Maternal Exposure to Diesel Exhaust Particles (DEPs) During Pregnancy and Adverse Pregnancy Outcomes: Focusing on the Effect of Particulate Matter on Trophoblast, Epithelial-Mesenchymal Transition
Abstract
1. Introduction
2. Materials and Methods
2.1. Diesel Exhaust Particles
2.2. Cell Culture
2.3. Cell Proliferation Assay
2.4. Wound-Healing Assay
2.5. Formation of Blastocyst-like Spheroids (BLS) by Trophoblast Cells
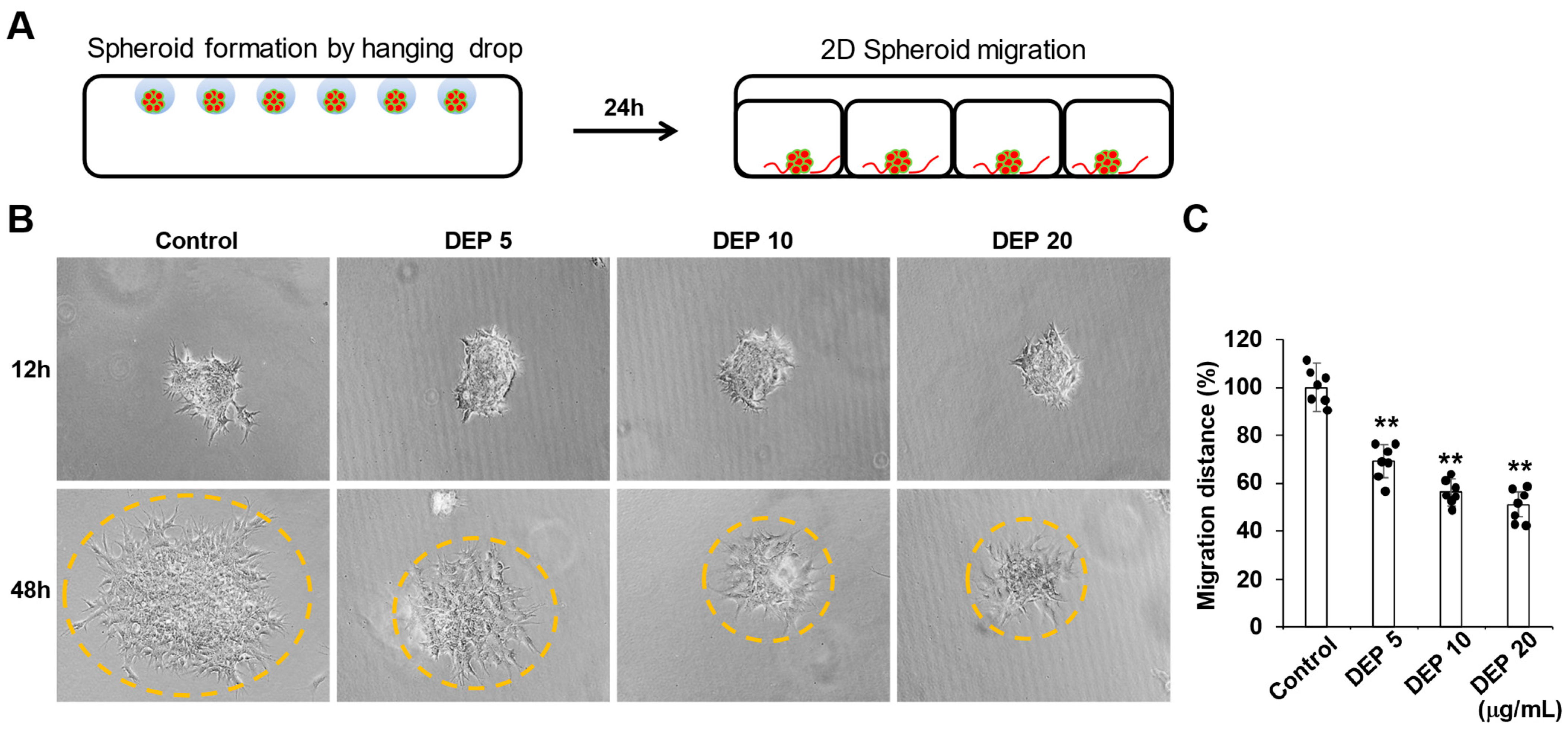
2.6. Two-Dimensional Trophoblast Migration
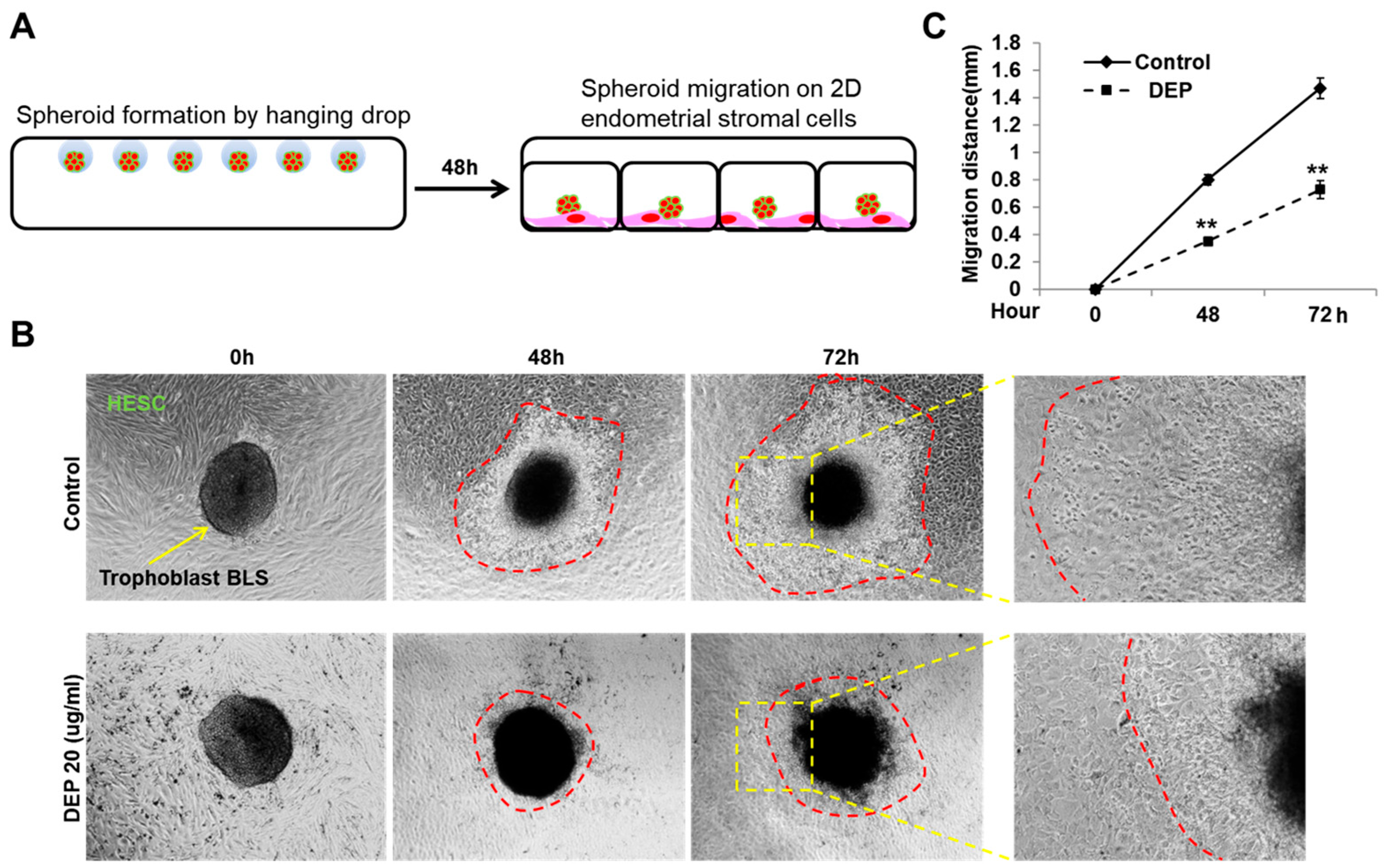
2.7. Two-Dimensional Trophoblast–Endometrial Cell Interactions
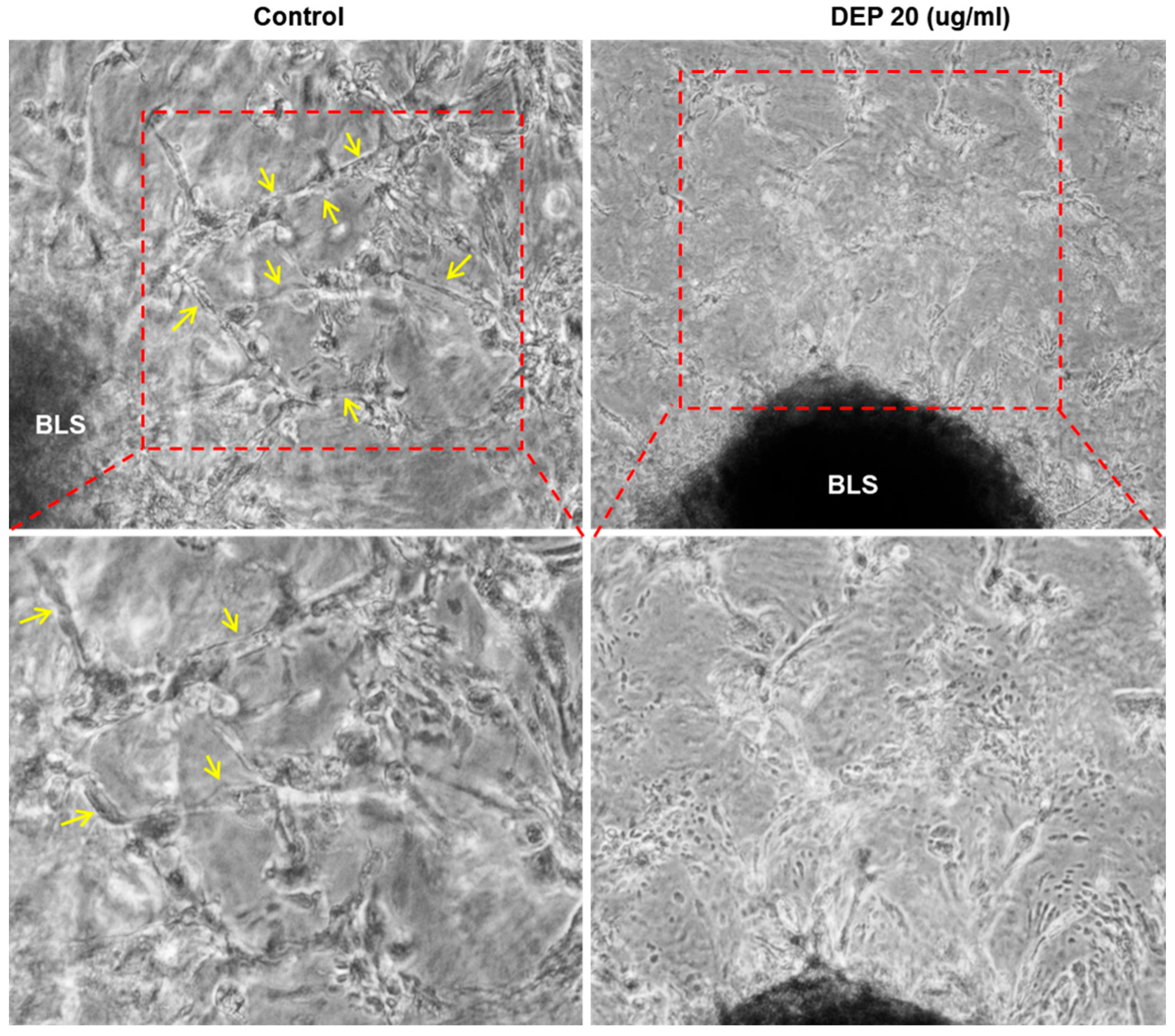
2.8. Three-Dimensional Trophoblast Invasion and Structure Formation
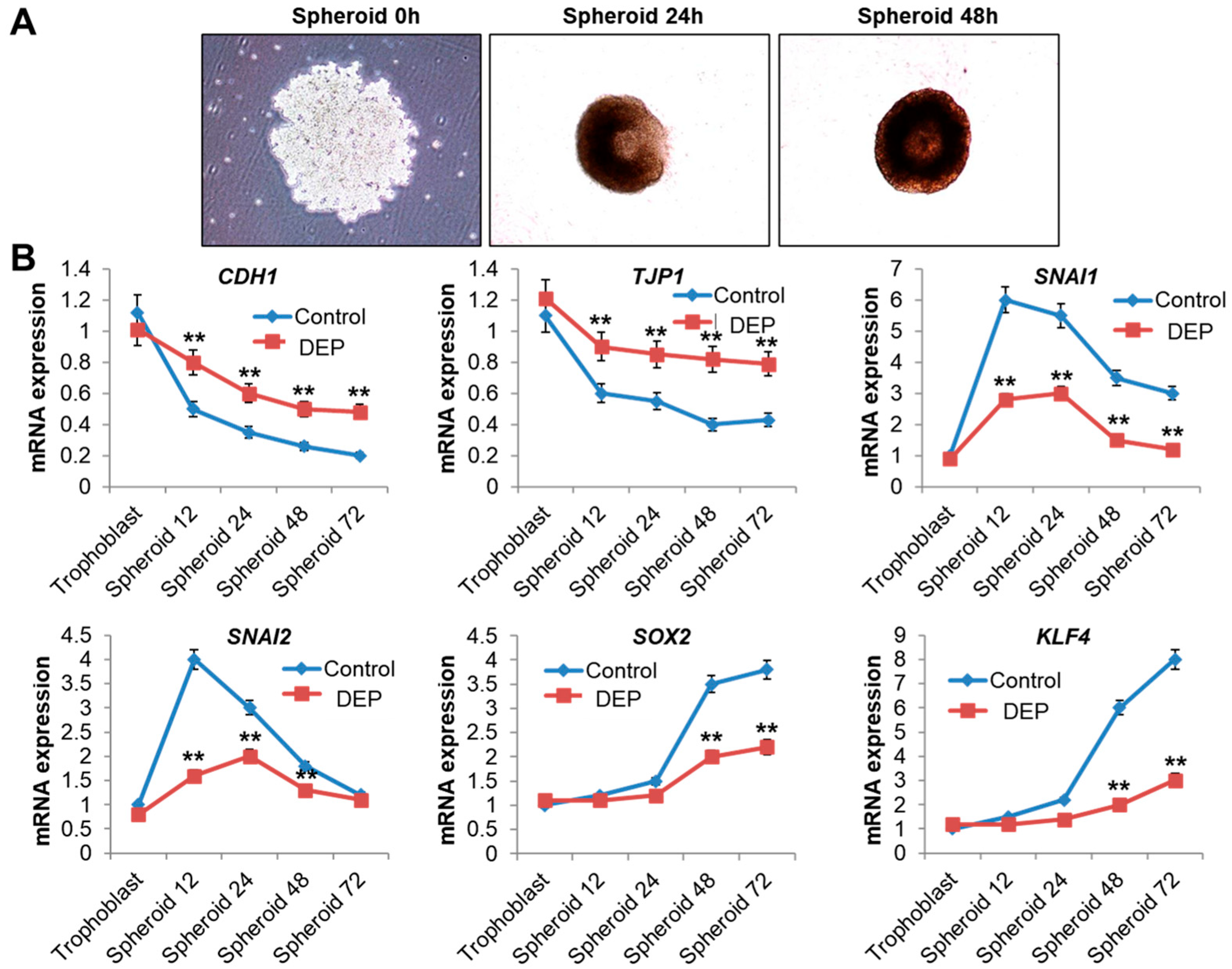
2.9. Epithelial-Mesenchymal Transition Assay
2.10. RT-qPCR
2.11. Statistical Analyses
3. Results
3.1. DEPs Decreased the Proliferation and Wound Healing of Sw71 Trophoblasts
3.2. DEP Decreased the Migration of Sw71 Trophoblasts
3.3. DEP Reduced the 2D Migration of Sw71 Cells on Endometrial Stromal Cells
3.4. DEPs Inhibited Trophoblast Invasion and 3D Structure Formation in 3D Culture Model with Endometrial Stromal Cells
3.5. DEP Inhibited Epithelial-Mesenchymal Transition
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| DEP | Diesel exhaust particles |
| PM2.5 | Fine particulate matter |
| HESC | Human endometrial stromal cell |
| Sw71 | Human first-trimester trophoblasts swan cell |
| EMT | Epithelial-mesenchymal transition |
References
- Agency, U.S.E.P. Particulate Matter (PM) Basics. Available online: https://www.epa.gov/pm-pollution/particulate-matter-pm-basics (accessed on 31 May 2023).
- Nemmar, A.; Hoet, P.H.; Vanquickenborne, B.; Dinsdale, D.; Thomeer, M.; Hoylaerts, M.F.; Vanbilloen, H.; Mortelmans, L.; Nemery, B. Passage of inhaled particles into the blood circulation in humans. Circulation 2002, 105, 411–414. [Google Scholar] [CrossRef]
- Feng, S.; Gao, D.; Liao, F.; Zhou, F.; Wang, X. The health effects of ambient PM2.5 and potential mechanisms. Ecotoxicol. Environ. Saf. 2016, 128, 67–74. [Google Scholar] [CrossRef]
- Jan, R.; Roy, R.; Bhor, R.; Pai, K.; Satsangi, P.G. Toxicological screening of airborne particulate matter in atmosphere of pune: Reactive oxygen species and cellular toxicity. Environ. Pollut. 2020, 261, 113724. [Google Scholar] [CrossRef]
- Pope, C.A., III; Bhatnagar, A.; McCracken, J.P.; Abplanalp, W.; Conklin, D.J.; O’Toole, T. Exposure to fine particulate air pollution is associated with endothelial injury and systemic inflammation. Circ. Res. 2016, 119, 1204–1214. [Google Scholar] [CrossRef]
- Olaniyan, T.; Pinault, L.; Li, C.; van Donkelaar, A.; Meng, J.; Martin, R.V.; Hystad, P.; Robichaud, A.; Ménard, R.; Tjepkema, M.; et al. Ambient air pollution and the risk of acute myocardial infarction and stroke: A national cohort study. Environ. Res. 2022, 204, 111975. [Google Scholar] [CrossRef]
- Farhadi, Z.; Abulghasem Gorgi, H.; Shabaninejad, H.; Aghajani Delavar, M.; Torani, S. Association between PM2.5 and risk of hospitalization for myocardial infarction: A systematic review and a meta-analysis. BMC Public Health 2020, 20, 314. [Google Scholar] [CrossRef]
- Wallwork, R.S.; Colicino, E.; Zhong, J.; Kloog, I.; Coull, B.A.; Vokonas, P.; Schwartz, J.D.; Baccarelli, A.A. Ambient fine particulate matter, outdoor temperature, and risk of metabolic syndrome. Am. J. Epidemiol. 2017, 185, 30–39. [Google Scholar] [CrossRef]
- Carey, I.M.; Anderson, H.R.; Atkinson, R.W.; Beevers, S.D.; Cook, D.G.; Strachan, D.P.; Dajnak, D.; Gulliver, J.; Kelly, F.J. Are noise and air pollution related to the incidence of dementia? A cohort study in london, england. BMJ Open 2018, 8, e022404. [Google Scholar] [CrossRef] [PubMed]
- Rezayat, A.A.; Niloufar, J.; Mir Nourbakhsh, S.H.; Hasheminezhad Hoseini, F.S.; Hooshmand, N.; Ghasemi Nour, M.; Handjani, F.; Tabrizi, R. The effect of air pollution on systemic lupus erythematosus: A systematic review and meta-analysis. Lupus 2022, 31, 1606–1618. [Google Scholar] [CrossRef] [PubMed]
- Lewtas, J. Air pollution combustion emissions: Characterization of causative agents and mechanisms associated with cancer, reproductive, and cardiovascular effects. Mutat. Res. 2007, 636, 95–133. [Google Scholar] [CrossRef] [PubMed]
- Wang, B.; Lau, Y.S.; Huang, Y.; Organ, B.; Chuang, H.C.; Ho, S.S.H.; Qu, L.; Lee, S.C.; Ho, K.F. Chemical and toxicological characterization of particulate emissions from diesel vehicles. J. Hazard. Mater. 2021, 405, 124613. [Google Scholar] [CrossRef] [PubMed]
- Hiura, T.S.; Kaszubowski, M.P.; Li, N.; Nel, A.E. Chemicals in diesel exhaust particles generate reactive oxygen radicals and induce apoptosis in macrophages. J. Immunol. 1999, 163, 5582–5591. [Google Scholar] [CrossRef]
- Gibbs, J.L.; Dallon, B.W.; Lewis, J.B.; Walton, C.M.; Arroyo, J.A.; Reynolds, P.R.; Bikman, B.T. Diesel exhaust particle exposure compromises alveolar macrophage mitochondrial bioenergetics. Int. J. Mol. Sci. 2019, 20, 5598. [Google Scholar] [CrossRef]
- Valentino, S.A.; Tarrade, A.; Aioun, J.; Mourier, E.; Richard, C.; Dahirel, M.; Rousseau-Ralliard, D.; Fournier, N.; Aubrière, M.C.; Lallemand, M.S.; et al. Maternal exposure to diluted diesel engine exhaust alters placental function and induces intergenerational effects in rabbits. Part. Fibre Toxicol. 2016, 13, 39. [Google Scholar] [CrossRef]
- Bongaerts, E.; Nawrot, T.S.; Wang, C.; Ameloot, M.; Bové, H.; Roeffaers, M.B.; Chavatte-Palmer, P.; Couturier-Tarrade, A.; Cassee, F.R. Placental-fetal distribution of carbon particles in a pregnant rabbit model after repeated exposure to diluted diesel engine exhaust. Part. Fibre Toxicol. 2023, 20, 20. [Google Scholar] [CrossRef]
- Thaver, S.; Foa, L.; Richards, S.M.; Lyons, A.B.; Zosky, G.R. In utero exposure to diesel exhaust particles, but not silica, alters post-natal immune development and function. Chemosphere 2021, 268, 129314. [Google Scholar] [CrossRef]
- Ma, X.L.; Li, X.; Tian, F.J.; Zeng, W.H.; Zhang, J.; Mo, H.Q.; Qin, S.; Sun, L.Q.; Zhang, Y.C.; Zhang, Y.; et al. Upregulation of rnd3 affects trophoblast proliferation, apoptosis, and migration at the maternal-fetal interface. Front. Cell Dev. Biol. 2020, 8, 153. [Google Scholar] [CrossRef]
- Liu, Y.; Wang, L.; Wang, F.; Li, C. Effect of fine particulate matter (PM2.5) on rat placenta pathology and perinatal outcomes. Med. Sci. Monit. 2016, 22, 3274–3280. [Google Scholar] [CrossRef] [PubMed]
- González-Comadran, M.; Jacquemin, B.; Cirach, M.; Lafuente, R.; Cole-Hunter, T.; Nieuwenhuijsen, M.; Brassesco, M.; Coroleu, B.; Checa, M.A. The effect of short term exposure to outdoor air pollution on fertility. Reprod. Biol. Endocrinol. 2021, 19, 151. [Google Scholar] [CrossRef]
- You, Y.; Stelzl, P.; Zhang, Y.; Porter, J.; Liu, H.; Liao, A.H.; Aldo, P.B.; Mor, G. Novel 3d in vitro models to evaluate trophoblast migration and invasion. Am. J. Reprod. Immunol. 2019, 81, e13076. [Google Scholar] [CrossRef] [PubMed]
- Tapia, V.L.; Vasquez, B.V.; Vu, B.; Liu, Y.; Steenland, K.; Gonzales, G.F. Association between maternal exposure to particulate matter (PM2.5) and adverse pregnancy outcomes in lima, peru. J. Expo. Sci. Environ. Epidemiol. 2020, 30, 689–697. [Google Scholar] [CrossRef]
- Sun, M.; Yan, W.; Fang, K.; Chen, D.; Liu, J.; Chen, Y.; Duan, J.; Chen, R.; Sun, Z.; Wang, X.; et al. The correlation between PM2.5 exposure and hypertensive disorders in pregnancy: A meta-analysis. Sci. Total Environ. 2020, 703, 134985. [Google Scholar] [CrossRef]
- Pedersen, M.; Stayner, L.; Slama, R.; Sørensen, M.; Figueras, F.; Nieuwenhuijsen, M.J.; Raaschou-Nielsen, O.; Dadvand, P. Ambient air pollution and pregnancy-induced hypertensive disorders: A systematic review and meta-analysis. Hypertension 2014, 64, 494–500. [Google Scholar] [CrossRef]
- Rammah, A.; Whitworth, K.W.; Han, I.; Chan, W.; Symanski, E. PM2.5 metal constituent exposure and stillbirth risk in harris county, texas. Environ. Res. 2019, 176, 108516. [Google Scholar] [CrossRef]
- Michikawa, T.; Morokuma, S.; Yamazaki, S.; Takami, A.; Sugata, S.; Yoshino, A.; Takeda, Y.; Nakahara, K.; Saito, S.; Hoshi, J.; et al. Exposure to chemical components of fine particulate matter and ozone, and placenta-mediated pregnancy complications in tokyo: A register-based study. J. Expo. Sci. Environ. Epidemiol. 2022, 32, 135–145. [Google Scholar] [CrossRef]
- Bové, H.; Bongaerts, E.; Slenders, E.; Bijnens, E.M.; Saenen, N.D.; Gyselaers, W.; Van Eyken, P.; Plusquin, M.; Roeffaers, M.B.J.; Ameloot, M.; et al. Ambient black carbon particles reach the fetal side of human placenta. Nat. Commun. 2019, 10, 3866. [Google Scholar] [CrossRef] [PubMed]
- Enquobahrie, D.A.; MacDonald, J.; Hussey, M.; Bammler, T.K.; Loftus, C.T.; Paquette, A.G.; Byington, N.; Marsit, C.J.; Szpiro, A.; Kaufman, J.D.; et al. Prenatal exposure to particulate matter and placental gene expression. Environ. Int. 2022, 165, 107310. [Google Scholar] [CrossRef] [PubMed]
- Brunst, K.J.; Sanchez-Guerra, M.; Chiu, Y.M.; Wilson, A.; Coull, B.A.; Kloog, I.; Schwartz, J.; Brennan, K.J.; Bosquet Enlow, M.; Wright, R.O.; et al. Prenatal particulate matter exposure and mitochondrial dysfunction at the maternal-fetal interface: Effect modification by maternal lifetime trauma and child sex. Environ. Int. 2018, 112, 49–58. [Google Scholar] [CrossRef] [PubMed]
- Chi, Y.; Huang, Q.; Lin, Y.; Ye, G.; Zhu, H.; Dong, S. Epithelial-mesenchymal transition effect of fine particulate matter from the yangtze river delta region in china on human bronchial epithelial cells. J. Environ. Sci. 2018, 66, 155–164. [Google Scholar] [CrossRef]
- Kirwa, K.; Feric, Z.; Manjourides, J.; Alshawabekeh, A.; Vega, C.M.V.; Cordero, J.F.; Meeker, J.D.; Suh, H.H. Preterm birth and PM2.5 in puerto rico: Evidence from the protect birth cohort. Environ. Health 2021, 20, 69. [Google Scholar] [CrossRef]
- Alman, B.L.; Stingone, J.A.; Yazdy, M.; Botto, L.D.; Desrosiers, T.A.; Pruitt, S.; Herring, A.H.; Langlois, P.H.; Nembhard, W.N.; Shaw, G.M.; et al. Associations between PM2.5 and risk of preterm birth among liveborn infants. Ann. Epidemiol. 2019, 39, 46–53.e2. [Google Scholar] [CrossRef]
- Miron-Celis, M.; Talarico, R.; Villeneuve, P.J.; Crighton, E.; Stieb, D.M.; Stanescu, C.; Lavigne, É. Critical windows of exposure to air pollution and gestational diabetes: Assessing effect modification by maternal pre-existing conditions and environmental factors. Environ. Health 2023, 22, 26. [Google Scholar] [CrossRef] [PubMed]
- Cheng, X.; Ji, X.; Yang, D.; Zhang, C.; Chen, L.; Liu, C.; Meng, X.; Wang, W.; Li, H.; Kan, H.; et al. Associations of PM2.5 exposure with blood glucose impairment in early pregnancy and gestational diabetes mellitus. Ecotoxicol. Environ. Saf. 2022, 232, 113278. [Google Scholar] [CrossRef] [PubMed]
- Weber, K.A.; Yang, W.; Lurmann, F.; Hammond, S.K.; Shaw, G.M.; Padula, A.M. Air pollution, maternal hypertensive disorders, and preterm birth. Environ. Epidemiol. 2019, 3, e062. [Google Scholar] [CrossRef]
- Savitz, D.A.; Elston, B.; Bobb, J.F.; Clougherty, J.E.; Dominici, F.; Ito, K.; Johnson, S.; McAlexander, T.; Ross, Z.; Shmool, J.L.; et al. Ambient fine particulate matter, nitrogen dioxide, and hypertensive disorders of pregnancy in new york city. Epidemiology 2015, 26, 748–757. [Google Scholar] [CrossRef] [PubMed]
- Gaskins, A.J.; Hart, J.E.; Chavarro, J.E.; Missmer, S.A.; Rich-Edwards, J.W.; Laden, F.; Mahalingaiah, S. Air pollution exposure and risk of spontaneous abortion in the nurses’ health study ii. Hum. Reprod. 2019, 34, 1809–1817. [Google Scholar] [CrossRef]
- Qin, Z.; Hou, H.; Fu, F.; Wu, J.; Han, B.; Yang, W.; Zhang, L.; Cao, J.; Jin, X.; Cheng, S.; et al. Fine particulate matter exposure induces cell cycle arrest and inhibits migration and invasion of human extravillous trophoblast, as determined by an itraq-based quantitative proteomics strategy. Reprod. Toxicol. 2017, 74, 10–22. [Google Scholar] [CrossRef]
- Raez-Villanueva, S.; Ma, C.; Kleiboer, S.; Holloway, A.C. The effects of electronic cigarette vapor on placental trophoblast cell function. Reprod. Toxicol. 2018, 81, 115–121. [Google Scholar] [CrossRef]
- Familari, M.; Nääv, Å.; Erlandsson, L.; de Iongh, R.U.; Isaxon, C.; Strandberg, B.; Lundh, T.; Hansson, S.R.; Malmqvist, E. Exposure of trophoblast cells to fine particulate matter air pollution leads to growth inhibition, inflammation and er stress. PLoS ONE 2019, 14, e0218799. [Google Scholar] [CrossRef]
- Nääv, Å.; Erlandsson, L.; Isaxon, C.; Åsander Frostner, E.; Ehinger, J.; Sporre, M.K.; Krais, A.M.; Strandberg, B.; Lundh, T.; Elmér, E.; et al. Urban PM2.5 induces cellular toxicity, hormone dysregulation, oxidative damage, inflammation, and mitochondrial interference in the hrt8 trophoblast cell line. Front. Endocrinol. 2020, 11, 75. [Google Scholar] [CrossRef]
- Gellersen, B.; Wolf, A.; Kruse, M.; Schwenke, M.; Bamberger, A.M. Human endometrial stromal cell-trophoblast interactions: Mutual stimulation of chemotactic migration and promigratory roles of cell surface molecules cd82 and ceacam1. Biol. Reprod. 2013, 88, 80. [Google Scholar] [CrossRef] [PubMed]
- Bengalli, R.; Zerboni, A.; Marchetti, S.; Longhin, E.; Priola, M.; Camatini, M.; Mantecca, P. In vitro pulmonary and vascular effects induced by different diesel exhaust particles. Toxicol. Lett. 2019, 306, 13–24. [Google Scholar] [CrossRef]
- Straszewski-Chavez, S.L.; Abrahams, V.M.; Alvero, A.B.; Aldo, P.B.; Ma, Y.; Guller, S.; Romero, R.; Mor, G. The isolation and characterization of a novel telomerase immortalized first trimester trophoblast cell line, swan 71. Placenta 2009, 30, 939–948. [Google Scholar] [CrossRef]
- Krikun, G.; Mor, G.; Alvero, A.; Guller, S.; Schatz, F.; Sapi, E.; Rahman, M.; Caze, R.; Qumsiyeh, M.; Lockwood, C.J. A novel immortalized human endometrial stromal cell line with normal progestational response. Endocrinology 2004, 145, 2291–2296. [Google Scholar] [CrossRef]
- Aplin, J.D.; Ruane, P.T. Embryo-epithelium interactions during implantation at a glance. J. Cell Sci. 2017, 130, 15–22. [Google Scholar] [CrossRef] [PubMed]
- Velicky, P.; Knofler, M.; Pollheimer, J. Function and control of human invasive trophoblast subtypes: Intrinsic vs. Maternal control. Cell Adh Migr. 2016, 10, 154–162. [Google Scholar] [CrossRef]
- Cao, Z.; Livas, T.; Kyprianou, N. Anoikis and emt: Lethal “liaisons” during cancer progression. Crit. Rev. Oncog. 2016, 21, 155–168. [Google Scholar] [CrossRef]
- Paoli, P.; Giannoni, E.; Chiarugi, P. Anoikis molecular pathways and its role in cancer progression. Biochim. Biophys. Acta 2013, 1833, 3481–3498. [Google Scholar] [CrossRef]
- Lunghi, L.; Ferretti, M.E.; Medici, S.; Biondi, C.; Vesce, F. Control of human trophoblast function. Reprod. Biol. Endocrinol. 2007, 5, 6. [Google Scholar] [CrossRef]
- DaSilva-Arnold, S.C.; Zamudio, S.; Al-Khan, A.; Alvarez-Perez, J.; Mannion, C.; Koenig, C.; Luke, D.; Perez, A.M.; Petroff, M.; Alvarez, M.; et al. Human trophoblast epithelial-mesenchymal transition in abnormally invasive placenta†. Biol. Reprod. 2018, 99, 409–421. [Google Scholar] [CrossRef] [PubMed]
- Kalluri, R.; Weinberg, R.A. The basics of epithelial-mesenchymal transition. J. Clin. Investig. 2009, 119, 1420–1428. [Google Scholar] [CrossRef]
- Oghbaei, F.; Zarezadeh, R.; Jafari-Gharabaghlou, D.; Ranjbar, M.; Nouri, M.; Fattahi, A.; Imakawa, K. Epithelial-mesenchymal transition process during embryo implantation. Cell Tissue Res. 2022, 388, 1–17. [Google Scholar] [CrossRef]
- Davies, J.E.; Pollheimer, J.; Yong, H.E.J.; Kokkinos, M.I.; Kalionis, B.; Knöfler, M.; Murthi, P. Epithelial-mesenchymal transition during extravillous trophoblast differentiation. Cell Adh Migr. 2016, 10, 310–321. [Google Scholar] [CrossRef]
- Lim, K.H.; Zhou, Y.; Janatpour, M.; McMaster, M.; Bass, K.; Chun, S.H.; Fisher, S.J. Human cytotrophoblast differentiation/invasion is abnormal in pre-eclampsia. Am. J. Pathol. 1997, 151, 1809–1818. [Google Scholar] [PubMed]
- Ge, H.; Yin, N.; Han, T.L.; Huang, D.; Chen, X.; Xu, P.; He, C.; Tong, C.; Qi, H. Interleukin-27 inhibits trophoblast cell invasion and migration by affecting the epithelial-mesenchymal transition in preeclampsia. Reprod. Sci. 2019, 26, 928–938. [Google Scholar] [CrossRef] [PubMed]
- Cui, J.; Yang, Z.; Ma, R.; He, W.; Tao, H.; Li, Y.; Zhao, Y. Placenta-targeted treatment strategies for preeclampsia and fetal growth restriction: An opportunity and major challenge. Stem Cell Rev. Rep. 2024, 20, 1501–1511. [Google Scholar] [CrossRef]
- Huppertz, B. Traditional and new routes of trophoblast invasion and their implications for pregnancy diseases. Int. J. Mol. Sci. 2019, 21, 289. [Google Scholar] [CrossRef]
- van den Brûle, F.; Berndt, S.; Simon, N.; Coulon, C.; Le Goarant, J.; Munaut, C.; Noël, A.; Frankenne, F.; Foidart, J.M. Trophoblast invasion and placentation: Molecular mechanisms and regulation. Chem. Immunol. Allergy 2005, 88, 163–180. [Google Scholar] [CrossRef] [PubMed]
- Yue, H.; Ji, X.; Zhang, Y.; Li, G.; Sang, N. Gestational exposure to PM2.5 impairs vascularization of the placenta. Sci. Total Environ. 2019, 665, 153–161. [Google Scholar] [CrossRef]
- Tosevska, A.; Ghosh, S.; Ganguly, A.; Cappelletti, M.; Kallapur, S.G.; Pellegrini, M.; Devaskar, S.U. Integrated analysis of an in vivo model of intra-nasal exposure to instilled air pollutants reveals cell-type specific responses in the placenta. Sci. Rep. 2022, 12, 8438. [Google Scholar] [CrossRef]
- Soto, S.F.; Melo, J.O.; Marchesi, G.D.; Lopes, K.L.; Veras, M.M.; Oliveira, I.B.; Souza, R.M.; de Castro, I.; Furukawa, L.N.S.; Saldiva, P.H.N.; et al. Exposure to fine particulate matter in the air alters placental structure and the renin-angiotensin system. PLoS ONE 2017, 12, e0183314. [Google Scholar] [CrossRef]
- Lafontaine, L.; Chaudhry, P.; Lafleur, M.J.; Van Themsche, C.; Soares, M.J.; Asselin, E. Transforming growth factor beta regulates proliferation and invasion of rat placental cell lines. Biol. Reprod. 2011, 84, 553–559. [Google Scholar] [CrossRef]
- Shibuya, M. Vascular endothelial growth factor and its receptor system: Physiological functions in angiogenesis and pathological roles in various diseases. J. Biochem. 2013, 153, 13–19. [Google Scholar] [CrossRef]
- Zuo, Q.; Huang, S.; Zou, Y.; Xu, Y.; Jiang, Z.; Zou, S.; Xu, H.; Sun, L. The lnc rna spry4-it1 modulates trophoblast cell invasion and migration by affecting the epithelial-mesenchymal transition. Sci. Rep. 2016, 6, 37183. [Google Scholar] [CrossRef]
- Wei, H.; Liang, F.; Cheng, W.; Zhou, R.; Wu, X.; Feng, Y.; Wang, Y. The mechanisms for lung cancer risk of PM2.5: Induction of epithelial-mesenchymal transition and cancer stem cell properties in human non-small cell lung cancer cells. Environ. Toxicol. 2017, 32, 2341–2351. [Google Scholar] [CrossRef]
- Xu, Z.; Ding, W.; Deng, X. PM2.5, fine particulate matter: A novel player in the epithelial-mesenchymal transition? Front. Physiol. 2019, 10, 1404. [Google Scholar] [CrossRef] [PubMed]
- de Melo, J.O.; Soto, S.F.; Katayama, I.A.; Wenceslau, C.F.; Pires, A.G.; Veras, M.M.; Furukawa, L.N.; de Castro, I.; Saldiva, P.H.; Heimann, J.C. Inhalation of fine particulate matter during pregnancy increased il-4 cytokine levels in the fetal portion of the placenta. Toxicol. Lett. 2015, 232, 475–480. [Google Scholar] [CrossRef] [PubMed]
- Wang, B.; Chan, Y.L.; Li, G.; Ho, K.F.; Anwer, A.G.; Smith, B.J.; Guo, H.; Jalaludin, B.; Herbert, C.; Thomas, P.S.; et al. Maternal particulate matter exposure impairs lung health and is associated with mitochondrial damage. Antioxidants 2021, 10, 1029. [Google Scholar] [CrossRef]
- Piao, M.J.; Ahn, M.J.; Kang, K.A.; Ryu, Y.S.; Hyun, Y.J.; Shilnikova, K.; Zhen, A.X.; Jeong, J.W.; Choi, Y.H.; Kang, H.K.; et al. Particulate matter 2.5 damages skin cells by inducing oxidative stress, subcellular organelle dysfunction, and apoptosis. Arch. Toxicol. 2018, 92, 2077–2091. [Google Scholar] [CrossRef]
- Bräuner, E.V.; Forchhammer, L.; Møller, P.; Simonsen, J.; Glasius, M.; Wåhlin, P.; Raaschou-Nielsen, O.; Loft, S. Exposure to ultrafine particles from ambient air and oxidative stress-induced DNA damage. Environ. Health Perspect. 2007, 115, 1177–1182. [Google Scholar] [CrossRef] [PubMed]
- Brunst, K.J.; Hsu, H.L.; Zhang, L.; Zhang, X.; Carroll, K.N.; Just, A.; Coull, B.A.; Kloog, I.; Wright, R.O.; Baccarelli, A.A.; et al. Prenatal particulate matter exposure and mitochondrial mutational load at the maternal-fetal interface: Effect modification by genetic ancestry. Mitochondrion 2022, 62, 102–110. [Google Scholar] [CrossRef]
- Janssen, B.G.; Munters, E.; Pieters, N.; Smeets, K.; Cox, B.; Cuypers, A.; Fierens, F.; Penders, J.; Vangronsveld, J.; Gyselaers, W.; et al. Placental mitochondrial DNA content and particulate air pollution during in utero life. Environ. Health Perspect. 2012, 120, 1346–1352. [Google Scholar] [CrossRef]
- Wylie, B.J.; Matechi, E.; Kishashu, Y.; Fawzi, W.; Premji, Z.; Coull, B.A.; Hauser, R.; Ezzati, M.; Roberts, D.J. Placental pathology associated with household air pollution in a cohort of pregnant women from dar es salaam, tanzania. Environ. Health Perspect. 2017, 125, 134–140. [Google Scholar] [CrossRef]
- Jaligama, S.; Patel, V.S.; Wang, P.; Sallam, A.; Harding, J.; Kelley, M.; Mancuso, S.R.; Dugas, T.R.; Cormier, S.A. Radical containing combustion derived particulate matter enhance pulmonary th17 inflammation via the aryl hydrocarbon receptor. Part. Fibre Toxicol. 2018, 15, 20. [Google Scholar] [CrossRef]
- Aguilera, J.; Han, X.; Cao, S.; Balmes, J.; Lurmann, F.; Tyner, T.; Lutzker, L.; Noth, E.; Hammond, S.K.; Sampath, V.; et al. Increases in ambient air pollutants during pregnancy are linked to increases in methylation of il4, il10, and ifnγ. Clin. Epigenet. 2022, 14, 40. [Google Scholar] [CrossRef]
- Miyata, R.; van Eeden, S.F. The innate and adaptive immune response induced by alveolar macrophages exposed to ambient particulate matter. Toxicol. Appl. Pharmacol. 2011, 257, 209–226. [Google Scholar] [CrossRef] [PubMed]
- Parikh, M.N.; Brokamp, C.; Rasnick, E.; Ding, L.; Mersha, T.B.; Bowers, K.; Folger, A.T. Epigenome-wide association of neonatal methylation and trimester-specific prenatal PM2.5 exposure. Environ. Epidemiol. 2022, 6, e227. [Google Scholar] [CrossRef] [PubMed]
- Szpak, D.; Grochowalski, A.; Chrząszcz, R.; Florek, E.; Jawień, W.; Undas, A. Tobacco smoke exposure and endothelial dysfunction in patients with advanced coronary artery disease. Pol. Arch. Med. Wewn. 2013, 123, 474–481. [Google Scholar] [CrossRef]
- Fongsodsri, K.; Chamnanchanunt, S.; Desakorn, V.; Thanachartwet, V.; Sahassananda, D.; Rojnuckarin, P.; Umemura, T. Particulate matter 2.5 and hematological disorders from dust to diseases: A systematic review of available evidence. Front. Med. 2021, 8, 692008. [Google Scholar] [CrossRef] [PubMed]
- Farahani, V.J.; Pirhadi, M.; Sioutas, C. Are standardized diesel exhaust particles (dep) representative of ambient particles in air pollution toxicological studies? Sci. Total Environ. 2021, 788, 147854. [Google Scholar] [CrossRef]
- Kim, W.; Cho, Y.; Song, M.K.; Lim, J.H.; Kim, J.Y.; Gye, M.C.; Ryu, J.C. Effect of particulate matter 2.5 on gene expression profile and cell signaling in jeg-3 human placenta cells. Environ. Toxicol. 2018, 33, 1123–1134. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Li, L.; Zhang, C.; Fu, H.; Yu, S.; Zhou, M.; Guo, J.; Fang, Z.; Li, A.; Zhao, M.; et al. PM2.5 leads to adverse pregnancy outcomes by inducing trophoblast oxidative stress and mitochondrial apoptosis via klf9/cyp1a1 transcriptional axis. eLife 2023, 12, e85944. [Google Scholar] [CrossRef]
- Alexandrova, M.; Manchorova, D.; You, Y.; Terzieva, A.; Dimitrova, V.; Mor, G.; Dimova, T. Validation of the sw71-spheroid model with primary trophoblast cells. Am. J. Reprod. Immunol. 2023, 90, e13800. [Google Scholar] [CrossRef]
- Gupta, S.K.; Malhotra, S.S.; Malik, A.; Verma, S.; Chaudhary, P. Cell signaling pathways involved during invasion and syncytialization of trophoblast cells. Am. J. Reprod. Immunol. 2016, 75, 361–371. [Google Scholar] [CrossRef]
- Dietrich, B.; Haider, S.; Meinhardt, G.; Pollheimer, J.; Knöfler, M. Wnt and notch signaling in human trophoblast development and differentiation. Cell Mol. Life Sci. 2022, 79, 292. [Google Scholar] [CrossRef]
- Subbalakshmi, A.R.; Sahoo, S.; McMullen, I.; Saxena, A.N.; Venugopal, S.K.; Somarelli, J.A.; Jolly, M.K. Klf4 induces mesenchymal-epithelial transition (met) by suppressing multiple emt-inducing transcription factors. Cancers 2021, 13, 5135. [Google Scholar] [CrossRef] [PubMed]
- Thiery, J.P. Epithelial-mesenchymal transitions in tumour progression. Nat. Rev. Cancer 2002, 2, 442–454. [Google Scholar] [CrossRef] [PubMed]


| CDH1 | F-aaagcctcaggtcataaaca | SOX2 | F-ccacctacagcatgtcctac |
| R-gttgggtcgttgtactgaat | R-gagtgggaggaagaggtaac | ||
| TJP1 | F-gagcacatggtgaaggtaat | KLF4 | F-ggcaaaacctacacaaagag |
| R-ctgaaagttgctggcttatt | R-gtagtgcctggtcagttcat | ||
| SNAI1 | F-cttcagtctcttccttggag | GAPDH | F-atggggaaggtgaaggtcg |
| R-gttgcagtatttgcagttga | R-ggggtcattgatggcaacaata | ||
| SNAI2 | F-tgtcataccacaaccagaga | ||
| R-agtatccggaaagaggagag |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hur, H.; Kwon, H.; Jung, Y.J.; Choi, E.; Shin, J.; Jo, S.; Lee, Y.; Kim, M.-A.; Maeng, Y.-S.; Kwon, J.-Y. Maternal Exposure to Diesel Exhaust Particles (DEPs) During Pregnancy and Adverse Pregnancy Outcomes: Focusing on the Effect of Particulate Matter on Trophoblast, Epithelial-Mesenchymal Transition. Cells 2025, 14, 1317. https://doi.org/10.3390/cells14171317
Hur H, Kwon H, Jung YJ, Choi E, Shin J, Jo S, Lee Y, Kim M-A, Maeng Y-S, Kwon J-Y. Maternal Exposure to Diesel Exhaust Particles (DEPs) During Pregnancy and Adverse Pregnancy Outcomes: Focusing on the Effect of Particulate Matter on Trophoblast, Epithelial-Mesenchymal Transition. Cells. 2025; 14(17):1317. https://doi.org/10.3390/cells14171317
Chicago/Turabian StyleHur, Hyewon, Hayan Kwon, Yun Ji Jung, Euna Choi, Joonggyeong Shin, Subin Jo, Yeji Lee, Min-A Kim, Yong-Sun Maeng, and Ja-Young Kwon. 2025. "Maternal Exposure to Diesel Exhaust Particles (DEPs) During Pregnancy and Adverse Pregnancy Outcomes: Focusing on the Effect of Particulate Matter on Trophoblast, Epithelial-Mesenchymal Transition" Cells 14, no. 17: 1317. https://doi.org/10.3390/cells14171317
APA StyleHur, H., Kwon, H., Jung, Y. J., Choi, E., Shin, J., Jo, S., Lee, Y., Kim, M.-A., Maeng, Y.-S., & Kwon, J.-Y. (2025). Maternal Exposure to Diesel Exhaust Particles (DEPs) During Pregnancy and Adverse Pregnancy Outcomes: Focusing on the Effect of Particulate Matter on Trophoblast, Epithelial-Mesenchymal Transition. Cells, 14(17), 1317. https://doi.org/10.3390/cells14171317







