]
- Tamm, T.; Grallert, A.; Grossman, E.P.; Alvarez-Tabares, I.; Stevens, F.E.; Hagan, I.M. Brr6 drives the Schizosaccharomyces pombe spindle pole body nuclear envelope insertion/extrusion cycle. J. Cell Biol. 2011, 195, 467–484. [Google Scholar] [CrossRef] [PubMed]
- Akey, C.W.; Radermacher, M. Architecture of the Xenopus nuclear pore complex revealed by three-dimensional cryo-electron microscopy. J. Cell Biol. 1993, 122, 1–19. [Google Scholar] [CrossRef] [PubMed]
- Löschberger, A.; van de Linde, S.; Dabauvalle, M.C.; Rieger, B.; Heilemann, M.; Krohne, G.; Sauer, M. Super-resolution imaging visualizes the eightfold symmetry of gp210 proteins around the nuclear pore complex and resolves the central channel with nanometer resolution. J. Cell Sci. 2012, 125, 570–575. [Google Scholar] [CrossRef] [PubMed]
- Bui, K.H.; von Appen, A.; DiGuilio, A.L.; Ori, A.; Sparks, L.; Mackmull, M.T.; Bock, T.; Hagen, W.; Andres-Pons, A.; Glavy, J.S.; et al. Integrated structural analysis of the human nuclear pore complex scaffold. Cell 2013, 155, 1233–1243. [Google Scholar] [CrossRef] [PubMed]
- Mi, L.; Goryaynov, A.; Lindquist, A.; Rexach, M.; Yang, W. Quantifying nucleoporin stoichiometry inside single nuclear pore complexes in vivo. Sci. Rep. 2015, 5, 9372. [Google Scholar] [CrossRef] [PubMed]
- Wozniak, R.W.; Blobel, G.; Rout, M.P. POM152 is an integral protein of the pore membrane domain of the yeast nuclear envelope. J. Cell Biol. 1994, 125, 31–42. [Google Scholar] [CrossRef] [PubMed]
- Chial, H.J.; Rout, M.P.; Giddings, T.H.; Winey, M. Saccharomyces cerevisiae Ndc1p is a shared component of nuclear pore complexes and spindle pole bodies. J. Cell Biol. 1998, 143, 1789–1800. [Google Scholar] [CrossRef] [PubMed]
- Miao, M.; Ryan, K.J.; Wente, S.R. The integral membrane protein Pom34p functionally links nucleoporin subcomplexes. Genetics 2006, 172, 1441–1457. [Google Scholar] [CrossRef] [PubMed]
- Alber, F.; Dokudovskaya, S.; Veenhoff, L.M.; Zhang, W.; Kipper, J.; Devos, D.; Suprapto, A.; Karni-Schmidt, O.; Williams, R.; Chait, B.T.; et al. The molecular architecture of the nuclear pore complex. Nature 2007, 450, 695–701. [Google Scholar] [CrossRef] [PubMed]
- Onischenko, E.; Stanton, L.H.; Madrid, A.S.; Kieselbach, T.; Weis, K. Role of the Ndc1 interaction network in yeast nuclear pore complex assembly and maintenance. J. Cell Biol. 2009, 185, 475–491. [Google Scholar] [CrossRef] [PubMed]
- Hinshaw, J.E.; Milligan, R.A. Nuclear pore complexes exceeding eightfold rotational symmetry. J. Struct. Biol. 2003, 141, 259–268. [Google Scholar] [CrossRef]
- D’Angelo, M.A.; Anderson, D.J.; Richard, E.; Hetzer, M.W. Nuclear pores form de novo from both sides of the nuclear envelope. Science 2006, 312, 440–443. [Google Scholar] [CrossRef] [PubMed]
- Otsuka, S.; Bui, K.H.; Schorb, M.; Hossain, M.J.; Politi, A.Z.; Koch, B.; Eltsov, M.; Beck, M.; Ellenberg, J. Nuclear pore assembly proceeds by an inside-out extrusion of the nuclear envelope. Elife 2016, 5. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Neuner, A.; Ruthnick, D.; Sachsenheimer, T.; Luchtenborg, C.; Brugger, B.; Schiebel, E. Brr6 and Brl1 locate to nuclear pore complex assembly sites to promote their biogenesis. J. Cell Biol. 2018, 217, 877–894. [Google Scholar] [CrossRef] [PubMed]
- Ito, T.; Chiba, T.; Ozawa, R.; Yoshida, M.; Hattori, M.; Sakaki, Y. A comprehensive two-hybrid analysis to explore the yeast protein interactome. Proc. Natl. Acad. Sci. USA 2001, 98, 4569–4574. [Google Scholar] [CrossRef] [PubMed]
- Costanzo, M.; VanderSluis, B.; Koch, E.N.; Baryshnikova, A.; Pons, C.; Tan, G.; Wang, W.; Usaj, M.; Hanchard, J.; Lee, S.D.; et al. A global genetic interaction network maps a wiring diagram of cellular function. Science 2016, 353. [Google Scholar] [CrossRef] [PubMed]
- Niepel, M.; Strambio-de-Castillia, C.; Fasolo, J.; Chait, B.T.; Rout, M.P. The nuclear pore complex-associated protein, Mlp2p, binds to the yeast spindle pole body and promotes its efficient assembly. J. Cell Biol. 2005, 170, 225–235. [Google Scholar] [CrossRef] [PubMed]
- Sezen, B.; Seedorf, M.; Schiebel, E. The SESA network links duplication of the yeast centrosome with the protein translation machinery. Genes Dev. 2009, 23, 1559–1570. [Google Scholar] [CrossRef] [PubMed]
- Witkin, K.L.; Friederichs, J.M.; Cohen-Fix, O.; Jaspersen, S.L. Changes in the nuclear envelope environment affect spindle pole body duplication in Saccharomyces cerevisiae. Genetics 2010, 186, 867–883. [Google Scholar] [CrossRef] [PubMed]
- Lo Presti, L.; Cockell, M.; Cerutti, L.; Simanis, V.; Hauser, P.M. Functional characterization of Pneumocystis carinii brl1 by transspecies complementation analysis. Eukaryot. Cell 2007, 6, 2448–2452. [Google Scholar] [CrossRef] [PubMed]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rüthnick, D.; Schiebel, E. Duplication and Nuclear Envelope Insertion of the Yeast Microtubule Organizing Centre, the Spindle Pole Body. Cells 2018, 7, 42. https://doi.org/10.3390/cells7050042
Rüthnick D, Schiebel E. Duplication and Nuclear Envelope Insertion of the Yeast Microtubule Organizing Centre, the Spindle Pole Body. Cells. 2018; 7(5):42. https://doi.org/10.3390/cells7050042
Chicago/Turabian StyleRüthnick, Diana, and Elmar Schiebel. 2018. "Duplication and Nuclear Envelope Insertion of the Yeast Microtubule Organizing Centre, the Spindle Pole Body" Cells 7, no. 5: 42. https://doi.org/10.3390/cells7050042
APA StyleRüthnick, D., & Schiebel, E. (2018). Duplication and Nuclear Envelope Insertion of the Yeast Microtubule Organizing Centre, the Spindle Pole Body. Cells, 7(5), 42. https://doi.org/10.3390/cells7050042




