TRPV1 and Endocannabinoids: Emerging Molecular Signals that Modulate Mammalian Vision
Abstract
:1. Introduction
2. TRPV1 in the Brain
2.1. TRPV1 Expression in the Brain
3. TRPV1 in the Retina
3.1. Overview of Retinal Anatomy and Early Visual Information Processing
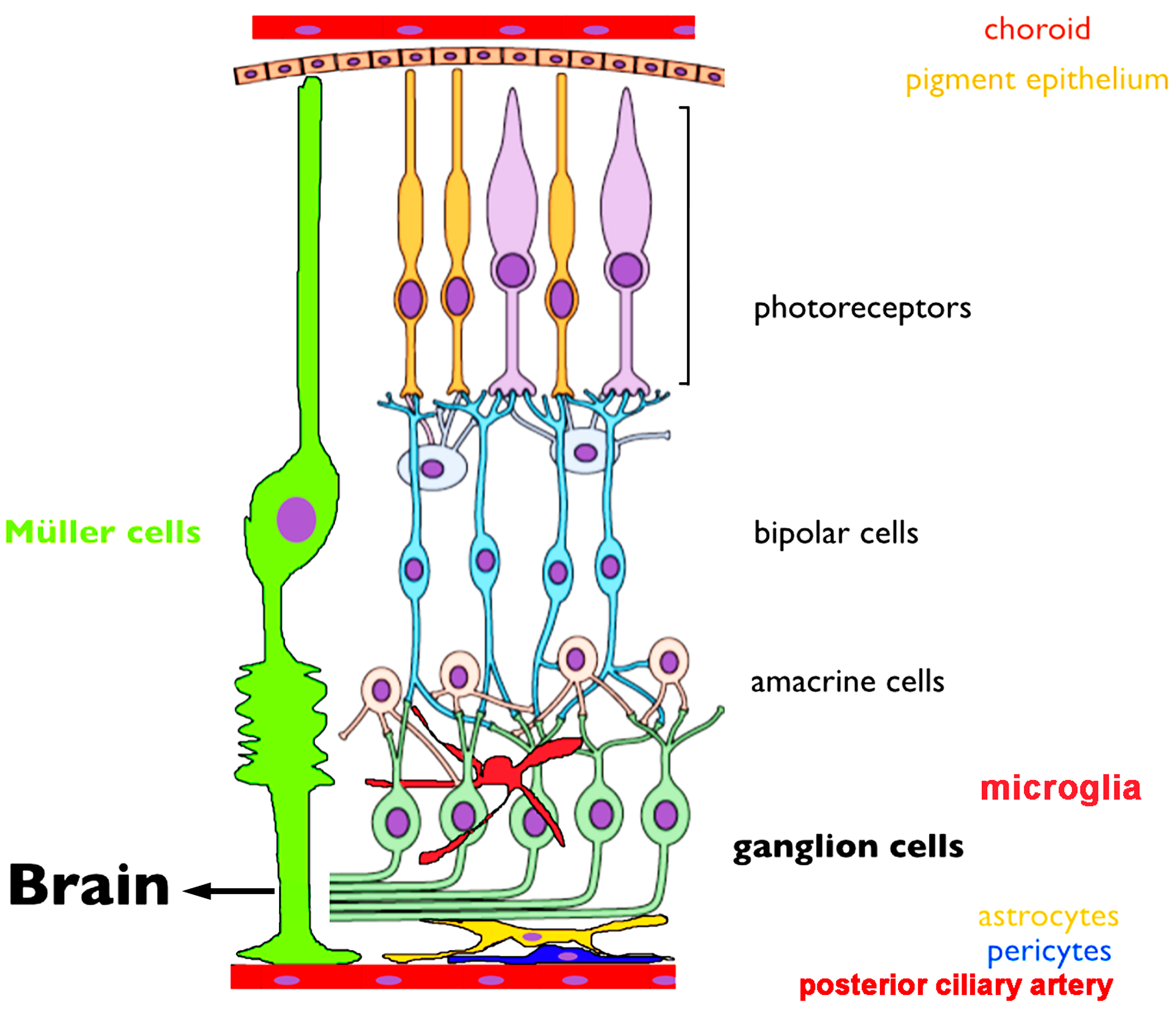
3.2. TRPV1 Channel Distribution in the Retina
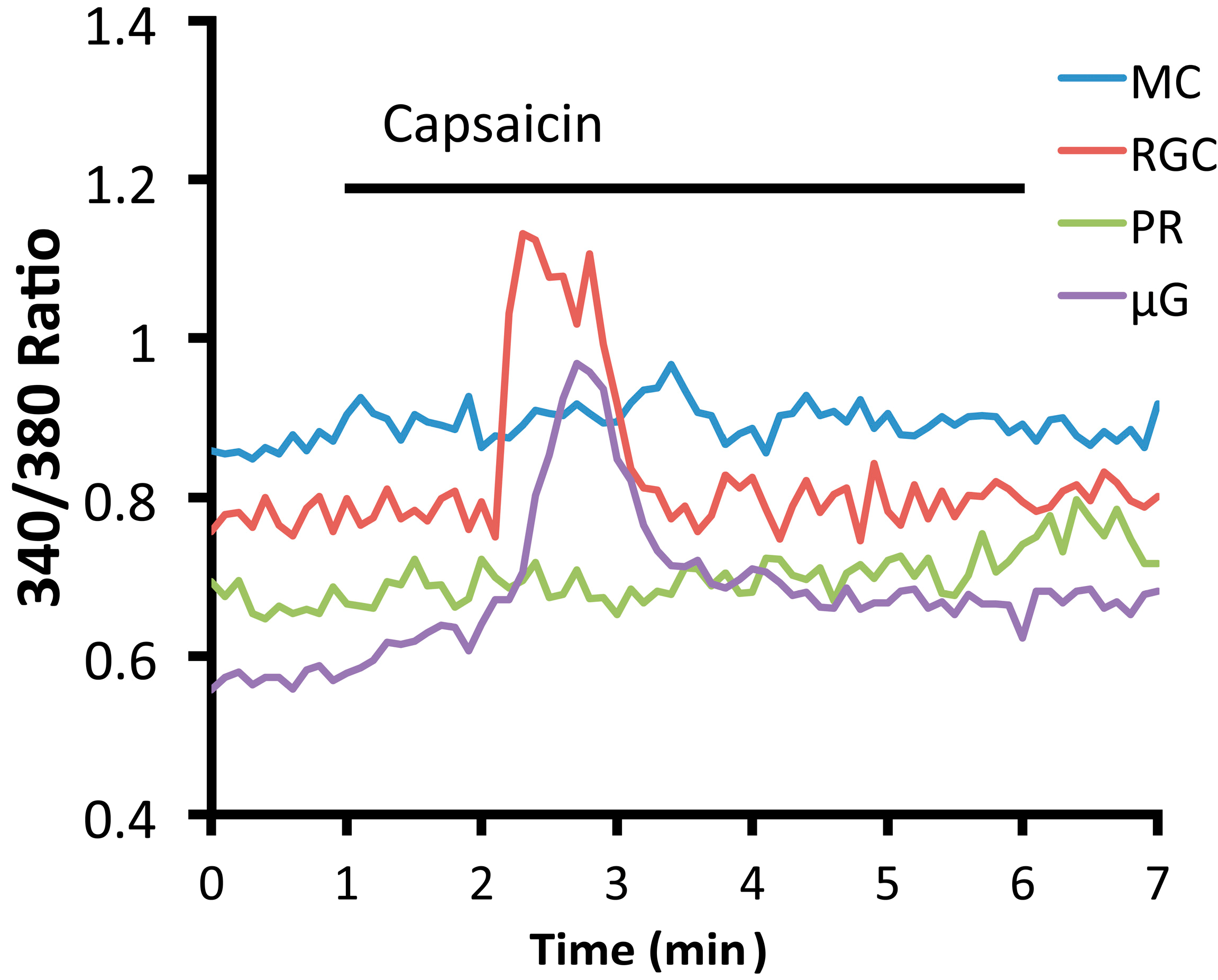
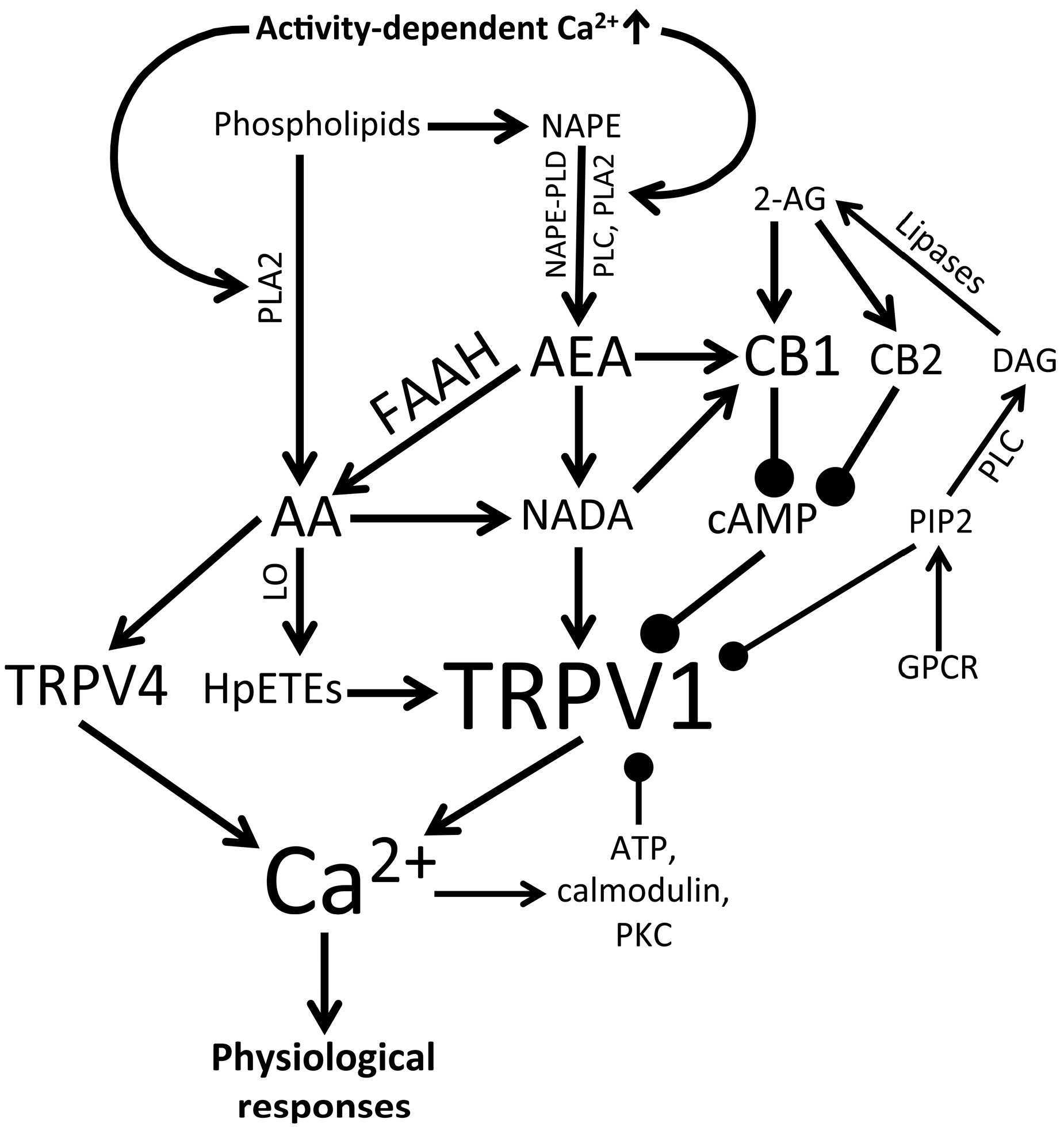
3.3. Endogenous and Synthetic TRPV1 Agonists and Retinal Signaling
3.4. Is TRPV1 Neurodegenerative or Neuroprotective?
3.5. TRPV1 and Retinal Vasoregulation
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Minke, B. Light-induced reduction in excitation efficiency in the trp mutant of Drosophila. J. Gen. Physiol. 1982, 79, 361–385. [Google Scholar] [CrossRef]
- Caterina, M.J.; Schumacher, M.A.; Tominaga, M.; Rosen, T.A.; Levine, J.D.; Julius, D. The capsaicin receptor: A heat-activated ion channel in the pain pathway. Nature 1997, 389, 816–824. [Google Scholar] [CrossRef]
- Clapham, D.E. TRP channels as cellular sensors. Nature 2003, 426, 517–524. [Google Scholar] [CrossRef]
- Suzuki, M.; Mizuno, A.; Kodaira, K.; Imai, M. Impaired pressure sensation in mice lacking TRPV4. J. Biol. Chem. 2003, 278, 22664–22668. [Google Scholar] [CrossRef]
- Watanabe, H.; Vriens, J.; Prenen, J.; Droogmans, G.; Voets, T.; Nilius, B. Anandamide and arachidonic acid use epoxyeicosatrienoic acid to activate TRPV4 channels. Nature 2003, 424, 434–438. [Google Scholar] [CrossRef]
- Kraft, R.; Harteneck, C. The mammalian melastatin-related transient receptor potential cation channels: An overview. Pflugers Arch. 2005, 451, 204–211. [Google Scholar] [CrossRef]
- Phelps, C.B.; Wang, R.R.; Choo, S.S.; Gaudet, R. Differential regulation of TRPV1, TRPV3, and TRPV4 sensitivity through a conserved binding site on the ankyrin repeat domain. J. Biol. Chem. 2010, 285, 731–740. [Google Scholar] [CrossRef]
- Nilius, B.; Owsianik, G. Transient receptor potential channelopathies. Pflugers Arch. 2010, 460, 437–450. [Google Scholar] [CrossRef]
- Kraft, R.; Grimm, C.; Grosse, K.; Hoffmann, A.; Sauerbruch, S.; Kettenmann, H.; Schultz, G.; Harteneck, C. Hydrogen peroxide and ADP-ribose induce TRPM2-mediated calcium influx and cation currents in microglia. Am. J. Physiol. Cell Physiol. 2004, 286, 129–137. [Google Scholar]
- Shen, Y.; Heimel, J.A.; Kamermans, M.; Peachey, N.S.; Gregg, R.G.; Nawy, S. A transient receptor potential-like channel mediates synaptic transmission in rod bipolar cells. J. Neurosci. 2009, 29, 6088–6093. [Google Scholar] [CrossRef]
- Gavva, N.R.; Bannon, A.W.; Surapaneni, S.; Hovland, D.N.; Lehto, S.G.; Gore, A.; Juan, T.; Deng, H.; Han, B.; Klionsky, L.; et al. The vanilloid receptor TRPV1 is tonically activated in vivo and involved in body temperature regulation. J. Neurosci. 2007, 27, 3366–3374. [Google Scholar] [CrossRef]
- Sudbury, J.R.; Ciura, S.; Sharif-Naeini, R.; Bourque, C.W. Osmotic and thermal control of magnocellular neurosecretory neurons-role of an N-terminal variant of TRPV1. Eur. J. Neurosci. 2010, 32, 2022–2030. [Google Scholar] [CrossRef]
- Dunn, K.M.; Hill-Eubanks, D.C.; Liedtke, W.B.; Nelson, M.T. TRPV4 channels stimulate Ca2+-induced Ca2+ release in astrocytic endfeet and amplify neurovascular coupling responses. Proc. Natl. Acad. Sci. U.S.A. 2013, 110, 6157–6162. [Google Scholar] [CrossRef]
- Filosa, J.A.; Yao, X.; Rath, G. TRPV4 and the regulation of vascular tone. J. Cardiovasc. Pharmacol. 2013, 61, 113–119. [Google Scholar] [CrossRef]
- Caterina, M.J.; Leffler, A.; Malmberg, A.B.; Martin, W.J.; Trafton, J.; Petersen-Zeitz, K.R.; Koltzenburg, M.; Basbaum, A.I.; Julius, D. Impaired nociception and pain sensation in mice lacking the capsaicin receptor. Sci. 2000, 288, 306–313. [Google Scholar] [CrossRef]
- Ward, N.J.; Ho, K.W.; Lambert, W.S.; Weitlauf, C.; Calkins, D.J. Absence of transient receptor potential vanilloid-1 accelerates stress-induced axonopathy in the optic projection. J. Neurosci. 2014, 34, 3161–3170. [Google Scholar] [CrossRef]
- Ryskamp, D.A.; Witkovsky, P.; Barabas, P.; Huang, W.; Koehler, C.; Akimov, N.P.; Lee, S.H.; Chauhan, S.; Xing, W.; Renteria, R.C.; et al. The polymodal ion channel transient receptor potential vanilloid 4 modulates calcium flux, spiking rate, and apoptosis of mouse retinal ganglion cells. J. Neurosci. 2011, 31, 7089–7101. [Google Scholar] [CrossRef]
- Gilliam, J.C.; Wensel, T.G. TRP channel gene expression in the mouse retina. Vis. Res. 2011, 51, 2440–2452. [Google Scholar] [CrossRef]
- Molnar, T.; Barabas, P.; Birnbaumer, L.; Punzo, C.; Kefalov, V.; Križaj, D. Store-operated channels regulate intracellular calcium in mammalian rods. J. Physiol. 2012, 590, 3465–3481. [Google Scholar] [CrossRef]
- Tóth, A.; Czikora, Á.; Pásztor, E.T.; Dienes, B.; Bai, P.; Csernoch, L.; Rutkai, I.; Csató, V.; Mányiné, I.S.; Pórszász, R.; et al. Vanilloid receptor-1 (TRPV1) expression and function in the vasculature of the rat. J. Histochem. Cytochem. 2014, 62, 129–144. [Google Scholar] [CrossRef]
- Sekaran, S.; Lall, G.S.; Ralphs, K.L.; Wolstenholme, A.J.; Lucas, R.J.; Foster, R.G.; Hankins, M.W. 2-Aminoethoxydiphenylborane is an acute inhibitor of directly photosensitive retinal ganglion cell activity in vitro and in vivo. J. Neurosci. 2007, 27, 3981–3986. [Google Scholar]
- Xue, T.; Do, M.T.H.; Riccio, A.; Jiang, Z.; Hsieh, J.; Wang, H.C.; Merbs, S.L.; Welsbie, D.S.; Yoshioka, T.; Weissgerber, P.; et al. Melanopsin signaling in mammalian iris and retina. Nature 2011, 479, 67–73. [Google Scholar] [CrossRef]
- Albert, E.S.; Bec, J.M.; Desmadryl, G.; Chekroud, K.; Travo, C.; Gaboyard, S.; Bardin, F.; Marc, I.; Dumas, M.; Lenaers, G.; et al. TRPV4 channels mediate the infared laser-evoked response in sensory neurons. J. Neurophysiol. 2012, 107, 3227–3234. [Google Scholar] [CrossRef]
- Montell, C.; Jones, K.; Hafen, E.; Rubin, G. Rescue of the Drosophila phototransduction mutation trp by germline transformation. Science 1985, 230, 1040–1043. [Google Scholar]
- Morgans, C.W.; Zhang, J.; Jeffrey, B.G.; Nelson, S.M.; Burke, N.S.; Duvoisin, R.M.; Brown, R.L. TRPM1 is required for the depolarizing light response in retinal ON-bipolar cells. Proc. Natl. Acad. Sci. U.S.A. 2009, 106, 19174–19178. [Google Scholar]
- Krol, J.; Busskamp, V.; Markiewicz, I.; Stadler, M.B.; Ribi, S.; Richter, J.; Duebel, J.; Bicker, S.; Fehling, H.J.; Schübeler, D.; et al. Characterizing light-regulated retinal microRNAs reveals rapid turnover as a common property of neuronal microRNAs. Cell 2010, 141, 618–631. [Google Scholar] [CrossRef]
- Moiseenkova-Bell, V.Y.; Stanciu, L.A.; Serysheva, I.I.; Tobe, B.J.; Wensel, T.G. Structure of TRPV1 channel revealed by electron cryomicroscopy. Proc. Natl. Acad. Sci. U.S.A. 2008, 105, 7451–7455. [Google Scholar] [CrossRef]
- Arniges, M.; Fernández-Fernández, J.M.; Albrecht, N.; Schaefer, M.; Valverde, M.A. Human TRPV4 channel splice variants revealed a key role of ankyrin domains in multimerization and trafficking. J. Biol. Chem. 2006, 281, 1580–1586. [Google Scholar] [CrossRef]
- Lishko, P.V.; Procko, E.; Jin, X.; Phelps, C.B.; Gaudet, R. The ankyrin repeats of TRPV1 bind multiple ligands and modulate channel sensitivity. Neuron 2007, 54, 905–918. [Google Scholar]
- Everaerts, W.; Nilius, B.; Owsianik, G. The vanilloid transient receptor potential channel TRPV4: From structure to disease. Prog. Biophys. Mol. Biol. 2010, 103, 2–17. [Google Scholar] [CrossRef]
- Tominaga, M.; Caterina, M.J.; Malmberg, A.B.; Rosen, T.A.; Gilbert, H.; Skinner, K.; Raumann, B.E.; Basbaum, A.I.; Julius, D. The cloned capsaicin receptor integrates multiple pain-producing stimuli. Neuron 1998, 21, 531–543. [Google Scholar] [CrossRef]
- Numazaki, M.; Tominaga, T.; Takeuchi, K.; Murayama, N.; Toyooka, H.; Tominaga, M. Structural determinant of TRPV1 desensitization interacts with calmodulin. Proc. Natl. Acad. Sci. U.S.A. 2003, 100, 8002–8006. [Google Scholar] [CrossRef]
- Wu, Z.Z.; Chen, S.R.; Pan, H.L. Transient receptor potential vanilloid type 1 activation down-regulates voltage-gated calcium channels through calcium-dependent calcineurin in sensory neurons. J. Biol. Chem. 2005, 280, 18142–18151. [Google Scholar] [CrossRef]
- Hellwig, N.; Albrecht, N.; Harteneck, C.; Schultz, G.; Schaefer, M. Homo- and heteromeric assembly of TRPV channel subunits. J. Cell Sci. 2005, 118, 917–928. [Google Scholar] [CrossRef]
- Prescott, E.D.; Julius, D. A modular PIP2 binding site as a determinant of capsaicin receptor. Science 2003, 300, 1284–1288. [Google Scholar] [CrossRef]
- Van der Stelt, M.; Di Marzo, V. Endovanilloids. Eur. J. Biochem. 2004, 271, 1827–1834. [Google Scholar]
- Woo, D.H.; Jung, S.J.; Zhu, M.H.; Park, C.K.; Kim, Y.H.; Oh, S.B.; Lee, C.J. Direct activation of transient receptor potential vanilloid 1 (TRPV1) by diacylglycerol (DAG). Mol. Pain 2008, 4, 1744–8069. [Google Scholar]
- Zhang, X.; Huang, J.; McNaughton, P.A. NGF rapidly increases membrane expression of TRPV1 heat-gated ion channels. Neuromol. Med. 2005, 24, 4211–4223. [Google Scholar]
- Sanz-Salvador, L.; Andrés-Borderia, A.; Ferrer-Montiel, A.; Planells-Cases, R. Agonist- and Ca2+-dependent desensitization of TRPV1 channel targets the receptor to lysosomes for degradation. J. Biol. Chem. 2012, 287, 19462–19471. [Google Scholar]
- Christoph, T.; Grünweller, A.; Mika, J.; Schäfer, M.K.; Wade, E.J.; Weihe, E.; Erdmann, V.A.; Frank, R.; Gillen, C.; Kurreck, J. Silencing of vanilloid receptor TRPV1 by RNAi reduces neuropathic and visceral pain in vivo. Biochem. Biophys. Res. Commun. 2006, 350, 238–243. [Google Scholar] [CrossRef]
- McNamara, F.N.; Randall, A.; Gunthorpe, M.J. Effects of piperine, the pungent component of black pepper, at the human vanilloid receptor (TRPV1). Br. J. Pharmacol. 2005, 144, 781–790. [Google Scholar] [CrossRef]
- Prager-Khoutorsky, M.; Khoutorsky, A.; Bourque, C.W. Unique Interweaved Microtubule Scaffold Mediates Osmosensory Transduction via Physical Interaction with TRPV1. Neuron 2014, 83, 866–878. [Google Scholar] [CrossRef]
- Immke, D.C.; Gavva, N.R. The TRPV1 receptor and nociception. Semin. Cell Dev. Biol. 2006, 17, 582–591. [Google Scholar] [CrossRef]
- Vigna, S.R.; Shahid, R.A.; Nathan, J.D.; McVey, D.C.; Liddle, R.A. Leukotriene B4 mediates inflammation via TRPV1 in duct obstruction-induced pancreatitis in rats. Pancreas 2011, 40, 708–714. [Google Scholar] [CrossRef]
- Bhave, G.; Hu, H.J.; Glauner, K.S.; Zhu, W.; Wang, H.; Brasier, D.J.; Oxford, G.S.; Gereau, R.W. Protein kinase C phosphorylation sensitizes but does not activate the capsaicin receptor transient receptor potential vanilloid 1 (TRPV1). Proc. Natl. Acad. Sci. U.S.A. 2003, 100, 12480–12485. [Google Scholar]
- Mandadi, S.; Tominaga, T.; Numazaki, M.; Murayama, N.; Saito, N.; Armati, P.J.; Roufogalis, B.D.; Tominaga, M. Increased sensitivity of desensitized TRPV1 by PMA occurs through PKCε-mediated phosphorylation at S800. Pain 2006, 123, 106–116. [Google Scholar] [CrossRef]
- Tominaga, M.; Wada, M.; Masu, M. Potentiation of capsaicin receptor activity by metabotropic ATP receptors as a possible mechanism for ATP-evoked pain and hyperalgesia. Proc. Natl. Acad. Sci. U.S.A. 2001, 98, 6951–6956. [Google Scholar] [CrossRef]
- Puntambekar, P.; Van Buren, J.; Raisinghani, M.; Premkumar, L.S.; Ramkumar, V. Direct interaction of adenosine with the TRPV1 channel protein. J. Neurosci. 2004, 29, 153–158. [Google Scholar]
- Nagy, I.; Sántha, P.; Jancsó, G.; Urbán, L. The role of the vanilloid (capsaicin) receptor (TRPV1) in physiology and pathology. Eur. J. Pharmacol. 2004, 500, 351–369. [Google Scholar]
- Sanchez, J.F.; Krause, J.E.; Cortright, D.N. The distribution and regulation of vanilloid receptor VR1 and VR1 5' splice variant RNA expression in rat. Neurosci. 2001, 107, 373–381. [Google Scholar]
- Cavanaugh, D.J.; Chesler, A.T.; Jackson, A.C.; Sigal, Y.M.; Yamanaka, H.; Grant, R.; O'Donnell, D.; Nicoll, R.A.; Shah, N.M.; Julius, D.; et al. TRPV1 reporter mice reveal highly restricted brain distribution and functional expression in arteriolar smooth muscle cells. J. Neurosci. 2011, 31, 5067–5077. [Google Scholar] [CrossRef]
- Han, L.; Ma, C.; Liu, Q.; Weng, H.-J.; Cui, Y.; Tang, Z.; Kim, Y.; Nie, H.; Qu, L.; Patel, K.N.; et al. A subpopulation of nociceptors specifically linked to itch. Nat. Neurosci. 2013, 16, 174–182. [Google Scholar]
- Di Marzo, V. Targeting the endocannabinoid system: To enhance or reduce? Nat. Rev. 2008, 7, 438–455. [Google Scholar]
- Kauer, J.A.; Gibson, H.E. Body-temperature maintenance as the predominant function of the vanilloid receptor TRPV1. Trends in Neurosci. 2009, 32, 215–224. [Google Scholar] [CrossRef]
- Martins, D.; Tavares, I.; Morgado, C. "Hotheaded": The role of TRPV1 in brain functions. Neuropharmacol. 2014, 85, 151–157. [Google Scholar] [CrossRef]
- Marsch, R.; Foeller, E.; Rammes, G.; Bunck, M.; Kössl, M.; Holsboer, F.; Zieglgänsberger, W.; Landgraf, R.; Lutz, B.; Wotjak, C.T. Reduced anxiety, conditioned fear, and hippocampal long-term potentiation in transient receptor potential vanilloid type 1 receptor-deficient mice. J. Neurosci. 2007, 27, 832–839. [Google Scholar] [CrossRef]
- Gibson, H.E.; Edwards, J.G.; Page, R.S.; Van Hook, M.J.; Kauer, J.A. TRPV1 channels mediate long-term depression at synapses on hippocampal interneurons. Neuron 2008, 57, 746–759. [Google Scholar]
- Adamczyk, P.; Miszkiel, J.; McCreary, A.C.; Filip, M.; Papp, M.; Przegaliński, E. The effects of cannabinoid CB1, CB2 and vanilloid TRPV1 receptor antagonists on cocaine addictive behavior in rats. Brain Res. 2012, 1444, 45–54. [Google Scholar]
- Osaka, T.; Kobayashi, A.; Lee, T.H.; Namba, Y.; Inoue, S.; Kimura, S. Lack of integrative control of heat production and heat loss after capsaicin administration. Pflugers Arch. 2000, 440, 440–445. [Google Scholar] [CrossRef]
- Dawbarn, D.; Harmar, A.J.; Pycock, C.J. Intranigral injection of capsaicin enhances motor activity and depletes nigral 5-hydroxytrptamine but not substance P. Neuropharmacol. 1981, 20, 341–346. [Google Scholar] [CrossRef]
- Szallasi, A.; Nilsson, S.; Farkas-Szallasi, T.; Blumberg, P.M.; Hökfelt, T.; Lundberg, J.M. Vanilloid (capsaicin) receptors in the rat: Distribution in the brain, regional differences in the spinal cord, axonal transport to the periphery, and depletion by systemic vanilloid treatment. Brain Res. 1995, 703, 175–183. [Google Scholar]
- Benninger, F.; Freund, T.F.; Hájos, N. Control of excitatory synaptic transmission by capsaicin is unaltered in TRPV1 vanilloid receptor knockout mice. Neurochem. Int. 2008, 52, 89–94. [Google Scholar] [CrossRef]
- Everaerts, W.; Sepúlveda, M.R.; Gevaert, T.; Roskams, T.; Nilius, B.; De Ridder, D. Where is TRPV1 expressed in the bladder, do we see the real channel? Naunyn Schmiedebergs Arch Pharmacol. 2009, 379, 421–425. [Google Scholar] [CrossRef]
- Brown, T.E.; Chirila, A.M.; Schrank, B.R.; Kauer, J.A. Loss of interneuron LTD and attenuated pyramidal cell LTP in TRPV1 and TRPV3 KO mice. Hippocampus 2013, 23, 662–671. [Google Scholar] [CrossRef]
- Ritter, S.; Dinh, T.T. Capsaicin-induced neuronal degeneration: Silver impregnation of cell bodies, axons, and terminals in the central nervous system of the adult rat. J. Comp. Neurol. 1988, 271, 79–90. [Google Scholar] [CrossRef]
- Ritter, S.; Dinh, T. Capsaicin-induced neuronal degeneration in the brain and retina of preweanling rats. J. Comp. Neurol. 1990, 296, 447–461. [Google Scholar] [CrossRef]
- Marc, R.E.; Jones, B.W.; Watt, C.B.; Anderson, J.R.; Sigulinsky, C.; Lauritzen, S. Retinal connectomics: Towards complete, accurate networks. Prog. Retin. Eye Res. 2013, 37, 141–162. [Google Scholar] [CrossRef]
- Bringmann, A.; Iandiev, I.; Pannicke, T.; Wurm, A.; Hollborn, M.; Wiedemann, P.; Osborne, N.N.; Reichenbach, A. Cellular signaling and factors involved in Müller cell gliosis: Neuroprotective and detrimental effects. Prog. Retin. Eye Res. 2009, 28, 423–451. [Google Scholar] [CrossRef]
- Cuenca, N.; Fernández-Sánchez, L.; Campello, L.; Maneu, V.; De la Villa, P.; Lax, P.; Pinilla, I. Cellular responses following retinal injuries and therapeutic approaches for neurodegenerative diseases. Prog. Retin. Eye Res. 2014, in press. [Google Scholar]
- Chalupa, L.M.; Werner, J.S. The Visual Neurosciences; MIT Press: Cambridge, MA, USA, 2003. [Google Scholar]
- Sappington, R.M.; Calkins, D.J. Contribution of TRPV1 to microglia-derived IL-6 and NFκB translocation with elevated hydrostatic pressure. Investig. Ophthalmol. Vis. Sci. 2008, 49, 3004–3017. [Google Scholar] [CrossRef]
- Sappington, R.M.; Sidorova, T.; Long, D.J.; Calkins, D.J. TRPV1: Contribution to retinal ganglion cell apoptosis and increased intracellular Ca2+ with exposure to hydrostatic pressure. Investig. Ophthalmol. Vis. Sci. 2009, 50, 717–728. [Google Scholar]
- Križaj, D.; Ryskamp, D.A.; Tian, N.; Tezel, G.; Mitchell, C.H.; Slepak, V.Z.; Shestopalov, V.I. From mechanosensitivity to inflammatory responses: New players in the pathology of glaucoma. Curr. Eye Res. 2014, 39, 105–119. [Google Scholar] [CrossRef]
- Szikra, T.; Cusato, K.; Thoreson, W.B.; Barabas, P.; Bartoletti, T.M.; Križaj, D. Depletion of calcium stores regulates calcium influx and signal transmission in rod photoreceptors. J. Physiol. 2008, 586, 4859–4875. [Google Scholar] [CrossRef]
- Beckel, J.M.; Argall, A.J.; Lim, J.C.; Xia, J.; Lu, W.; Coffey, E.E.; Macarak, E.J.; Shahidullah, M.; Delamere, N.A.; Zode, G.S.; et al. Mechanosensitive release of adenosine 5'-triphosphate through pannexin channels and mechanosensitive upregulation of pannexin channels in optic nerve head astrocytes: A mechanism for purinergic involvement in chronic strain. Glia 2014, 62, 1486–1501. [Google Scholar] [CrossRef]
- Ritter, S.; Dinh, T. Age-related changes in capsaicin-induced degeneration in rat brain. J. Comp. Neurol. 1992, 318, 103–116. [Google Scholar] [CrossRef]
- Ritter, S.; Dinh, T.T. Prior optic nerve transection reduces capsaicin-induced degeneration in rat subcortical visual structures. J. Comp. Neurol. 1991, 308, 79–90. [Google Scholar] [CrossRef]
- Jancsó, G.; Király, E.; Jancsó-Gábor, A. Direct evidence for an axonal site of action of capsaicin. Naunyn Schmiedebergs Arch. Pharmacol. 1980, 313, 91–94. [Google Scholar] [CrossRef]
- Kim, C.S.; Kawada, T.; Kim, B.S.; Han, I.S.; Choe, S.Y.; Kurata, T.; Yu, R. Capsaicin exhibits anti-inflammatory property by inhibiting IκB-a degradation in LPS-stimulated peritoneal macrophages. Cell Signal. 2003, 15, 299–306. [Google Scholar] [CrossRef]
- Costa, R.M.; Liu, L.; Nicolelis, M.A.L.; Simon, S.A. Gustatory effects of capsaicin that are independent of TRPV1 receptors. Chem. Senses 2005, 30, i198–i200. [Google Scholar] [CrossRef]
- Athanasiou, A.; Smith, P.A.; Vakilpour, S.; Kumaran, N.M.; Turner, A.E.; Bagiokou, D.; Layfield, R.; Ray, D.E.; Westwell, A.D.; Alexander, S.P.; et al. Vanilloid receptor agonists and antagonists are mitochondrial inhibitors: How vanilloids cause non-vanilloid receptor mediated cell death. Biochem. Biophys. Res. Commun. 2007, 354, 50–55. [Google Scholar]
- Di Marzo, V.; De Petrocellis, L.; Fezza, F.; Ligresti, A.; Bisogno, T. Anandamide receptors. Prostaglandins Leukot. Essent. Fat. Acids 2002, 66, 377–391. [Google Scholar] [CrossRef]
- Xu, Y.; Dhingra, A.; Fina, M.E.; Koike, C.; Furukawa, T.; Vardi, N. mGluR6 deletion renders the TRPM1 channel in retina inactive. J. Neurophysiol. 2012, 107, 948–957. [Google Scholar]
- Docherty, R.J.; Yeat, J.C.; Piper, A.S. Capsazepine block of voltage-activated calcium channels in adult rat dorsal root ganglion neurones in culture. Br. J. Pharmacol. 1997, 121, 1461–1467. [Google Scholar]
- Liu, L.; Simon, S.A. Capsazepine, a vanilloid receptor antagonist, inhibits nicotinic acetylcholine receptors in rat trigeminal ganglia. Neurosci. Lett. 1997, 228, 29–32. [Google Scholar] [CrossRef]
- Ray, A.M.; Benham, C.D.; Roberts, J.C.; Gill, C.H.; Lanneau, C.; Gitterman, D.P.; Harries, M.; Davis, J.B.; Davies, C.H. Capsazepine protects against neuronal injury caused by oxygen glucose deprivation by inhibition I(h). J. Neurosci. 2003, 23, 10146–10153. [Google Scholar]
- Yamamura, H.; Ugawa, S.; Ueda, T.; Nagao, M.; Shimada, S. Capsazepine is a novel activator of the δ subunit of the human epithelial Na+ channel. J. Biol. Chem. 2004, 279, 44483–44489. [Google Scholar]
- Nucci, C.; Gasperi, V.; Tartaglione, R.; Cerulli, A.; Terrinoni, A.; Bari, M.; De Simone, C.; Agrò, A.F.; Morrone, L.A.; Corasaniti, M.T.; et al. Involvement of the endocannabinoid system in retinal damage after high intraocular pressure-induced ischemia in rats. Investig. Ophthalmol. Vis. Sci. 2007, 48, 2997–3004. [Google Scholar] [CrossRef]
- Martinez-Garcia, M.C.; Martinez, T.; Pañeda, C.; Gallego, P.; Jimenez, A.I.; Merayo, J. Differential expression and localization of transient receptor potential vanilloid 1 in rabbit and human eyes. Histol. Histopathol. 2013, 28, 1507–1516. [Google Scholar]
- Zimov, S.; Yazulla, S. Localization of vanilloid receptor 1 (TRPV1/VR1)-like immunoreactivity in goldfish and zebrafish retinas: Restriction to photoreceptor synaptic ribbons. J. Neurocytol. 2004, 33, 441–452. [Google Scholar] [CrossRef]
- Zimov, S.; Yazulla, S. Vanilloid receptor 1 (TRPV1/VR1) co-localizes with fatty acid amide hydrolase (FAAH) in retinal amacrine cells. Vis. Neurosci. 2007, 24, 581–591. [Google Scholar]
- Leonelli, M.; Martins, D.O.; Kihara, A.H.; Britto, L.R. Ontogenetic expression of the vanilloid receptors TRPV1 and TRPV2 in the rat retina. Int. J. Dev. Neurosci. 2009, 27, 709–718. [Google Scholar]
- Leonelli, M.; Martins, D.O.; Britto, L.R. TRPV1 receptors modulate retinal development. Int. J. Dev. Neurosci. 2011, 29, 405–413. [Google Scholar] [CrossRef]
- Leonelli, M.; Martins, D.O.; Britto, L.R. TRPV1 receptors are involved in protein nitration and Müller cell reaction in the acutely axotomized rat retina. Exp. Eye Res. 2010, 91, 755–768. [Google Scholar] [CrossRef]
- Maione, S.; Cristino, L.; Migliozzi, A.L.; Georgiou, A.L.; Starowicz, K.; Salt, T.E.; Di Marzo, V. TRPV1 channels control synaptic plasticity in the developing superior colliculus. J. Physiol. 2009, 587, 2521–2535. [Google Scholar]
- Fan, S.F.; Yazulla, S. Biphasic modulation of voltage-dependent currents of retinal cones by cannabinoid CB1 receptor agonist WIN 55212-2. Vis. Neurosci. 2003, 20, 177–188. [Google Scholar]
- Warrier, A.; Wilson, M. Endocannabinoid signaling regulates spontaneous transmitter release from embryonic retinal amacrine cells. Vis. Neurosci. 2007, 24, 25–35. [Google Scholar]
- Yazulla, S. Endocannabinoids in the retina: From marijuana to neuroprotection. Prog. Retin. Eye Res. 2008, 27, 501–526. [Google Scholar] [CrossRef]
- Straiker, A.; Stella, N.; Piomelli, D.; Mackie, K.; Karten, H.J.; Maguire, G. Cannabinoid CB1 receptors and ligands in vertebrate retina: Localization and function of an endogenous signaling system. Proc. natl. Acad. Sci. U.S.A. 1999, 96, 14565–14570. [Google Scholar] [CrossRef]
- Glaser, S.T.; Deutsch, D.G.; Studholme, K.M.; Zimov, S.; Yazulla, S. Endocannabinoids in the intact retina: 3 H-anandamide uptake, fatty acid amide hydrolase immunoreactivity and hydrolysis of anandamide. Vis. Neurosci. 2005, 22, 693–705. [Google Scholar]
- Ho, K.W.; Lambert, W.S.; Calkins, D.J. Activation of TRPV1 cation channel contributes to stress-induced astrocyte migration. Glia 2014. Epub ahead of print. [Google Scholar]
- Jo, A.O.; Ryskamp, D.A.; Redmon, S.; Barabas, P.; Križaj, D. Nonretrograde endocannabinoid signaling modulates retinal ganglion cell calcium homeostasis through the TRPV1cation channel. Investig. Ophtalmol. Vis. Sci. 2014, 55. E-Abstract 3021. [Google Scholar]
- Szikra, T.; Barabas, P.; Bartoletti, T.M.; Huang, W.; Akopian, A.; Thoreson, W.B.; Križaj, D. Calcium homeostasis and cone signaling are regulated by interactions between calcium stores and plasma membrane ion channels. PLoS One 2009, 4, e6723. [Google Scholar]
- Ahluwalia, J.; Urban, L.; Bevan, S.; Nagy, I. Anandamide regulates neuropeptide release from capsaicin-sensitive primary sensory neurons by activating both the cannabinoid 1 receptor and the vanilloid receptor 1 in vitro. Eur. J. Neurosci. 2003, 12, 2611–2618. [Google Scholar] [CrossRef]
- Kishimoto, Y.; Kano, M. Endogenous cannabinoid signaling through the CB1 receptor is essential for cerebellum-dependent discrete motor learning. J. Neurosci. 2006, 26, 8829–8837. [Google Scholar] [CrossRef]
- Cadas, H.; Gaillet, S.; Beltramo, M.; Venance, L.; Piomelli, D. Biosynthesis of an endogenous cannabinoid precursor in neurons and its control by calcium and cAMP. J. Neurosci. 1996, 16, 3934–3942. [Google Scholar]
- De Petrocellis, L.; Schiano, M.A.; Imperatore, R.; Cristino, L.; Starowicz, K.; Di Marzo, V. A re-evaluation of 9-HODE activity at TRPV1 channels in comparison with anandamide: Enantioselectivity and effects at other TRP channels and in sensory neurons. Br. J. Pharmacol. 2012, 167, 1643–1651. [Google Scholar] [CrossRef]
- Tóth, B.I.; Dobrosi, N.; Dajnoki, A.; Czifra, G.; Oláh, A.; Szöllosi, A.G.; Juhász, I.; Sugawara, K.; Paus, R.; Bíró, T. Endocannabinoids modulate human epidermal keratinocyte proliferation and survival via the sequential engagement of cannabinoid receptor-1 and transient receptor potential vanilloid-1. J. Invest. Dermatol. 2011, 131, 1095–1104. [Google Scholar] [CrossRef]
- Jeske, N.A.; Patwardhan, A.M.; Gamper, N.; Price, T.J.; Akopian, A.N.; Hargreaves, K.M. Cannabinoid WIN 55,212–2 regulates TRPV1 phosphorylation in sensory neurons. J. Biol. Chem. 2006, 281, 32789–32890. [Google Scholar]
- Buckley, N.E.; Hansson, S.; Harta, G.; Mezey, E. Expression of the CB1 and CB2 receptor messenger RNAs during embryonic development in the rat. Neuroscience 1998, 82, 1131–1149. [Google Scholar]
- Porcella, A.; Maxia, C.; Gessa, G.L.; Pani, L. The human eye expresses high levels of CB1 cannabinoid receptor mRNA and protein. Eur. J. Neurosci. 2000, 12, 1123–1127. [Google Scholar] [CrossRef]
- Hu, S.S.J.; Arnold, A.; Hutchens, J.M.; Radicke, J.; Cravatt, B.F.; Wager-Miller, J.; Mackie, K.; Straiker, A. Architecture of cannabinoid signaling in mouse retina. J. Comp. Neurol. 2010, 518, 3848–3866. [Google Scholar] [CrossRef]
- Wilkinson-Berka, J.L.; Alousis, N.S.; Kelly, D.J.; Gilbert, R.E. COX-2 inhibition and retinal angiogenesis in a mouse model of retinopathy of prematurity. Invest. Ophthalmol. Vis. Sci. 2003, 44, 974–979. [Google Scholar] [CrossRef]
- Struik, M.L.; Yazulla, S.; Kamermans, M. Cannabinoid agonist WIN 55212-2 speeds up the cone response to light offset in goldfish retina. Vis. Neurosci. 2006, 23, 285–293. [Google Scholar]
- Matias, I.; Wang, J.W.; Moriello, A.S.; Nieves, A.; Woodward, D.F.; Di Marzo, V. Changes in endocannabinoid and palmitoylethanolamide levels in eye tissues of patients with diabetic retinopathy and age-related macular degeneration. Prostaglandins Leukot. Essent. Fat. Acids 2006, 75, 413–418. [Google Scholar] [CrossRef]
- Kumar, R.N.; Chambers, W.A.; Pertwee, R.G. Pharmacological actions and therapeutic uses of cannabis and cannabinoids. Anaesthesia 2001, 56, 1059–1068. [Google Scholar] [CrossRef]
- Hwang, S.W.; Cho, H.; Kwak, J.; Lee, S.Y.; Kang, C.J.; Jung, J.; Cho, S.; Min, K.H.; Suh, Y.G.; Kim, D.; et al. Direct activation of capsaicin receptors by products of lipoxygenases: Endogenous capsaicin-like substances. Proc. Natl. Acad. Sci. U.S.A. 2000, 97, 6155–6160. [Google Scholar] [CrossRef]
- Piomelli, D. The ligand that came from within. Trends in Pharmacol. Sci. 2001, 22, 17–19. [Google Scholar] [CrossRef]
- Russo, E.B.; Merzouki, A.; Mesa, J.M.; Frey, K.A.; Bach, P.J. Cannabis improves night vision: A case study of dark adaptometry and scotopic sensitivity in kif smokers of the Rif mountains of northern Morocco. J. Ethnopharmacol. 2004, 93, 99–104. [Google Scholar] [CrossRef]
- Adams, A.J.; Brown, B.; Haegerstrom-Portnoy, G.; Flom, M.C.; Jones, R.T. Evidence for acute effects of alcohol and marijuana on color discrimination. Percept. Psychophys. 1976, 20, 119–124. [Google Scholar] [CrossRef]
- Dawson, W.W.; Jimenez-Antillon, C.F.; Perez, J.M.; Zeskind, J.A. Marijuana and vision—after ten years’ use in Costa Rica. Investig. Ophtalmol. Vis. Sci. 1977, 16, 689–699. [Google Scholar]
- Straiker, A.; Sullivan, J.M. Cannabinoid receptor activation differentially modulates ion channels in photoreceptors of the tiger salamander. J. Neurophysiol. 2003, 89, 2647–2654. [Google Scholar] [CrossRef]
- Lalonde, M.R.; Jollimore, C.A.B.; Stevens, K.; Barnes, S.; Kelly, M.E.M. Cannabinoid receptor-mediated inhibition of calcium signaling in rat retinal ganglion cells. Mol. Vis. 2006, 12, 1160–1166. [Google Scholar]
- Middleton, T.P.; Protti, D.A. Cannabinoids modulate spontaneous synaptic activity in retinal ganglion cells. Vis. Neurosci. 2011, 28, 393–402. [Google Scholar] [CrossRef]
- El-Remessy, A.B.; Khalil, I.E.; Matragoon, S.; Abou-Mohamed, G.; Tsai, N.J.; Roon, P.; Caldwell, R.B.; Caldwell, R.W.; Green, K.; Liou, G.I. Neuroprotective effect of (-)Δ9-tetrahydrocannabinol and cannabidiol in N-Methyl-d-Aspartate-induced retinal neurotoxicity. Am. J. Pathol. 2003, 163, 1997–2008. [Google Scholar] [CrossRef]
- Opere, C.A.; Zheng, W.D.; Zhao, M.; Lee, J.S.; Kulkarni, K.H.; Ohia, S.E. Inhibition of potassium- and ischemia-evoked [3H] D-aspartate release from isolated bovine retina by cannabinoids. Curr. Eye Res. 2006, 31, 645–653. [Google Scholar] [CrossRef]
- Lax, P.; Esquiva, G.; Altavilla, C.; Cuenca, N. Neuroprotective effect of the cannabinoid agonist HU210 on retinal degeneration. Exp. Eye Res. 2014, 120, 175–185. [Google Scholar] [CrossRef]
- Chen, J.; Matias, I.; Dinh, T.; Lu, T.; Venezia, S.; Nieves, A.; Woodward, D.F.; Di Marzo, V. Finding of endocannabinoids in human eye tissues: Implications for glaucoma. Biochem. Biophys. Res. Commun. 2005, 330, 1062–1067. [Google Scholar] [CrossRef]
- Slusar, J.E.; Cairns, E.A.; Szczesniak, A.M.; Bradshaw, H.B.; Di Polo, A.; Kelly, M.E. The fatty acid amide hydrolase inhibitor, URB597, promotes retinal ganglion cell neuroprotection in a rat model of optic nerve axotomy. Neuropharmacology 2013, 72, 116–125. [Google Scholar] [CrossRef]
- Millns, P.J.; Chimenti, M.; Ali, N.; Ryland, E.; de Lago, E.; Fernandez-Ruiz, J.; Chapman, V.; Kendall, D.A. Effects of inhibition of fatty acid amide hydrolase vs the anandamide membrane transporter on TRPV1-mediated calcium responses in adult DRG neurons; the role of CB receptors. Eur. J. Neurosci. 2006, 24, 3489–3495. [Google Scholar] [CrossRef]
- Shin, C.Y.; Shin, J.; Kim, B.M.; Wang, M.H.; Jang, J.H.; Surh, Y.J.; Oh, U. Essential role of mitochondrial permeability transition in vanilloid receptor 1-dependent cell death of sensory neurons. Mol. Cell Neurosci. 2003, 24, 57–68. [Google Scholar] [CrossRef]
- Kim, S.R.; Lee, D.Y.; Chung, E.S.; Oh, U.; Kim, S.U.; Jin, B.K. Transient receptor potential vanilloid subtype 1 mediates cell death of mesencephalic dopaminergic neurons in vivo and in vitro. J. Neurosci. 2005, 25, 662–671. [Google Scholar] [CrossRef]
- Kim, S.R.; Kim, S.U.; Oh, U.; Jin, B.K. Transient receptor potential vanilloid subtype 1 mediates microglial cell death in vivo and in vitro via Ca2+-mediated mitochondrial damage and cytochrome c release. J. Immunol. 2006, 177, 4322–4329. [Google Scholar] [CrossRef]
- Shirakawa, H.; Yamaoka, T.; Sanpei, K.; Sasaoka, H.; Nakagawa, T.; Kaneko, S. TRPV1 stimulation triggers apoptotic cell death of rat cortical neurons. Biochem. Biophys. Res. Commun. 2008, 377, 1211–1215. [Google Scholar] [CrossRef]
- Birder, L.A.; Nakamura, N.Y.; Kiss, S.; Nealen, M.L.; Barrick, S.; Kanai, A.J.; Wang, E.; Ruiz, G.; de Groat, W.C.; Apodaca, G.; et al. Altered urinary bladder function in mice lacking the vanilloid receptor TRPV1. Nat. Neurosci. 2002, 5, 856–860. [Google Scholar] [CrossRef]
- Eijkelkamp, N.; Quick, K.; Wood, J.N. Transient receptor potential channels and mechanosensation. Neurosci. 2013, 36, 519–546. [Google Scholar]
- Ho, K.W.; Ward, N.J.; Calkins, D.J. TRPV1: A stress response protein in the central nervous system. Am. J. Neurodegener. Dis. 2012, 1, 1–14. [Google Scholar]
- Sanderson, J.; Rhodes, J.; Osborne, A.; Broadway, D. Increased hydrostatic pressure does not cause loss of retinal ganglion cell viability in human organotypic retinal cultures. Acta Ophthalmol. 2011, 89. [Google Scholar] [CrossRef]
- Burgoyne, C.F. A biomechanical paradigm for axonal insult within the optic nerve head in aging and glaucoma. Exp. Eye Res. 2011, 93, 120–132. [Google Scholar] [CrossRef]
- Veldhuis, W.B.; van der Stelt, M.; Wadman, M.W.; van Zadelhoff, G.; Maccarrone, M.; Fezza, F.; Veldink, G.A.; Vliegenthart, J.F.G.; Bär, P.R.; Nicolay, K.; et al. Neuroprotection by the endogenous cannabinoid anandamide and arvanil against in vivo excitotoxicity in the rat: Role of vanilloid receptors and lipoxygenases. J. Neurosci. 2003, 23, 4127–4133. [Google Scholar]
- Sappington, R.M.; Chan, M.; Calkins, D.J. Interleukin-6 protects retinal ganglion cells from pressure-induced death. Investig. Ophthalmol. Vis. Sci. 2006, 47, 2932–2942. [Google Scholar] [CrossRef]
- Sakamoto, K.; Kuroki, T.; Okuno, Y.; Sekiya, H.; Watanabe, A.; Sagawa, T.; Ito, H.; Mizuta, A.; Mori, A.; Nakahara, T.; et al. Activation of the TRPV1 channel attenuates N-methyl-D-aspartic acid-induced neuronal injury in the rat retina. Eur. J. Pharmacol. 2014, 733, 13–22. [Google Scholar] [CrossRef]
- Bronzetti, E.; Artico, M.; Koyacs, I.; Felici, L.M.; Magliulo, G.; Vignone, D.; D'Ambrosio, A.; Forte, F.; De Liddo, R.; Feher, J. Expression of neurotransmitters and neurotrophins in neurogenic inflammation of the rat retina. Eur. J. Histochem. 2007, 51, 251–260. [Google Scholar]
- Yang, J.H.; Guo, Z.; Zhang, T.; Meng, X.X.; Sun, T.; Wu, J. STZ treatment induced apoptosis of retinal cells and effect of up-regulation of calcitonin gene related peptide in rats. J. Diabetes Complicat. 2013, 27, 531–537. [Google Scholar] [CrossRef]
- Ching, L.C.; Kou, Y.R.; Shyue, S.K.; Su, K.H.; Wei, J.; Cheng, L.C.; Yu, Y.B.; Pan, C.C.; Lee, T.S. Molecular mechanisms of activation of endothelial nitric oxide synthase mediated by transient receptor potential vanilloid type 1. Cardiovasc. Res. 2011, 91, 492–501. [Google Scholar] [CrossRef]
- Martin, E.; Dahan, D.; Cardouat, G.; Gillibert-Duplantier, J.; Marthan, R.; Savineau, J.P.; Ducret, T. Involvement of TRPV1 and TRPV4 channels in migration of rat pulmonary arterial smooth muscle cells. Pflugers Arch. 2012, 464, 261–272. [Google Scholar] [CrossRef]
- Donnerer, J.; Lembeck, F. Analysis of the effects of intravenously injected capsaicin in the rat. Naunyn Schmiedebergs Arch. Pharmacol. 1982, 320, 54–57. [Google Scholar] [CrossRef]
- Wang, L.; Wang, D.H. TRPV1 gene knockout impairs post ischemic recovery in isolated perfused heart in mice. Circ. 2005, 112, 3617–3623. [Google Scholar] [CrossRef]
- Kark, T.; Bagi, Z.; Lizanecz, E.; Pásztor, E.T.; Erdei, N.; Czikora, A.; Papp, Z.; Edes, I.; Pórszász, R.; Tóth, A. Tissue-specific regulation of microvascular diameter: Opposite functional roles of neuronal and smooth muscle located vanilloid receptor-1. Mol. Pharmacol. 2008, 73, 1405–1412. [Google Scholar] [CrossRef]
- Guarini, G.; Ohanyan, V.A.; Kmetz, J.G.; DelloStritto, D.J.; Thoppil, R.J.; Thodeti, C.K.; Meszaros, J.G.; Damron, D.S.; Bratz, I.N. Disruption of TRPV1-mediated coupling of coronary blood flow to cardiac metabolism in diabetic mice: Role of nitric oxide and BK channels. Am. J. Physiol. Heart Circ. Physiol. 2012, 303, H216–H223. [Google Scholar] [CrossRef]
- Leonelli, M.; Martins, D.O.; Britto, L.R. Retinal cell death induced by TRPV1 activation involves NMDA signaling and upregulation of nitric oxide synthases. Cell Mol. Neurobiol. 2013, 33, 379–392. [Google Scholar] [CrossRef]
- Prieto, D.; Benedito, S.; Nielsen, P.J.; Nyborg, N.C.B. Calcitonin gene-related peptide is a potent vasodilator of bovine retinal arteries in vitro. Exp. Eye Res. 1991, 53, 399–405. [Google Scholar] [CrossRef]
- Samudre, S.S.; Nicholls, M.; Williams, P.B.; Lattanzio, F.A. Endocannabinoid analogs reduce human retinal vascular endothelial cell proliferation. FASEB J. 2009, 23. E-Abstract LB404. [Google Scholar]
- Turner, H.; Fleig, A.; Stokes, A.; Kinet, J.P.; Penner, R. Discrimination of intracellular calcium store subcompartments using TRPV1 (transient receptor potential channel, vanilloid subfamily member 1) release channel activity. Biochem. J. 2003, 371, 341–350. [Google Scholar] [CrossRef]
- Križaj, D.; Bao, J.X.; Schmitz, Y.; Witkovsky, P.; Copenhagen, D.R. Caffeine-sensitive calcium stores regulate synaptic transmission from retinal rod photoreceptors. J. Neurosci. 1999, 19, 7249–7261. [Google Scholar]
- Chen, M.; Križaj, D.; Thoreson, W.B. Intracellular calcium stores drive slow non-ribbon vesicle release from rod photoreceptors. Front. Cell. Neurosci. 2014, 8. [Google Scholar] [CrossRef]
- Križaj, D.; Copenhagen, D.R. Calcium regulation in photoreceptors. Front. Biosci. 2002, 7, d2023–d2044. [Google Scholar] [CrossRef]
- Fain, G.L.; Granda, A.M.; Maxwell, J.M. Voltage signal of photoreceptors at visual threshold. Nature 1977, 265, 181–183. [Google Scholar]
- Thoreson, W.B.; Rabi, K.; Townes-Anderson, E.; Heidelberger, R. A highly Ca2+-sensitive pool of vesicles contributes to linearity at the rod photoreceptor ribbon synapse. Neuron 2004, 42, 595–605. [Google Scholar]
- Ciura, S.; Liedtke, W.; Bourque, C.W. Hypertonicity sensing in organum vasculosum lamina terminalis neurons: A mechanical process involving TRPV1 but not TRPV4. J. Neurosci. 2011, 31, 14669–14676. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Ryskamp, D.A.; Redmon, S.; Jo, A.O.; Križaj, D. TRPV1 and Endocannabinoids: Emerging Molecular Signals that Modulate Mammalian Vision. Cells 2014, 3, 914-938. https://doi.org/10.3390/cells3030914
Ryskamp DA, Redmon S, Jo AO, Križaj D. TRPV1 and Endocannabinoids: Emerging Molecular Signals that Modulate Mammalian Vision. Cells. 2014; 3(3):914-938. https://doi.org/10.3390/cells3030914
Chicago/Turabian StyleRyskamp, Daniel A., Sarah Redmon, Andrew O. Jo, and David Križaj. 2014. "TRPV1 and Endocannabinoids: Emerging Molecular Signals that Modulate Mammalian Vision" Cells 3, no. 3: 914-938. https://doi.org/10.3390/cells3030914
APA StyleRyskamp, D. A., Redmon, S., Jo, A. O., & Križaj, D. (2014). TRPV1 and Endocannabinoids: Emerging Molecular Signals that Modulate Mammalian Vision. Cells, 3(3), 914-938. https://doi.org/10.3390/cells3030914



