Hematopoietic Cells from Pluripotent Stem Cells: Hope and Promise for the Treatment of Inherited Blood Disorders
Abstract
1. Introduction
2. Genetic and Genomic Hematologic Diseases
3. Innovative Cell Therapy Approaches for Inherited Blood Disorders
3.1. Pluripotent Stem Cells
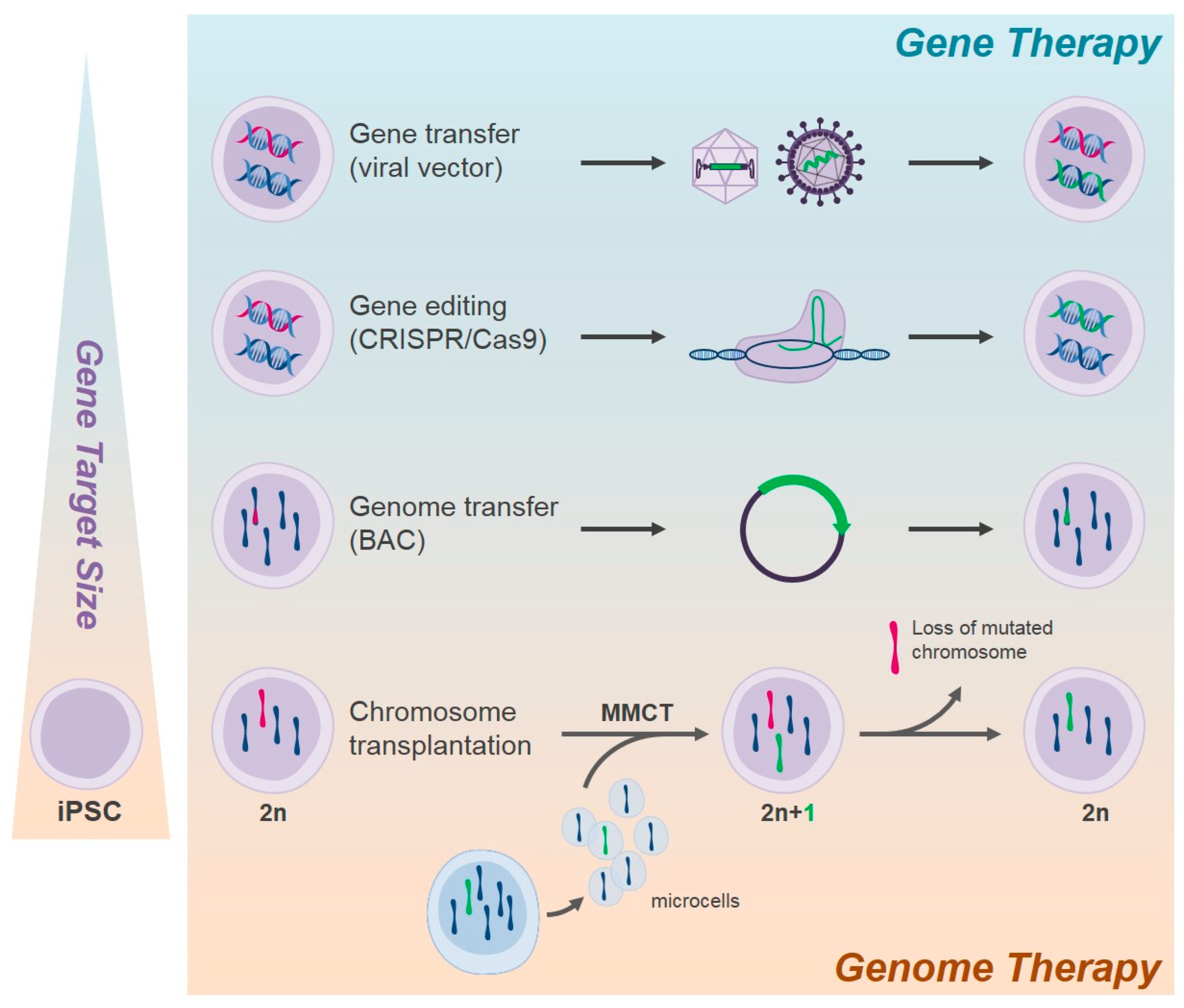
3.2. Gene Therapy of Pluripotent Stem Cells
3.3. iPSC Biobanks
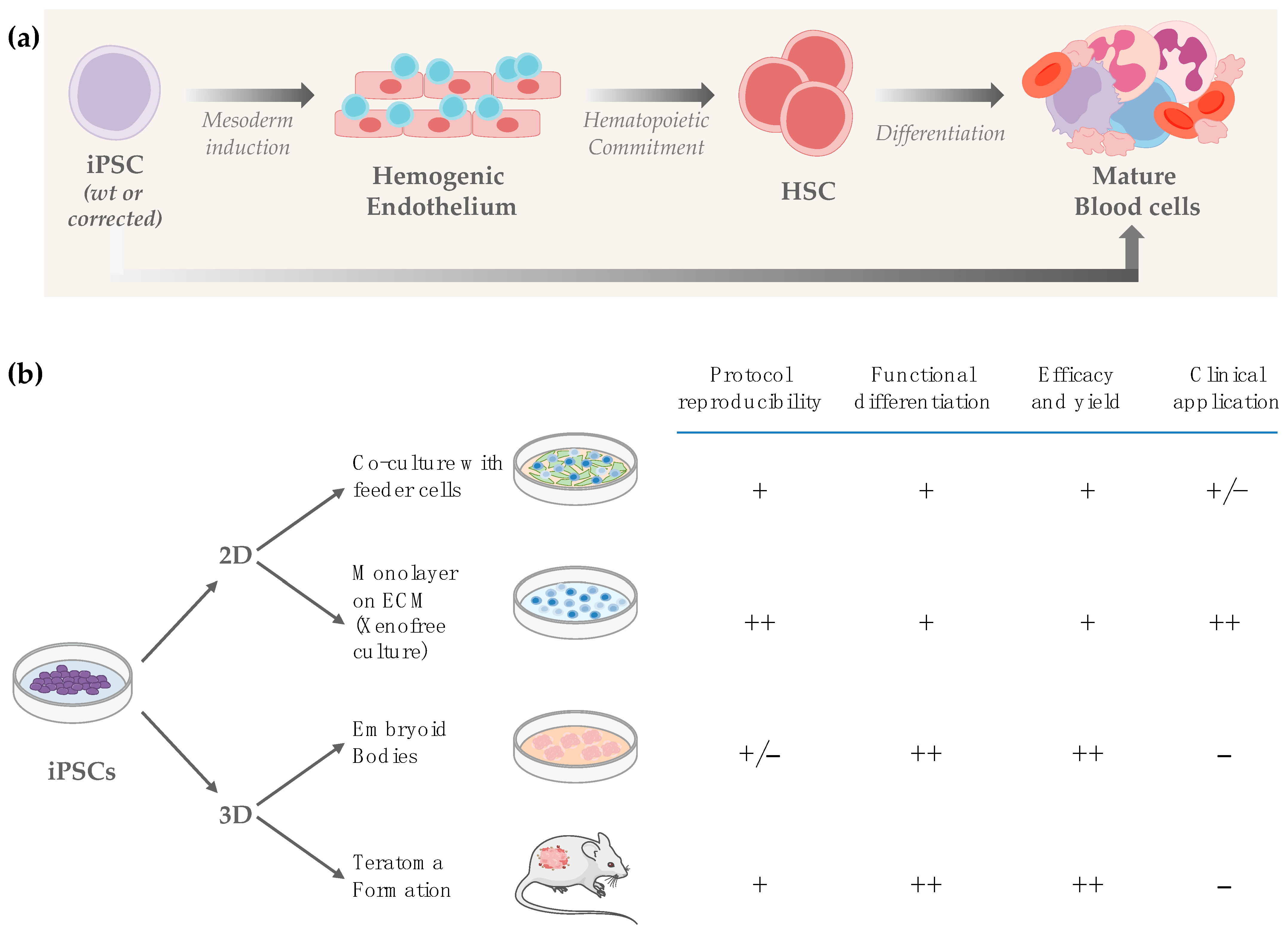
4. Strategies to Generate Hematopoietic Stem and Progenitor Cells from Pluripotent Stem Cells
4.1. Two-Dimensional (2D) In Vitro Differentiation
4.2. Three-Dimensional (3D) In Vitro Differentiation
4.3. In Vivo Approaches
5. Generation of Terminally Differentiated Hematopoietic Cells for Therapeutic Purposes
5.1. Platelets
5.2. Erythrocytes
5.3. Osteoclasts
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
Abbreviations
| Pt | patient |
| Ery | erythroid |
| PL | platelets lysate |
| Hematop. | Hematopoietic |
| BFU-E | burst-forming unit-Ery |
| Diff | differentiation |
| Plt | platelets |
| HC | hematopoietic cells |
| Prog | progenitors |
References
- Cavazzana, M.; Antoniani, C.; Miccio, A. Gene Therapy for β-Hemoglobinopathies. Mol. Ther. 2017, 25, 1142–1154. [Google Scholar] [CrossRef]
- Amaya-Uribe, L.; Rojas, M.; Azizi, G.; Anaya, J.M.; Gershwin, M.E. Primary immunodeficiency and autoimmunity: A comprehensive review. J. Autoimmun. 2019, 99, 52–72. [Google Scholar] [CrossRef] [PubMed]
- Firth, H.V.; Richards, S.M.; Bevan, A.P.; Clayton, S.; Corpas, M.; Rajan, D.; Van Vooren, S.; Moreau, Y.; Pettett, R.M.; Carter, N.P. DECIPHER: Database of Chromosomal Imbalance and Phenotype in Humans Using Ensembl Resources. Am. J. Hum. Genet. 2009, 84, 524–533. [Google Scholar] [CrossRef]
- Castagnoli, R.; Delmonte, O.M.; Calzoni, E.; Notarangelo, L.D. Hematopoietic Stem Cell Transplantation in Primary Immunodeficiency Diseases: Current Status and Future Perspectives. Front. Pediatr. 2019, 7, 295. [Google Scholar] [CrossRef] [PubMed]
- Ferrari, G.; Thrasher, A.J.; Aiuti, A. Gene therapy using haematopoietic stem and progenitor cells. Nat. Rev. Genet. 2021, 22, 216–234. [Google Scholar] [CrossRef] [PubMed]
- Demirci, S.; Leonard, A.; Tisdale, J.F. Hematopoietic stem cells from pluripotent stem cells: Clinical potential, challenges, and future perspectives. Stem Cells Transl. Med. 2020, 9, 1549–1557. [Google Scholar] [CrossRef]
- Ebrahimi, M.; Forouzesh, M.; Raoufi, S.; Ramazii, M.; Ghaedrahmati, F.; Farzaneh, M. Differentiation of human induced pluripotent stem cells into erythroid cells. Stem Cell Res. Ther. 2020, 11, 483. [Google Scholar] [CrossRef] [PubMed]
- Evans, A.L.; Dalby, A.; Foster, H.R.; Howard, D.; Waller, A.K.; Taimoor, M.; Lawrence, M.; Mookerjee, S.; Lehmann, M.; Burton, A.; et al. Transfer to the clinic: Refining forward programming of hPSCs to megakaryocytes for platelet production in bioreactors. Blood Adv. 2021, 5, 1977–1990. [Google Scholar] [CrossRef] [PubMed]
- Chang, C.J.; Bouhassira, E.E. Zinc-finger nuclease-mediated correction of α-thalassemia in iPS cells. Blood 2012, 120, 3906–3914. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.; Wang, Y.; Yan, W.; Smith, C.; Ye, Z.; Wang, J.; Gao, Y.; Mendelsohn, L.; Cheng, L. Production of Gene-Corrected Adult Beta Globin Protein in Human Erythrocytes Differentiated from Patient iPSCs After Genome Editing of the Sickle Point Mutation. Stem Cells 2015, 33, 1470–1479. [Google Scholar] [CrossRef] [PubMed]
- Ingrungruanglert, P.; Amarinthnukrowh, P.; Rungsiwiwut, R.; Maneesri-le Grand, S.; Sosothikul, D.; Suphapeetiporn, K.; Israsena, N.; Shotelersuk, V. Wiskott-Aldrich syndrome iPS cells produce megakaryocytes with defects in cytoskeletal rearrangement and proplatelet formation. Thromb. Haemost. 2015, 113, 792–805. [Google Scholar] [CrossRef] [PubMed]
- Menon, T.; Firth, A.L.; Scripture-Adams, D.D.; Galic, Z.; Qualls, S.J.; Gilmore, W.B.; Ke, E.; Singer, O.; Anderson, L.S.; Bornzin, A.R.; et al. Lymphoid regeneration from gene-corrected SCID-X1 subject-derived iPSCs. Cell Stem Cell 2015, 16, 367–372. [Google Scholar] [CrossRef] [PubMed]
- Park, C.Y.; Kim, D.H.; Son, J.S.; Sung, J.J.; Lee, J.; Bae, S.; Kim, J.H.; Kim, D.W.; Kim, J.S. Functional Correction of Large Factor VIII Gene Chromosomal Inversions in Hemophilia A Patient-Derived iPSCs Using CRISPR-Cas9. Cell Stem Cell 2015, 17, 213–220. [Google Scholar] [CrossRef]
- Laskowski, T.J.; Van Caeneghem, Y.; Pourebrahim, R.; Ma, C.; Ni, Z.; Garate, Z.; Crane, A.M.; Li, X.S.; Liao, W.; Gonzalez-Garay, M.; et al. Gene Correction of iPSCs from a Wiskott-Aldrich Syndrome Patient Normalizes the Lymphoid Developmental and Functional Defects. Stem Cell Rep. 2016, 7, 139–148. [Google Scholar] [CrossRef]
- Wu, Y.; Hu, Z.; Li, Z.; Pang, J.; Feng, M.; Hu, X.; Wang, X.; Lin-Peng, S.; Liu, B.; Chen, F.; et al. In situ genetic correction of F8 intron 22 inversion in hemophilia A patient-specific iPSCs. Sci. Rep. 2016, 6, 18865. [Google Scholar] [CrossRef] [PubMed]
- He, Q.; Wang, H.H.; Cheng, T.; Yuan, W.P.; Ma, Y.P.; Jiang, Y.P.; Ren, Z.H. Genetic Correction and Hepatic Differentiation of Hemophilia B-specific Human Induced Pluripotent Stem Cells. Chin. Med. Sci. J. 2017, 32, 135–144. [Google Scholar] [PubMed]
- Lyu, C.; Shen, J.; Wang, R.; Gu, H.; Zhang, J.; Xue, F.; Liu, X.; Liu, W.; Fu, R.; Zhang, L.; et al. Targeted genome engineering in human induced pluripotent stem cells from patients with hemophilia B using the CRISPR-Cas9 system. Stem Cell Res. Ther. 2018, 9, 92. [Google Scholar] [CrossRef]
- Olgasi, C.; Talmon, M.; Merlin, S.; Cucci, A.; Richaud-Patin, Y.; Ranaldo, G.; Colangelo, D.; Di Scipio, F.; Berta, G.N.; Borsotti, C.; et al. Patient-Specific iPSC-Derived Endothelial Cells Provide Long-Term Phenotypic Correction of Hemophilia, A. Stem Cell Rep. 2018, 11, 1391–1406. [Google Scholar] [CrossRef]
- Wattanapanitch, M.; Damkham, N.; Potirat, P.; Trakarnsanga, K.; Janan, M.; U-Pratya, Y.; Kheolamai, P.; Klincumhom, N.; Issaragrisil, S. One-step genetic correction of hemoglobin E/beta-thalassemia patient-derived iPSCs by the CRISPR/Cas9 system. Stem Cell Res. Ther. 2018, 9, 46. [Google Scholar] [CrossRef]
- Ma, H.; Morey, R.; O'Neil, R.C.; He, Y.; Daughtry, B.; Schultz, M.D.; Hariharan, M.; Nery, J.R.; Castanon, R.; Sabatini, K.; et al. Abnormalities in human pluripotent cells due to reprogramming mechanisms. Nature 2014, 511, 177–183. [Google Scholar] [CrossRef]
- Tan, E.P.; Li, Y.; Velasco-Herrera, M.e.C.; Yusa, K.; Bradley, A. Off-target assessment of CRISPR-Cas9 guiding RNAs in human iPS and mouse ES cells. Genesis 2015, 53, 225–236. [Google Scholar] [CrossRef]
- Paulis, M.; Castelli, A.; Susani, L.; Lizier, M.; Lagutina, I.; Focarelli, M.L.; Recordati, C.; Uva, P.; Faggioli, F.; Neri, T.; et al. Chromosome transplantation as a novel approach for correcting complex genomic disorders. Oncotarget 2015, 6, 35218–35230. [Google Scholar] [CrossRef]
- Paulis, M.; Susani, L.; Castelli, A.; Suzuki, T.; Hara, T.; Straniero, L.; Duga, S.; Strina, D.; Mantero, S.; Caldana, E.; et al. Chromosome Transplantation: A Possible Approach to Treat Human X-linked Disorders. Mol. Ther. Methods Clin. Dev. 2020, 17, 369–377. [Google Scholar] [CrossRef]
- Deuse, T.; Hu, X.; Gravina, A.; Wang, D.; Tediashvili, G.; De, C.; Thayer, W.O.; Wahl, A.; Garcia, J.V.; Reichenspurner, H.; et al. Hypoimmunogenic derivatives of induced pluripotent stem cells evade immune rejection in fully immunocompetent allogeneic recipients. Nat. Biotechnol. 2019, 37, 252–258. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.Y.; Liu, C.L.; Ting, C.Y.; Chiu, Y.T.; Cheng, Y.C.; Nicholson, M.W.; Hsieh, P.C.H. Human iPSC banking: Barriers and opportunities. J. Biomed. Sci. 2019, 26, 87. [Google Scholar] [CrossRef] [PubMed]
- Tursky, M.L.; Loi, T.H.; Artuz, C.M.; Alateeq, S.; Wolvetang, E.J.; Tao, H.; Ma, D.D.; Molloy, T.J. Direct Comparison of Four Hematopoietic Differentiation Methods from Human Induced Pluripotent Stem Cells. Stem Cell Rep. 2020, 15, 735–748. [Google Scholar] [CrossRef]
- Jackson, M.; Marks, L.; May, G.H.W.; Wilson, J.B. The genetic basis of disease. Essays Biochem. 2018, 62, 643–723. [Google Scholar] [CrossRef] [PubMed]
- Rae, J.; Newburger, P.E.; Dinauer, M.C.; Noack, D.; Hopkins, P.J.; Kuruto, R.; Curnutte, J.T. X-Linked chronic granulomatous disease: Mutations in the CYBB gene encoding the gp91-phox component of respiratory-burst oxidase. Am. J. Hum. Genet. 1998, 62, 1320–1331. [Google Scholar] [CrossRef]
- Guna, A.; Butcher, N.J.; Bassett, A.S. Comparative mapping of the 22q11.2 deletion region and the potential of simple model organisms. J. Neurodev. Disord. 2015, 7, 18. [Google Scholar] [CrossRef]
- Rosa, R.F.; Rosa, R.C.; Dos Santos, P.P.; Zen, P.R.; Paskulin, G.A. Hematological abnormalities and 22q11.2 deletion syndrome. Rev. Bras. Hematol Hemoter. 2011, 33, 151–154. [Google Scholar] [CrossRef] [PubMed]
- Nalepa, G.; Clapp, D.W. Fanconi anaemia and cancer: An intricate relationship. Nat. Rev. Cancer 2018, 18, 168–185. [Google Scholar] [CrossRef]
- Ceccaldi, R.; Sarangi, P.; D'Andrea, A.D. The Fanconi anaemia pathway: New players and new functions. Nat. Rev. Mol. Cell Biol. 2016, 17, 337–349. [Google Scholar] [CrossRef]
- Moreno, O.M.; Paredes, A.C.; Suarez-Obando, F.; Rojas, A. An update on Fanconi anemia: Clinical, cytogenetic and molecular approaches (Review). Biomed. Rep. 2021, 15, 74. [Google Scholar] [CrossRef] [PubMed]
- Morgan, N.V.; Tipping, A.J.; Joenje, H.; Mathew, C.G. High frequency of large intragenic deletions in the Fanconi anemia group A gene. Am. J. Hum. Genet. 1999, 65, 1330–1341. [Google Scholar] [CrossRef] [PubMed]
- De Rocco, D.; Bottega, R.; Cappelli, E.; Cavani, S.; Criscuolo, M.; Nicchia, E.; Corsolini, F.; Greco, C.; Borriello, A.; Svahn, J.; et al. Molecular analysis of Fanconi anemia: The experience of the Bone Marrow Failure Study Group of the Italian Association of Pediatric Onco-Hematology. Haematologica 2014, 99, 1022–1031. [Google Scholar] [CrossRef] [PubMed]
- Yeh, Y.H.; Hsieh, M.Y.; Lee, W.I.; Huang, J.L.; Chen, L.C.; Yeh, K.W.; Ou, L.S.; Yao, T.C.; Wu, C.Y.; Lin, S.J. Distinct Clinical Features and Novel Mutations in Taiwanese Patients With X-Linked Agammaglobulinemia. Front. Immunol. 2020, 11, 2001. [Google Scholar] [CrossRef]
- Sedivá, A.; Smith, C.I.; Asplund, A.C.; Hadac, J.; Janda, A.; Zeman, J.; Hansíková, H.; Dvoráková, L.; Mrázová, L.; Velbri, S.; et al. Contiguous X-chromosome deletion syndrome encompassing the BTK, TIMM8A, TAF7L, and DRP2 genes. J. Clin. Immunol. 2007, 27, 640–646. [Google Scholar] [CrossRef] [PubMed]
- Xiang, Y.; Sugimura, R. Single-Cell Approaches to Deconvolute the Development of HSCs. Cells 2021, 10, 2876. [Google Scholar] [CrossRef]
- Takahashi, K.; Yamanaka, S. Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell 2006, 126, 663–676. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, K.; Tanabe, K.; Ohnuki, M.; Narita, M.; Ichisaka, T.; Tomoda, K.; Yamanaka, S. Induction of pluripotent stem cells from adult human fibroblasts by defined factors. Cell 2007, 131, 861–872. [Google Scholar] [CrossRef]
- Liu, G.; David, B.T.; Trawczynski, M.; Fessler, R.G. Advances in Pluripotent Stem Cells: History, Mechanisms, Technologies, and Applications. Stem Cell Rev. Rep. 2020, 16, 3–32. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Zheng, C.G.; Jiang, Y.; Zhang, J.; Chen, J.; Yao, C.; Zhao, Q.; Liu, S.; Chen, K.; Du, J.; et al. Genetic correction of β-thalassemia patient-specific iPS cells and its use in improving hemoglobin production in irradiated SCID mice. Cell Res. 2012, 22, 637–648. [Google Scholar] [CrossRef]
- Tubsuwan, A.; Abed, S.; Deichmann, A.; Kardel, M.D.; Bartholomä, C.; Cheung, A.; Negre, O.; Kadri, Z.; Fucharoen, S.; von Kalle, C.; et al. Parallel assessment of globin lentiviral transfer in induced pluripotent stem cells and adult hematopoietic stem cells derived from the same transplanted β-thalassemia patient. Stem Cells 2013, 31, 1785–1794. [Google Scholar] [CrossRef] [PubMed]
- Hanna, J.; Wernig, M.; Markoulaki, S.; Sun, C.W.; Meissner, A.; Cassady, J.P.; Beard, C.; Brambrink, T.; Wu, L.C.; Townes, T.M.; et al. Treatment of sickle cell anemia mouse model with iPS cells generated from autologous skin. Science 2007, 318, 1920–1923. [Google Scholar] [CrossRef]
- Mandai, M.; Kurimoto, Y.; Takahashi, M. Autologous Induced Stem-Cell-Derived Retinal Cells for Macular Degeneration. N. Engl. J. Med. 2017, 377, 792–793. [Google Scholar] [CrossRef] [PubMed]
- Neri, T.; Muggeo, S.; Paulis, M.; Caldana, M.E.; Crisafulli, L.; Strina, D.; Focarelli, M.L.; Faggioli, F.; Recordati, C.; Scaramuzza, S.; et al. Targeted Gene Correction in Osteopetrotic-Induced Pluripotent Stem Cells for the Generation of Functional Osteoclasts. Stem Cell Rep. 2015, 5, 558–568. [Google Scholar] [CrossRef] [PubMed]
- Laugsch, M.; Rostovskaya, M.; Velychko, S.; Richter, C.; Zimmer, A.; Klink, B.; Schröck, E.; Haase, M.; Neumann, K.; Thieme, S.; et al. Functional Restoration of gp91phox-Oxidase Activity by BAC Transgenesis and Gene Targeting in X-linked Chronic Granulomatous Disease iPSCs. Mol. Ther. 2016, 24, 812–822. [Google Scholar] [CrossRef] [PubMed]
- Chang, C.W.; Lai, Y.S.; Westin, E.; Khodadadi-Jamayran, A.; Pawlik, K.M.; Lamb, L.S.; Goldman, F.D.; Townes, T.M. Modeling Human Severe Combined Immunodeficiency and Correction by CRISPR/Cas9-Enhanced Gene Targeting. Cell Rep. 2015, 12, 1668–1677. [Google Scholar] [CrossRef]
- Merling, R.K.; Sweeney, C.L.; Chu, J.; Bodansky, A.; Choi, U.; Priel, D.L.; Kuhns, D.B.; Wang, H.; Vasilevsky, S.; De Ravin, S.S.; et al. An AAVS1-targeted minigene platform for correction of iPSCs from all five types of chronic granulomatous disease. Mol. Ther. 2015, 23, 147–157. [Google Scholar] [CrossRef]
- Jalil, S.; Keskinen, T.; Maldonado, R.; Sokka, J.; Trokovic, R.; Otonkoski, T.; Wartiovaara, K. Simultaneous high-efficiency base editing and reprogramming of patient fibroblasts. Stem Cell Rep. 2021, 16, 3064–3075. [Google Scholar] [CrossRef]
- Dever, D.P.; Bak, R.O.; Reinisch, A.; Camarena, J.; Washington, G.; Nicolas, C.E.; Pavel-Dinu, M.; Saxena, N.; Wilkens, A.B.; Mantri, S.; et al. CRISPR/Cas9 β-globin gene targeting in human haematopoietic stem cells. Nature 2016, 539, 384–389. [Google Scholar] [CrossRef] [PubMed]
- Pavel-Dinu, M.; Wiebking, V.; Dejene, B.T.; Srifa, W.; Mantri, S.; Nicolas, C.E.; Lee, C.; Bao, G.; Kildebeck, E.J.; Punjya, N.; et al. Gene correction for SCID-X1 in long-term hematopoietic stem cells. Nat. Commun. 2019, 10, 1634. [Google Scholar] [CrossRef]
- Salisbury-Ruf, C.T.; Larochelle, A. Advances and Obstacles in Homology-Mediated Gene Editing of Hematopoietic Stem Cells. J. Clin. Med. 2021, 10, 513. [Google Scholar] [CrossRef]
- Gentner, B.; Naldini, L. In Vivo Selection for Gene-Corrected HSPCs Advances Gene Therapy for a Rare Stem Cell Disease. Cell Stem Cell 2019, 25, 592–593. [Google Scholar] [CrossRef]
- Osborn, M.; Lonetree, C.L.; Webber, B.R.; Patel, D.; Dunmire, S.; McElroy, A.N.; DeFeo, A.P.; MacMillan, M.L.; Wagner, J.; Balzar, B.R.; et al. CRISPR/Cas9 Targeted Gene Editing and Cellular Engineering in Fanconi Anemia. Stem Cells Dev. 2016, 25, 1591–1603. [Google Scholar] [CrossRef] [PubMed]
- Skvarova Kramarzova, K.; Osborn, M.J.; Webber, B.R.; DeFeo, A.P.; McElroy, A.N.; Kim, C.J.; Tolar, J. CRISPR/Cas9-Mediated Correction of the FANCD1 Gene in Primary Patient Cells. Int. J. Mol. Sci. 2017, 18, 1269. [Google Scholar] [CrossRef]
- Capo, V.; Penna, S.; Merelli, I.; Barcella, M.; Scala, S.; Basso-Ricci, L.; Draghici, E.; Palagano, E.; Zonari, E.; Desantis, G.; et al. Expanded circulating hematopoietic stem/progenitor cells as novel cell source for the treatment of TCIRG1 osteopetrosis. Haematologica 2021, 106, 74–86. [Google Scholar] [CrossRef]
- Castelli, A.; Susani, L.; Menale, C.; Muggeo, S.; Caldana, E.; Strina, D.; Cassani, B.; Recordati, C.; Scanziani, E.; Ficara, F.; et al. Chromosome Transplantation: Correction of the Chronic Granulomatous Disease Defect in Mouse Induced Pluripotent Stem Cells. Stem Cells 2019, 37, 876–887. [Google Scholar] [CrossRef]
- Julien, E.; El Omar, R.; Tavian, M. Origin of the hematopoietic system in the human embryo. FEBS Lett. 2016, 590, 3987–4001. [Google Scholar] [CrossRef]
- Hansen, M.; von Lindern, M.; van den Akker, E.; Varga, E. Human-induced pluripotent stem cell-derived blood products: State of the art and future directions. FEBS Lett. 2019, 593, 3288–3303. [Google Scholar] [CrossRef] [PubMed]
- Buchrieser, J.; James, W.; Moore, M.D. Human Induced Pluripotent Stem Cell-Derived Macrophages Share Ontogeny with MYB-Independent Tissue-Resident Macrophages. Stem Cell Rep. 2017, 8, 334–345. [Google Scholar] [CrossRef]
- Creamer, J.P.; Dege, C.; Ren, Q.; Ho, J.T.K.; Valentine, M.C.; Druley, T.E.; Sturgeon, C.M. Human definitive hematopoietic specification from pluripotent stem cells is regulated by mesodermal expression of CDX4. Blood 2017, 129, 2988–2992. [Google Scholar] [CrossRef] [PubMed]
- French, A.; Yang, C.T.; Taylor, S.; Watt, S.M.; Carpenter, L. Human induced pluripotent stem cell-derived B lymphocytes express sIgM and can be generated via a hemogenic endothelium intermediate. Stem Cells Dev. 2015, 24, 1082–1095. [Google Scholar] [CrossRef] [PubMed]
- Böiers, C.; Richardson, S.E.; Laycock, E.; Zriwil, A.; Turati, V.A.; Brown, J.; Wray, J.P.; Wang, D.; James, C.; Herrero, J.; et al. A Human IPS Model Implicates Embryonic B-Myeloid Fate Restriction as Developmental Susceptibility to B Acute Lymphoblastic Leukemia-Associated ETV6-RUNX1. Dev. Cell. 2018, 44, 362–377.e7. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Wu, B.; Weng, Q.; Hu, F.; Lin, Y.; Xia, C.; Peng, H.; Wang, Y.; Liu 1, X.; Liu, L.; et al. Regeneration of immunocompetent B lymphopoiesis from pluripotent stem cells guided by transcription factors. Cell Mol. Immunol. 2021. [Google Scholar] [CrossRef]
- Guo, R.; Hu, F.; Weng, Q.; Lv, C.; Wu, H.; Liu, L.; Li, Z.; Zeng, Y.; Bai, Z.; Zhang, M.; et al. Guiding T lymphopoiesis from pluripotent stem cells by defined transcription factors. Cell Res. 2020, 30, 21–33. [Google Scholar] [CrossRef]
- Sugimura, R.; Jha, D.K.; Han, A.; Soria-Valles, C.; da Rocha, E.L.; Lu, Y.F.; Goettel, J.A.; Serrao, E.; Rowe, R.G.; Malleshaiah, M.; et al. Haematopoietic stem and progenitor cells from human pluripotent stem cells. Nature 2017, 545, 432–438. [Google Scholar] [CrossRef]
- Lange, L.; Hoffmann, D.; Schwarzer, A.; Ha, T.C.; Philipp, F.; Lenz, D.; Morgan, M.; Schambach, A. Inducible Forward Programming of Human Pluripotent Stem Cells to Hemato-endothelial Progenitor Cells with Hematopoietic Progenitor Potential. Stem Cell Rep. 2020, 15, 274. [Google Scholar] [CrossRef]
- Ackermann, M.; Liebhaber, S.; Klusmann, J.H.; Lachmann, N. Lost in translation: Pluripotent stem cell-derived hematopoiesis. EMBO Mol. Med. 2015, 7, 1388–1402. [Google Scholar] [CrossRef]
- Wahlster, L.; Daley, G.Q. Progress towards generation of human haematopoietic stem cells. Nat. Cell Biol. 2016, 18, 1111–1117. [Google Scholar] [CrossRef]
- Mills, J.A.; Paluru, P.; Weiss, M.J.; Gadue, P.; French, D.L. Hematopoietic differentiation of pluripotent stem cells in culture. Methods Mol. Biol. 2014, 1185, 181–194. [Google Scholar] [PubMed]
- Vodyanik, M.A.; Thomson, J.A.; Slukvin, I.I. Leukosialin (CD43) defines hematopoietic progenitors in human embryonic stem cell differentiation cultures. Blood 2006, 108, 2095–2105. [Google Scholar] [CrossRef] [PubMed]
- Ledran, M.H.; Krassowska, A.; Armstrong, L.; Dimmick, I.; Renström, J.; Lang, R.; Yung, S.; Santibanez-Coref, M.; Dzierzak, E.; Stojkovic, M.; et al. Efficient hematopoietic differentiation of human embryonic stem cells on stromal cells derived from hematopoietic niches. Cell Stem Cell 2008, 3, 85–98. [Google Scholar] [CrossRef] [PubMed]
- Kennedy, M.; Awong, G.; Sturgeon, C.M.; Ditadi, A.; LaMotte-Mohs, R.; Zúñiga-Pflücker, J.C.; Keller, G. T lymphocyte potential marks the emergence of definitive hematopoietic progenitors in human pluripotent stem cell differentiation cultures. Cell Rep. 2012, 2, 1722–1735. [Google Scholar] [CrossRef] [PubMed]
- Ruiz, J.P.; Chen, G.; Haro Mora, J.J.; Keyvanfar, K.; Liu, C.; Zou, J.; Beers, J.; Bloomer, H.; Qanash, H.; Uchida, N.; et al. Robust generation of erythroid and multilineage hematopoietic progenitors from human iPSCs using a scalable monolayer culture system. Stem Cell Res. 2019, 41, 101600. [Google Scholar] [CrossRef] [PubMed]
- Mora-Roldan, G.A.; Ramirez-Ramirez, D.; Pelayo, R.; Gazarian, K. Assessment of the Hematopoietic Differentiation Potential of Human Pluripotent Stem Cells in 2D and 3D Culture Systems. Cells 2021, 10, 2858. [Google Scholar] [CrossRef]
- Sturgeon, C.M.; Ditadi, A.; Awong, G.; Kennedy, M.; Keller, G. Wnt signaling controls the specification of definitive and primitive hematopoiesis from human pluripotent stem cells. Nat. Biotechnol. 2014, 32, 554–561. [Google Scholar] [CrossRef]
- Nafria, M.; Bonifer, C.; Stanley, E.G.; Ng, E.S.; Elefanty, A.G. Protocol for the Generation of Definitive Hematopoietic Progenitors from Human Pluripotent Stem Cells. STAR Protoc. 2020, 1, 100130. [Google Scholar] [CrossRef]
- Garcia-Alegria, E.; Potts, B.; Menegatti, S.; Kouskoff, V. differentiation of human embryonic stem cells to hemogenic endothelium and blood progenitors via embryoid body formation. STAR Protoc. 2021, 2, 100367. [Google Scholar] [CrossRef]
- Takayama, N.; Nishikii, H.; Usui, J.; Tsukui, H.; Sawaguchi, A.; Hiroyama, T.; Eto, K.; Nakauchi, H. Generation of functional platelets from human embryonic stem cells in vitro via ES-sacs, VEGF-promoted structures that concentrate hematopoietic progenitors. Blood 2008, 111, 5298–5306. [Google Scholar] [CrossRef]
- Martin, R.M.; Ikeda, K.; Cromer, M.K.; Uchida, N.; Nishimura, T.; Romano, R.; Tong, A.J.; Lemgart, V.T.; Camarena, J.; Pavel-Dinu, M.; et al. Highly Efficient and Marker-free Genome Editing of Human Pluripotent Stem Cells by CRISPR-Cas9 RNP and AAV6 Donor-Mediated Homologous Recombination. Cell Stem Cell 2019, 24, 821–828.e5. [Google Scholar] [CrossRef] [PubMed]
- McDonald, D.; Wu, Y.; Dailamy, A.; Tat, J.; Parekh, U.; Zhao, D.; Hu, M.; Tipps, A.; Zhang, K.; Mali, P. Defining the Teratoma as a Model for Multi-lineage Human Development. Cell 2020, 183, 1402–1419.e18. [Google Scholar] [CrossRef] [PubMed]
- Tsukada, M.; Ota, Y.; Wilkinson, A.C.; Becker, H.J.; Osato, M.; Nakauchi, H.; Yamazaki, S. In Vivo Generation of Engraftable Murine Hematopoietic Stem Cells by Gfi1b, c-Fos, and Gata2 Overexpression within Teratoma. Stem Cell Rep. 2017, 9, 1024–1033. [Google Scholar] [CrossRef] [PubMed]
- Deng, J.; Lancelot, M.; Jajosky, R.; Deng, Q.; Deeb, K.; Saakadze, N.; Gao, Y.; Jaye, D.; Liu, S.; Stowell, R.S.; et al. Erythropoietic properties of human induced pluripotent stem cells-derived red blood cells in immunodeficient mice. Am. J. Hematol. 2021, 1, 194–202. [Google Scholar] [CrossRef]
- Garate, Z.; Quintana-Bustamante, O.; Crane, A.M.; Olivier, E.; Poirot, L.; Galetto, R.; Kosinski, P.; Hill, C.; Kung, C.; XAgirre, X.; et al. Generation of a High Number of Healthy Erythroid Cells from Gene-Edited Pyruvate Kinase Deficiency Patient-Specific Induced Pluripotent Stem Cells. Stem Cell Rep. 2015, 5, 1053–1066. [Google Scholar] [CrossRef] [PubMed]
- Pandey, P.; Zhang, N.; Curtis, B.R.; Newman, P.J.; Denomme, G.A. Generation of ‘designer erythroblasts’ lacking one or more blood group systems from CRISPR/Cas9 gene-edited human-induced pluripotent stem cells. J. Cell Mol. Med. 2021, 25, 9340–9349. [Google Scholar] [CrossRef] [PubMed]
- Cappariello, A.; Berardi, A.C.; Peruzzi, B.; Del Fattore, A.; Ugazio, A.; Bottazzo, G.F.; Teti, A. Committed osteoclast precursors colonize the bone and improve the phenotype of a mouse model of autosomal recessive osteopetrosis. J. Bone Miner. Res. 2010, 25, 106–113. [Google Scholar] [CrossRef]
- Root, S.H.; Aguila, H.L. Novel population of human monocyte and osteoclast progenitors from pluripotent stem cells and peripheral blood. Blood Adv. 2021, 5, 4435–4446. [Google Scholar] [CrossRef]
- Lanzi, G.; Ferraro, R.M.; Masneri, S.; Piovani, G.; Barisani, C.; Sobacchi, C.; Villa, A.; Vezzoni, P.; Giliani, S. Generation of 3 clones of induced pluripotent stem cells (iPSCs) from a patient affected by Autosomal Recessive Osteopetrosis due to mutations in TCIRG1 gene. Stem Cell Res. 2020, 42, 101660. [Google Scholar] [CrossRef]
- Chen, W.; Twaroski, K.; Eide, C.; Riddle, M.J.; Orchard, P.J.; Tolar, J. TCIRG1 Transgenic Rescue of Osteoclast Function Using Induced Pluripotent Stem Cells Derived from Patients with Infantile Malignant Autosomal Recessive Osteopetrosis. J. Bone Jt. Surg. Am. 2019, 101, 1939–1947. [Google Scholar] [CrossRef] [PubMed]
- Xian, X.; Moraghebi, R.; Löfvall, H.; Fasth, A.; Henriksen, K.; Richter, J.; Woods, N.B.; Moscatelli, I. Generation of gene-corrected functional osteoclasts from osteopetrotic induced pluripotent stem cells. Stem Cell Res. Ther. 2020, 11, 179. [Google Scholar] [CrossRef] [PubMed]
- Palagano, E.; Muggeo, S.; Crisafulli, L.; Tourkova, I.L.; Strina, D.; Mantero, S.; Fontana, E.; Locatelli, S.L.; Monari, M.; Morenghi, E.; et al. Generation of an immunodeficient mouse model of tcirg1-deficient autosomal recessive osteopetrosis. Bone Rep. 2020, 12, 100242. [Google Scholar] [CrossRef] [PubMed]
| Final Cell Type | PSCs | Differentiation Protocol Steps | Validation | Ref. |
|---|---|---|---|---|
| RBCs | iPSCs from primary erythroblasts | 1. STEMdiff to HSPCs 2. Ery expansion 3. hPL or plasma | FACS; transfusion in NSG mice pre-treated with CL and CVF | [84] |
| MSC-iPS1 | 1. 3D mesoderm induction 2. Hematop. specification 3. Culture with OP9-DL1 | BFU-E, analysis of globin gene expression | [77] | |
| OT1-1 | 1. 2D diff to HSPCs 2. Ery diff and expansion | FACS, morphology, enucleation | [86] | |
| iPSCs from sickle cell anemia pt dermal fibroblasts | 1. 3D (ES sacs) 2. CD34+ isolation 3. Ery diff | FACS | [81] | |
| PLT | PSCs from cell repositories | 1. 2D mesoderm induction 2. Mk diff 3. Plt release in bioreactor | FACS; in vitro thrombus formation, transfusion in NRG/J mice | [8] |
| T and NK cells | MSC-iPS1 | 1. 3D mesoderm induction 2. 2D hematop specification 3. Culture with OP9-DL4 | FACS | [77] |
| OCs | iPSCs from pt skin fibroblasts | 1. 2D OP9 co-culture to HC 2. Myelo-prog. expansion 3. OC prog. expansion 4. OC maturation medium | TRAP staining, morphology, bone resorption | [90] |
| iPSCs from pt skin fibroblasts | 1. 3D mesoderm induction 2. monocyte diff medium 3. OC diff on bone slices | CTX-I release, TRAP staining, resorption | [91] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rao, I.; Crisafulli, L.; Paulis, M.; Ficara, F. Hematopoietic Cells from Pluripotent Stem Cells: Hope and Promise for the Treatment of Inherited Blood Disorders. Cells 2022, 11, 557. https://doi.org/10.3390/cells11030557
Rao I, Crisafulli L, Paulis M, Ficara F. Hematopoietic Cells from Pluripotent Stem Cells: Hope and Promise for the Treatment of Inherited Blood Disorders. Cells. 2022; 11(3):557. https://doi.org/10.3390/cells11030557
Chicago/Turabian StyleRao, Ilaria, Laura Crisafulli, Marianna Paulis, and Francesca Ficara. 2022. "Hematopoietic Cells from Pluripotent Stem Cells: Hope and Promise for the Treatment of Inherited Blood Disorders" Cells 11, no. 3: 557. https://doi.org/10.3390/cells11030557
APA StyleRao, I., Crisafulli, L., Paulis, M., & Ficara, F. (2022). Hematopoietic Cells from Pluripotent Stem Cells: Hope and Promise for the Treatment of Inherited Blood Disorders. Cells, 11(3), 557. https://doi.org/10.3390/cells11030557






