Alteration of Immunoregulatory Patterns and Survival Advantage of Key Cell Types in Food Allergic Children
Abstract
1. Introduction
2. Methods
2.1. Study Population
2.2. Allergens
2.3. Plasma Collection and Isolation and Culture of Peripheral Blood Mononuclear Cells
2.4. Antibodies
2.5. Cell Surface and Intracellular Staining
2.6. Detection of sCD23 and Cytokine Levels
2.7. Statistics
3. Results
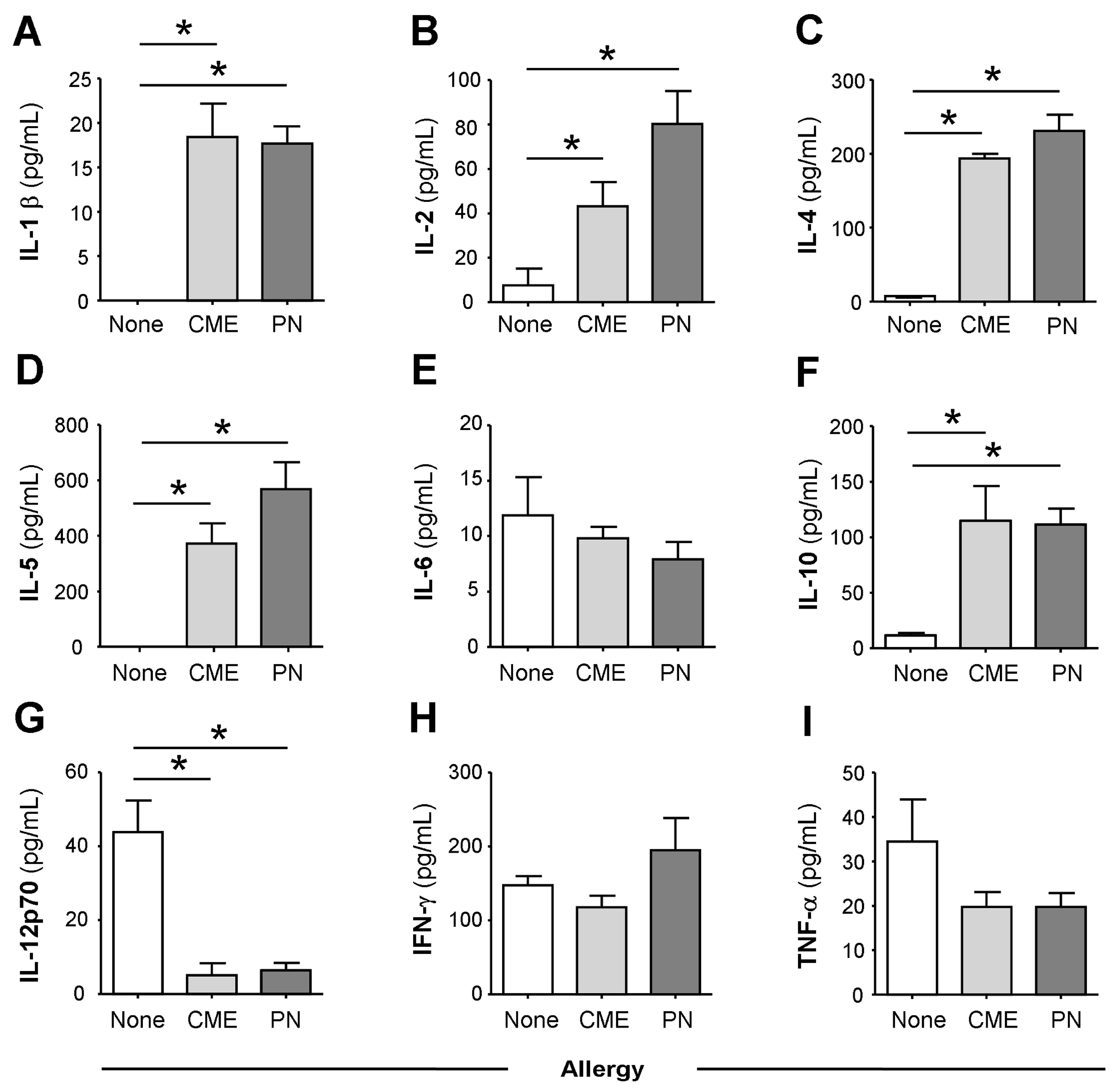
3.1. Constitutive Plasma Cytokines in Food Allergy
3.2. Spontaneous In Vitro sCD23 Release and Cytokine Secretion by MNC in Food Allergy
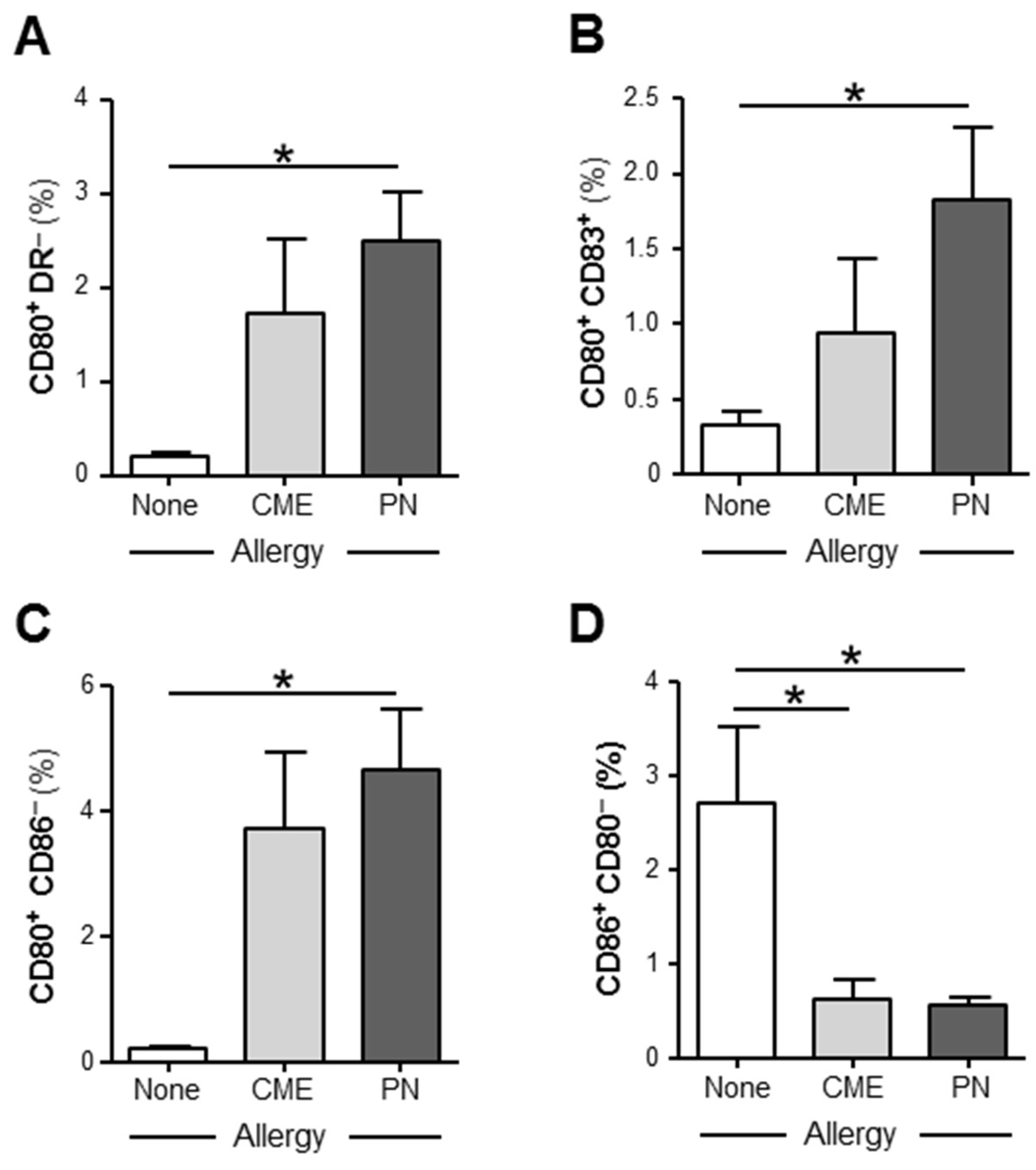
3.3. Allergy-Associated Abnormalities in the Expression of Costimulatory Molecules
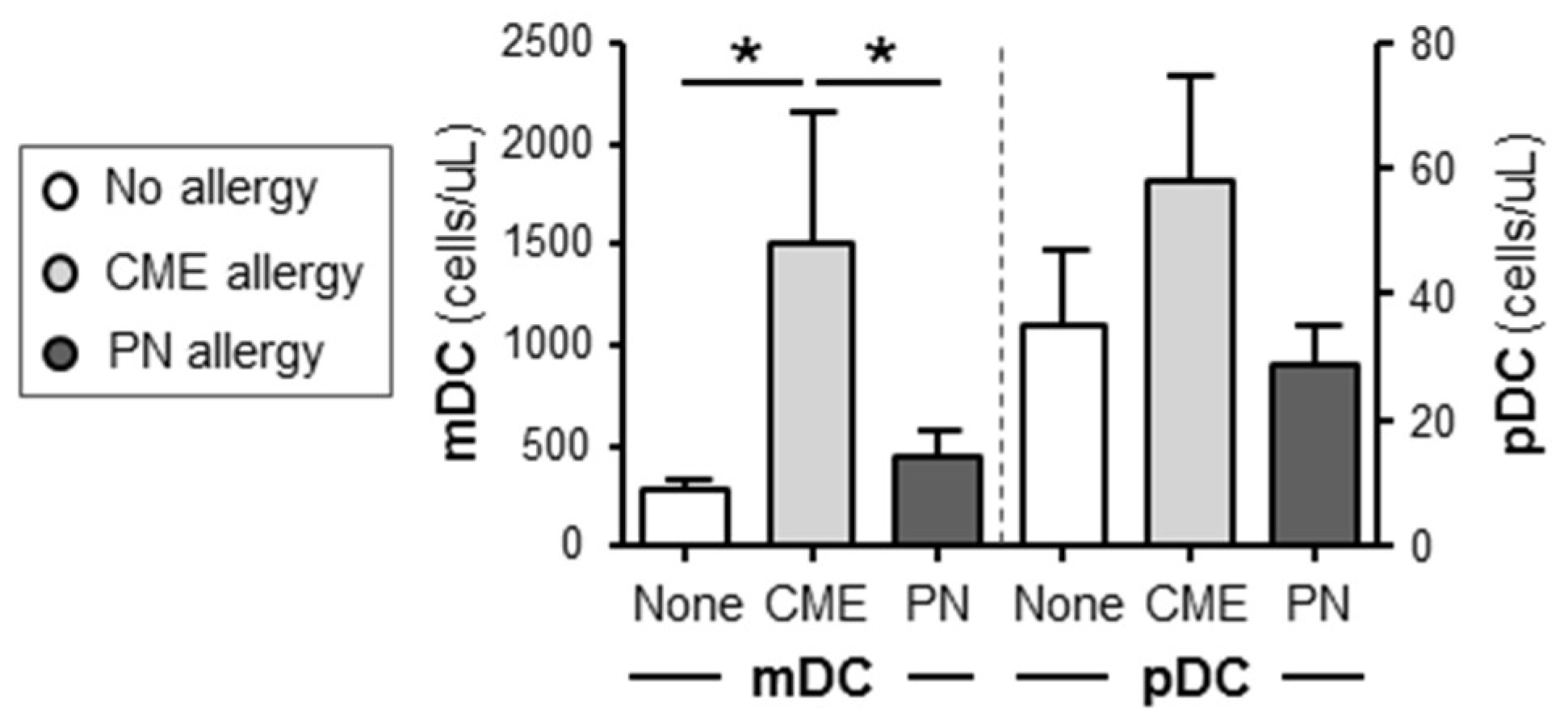
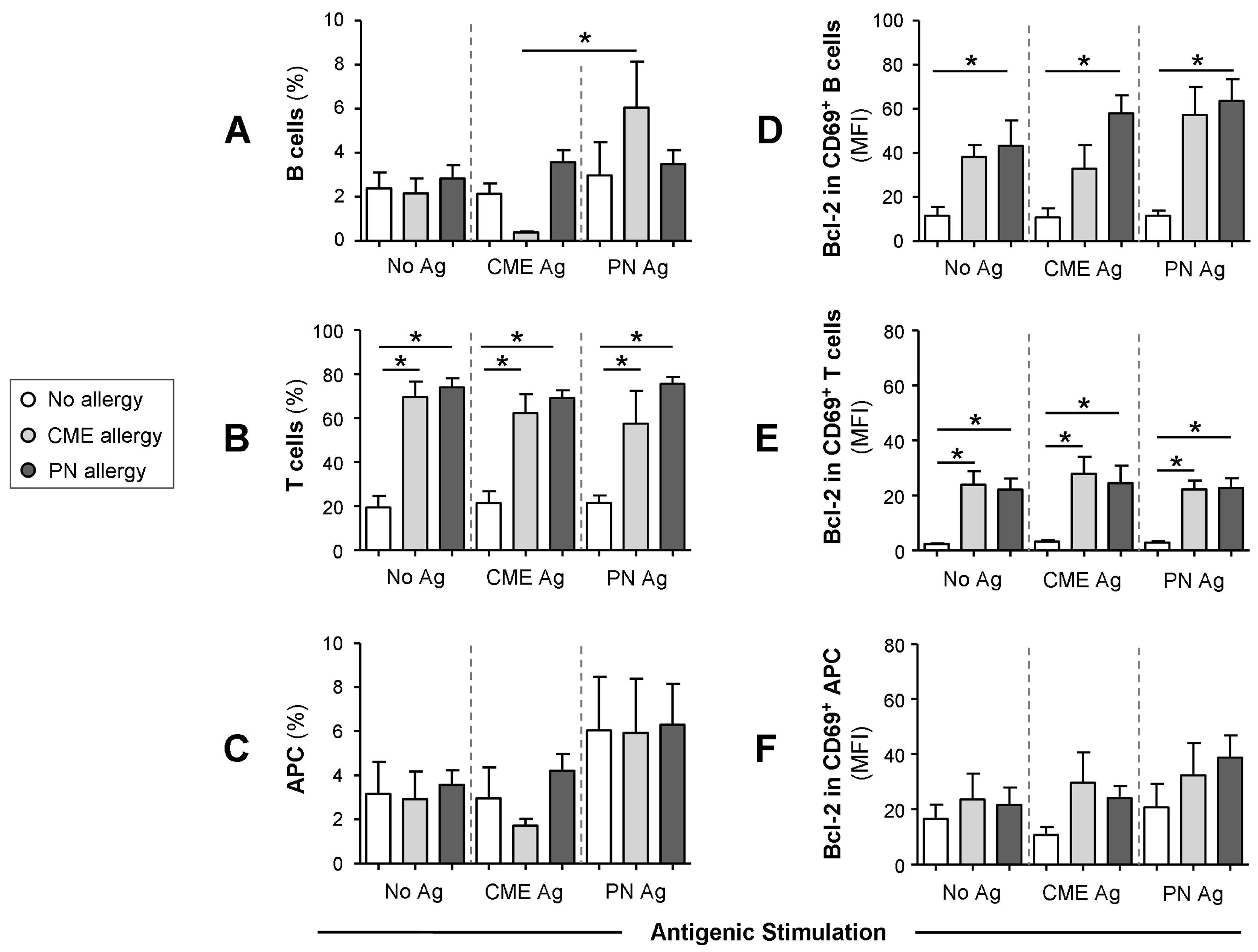
3.4. Differences in the Numbers of Immune Cell Subsets
3.5. Survival Advantage of Immune Cell Subtypes from Allergic Children
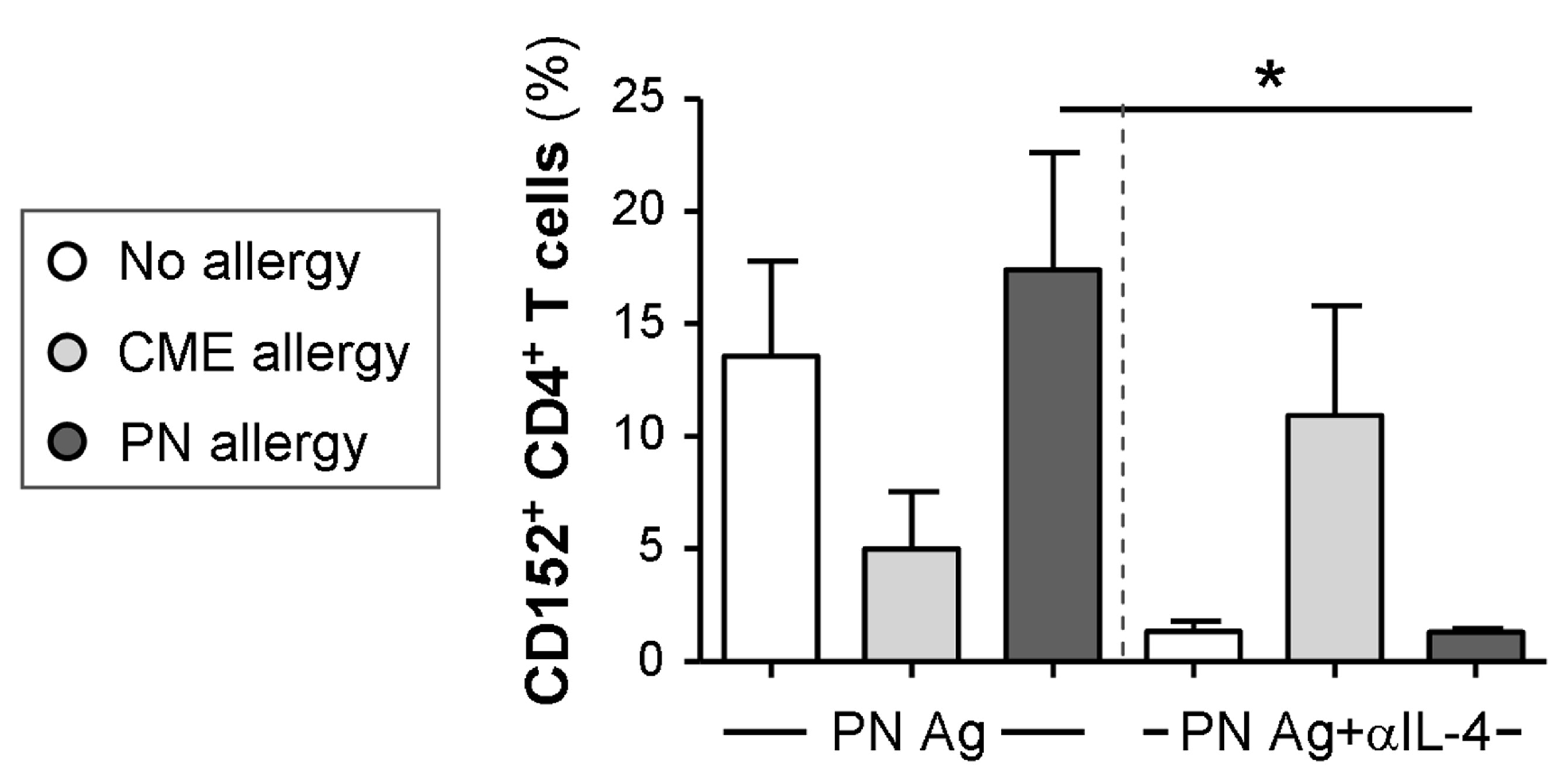
3.6. IL-4-Mediated Regulation of CD152 Expression
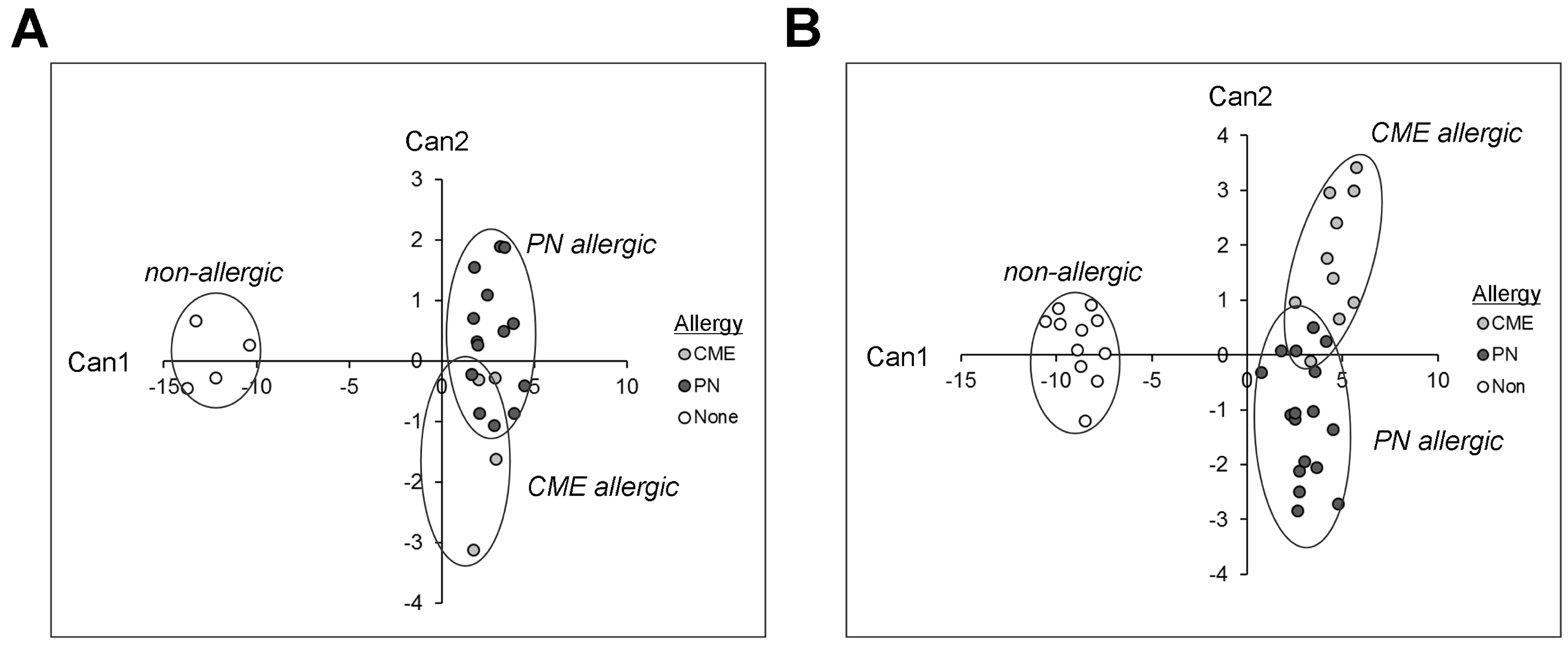
3.7. Canonical Discriminant Analysis
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
References
- Ling, Z.; Li, Z.; Liu, X.; Cheng, Y.; Luo, Y.; Tong, X.; Yuan, L.; Wang, Y.; Sun, J.; Li, L.; et al. Altered Fecal Microbiota Composition Associated with Food Allergy in Infants. Appl. Environ. Microbiol. 2014, 80, 2546–2554. [Google Scholar] [CrossRef] [PubMed]
- Tomicić, S.; Fälth-Magnusson, K.; Böttcher, M.F. Dysregulated Th1 and Th2 responses in food-allergic children—Does elimination diet contribute to the dysregulation? Pediatr. Allergy Immunol. 2010, 4, 649–655. [Google Scholar] [CrossRef] [PubMed]
- Chatila, T. Regulatory T cells: Key Players in Tolerance and Autoimmunity. Endocrinol. Metab. Clin. N. Am. 2009, 38, 265–272. [Google Scholar] [CrossRef] [PubMed]
- Kwon, H.K.; Lee, C.G.; So, J.S.; Chae, C.S.; Hwang, J.S.; Sahoo, A.; Nam, J.H.; Rhee, J.H.; Hwang, K.C.; Im, S.H. Generation of regulatory dendritic cells and CD4+Foxp3+ T cells by probiotics administration suppresses immune disorders. Proc. Natl. Acad. Sci. USA 2010, 107, 2159–2164. [Google Scholar] [CrossRef] [PubMed]
- Ruiter, B.; Shreffler, W.G. The role of dendritic cells in food allergy. J. Allergy Clin. Immunol. 2012, 129, 921–928. [Google Scholar] [CrossRef]
- Pabst, O.; Mowat, A.M. Oral tolerance to food protein. Mucosal Immunol. 2012, 5, 232–239. [Google Scholar] [CrossRef]
- Ogrodowczyk, A.; Markiewicz, L.; Wróblewska, B. Mutations in the filaggrin gene and food allergy. Prz. Gastroenterol. 2014, 9, 200–207. [Google Scholar] [CrossRef]
- Frischmeyer-Guerrerio, P.A.; Guerrerio, A.L.; Chichester, K.L.; Bieneman, A.P.; Hamilton, R.A.; Wood, R.A.; Schroeder, J.T. Dendritic cell and T cell responses in children with food allergy. Clin. Exp. Allergy 2011, 41, 61–71. [Google Scholar] [CrossRef][Green Version]
- Boesteanu, A.C.; Katsikis, P.D. Memory T cells need CD28 costimulation to remember. Semin. Immunol. 2009, 21, 69–77. [Google Scholar] [CrossRef]
- Borriello, F.; Sethna, M.P.; Boyd, S.D.; Schweitzer, A.N.; Tivol, E.A.; Jacoby, D.; Strom, T.B.; Simpson, E.M.; Freeman, G.J.; Sharpe, A.H. B7-1 and B7-2 Have Overlapping, Critical Roles in Immunoglobulin Class Switching and Germinal Center Formation. Immunity 1997, 6, 303–313. [Google Scholar] [CrossRef]
- van Wijk, F.; Nierkens, S.; de Jong, W.; Wehrens, E.J.; Boon, L.; van Kooten, P.; Knippels, L.M.; Pieters, R. The CD28/CTLA-4-B7 signaling pathway is involved in both allergic sensitization and tolerance induction to orally administered peanut proteins. J. Immunol. 2007, 178, 6894–6900. [Google Scholar] [CrossRef] [PubMed]
- Lenschow, D.J.; Walunas, T.L.; Bluestone, J.A. CD28/B7 system of T cell costimulation. Ann. Rev. Immunol. 1996, 14, 233–258. [Google Scholar] [CrossRef] [PubMed]
- van Wijk, F.; Hoeks, S.; Nierkens, S.; Koppelman, S.J.; van Kooten, P.; Boon, L.; Knippels, L.M.; Pieters, R. CTLA-4 signaling regulates the intensity of hypersensitivity responses to food antigens, but is not decisive in the induction of sensitization. J. Immunol. 2005, 174, 174–179. [Google Scholar] [CrossRef] [PubMed]
- Grohmann, U.; Orabona, C.; Fallarino, F.; Vacca, C.; Calcinaro, F.; Falorni, A.; Candeloro, P.; Belladonna, M.L.; Bianchi, R.; Fioretti, M.C.; et al. CTLA-4–Ig regulates tryptophan catabolism in vivo. Nat. Immunol. 2002, 3, 1097–1101. [Google Scholar] [CrossRef] [PubMed]
- Jeannin, P.; Delneste, Y.; Lecoanet-Henchoz, S.; Gauchat, J.-F.; Jonathan Ellis, J.; Bonnefoy, J.-Y. CD86 (B7.2) on human B cells. A functional role in proliferation and selective differentiation into IgE and IgG4-producing cells. J. Biol. Chem. 1997, 272, 15613–15619. [Google Scholar] [CrossRef] [PubMed]
- Cooper, A.M.; Hobson, P.S.; Jutton, M.R.; Kao, M.W.; Drung, B.; Schmidt, B.; Fear, D.J.; Beavil, A.J.; McDonnel, J.M.; Sutton, B.J.; et al. Soluble CD23 Controls IgE Synthesis and Homeostasis in Human B Cells. J. Immunol. 2012, 188, 3199–3207. [Google Scholar] [CrossRef] [PubMed]
- Gould, H.J.; Sutton, B.J. IgE in allergy and asthma today. Nat. Rev. Immunol. 2008, 8, 205–217. [Google Scholar] [CrossRef]
- Edkins, A.L.; Borland, G.; Acharya, M.; Cogdell, R.J.; Ozanne, B.W.; Cushley, W. Differential regulation of monocyte cytokine release by αV and β2 integrins that bind CD23. Immunology 2012, 136, 241–251. [Google Scholar] [CrossRef]
- Lindsey, J.; White, L.J.; Bradford, W.; Ozanne, B.W.; Pierre Graber, P.; Jean-Pierre Aubry, J.-P.; Jean-Yves Bonnefoy, J.Y.; William Cushley, W. Inhibition of Apoptosis in a Human Pre-B–Cell Line by CD23 Is Mediated Via a Novel Receptor. Blood 1997, 90, 234–243. [Google Scholar]
- Liu, Y.J.; Cairns, J.A.; Holder, M.J.; Abbot, S.D.; Jansen, K.U.; Bonnefoy, J.Y.; Gordon, J.; MacLennan, I.C. Recombinant 25-kDa CD23 and interleukin 1 alpha promote the survival of germinal center B cells: Evidence for bifurcation in the development of centrocytes rescued from apoptosis. Eur. J. Immunol. 1991, 21, 1107–1114. [Google Scholar] [CrossRef]
- Cory, S. Regulation of lymphocyte survival by the BCL-2 gene family. Ann. Rev. Immunol. 1995, 13, 513–543. [Google Scholar] [CrossRef] [PubMed]
- Graninger, W.B.; Steiner, C.W.; Graninger, M.T.; Aringer, M.; Smolen, J.S. Cytokine regulation of apoptosis and Bcl-2 expression in lymphocytes of patients with systemic lupus erythematosus. Cell Death Differ. 2000, 7, 966–972. [Google Scholar] [CrossRef] [PubMed]
- Aronica, M.A.; Goenka, S.; Boothby, M. IL-4-dependent induction of BCL-2 and BCL-X(L)IN activated T lymphocytes through a STAT6- and pi 3-kinase-independent pathway. Cytokine 2000, 12, 578–587. [Google Scholar] [CrossRef]
- Man, A.L.; Bertelli, E.; Regoli, M.; Chambers, S.J.; Nicoletti, C. Antigen-specific T cell–mediated apoptosis of dendritic cells is impaired in a mouse model of food allergy. J. Allergy Clin. Immunol. 2004, 113, 965–972. [Google Scholar] [CrossRef] [PubMed]
- Arques, J.L.; Regoli, M.; Bertelli, E.; Nicoletti, C. Persistence of apoptosis-resistant T cell-activating dendritic cells promotes T helper type-2 response and IgE antibody production. Mol. Immunol. 2008, 45, 2177–2186. [Google Scholar] [CrossRef]
- Sicherer, S.H. Food allergy from infancy through adulthood. J. Allergy Clin. Immunol. 2020, 8, 1854–1864. [Google Scholar] [CrossRef]
- Clemente, A.; Chambers, S.J.; Lodi, F.; Nicoletti, C.; Brett, G.M. Use of indirect competitive ELISA for the detection of Brazil nut in food products. Food Control. 2004, 15, 65–69. [Google Scholar] [CrossRef]
- Ivory, K.; Chambers, S.J.; Pin, C.; Prieto, E.; Arques, J.L.; Nicoletti, C. Oral delivery of Lactobacillus casei Shirota modifies allergen-specific immune responses in allergic rhinitis. Clin. Exp. Allergy 2008, 38, 1282–1289. [Google Scholar] [CrossRef]
- Riemann, D.; Turzer, S.; Ganchev, G.; Schütte, W.; Seliger, B.; Möller, M. Monitoring Blood Immune Cells in Patients with Advanced Small Cell Lung Cancer Undergoing a Combined Immune Checkpoint Inhibitor/Chemotherapy. Biomolecules 2023, 13, 190. [Google Scholar] [CrossRef]
- Uchida, Y.; Kurasawa, K.; Nakajima, H.; Nakagawa, N.; Tanabe, E.; Sueishi, M.; Saito, Y.; Iwamoto, I. Increase of dendritic cells of type 2 (DC2) by altered response to IL-4 in atopic patients. J. Allergy Clin. Immunol. 2001, 108, 1005–1011. [Google Scholar] [CrossRef]
- Sancho, D.; Gomez, M.; Sanchez-Madrid, F. CD69 is an immunoregulatory molecule induced following activation. Trends Immunol. 2005, 26, 136–140. [Google Scholar] [CrossRef]
- Pandiyan, P.; Gärtner, G.; Soezeri, O.; Radbruch, A.; Schulze-Osthoff, K.; Brunner-Weinzierl, M.C. CD152 (CTLA-4) determines the unequal resistance of Th1 and Th2 cells against activation-induced cell death by a mechanism requiring PI3 kinase function. J. Exp. Med. 2004, 199, 831–842. [Google Scholar] [CrossRef]
- Rudd, C.E.; Taylor, A.; Schneider, H. CD28 and CTLA-4 coreceptor expression and signal transduction. Immunol. Rev. 2009, 229, 12–26. [Google Scholar] [CrossRef]
- Alegre, M.L.; Noel, P.J.; Eisfelder, B.J.; Chuang, E.; Clark, M.R.; Reiner, S.L.; Thompson, C.B. Regulation of surface and intracellular expression of CTLA4 on mouse T cells. J. Immunol. 1996, 157, 4762–4770. [Google Scholar] [CrossRef]
- Pioli, C.; Gatta, L.; Ubaldi, V.; Doria, G. Inhibition of IgG1 and IgE production by stimulation of the B cell CTLA-4 receptor. J. Immunol. 2000, 165, 5530–5536. [Google Scholar] [CrossRef]
- Akdis, C.A.; Blesken, T.; Akdis, M.; Wüthrich, B.; Blaser, K. Role of interleukin-10 in specific immunotherapy. J. Clin. Investig. 1998, 102, 98–106. [Google Scholar] [CrossRef]
- Dang, T.D.; Tang, M.L.K.; Koplin, J.J.; Licciardi, P.V.; Eckert, J.K.; Tan, T.; Gurin, L.C.; Ponsonby, A.-L.; Allen, K.L. Characterization of plasma cytokines in an infant population cohort of challenge-proven food allergy. Allergy 2013, 68, 1333–1340. [Google Scholar] [CrossRef]
- Schmidt-Weber, C.B.; Alexander, S.I.; Henault, L.E.; James, L.; Lichtman, A.H. IL-4 Enhances IL-10 Gene Expression in Murine Th2 Cells in the Absence of TCR Engagement. J. Immunol. 1999, 162, 238–244. [Google Scholar] [CrossRef]
- Lotoski, L.C.; Simons, F.E.R.; Chooniedass, R.; Liem, J.; Ostopowich Becker, A.B.; HayGlass, K.T. Are Plasma IL-10 Levels a Useful Marker of Human Clinical Tolerance in Peanut Allergy? PLoS ONE 2010, 5, e11192. [Google Scholar] [CrossRef] [PubMed]
- Stone, S.F.; Cotterell, C.; Isbister, G.K.; Holdgate, A.; Brown, S.G. Elevated serum cytokines during human anaphylaxis: Identification of potential mediators of acute allergic reactions. J. Allergy Clin. Immunol. 2009, 124, 786–792. [Google Scholar] [CrossRef] [PubMed]
- Palma, J.P.; Yauch, R.L.; Kang, H.K.; Lee, H.G.; Kim, B.S. Preferential induction of IL-10 in APC correlates with a switch from Th1 to Th2 response following infection with a low pathogenic variant of Theiler’s virus. J. Immunol. 2002, 168, 4221–4230. [Google Scholar] [CrossRef] [PubMed]
- Laouini, D.; Alenius, H.; Bryce, P.; Oettgen, H.; Tsitsikov, E.; Geha, R.S. IL-10 is critical for Th2 responses in a murine model of allergic dermatitis. J. Clin. Investig. 2003, 112, 1058–1066. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, N.; Nagumo, H.; Agematsu, K. IL-10 enhances B-cell IgE synthesis by promoting differentiation into plasma cells, a process that is inhibited by CD27/CD70 interaction. Clin. Exp. Immunol. 2002, 129, 446–452. [Google Scholar] [CrossRef] [PubMed]
- Punnonen, J.; Kainulainen, L.; Ruuskanen, O.; Nikoskelainen, J.; Arvilommi, H. IL-4 synergizes with IL-10 and anti-CD40 MoAbs to induce B-cell differentiation in patients with common variable immunodeficiency. Scand. J. Immunol. 1997, 45, 203–212. [Google Scholar] [CrossRef] [PubMed]
- Uejima, Y.; Takahashi, K.; Komoriya, K.; Kurozumi, S.; Ochs, H.D. Effect of interleukin-10 on anti-CD40- and interleukin-4-induced immunoglobulin E production by human lymphocytes. Int. Arch. Allergy Appl. Immunol. 1996, 110, 225–232. [Google Scholar] [CrossRef] [PubMed]
- Nakae, S.; Komiyama, Y.; Yokoyama, H.; Nambu, A.; Umeda, M.; Iwase, M.; Homma, I.; Sudo, K.; Horai, R.; Asano, M.; et al. IL-1 is required for allergen-specific Th2 cell activation and the development of airway hypersensitivity response. Int. Immunol. 2003, 15, 483–490. [Google Scholar] [CrossRef] [PubMed]
- Dinarello, C.A. Biologic basis for interleukin-1 in disease. Blood 1996, 87, 2095–2147. [Google Scholar] [CrossRef] [PubMed]
- van Haelst Pisani, C.; Kovach, J.S.; Kita, H.; Leiferman, K.M.; Gleich, G.J.; Silver, J.E.; Dennin, R.; Abrams, J.S. Administration of interleukin-2 (IL-2) results in increased plasma concentrations of IL-5 and eosinophilia in patients with cancer. Blood 1991, 78, 1538–1544. [Google Scholar] [CrossRef]
- Blumchen, K.; Ulbricht, H.; Staden, U.; Dobberstein, K.; Beschorner, J.; de Oliveira, L.C.; Shreffler, W.G.; Sampson, H.A.; Niggemann, B.; Wahn, U.; et al. Oral peanut immunotherapy in children with peanut anaphylaxis. J. Allergy Clin. Immunol. 2010, 126, 83–91. [Google Scholar] [CrossRef]
- Kouro, T.; Takatsu, K. IL-5- and eosinophil-mediated inflammation: From discovery to therapy. Int. Immunol. 2009, 2, 1303–1309. [Google Scholar] [CrossRef]
- Bublin, M.; Breiteneder, H. Cross-Reactivity of Peanut Allergens. Curr. Allergy Asthma Rep. 2014, 14, 426. [Google Scholar] [CrossRef]
- Rozenfeld, P.; Docena, G.H.; Añón, M.C.; Fossati, C.A. Detection and identification of a soy protein component that cross-reacts with caseins from cow’s milk. Clin. Exp. Immunol. 2002, 130, 49–58. [Google Scholar] [CrossRef] [PubMed]
- Popescu, F.-D. Cross-reactivity between aeroallergens and food allergens. World J. Methodol. 2015, 5, 31–50. [Google Scholar] [CrossRef] [PubMed]
- Sansom, D.M.; Manzotti, C.N.; Zheng, Y. What’s the difference between CD80 and CD86? Trends Immunol. 2003, 24, 313–318. [Google Scholar] [CrossRef]
- Casas, R.; Skarsvik, S.; Lindström, A.; Zetterström, O.; Duchén, K. Impaired maturation of monocyte-derived dendritic cells from birch allergic individuals in association with birch-specific immune responses. Scand. J. Immunol. 2007, 66, 591–598. [Google Scholar] [CrossRef]
- Hubo, M.; Trinschek, B.; Kryczanowsky, F.; Tuettenberg, A.; Steinbrink, K.; Jonuleit, H. Costimulatory Molecules on Immunogenic Versus Tolerogenic Human Dendritic Cells. Front. Immunol. 2013, 4, 82. [Google Scholar] [CrossRef]
- Singh, R.; Letai, A.; Sarosiek, K. Regulation of apoptosis in health and disease: The balancing act of BCL-2 family proteins. Nat. Rev. Mol. Cell Biol. 2019, 20, 175–193. [Google Scholar] [CrossRef]
- Chambers, S.J.; Bertelli, E.; Winterbone, M.S.; Regoli, M.; Man, A.L.; Nicoletti, C. Adoptive transfer of dendritic cells from allergic mice induces specific immunoglobulin E antibody in naïve recipients in absence of antigen challenge without altering the T helper 1/T helper 2 balance. Immunology 2004, 112, 72–79. [Google Scholar] [CrossRef]
- Berin, M.C.; Sampson, H.A. Food allergy: An enigmatic epidemic. Trends Immunol. 2013, 34, 390–397. [Google Scholar] [CrossRef]
- Mudter, J.; Neurath, M.F. Apoptosis of T cells and the control of inflammatory bowel disease: Therapeutic implications. Gut 2007, 56, 293–303. [Google Scholar] [CrossRef] [PubMed]


| Variable | No Allergy | CME Allergy | PN Allergy |
|---|---|---|---|
| Constant | −67.44 | −198.85 | −218.99 |
| *Ln IL-2 | 9.66 | 14.39 | 16.71 |
| Ln IL-4 | 39.86 | 66.13 | 69.06 |
| Ln IL-5 | −9.99 | 8.74 | 9.78 |
| Ln IL-1β | −19.71 | −10.74 | −13.80 |
| Ln IL-10 | 4.98 | −4.57 | −5.22 |
| Ln IL-12p70 | 5.93 | −2.47 | −1.79 |
| Variable | No Allergy | CME Allergy | PN Allergy |
|---|---|---|---|
| Constant | −0.35 | −19.39 | −16.52 |
| *Ln IL-2 | −0.01 | 1.33 | 1.29 |
| Ln IL-4 | 0.33 | 1.41 | 1.57 |
| Ln IL-5 | 0.05 | 3.99 | 3.92 |
| Ln IFN-γ | 0.26 | −0.65 | −0.63 |
| Ln IL-10 | −0.18 | 2.19 | 1.46 |
| Ln IL-12p70 | −0.16 | −3.22 | −2.76 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ivory, K.; Angotti, R.; Messina, M.; Bonente, D.; Paternostro, F.; Gulisano, M.; Nicoletti, C. Alteration of Immunoregulatory Patterns and Survival Advantage of Key Cell Types in Food Allergic Children. Cells 2023, 12, 2736. https://doi.org/10.3390/cells12232736
Ivory K, Angotti R, Messina M, Bonente D, Paternostro F, Gulisano M, Nicoletti C. Alteration of Immunoregulatory Patterns and Survival Advantage of Key Cell Types in Food Allergic Children. Cells. 2023; 12(23):2736. https://doi.org/10.3390/cells12232736
Chicago/Turabian StyleIvory, Kamal, Rossella Angotti, Mario Messina, Denise Bonente, Ferdinando Paternostro, Massimo Gulisano, and Claudio Nicoletti. 2023. "Alteration of Immunoregulatory Patterns and Survival Advantage of Key Cell Types in Food Allergic Children" Cells 12, no. 23: 2736. https://doi.org/10.3390/cells12232736
APA StyleIvory, K., Angotti, R., Messina, M., Bonente, D., Paternostro, F., Gulisano, M., & Nicoletti, C. (2023). Alteration of Immunoregulatory Patterns and Survival Advantage of Key Cell Types in Food Allergic Children. Cells, 12(23), 2736. https://doi.org/10.3390/cells12232736







