Multiple Forms of Neural Cell Death in the Cyclical Brain Degeneration of A Colonial Chordate
Abstract
1. Introduction
2. Materials and Methods
2.1. Mariculture, Sample Collection
2.2. Behavioral Test
2.3. Histology and Transmission Electron Microscopy (TEM)
2.4. Neural Complex Three-Dimensional Reconstructions
2.5. Bioinformatic Analyses
2.5.1. Gene Counts
2.5.2. Gene Ontology
2.5.3. Heatmaps
3. Results
3.1. Adult Zooid Resorption Is Accompanied by Behavioral Impairment at Takeover
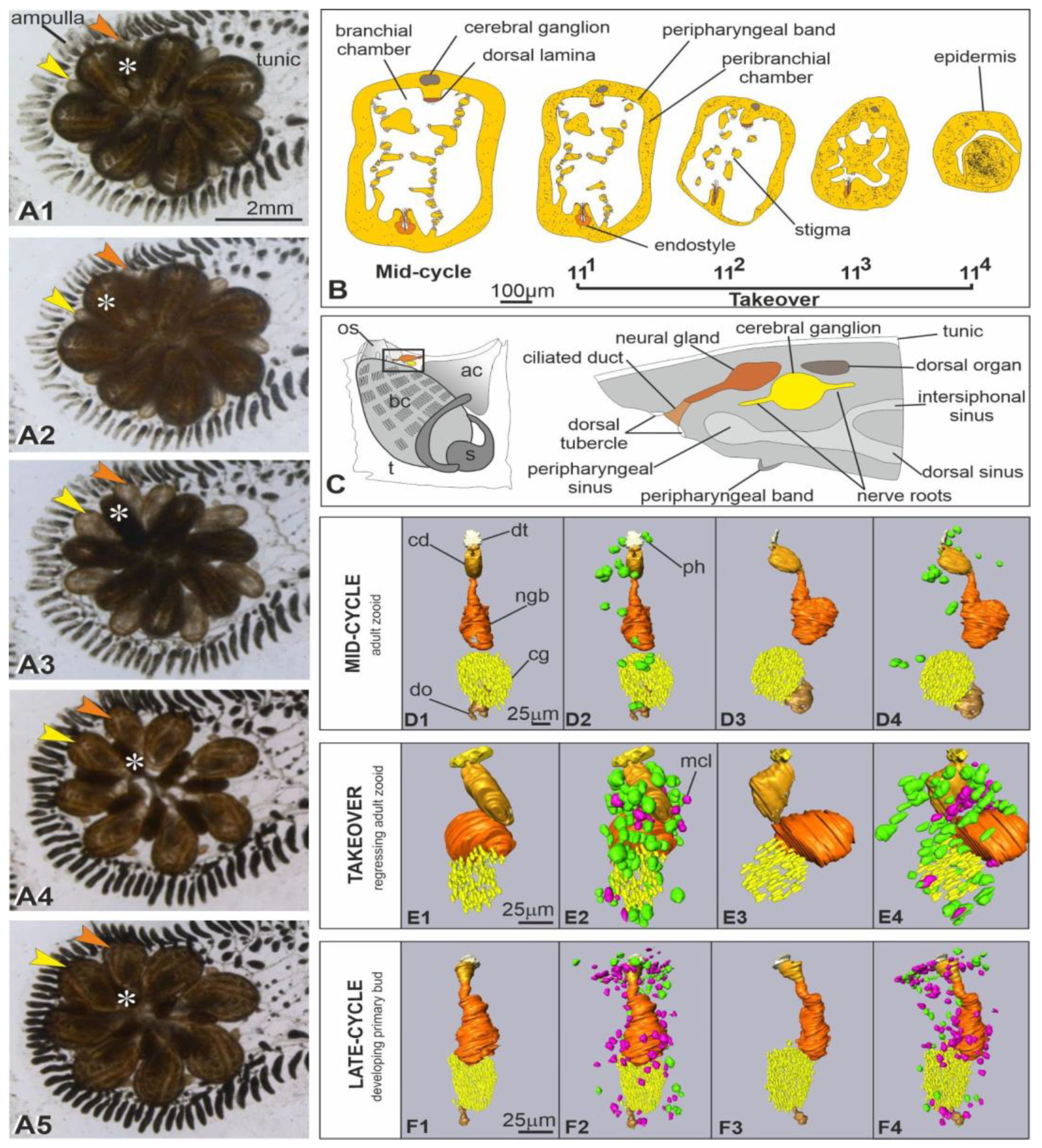
3.2. At Takeover, the Neural Complex Progressively Is Infiltrated by Immunocytes, Reduces in Size and Loses Its Organization
3.3. Apoptosis, Regulated Necrosis, and Autophagy Characterize the Neural Complex Degeneration
3.4. Transcriptome Analyses Suggest That Apoptosis, Necroptosis, Lysosomal Cell Death, and Autophagy Are Involved in Neurodegeneration
3.5. Genes Associated with Conformational Disorders Are Differentially Expressed during Neurodegeneration
4. Discussion
4.1. Cell Death in B. schlosseri: A Cyclical Natural Event for Colony Survival
4.2. Apoptosis, Necrosis, and Autophagy Are Displayed by the Degenerating Neural Complex
4.3. A Conformational Disorder-Like Process Characterizes Neurodegeneration in B. schlosseri
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Fricker, M.; Tolkovsky, A.M.; Borutaite, V.; Coleman, M.; Brown, G.C. Neuronal Cell Death. Physiol. Rev. 2018, 98, 813–880. [Google Scholar] [CrossRef] [PubMed]
- Hollville, E.; Romero, S.E.; Deshmukh, M. Apoptotic cell death regulation in neurons. FEBS J. 2019, 286, 3276–3298. [Google Scholar] [CrossRef] [PubMed]
- D’Arcy, M.S. Cell death: A review of the major forms of apoptosis, necrosis and autophagy. Cell Biol. Int. 2019, 43, 582–592. [Google Scholar] [CrossRef]
- Kerr, J.F.; Wyllie, A.H.; Currie, A.R. Apoptosis: A basic biological phenomenon with wide-ranging implications in tissue kinetics. Br. J. Cancer 1972, 26, 239–257. [Google Scholar] [CrossRef]
- Delsuc, F.; Philippe, H.; Tsagkogeorga, G.; Simion, P.; Tilak, M.-K.; Turon, X.; López-Legentil, S.; Piette, J.; Lemaire, P.; Douzery, E.J.P. A phylogenomic framework and timescale for comparative studies of tunicates. BMC Biol. 2018, 16, 39. [Google Scholar] [CrossRef] [PubMed]
- Voskoboynik, A.; Neff, N.F.; Sahoo, D.; Newman, A.M.; Pushkarev, D.; Koh, W.; Passarelli, B.; Fan, H.C.; Mantalas, G.L.; Palmeri, K.J.; et al. The genome sequence of the colonial chordate, Botryllus schlosseri. eLife 2013, 2, e00569. [Google Scholar] [CrossRef] [PubMed]
- Anselmi, C.; Kowarsky, M.; Gasparini, F.; Caicci, F.; Ishizuka, K.J.; Palmeri, K.J.; Raveh, T.; Sinha, R.; Neff, N.; Quake, S.R.; et al. Two distinct evolutionary conserved neural degeneration pathways characterized in a colonial chordate. Proc. Natl. Acad. Sci. USA 2022, 119, e2203032119. [Google Scholar] [CrossRef]
- Galluzzi, L.; Vitale, I.; Aaronson, S.A.; Abrams, J.M.; Adam, D.; Agostinis, P.; Alnemri, E.S.; Altucci, L.; Amelio, I.; Andrews, D.W.; et al. Molecular mechanisms of cell death: Recommendations of the Nomenclature Committee on Cell Death 2018. Cell Death Differ. 2018, 25, 486–541. [Google Scholar] [CrossRef]
- Lauzon, R.J.; Patton, C.W.; Weissman, I.L. A morphological and immunohistochemical study of programmed cell death in Botryllus schlosseri (Tunicata, Ascidiacea). Cell Tissue Res. 1993, 272, 115–127. [Google Scholar] [CrossRef]
- Lauzon, R.J.; Rinkevich, B.; Patton, C.W.; Weissman, I.L. A morphological study of nonrandom senescence in a colonial urochordate. Biol. Bull. 2000, 198, 367–378. [Google Scholar] [CrossRef]
- Ballarin, L.; Schiavon, F.; Manni, L. Natural apoptosis during the blastogenetic cycle of the colonial ascidian Botryllus schlosseri: A morphological analysis. Zool. Sci. 2010, 27, 96–102. [Google Scholar] [CrossRef] [PubMed]
- Gasparini, F.; Skobo, T.; Benato, F.; Gioacchini, G.; Voskoboynik, A.; Carnevali, O.; Manni, L.; Dalla Valle, L. Characterization of Ambra1 in asexual cycle of a non-vertebrate chordate, the colonial tunicate Botryllus schlosseri, and phylogenetic analysis of the protein group in Bilateria. Mol. Phylogenet. Evol. 2016, 95, 46–57. [Google Scholar] [CrossRef] [PubMed]
- Kawamura, K.; Yoshida, T.; Sekida, S. Autophagic dedifferentiation induced by cooperation between TOR inhibitor and retinoic acid signals in budding tunicates. Dev. Biol. 2018, 433, 384–393. [Google Scholar] [CrossRef] [PubMed]
- Jeffery, W.R.; Gorički, Š. Apoptosis is a generator of Wnt-dependent regeneration and homeostatic cell renewal in the ascidian Ciona. Biol. Open 2021, 10, bio058526. [Google Scholar] [CrossRef] [PubMed]
- Tiozzo, S.; Ballarin, L.; Burighel, P.; Zaniolo, G. Programmed cell death in vegetative development: Apoptosis during the colonial life cycle of the ascidian Botryllus schlosseri. Tissue Cell 2006, 38, 193–201. [Google Scholar] [CrossRef]
- Papadogiannis, V.; Pennati, A.; Parker, H.J.; Rothbächer, U.; Patthey, C.; Bronner, M.E.; Shimeld, S.M. Hmx gene conservation identifies the origin of vertebrate cranial ganglia. Nature 2022, 605, 701–705. [Google Scholar] [CrossRef]
- Schiaffino, S.; Burighel, P.; Nunzi, M.G. Involution of the caudal musculature during metamorphosis in the ascidian, Botryllus schlosseri. Cell Tissue Res. 1974, 153, 293–305. [Google Scholar] [CrossRef]
- Chambon, J.-P.; Soule, J.; Pomies, P.; Fort, P.; Sahuquet, A.; Alexandre, D.; Mangeat, P.-H.; Baghdiguian, S. Tail regression in Ciona intestinalis (Prochordate) involves a Caspase-dependent apoptosis event associated with ERK activation. Development 2002, 129, 3105–3114. [Google Scholar] [CrossRef]
- Krasovec, G.; Robine, K.; Quéinnec, E.; Karaiskou, A.; Chambon, J.P. Ci-hox12 tail gradient precedes and participates in the control of the apoptotic-dependent tail regression during Ciona larva metamorphosis. Dev. Biol. 2019, 448, 237–246. [Google Scholar] [CrossRef]
- Comes, S.; Locascio, A.; Silvestre, F.; d’Ischia, M.; Russo, G.L.; Tosti, E.; Branno, M.; Palumbo, A. Regulatory roles of nitric oxide during larval development and metamorphosis in Ciona intestinalis. Dev. Biol. 2007, 306, 772–784. [Google Scholar] [CrossRef]
- Rinkevich, B.; Lauzon, R.J.; Brown, B.W.; Weissman, I.L. Evidence for a programmed life span in a colonial protochordate. Proc. Natl. Acad. Sci. USA 1992, 89, 3546–3550. [Google Scholar] [CrossRef]
- Voskoboynik, A.; Rinkevich, B.; Weiss, A.; Moiseeva, E.; Reznick, A.Z. Macrophage involvement for successful degeneration of apoptotic organs in the colonial urochordate Botryllus schlosseri. J. Exp. Biol. 2004, 207, 2409–2416. [Google Scholar] [CrossRef]
- Cima, F.; Ballarin, L. Apoptosis and pattern of Bcl-2 and Bax expression in the alimentary tract during the colonial blastogenetic cycle of Botryllus schlosseri (Urochordata, Ascidiacea). Ital. J. Zool. 2009, 76, 28–42. [Google Scholar] [CrossRef]
- Franchi, N.; Ballin, F.; Manni, L.; Schiavon, F.; Basso, G.; Ballarin, L. Recurrent phagocytosis-induced apoptosis in the cyclical generation change of the compound ascidian Botryllus schlosseri. Dev. Comp. Immunol. 2016, 62, 8–16. [Google Scholar] [CrossRef] [PubMed]
- Sabbadin, A. The compound ascidian Botryllus-schlosseri in the field and in the laboratory. Pubbl. Della Stn. Zool. Napoli 1969, 37, 62–72. [Google Scholar]
- Lauzon, R.J.; Ishizuka, K.J.; Weissman, I.L. Cyclical generation and degeneration of organs in a colonial urochordate involves crosstalk between old and new: A model for development and regeneration. Dev. Biol. 2002, 249, 333–348. [Google Scholar] [CrossRef]
- Chadwick-Furman, N.E.; Weissman, I.L. Life histories and senescence of Botryllus schlosseri (Chordata, Ascidiacea) in Monterey Bay. Biol. Bull. 1995, 189, 36–41. [Google Scholar] [CrossRef][Green Version]
- Brunetti, R.; Copello, M. Growth and senescence in colonies of Botryllus schlosseri (pallas) (ascidiacea). Bolletino Zool. 1978, 45, 359–364. [Google Scholar] [CrossRef]
- Kowarsky, M.; Anselmi, C.; Hotta, K.; Burighel, P.; Zaniolo, G.; Caicci, F.; Rosental, B.; Neff, N.F.; Ishizuka, K.J.; Palmeri, K.J.; et al. Sexual and asexual development: Two distinct programs producing the same tunicate. Cell Rep. 2021, 34, 108681. [Google Scholar] [CrossRef]
- Di Bartolomeo, S.; Corazzari, M.; Nazio, F.; Oliverio, S.; Lisi, G.; Antonioli, M.; Pagliarini, V.; Matteoni, S.; Fuoco, C.; Giunta, L.; et al. The dynamic interaction of AMBRA1 with the dynein motor complex regulates mammalian autophagy. J. Cell Biol. 2010, 191, 155–168. [Google Scholar] [CrossRef]
- He, C.; Levine, B. The Beclin 1 interactome. Curr. Opin. Cell Biol. 2010, 22, 140–149. [Google Scholar] [CrossRef] [PubMed]
- Fimia, G.M.; Stoykova, A.; Romagnoli, A.; Giunta, L.; Di Bartolomeo, S.; Nardacci, R.; Corazzari, M.; Fuoco, C.; Ucar, A.; Schwartz, P.; et al. Ambra1 regulates autophagy and development of the nervous system. Nature 2007, 447, 1121–1125. [Google Scholar] [CrossRef]
- Burighel, P.; Lane, N.J.; Zaniolo, G.; Manni, L. Neurogenic role of the neural gland in the development of the ascidian, Botryllus schlosseri (Tunicata, Urochordata). J. Comp. Neurol. 1998, 394, 230–241. [Google Scholar] [CrossRef]
- Burighel, P.; Sorrentino, M.; Zaniolo, G.; Thorndyke, M.C.; Manni, L. The peripheral nervous system of an ascidian, Botryllus schlosseri, as revealed by cholinesterase activity. Invertebr. Biol. 2001, 120, 185–198. [Google Scholar] [CrossRef]
- Ruppert, E.E. Structure, Ultrastructure and Function of the Neural Gland Complex of Ascidia interrupta (Chordata, Ascidiacea): Clarification of Hypotheses Regarding the Evolution of the Vertebrate Anterior Pituitary. Acta Zool. 1990, 71, 135–149. [Google Scholar] [CrossRef]
- Deyts, C.; Casane, D.; Vernier, P.; Bourrat, F.; Joly, J.-S. Morphological and gene expression similarities suggest that the ascidian neural gland may be osmoregulatory and homologous to vertebrate peri-ventricular organs. Eur. J. Neurosci. 2006, 24, 2299–2308. [Google Scholar] [CrossRef]
- Hetz, C.; Zhang, K.; Kaufman, R.J. Mechanisms, regulation and functions of the unfolded protein response. Nat. Rev. Mol. Cell Biol. 2020, 21, 421–438. [Google Scholar] [CrossRef]
- González-Sanmiguel, J.; Schuh, C.M.A.P.; Muñoz-Montesino, C.; Contreras-Kallens, P.; Aguayo, L.G.; Aguayo, S. Complex Interaction between Resident Microbiota and Misfolded Proteins: Role in Neuroinflammation and Neurodegeneration. Cells 2020, 9, 2476. [Google Scholar] [CrossRef]
- Gupta, R.; Ambasta, R.K.; Kumar, P. Autophagy and apoptosis cascade: Which is more prominent in neuronal death? Cell. Mol. Life Sci. 2021, 78, 8001–8047. [Google Scholar] [CrossRef]
- Vascellari, S.; Manzin, A. Parkinson’s disease: A prionopathy? Int. J. Mol. Sci. 2021, 22, 8022. [Google Scholar] [CrossRef]
- Freundt, E.C.; Maynard, N.; Clancy, E.K.; Roy, S.; Bousset, L.; Sourigues, Y.; Covert, M.; Melki, R.; Kirkegaard, K.; Brahic, M. Neuron-to-neuron transmission of α-synuclein fibrils through axonal transport. Ann. Neurol. 2012, 72, 517–524. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.-J.; Desplats, P.; Sigurdson, C.; Tsigelny, I.; Masliah, E. Cell-to-cell transmission of non-prion protein aggregates. Nat. Rev. Neurol. 2010, 6, 702–706. [Google Scholar] [CrossRef]
- Ballarin, L.; Menin, A.; Tallandini, L.; Matozzo, V.; Burighel, P.; Basso, G.; Fortunato, E.; Cima, F. Haemocytes and blastogenetic cycle in the colonial ascidian Botryllus schlosseri: A matter of life and death. Cell Tissue Res. 2008, 331, 555–564. [Google Scholar] [CrossRef]
- Manni, L.; Gasparini, F.; Hotta, K.; Ishizuka, K.J.; Ricci, L.; Tiozzo, S.; Voskoboynik, A.; Dauga, D. Ontology for the asexual development and anatomy of the colonial chordate Botryllus schlosseri. PLoS ONE 2014, 9, e96434. [Google Scholar] [CrossRef]
- Köster, J.; Rahmann, S. Snakemake—A scalable bioinformatics workflow engine. Bioinformatics 2012, 28, 2520–2522. [Google Scholar] [CrossRef] [PubMed]
- Ben-Ari Fuchs, S.; Lieder, I.; Stelzer, G.; Mazor, Y.; Buzhor, E.; Kaplan, S.; Bogoch, Y.; Plaschkes, I.; Shitrit, A.; Rappaport, N.; et al. Geneanalytics: An integrative gene set analysis tool for next generation sequencing, rnaseq and microarray data. OMICS 2016, 20, 139–151. [Google Scholar] [CrossRef] [PubMed]
- Love, M.I.; Huber, W.; Anders, S. Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol. 2014, 15, 550. [Google Scholar] [CrossRef]
- Sabbadin, A. Osservazioni sullo sviluppo, l’accrescimento e la riproduzione di Botryllus schlosseri (Pallas), in condizioni di laboratorio. Bolletino Zool. 1955, 22, 243–263. [Google Scholar] [CrossRef]
- Burighel, P.; Schiavinato, A. Degenerative regression of the digestive tract in the colonial ascidian Botryllus schlosseri (Pallas). Cell Tissue Res. 1984, 235, 309–318. [Google Scholar] [CrossRef]
- Brunetti, R.; Burighel, P. Sviluppo dell’apparato vascolare coloniale in Botryllus schlosseri (Pallas). Staz. Zoolo. Napoli 1969, 37, 137–148. [Google Scholar]
- Manni, L.; Anselmi, C.; Burighel, P.; Martini, M.; Gasparini, F. Differentiation and induced sensorial alteration of the coronal organ in the asexual life of a tunicate. Integr. Comp. Biol. 2018, 58, 317–328. [Google Scholar] [CrossRef] [PubMed]
- Anselmi, C.; Kowarsky, M.; Gasparini, F.; Caicci, F.; Ishizuka, K.J.; Palmeri, K.J.; Sinha, R.A.; Neff, N.; Quake, S.R.; Weissman, I.L.; et al. Revealing conserved mechanisms of neurodegeneration in a colonial chordate. BioRxiv 2021. [Google Scholar] [CrossRef]
- Cho, H.; Chung, J.-Y.; Song, K.-H.; Noh, K.H.; Kim, B.W.; Chung, E.J.; Ylaya, K.; Kim, J.H.; Kim, T.W.; Hewitt, S.M.; et al. Apoptosis inhibitor-5 overexpression is associated with tumor progression and poor prognosis in patients with cervical cancer. BMC Cancer 2014, 14, 545. [Google Scholar] [CrossRef]
- Gao, Z.; Man, X.; Li, Z.; Bi, J.; Liu, X.; Li, Z.; Li, J.; Zhang, Z.; Kong, C. PLK1 promotes proliferation and suppresses apoptosis of renal cell carcinoma cells by phosphorylating MCM3. Cancer Gene Ther. 2020, 27, 412–423. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Kuang, Y.; Ma, K.; Li, Y.; Liu, X.; Shi, Y.; Wu, X. Gclc overexpression inhibits apoptosis of bone marrow mesenchymal stem cells through the PI3K/AKT/Foxo1 pathway to alleviate inflammation in acute lung injury. Int. Immunopharmacol. 2022, 110, 109017. [Google Scholar] [CrossRef]
- Silke, J.; Meier, P. Inhibitor of apoptosis (IAP) proteins-modulators of cell death and inflammation. Cold Spring Harb. Perspect. Biol. 2013, 5, a008730. [Google Scholar] [CrossRef]
- Cheung, C.H.A.; Chang, Y.-C.; Lin, T.-Y.; Cheng, S.M.; Leung, E. Anti-apoptotic proteins in the autophagic world: An update on functions of XIAP, Survivin, and BRUCE. J. Biomed. Sci. 2020, 27, 31. [Google Scholar] [CrossRef]
- Xu, C.; Du, Z.; Ren, S.; Liang, X.; Li, H. MiR-129-5p sensitization of lung cancer cells to etoposide-induced apoptosis by reducing YWHAB. J. Cancer 2020, 11, 858–866. [Google Scholar] [CrossRef]
- O’Brien, M.A.; Kirby, R. Apoptosis: A review of pro-apoptotic and anti-apoptotic pathways and dysregulation in disease. J. Veter. Emer. Crit. 2008, 18, 572–585. [Google Scholar] [CrossRef]
- Su, L.; Pan, P.; Yan, P.; Long, Y.; Zhou, X.; Wang, X.; Zhou, R.; Wen, B.; Xie, L.; Liu, D. Role of vimentin in modulating immune cell apoptosis and inflammatory responses in sepsis. Sci. Rep. 2019, 9, 5747. [Google Scholar] [CrossRef]
- Goedert, M. NEURODEGENERATION. Alzheimer’s and Parkinson’s diseases: The prion concept in relation to assembled Aβ, tau, and α-synuclein. Science 2015, 349, 1255555. [Google Scholar] [CrossRef]
- Duyckaerts, C.; Clavaguera, F.; Potier, M.-C. The prion-like propagation hypothesis in Alzheimer’s and Parkinson’s disease. Curr. Opin. Neurol. 2019, 32, 266–271. [Google Scholar] [CrossRef] [PubMed]
- Boeve, B.F.; Boxer, A.L.; Kumfor, F.; Pijnenburg, Y.; Rohrer, J.D. Advances and controversies in frontotemporal dementia: Diagnosis, biomarkers, and therapeutic considerations. Lancet Neurol. 2022, 21, 258–272. [Google Scholar] [CrossRef] [PubMed]
- Liberski, P.P.; Jeffrey, M. Tubulovesicular structures: What are they really? Microsc. Res. Tech. 2000, 50, 46–57. [Google Scholar] [CrossRef]
- Liberski, P. The tubulovesicular structures—The ultrastructural hallmark for all prion diseases. Acta Neurobiol. Exp. (Wars) 2008, 68, 113–121. [Google Scholar]
- Sigurdson, C.J.; Bartz, J.C.; Glatzel, M. Cellular and molecular mechanisms of prion disease. Annu. Rev. Pathol. 2019, 14, 497–516. [Google Scholar] [CrossRef]
- Schweichel, J.U.; Merker, H.J. The morphology of various types of cell death in prenatal tissues. Teratology 1973, 7, 253–266. [Google Scholar] [CrossRef] [PubMed]
- Thompson, S.H.; Anselmi, C.; Ishizuka, K.J.; Palmeri, K.J.; Voskoboynik, A. Contributions from both the brain and the vascular network guide behavior in the colonial tunicate Botryllus schlosseri. J. Exp. Biol. 2022, 225, jeb244491. [Google Scholar] [CrossRef] [PubMed]
- Manni, L.; Anselmi, C.; Cima, F.; Gasparini, F.; Voskoboynik, A.; Martini, M.; Peronato, A.; Burighel, P.; Zaniolo, G.; Ballarin, L. Sixty years of experimental studies on the blastogenesis of the colonial tunicate Botryllus schlosseri. Dev. Biol. 2019, 448, 293–308. [Google Scholar] [CrossRef] [PubMed]
- Gasparini, F.; Caicci, F.; Rigon, F.; Zaniolo, G.; Manni, L. Testing an unusual in vivo vessel network model: A method to study angiogenesis in the colonial tunicate Botryllus schlosseri. Sci. Rep. 2014, 4, 6460. [Google Scholar] [CrossRef]
- Gordon, T.; Zaquin, T.; Kowarsky, M.A.; Voskoboynik, Y.; Hendin, N.; Wurtzel, O.; Caicci, F.; Manni, L.; Voskoboynik, A.; Shenkar, N. Stemness activity underlying whole brain regeneration in a basal chordate. Cells 2022, 11, 3727. [Google Scholar] [CrossRef] [PubMed]
- Zaniolo, G.; Lane, N.J.; Burighel, P.; Manni, L. Development of the motor nervous system in ascidians. J. Comp. Neurol. 2002, 443, 124–135. [Google Scholar] [CrossRef] [PubMed]
- Goldstein, O.; Mandujano-Tinoco, E.A.; Levy, T.; Talice, S.; Raveh, T.; Gershoni-Yahalom, O.; Voskoboynik, A.; Rosental, B. Botryllus schlosseri as a Unique Colonial Chordate Model for the Study and Modulation of Innate Immune Activity. Mar. Drugs 2021, 19, 454. [Google Scholar] [CrossRef] [PubMed]
- Rinkevich, B.; Weissman, I.L. Botryllus schlosseri (Tunicata) whole colony irradiation: Do senescent zooid resorption and immunological resorption involve similar recognition events? J. Exp. Zool. 1990, 253, 189–201. [Google Scholar] [CrossRef]
- Corey, D.M.; Rosental, B.; Kowarsky, M.; Sinha, R.; Ishizuka, K.J.; Palmeri, K.J.; Quake, S.R.; Voskoboynik, A.; Weissman, I.L. Developmental cell death programs license cytotoxic cells to eliminate histocompatible partners. Proc. Natl. Acad. Sci. USA 2016, 113, 6520–6525. [Google Scholar] [CrossRef]
- Goodbody, I. The physiology of ascidians. In Advances in Marine Biology; Elsevier: Amsterdam, The Netherlands, 1975; Volume 12, pp. 1–149. ISBN 9780120261123. [Google Scholar]
- Bone, Q.; Whitear, M. A note on the innervation of the pericardium inCiona. Publ. Stn. Zool. Napoli 1958, 30, 337–341. [Google Scholar]
- Cima, F.; Manni, L.; Basso, G.; Fortunato, E.; Accordi, B.; Schiavon, F.; Ballarin, L. Hovering between death and life: Natural apoptosis and phagocytes in the blastogenetic cycle of the colonial ascidian Botryllus schlosseri. Dev. Comp. Immunol. 2010, 34, 272–285. [Google Scholar] [CrossRef]
- Xu, P.-X.; Zheng, W.; Huang, L.; Maire, P.; Laclef, C.; Silvius, D. Six1 is required for the early organogenesis of mammalian kidney. Development 2003, 130, 3085–3094. [Google Scholar] [CrossRef]
- Campagna, D.; Gasparini, F.; Franchi, N.; Vitulo, N.; Ballin, F.; Manni, L.; Valle, G.; Ballarin, L. Transcriptome dynamics in the asexual cycle of the chordate Botryllus schlosseri. BMC Genom. 2016, 17, 275. [Google Scholar] [CrossRef]
- Repnik, U.; Stoka, V.; Turk, V.; Turk, B. Lysosomes and lysosomal cathepsins in cell death. Biochim. Biophys. Acta 2012, 1824, 22–33. [Google Scholar] [CrossRef]
- Rosner, A.; Kravchenko, O.; Rinkevich, B. IAP genes partake weighty roles in the astogeny and whole body regeneration in the colonial urochordate Botryllus schlosseri. Dev. Biol. 2019, 448, 320–341. [Google Scholar] [CrossRef] [PubMed]
- Frazzi, R. BIRC3 and BIRC5: Multi-faceted inhibitors in cancer. Cell Biosci. 2021, 11, 8. [Google Scholar] [CrossRef] [PubMed]
- Ballarin, L.; Burighel, P.; Cima, F. A tale of death and life: Natural apoptosis in the colonial ascidian Botryllus schlosseri (Urochordata, Ascidiacea). Curr. Pharm. Des. 2008, 14, 138–147. [Google Scholar] [CrossRef] [PubMed]
- Voskoboynik, A.; Newman, A.M.; Corey, D.M.; Sahoo, D.; Pushkarev, D.; Neff, N.F.; Passarelli, B.; Koh, W.; Ishizuka, K.J.; Palmeri, K.J.; et al. Identification of a colonial chordate histocompatibility gene. Science 2013, 341, 384–387. [Google Scholar] [CrossRef] [PubMed]
- Ballarin, L.; Menin, A.; Franchi, N.; Bertoloni, G.; Cima, F. Morula cells and non-self recognition in the compound ascidian Botryllus schlosseri. Invertebr. Surviv. J. 2005, 2, 1–5. [Google Scholar]
- Shlomovitz, I.; Speir, M.; Gerlic, M. Flipping the dogma—Phosphatidylserine in non-apoptotic cell death. Cell Commun. Signal. 2019, 17, 139. [Google Scholar] [CrossRef] [PubMed]
- Cima, F.; Basso, G.; Ballarin, L. Apoptosis and phosphatidylserine-mediated recognition during the take-over phase of the colonial life-cycle in the ascidian Botryllus schlosseri. Cell Tissue Res. 2003, 312, 369–376. [Google Scholar] [CrossRef]
- Menin, A.; Ballarin, L. Immunomodulatory molecules in the compound ascidian Botryllus schlosseri: Evidence from conditioned media. J. Invertebr. Pathol. 2008, 99, 275–280. [Google Scholar] [CrossRef] [PubMed]
- Peronato, A.; Drago, L.; Rothbächer, U.; Macor, P.; Ballarin, L.; Franchi, N. Complement system and phagocytosis in a colonial protochordate. Dev. Comp. Immunol. 2020, 103, 103530. [Google Scholar] [CrossRef] [PubMed]
- Franchi, N.; Ballarin, L. Preliminary characterization of complement in a colonial tunicate: C3, Bf and inhibition of C3 opsonic activity by compstatin. Dev. Comp. Immunol. 2014, 46, 430–438. [Google Scholar] [CrossRef]
- Li, B.; Chen, M.; Zhu, C. Neuroinflammation in prion disease. Int. J. Mol. Sci. 2021, 22, 2196. [Google Scholar] [CrossRef] [PubMed]
- Franchi, N.; Ballarin, L.; Peronato, A.; Cima, F.; Grimaldi, A.; Girardello, R.; de Eguileor, M. Functional amyloidogenesis in immunocytes from the colonial ascidian Botryllus schlosseri: Evolutionary perspective. Dev. Comp. Immunol. 2019, 90, 108–120. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Wang, B.; Wang, Z.; Guo, Q.; Tabuchi, K.; Hammer, R.E.; Südhof, T.C.; Zheng, H. Soluble amyloid precursor protein (APP) regulates transthyretin and Klotho gene expression without rescuing the essential function of APP. Proc. Natl. Acad. Sci. USA 2010, 107, 17362–17367. [Google Scholar] [CrossRef] [PubMed]
- Turner, P.R.; O’Connor, K.; Tate, W.P.; Abraham, W.C. Roles of amyloid precursor protein and its fragments in regulating neural activity, plasticity and memory. Prog. Neurobiol. 2003, 70, 1–32. [Google Scholar] [CrossRef]
- Nikolaev, A.; McLaughlin, T.; O’Leary, D.D.M.; Tessier-Lavigne, M. APP binds DR6 to trigger axon pruning and neuron death via distinct caspases. Nature 2009, 457, 981–989. [Google Scholar] [CrossRef]
- Ma, J.; Wang, F. Prion disease and the “protein-only hypothesis”. Essays Biochem. 2014, 56, 181–191. [Google Scholar] [CrossRef]
- Woerman, A.L.; Watts, J.C.; Aoyagi, A.; Giles, K.; Middleton, L.T.; Prusiner, S.B. α-Synuclein: Multiple System Atrophy Prions. Cold Spring Harb. Perspect. Med. 2018, 8, a024588. [Google Scholar] [CrossRef]
- Espay, A.J.; Okun, M.S. Abandoning the proteinopathy paradigm in parkinson disease. JAMA Neurol. 2022, 80, 123. [Google Scholar] [CrossRef]
- Lang, A.E.; Siderowf, A.D.; Macklin, E.A.; Poewe, W.; Brooks, D.J.; Fernandez, H.H.; Rascol, O.; Giladi, N.; Stocchi, F.; Tanner, C.M.; et al. SPARK Investigators Trial of cinpanemab in early parkinson’s disease. N. Engl. J. Med. 2022, 387, 408–420. [Google Scholar] [CrossRef]
- Pagano, G.; Boess, F.G.; Taylor, K.I.; Ricci, B.; Mollenhauer, B.; Poewe, W.; Boulay, A.; Anzures-Cabrera, J.; Vogt, A.; Marchesi, M.; et al. Prasinezumab Study Group A phase II study to evaluate the safety and efficacy of prasinezumab in early parkinson’s disease (PASADENA): Rationale, design, and baseline data. Front. Neurol. 2021, 12, 705407. [Google Scholar] [CrossRef]
- Virata, M.J.; Zeller, R.W. Ascidians: An invertebrate chordate model to study Alzheimer’s disease pathogenesis. Dis. Model. Mech. 2010, 3, 377–385. [Google Scholar] [CrossRef] [PubMed]
- Horsager, J.; Andersen, K.B.; Knudsen, K.; Skjærbæk, C.; Fedorova, T.D.; Okkels, N.; Schaeffer, E.; Bonkat, S.K.; Geday, J.; Otto, M.; et al. Brain-first versus body-first Parkinson’s disease: A multimodal imaging case-control study. Brain 2020, 143, 3077–3088. [Google Scholar] [CrossRef]
- Olanow, C.W.; Brundin, P. Parkinson’s disease and alpha synuclein: Is Parkinson’s disease a prion-like disorder? Mov. Disord. 2013, 28, 31–40. [Google Scholar] [CrossRef] [PubMed]
- Uemura, N.; Yagi, H.; Uemura, M.T.; Hatanaka, Y.; Yamakado, H.; Takahashi, R. Correction to: Inoculation of α-synuclein preformed fibrils into the mouse gastrointestinal tract induces Lewy body-like aggregates in the brainstem via the vagus nerve. Mol. Neurodegener. 2019, 14, 31. [Google Scholar] [CrossRef] [PubMed]
- Okuzumi, A.; Kurosawa, M.; Hatano, T.; Takanashi, M.; Nojiri, S.; Fukuhara, T.; Yamanaka, T.; Miyazaki, H.; Yoshinaga, S.; Furukawa, Y.; et al. Rapid dissemination of alpha-synuclein seeds through neural circuits in an in-vivo prion-like seeding experiment. Acta Neuropathol. Commun. 2018, 6, 96. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.; Kwon, S.-H.; Kam, T.-I.; Panicker, N.; Karuppagounder, S.S.; Lee, S.; Lee, J.H.; Kim, W.R.; Kook, M.; Foss, C.A.; et al. Transneuronal Propagation of Pathologic α-Synuclein from the Gut to the Brain Models Parkinson’s Disease. Neuron 2019, 103, 627–641. [Google Scholar] [CrossRef]
- Luk, K.C.; Kehm, V.; Carroll, J.; Zhang, B.; O’Brien, P.; Trojanowski, J.Q.; Lee, V.M.-Y. Pathological α-synuclein transmission initiates Parkinson-like neurodegeneration in nontransgenic mice. Science 2012, 338, 949–953. [Google Scholar] [CrossRef]






| Sub-Stage | Duration | Description |
|---|---|---|
| 111 | 2–4 h | Onset of takeover, recognizable by adult zooid siphon retraction and closure |
| 112 | 4–7 h | General shrinkage of regressing zooids that progressively reduce their dimension |
| 113 | 12–16 h | Further contraction of regressing zooids that in length are about half of their primary buds; inner organs, other than the beating heart, are no longer recognizable; zooids converge progressively toward the system center, in ventral position with respect to their growing primary buds |
| 114 | 6–9 h | Regressing zooids are spherical dark masses at the system center; their hearts do not beat |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Anselmi, C.; Caicci, F.; Bocci, T.; Guidetti, M.; Priori, A.; Giusti, V.; Levy, T.; Raveh, T.; Voskoboynik, A.; Weissman, I.L.; et al. Multiple Forms of Neural Cell Death in the Cyclical Brain Degeneration of A Colonial Chordate. Cells 2023, 12, 1041. https://doi.org/10.3390/cells12071041
Anselmi C, Caicci F, Bocci T, Guidetti M, Priori A, Giusti V, Levy T, Raveh T, Voskoboynik A, Weissman IL, et al. Multiple Forms of Neural Cell Death in the Cyclical Brain Degeneration of A Colonial Chordate. Cells. 2023; 12(7):1041. https://doi.org/10.3390/cells12071041
Chicago/Turabian StyleAnselmi, Chiara, Federico Caicci, Tommaso Bocci, Matteo Guidetti, Alberto Priori, Veronica Giusti, Tom Levy, Tal Raveh, Ayelet Voskoboynik, Irving L. Weissman, and et al. 2023. "Multiple Forms of Neural Cell Death in the Cyclical Brain Degeneration of A Colonial Chordate" Cells 12, no. 7: 1041. https://doi.org/10.3390/cells12071041
APA StyleAnselmi, C., Caicci, F., Bocci, T., Guidetti, M., Priori, A., Giusti, V., Levy, T., Raveh, T., Voskoboynik, A., Weissman, I. L., & Manni, L. (2023). Multiple Forms of Neural Cell Death in the Cyclical Brain Degeneration of A Colonial Chordate. Cells, 12(7), 1041. https://doi.org/10.3390/cells12071041







