The Vitamin D Receptor as a Potential Target for the Treatment of Age-Related Neurodegenerative Diseases Such as Alzheimer’s and Parkinson’s Diseases: A Narrative Review
Abstract
1. Introduction
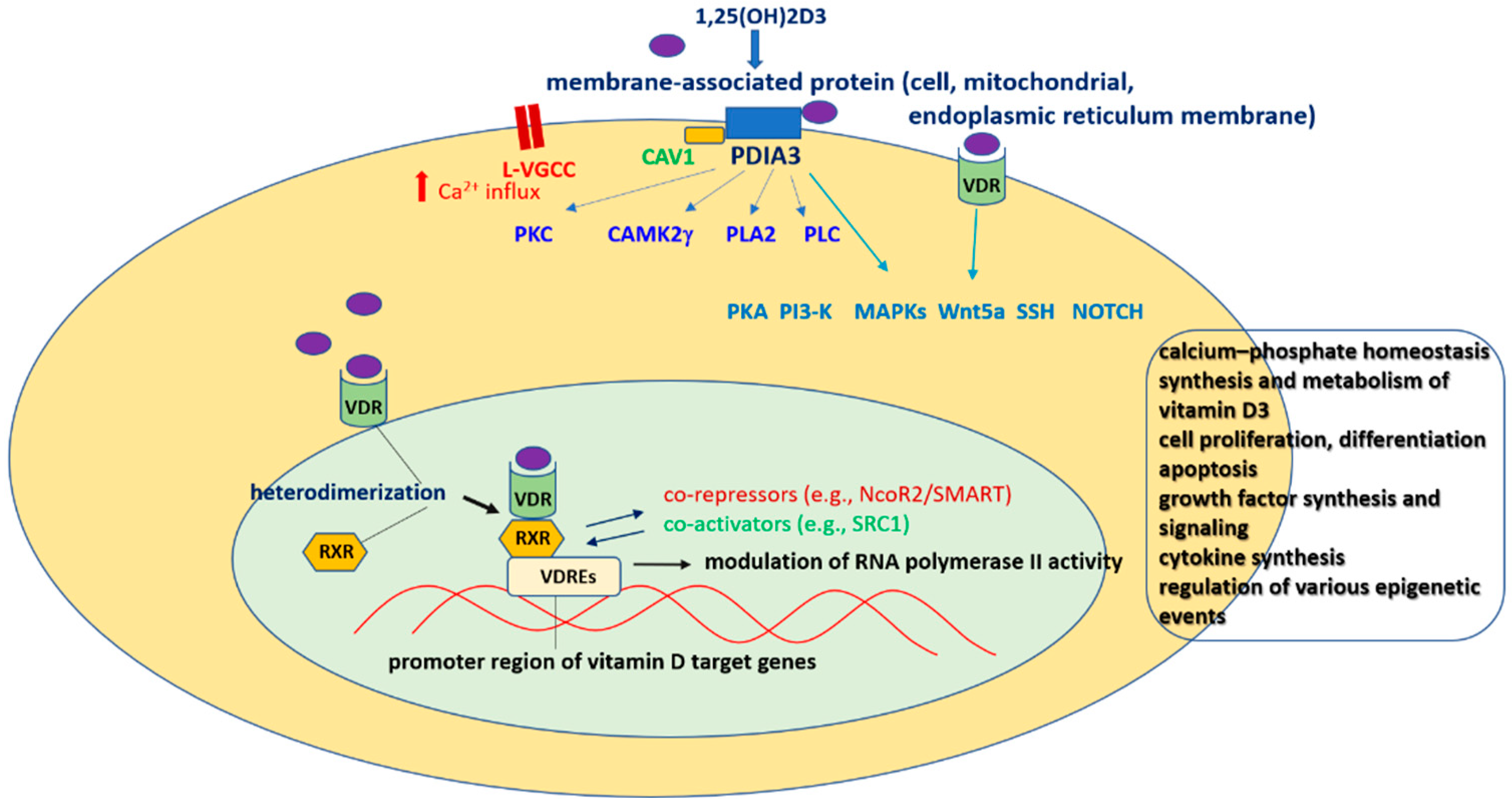
Vitamin D and Molecular Characteristics of Its Genomic and Nongenomic Action
2. The Brain Expression of the VDR
Brain Aging
3. Alzheimer’s Disease
3.1. Experimental Studies
| AD Model | Vitamin D Administration | Effects | References |
|---|---|---|---|
| Aβ(1–42) peptide-induced toxicity in differentiated SH-SY5Y cells | calcitriol (100 nM)/24 h prior to Aβ(1−42) peptide (1 μM)/6 h | ↑ cell viability ↑ sphingosine-1-phosphate, sphingosine kinase ↓ p38MAPK/ATF4 axis | [97] |
| Aβ(25–35) peptide induced toxicity in SH-SY5Y cells | calcitriol (0.1 or 10 nM)/24 h after Aβ(25–35) peptide | ↑ cell viability ↑ VDR protein expression ↑ GDNF expression ↓ cell apoptosis ↓ ROS level ↓ p-tau/tau ↑ p-Akt/Akt | [98] |
| Aβ(1–42) peptide-induced toxicity in primary cortical neuronal cultures | vitamin D3 (1 nM), curcumin (5 μM), vitamin D3 + curcumin Aβ(1–42) (1 µM)/72 h | ↓ lipid peroxidation ↑ reduced GSH ↑ GST enzyme level ↑ NGF | [99] |
| SH-SY5Y cells | calcitriol (10–500 nM)/48 h | ↑ IL-34 mRNA and protein ↑ VDR expression | [109] |
| SH-SY5Y wt cells, SH-SY5Y APP695 cells and N2a cells | calcifediol, maxacalcitol, calcipotriol, alfacalcidol, paricalcitol, doxercalciferol (100 nM)/24 h | ↓ Aβ-production ↑ Aβ-degradation ↓ γ-secretase activity ↓ β-secretase 1 ↓ IL-1β | [113] |
| Aβ(25–35)-induced damage of BV-2 microglia cells | ergocalciferol (1 μM)/1 h prior to Aβ(25–35) (20 μM)/24 h | ↓ ROS, IL-6, IL-1β, TNF-α, iNOS, COX-2 and NF-κB activation | [110] |
| BBB model (bEnd.3 cells) HepG2 | calcitriol (1–100 nM)/control or hypoxia/60 h/+ Aβ(1–40) (100 nM) calcitriol (1–100 nM)/6 h/ + Aβ(1–40) (115 nM) | ↓ cerebral Aβ1–40 level ↑ LRP-1 expression ↑ VDR expression ↓ RAGE expression ↑ Aβ1–40 uptake | [59] |
| Hydrogen peroxide-induced damage of HUVEC line | vitamin D and VDR ligand (ZK191784) + hydrogen peroxide (200 μM)/20 min | ↓ apoptosis-related gene expression, ↑ pro-autophagic Beclin 1 and the phosphorylation of ERK1/2 and Akt, ↑ mitochondrial potential ↓ cytochrome C release and caspase activation | [122] |
| NO-deprived BMECs | - | ↑ APP, BACE1, Aβ(1–41) and Aβ(1–42) peptides | [120] |
| AD Model | Vitamin D Administration | Effects | References |
|---|---|---|---|
| The human amyloid precursor protein-expressing models-Tg2576 and TgCRND8 mice | calcitriol (2.5 μg/kg, i.p., q2d × 4) | ↑ brain P-glycoprotein ↓ soluble and insoluble plaque-associated Aβ ↑ conditioned fear memory | [96] |
| Intrahippocampal injection of Aβ(1−42) in rats Intraventricular injection of Aβ(1−42) in mice | calcitriol (1 μg/kg)/day/6 consecutive days before the hippocampal delivery of aggregated Aβ(1−42) and at days 8,11,13,15,18 after Aβ(1−42) calcitriol (1 μg/kg)/day/5 days before Aβ(1−42) and at days 8 and 12 after Aβ(1−42) | ↓ neuronal damage in CA1 ↑ neurogenesis in dentate gyrus | [97] |
| Rats at different ages (5, 12 and 24 months) | cholecalciferol (500 IU/kg, orally)/day/5 weeks | ↑ brain BDNF level, AChE, antioxidant enzymes; ↓ malondialdehyde level, caspase-3 activity | [94] |
| APPswe/PS1E9 and C57BL/6J mice | calcitriol (100 ng/kg), i.p./daily/6 weeks | ↑ learning and memory abilities ↓ morphological defects in hippocampal neurons ↓ caspase-3, Bax ↑ Bcl-2, VDR, p-ERK1 | [100] |
| AβPP-PS1-transgenic mice | cholecalciferol (control diet, 2.4 IU/g), (Surplus vitamin D diet ~12 IU/g) or (vitamin D deficient diet, 0 IU/g) | ↓ the number of amyloid plaques, Aβ peptides, TNF-α ↑ NGF | [101] |
| Two-month-old wild-type (B6C3) and AD transgenic (APPSwe/PS1dE9) mice | Diet deficient in vitamin D2 or a diet supplemented with vitamin-D2-enriched mushrooms (VDM), containing 160.2 mg/kg (54 IU/kg) vitamin D2/7 months | ↑ learning and memory ↓ amyloid plaque load and glial fibrillary acidic protein ↑ interleukin-10 in the brain of VDM-fed wild type and AD transgenic mice | [102] |
| Intracerebroventricular STZ injection in rats | STZ (1.5 mg/kg)/first and third day cholecalciferol (42 IU, oral gavage)/day/pre-treatment 7 days before STZ/21 days and post-treatment 24 h after STZ/21 days | ↑ spatial learning and memory functions ↓ neuronal oxidative stress ↓ mitochondrial aberrations ↑ cholinergic functions ↓ hippocampal neuroinflammatory response ↓ neuronal death in cortex and hippocampus | [104] |
| LPS-induced hippocampal damage in rats | Maxacalcitol (1 μg/kg, i.p.)/twice a day/4 weeks LPS (0.8 mg/kg, i.p./once a week/3 weeks before maxacalcitol | ↓ TNF-α, MDA, ↑ Nrf2 ↑ IL-10, HO-1, GSH ↓ Aβ, p-Tau ↓ MAPK-p38, ERK1/2 ↓ neuronal damage in CA1 ↑ cognitive functions | [105] |
| APP/PS1-transgenic mice | Paricalcitol (200 ng/kg)/once every two days/15 weeks | ↓ the iron accumulation in the cortex and hippocampus ↓ Transferrin receptor (TFR) and iron-regulatory protein 2 (IRP2) expression ↓ phosphorylation of Tau at Ser396 and Thr181 sites via inhibiting the GSK3β phosphorylation (Tyr216) | [106] |
| APPswe/PS1E9-transgenic mice | eriodictyol (50 mg/kg, i.p.)/3 times per week | ↑ Nrf2/HO-1 ↑ VDR expression ↓ ferroptosis | [107] |
| D-galactose-induced oxidative stress in mice | cholecalciferol (100 μg/kg i.p.)/3 times a week/4 weeks | ↑ the neuronal synapse and memory ↓ oxidative stress ↑ NRF-2, HO-1 ↓ NF-κB, TNF-α, IL-1β | [103] |
| mouse brain homogenates from WT and vitamin D deficient C57BL/6 mice | calcifediol, maxacalcitol, calcipotriol, alfacalcidol, paricalcitol, doxercalciferol (100 nM)/24 h | ↑ Aβ-degradation ↓ β-secretase 1 | [113] |
| 5XFAD transgenic mice (Transcriptomic analysis) | cholecalciferol (7500 IU/kg)/5 months | improved dysregulated pathways related to immune, inflammatory response, neurotransmitter activity, endothelial, vascular processes, hormonal alterations | [114] |
| Kun Ming mice | calciriol (2.5 μg/kg, i.p./day/14 days | ↑ LRP-1 expression ↑ VDR expression | [59] |
| Aging model in C57BL/6 fed a normal or high-fat/high-cholesterol diet | calcitriol (2.5 μg/kg, i.p.)/day/week | ↑ P-glycoprotein expression | [93] |
| APP/PS1-transgenic mice | Paricalcitol (200 ng/kg, i.p.)/once every two days/15 weeks | ↓ Aβ generation and neuronal loss ↑ LRP-1 expression ↑ lysosomal degradation of BACE1 ↓ 8-hydroxyguanosine generation in neuronal mitochondria resulting in the attenuation of calpain-1-mediated neuronal loss | [117] |
| LPS-treated rats | LPS (0.8 mg/kg, i.p.)/once a week/3 weeks + vitamin D3 (1 μg/kg)/twice a day/4 weeks and/or exercise (30 min/once daily/4 weeks | ↑ cognitive dysfunction ↓ IL-6, MDA, Aβ, tau proteins levels ↑ IL-10, GSH, AChE, dopamine, BDNF, NGF | [102] |
| UCMS rat model | Vitamin D3 (100, 1000, 10,000 IU/kg, i.p.)/4 weeks during UCMS | ↓ corticosterone ↑ spatial learning and memory ↓ oxidative stress ↓ Aβ ↑ BDNF | [118] |
| eNOS-deficient mice | - | ↑ APP, BACE1, Aβ peptides | [120] |
| APP/PS-1 double-transgenic (2xTg-AD) mice | - | ↓ PGC-1α ↓ VDR | [123] |
| Intrahippocampal Aβ(1–40) peptide injection in rats | calcitriol (1 µg/mL×1 mL/kg, i.p.)/day/14 days | ↑ learning and memory ↓ neuronal loss and oxidative stress (↓ MDA, ↑ SOD) | [111] |
| Intrahippocampal injection of Aβ(1–40) peptide in rats | cholecalciferol (5 μg/kg/day, i.p.)/2 weeks | ↓ Aβ-induced memory impairment ↑ antioxidant markers (TAC, TTG) ↓ stress oxidative biomarkers (LPO, DNA damage) | [112] |
| Double-transgenic APP/PS1 mice | vitamin D3-sufficient diet (600 IU/Kg of cholecalciferol) vitamin D3-supplemented diet (8044 IU/Kg of cholecalciferol) | ↓ serum level of vit. D3 ? ↑ Aβ deposition ? ↑ cognitive impairments ? | [127] |
| Transgenic hemizygous 5xFAD mice | vitamin-D-deficient diet cholecalciferol (410 ng/g, i.p.) | ↑ Aβ production and deposits, astrogliosis, memory impairments- restored after cholecalciferol administration | [115] |
3.2. Clinical Studies
4. Parkinson’s Disease
4.1. Experimental Studies
| PD Model | Vitamin D Administration | Effects | References |
|---|---|---|---|
| 6-OHDA-induced neurotoxicity in rat | calcitriol (1 μg/mL × 1 mL/kg, i.p. per day)/8 consecutive days | ↑ locomotor activity ↑ DA, DOPAC, HVA | [163] |
| 6-OHDA-induced lesion in rat | calcitriol (1 μg/kg)/day by gavage/7 days before 6-OHDA-induced lesion calcitriol (1 μg/kg)/day by gavage/14 days after 6-OHDA-induced lesion | ↓ behavioral changes ↓ DA depletion ↑ neuroprotective effects ↑ oxygen consumption rate ↓ mitochondria swelling, H2O2 production, SOD activity ↑ TH, DAT, VDAC, Hsp60 expression | [170] |
| 6-OHDA-induced lesion in mouse | calcitriol (2.56 μg/kg, i.p.)/once every two days (total 4 times) | ↓ dopaminergic loss ↓ neuroinflammation ↑ transcriptional VDR, VDR target genes ↑ endothelial P-glycoprotein | [171] |
| 6-OHDA -induced lesion in young adult (4 month old), middle-aged (14 month old) and aged (22 month old) rat | calcitriol (1.0 μg/kg, s.c.)/4 weeks after 6-OHDA (12 μg)/once a day/8 consecutive days | ↑ overflow of DA from the lesioned striatum, ↑ striatal DA in the young adult and middle-aged rats ↑ DA in substantia nigra in all age groups | [169] |
| 6-OHDA-induced lesion in mouse | cholecalciferol and/or L-DOPA/2 weeks post lesion | ↓ behavioral deficits, ↓ protein associated with dopamine metabolism ↓ biomarkers of oxidative stress ↑ contralateral wall touches, exploratory motor and cognitive activities ↑ TH, DAT, BDNF expression of, while ↓ MAO-B, CD11b, IL-1β and p47phox expression | [172] |
| 6-OHDA-induced lesion in rat | cholecalciferol (1 µg/kg)/21 days, in the absence and presence of physical exercise on a treadmill (30 min, speed of 20 cm/s, once a day/21 days/24 h after 6-OHDA | ↑ behavioral changes ↑ DA, DOPAC, TH, DAT ↑ VDR ↓ oxidative stress ↓ nitrite | [173] |
4.2. Clinical Studies
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Hou, Y.; Dan, X.; Babbar, M.; Wei, Y.; Hasselbalch, S.G.; Croteau, D.L.; Bohr, V.A. Ageing as a risk factor for neurodegenerative disease. Nat. Rev. Neurol. 2019, 15, 565–581. [Google Scholar] [CrossRef]
- Ellul, M.A.; Benjamin, L.; Singh, B.; Lant, S.; Michael, B.D.; Easton, A.; Kneen, R.; Defres, S.; Sejvar, J.; Solomon, T. Neurological associations of COVID-19. Lancet Neurol. 2020, 19, 767–783. [Google Scholar] [CrossRef]
- Roy, D.; Ghosh, R.; Dubey, S.; Dubey, M.J.; Benito-Leon, J.; Kanti Ray, B. Neurological and Neuropsychiatric Impacts of COVID-19 Pandemic. Can. J. Neurol. Sci. 2021, 48, 9–24. [Google Scholar] [CrossRef]
- Sankowski, R.; Mader, S.; Valdés-Ferrer, S.I. Systemic inflammation and the brain: Novel roles of genetic, molecular, and environmental cues as drivers of neurodegeneration. Front. Cell. Neurosci. 2015, 9, 28. [Google Scholar] [CrossRef]
- Higgins, V.; Sohaei, D.; Diamandis, E.P.; Prassas, I. COVID-19: From an acute to chronic disease? Potential long-term health consequences. Crit. Rev. Clin. Lab. Sci. 2021, 58, 297–310. [Google Scholar] [CrossRef]
- Ahuja, M.; Buabeid, M.; Abdel-Rahman, E.; Majrashi, M.; Parameshwaran, K.; Amin, R.; Ramesh, S.; Thiruchelvan, K.; Pondugula, S.; Suppiramaniam, V.; et al. Immunological alteration & toxic molecular inductions leading to cognitive impairment & neurotoxicity in transgenic mouse model of Alzheimer’s disease. Life Sci. 2017, 177, 49–59. [Google Scholar] [CrossRef]
- Khan, S.; Barve, K.H.; Kumar, M.S. Recent Advancements in Pathogenesis, Diagnostics and Treatment of Alzheimer’s Disease. Curr. Neuropharmacol. 2020, 18, 1106–1125. [Google Scholar] [CrossRef]
- Ladak, A.A.; Enam, S.A.; Ibrahim, M.T. A Review of the Molecular Mechanisms of Traumatic Brain Injury. World Neurosurg. 2019, 131, 126–132. [Google Scholar] [CrossRef]
- Ramanan, V.K.; Saykin, A.J. Pathways to neurodegeneration: Mechanistic insights from GWAS in Alzheimer’s disease, Parkinson’s disease, and related disorders. Am. J. Neurodegener. Dis. 2013, 2, 145–175. [Google Scholar]
- Raza, C.; Anjum, R.; Shakeel, N.U.A. Parkinson’s disease: Mechanisms, translational models and management strategies. Life Sci. 2019, 226, 77–90. [Google Scholar] [CrossRef]
- Sekerdag, E.; Solaroglu, I.; Gursoy-Ozdemir, Y. Cell Death Mechanisms in Stroke and Novel Molecular and Cellular Treatment Options. Curr. Neuropharmacol. 2018, 16, 1396–1415. [Google Scholar] [CrossRef]
- Trojan, E.; Tylek, K.; Schröder, N.; Kahl, I.; Brandenburg, L.O.; Mastromarino, M.; Leopoldo, M.; Basta-Kaim, A.; Lacivita, E. The N-Formyl Peptide Receptor 2 (FPR2) Agonist MR-39 Improves Ex Vivo and In Vivo Amyloid Beta (1–42)-Induced Neuroinflammation in Mouse Models of Alzheimer’s Disease. Mol. Neurobiol. 2021, 58, 6203–6221. [Google Scholar] [CrossRef]
- Di Pietro, V.; Yakoub, K.M.; Caruso, G.; Lazzarino, G.; Signoretti, S.; Barbey, A.K.; Tavazzi, B.; Lazzarino, G.; Belli, A.; Amorini, A.M. Antioxidant Therapies in Traumatic Brain Injury. Antioxidants 2020, 9, 260. [Google Scholar] [CrossRef]
- Jelinek, M.; Jurajda, M.; Duris, K. Oxidative stress in the brain: Basic concepts and treatment strategies in stroke. Antioxidants 2021, 10, 1886. [Google Scholar] [CrossRef] [PubMed]
- Niedzielska, E.; Smaga, I.; Gawlik, M.; Moniczewski, A.; Stankowicz, P.; Pera, J.; Filip, M. Oxidative Stress in Neurodegenerative Diseases. Mol. Neurobiol. 2016, 53, 4094–4125. [Google Scholar] [CrossRef]
- Rekatsina, M.; Paladini, A.; Piroli, A.; Zis, P.; Pergolizzi, J.V.; Varrassi, G. Pathophysiology and Therapeutic Perspectives of Oxidative Stress and Neurodegenerative Diseases: A Narrative Review. Adv. Ther. 2020, 37, 113–139. [Google Scholar] [CrossRef] [PubMed]
- Singh, N.; McMahon, H.; Bilderbeck, A.; Reed, Z.E.; Tunbridge, E.; Brett, D.; Geddes, J.R.; Churchill, G.C.; Goodwin, G.M. Plasma glutathione suggests oxidative stress is equally present in early- and late-onset bipolar disorder. Bipolar Disord. 2019, 21, 61–67. [Google Scholar] [CrossRef]
- Cobley, J.N.; Fiorello, M.L.; Bailey, D.M. 13 Reasons Why the Brain Is Susceptible to Oxidative Stress. Redox Biol. 2018, 15, 490–503. [Google Scholar] [CrossRef]
- Jakaria, M.; Belaidi, A.A.; Bush, A.I.; Ayton, S. Ferroptosis as a mechanism of neurodegeneration in Alzheimer’s disease. J. Neurochem. 2021, 159, 804–825. [Google Scholar] [CrossRef]
- Ward, R.J.; Zucca, F.A.; Duyn, J.H.; Crichton, R.R.; Zecca, L. The role of iron in brain ageing and neurodegenerative disorders. Lancet Neurol. 2014, 13, 1045–1060. [Google Scholar] [CrossRef]
- Chi, H.; Chang, H.-Y.; Sang, T.-K. Neuronal Cell Death Mechanisms in Major Neurodegenerative Diseases. Int. J. Mol. Sci. 2018, 19, 3082. [Google Scholar] [CrossRef]
- Fricker, M.; Tolkovsky, A.M.; Borutaite, V.; Coleman, M.; Brown, G.C. Neuronal cell death. Physiol. Rev. 2018, 98, 813–880. [Google Scholar] [CrossRef]
- Tang, D.; Kang, R.; Berghe, T.V.; Vandenabeele, P.; Kroemer, G. The molecular machinery of regulated cell death. Cell Res. 2019, 29, 347–364. [Google Scholar] [CrossRef]
- Ghavami, S.; Shojaei, S.; Yeganeh, B.; Ande, S.R.; Jangamreddy, J.R.; Mehrpour, M.; Christoffersson, J.; Chaabane, W.; Moghadam, A.R.; Kashani, H.H.; et al. Autophagy and apoptosis dysfunction in neurodegenerative disorders. Prog. Neurobiol. 2014, 112, 24–49. [Google Scholar] [CrossRef]
- Vanden Berghe, T.; Kaiser, W.J.; Bertrand, M.J.M.; Vandenabeele, P. Molecular crosstalk between apoptosis, necroptosis, and survival signaling. Mol. Cell. Oncol. 2015, 2, e975093. [Google Scholar] [CrossRef]
- Jantas, D.; Lasoń, W. Preclinical evidence for the interplay between oxidative stress and rip1-dependent cell death in neurodegeneration: State of the art and possible therapeutic implications. Antioxidants 2021, 10, 1518. [Google Scholar] [CrossRef]
- Marogianni, C.; Sokratous, M.; Dardiotis, E.; Hadjigeorgiou, G.M.; Bogdanos, D.; Xiromerisiou, G. Neurodegeneration and inflammation—An interesting interplay in parkinson’s disease. Int. J. Mol. Sci. 2020, 21, 8421. [Google Scholar] [CrossRef]
- Tylek, K.; Trojan, E.; Regulska, M.; Lacivita, E.; Leopoldo, M.; Basta-Kaim, A. Formyl peptide receptor 2, as an important target for ligands triggering the inflammatory response regulation: A link to brain pathology. Pharmacol. Rep. 2021, 73, 1004–1019. [Google Scholar] [CrossRef]
- Djajadikerta, A.; Keshri, S.; Pavel, M.; Prestil, R.; Ryan, L.; Rubinsztein, D.C. Autophagy Induction as a Therapeutic Strategy for Neurodegenerative Diseases. J. Mol. Biol. 2020, 432, 2799–2821. [Google Scholar] [CrossRef]
- Guo, F.; Liu, X.; Cai, H.; Le, W. Autophagy in neurodegenerative diseases: Pathogenesis and therapy. Brain Pathol. 2018, 28, 3–13. [Google Scholar] [CrossRef]
- Park, H.; Kam, T.I.; Dawson, T.M.; Dawson, V.L. Poly (ADP-Ribose) (PAR)-Dependent Cell Death in Neurodegenerative Diseases, 1st ed.; Elsevier Inc.: Amsterdam, The Netherlands, 2020; Volume 353, ISBN 9780128201350. [Google Scholar]
- Mo, Z.; Tang, C.; Li, H.; Lei, J.; Zhu, L.; Kou, L.; Li, H.; Luo, S.; Li, C.; Chen, W.; et al. Eicosapentaenoic acid prevents inflammation induced by acute cerebral infarction through inhibition of NLRP3 inflammasome activation. Life Sci. 2020, 242, 117133. [Google Scholar] [CrossRef]
- Acioglu, C.; Li, L.; Elkabes, S. Contribution of astrocytes to neuropathology of neurodegenerative diseases. Brain Res. 2021, 1758, 147291. [Google Scholar] [CrossRef]
- Fakhoury, M. Microglia and astrocytes in Alzheimer’s disease: Implications for therapy. Curr. Neuropharmacol. 2017, 15, 508–518. [Google Scholar] [CrossRef]
- Kam, T.I.; Hinkle, J.T.; Dawson, T.M.; Dawson, V.L. Microglia and astrocyte dysfunction in parkinson’s disease. Neurobiol. Dis. 2020, 144, 105028. [Google Scholar] [CrossRef]
- Kuter, K.Z.; Cenci, M.A.; Carta, A.R. The Role of Glia in Parkinson’s Disease: Emerging Concepts and Therapeutic Applications, 1st ed.; Elsevier B.V.: Amsterdam, The Netherlands, 2020; Volume 252, ISBN 9780444642608. [Google Scholar]
- Tang, W.C.; Hsu, Y.C.; Wang, C.C.; Hu, C.Y.; Chio, C.C.; Kuo, J.R. Early electroacupuncture treatment ameliorates neuroinflammation in rats with traumatic brain injury. BMC Complement. Altern. Mzed. 2016, 16, 470. [Google Scholar] [CrossRef]
- Devos, D.; Hirsch, E.; Wyse, R. Seven Solutions for Neuroprotection in Parkinson’s Disease. Mov. Disord. 2021, 36, 306–316. [Google Scholar] [CrossRef]
- Jayaraj, R.L.; Azimullah, S.; Beiram, R.; Jalal, F.Y.; Rosenberg, G.A. Neuroinflammation: Friend and foe for ischemic stroke. J. Neuroinflammation 2019, 16, 142. [Google Scholar] [CrossRef]
- Kwon, H.S.; Koh, S.H. Neuroinflammation in neurodegenerative disorders: The roles of microglia and astrocytes. Transl. Neurodegener. 2020, 9, 42. [Google Scholar] [CrossRef]
- Lünemann, J.D.; Malhotra, S.; Shinohara, M.L.; Montalban, X.; Comabella, M. Targeting Inflammasomes to Treat Neurological Diseases. Ann. Neurol. 2021, 90, 177–188. [Google Scholar] [CrossRef]
- Shi, Y.; Manis, M.; Long, J.; Wang, K.; Sullivan, P.M.; Serrano, J.R.; Hoyle, R.; Holtzman, D.M. Microglia drive APOE-dependent neurodegeneration in a tauopathy mouse model. J. Exp. Med. 2019, 216, 2546–2561. [Google Scholar] [CrossRef]
- Schimmel, S.; Acosta, S.; Lozano, D. Neuroinflammation in traumatic brain injury: A chronic response to an acute injury. Brain Circ. 2017, 3, 135. [Google Scholar] [CrossRef]
- Trojan, E.; Leśkiewicz, M.; Lacivita, E.; Leopoldo, M.; Basta-Kaim, A. The Formyl Peptide Receptor 2 as a Target for Promotion of Resolution of Inflammation. Curr. Neuropharmacol. 2022, 20, 1–6. [Google Scholar] [CrossRef]
- Boese, A.C.; Hamblin, M.H.; Lee, J.P. Neural stem cell therapy for neurovascular injury in Alzheimer’s disease. Exp. Neurol. 2020, 324, 113112. [Google Scholar] [CrossRef]
- Cai, W.; Zhang, K.; Li, P.; Zhu, L.; Xu, J.; Yang, B.; Hu, X.; Lu, Z.; Chen, J. Dysfunction of the neurovascular unit in ischemic stroke and neurodegenerative diseases: An aging effect. Ageing Res. Rev. 2017, 34, 77–87. [Google Scholar] [CrossRef]
- Iadecola, C. The Neurovascular Unit Coming of Age: A Journey through Neurovascular Coupling in Health and Disease. Neuron 2017, 96, 17–42. [Google Scholar] [CrossRef]
- Lok, J.; Wang, X.S.; Xing, C.H.; Maki, T.K.; Wu, L.M.; Guo, S.Z.; Noviski, N.; Arai, K.; Whalen, M.J.; Lo, E.H.; et al. Targeting the Neurovascular Unit in Brain Trauma. CNS Neurosci. Ther. 2015, 21, 304–308. [Google Scholar] [CrossRef]
- Abbott, N.J.; Rönnbäck, L.; Hansson, E. Astrocyte-endothelial interactions at the blood-brain barrier. Nat. Rev. Neurosci. 2006, 7, 41–53. [Google Scholar] [CrossRef]
- Kim, J.H.; Kim, J.H.; Park, J.A.; Lee, S.-W.; Kim, W.J.; Yu, Y.S.; Kim, K.-W. Blood-neural barrier: Intercellular communication at glio-vascular interface. J. Biochem. Mol. Biol. 2006, 39, 339–345. [Google Scholar] [CrossRef]
- Bell, A.H.; Miller, S.L.; Castillo-Melendez, M.; Malhotra, A. The Neurovascular Unit: Effects of Brain Insults during the Perinatal Period. Front. Neurosci. 2020, 13, 1452. [Google Scholar] [CrossRef]
- De la Torre, J.C. Are Major Dementias Triggered by Poor Blood Flow to the Brain? Theoretical Considerations. J. Alzheimers Dis. 2017, 57, 353–371. [Google Scholar] [CrossRef]
- Parkes, I.; Chintawar, S.; Zameel Cader, M. Neurovascular dysfunction in dementia—Human cellular models and molecular mechanisms. Clin. Sci. 2018, 132, 399–418. [Google Scholar] [CrossRef]
- Lang, F.; Ma, K.; Leibrock, C.B. 1,25(OH)2D3 in Brain Function and Neuropsychiatric Disease. Neurosignals 2019, 27, 40–49. [Google Scholar] [CrossRef]
- Lasoń, W.; Jantas, D.; Leśkiewicz, M.; Regulska, M.; Basta-Kaim, A. Vitamin D3 and Ischemic Stroke: A Narrative Review. Antioxidants 2022, 11, 2120. [Google Scholar] [CrossRef]
- Baas, D.; Prüfer, K.; Ittel, M.E.; Kuchler-Bopp, S.; Labourdette, G.; Sarliève, L.L.; Brachet, P. Rat oligodendrocytes express the vitamin D3 receptor and respond to 1,25-dihydroxyvitamin D3. Glia 2000, 31, 59–68. [Google Scholar] [CrossRef]
- Zorrilla Veloz, R.I.; McKenzie, T.; Palacios, B.E.; Hu, J. Nuclear hormone receptors in demyelinating diseases. J. Neuroendocrinol. 2022, 34, e13171. [Google Scholar] [CrossRef]
- Gáll, Z.; Székely, O. Role of vitamin d in cognitive dysfunction: New molecular concepts and discrepancies between animal and human findings. Nutrients 2021, 13, 3672. [Google Scholar] [CrossRef]
- Guo, Y.-X.; He, L.-Y.; Zhang, M.; Wang, F.; Liu, F.; Peng, W.-X. 1,25-Dihydroxyvitamin D3 regulates expression of LRP1 and RAGE in vitro and in vivo, enhancing Aβ1–40 brain-to-blood efflux and peripheral uptake transport. Neuroscience 2016, 322, 28–38. [Google Scholar] [CrossRef]
- Christakos, S.; Dhawan, P.; Verstuyf, A.; Verlinden, L.; Carmeliet, G. Vitamin D: Metabolism, Molecular Mechanism of Action, and Pleiotropic Effects. Physiol. Rev. 2016, 96, 365–408. [Google Scholar] [CrossRef]
- Yasmin, R.; Williams, R.M.; Xu, M.; Noy, N. Nuclear import of the retinoid X receptor, the vitamin D receptor, and their mutual heterodimer. J. Biol. Chem. 2005, 280, 40152–40160. [Google Scholar] [CrossRef]
- Dos Reis, G.V.O.P.; Gontijo, N.A.; Rodrigues, K.F.; Alves, M.T.; Ferreira, C.N.; Gomes, K.B. Vitamin D receptor polymorphisms and the polycystic ovary syndrome: A systematic review. J. Obstet. Gynaecol. Res. 2017, 43, 436–446. [Google Scholar] [CrossRef]
- Bouillon, R.; Carmeliet, G.; Verlinden, L.; Van Etten, E.; Verstuyf, A.; Luderer, H.F.; Lieben, L.; Mathieu, C.; Demay, M. Vitamin D and human health: Lessons from vitamin D receptor null mice. Endocr. Rev. 2008, 29, 726–776. [Google Scholar] [CrossRef]
- Gil, Á.; Plaza-Diaz, J.; Mesa, M.D. Vitamin D: Classic and Novel Actions. Ann. Nutr. Metab. 2018, 72, 87–95. [Google Scholar] [CrossRef]
- Wimalawansa, S.J. Vitamin D Deficiency: Effects on Oxidative Stress, Epigenetics, Gene Regulation, and Aging. Biology 2019, 8, 30. [Google Scholar] [CrossRef]
- Prüfer, K.; Veenstra, T.D.; Jirikowski, G.F.; Kumar, R. Distribution of 1,25-dihydroxyvitamin D3 receptor immunoreactivity in the rat brain and spinal cord. J. Chem. Neuroanat. 1999, 16, 135–145. [Google Scholar] [CrossRef]
- Smolders, J.; Schuurman, K.G.; Van Strien, M.E.; Melief, J.; Hendrickx, D.; Hol, E.M.; Van Eden, C.; Luchetti, S.; Huitinga, I. Expression of vitamin D receptor and metabolizing enzymes in multiple sclerosis-affected brain tissue. J. Neuropathol. Exp. Neurol. 2013, 72, 91–105. [Google Scholar] [CrossRef]
- Landel, V.; Stephan, D.; Cui, X.; Eyles, D.; Feron, F. Differential expression of vitamin D-associated enzymes and receptors in brain cell subtypes. J. Steroid Biochem. Mol. Biol. 2018, 177, 129–134. [Google Scholar] [CrossRef]
- Stumpf, W.E.; O’Brien, L.P. 1,25(OH)2 vitamin D3 sites of action in the brain. Histochemistry 1987, 87, 393–406. [Google Scholar] [CrossRef]
- Taniura, H.; Ito, M.; Sanada, N.; Kuramoto, N.; Ohno, Y.; Nakamichi, N.; Yoneda, Y. Chronic vitamin D3 treatment protects against neurotoxicity by glutamate in association with upregulation of vitamin D receptor mRNA expression in cultured rat cortical neurons. J. Neurosci. Res. 2006, 83, 1179–1189. [Google Scholar] [CrossRef]
- Eyles, D.W.; Smith, S.; Kinobe, R.; Hewison, M.; McGrath, J.J. Distribution of the Vitamin D receptor and 1α-hydroxylase in human brain. J. Chem. Neuroanat. 2005, 29, 21–30. [Google Scholar] [CrossRef]
- Féron, F.; Burne, T.H.J.; Brown, J.; Smith, E.; McGrath, J.J.; Mackay-Sim, A.; Eyles, D.W. Developmental Vitamin D3 deficiency alters the adult rat brain. Brain Res. Bull. 2005, 65, 141–148. [Google Scholar] [CrossRef]
- Eyles, D.; Almeras, L.; Benech, P.; Patatian, A.; Mackay-Sim, A.; McGrath, J.; Féron, F. Developmental vitamin D deficiency alters the expression of genes encoding mitochondrial, cytoskeletal and synaptic proteins in the adult rat brain. J. Steroid Biochem. Mol. Biol. 2007, 103, 538–545. [Google Scholar] [CrossRef]
- Karnopp, T.E.; Freitas, E.C.; Rieger, A.; Chapacais, G.F.; Monticielo, O.A. Higher IgG level correlated with vitamin D receptor in the hippocampus of a pristane-induced lupus model. Clin. Rheumatol. 2022, 41, 1859–1866. [Google Scholar] [CrossRef]
- Jiang, H.; Yang, X.; Wang, Y.; Zhou, C. Vitamin D Protects against Traumatic Brain Injury via Modulating TLR4/MyD88/NF-κB Pathway-Mediated Microglial Polarization and Neuroinflammation. Biomed Res. Int. 2022, 2022, 3363036. [Google Scholar] [CrossRef]
- Squier, T.C.E.P. Wohlfarth-Handbook of Magnetic Materials, Volume Volume 1-North Holland (1986).pdf; North Holland Publishing: Amsterdam, The Netherlands, 2001; Volume 36, pp. 1539–1550. [Google Scholar]
- Schmauck-Medina, T.; Molière, A.; Lautrup, S.; Zhang, J.; Chlopicki, S.; Madsen, H.B.; Cao, S.; Soendenbroe, C.; Mansell, E.; Vestergaard, M.B.; et al. New hallmarks of ageing: A 2022 Copenhagen ageing meeting summary. Aging 2022, 14, 6829–6839. [Google Scholar] [CrossRef]
- Molinari, C.; Morsanuto, V.; Ghirlanda, S.; Ruga, S.; Notte, F.; Gaetano, L.; Uberti, F. Role of combined lipoic acid and vitamin D3 on astrocytes as a way to prevent brain ageing by induced oxidative stress and iron accumulation. Oxid. Med. Cell. Longev. 2019, 2019, 2843121. [Google Scholar] [CrossRef]
- Berridge, M.J. Vitamin D deficiency accelerates ageing and age-related diseases: A novel hypothesis. J. Physiol. 2017, 595, 6825–6836. [Google Scholar] [CrossRef]
- Tsoukalas, D.; Buga, A.M.; Docea, A.O.; Sarandi, E.; Mitrut, R.; Renieri, E.; Spandidos, D.A.; Rogoveanu, I.; Cercelaru, L.; Niculescu, M.; et al. Reversal of brain aging by targeting telomerase: A nutraceutical approach. Int. J. Mol. Med. 2021, 48, 199. [Google Scholar] [CrossRef]
- Gonzalez-Suarez, I.; Redwood, A.B.; Grotsky, D.A.; Neumann, M.A.; Cheng, E.H.Y.; Stewart, C.L.; Dusso, A.; Gonzalo, S. A new pathway that regulates 53BP1 stability implicates Cathepsin L and vitamin D in DNA repair. EMBO J. 2011, 30, 3383–3396. [Google Scholar] [CrossRef]
- Ashapkin, V.V.; Kutueva, L.I.; Kurchashova, S.Y.; Kireev, I.I. Are there common mechanisms between the Hutchinson-Gilford progeria syndrome and natural aging? Front. Genet. 2019, 10, 455. [Google Scholar] [CrossRef]
- Berridge, M.J. Vitamin D, reactive oxygen species and calcium signalling in ageing and disease. Philos. Trans. R. Soc. B Biol. Sci. 2016, 371, 20150434. [Google Scholar] [CrossRef]
- Drmaku-Sopjani, M.; Kurti, F.; Xuan, N.T.; Sopjani, M. Klotho-Dependent role of 1,25(oh)2d3 in the brain. NeuroSignals 2021, 29, 14–23. [Google Scholar] [CrossRef]
- Tuohimaa, P. Vitamin D and aging. J. Steroid Biochem. Mol. Biol. 2009, 114, 78–84. [Google Scholar] [CrossRef]
- Keisala, T.; Minasyan, A.; Lou, Y.R.; Zou, J.; Kalueff, A.V.; Pyykkö, I.; Tuohimaa, P. Premature aging in vitamin D receptor mutant mice. J. Steroid Biochem. Mol. Biol. 2009, 115, 91–97. [Google Scholar] [CrossRef]
- Messing, J.; Heuberger, R.; Schisa, J. Effect of Vitamin D3 on Lifespan in Caenorhabditis elegans. Curr. Aging Sci. 2014, 6, 220–224. [Google Scholar] [CrossRef]
- Mark, K.A.; Dumas, K.J.; Bhaumik, D.; Schilling, B.; Davis, S.; Oron, T.R.; Sorensen, D.J.; Lucanic, M.; Brem, R.B.; Melov, S.; et al. Vitamin D Promotes Protein Homeostasis and Longevity via the Stress Response Pathway Genes skn-1, ire-1, and xbp-1. Cell Rep. 2016, 17, 1227–1237. [Google Scholar] [CrossRef]
- Lee, V.; Rekhi, E.; Hoh Kam, J.; Jeffery, G. Vitamin D rejuvenates aging eyes by reducing inflammation, clearing amyloid beta and improving visual function. Neurobiol. Aging 2012, 33, 2382–2389. [Google Scholar] [CrossRef]
- Lazzara, F.; Conti, F.; Platania, C.B.M.; Eandi, C.M.; Drago, F.; Bucolo, C. Effects of Vitamin D3 and Meso-Zeaxanthin on Human Retinal Pigmented Epithelial Cells in Three Integrated in vitro Paradigms of Age-Related Macular Degeneration. Front. Pharmacol. 2021, 12, 2849. [Google Scholar] [CrossRef]
- Ashok, A.; Singh, N.; Chaudhary, S.; Bellamkonda, V.; Kritikos, A.E.; Wise, A.S.; Rana, N.; McDonald, D.; Ayyagari, R. Retinal degeneration and alzheimer’s disease: An evolving link. Int. J. Mol. Sci. 2020, 21, 7290. [Google Scholar] [CrossRef]
- Moore, M.E.; Piazza, A.; McCartney, Y.; Lynch, M.A. Evidence that vitamin D3 reverses age-related inflammatory changes in the rat hippocampus. Biochem. Soc. Trans. 2005, 33, 573–577. [Google Scholar] [CrossRef]
- Peng, H.B.; Bukuroshi, P.; Durk, M.R.; Grootendorst, P.; Yan, X.; Pan, S.R.; de Lannoy, I.A.M.; Pang, K.S. Impact of age, hypercholesterolemia, and the vitamin D receptor on brain endogenous β-amyloid peptide accumulation in mice. Biopharm. Drug Dispos. 2021, 42, 372–388. [Google Scholar] [CrossRef]
- Khairy, E.Y.; Attia, M.M. Protective effects of vitamin D on neurophysiologic alterations in brain aging: Role of brain-derived neurotrophic factor (BDNF). Nutr. Neurosci. 2021, 24, 650–659. [Google Scholar] [CrossRef]
- Gómez-Oliva, R.; Geribaldi-Doldán, N.; Domínguez-García, S.; Carrascal, L.; Verástegui, C.; Nunez-Abades, P.; Castro, C. Vitamin D deficiency as a potential risk factor for accelerated aging, impaired hippocampal neurogenesis and cognitive decline: A role for Wnt/β-catenin signaling. Aging 2020, 12, 13824–13844. [Google Scholar] [CrossRef]
- Durk, M.R.; Han, K.; Chow, E.C.Y.; Ahrens, R.; Henderson, J.T.; Fraser, P.E.; Pang, K.S. 1α, 25-dihydroxyvitamin D3 reduces cerebral Amyloid-β accumulation and improves cognition in mouse models of Alzheimer’s disease. J. Neurosci. 2014, 34, 7091–7101. [Google Scholar] [CrossRef]
- Pierucci, F.; Garcia-Gil, M.; Frati, A.; Bini, F.; Martinesi, M.; Vannini, E.; Mainardi, M.; Luzzati, F.; Peretto, P.; Caleo, M.; et al. Vitamin D3 protects against Aβ peptide cytotoxicity in differentiated human neuroblastoma SH- SY5Y cells: A role for S1P1/p38MAPK/ATF4 axis. Neuropharmacology 2017, 116, 328–342. [Google Scholar] [CrossRef]
- Lin, C.-I.; Chang, Y.; Kao, N.; Lee, W.; Cross, T.; Lin, S. 1,25(OH)2D3 Alleviates Aβ(25–35)-Induced Tau Hyperphosphorylation, Excessive Reactive Oxygen Species, and Apoptosis through Interplay with Glial Cell Line-Derived Neurotrophic Factor Signaling in SH-SY5Y Cells. Int. J. Mol. Sci. 2020, 21, 4215. [Google Scholar] [CrossRef]
- Alamro, A.A.; Alsulami, E.A.; Almutlaq, M.; Alghamedi, A.; Alokail, M.; Haq, S.H. Therapeutic Potential of Vitamin D and Curcumin in an In Vitro Model of Alzheimer Disease. J. Cent. Nerv. Syst. Dis. 2020, 12. [Google Scholar] [CrossRef]
- Bao, Z.; Wang, X.; Li, Y.; Feng, F. Vitamin D Alleviates Cognitive Dysfunction by Activating the VDR/ERK1/2 Signaling Pathway in an Alzheimer’s Disease Mouse Model. Neuroimmunomodulation 2021, 27, 178–185. [Google Scholar] [CrossRef]
- Yu, J.; Gattoni-Celli, M.; Zhu, H.; Bhat, N.R.; Sambamurti, K.; Gattoni-Celli, S.; Kindy, M.S. Vitamin D3-Enriched Diet Correlates with a Decrease of Amyloid Plaques in the Brain of AβPP Transgenic Mice. J. Alzheimer’s Dis. 2011, 25, 295–307. [Google Scholar] [CrossRef]
- Bennett, L.; Kersaitis, C.; Macaulay, S.L.; Münch, G.; Niedermayer, G.; Nigro, J.; Payne, M.; Sheean, P.; Vallotton, P.; Zabaras, D.; et al. Vitamin D2-Enriched Button Mushroom (Agaricus bisporus) Improves Memory in Both Wild Type and APPswe/PS1dE9 Transgenic Mice. PLoS ONE 2013, 8, e76362. [Google Scholar] [CrossRef]
- Ali, A.; Shah, S.A.; Zaman, N.; Uddin, M.N.; Khan, W.; Ali, A.; Riaz, M.; Kamil, A. Vitamin D exerts neuroprotection via SIRT1/nrf-2/ NF-kB signaling pathways against D-galactose-induced memory impairment in adult mice. Neurochem. Int. 2021, 142, 104893. [Google Scholar] [CrossRef]
- Yamini, P.; Ray, R.S.; Chopra, K. Vitamin D3 attenuates cognitive deficits and neuroinflammatory responses in ICV-STZ induced sporadic Alzheimer’s disease. Inflammopharmacology 2018, 26, 39–55. [Google Scholar] [CrossRef]
- Saad El-Din, S.; Rashed, L.; Medhat, E.; Emad Aboulhoda, B.; Desoky Badawy, A.; Mohammed ShamsEldeen, A.; Abdelgwad, M. Active form of vitamin D analogue mitigates neurodegenerative changes in Alzheimer’s disease in rats by targeting Keap1/Nrf2 and MAPK-38p/ERK signaling pathways. Steroids 2020, 156, 108586. [Google Scholar] [CrossRef]
- Wu, T.Y.; Zhao, L.X.; Zhang, Y.H.; Fan, Y.G. Activation of vitamin D receptor inhibits Tau phosphorylation is associated with reduction of iron accumulation in APP/PS1 transgenic mice. Neurochem. Int. 2022, 153, 105260. [Google Scholar] [CrossRef]
- Li, L.; Li, W.J.; Zheng, X.R.; Liu, Q.L.; Du, Q.; Lai, Y.J.; Liu, S.Q. Eriodictyol ameliorates cognitive dysfunction in APP/PS1 mice by inhibiting ferroptosis via vitamin D receptor-mediated Nrf2 activation. Mol. Med. 2022, 28, 11. [Google Scholar] [CrossRef]
- Gombash, S.E.; Lee, P.W.; Sawdai, E.; Lovett-Racke, A.E. Vitamin D as a Risk Factor for Multiple Sclerosis: Immunoregulatory or Neuroprotective? Front. Neurol. 2022, 13, 895. [Google Scholar] [CrossRef]
- Zhang, D.; Li, M.; Dong, Y.; Zhang, X.; Liu, X.; Chen, Z.; Zhu, Y.; Wang, H.; Liu, X.; Zhu, J.; et al. 1α,25-Dihydroxyvitamin D3 up-regulates IL-34 expression in SH-SY5Y neural cells. Innate Immun. 2017, 23, 584–591. [Google Scholar] [CrossRef]
- Raha, S.; Lee, H.J.; Yumnam, S.; Hong, G.E.; Venkatarame Gowda Saralamma, V.; Ha, Y.L.; Kim, J.O.; Kim, Y.S.; Heo, J.D.; Lee, S.J.; et al. Vitamin D2 suppresses amyloid-β 25–35 induced microglial activation in BV2 cells by blocking the NF-κB inflammatory signaling pathway. Life Sci. 2016, 161, 37–44. [Google Scholar] [CrossRef]
- Mehrabadi, S.; Sadr, S.S. Administration of Vitamin D3 and E supplements reduces neuronal loss and oxidative stress in a model of rats with Alzheimer’s disease. Neurol. Res. 2020, 42, 862–868. [Google Scholar] [CrossRef]
- Mehri, N.; Haddadi, R.; Ganji, M.; Shahidi, S.; Soleimani Asl, S.; Taheri Azandariani, M.; Ranjbar, A. Effects of vitamin D in an animal model of Alzheimer’s disease: Behavioral assessment with biochemical investigation of Hippocampus and serum. Metab. Brain Dis. 2020, 35, 263–274. [Google Scholar] [CrossRef]
- Grimm, M.O.W.; Thiel, A.; Lauer, A.A.; Winkler, J.; Lehmann, J.; Regner, L.; Nelke, C.; Janitschke, D.; Benoist, C.; Streidenberger, O.; et al. Vitamin D and its analogues decrease amyloid-β (Aβ) formation and increase Aβ-degradation. Int. J. Mol. Sci. 2017, 18, 2764. [Google Scholar] [CrossRef]
- Landel, V.; Millet, P.; Baranger, K.; Loriod, B.; Féron, F. Vitamin D interacts with Esr1 and Igf1 to regulate molecular pathways relevant to Alzheimer’s disease. Mol. Neurodegener. 2016, 11, 22. [Google Scholar] [CrossRef]
- Kang, J.; Park, M.; Lee, E.; Jung, J.; Kim, T. The Role of Vitamin D in Alzheimer’s Disease: A Transcriptional Regulator of Amyloidopathy and Gliopathy. Biomedicines 2022, 10, 1824. [Google Scholar] [CrossRef]
- Patel, P.; Shah, J. Role of Vitamin D in Amyloid clearance via LRP-1 upregulation in Alzheimer’s disease: A potential therapeutic target? J. Chem. Neuroanat. 2017, 85, 36–42. [Google Scholar] [CrossRef]
- Fan, Y.G.; Guo, T.; Han, X.R.; Liu, J.L.; Cai, Y.T.; Xue, H.; Huang, X.S.; Li, Y.C.; Wang, Z.Y.; Guo, C. Paricalcitol accelerates BACE1 lysosomal degradation and inhibits calpain-1 dependent neuronal loss in APP/PS1 transgenic mice. EBioMedicine 2019, 45, 393–407. [Google Scholar] [CrossRef]
- Medhat, E.; Rashed, L.; Abdelgwad, M.; Aboulhoda, B.E.; Khalifa, M.M.; El-Din, S.S. Exercise enhances the effectiveness of vitamin D therapy in rats with Alzheimer’s disease: Emphasis on oxidative stress and inflammation. Metab. Brain Dis. 2020, 35, 111–120. [Google Scholar] [CrossRef]
- Bakhtiari-Dovvombaygi, H.; Izadi, S.; Zare, M.; Asgari Hassanlouei, E.; Dinpanah, H.; Ahmadi-Soleimani, S.M.; Beheshti, F. Vitamin D3 administration prevents memory deficit and alteration of biochemical parameters induced by unpredictable chronic mild stress in rats. Sci. Rep. 2021, 11, 16271. [Google Scholar] [CrossRef]
- Katusic, Z.S.; Austin, S.A. Endothelial nitric oxide: Protector of a healthy mind. Eur. Heart J. 2014, 35, 888–894. [Google Scholar] [CrossRef]
- Kim, H.; Shin, J.Y.; Lee, Y.S.; Yun, S.P.; Maeng, H.J.; Lee, Y. Brain endothelial p-glycoprotein level is reduced in parkinson’s disease via a vitamin d receptor-dependent pathway. Int. J. Mol. Sci. 2020, 21, 8538. [Google Scholar] [CrossRef]
- Uberti, F.; Lattuada, D.; Morsanuto, V.; Nava, U.; Bolis, G.; Vacca, G.; Squarzanti, D.F.; Cisari, C.; Molinari, C. Vitamin D protects human endothelial cells from oxidative stress through the autophagic and survival pathways. J. Clin. Endocrinol. Metab. 2014, 99, 1367–1374. [Google Scholar] [CrossRef]
- Wang, J.; Guo, M.N.; Liu, Z.Z.; Ma, S.F.; Liu, W.J.; Qian, J.J.; Zhang, W.N. PGC-1α reduces Amyloid-β deposition in Alzheimer’s disease: Effect of increased VDR expression. Neurosci. Lett. 2021, 744, 135598. [Google Scholar] [CrossRef]
- Lai, R.H.; Hsu, Y.Y.; Shie, F.S.; Huang, C.C.; Chen, M.H.; Juang, J.L. Non-genomic rewiring of vitamin D receptor to p53 as a key to Alzheimer’s disease. Aging Cell 2021, 20, e13509. [Google Scholar] [CrossRef]
- Gombart, A.F.; Michels, A.J.; Eggersdorfer, M. There is no evidence that vitamin D supplementation drives the progression of Alzheimer’s disease. Aging Cell 2022, e13758. [Google Scholar] [CrossRef]
- Vieth, R. Mistakes in terminology cause false conclusions: Vitamin D does not increase the risk of dementia. Aging Cell 2022, 21, e13722. [Google Scholar] [CrossRef]
- Lai, R.H.; Hsu, C.C.; Yu, B.H.; Lo, Y.R.; Hsu, Y.Y.; Chen, M.H.; Juang, J.L. Vitamin D supplementation worsens Alzheimer’s progression: Animal model and human cohort studies. Aging Cell 2022, 21, e13670. [Google Scholar] [CrossRef]
- Holick, M.F.; Binkley, N.C.; Bischoff-Ferrari, H.A.; Gordon, C.M.; Hanley, D.A.; Heaney, R.P.; Murad, M.H.; Weaver, C.M. Evaluation, treatment, and prevention of vitamin D deficiency: An endocrine society clinical practice guideline. J. Clin. Endocrinol. Metab. 2011, 96, 1911–1930. [Google Scholar] [CrossRef]
- Barichella, M.; Garrì, F.; Caronni, S.; Bolliri, C.; Zocchi, L.; Macchione, M.C.; Ferri, V.; Calandrella, D.; Pezzoli, G. Vitamin D Status and Parkinson’s Disease. Brain Sci. 2022, 12, 790. [Google Scholar] [CrossRef]
- Giustina, A.; Adler, R.A.; Binkley, N.; Bollerslev, J.; Bouillon, R.; Dawson-Hughes, B.; Ebeling, P.R.; Feldman, D.; Formenti, A.M.; Lazaretti-Castro, M.; et al. Consensus statement from 2nd International Conference on Controversies in Vitamin D. Rev. Endocr. Metab. Disord. 2020, 21, 89–116. [Google Scholar] [CrossRef]
- Shah, I.; Petroczi, A.; Tabet, N.; Klugman, A.; Isaac, M.; Naughton, D.P. Low 25OH Vitamin D2 Levels Found in Untreated Alzheimer’s Patients, Compared to Acetylcholinesterase-Inhibitor Treated and Controls. Curr. Alzheimer Res. 2012, 9, 1069–1076. [Google Scholar] [CrossRef]
- Shah, I.; Petroczi, A.; Naughton, D.P. Exploring the role of Vitamin D in type 1 diabetes, rheumatoid arthritis, and alzheimer disease: New insights from accurate analysis of 10 forms. J. Clin. Endocrinol. Metab. 2014, 99, 808–816. [Google Scholar] [CrossRef]
- Soni, M.; Kos, K.; Lang, I.A.; Jones, K.; Melzer, D.; Llewellyn, D.J. Vitamin D and cognitive function. Scand. J. Clin. Lab. Invest. 2012, 72, 79–82. [Google Scholar] [CrossRef]
- Lee, D.M.; Tajar, A.; Ulubaev, A.; Pendleton, N.; O’Neill, T.W.; O’Connor, D.B.; Bartfai, G.; Boonen, S.; Bouillon, R.; Casanueva, F.F.; et al. Association between 25-hydroxyvitamin D levels and cognitive performance in middle-aged and older European men. J. Neurol. Neurosurg. Psychiatry 2009, 80, 722–729. [Google Scholar] [CrossRef]
- Littlejohns, T.J.; Henley, W.E.; Lang, I.A.; Annweiler, C.; Beauchet, O.; Chaves, P.H.M.; Fried, L.; Kestenbaum, B.R.; Kuller, L.H.; Langa, K.M.; et al. Vitamin D and the risk of dementia and Alzheimer disease. Neurology 2014, 83, 920–928. [Google Scholar] [CrossRef]
- Aspell, N.; Lawlor, B.; O’Sullivan, M. Is there a role for Vitamin D in supporting cognitive function as we age? Proc. Nutr. Soc. 2018, 77, 124–134. [Google Scholar] [CrossRef]
- Jayedi, A.; Rashidy-Pour, A.; Shab-Bidar, S. Vitamin D status and risk of dementia and Alzheimer’s disease: A meta-analysis of dose-response †. Nutr. Neurosci. 2019, 22, 750–759. [Google Scholar] [CrossRef]
- Chai, B.; Gao, F.; Wu, R.; Dong, T.; Gu, C.; Lin, Q.; Zhang, Y. Vitamin D deficiency as a risk factor for dementia and Alzheimer’s disease: An updated meta-analysis. BMC Neurol. 2019, 19, 284. [Google Scholar] [CrossRef]
- Overman, M.J.; Pendleton, N.; O’Neill, T.W.; Bartfai, G.; Casanueva, F.F.; Finn, J.D.; Forti, G.; Rastrelli, G.; Giwercman, A.; Han, T.S.; et al. Evaluation of cognitive subdomains, 25-hydroxyvitamin D, and 1,25-dihydroxyvitamin D in the European Male Ageing Study. Eur. J. Nutr. 2017, 56, 2093–2103. [Google Scholar] [CrossRef]
- Olsson, E.; Byberg, L.; Karlström, B.; Cederholm, T.; Melhus, H.; Sjögren, P.; Kilander, L. Vitamin D is not associated with incident dementia or cognitive impairment: An 18-y follow-up study in community-living old men. Am. J. Clin. Nutr. 2017, 105, 936–943. [Google Scholar] [CrossRef]
- Yang, K.; Chen, J.; Li, X.; Zhou, Y. Vitamin D concentration and risk of Alzheimer disease. Medicine 2019, 98, e16804. [Google Scholar] [CrossRef]
- Panza, F.; La Montagna, M.; Lampignano, L.; Zupo, R.; Bortone, I.; Castellana, F.; Sardone, R.; Borraccino, L.; Dibello, V.; Resta, E.; et al. Vitamin D in the development and progression of alzheimer’s disease: Implications for clinical management. Expert Rev. Neurother. 2021, 21, 287–301. [Google Scholar] [CrossRef]
- Shea, M.K.; Fu, X.; Barger, K.; James, B.D.; Dawson-hughes, B.; Leurgans, S.E.; Holland, T.M.; Agarwal, P.; Heger, N.E.; Schneider, J.A.; et al. Brain vitamin D forms, cognitive decline, and neuropathology in community-dwelling older adults. Alzheimer’s Dement. 2022, 1–8. [Google Scholar] [CrossRef]
- Annweiler, C.; Fantino, B.; Parot-Schinkel, E.; Thiery, S.; Gautier, J.; Beauchet, O. Alzheimer’s disease—Input of vitamin D with mEmantine assay (AD-IDEA trial): Study protocol for a randomized controlled trial. Trials 2011, 12, 230. [Google Scholar] [CrossRef]
- Lu’O’Ng, K.V.Q.; Nguyên, L.T.H. The beneficial role of vitamin D in Alzheimer’s disease. Am. J. Alzheimers. Dis. Other Demen. 2011, 26, 511–520. [Google Scholar] [CrossRef]
- Stein, M.S.; Scherer, S.C.; Ladd, K.S.; Harrison, L.C. A randomized controlled trial of high-dose vitamin D2 followed by intranasal insulin in Alzheimer’s disease. J. Alzheimer’s Dis. 2011, 26, 477–484. [Google Scholar] [CrossRef]
- Jia, J.; Hu, J.; Huo, X.; Miao, R.; Zhang, Y.; Ma, F. Effects of Vitamin D supplementation on cognitive function and blood Aβ-related biomarkers in older adults with Alzheimer’s disease: A randomised, double-blind, placebo-controlled trial. J. Neurol. Neurosurg. Psychiatry 2019, 90, 1347–1352. [Google Scholar] [CrossRef]
- Marazziti, D.; Parra, E.; Palermo, S.; Barberi, F.M.; Buccianelli, B.; Ricciardulli, S.; Cappelli, A.; Mucci, F.; Dell’Osso, L. Vitamin D: A Pleiotropic Hormone with Possible Psychotropic Activities. Curr. Med. Chem. 2021, 28, 3843–3864. [Google Scholar] [CrossRef]
- SanMartín, C.D.; Henriquez, M.; Chacon, C.; Ponce, D.P.; Salech, F.; Rogers, N.K.; Behrens, M.I. Vitamin D Increases Aβ140 Plasma Levels and Protects Lymphocytes from Oxidative Death in Mild Cognitive Impairment Patients. Curr. Alzheimer Res. 2017, 15, 561–569. [Google Scholar] [CrossRef]
- Buell, J.S.; Weiner, D.E.; Tucker, L.; Usda, J.M. Cerebrovascular Pathology in Elders Receiving Home Services. Am. Acad. Neurol. 2010, 74, 18–26. [Google Scholar]
- Afzal, S.; Bojesen, S.E.; Nordestgaard, B.G. Reduced 25-hydroxyvitamin D and risk of Alzheimer’s disease and vascular dementia. Alzheimer’s Dement. 2014, 10, 296–302. [Google Scholar] [CrossRef]
- Gezen-Ak, D.; Dursun, E.; Ertan, T.; Hanaǧasi, H.; Gürvit, H.; Emre, M.; Eker, E.; Öztürk, M.; Engin, F.; Yilmazer, S. Association between vitamin D receptor gene polymorphism and Alzheimer’s disease. Tohoku J. Exp. Med. 2007, 212, 275–282. [Google Scholar] [CrossRef]
- Liu, H.; He, Y.; Beck, J.; Silva Teixeira, S.; Harrison, K.; Xu, Y.; Sisley, S. Defining vitamin D receptor expression in the brain using a novel <scp> VDR Cre </scp> mouse. J. Comp. Neurol. 2021, 529, 2362–2375. [Google Scholar] [CrossRef]
- Łaczmański, Ł.; Jakubik, M.; Bednarek-Tupikowska, G.; Rymaszewska, J.; Słoka, N.; Lwow, F. Vitamin D receptor gene polymorphisms in Alzheimer’s disease patients. Exp. Gerontol. 2015, 69, 142–147. [Google Scholar] [CrossRef]
- Dimitrakis, E.; Katsarou, M.-S.; Lagiou, M.; Papastefanopoulou, V.; Spandidos, D.; Tsatsakis, A.; Papageorgiou, S.; Moutsatsou, P.; Antoniou, K.; Kroupis, C.; et al. Association of vitamin D receptor gene haplotypes with late-onset Alzheimer’s disease in a Southeastern European Caucasian population. Exp. Ther. Med. 2022, 24, 584. [Google Scholar] [CrossRef]
- Zhang, K.; Ma, X.; Zhang, R.; Liu, Z.; Jiang, L.; Qin, Y.; Zhang, D.; Tian, P.; Gao, Z.Y.; Zhang, N.; et al. Crosstalk between Gut Microflora and Vitamin D Receptor SNPs Are Associated with the Risk of Amnestic Mild Cognitive Impairment in a Chinese Elderly Population. J. Alzheimer’s Dis. 2022, 88, 357–373. [Google Scholar] [CrossRef]
- Soares, J.Z.; Valeur, J.; Benth, J.Š.; Knapskog, A.B.; Selbæk, G.; Arefi, G.; Gilfillan, G.D.; Tollisen, A.; Bogdanovic, N.; Pettersen, R. Vitamin D in Alzheimer’s Disease: Low Levels in Cerebrospinal Fluid Despite Normal Amounts in Serum. J. Alzheimer’s Dis. 2022, 86, 1301–1314. [Google Scholar] [CrossRef]
- Arévalo, N.B.; Castillo-Godoy, D.P.; Espinoza-Fuenzalida, I.; Rogers, N.K.; Farias, G.; Delgado, C.; Henriquez, M.; Herrera, L.; Behrens, M.I.; Sanmartín, C.D. Association of Vitamin D Receptor Polymorphisms with Amyloid-β Transporters Expression and Risk of Mild Cognitive Impairment in a Chilean Cohort. J. Alzheimer’s Dis. 2021, 82, S283–S297. [Google Scholar] [CrossRef]
- De Oliveira, A.C.R.; Magalhães, C.A.; Loures, C.M.G.; Fraga, V.G.; de Souza, L.C.; Guimarães, H.C.; Cintra, M.T.G.; Bicalho, M.A.; Sousa, M.C.R.; Silveira, J.N.; et al. Bsmi polymorphism in the vitamin d receptor gene is associated with 25-hydroxy vitamin D levels in individuals with cognitive decline. Arq. Neuropsiquiatr. 2018, 76, 760–766. [Google Scholar] [CrossRef]
- Kalia, L.V.; Lang, A.E. Parkinson’s disease. Lancet 2015, 386, 896–912. [Google Scholar] [CrossRef]
- Airavaara, M.; Parkkinen, I.; Konovalova, J.; Albert, K.; Chmielarz, P.; Domanskyi, A. Back and to the Future: From Neurotoxin-Induced to Human Parkinson’s Disease Models. Curr. Protoc. Neurosci. 2020, 91, e88. [Google Scholar] [CrossRef]
- Ibi, M.; Sawada, H.; Nakanishi, M.; Kume, T.; Katsuki, H.; Kaneko, S.; Shimohama, S.; Akaike, A. Protective effects of 1α,25-(OH)2D3 against the neurotoxicity of glutamate and reactive oxygen species in mesencephalic culture. Neuropharmacology 2001, 40, 761–771. [Google Scholar] [CrossRef]
- Wang, J.Y.; Wu, J.N.; Cherng, T.L.; Hoffer, B.J.; Chen, H.H.; Borlongan, C.V.; Wang, Y. Vitamin D3 attenuates 6-hydroxydopamine-induced neurotoxicity in rats. Brain Res. 2001, 904, 67–75. [Google Scholar] [CrossRef]
- Shinpo, K.; Kikuchi, S.; Sasaki, H.; Moriwaka, F.; Tashiro, K. Effect of 1,25-dihydroxyvitamin D3 on cultured mesencephalic dopaminergic neurons to the combined toxicity caused by L-buthionine sulfoximine and 1-methyl-4-phenylpyridine. J. Neurosci. Res. 2000, 62, 374–382. [Google Scholar] [CrossRef]
- Jang, W.; Park, H.H.; Lee, K.Y.; Lee, Y.J.; Kim, H.T.; Koh, S.H. 1,25-dyhydroxyvitamin D3 Attenuates l-DOPA-Induced Neurotoxicity in Neural Stem Cells. Mol. Neurobiol. 2015, 51, 558–570. [Google Scholar] [CrossRef]
- Jang, W.; Kim, H.J.; Li, H.; Jo, K.D.; Lee, M.K.; Song, S.H.; Yang, H.O. 1,25-Dyhydroxyvitamin D3 attenuates rotenone-induced neurotoxicity in SH-SY5Y cells through induction of autophagy. Biochem. Biophys. Res. Commun. 2014, 451, 142–147. [Google Scholar] [CrossRef]
- Orme, R.P.; Bhangal, M.S.; Fricker, R.A. Calcitriol Imparts Neuroprotection In Vitro to Midbrain Dopaminergic Neurons by Upregulating GDNF Expression. PLoS ONE 2013, 8, e62040. [Google Scholar] [CrossRef]
- Zhang, Y.; Ji, W.; Zhang, S.; Gao, N.; Xu, T.; Wang, X.; Zhang, M. Vitamin D Inhibits the Early Aggregation of α-Synuclein and Modulates Exocytosis Revealed by Electrochemical Measurements. Angew. Chem. —Int. Ed. 2022, 61, e202111853. [Google Scholar] [CrossRef]
- Cass, W.A.; Peters, L.E. Reduced ability of calcitriol to promote augmented dopamine release in the lesioned striatum of aged rats. Neurochem. Int. 2017, 108, 222–229. [Google Scholar] [CrossRef]
- Lima, L.A.R.; Lopes, M.J.P.; Costa, R.O.; Lima, F.A.V.; Neves, K.R.T.; Calou, I.B.F.; Andrade, G.M.; Viana, G.S.B. Vitamin D protects dopaminergic neurons against neuroinflammation and oxidative stress in hemiparkinsonian rats. J. Neuroinflamm. 2018, 15, 249. [Google Scholar] [CrossRef]
- Kim, H.A.; Perrelli, A.; Ragni, A.; Retta, F.; De Silva, T.M.; Sobey, C.G.; Retta, S.F. Vitamin D deficiency and the risk of cerebrovascular disease. Antioxidants 2020, 9, 327. [Google Scholar] [CrossRef]
- Bayo-Olugbami, A.; Nafiu, A.B.; Amin, A.; Ogundele, O.M.; Lee, C.C.; Owoyele, B.V. Vitamin D attenuated 6-OHDA-induced behavioural deficits, dopamine dysmetabolism, oxidative stress, and neuro-inflammation in mice. Nutr. Neurosci. 2022, 25, 823–834. [Google Scholar] [CrossRef]
- Da Costa, R.O.; Gadelha-Filho, C.V.J.; de Aquino, P.E.A.; Lima, L.A.R.; de Lucena, J.D.; Ribeiro, W.L.C.; Lima, F.A.V.; Neves, K.R.T.; de Barros Viana, G.S. Vitamin D (VD3) Intensifies the Effects of Exercise and Prevents Alterations of Behavior, Brain Oxidative Stress, and Neuroinflammation, in Hemiparkinsonian Rats. Neurochem. Res. 2022, 48, 142–160. [Google Scholar] [CrossRef]
- Vinh Quôc Luong, K.; Thi Hoàng Nguyên, L. Vitamin D and Parkinson’s disease. J. Neurosci. Res. 2012, 90, 2227–2236. [Google Scholar] [CrossRef]
- Lien, C.Y.; Lu, C.H.; Chang, C.C.; Chang, W.N. Correlation between hypovitaminosis d and nutritional status with the severity of clinical symptoms and impaired cognitive function in patients with parkinson’s disease. Acta Neurol. 2021, 30, 63–73. [Google Scholar]
- Peterson, A.L. A review of vitamin D and Parkinson’s disease. Maturitas 2014, 78, 40–44. [Google Scholar] [CrossRef]
- Wang, L.; Evatt, M.L.; Maldonado, L.G.; Perry, W.R.; Ritchie, J.C.; Beecham, G.W.; Martin, E.R.; Haines, J.L.; Pericak-Vance, M.A.; Vance, J.M.; et al. Vitamin D from different sources is inversely associated with Parkinson disease. Mov. Disord. 2015, 30, 560–566. [Google Scholar] [CrossRef]
- Peterson, A.L.; Murchison, C.; Zabetian, C.; Leverenz, J.B.; Watson, G.S.; Montine, T.; Carney, N.; Bowman, G.L.; Edwards, K.; Quinn, J.F. Memory, mood, and vitamin d in persons with parkinson’s disease. J. Park. Dis. 2013, 3, 547–555. [Google Scholar] [CrossRef]
- Zhao, X.; Zhang, M.; Li, C.; Jiang, X.; Su, Y.; Zhang, Y. Benefits of Vitamins in the Treatment of Parkinson’s Disease. Oxid. Med. Cell. Longev. 2019, 2019, 9426867. [Google Scholar] [CrossRef]
- Fullard, M.E.; Duda, J.E. A Review of the Relationship between Vitamin D and Parkinson Disease Symptoms. Front. Neurol. 2020, 11, 454. [Google Scholar] [CrossRef]
- Zhang, H.J.; Zhang, J.R.; Mao, C.J.; Li, K.; Wang, F.; Chen, J.; Liu, C.F. Relationship between 25-Hydroxyvitamin D, bone density, and Parkinson’s disease symptoms. Acta Neurol. Scand. 2019, 140, 274–280. [Google Scholar] [CrossRef]
- Rektor, I.; Rektorová, I.; Kubová, D. Vascular parkinsonism—An update. J. Neurol. Sci. 2006, 248, 185–191. [Google Scholar] [CrossRef]
- Narasimhan, M.; Schwartz, R.; Halliday, G. Parkinsonism and cerebrovascular disease. J. Neurol. Sci. 2022, 433, 120011. [Google Scholar] [CrossRef]
- Seals, D.R.; Jablonski, K.L.; Donato, A.J. Aging and vascular endothelial function in humans. Clin. Sci. 2011, 120, 357–375. [Google Scholar] [CrossRef]
- Butler, M.W.; Burt, A.; Edwards, T.L.; Zuchner, S.; Scott, W.K.; Martin, E.R.; Vance, J.M.; Wang, L. Vitamin D Receptor Gene as a Candidate Gene for Parkinson Disease. Ann. Hum. Genet. 2011, 75, 201–210. [Google Scholar] [CrossRef]
- Lv, L.; Tan, X.; Peng, X.; Bai, R.; Xiao, Q.; Zou, T.; Tan, J.; Zhang, H.; Wang, C. The relationships of vitamin D, vitamin D receptor gene polymorphisms, and vitamin D supplementation with Parkinson’s disease. Transl. Neurodegener. 2020, 9, 34. [Google Scholar] [CrossRef]
- Hu, W.; Wang, L.; Chen, B.; Wang, X. Vitamin D receptor rs2228570 polymorphism and Parkinson’s disease risk in a Chinese population. Neurosci. Lett. 2020, 717, 134722. [Google Scholar] [CrossRef]
- Geng, J.; Zhang, J.; Yao, F.; Liu, X.; Liu, J.; Huang, Y. A systematic review and meta-analysis of the associations of vitamin D receptor genetic variants with two types of most common neurodegenerative disorders. Aging Clin. Exp. Res. 2020, 32, 21–27. [Google Scholar] [CrossRef]
- Agliardi, C.; Guerini, F.R.; Zanzottera, M.; Bolognesi, E.; Meloni, M.; Riboldazzi, G.; Zangaglia, R.; Sturchio, A.; Casali, C.; Di Lorenzo, C.; et al. The VDR FokI (rs2228570) polymorphism is involved in Parkinson’s disease. J. Neurol. Sci. 2021, 428, 117606. [Google Scholar] [CrossRef]
- Török, R.; Török, N.; Szalardy, L.; Plangar, I.; Szolnoki, Z.; Somogyvari, F.; Vecsei, L.; Klivenyi, P. Association of vitamin D receptor gene polymorphisms and Parkinson’s disease in Hungarians. Neurosci. Lett. 2013, 551, 70–74. [Google Scholar] [CrossRef]
- Suzuki, M.; Yoshioka, M.; Hashimoto, M.; Murakami, M.; Kawasaki, K.; Noya, M.; Takahashi, D.; Urashima, M. 25-hydroxyvitamin D, vitamin D receptor gene polymorphisms, and severity of Parkinson’s disease. Mov. Disord. 2012, 27, 264–271. [Google Scholar] [CrossRef]
- Tanaka, K.; Miyake, Y.; Fukushima, W.; Kiyohara, C.; Sasaki, S.; Tsuboi, Y.; Oeda, T.; Shimada, H.; Kawamura, N.; Sakae, N.; et al. Vitamin D receptor gene polymorphisms, smoking, and risk of sporadic Parkinson’s disease in Japan. Neurosci. Lett. 2017, 643, 97–102. [Google Scholar] [CrossRef]
- Gatto, N.M.; Paul, K.C.; Sinsheimer, J.S.; Bronstein, J.M.; Bordelon, Y.; Rausch, R.; Ritz, B. Vitamin D receptor gene polymorphisms and cognitive decline in Parkinson’s disease. J. Neurol. Sci. 2016, 370, 100–106. [Google Scholar] [CrossRef]
- Gatto, N.M.; Sinsheimer, J.S.; Cockburn, M.; Escobedo, L.A.; Bordelon, Y.; Ritz, B. Vitamin D receptor gene polymorphisms and Parkinson’s disease in a population with high ultraviolet radiation exposure. J. Neurol. Sci. 2015, 352, 88–93. [Google Scholar] [CrossRef]
- Redenšek, S.; Kristanc, T.; Blagus, T.; Trošt, M.; Dolžan, V. Genetic Variability of the Vitamin D Receptor Affects Susceptibility to Parkinson’s Disease and Dopaminergic Treatment Adverse Events. Front. Aging Neurosci. 2022, 14, 853277. [Google Scholar] [CrossRef]
- Gao, J.; Teng, J.; Liu, Z.; Cai, M.; Xie, A. Association between vitamin D receptor polymorphisms and susceptibility to Parkinson’s disease: An updated meta-analysis. Neurosci. Lett. 2020, 720, 134778. [Google Scholar] [CrossRef]
- Iacopetta, K.; Collins-Praino, L.E.; Buisman-Pijlman, F.T.A.; Liu, J.; Hutchinson, A.D.; Hutchinson, M.R. Are the protective benefits of vitamin D in neurodegenerative disease dependent on route of administration? A systematic review. Nutr. Neurosci. 2020, 23, 251–280. [Google Scholar] [CrossRef]
- Bivona, G.; Gambino, C.M.; Iacolino, G.; Ciaccio, M. Vitamin D and the nervous system. Neurol. Res. 2019, 41, 827–835. [Google Scholar] [CrossRef]
- Morello, M.; Pieri, M.; Zenobi, R.; Talamo, A.; Stephan, D.; Landel, V.; Féron, F.; Millet, P. The Influence of Vitamin D on Neurodegeneration and Neurological Disorders: A Rationale for its Physio-pathological Actions. Curr. Pharm. Des. 2020, 26, 2475–2491. [Google Scholar] [CrossRef]
- Ibe, K.; Nakada, H.; Ohgami, M.; Yamada, T.; Okamoto, S. Design, synthesis, and properties of des-D-ring interphenylene derivatives of 1α,25-Dihydroxyvitamin D3. Eur. J. Med. Chem. 2022, 243, 114795. [Google Scholar] [CrossRef]
- Maestro, M.A.; Seoane, S. The Centennial Collection of VDR Ligands: Metabolites, Analogs, Hybrids and Non-Secosteroidal Ligands. Nutrients 2022, 14, 4927. [Google Scholar] [CrossRef]
- Rcom-H’cheo-Gauthier, A.N.; Meedeniya, A.C.B.; Pountney, D.L. Calcipotriol inhibits α-synuclein aggregation in SH-SY5Y neuroblastoma cells by a Calbindin-D28k-dependent mechanism. J. Neurochem. 2017, 141, 263–274. [Google Scholar] [CrossRef]

| PD Models | Vitamin D Administration | Effects | References |
|---|---|---|---|
| Glutamate, 6-OHDA and MPP+-induced toxicity in rat mesencephalic culture | calcitriol (10–100 nM) | ↑ cell viability | [162] |
| H2O2 and 6-OHDA-induced damage of primary cultures of rat ventral mesencephalon | calcitriol (0.1 n)/on 7DIV/before toxins | ↑ cell viability | [163] |
| BSO and MPP+-induced neurotoxicity in rat cultured mesencephalic neurons | calcitriol (1–100 nM) | ↑ neuron survival and neurite extension, ↓ ROS and gluthatione depletion | [164] |
| L-DOPA-induced neural stem cells (NSCs) injury | calcitriol (10–1000 nM) | ↓ free radicals, ↑ cell viability and proliferation, ↑ prosurvival signaling, including activation of the PI3K pathway, and reducing oxidative stress | [165] |
| Rotenone-induced neurotoxicity in SH-SY5Y cells | calcitriol (2.5–10 μM) | ↓ reactive oxygen species levels, ↑ levels of intracellular signaling proteins associated with cell survival; ↑ levels of autophagy markers (LC3, beclin-1, and AMPK) | [166] |
| Rat primary fetal ventral mesencephalic cultures of dopamine neurons | calcitriol (100 pM-100 nM)/7 days | ↑ numbers of rat primary dopamine neurons ↑ GDNF expression ↓ dopamine neurons apoptosis | [167] |
| α-Syn-induced aggregation in SH-SY5Y cells | α-Syn oligomers +vitamin D (4 μM)/36 h | ↑ cell viability ↓ cytotoxicity of α-syn oligomers ↓ α-Syn aggregation ↓ ROS | [168] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lasoń, W.; Jantas, D.; Leśkiewicz, M.; Regulska, M.; Basta-Kaim, A. The Vitamin D Receptor as a Potential Target for the Treatment of Age-Related Neurodegenerative Diseases Such as Alzheimer’s and Parkinson’s Diseases: A Narrative Review. Cells 2023, 12, 660. https://doi.org/10.3390/cells12040660
Lasoń W, Jantas D, Leśkiewicz M, Regulska M, Basta-Kaim A. The Vitamin D Receptor as a Potential Target for the Treatment of Age-Related Neurodegenerative Diseases Such as Alzheimer’s and Parkinson’s Diseases: A Narrative Review. Cells. 2023; 12(4):660. https://doi.org/10.3390/cells12040660
Chicago/Turabian StyleLasoń, Władysław, Danuta Jantas, Monika Leśkiewicz, Magdalena Regulska, and Agnieszka Basta-Kaim. 2023. "The Vitamin D Receptor as a Potential Target for the Treatment of Age-Related Neurodegenerative Diseases Such as Alzheimer’s and Parkinson’s Diseases: A Narrative Review" Cells 12, no. 4: 660. https://doi.org/10.3390/cells12040660
APA StyleLasoń, W., Jantas, D., Leśkiewicz, M., Regulska, M., & Basta-Kaim, A. (2023). The Vitamin D Receptor as a Potential Target for the Treatment of Age-Related Neurodegenerative Diseases Such as Alzheimer’s and Parkinson’s Diseases: A Narrative Review. Cells, 12(4), 660. https://doi.org/10.3390/cells12040660






