Genetic and Functional Differences between Duplicated Zebrafish Genes for Human SCN1A
Abstract
:1. Introduction
2. Materials and Methods
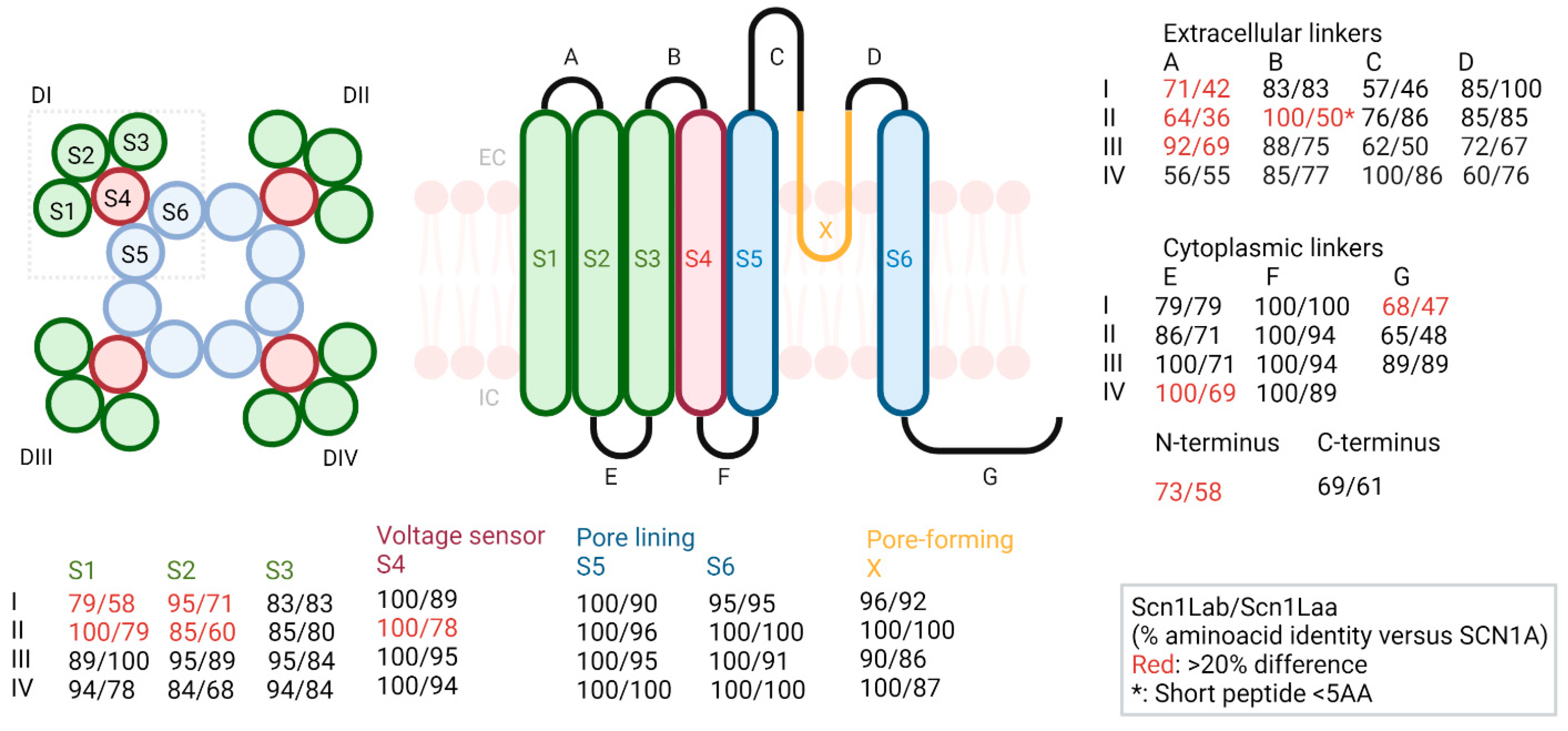
2.1. Alignment
2.2. Zebrafish Maintenance & Ethics Statement
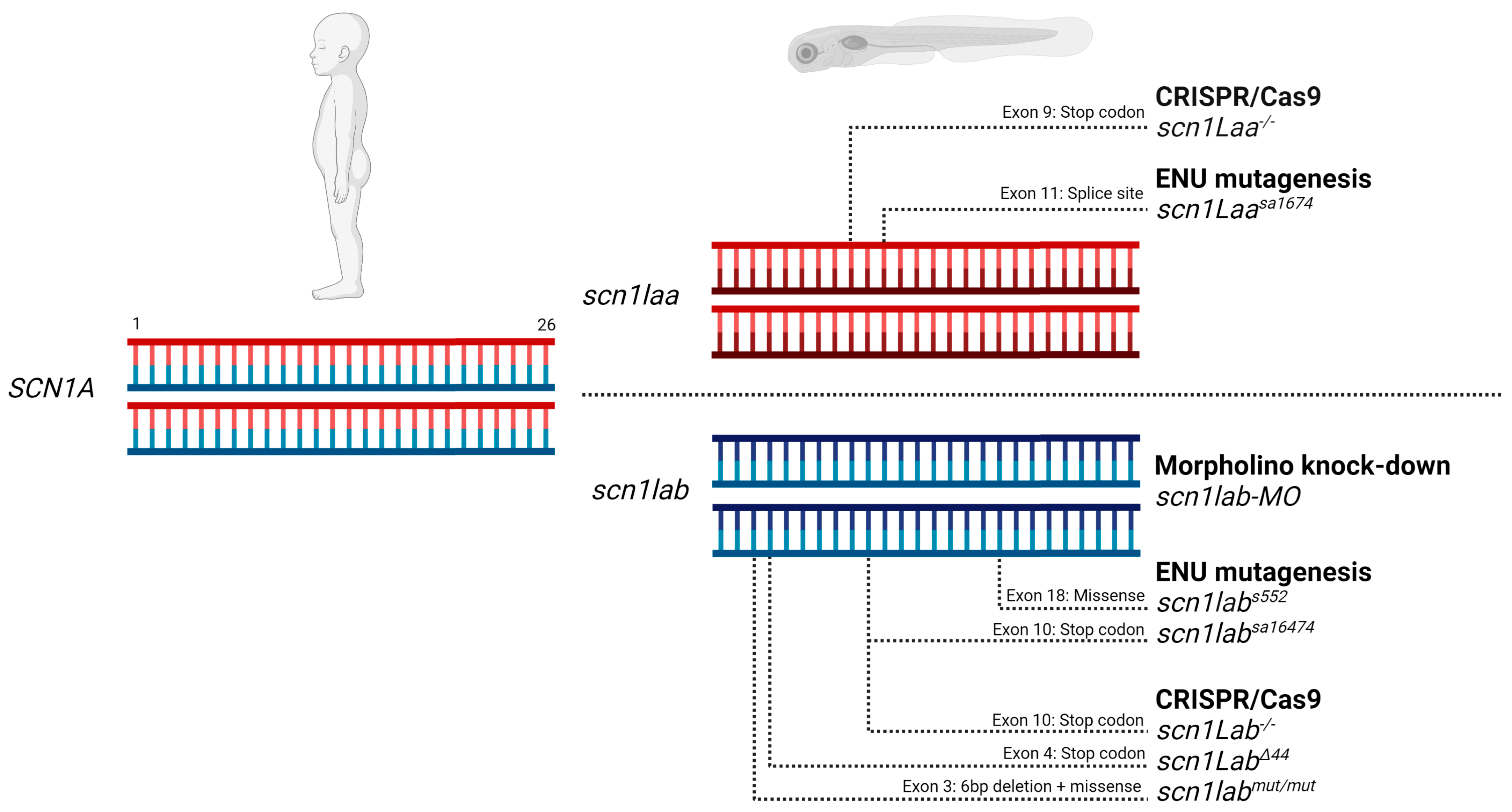
2.3. Scn1laa Knockout Strain Generation
2.4. Locomotor Assays
2.5. Local Field Potential Recordings
3. Results
3.1. Structural Alignment
3.2. Scn1laa Knockout Phenotype
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Tiraboschi, E.; Martina, S.; Ent, W.; Grzyb, K.; Gawel, K.; Cordero-Maldonado, M.L.; Poovathingal, S.K.; Heintz, S.; Satheesh, S.V.; Brattespe, J.; et al. New insights into the early mechanisms of epileptogenesis in a zebrafish model of Dravet syndrome. Epilepsia 2020, 61, 549–560. [Google Scholar] [CrossRef] [PubMed]
- Grone, B.P.; Qu, T.; Baraban, S.C. Behavioral Comorbidities and Drug Treatments in a Zebrafish scn1lab Model of Dravet Syndrome. Eneuro 2017, 4, ENEURO.0066-17.2017. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, Y.; Kecskés, A.; Copmans, D.; Langlois, M.; Crawford, A.D.; Ceulemans, B.; Lagae, L.; de Witte, P.A.M.; Esguerra, C.V. Pharmacological Characterization of an Antisense Knockdown Zebrafish Model of Dravet Syndrome: Inhibition of Epileptic Seizures by the Serotonin Agonist Fenfluramine. PLoS ONE 2015, 10, e0125898. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Baraban, S.C.; Dinday, M.T.; Hortopan, G.A. Drug screening in Scn1a zebrafish mutant identifies clemizole as a potential Dravet syndrome treatment. Nat. Commun. 2013, 4, 1–10. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Weuring, W.J.; Singh, S.; Volkers, L.; Rook, M.B.; van’t Slot, R.H.; Bosma, M.; Inserra, M.; Vetter, I.; Verhoeven-Duif, N.M.; Braun, K.P.J.; et al. NaV1.1 and NaV1.6 selective compounds reduce the behavior phenotype and epileptiform activity in a novel zebrafish model for Dravet Syndrome. PLoS ONE 2020, 15, e0219106. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Weuring, W.J.; Dilevska, I.; Hoekman, J.; van de Vondervoort, J.; Koetsier, M.; van’t Slot, R.H.; Braun, K.P.; Koeleman, B.P. CRISPRa-Mediated Upregulation of scn1laa During Early Development Causes Epileptiform Activity and dCas9-Associated Toxicity. CRISPR J. 2021, 4, 575–582. [Google Scholar] [CrossRef] [PubMed]
- Novak, A.E.; Taylor, A.D.; Pineda, R.H.; Lasda, E.L.; Wright, M.A.; Ribera, A.B. Embryonic and larval expression of zebrafish voltage-gated sodium channel α-subunit genes. Dev. Dyn. 2006, 235, 1962–1973. [Google Scholar] [CrossRef] [PubMed]
- Griffin, A.; Hamling, K.R.; Knupp, K.; Hong, S.; Lee, L.P.; Baraban, S.C. Clemizole and modulators of serotonin signalling suppress seizures in Dravet syndrome. Brain 2017, 140, 669–683. [Google Scholar] [CrossRef]
- Eimon, P.M.; Ghannad-Rezaie, M.; de Rienzo, G.; Allalou, A.; Wu, Y.; Gao, M.; Roy, A.; Skolnick, J.; Yanik, M.F. Brain activity patterns in high-throughput electrophysiology screen predict both drug efficacies and side effects. Nat. Commun. 2018, 9, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Kroll, F.; Powell, G.T.; Ghosh, M.; Gestri, G.; Antinucci, P.; Hearn, T.J.; Tunbak, H.; Lim, S.; Dennis, H.W.; Fernandez, J.M.; et al. A simple and effective F0 knockout method for rapid screening of behaviour and other complex phenotypes. ELife 2021, 10, e59683. [Google Scholar] [CrossRef] [PubMed]
- Griffin, A.; Carpenter, C.; Liu, J.; Paterno, R.; Grone, B.; Hamling, K.; Moog, M.; Dinday, M.T.; Figueroa, F.; Anvar, M.; et al. Phenotypic analysis of catastrophic childhood epilepsy genes. Commun. Biol. 2021, 4, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Novak, A.E.; Jost, M.C.; Lu, Y.; Taylor, A.D.; Zakon, H.H.; Ribera, A.B. Gene duplications and evolution of vertebrate voltage-gated sodium channels. J. Mol. Evol. 2006, 63, 208–221. [Google Scholar] [CrossRef] [PubMed]
- Kiernan, M.C.; Mogyoros, I.; Burke, D. Differences in the recovery of excitability in sensory and motor axons of human median nerve. Brain 1996, 119, 1099–1105. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Waterhouse, A.M.; Procter, J.B.; Martin, D.M.; Clamp, M.; Barton, G.J. Jalview Version 2—A multiple sequence alignment editor and analysis workbench. Bioinformatics 2009, 25, 1189–1191. [Google Scholar] [CrossRef] [Green Version]
- UniProt Website Home Page. 2021. Available online: https://www.uniprot.org (accessed on 29 December 2021).
- Ekberg, J.; Adams, D.J. Neuronal voltage-gated sodium channel subtypes: Key roles in inflammatory and neuropathic pain. Int. J. Biochem. Cell Biol. 2010, 38, 2005–2010. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, F.; Tao, J.; Ye, P.; Ji, Y. Mining the Virgin Land of Neurotoxicology: A Novel Paradigm of Neurotoxic Peptides Action on Glycosylated Voltage-Gated Sodium Channels. J. Toxicol. 2012, 2012, 843787. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- de Bruijn, E.; Cuppen, E.; Feitsma, H. Highly Efficient ENU Mutagenesis in Zebrafish; Lieschke, G., Oates, A., Kawakami, K., Eds.; Humana Press: Clifton, NJ, USA, 2009; Volume 546. [Google Scholar] [CrossRef]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Weuring, W.J.; Hoekman, J.W.; Braun, K.P.J.; Koeleman, B.P.C. Genetic and Functional Differences between Duplicated Zebrafish Genes for Human SCN1A. Cells 2022, 11, 454. https://doi.org/10.3390/cells11030454
Weuring WJ, Hoekman JW, Braun KPJ, Koeleman BPC. Genetic and Functional Differences between Duplicated Zebrafish Genes for Human SCN1A. Cells. 2022; 11(3):454. https://doi.org/10.3390/cells11030454
Chicago/Turabian StyleWeuring, Wout J., Jos W. Hoekman, Kees P. J. Braun, and Bobby P. C. Koeleman. 2022. "Genetic and Functional Differences between Duplicated Zebrafish Genes for Human SCN1A" Cells 11, no. 3: 454. https://doi.org/10.3390/cells11030454
APA StyleWeuring, W. J., Hoekman, J. W., Braun, K. P. J., & Koeleman, B. P. C. (2022). Genetic and Functional Differences between Duplicated Zebrafish Genes for Human SCN1A. Cells, 11(3), 454. https://doi.org/10.3390/cells11030454





