Biotechnology of the Tree Fern Cyathea smithii (J.D. Hooker; Soft Tree Fern, Katote) II Cell Suspension Culture: Focusing on Structure and Physiology in the Presence of 2,4-D and BAP
Abstract
1. Introduction
2. Materials and Methods
2.1. Medium Description
2.2. Culture Initiation and Cell Suspension Establishment
2.3. Preparation of Living and Fixed Specimens for Bright-Field Light Microscopy
2.4. Scanning Electron Microscopy (SEM) Preparation
2.5. Semi-Thin Specimen Preparation
2.6. Preparation of Living Specimens for Confocal Microscopy
2.7. Acriflavine Nuclei Staining of Cell Suspension and Determination of Nuclear DNA Content
2.8. Cell Suspension Maintenance
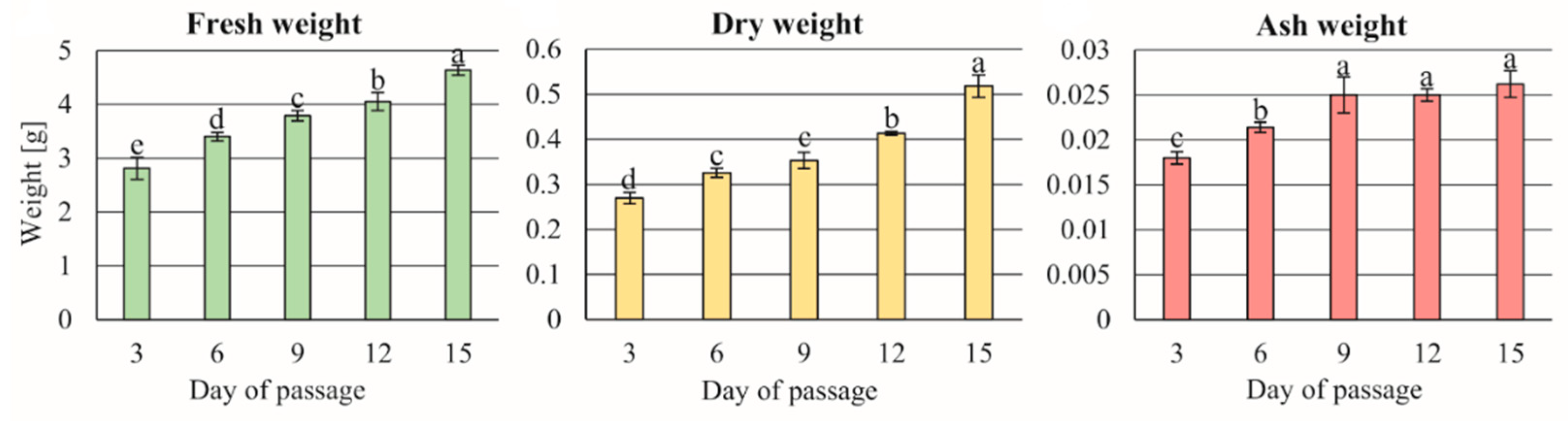
2.9. Fresh, Dry, and Ash Matter Evaluation
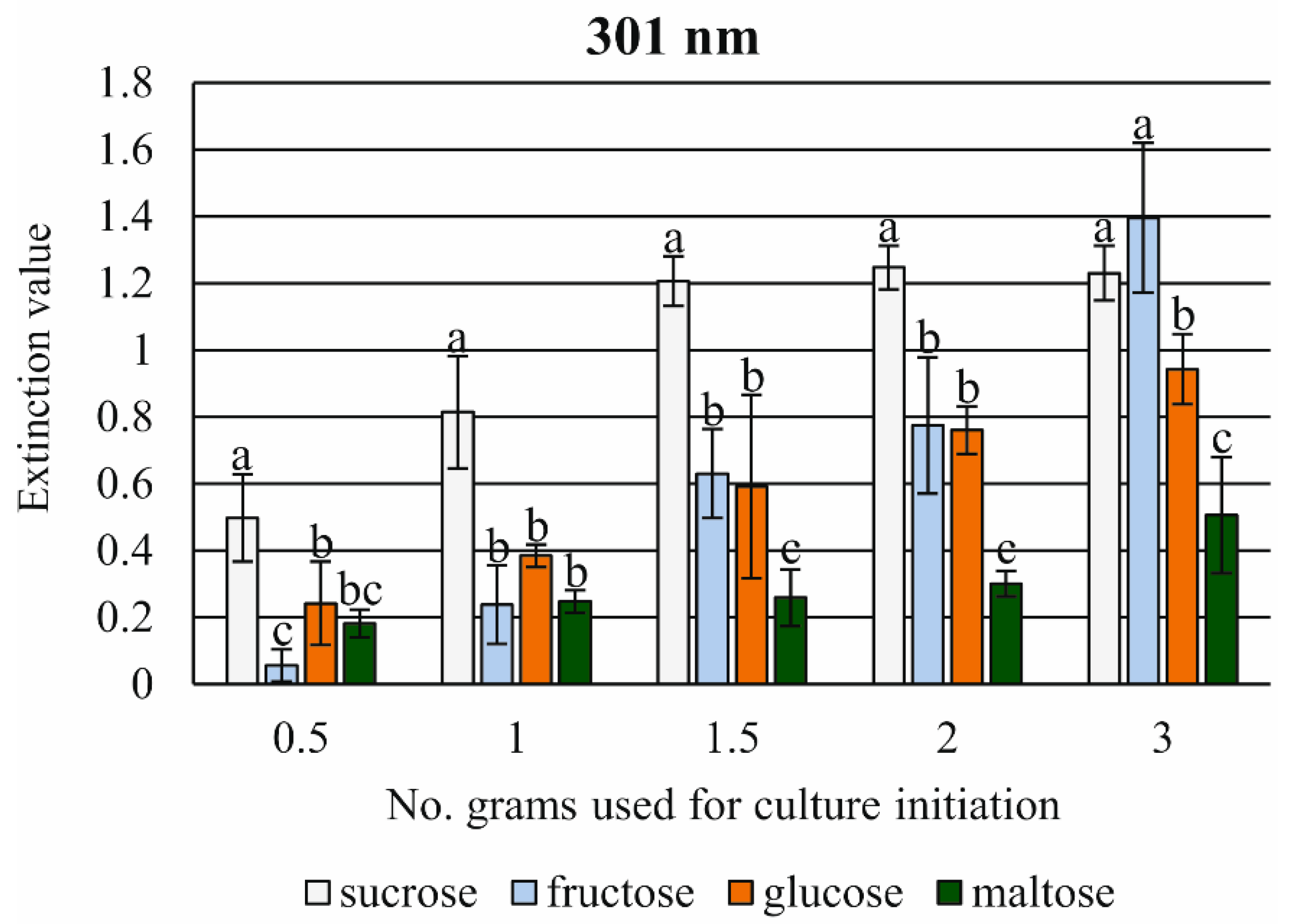
2.10. Measurements of the Growth Kinetics of Cell Suspensions Depending on the Type of Energy Source
2.11. Measurements of Quercetin Produced by the Cell Suspension during the 15-Day Subculture in the Post-Culture Medium
2.12. Effectivity of Ethylene Production in Relation to the Fresh Weight of Initial Tissue
2.13. Effectivity of Ethylene Production According to the Length of Time
2.14. Antibacterial Activity of Extracts of Cell Suspension
2.14.1. Antibacterial Character Determined via Agar Disc Diffusion Assay
2.14.2. MIC (Minimum Inhibitory Concentration) and MBC (Minimal Bactericidal Concentration) Analyses
3. Results
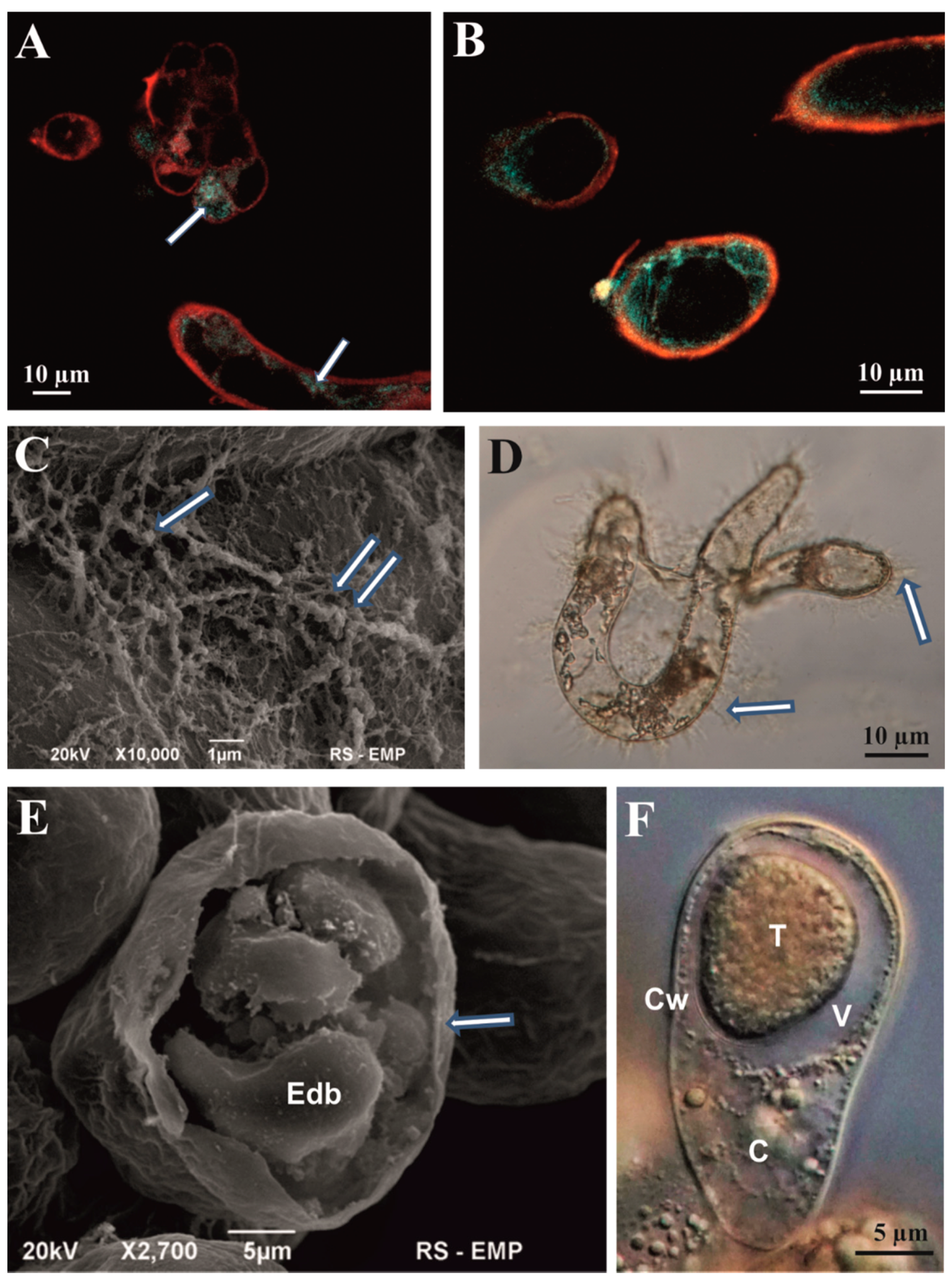
3.1. Microscopic Analysis of Cell Aggregates
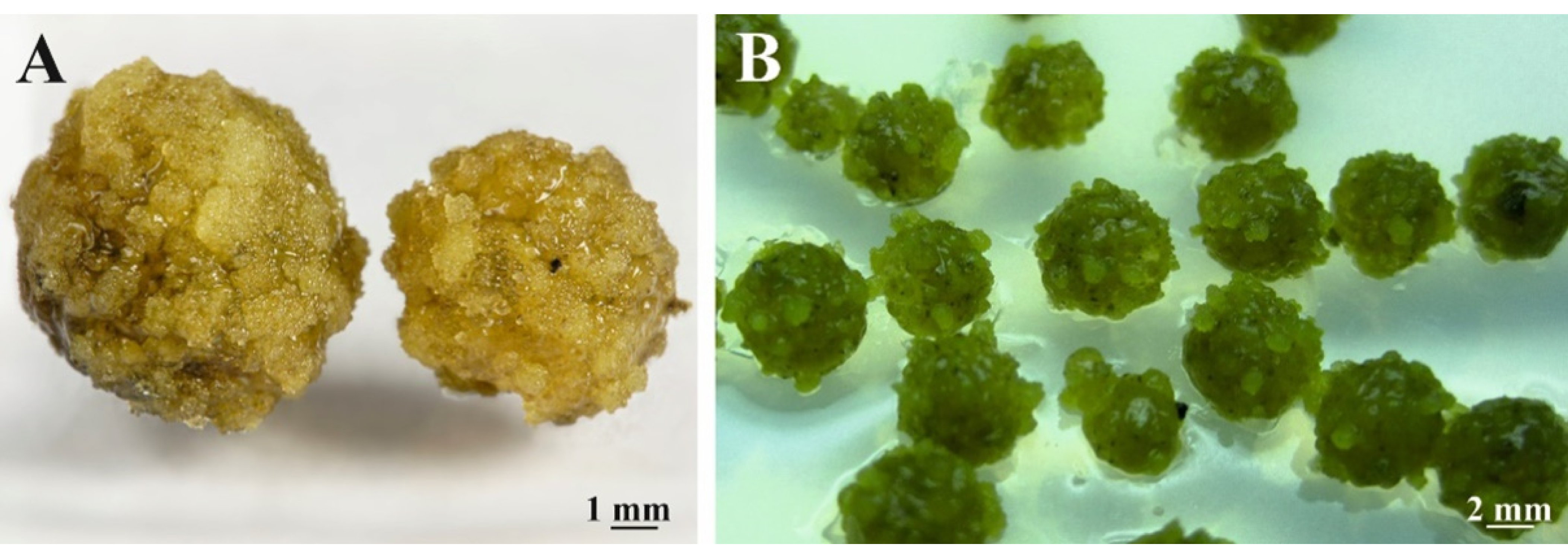
3.2. Morphogenic Potential Evaluation of Cell Suspensions
3.3. Measurement of Cell Suspension Kinetics
3.4. Nuclear DNA Content of Cell Suspension Determined with the Help of DNA-Specific Fluorochrome Acriflavine with Acetic Hydrolysis
3.5. Effectivity of ETH Production in Relation to the Fresh Weight of Initial Tissue of Subculture
3.6. Effectivity of ETH Production According to the Length of Time
3.7. Antibacterial Activity of C. smithii Extracts Tested with a Modified Disc Diffusion Method
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Large, M.F.; Braggins, J.E. Tree Ferns; CSIRO Publishing: Collingwood, VIC, Australia, 2004; pp. 1–359. [Google Scholar]
- Jones, D.L. Encyclopedia of Ferns; Timber Press: Portland, OR, USA, 1987; p. 254. [Google Scholar]
- Finne, J.F.; van Staden, J. Multiplication of the tree fern Cyathea dregei. HortScience 1987, 22, 665. [Google Scholar]
- Goller, K.; Rybczyński, J.J. In vitro culture used for woody fern Cyathea australis (R.Br.) Domin. vegetative propagation. Acta Soc. Bot. Pol. 1995, 64, 13–17. [Google Scholar] [CrossRef][Green Version]
- Goller, K.; Rybczyński, J.J. Gametophyte and sporophyte of tree-ferns in vitro culture. Acta Soc. Bot. Pol. 2007, 76, 193–199. [Google Scholar] [CrossRef]
- Rechenmacher, C.; Schmitt, J.L.; Droste, A. Spore germination and gametophyte development of Cyathea atrovirens (Langsd. & Fisch.) Domin (Cyatheaceae) under different pH conditions. Braz. J. Biol. 2010, 70, 4. [Google Scholar]
- De Vargas, I.B.; Droste, A. In vitro propagation of Cyathea atrovirens (Cyatheaceae): Spore storage and sterilization conditions. Rev. Biol. Trop. 2014, 62, 299–308. [Google Scholar] [CrossRef]
- Shukla, S.P.; Khare, P.B. In vitro shoot regeneration via caulogenesis in fern Pteris vittata L. J. Environ. Biol. 2012, 33, 683–687. [Google Scholar]
- Parajuli, J.; Joshi, S. In vitro studies of effect of growth hormones on sporophyte development of Cyathea spinulosa. Intern. J. Biodiv. Concerv. 2014, 6, 247–255. [Google Scholar] [CrossRef][Green Version]
- Das, S.; Choudhury, M.D.; Mazumder, P.B. In vitro propagation of Cyathea gigantea (Will Ex. Hook)—A tree fern. IJRSR 2013, 4, 221–224. [Google Scholar]
- Shimabukuro, A.E. The effect of sterilization and storage conditions on the viability of the spores of Cyathea delgadii. Am. Fern J. 1998, 88, 72–80. [Google Scholar] [CrossRef]
- Mikuła, A.; Grzyb, M.; Tomiczak, K.; Rybczyński, J.J. Experimental and practical application of fern somatic embryogenesis. In Current Advances in Fern Research; Fernández, H., Ed.; Springer: Berlin/Heidelberg, Germany, 2018; pp. 121–137. [Google Scholar] [CrossRef]
- Domżalska, L.; Kędracka-Krok, S.; Jankowska, U.; Grzyb, M.; Sobczak, M.; Rybczyński, J.J.; Mikuła, A. Proteomic analysis of stipe explants reveals differentially expressed proteins involved in early direct somatic embryogenesis of the tree-fern Cyathea delgadii Sternb. Plant Sci. 2017, 258, 61–76. [Google Scholar] [CrossRef]
- Tomaszewicz, W.; Cio, C.M.; Dos Santos Szewczyk, K.; Grzyb, M.; Pietrzak, W.; Pawłowska, B.; Mikuła, A. Enhancing in vitro production of the tree fern Cyathea delgadii and modifying secondary metabolite profiles by LED Lighting. Cells 2022, 11, 486. [Google Scholar] [CrossRef] [PubMed]
- Byrne, T.E.; Caponetti, J.D. (a) Morphogenesis in three cultivars of Boston fern. II. Callus production from stolon tips and plantlet differentiation from callus. Am. Fern J. 1992, 82, 1–11. [Google Scholar] [CrossRef]
- Byrne, T.E.; Caponetti, J.D. (b) Morphogenesis in three cultivars of Boston Fern. III. Callus production and plantlet differentiation from cell suspensions. Am. Fern J. 1992, 82, 12–22. [Google Scholar] [CrossRef]
- Kwa, S.-H.; Wee, Y.-C.; Lim, T.-M.; Kumar, P.P. Morphogenetic plasticity of callus reinitiated from cell suspension cultures of the fern Platycerium coronarium. Plant Cell. Tiss. Org. Cult. 1997, 48, 37–44. [Google Scholar] [CrossRef]
- Teng, W.-L. Activated charcoal affects morphogenesis and enhances sporophyte regeneration during leaf cell suspension culture of Platycerium bifurcatum. Plant Cell Rep. 1997, 17, 77–83. [Google Scholar] [CrossRef] [PubMed]
- Teng, W.-L.; Teng, M.-C. In vitro regeneration patterns of Platycerium bifurcatum leaf cell suspension culture. Plant Cell Rep. 1997, 16, 820–824. [Google Scholar] [CrossRef]
- Riviera, A.; Conde, P.; Cañal, M.J.; Fernández, H. Biotechnology and apogamy in Dropteris affinis spp. Affinis: The influence of tissue homogenization, auxins, cytokinins, gibberellic acid and polyamins. In Current Advances in Fern Research; Fernández, H., Ed.; Springer: Berlin/Heidelberg, Germany, 2018; pp. 139–152. [Google Scholar]
- Hagel, J.J.; Page, J.E.; Facchini, R.J. Impact of whole-genome and expressed sequence tag databases on the study of plant secondary metabolism. In Medicinal Plant Biotechnology; Kayser, O., Uax, W., Eds.; Wiley online Library: Hoboken, NJ, USA, 2007; Volume 1, pp. 203–236. [Google Scholar]
- Harada, T.; Saikai, Y. Distribution of flavonoids in ferns (2). Pharmaceutical studies on ferns VIII. Pharm. Bull. 1955, 3, 469–472. [Google Scholar] [CrossRef][Green Version]
- Kale, M. Qualitative and quantitative analysis of Cyathea giganthea ferns from south western Ghats. Res. J. Pharm. Biol. Chem. Sci. 2015, 1, 183–187. [Google Scholar] [CrossRef]
- Soeder, R.W.; Babb, M.S. Flavonoids in tree ferns. Phytochemistry 1972, 11, 3079–3080. [Google Scholar] [CrossRef]
- Hiraoka, A.; Masegawa, M. Flavonoides from five Cyathea species. Bot. Mag. Tokyo 1975, 88, 127–130. [Google Scholar] [CrossRef]
- Hiraoka, A.; Maeda, M. A new acylated flavonol glycoside from Cyathea contaminans Copel and its distribution in the pterydophyta. Chem. Pharm. Bull. 1979, 27, 3130–3136. [Google Scholar] [CrossRef]
- Janakiraman, N.; Johnson, M.A. Inter specific variation studies on Cyathea species using phytochemical and fluorescence analysis. J. Pharmacol. Toxicol. Stud. 2015, 3, 11–17. [Google Scholar]
- Janakiraman, N.; Johnson, M.A. CS-MS analysis of ethanolic extracts of Cyathea nilgirensis, C. gigantea and C. crinite. Egypt. Pharm. J. 2016, 15, 43. [Google Scholar] [CrossRef]
- Markham, K.R.; Mabry, T.J. Ultraviolet-visible and protonmagnetic resonance spectroscopy of flavonoids. In Flavonoids; Harborne, J.B., Mabry, T.J., Eds.; Springer Science Business Media B.V.: Berlin/Heidelberg, Germany, 1975; Chapter II; pp. 45–77. [Google Scholar]
- Muir, W.H.A.; Hildebrand, A.C.; Riker, A.J. Plant tissue culture produced from single isolated cells. Science 1954, 119, 877–878. [Google Scholar] [CrossRef]
- Muir, W.H.A.; Hildebrand, A.C.; Riker, A.J. The preparation, isolation and growth in culture of single cells from higher plants. Am. J. Bot. 1958, 45, 589–597. [Google Scholar] [CrossRef]
- Nikell, L.G. The continuous submerged cultivation of plant cell tissue as single cell. Proc. Natl. Acad. Sci. USA 1956, 42, 848–850. [Google Scholar] [CrossRef]
- De Ropp, R.S. The growth and behavior in vitro of isolated plant cells. Proc. R. Soc. Lond. B 1955, 144, 86–93. [Google Scholar]
- Steward, F.C.; Caplin, S.M.; Millar, F.K. Investigations on growth and metabolism of plant cells. I. New techniques for the investigation of metabolism, nutrition and growth in undifferentiated cells. Ann. Bot. 1952, 16, 57–79. [Google Scholar] [CrossRef]
- Steward, F.C.; Mapes, M.O.; Smith, J. Growth and organized development of cultured cells. I. Growth and division of freely suspended cells. Am. J. Bot. 1958, 45, 693–703. [Google Scholar] [CrossRef]
- Torrey, J.G.; Shigemura, Y. Growth and controlled morphogenesis in pea root callus tissue grown in liquid media. Am. J. Bot. 1957, 44, 334–344. [Google Scholar] [CrossRef]
- Vasil, I.K.; Vasil, V. Advances in cereal protoplast research. Physiol. Plant. 1992, 85, 279–283. [Google Scholar] [CrossRef]
- Oleszczuk, S.; Sowa, S.; Zimny, J. Direct embryogenesis and green plant regeneration from isolated microspore of hexaploidy Triticosecale (Wittm) cv. Bogo. Plant Cell Rep. 2004, 22, 85–93. [Google Scholar] [CrossRef] [PubMed]
- Vetter, J. Secondary metabolites of fern. In Current Advances in Fern Research; Fernandez, H., Ed.; Springer: Berlin/Heidelberg, Germany, 2018; pp. 305–327. [Google Scholar] [CrossRef]
- Santos, R.B.; Abranches, R.; Fischer, R.; Sack, M.; Holland, T. Putting the spotlight back on plant suspension cultures. Front. Plant Sci. 2016, 7, 297. [Google Scholar] [CrossRef] [PubMed]
- Rybczynski, J.J.; Tomiczak, K.; Grzyb, M.; Mikuła, A. Morphogenic events in ferns: Single and multicellural explant in vitro. In Current Advances in Fern Research; Fernandez, H., Ed.; Springer: Berlin/Heidelberg, Germany, 2018; pp. 99–120. [Google Scholar]
- Herbert, R.J.; Vilhar, B.; Evett, C.; Orchard, C.B.; Rogers, H.J.; Davies, M.S.; Francis, D. Ethylene induces cell death at particular phases of the cell cycle in the tobacco TBY-2 cell line. J. Exp. Bot. 2009, 52, 1615–1623. [Google Scholar]
- Schulte, A.; Schilling, J.V.; Nolten, J.; Korona, A.; Krömke, H.; Vennekötter, J.-B.; Schillheim, B.; Wessling, M.; Conrath, U.; Büchs, J. Parallel online determination of ethylene released rate by shaken parsley cell cultures using a modified RAMOS device. BMC Plant Biol. 2018, 18, 101. [Google Scholar] [CrossRef]
- Love, J.; Björklund, S.; Vahala, J.; Hertzberg, M.; Kangasjārvi, J.; Sundberg, B. Ethylene is an endogenous stimulator of cell division in the cambial meristem of Populus. Proc. Natl. Acad. Sci. USA 2009, 106, 5984–5989. [Google Scholar] [CrossRef]
- LaRue, T.A.G.; Gamborg, O.L. Ethylene production by plant cell culture. Variations in production during growing cycle and different plant species. Plant Physiol. 1971, 48, 394–398. [Google Scholar] [CrossRef][Green Version]
- Gamborg, O.L.; LaRue, T.A.G. Ethylene production by plant cell cultures: The effect of auxins, abscisic acid, and kinetin on ethylene production in suspension cultures of rose and ruta cells. Plant Physiol. 1971, 48, 399–401. [Google Scholar] [CrossRef][Green Version]
- Liderman, M.; Wang, S.Y.; Owens, L.D. Ethylene production by callus and cell suspension cells from cortex tissue of postclimacterie apples. Plant Physiol. 1979, 63, 811–815. [Google Scholar] [CrossRef]
- Biddington, N.L. The influence of ethylene in plant tissue. Plant Growth Regul. 1992, 11, 173–187. [Google Scholar] [CrossRef]
- Vajravel, S.; Sirin, S.; Kosourov, S.; Allahverdiyeva, Y. Towards sustainable ethylene production with cyanobacterial artificial biofilms. Green Chem. 2020, 22, 6404–6414. [Google Scholar] [CrossRef]
- Edwards, M.E.; Miller, J.H. Growth regulation by ethylene in fern gametophytes. III. Inhibition of spore germination. Am. J. Bot. 1972, 59, 458–546. [Google Scholar] [CrossRef]
- Fisher, R.W.; Miller, J.H. Growth regulation by ethylene in fern gametophytes. V. Ethylene and the early events of spore germination. Am. J. Bot. 1978, 65, 334–339. [Google Scholar] [CrossRef]
- Kwa, S.H.; Wee, Y.-C.; Kumar, P.P. Role of ethylene in the production of sporophytes from Platycerium coronarium (Koenig) Desv. frond and rhizome pieces cultured in vitro. J. Plant Growth Regul. 1995, 14, 183–189. [Google Scholar] [CrossRef]
- Bui, L.T.; Hurst, A.; Irish, E.E.; Cheng, C.-L. The effects of sugars and ethylene on apospory and regeneration in Ceratopteris richardii. Am. J. Plant Sci. 2012, 3, 953–961. [Google Scholar] [CrossRef][Green Version]
- Murashige, T.; Skoog, F. A revised medium for rapid growth and bioassays with tobacco tissue cultures. Physiol. Plant. 1962, 15, 473–492. [Google Scholar] [CrossRef]
- Fiuk, A.; Rybczyński, J.J. Factor influencing efficiency of somatic embryogenesis of Gentiana kurroo (Royle) cell suspension. Plant Biotechnol. Rev. 2008, 2, 33–39. [Google Scholar] [CrossRef]
- Raju, N.B. A simple fluorescent staining method for meiotic chromosomes of Neurospora. Mycologia 1986, 78, 901–906. [Google Scholar] [CrossRef]
- Dmuchowski, W.; Brągoszewska, P.; Gozdowski, D.; Baczewska-Dąbrowska, A.B.; Chojnacki, T.; Jóźwiak, A. Strategy of Ginkgo biloba L. in the mitigation of salt stress in the urban environment. Urban For. Urban Green. 2019, 38, 223–323. [Google Scholar] [CrossRef]
- Markham, K.R.; Mabry, T.J. Ultraviolet-visible and proton magnetic resonance spectroscopy of flavonoids. In Harborne; Mabry, T.J., Mabry, H., Eds.; The Flavonoids, Chapman and Hall: London, UK, 1975; pp. 45–77. [Google Scholar]
- Murray, P.R.; Baron, E.J.; Pfaller, M.A.; Tenover, F.C.; Yolke, R.H. Manuel of Clinical Microbiology, 6th ed.; ASM: Washington, DC, USA, 1995; p. 300. [Google Scholar]
- Méndez, C.C.; Serrano, M.C.; Valverde, A.; Pemán, J.; Almeida, C.; Mazuelos, S.M. Comparison of E-Test®, disk diffusion and a modified CLSI broth microdilution (M 38-A) method for in vitro testing of itraconazole, fluconazole and voriconazole against dermatophytes. Med. Mycol. 2008, 46, 119–123. [Google Scholar] [CrossRef]
- O’Neill, A.; Chopra, I. Preclinical evaluation of novel antibacterial agents by microbiological and molecular techniques. Expert Opin. Investig. Drugs. 2004, 13, 1045–1063. [Google Scholar] [CrossRef] [PubMed]
- Mehra, P.N.; Palta, H.K. In vitro controlled differentiation of the root callus of Cyclosorus dentatus. Phytomorphology 1971, 21, 367–375. [Google Scholar]
- Cheema, H. K In vitro studies on callus induction and differentiation of gametophytes and sporophytes in ferns. In National Seminar on Progress in Botanical Research; Verna, S.C., Sareen, T.S., Eds.; Penjab University: Chandigarh, India, 1983; pp. 44–46. [Google Scholar]
- Hedge, S.; Menon, V.K.; Noronha, R.D.; D’Souza, L. Callus culture and unconventional pattern of sporophyte regeneration in Drynaria quercifolia—A medicinal fern. In Vitro Cell. Dev. Biol. Plant 2006, 42, 508–513. [Google Scholar] [CrossRef]
- Kretzschmar, F.S.; Oliveira, C.J.F., Jr.; Marcia, R.; Brag, M.R. Differential sugar uptake by cell suspension cultures of Rudgea jasminoides, a tropical woody Rubiaceae. In Vitro Cell. Dev. Biol. Plant 2007, 43, 71–78. [Google Scholar] [CrossRef]
- Prakash, P.; Kumar, R.W.; Joy, I.V.; Thorpe, T.A. Ethylene and carbon dioxide accumulation, and growth of cell suspension cultures of Picea glauca (white sprouse). J. Plant Physiol. 1990, 135, 592–596. [Google Scholar]
- Doležal, J.; Binarová, P.; Lucretti, S. Analysis of nuclear DNA content in plant cells by flow cytometry. Biol. Plant. 1989, 231, 113–120. [Google Scholar]
- Karlsson, S.B.; Vasil, I.K. Growth, cytology and flow cytometry of embryogenic cell suspension cultures of Panicum maximum Jacq. and Pennisetum purpureum Schum. J. Plant Physiol. 1986, 123, 211–227. [Google Scholar] [CrossRef]
- Brillouet, J.M.; Romieu, C.; Schoefs, B.; Solymosi, K.; Cheynier, V.; Fulcrand, H.; Vertdeil, J.L.; Conéjéro, G. The tannosome is an organelle forming condensed tannins in the chlorophyllous organs of Tracheophyta. Ann. Bot. 2013, 112, 1003–1014. [Google Scholar] [CrossRef]
- Park, J.S.; Rho, H.S.; Kim, D.H.; Chang, I.S. Enzymatic preparation of kaempferol from green tea seed and its antioxidant activity. J. Agric. Food Chem. 2006, 54, 2951–2956. [Google Scholar] [CrossRef]
- Popielarska-Konieczna, M.; Kozieradzka-Kiszkurno, M.; Świerczyńska, J.; Góralski, H.; Ślesak, G.; Bohdanowicz, J. Ultrastructure and histochemical analysis of extracellular matrix surface network in kiwifruit endosperm-derived callus culture. Plant Cell Rep. 2008, 27, 1137–1145. [Google Scholar] [CrossRef]
- Konieczny, R.; Bohdanowicz, J.; Czaplicki, A.Z.; Przywara, L. Extracellular matrix surface network during plant regeneration in wheat anther culture. Plant Cell Tiss. Org. Cult. 2005, 83, 201–208. [Google Scholar] [CrossRef]
- Joyce, B.L.; Eda, S.; Dunlap, J.; Steward, C.N., Jr. Morphology and ploidy level determination of Pteris vittata callus during induction and regeneration. BMC Biotechnol. 2014, 14, 96. [Google Scholar] [CrossRef] [PubMed]
- Xia, X.; Cao, J.; Zheng, Y.; Wang, Q.; Xiao, J. Flavonid concentrations and bioactivity of flavonoid extracts from 19 species of ferns from China. Ind. Crops Prod. 2014, 58, 91–98. [Google Scholar] [CrossRef]
- Winked-Shirley, B. Biosynthesis of flavonoids and effects of stress. Curr. Opin. Plant Biol. 2002, 5, 218–223. [Google Scholar] [CrossRef]
- Bharii, M. Studies on phytochemical analysis and screening for active compounds in some ferns of Ranchi and Latehar districts. Int. J. Acad. Res. Dev. 2018, 3, 33. [Google Scholar]
- Umiokalsom, Y.; Harborne, J.B. Flavonoid distribution in Aslpenioid ferns. Partanika 1991, 14, 297–300. [Google Scholar]
- Baek, N.-I.; Kennelly, E.J.; Kardono, L.B.S.; Tsauri, S.; Padmawinata, K.; Soejarto, D.D.; Kinghorn, A.D. Flavonods and a proanthrocyanidin from rhizomes of Selliguea feei. Phytochemistry 1994, 36, 513–518. [Google Scholar] [CrossRef]
- Slanina, J.; Táborská, E.; Bochořáková, H.; Slaninová, I.; Humpa, R.; Robinson, U.E.; Schram, K.H. New and facile method of preparation of the anti-HIV-1 agent, 1,3-dicaffeoylquinic acid. Tetrahedron Lett. 2001, 42, 3383–3385. [Google Scholar] [CrossRef]
- Cao, H.; Chai, T.T.; Wang, X.; Morais-Braga, M.F.B.; Yang, J.H.; Wong, F.C.; Wang, R.; Yao, H.; Cao, J.; Cornara, L.; et al. Phytochemicals from fern species: Potential for medicinal application. Phytochem. Rev. 2017, 16, 379–440. [Google Scholar] [CrossRef]
- Lai, H.Y.; Lim, Y.Y. Evaluation of antioxidant activities of the methanolic extracts of selected ferns in Malaysia. Int. J. Environ. Sci. Dev. 2011, 2, 442–447. [Google Scholar] [CrossRef]
- Nath, K.; Talukdar, A.D.; Bhattacharya, M.K.; Bhowmik, D.; Chetri, S.; Choudhury, D.; Mitra, A.; Bhattachajee, A. Antibacterial activity of certain ferns against multi drag resistant organisms. J. Nat. Rem. 2017, 17, 144–153. [Google Scholar]
- Banerjee, P.D.; Sen, S.P. Antybiotic activity of Pteridophytes. Econ. Bot. 1980, 34, 284–298. [Google Scholar] [CrossRef]
- Longtine, C.; Tejedor, A. Antimicrobial activity of ethanolic and aqueous extracts of medicinally used tree ferns Alsophila cuspidata and Cyathea microdonta. Acta Bot. Malacit. 2017, 42, 119–123. [Google Scholar] [CrossRef]
- Rohini, S.M. Evaluation of antimicrobial and anti-inflammatory activities of n-hexane fraction of Cyathea nilgirensis Holttum. Int. J. Pharm. Bio Sci. 2016, 7, 748–755. [Google Scholar]
- Nath, K.; Talukdar, A.D.; Bhattacharya, M.K.; Bhowmik, D.; Chetri, S.; Choudhury, D.; Mitra, A.; Choudhury, N.A. Cyathea gigantea (Cyatheaceae) as an antimicrobial agent against multidrug resistant organisms. BMC Complement. Altern. Med. 2019, 19, 279. [Google Scholar] [CrossRef] [PubMed]
- Faizal, A.; Taufik, I.; Rachmani, A.F.; Azar, A.W.P. Antioxidant and antibacterial properties of tree fern Cyathea contaminans. Biodiversitas J. Biol. Divers. 2020, 21, 2201–2205. [Google Scholar]






| Sample No. | Time of Cultivation (Days) | Amount of ETH (ppm) | Amount of ETH per mg FW | Amount of ETH per mg DW | Amount ETH per mg of ASH |
|---|---|---|---|---|---|
| 1. | 3 | 5.64 ± 0.00 | 1.77 ± 0.05 a | 17.80 ± 0.40 a | 0.0035 ± 0.0002 a |
| 2. | 6 | 6.67 ± 0.00 a | 1.34 ± 0.04 b | 17.88 ± 3.12 a,b | 0.0042 ± 0.0001 b |
| 3. | 9 | 5.34 ± 0.00 | 1.63 ± 0.04 a,b,c | 16.02 ± 4.39 a,b,c | 0.0046 ± 0.0003 c |
| 4. | 12 | 5.56 ± 0.00 | 0.53 ± 0.02 c | 6.02 ± 0.29 a | 0.0096 ± 0.0007 |
| 5. | 15 | 6.68 ± 0.00 a | 1.51 ± 0.48 a,b,c | 23.30 ± 6.85 a,b,c | 0.0044 ± 0.0016 a,b,c |
| Sample No. | Initial Weight (g) | Amount of ETH (ppm) | Amount of ETH per mg FW | Amount of ETH per mg DW | Amount ETH per mg of ASH |
|---|---|---|---|---|---|
| 1. | 0.5 | 5.25 ± 0.00 | 7.05 ± 0.25 | 58.00 ± 3.05 | 0.0006 ± 0.0001 |
| 2. | 1.0 | 5.46 ± 0.00 | 3.25 ± 0.04 | 26.05 ± 0.68 | 0.0021 ± 0.0001 |
| 3. | 1.5 | 5.51 ± 0.00 | 2.20 ± 0.10 | 16.50 ± 0.95 a | 0.0031 ± 0.0001 |
| 4. | 2.0 | 5.53 ± 0.00 | 1.20 ± 0.04 | 12.50 ± 0.31 a | 0.0045 ± 0.0002 |
| 5. | 3.0 | 4.55 ± 0.00 | 0.61 ± 0.48 | 10.25 ± 4.68 a | 0.0083 ± 0.0004 |
| Sample | Staphylo coccus aureus | S. epidermidis | Escherichia coli | Pseudo monas aeruginosa | S. mutans | Propioni bacterium acnes PCM 2400 | P. acnes PCM 2334 | S. sanguinis PCM 2335 |
|---|---|---|---|---|---|---|---|---|
| Aerobic Gram-Positive | Aerobic Gram-Negative | Microaerobic Gram-Positive | ||||||
| Standards | ||||||||
| quercetin | 0 | 0 | 0 | 0 | 0 | 0 | 4 | 0 |
| luteolin | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| gallic acid | 15 | 36 | 12 | 10 | 16 | 20 | 22 | 18 |
| p-coumaric acid | 0 | 0 | 8 | 6 | 0 | 0 | 0 | 0 |
| caffeic acid | 0 | 28 | 4 | 4 | 0 | 0 | 0 | 0 |
| vanillic acid | 0 | 0 | 4 | 4 | 0 | 0 | 0 | 0 |
| Samples of Cyathea smithii | ||||||||
| In vitro * | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Green house ** | 0 | 0 | 0 | 0 | 0 | 6 | 6 | 0 |
| Cell suspension *** | 15 | 10 | 40 | 36 | 19 | 18 | 16 | 22 |
| Sample | S. aureus ATCC 25923 | MBC/MIC | S. epidermidis ATCC 12228 | MBC/MIC | E. coli ATCC 25992 | MBC/MIC | P. aeruginosa ATCC 27853 | MBC/MIC | S. mutans PCM 2502 | MBC/MIC | P. acnes PCM 2400 | MBC/MIC | P. acnes PCM 2334 | MBC/MIC | S. sanguinis PCM 2335 | MBC/MIC |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Cell * | 62.5 | 8 | 125 | 16 | 7.8 | 16 | 62.5 | 8 | 62.5 | 4 | 62.5 | 4 | 62.5 | 4 | 15.6 | 2 |
| Gallic acid | 125 | 16 | 31.25 | 8 | 500 | 8 | 1000 | - | 125 | 8 | 62.5 | 8 | 62.5 | 8 | 62.5 | 8 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rybczyński, J.J.; Kaźmierczak, A.; Dos Santos Szewczyk, K.; Tomaszewicz, W.; Miazga-Karska, M.; Mikuła, A. Biotechnology of the Tree Fern Cyathea smithii (J.D. Hooker; Soft Tree Fern, Katote) II Cell Suspension Culture: Focusing on Structure and Physiology in the Presence of 2,4-D and BAP. Cells 2022, 11, 1396. https://doi.org/10.3390/cells11091396
Rybczyński JJ, Kaźmierczak A, Dos Santos Szewczyk K, Tomaszewicz W, Miazga-Karska M, Mikuła A. Biotechnology of the Tree Fern Cyathea smithii (J.D. Hooker; Soft Tree Fern, Katote) II Cell Suspension Culture: Focusing on Structure and Physiology in the Presence of 2,4-D and BAP. Cells. 2022; 11(9):1396. https://doi.org/10.3390/cells11091396
Chicago/Turabian StyleRybczyński, Jan J., Andrzej Kaźmierczak, Katarzyna Dos Santos Szewczyk, Wojciech Tomaszewicz, Małgorzata Miazga-Karska, and Anna Mikuła. 2022. "Biotechnology of the Tree Fern Cyathea smithii (J.D. Hooker; Soft Tree Fern, Katote) II Cell Suspension Culture: Focusing on Structure and Physiology in the Presence of 2,4-D and BAP" Cells 11, no. 9: 1396. https://doi.org/10.3390/cells11091396
APA StyleRybczyński, J. J., Kaźmierczak, A., Dos Santos Szewczyk, K., Tomaszewicz, W., Miazga-Karska, M., & Mikuła, A. (2022). Biotechnology of the Tree Fern Cyathea smithii (J.D. Hooker; Soft Tree Fern, Katote) II Cell Suspension Culture: Focusing on Structure and Physiology in the Presence of 2,4-D and BAP. Cells, 11(9), 1396. https://doi.org/10.3390/cells11091396






