How Cargo Identity Alters the Uptake of Cell-Penetrating Peptide (CPP)/Cargo Complexes: A Study on the Effect of Net Cargo Charge and Length
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals and Reagents
2.2. Peptide Synthesis and Purification
2.3. Circular Dichroism
2.4. Cell Culture and Lysate Generation
2.5. Quantification of CPP and CPP/Cargo Uptake
2.6. Cell Viability Assay
2.7. Fluorescent Microscopy
3. Results and Discussion
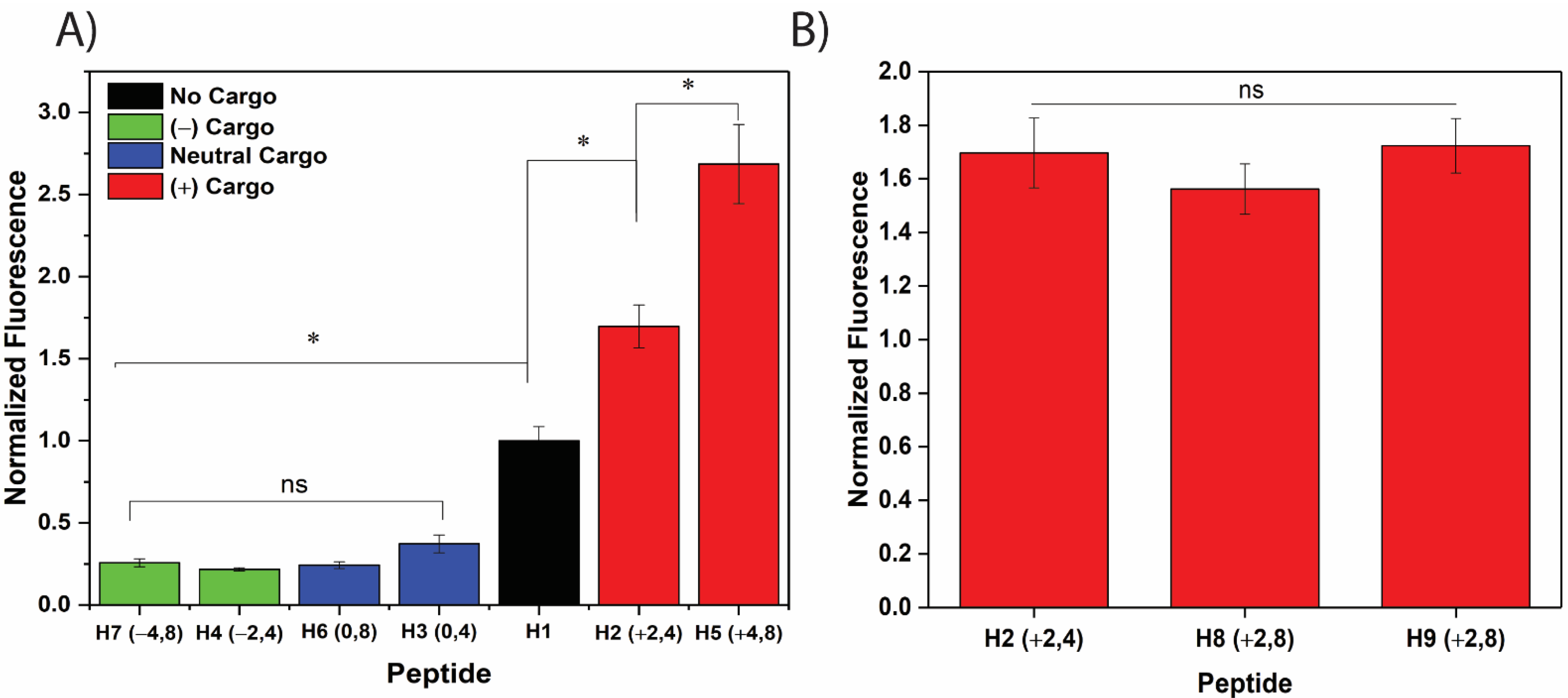
3.1. Net Positive Cargo Charge Enhances the Cellular Uptake of the CPP/Cargo Complex
3.2. Entry of the CPP/Cargo Complex into Intact Cells Does Not Significantly Diminish Cellular Viability
3.3. CPP/Cargo Complexes Exhibited a Non-Uniform Fluorescence Distribution in Intact HeLa Cells
3.4. Net Cargo Charge, but Not Net Cargo Length, Enhance CPP/Cargo Complex Uptake in Both Structured and Unstructured CPPs
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- De Jong, H.; Bonger, K.M.; Lowik, D.W.P.M. Activatable Cell-Penetrating Peptides: 15 Years of Research. RSC Chem. Biol. 2020, 1, 192–203. [Google Scholar] [CrossRef] [PubMed]
- Park, S.E.; Sajid, M.I.; Parang, K.; Tiwari, R.K. Cyclic Cell-Penetrating Peptides as Efficient Intracellular Drug Delivery Tools. Mol. Pharm. 2019, 16, 3727–3743. [Google Scholar] [CrossRef] [PubMed]
- Wang, F.; Wang, Y.; Zhang, X.; Zhang, W.; Guo, S.; Jin, F. Recent progress of cell-penetrating peptides as new carriers for intracellular cargo delivery. J. Control. Release 2014, 174, 126–136. [Google Scholar] [CrossRef]
- Borrelli, A.; Tornesello, A.L.; Tornesello, M.L.; Buonaguro, F.M. Cell Penetrating Peptides as Molecular Carriers for Anti-Cancer Agents. Molecules 2018, 23, 295. [Google Scholar] [CrossRef] [PubMed]
- Kurrikoff, K.; Vunk, B.; Langel, U. Status update in the use of cell-penetrating peptides for the delivery of macromolecular therapeutics. Expert Opin. Biol. Ther. 2021, 21, 361–370. [Google Scholar] [CrossRef]
- Dinca, A.; Chien, W.M.; Chin, M.T. Intracellular Delivery of Proteins with Cell-Penetrating Peptides for Therapeutic Uses in Human Disease. Int. J. Mol. Sci. 2016, 17, 263. [Google Scholar] [CrossRef]
- Liao, H.W.; Li, X.; Zhao, L.Z.; Wang, Y.L.; Wang, X.D.; Wu, Y.; Zhou, X.; Fu, W.; Liu, L.; Hu, H.G.; et al. A PROTAC peptide induces durable beta-catenin degradation and suppresses Wnt-dependent intestinal cancer. Cell Discov. 2020, 6, 1–12. [Google Scholar] [CrossRef]
- McCarthy, H.O.; McCaffrey, J.; McCrudden, C.M.; Zholobenko, A.; Ali, A.A.; McBride, J.W.; Massey, A.S.; Pentlavalli, S.; Chen, K.H.; Cole, G.; et al. Development and characterization of self-assembling nanoparticles using a bio-inspired amphipathic peptide for gene delivery. J. Control. Release 2014, 189, 141–149. [Google Scholar] [CrossRef]
- Perillo, E.; Herve-Aubert, K.; Allard-Vannier, E.; Falanga, A.; Galdiero, S.; Chourpa, I. Synthesis and in vitro evaluation of fluorescent and magnetic nanoparticles functionalized with a cell penetrating peptide for cancer theranosis. J. Colloid Interface Sci. 2017, 499, 209–217. [Google Scholar] [CrossRef]
- Green, M.; Loewenstein, P.M. Autonomous functional domains of chemically synthesized human immunodeficiency virus tat trans-activator protein. Cell 1988, 55, 1179–1188. [Google Scholar] [CrossRef]
- Viscidi, R.P.; Mayur, K.; Lederman, H.M.; Frankel, A.D. Inhibition of antigen-induced lymphocyte proliferation by Tat protein from HIV-1. Science 1989, 246, 1606–1608. [Google Scholar] [CrossRef]
- Joliot, A.; Pernelle, C.; Deagostinibazin, H.; Prochiantz, A. Antennapedia Homeobox Peptide Regulates Neural Morphogenesis. Proc. Natl. Acad. Sci. USA 1991, 88, 1864–1868. [Google Scholar] [CrossRef]
- Derossi, D.; Joliot, A.H.; Chassaing, G.; Prochiantz, A. The 3rd Helix of the Antennapedia Homeodomain Translocates through Biological-Membranes. J. Biol. Chem. 1994, 269, 10444–10450. [Google Scholar] [CrossRef]
- Vives, E.; Brodin, P.; Lebleu, B. A truncated HIV-1 Tat protein basic domain rapidly translocates through the plasma membrane and accumulates in the cell nucleus. J. Biol. Chem. 1997, 272, 16010–16017. [Google Scholar] [CrossRef] [PubMed]
- Bohmova, E.; Machova, D.; Pechar, M.; Pola, R.; Venclikova, K.; Janouskova, O.; Etrych, T. Cell-penetrating peptides: A useful tool for the delivery of various cargoes into cells. Physiol. Res. 2018, 67, S267–S279. [Google Scholar] [CrossRef]
- Elmquist, A.; Hansen, M.; Langel, Ü. Structure–activity relationship study of the cell-penetrating peptide pVEC. Biochim. Biophys. Acta (BBA) Biomembr. 2006, 1758, 721–729. [Google Scholar] [CrossRef] [PubMed]
- Morris, M.C.; Deshayes, S.; Heitz, F.; Divita, G. Cell-penetrating peptides: From molecular mechanisms to therapeutics. Biol. Cell 2008, 100, 201–217. [Google Scholar] [CrossRef]
- Vaithiyanathan, M.; Hymel, H.C.; Safa, N.; Sanchez, O.M.; Pettigrew, J.H.; Kirkpatrick, C.S.; Gauthier, T.J.; Melvin, A.T. Kinetic analysis of cellular internalization and expulsion of unstructured D-chirality cell penetrating peptides. Aiche J. 2021, 67, e17087. [Google Scholar] [CrossRef]
- Hanna, S.E.; Mozaffari, S.; Tiwari, R.K.; Parang, K. Comparative Molecular Transporter Efficiency of Cyclic Peptides Containing Tryptophan and Arginine Residues. ACS Omega 2018, 3, 16281–16291. [Google Scholar] [CrossRef]
- Qian, Z.; Martyna, A.; Hard, R.L.; Wang, J.; Appiah-Kubi, G.; Coss, C.; Phelps, M.A.; Rossman, J.S.; Pei, D. Discovery and Mechanism of Highly Efficient Cyclic Cell-Penetrating Peptides. Biochemistry 2016, 55, 2601–2612. [Google Scholar] [CrossRef]
- Moiola, M.; Memeo, M.G.; Quadrelli, P. Stapled Peptides-A Useful Improvement for Peptide-Based Drugs. Molecules 2019, 24, 3654. [Google Scholar] [CrossRef] [PubMed]
- Cline, L.L.; Waters, M.L. The structure of well-folded beta-hairpin peptides promotes resistance to peptidase degradation. Biopolymers 2009, 92, 502–507. [Google Scholar] [CrossRef] [PubMed]
- Safa, N.; Anderson, J.C.; Vaithiyanathan, M.; Pettigrew, J.H.; Pappas, G.A.; Liu, D.; Gauthier, T.J.; Melvin, A.T. CPProtectides: Rapid uptake of well-folded beta-hairpin peptides with enhanced resistance to intracellular degradation. Pept. Sci. 2019, 111, e24092. [Google Scholar] [CrossRef] [PubMed]
- Jin, J.M.; Wu, Y.; Chen, J.J.; Shen, Y.W.; Zhang, L.J.; Zhang, H.; Chen, L.L.; Yuan, H.B.; Chen, H.Z.; Zhang, W.D.; et al. The peptide PROTAC modality: A novel strategy for targeted protein ubiquitination. Theranostics 2020, 10, 10141–10153. [Google Scholar] [CrossRef] [PubMed]
- Ottis, P.; Crews, C.M. Proteolysis-Targeting Chimeras: Induced Protein Degradation as a Therapeutic Strategy. ACS Chem. Biol. 2017, 12, 892–898. [Google Scholar] [CrossRef]
- Pazos, E.; Vazquez, O.; Mascarenas, J.L.; Vazquez, M.E. Peptide-based fluorescent biosensors. Chem. Soc. Rev. 2009, 38, 3348–3359. [Google Scholar] [CrossRef]
- Safa, N.; Vaithiyanathan, M.; Sombolestani, S.; Charles, S.; Melvin, A.T. Population-based analysis of cell-penetrating peptide uptake using a microfluidic droplet trapping array. Anal. Bioanal. Chem. 2019, 411, 2729–2741. [Google Scholar] [CrossRef]
- Hallbrink, M.; Floren, A.; Elmquist, A.; Pooga, M.; Bartfai, T.; Langel, U. Cargo delivery kinetics of cell-penetrating peptides. Biochim. Biophys. Acta 2001, 1515, 101–109. [Google Scholar] [CrossRef]
- Jones, S.W.; Christison, R.; Bundell, K.; Voyce, C.J.; Brockbank, S.M.; Newham, P.; Lindsay, M.A. Characterisation of cell-penetrating peptide-mediated peptide delivery. Br. J. Pharmacol. 2005, 145, 1093–1102. [Google Scholar] [CrossRef]
- Patel, S.G.; Sayers, E.J.; He, L.; Narayan, R.; Williams, T.L.; Mills, E.M.; Allemann, R.K.; Luk, L.Y.P.; Jones, A.T.; Tsai, Y.H. Cell-penetrating peptide sequence and modification dependent uptake and subcellular distribution of green florescent protein in different cell lines. Sci. Rep. 2019, 9, 6298. [Google Scholar] [CrossRef]
- Khayyatnejad Shoushtari, S.; Zoghebi, K.; Sajid, M.I.; Tiwari, R.K.; Parang, K. Hybrid Cyclic-Linear Cell-Penetrating Peptides Containing Alternative Positively Charged and Hydrophobic Residues as Molecular Transporters. Mol. Pharm. 2021, 18, 3909–3919. [Google Scholar] [CrossRef] [PubMed]
- Futaki, S.; Suzuki, T.; Ohashi, W.; Yagami, T.; Tanaka, S.; Ueda, K.; Sugiura, Y. Arginine-rich peptides. An abundant source of membrane-permeable peptides having potential as carriers for intracellular protein delivery. J. Biol. Chem. 2001, 276, 5836–5840. [Google Scholar] [CrossRef] [PubMed]
- Tunnemann, G.; Ter-Avetisyan, G.; Martin, R.M.; Stockl, M.; Herrmann, A.; Cardoso, M.C. Live-cell analysis of cell penetration ability and toxicity of oligo-arginines. J. Pept. Sci. 2008, 14, 469–476. [Google Scholar] [CrossRef] [PubMed]
- Zachowski, A. Phospholipids in animal eukaryotic membranes: Transverse asymmetry and movement. Biochem. J. 1993, 294 Pt 1, 1. [Google Scholar] [CrossRef]
- Utsugi, T.; Schroit, A.J.; Connor, J.; Bucana, C.D.; Fidler, I.J. Elevated expression of phosphatidylserine in the outer membrane leaflet of human tumor cells and recognition by activated human blood monocytes. Cancer Res. 1991, 51, 3062–3066. [Google Scholar] [PubMed]
- Amand, H.L.; Fant, K.; Nordén, B.; Esbjörner, E.K. Stimulated endocytosis in penetratin uptake: Effect of arginine and lysine. Biochem. Biophys. Res. Commun. 2008, 371, 621–625. [Google Scholar] [CrossRef]
- El-Sayed, N.S.; Miyake, T.; Shirazi, A.N.; Park, S.E.; Clark, J.; Buchholz, S.; Parang, K.; Tiwari, R. Design, Synthesis, and Evaluation of Homochiral Peptides Containing Arginine and Histidine as Molecular Transporters. Molecules 2018, 23, 1590. [Google Scholar] [CrossRef]
- Su, Y.; Doherty, T.; Waring, A.J.; Ruchala, P.; Hong, M. Roles of arginine and lysine residues in the translocation of a cell-penetrating peptide from (13)C, (31)P, and (19)F solid-state NMR. Biochemistry 2009, 48, 4587–4595. [Google Scholar] [CrossRef]
- Fei, L.; Ren, L.; Zaro, J.L.; Shen, W.C. The influence of net charge and charge distribution on cellular uptake and cytosolic localization of arginine-rich peptides. J. Drug Target. 2011, 19, 675–680. [Google Scholar] [CrossRef]
- Sun, C.; Shen, W.-C.; Tu, J.; Zaro, J.L. Interaction between Cell-Penetrating Peptides and Acid-Sensitive Anionic Oligopeptides as a Model for the Design of Targeted Drug Carriers. Mol. Pharm. 2014, 11, 1583–1590. [Google Scholar] [CrossRef]
- Fujita, H.; Yamagishi, M.; Kida, Y.; Sakaguchi, M. Positive charges on the translocating polypeptide chain arrest movement through the translocon. J. Cell Sci. 2011, 124, 4184–4193. [Google Scholar] [CrossRef]
- El-Andaloussi, S.; Jarver, P.; Johansson, H.J.; Langel, U. Cargo-dependent cytotoxicity and delivery efficacy of cell-penetrating peptides: A comparative study. Biochem. J. 2007, 407, 285–292. [Google Scholar] [CrossRef] [PubMed]
- Cardozo, A.K.; Buchillier, V.; Mathieu, M.; Chen, J.; Ortis, F.; Ladriere, L.; Allaman-Pillet, N.; Poirot, O.; Kellenberger, S.; Beckmann, J.S.; et al. Cell-permeable peptides induce dose- and length-dependent cytotoxic effects. Biochim. Biophys. Acta 2007, 1768, 2222–2234. [Google Scholar] [CrossRef]
- Deprey, K.; Becker, L.; Kritzer, J.; Pluckthun, A. Trapped! A Critical Evaluation of Methods for Measuring Total Cellular Uptake versus Cytosolic Localization. Bioconjug. Chem. 2019, 30, 1006–1027. [Google Scholar] [CrossRef]
- Kovarik, M.L.; Allbritton, N.L. Measuring enzyme activity in single cells. Trends Biotechnol. 2011, 29, 222–230. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Proctor, A.; Wang, Q.; Lawrence, D.S.; Allbritton, N.L. Metabolism of peptide reporters in cell lysates and single cells. Analyst 2012, 137, 3028–3038. [Google Scholar] [CrossRef] [PubMed]
- Kristensen, M.; Birch, D.; Mørck Nielsen, H. Applications and Challenges afor Use of Cell-Penetrating Peptides as Delivery Vectors for Peptide and Protein Cargos. Int. J. Mol. Sci. 2016, 17, 185. [Google Scholar] [CrossRef] [PubMed]




| Name | Sequence | Cargo Charge | Cargo Length |
|---|---|---|---|
| H1 | RWVRVpGO(FAM)WIRQ | ||
| H2 | RWVRVpGO(FAM)WIRQ-GRGR | +2 | 4-mer |
| H3 | RWVRVpGO(FAM)WIRQ-GGGG | 0 | 4-mer |
| H4 | RWVRVpGO(FAM)WIRQ-GEGE | −2 | 4-mer |
| H5 | RWVRVpGO(FAM)WIRQ-GRGRGRGR | +4 | 8-mer |
| H6 | RWVRVpGO(FAM)WIRQ-GGGGGGGG | 0 | 8-mer |
| H7 | RWVRVpGO(FAM)WIRQ-GEGEGEGE | −4 | 8-mer |
| H8 | RWVRVpGO(FAM)WIRQ-GGGGGRGR | +2 | 8-mer |
| H9 | RWVRVpGO(FAM)WIRQ-GRGRGGGG | +2 | 8-mer |
| R1 | FAM-RRRRRRRRR | ||
| R2 | FAM-RRRRRRRRR-GRGR | +2 | 4-mer |
| R3 | FAM-RRRRRRRRR-GRGRGRGR | +4 | 8-mer |
| R4 | FAM-RRRRRRRRR-GGGGGGGG | 0 | 8-mer |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hymel, H.C.; Rahnama, A.; Sanchez, O.M.; Liu, D.; Gauthier, T.J.; Melvin, A.T. How Cargo Identity Alters the Uptake of Cell-Penetrating Peptide (CPP)/Cargo Complexes: A Study on the Effect of Net Cargo Charge and Length. Cells 2022, 11, 1195. https://doi.org/10.3390/cells11071195
Hymel HC, Rahnama A, Sanchez OM, Liu D, Gauthier TJ, Melvin AT. How Cargo Identity Alters the Uptake of Cell-Penetrating Peptide (CPP)/Cargo Complexes: A Study on the Effect of Net Cargo Charge and Length. Cells. 2022; 11(7):1195. https://doi.org/10.3390/cells11071195
Chicago/Turabian StyleHymel, Hannah C., Alireza Rahnama, Olivia M. Sanchez, Dong Liu, Ted J. Gauthier, and Adam T. Melvin. 2022. "How Cargo Identity Alters the Uptake of Cell-Penetrating Peptide (CPP)/Cargo Complexes: A Study on the Effect of Net Cargo Charge and Length" Cells 11, no. 7: 1195. https://doi.org/10.3390/cells11071195
APA StyleHymel, H. C., Rahnama, A., Sanchez, O. M., Liu, D., Gauthier, T. J., & Melvin, A. T. (2022). How Cargo Identity Alters the Uptake of Cell-Penetrating Peptide (CPP)/Cargo Complexes: A Study on the Effect of Net Cargo Charge and Length. Cells, 11(7), 1195. https://doi.org/10.3390/cells11071195






