Beyond New Neurons in the Adult Hippocampus: Imipramine Acts as a Pro-Astrogliogenic Factor and Rescues Cognitive Impairments Induced by Stress Exposure
Abstract
:1. Introduction
2. Materials and Methods
2.1. Animals
2.2. Unpredictable Chronic Mild Stress—uCMS- and Drug Treatment
2.3. Behavioral Analysis
2.3.1. Forced Swim Test (FST)
2.3.2. Sucrose Consumption Test (SCT)
2.3.3. Open Field Test
2.3.4. Novel Object Recognition (NOR)
2.4. Serum Corticosterone Levels Measurement
2.5. Hippocampal DG Primary Cultures
2.6. Immunostaining Procedures
2.6.1. In Vivo
2.6.2. In Vitro
2.7. Morphological Analyses
2.8. RT-PCR Measurements
2.9. Statistical Analysis
3. Results
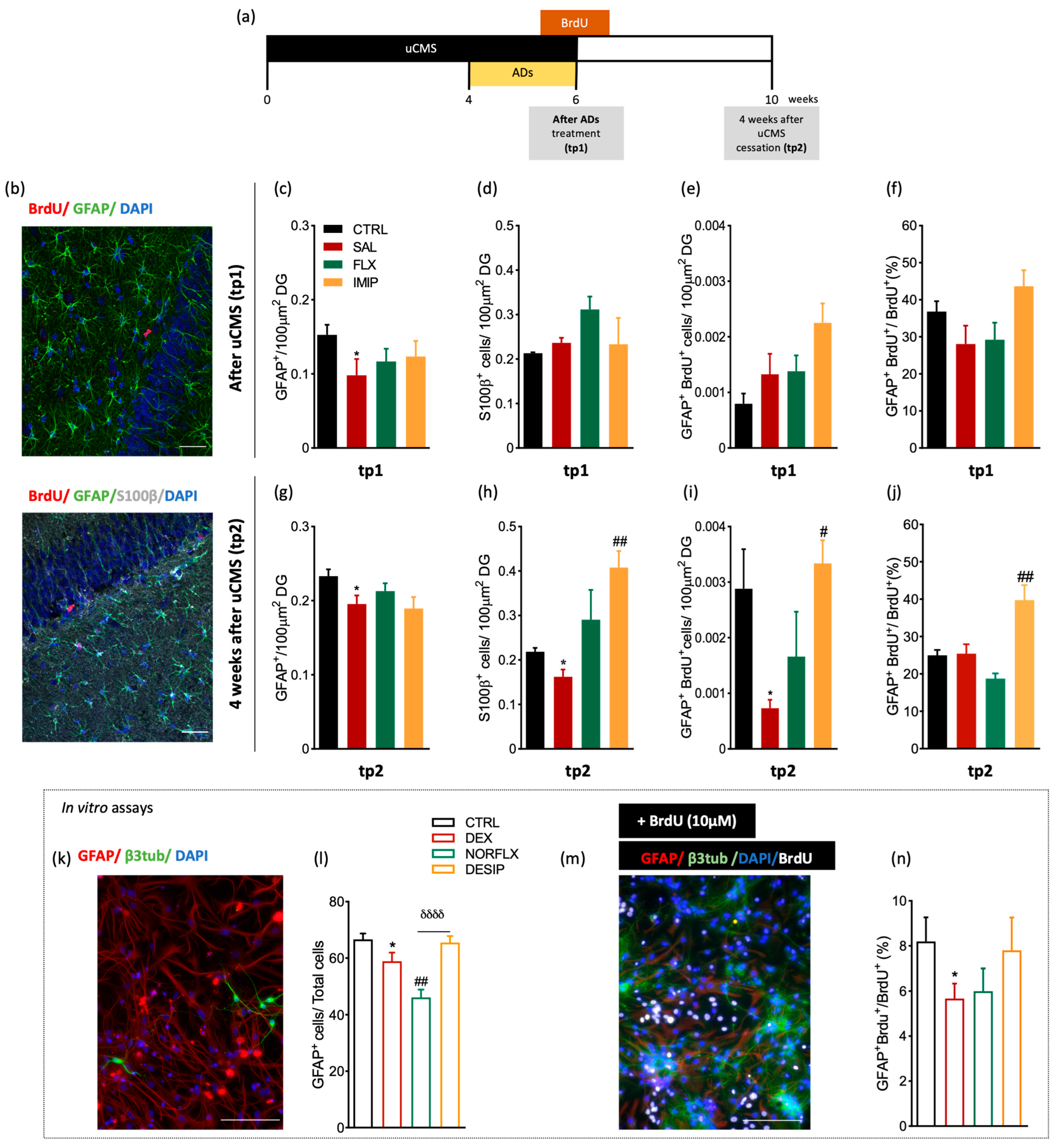
3.1. Imipramine Induces the Generation of New Astrocytes in the Hippocampal Dentate Gyrus
3.2. Expression of Astrocytes’ Mediator Factors
3.3. Fluoxetine Induces Hypertrophy of Resident and Newborn Astrocytes in the Hippocampal Dentate Gyrus
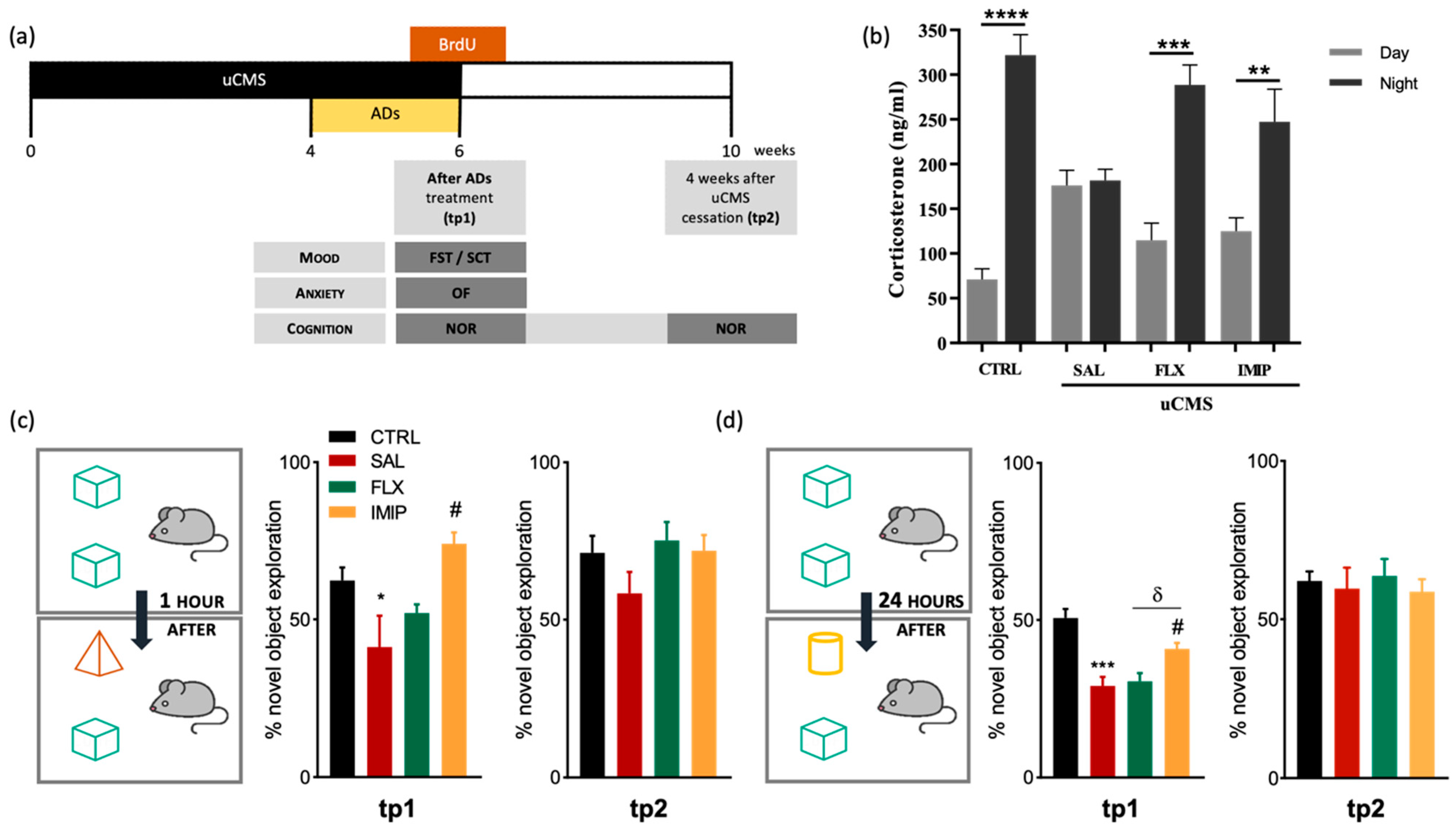
3.4. Imipramine, but Not Fluoxetine, Rescues Cognitive Impairments Induced by Stress Exposure
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Sahay, A.; Hen, R. Adult Hippocampal Neurogenesis in Depression. Nat. Neurosci. 2007, 10, 1110–1115. [Google Scholar] [CrossRef] [PubMed]
- Maletic, V.; Robinson, M.; Oakes, T.; Iyengar, S.; Ball, S.G.; Russell, J. Neurobiology of Depression: An Integrated View of Key Findings. Int. J. Clin. Pract. 2007, 61, 2030–2040. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Duman, R.S.; Monteggia, L.M. A Neurotrophic Model for Stress-Related Mood Disorders. Biol. Psychiatry 2006, 59, 1116–1127. [Google Scholar] [CrossRef] [PubMed]
- Patrício, P.; Mateus-Pinheiro, A.; Sousa, N.; Pinto, L. Re-Cycling Paradigms: Cell Cycle Regulation in Adult Hippocampal Neurogenesis and Implications for Depression. Mol. Neurobiol. 2013, 48, 84–96. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Radley, J.J.; Sisti, H.M.; Hao, J.; Rocher, A.B.; McCall, T.; Hof, P.R.; McEwen, B.S.; Morrison, J.H. Chronic Behavioral Stress Induces Apical Dendritic Reorganization in Pyramidal Neurons of the Medial Prefrontal Cortex. Neuroscience 2004, 125, 1–6. [Google Scholar] [CrossRef]
- Lucassen, P.; Heine, V.; Muller, M.; van der Beek, E.; Wiegant, V.; Ron De Kloet, E.; Joels, M.; Fuchs, E.; Swaab, D.; Czeh, B. Stress, Depression and Hippocampal Apoptosis. CNS Neurol. Disord.—Drug Targets 2008, 5, 531–546. [Google Scholar] [CrossRef]
- Rajkowska, G.; O’Dwyer, G.; Teleki, Z.; Stockmeier, C.A.; Miguel-Hidalgo, J.J. GABAergic Neurons Immunoreactive for Calcium Binding Proteins Are Reduced in the Prefrontal Cortex in Major Depression. Neuropsychopharmacology 2007, 32, 471–482. [Google Scholar] [CrossRef] [Green Version]
- Rajkowska, G.; Miguel-Hidalgo, J. Gliogenesis and Glial Pathology in Depression. CNS Neurol. Disord.—Drug Targets 2008, 6, 219–233. [Google Scholar] [CrossRef] [Green Version]
- Liu, R.J.; Aghajanian, G.K. Stress Blunts Serotonin- and Hypocretin-Evoked EPSCs in Prefrontal Cortex: Role of Corticosterone-Mediated Apical Dendritic Atrophy. Proc. Natl. Acad. Sci. USA 2008, 105, 359–364. [Google Scholar] [CrossRef] [Green Version]
- Banasr, M.; Duman, R.S. Glial Loss in the Prefrontal Cortex Is Sufficient to Induce Depressive-like Behaviors. Biol. Psychiatry 2008, 64, 863–870. [Google Scholar] [CrossRef] [Green Version]
- Maciag, D.; Hughes, J.; O’Dwyer, G.; Pride, Y.; Stockmeier, C.A.; Sanacora, G.; Rajkowska, G. Reduced Density of Calbindin Immunoreactive GABAergic Neurons in the Occipital Cortex in Major Depression: Relevance to Neuroimaging Studies. Biol. Psychiatry 2010, 67, 465–470. [Google Scholar] [CrossRef] [Green Version]
- Abraham, I.; Juhasz, G.; Kekesi, K.A.; Kovacs, K.J. Corticosterone Peak Is Responsible for Stress-Induced Elevation of Glutamate in the Hippocampus. Stress 1998, 2, 171–181. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.; Duan, W.; Mattson, M.P. Evidence That Brain-Derived Neurotrophic Factor Is Required for Basal Neurogenesis and Mediates, in Part, the Enhancement of Neurogenesis by Dietary Restriction in the Hippocampus of Adult Mice. J. Neurochem. 2002, 82, 1367–1375. [Google Scholar] [CrossRef]
- Sairanen, M.; Lucas, G.; Ernfors, P.; Castrén, M.; Castrén, E. Brain-Derived Neurotrophic Factor and AntidepressantDrugs Have Different but Coordinated Effects on Neuronal Turnover, Proliferation, and Survival in the Adult Dentate Gyrus. J. Neurosci. 2005, 25, 1089–1094. [Google Scholar] [CrossRef] [Green Version]
- Bessa, J.M.; Mesquita, A.R.; Oliveira, M.; Pêgo, J.M.; Cerqueira, J.J.; Palha, J.A.; Almeida, O.F.X.; Sousa, N. A Trans-Dimensional Approach to the Behavioral Aspects of Depression. Front. Behav. Neurosci. 2009, 3. [Google Scholar] [CrossRef] [Green Version]
- Mateus-Pinheiro, A.; Pinto, L.; Bessa, J.M.; Morais, M.; Alves, N.D.; Monteiro, S.; Patrício, P.; Almeida, O.F.X.; Sousa, N. Sustained Remission from Depressive-like Behavior Depends on Hippocampal Neurogenesis. Transl. Psychiatry 2013, 3, e210. [Google Scholar] [CrossRef]
- Mateus-Pinheiro, A.; Patrício, P.; Bessa, J.M.; Sousa, N.; Pinto, L. Cell Genesis and Dendritic Plasticity: A Neuroplastic Pas de Deux in the Onset and Remission from Depression. Mol. Psychiatry 2013, 18, 748–750. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Manji, H.K.; Moore, G.J.; Rajkowska, G.; Chen, G. Neuroplasticity and Cellular Resilience in Mood Disorders. Mol. Psychiatry 2000, 5, 578–593. [Google Scholar] [CrossRef] [Green Version]
- Bélair, E.L.; Vallée, J.; Robitaille, R. In Vivo Long-Term Synaptic Plasticity of Glial Cells. J. Physiol. 2010, 588, 1039–1056. [Google Scholar] [CrossRef] [PubMed]
- Ben Achour, S.; Pascual, O. Glia: The Many Ways to Modulate Synaptic Plasticity. Neurochem. Int. 2010, 57, 440–445. [Google Scholar] [CrossRef]
- Miguel-Hidalgo, J.J.; Waltzer, R.; Whittom, A.A.; Austin, M.C.; Rajkowska, G.; Stockmeier, C.A. Glial and Glutamatergic Markers in Depression, Alcoholism, and Their Comorbidity. J. Affect. Disord. 2010, 127, 230–240. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Verkhratsky, A.; Rodríguez, J.J.; Steardo, L. Astrogliopathology: A Central Element of Neuropsychiatric Diseases? Neuroscientist 2014, 20, 576–588. [Google Scholar] [CrossRef] [PubMed]
- Verkhratsky, A.; Rodríguez, J.J.; Parpura, V. Astroglia in Neurological Diseases. Future Neurol. 2013, 8, 149–158. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kofuji, P.; Araque, A. Astrocytes and Behavior. Annu. Rev. Neurosci. 2021, 44, 49–67. [Google Scholar] [CrossRef]
- Kol, A.; Goshen, I. The Memory Orchestra: The Role of Astrocytes and Oligodendrocytes in Parallel to Neurons. Curr. Opin. Neurobiol. 2021, 67, 131–137. [Google Scholar] [CrossRef]
- Saavedra, L.M.; Hernández-Velázquez, M.G.; Madrigal, S.; Ochoa-Zarzosa, A.; Torner, L. Long-Term Activation of Hippocampal Glial Cells and Altered Emotional Behavior in Male and Female Adult Rats after Different Neonatal Stressors. Psychoneuroendocrinology 2021, 126, 105164. [Google Scholar] [CrossRef]
- Semyanov, A.; Verkhratsky, A. Astrocytic Processes: From Tripartite Synapses to the Active Milieu. Trends Neurosci. 2021, 44, 781–792. [Google Scholar] [CrossRef]
- Codeluppi, S.A.; Chatterjee, D.; Prevot, T.D.; Bansal, Y.; Misquitta, K.A.; Sibille, E.; Banasr, M. Chronic Stress Alters Astrocyte Morphology in Mouse Prefrontal Cortex. Int. J. Neuropsychopharmacol. 2021, 24, 842–853. [Google Scholar] [CrossRef]
- Luarte, A.; Cisternas, P.; Caviedes, A.; Batiz, L.F.; Lafourcade, C.; Wyneken, U.; Henzi, R. Astrocytes at the Hub of the Stress Response: Potential Modulation of Neurogenesis by MiRNAs in Astrocyte-Derived Exosomes. Stem Cells Int. 2017, 2017, e1719050. [Google Scholar] [CrossRef] [Green Version]
- Murphy-Royal, C.; Johnston, A.D.; Boyce, A.K.J.; Diaz-Castro, B.; Institoris, A.; Peringod, G.; Zhang, O.; Stout, R.F.; Spray, D.C.; Thompson, R.J.; et al. Stress Gates an Astrocytic Energy Reservoir to Impair Synaptic Plasticity. Nat. Commun. 2020, 11, 2014. [Google Scholar] [CrossRef] [Green Version]
- Naskar, S.; Chattarji, S. Stress Elicits Contrasting Effects on the Structure and Number of Astrocytes in the Amygdala versus Hippocampus. eNeuro 2019, 6, ENEURO.0338-18.2019. [Google Scholar] [CrossRef] [PubMed]
- Theis, M.; Jauch, R.; Zhuo, L.; Speidel, D.; Wallraff, A.; Döring, B.; Frisch, C.; Söhl, G.; Teubner, B.; Euwens, C.; et al. Accelerated Hippocampal Spreading Depression and Enhanced Locomotory Activity in Mice with Astrocyte-Directed Inactivation of Connexin43. J. Neurosci. 2003, 23, 766–776. [Google Scholar] [CrossRef] [PubMed]
- Escartin, C.; Galea, E.; Lakatos, A.; O’Callaghan, J.P.; Petzold, G.C.; Serrano-Pozo, A.; Steinhäuser, C.; Volterra, A.; Carmignoto, G.; Agarwal, A.; et al. Reactive Astrocyte Nomenclature, Definitions, and Future Directions. Nat. Neurosci. 2021, 24, 312–325. [Google Scholar] [CrossRef] [PubMed]
- Czéh, B.; Simon, M.; Schmelting, B.; Hiemke, C.; Fuchs, E. Astroglial Plasticity in the Hippocampus Is Affected by Chronic Psychosocial Stress and Concomitant Fluoxetine Treatment. Neuropsychopharmacology 2006, 31, 1616–1626. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Czéh, B.; Müller-Keuker, J.I.H.; Rygula, R.; Abumaria, N.; Hiemke, C.; Domenici, E.; Fuchs, E. Chronic Social Stress Inhibits Cell Proliferation in the Adult Medial Prefrontal Cortex: Hemispheric Asymmetry and Reversal by Fluoxetine Treatment. Neuropsychopharmacology 2007, 32, 1490–1503. [Google Scholar] [CrossRef] [Green Version]
- Banasr, M.; Duman, R. Regulation of Neurogenesis and Gliogenesis by Stress and Antidepressant Treatment. CNS Neurol. Disord.—Drug Targets 2008, 6, 311–320. [Google Scholar] [CrossRef]
- Banasr, M.; Valentine, G.W.; Li, X.Y.; Gourley, S.L.; Taylor, J.R.; Duman, R.S. Chronic Unpredictable Stress Decreases Cell Proliferation in the Cerebral Cortex of the Adult Rat. Biol. Psychiatry 2007, 62, 496–504. [Google Scholar] [CrossRef]
- Gosselin, R.D.; Gibney, S.; O’Malley, D.; Dinan, T.G.; Cryan, J.F. Region Specific Decrease in Glial Fibrillary Acidic Protein Immunoreactivity in the Brain of a Rat Model of Depression. Neuroscience 2009, 159, 915–925. [Google Scholar] [CrossRef]
- Rajkowska, G.; Selemon, L.D.; Goldman-Rakic, P.S. Neuronal and Glial Somal Size in the Prefrontal Cortex: A Postmortem Morphometric Study of Schizophrenia and Huntington Disease. Arch. Gen. Psychiatry 1998, 55, 215–224. [Google Scholar] [CrossRef]
- Öngür, D.; Drevets, W.C.; Price, J.L. Glial Reduction in the Subgenual Prefrontal Cortex in Mood Disorders. Proc. Natl. Acad. Sci. USA 1998, 95, 13290–13295. [Google Scholar] [CrossRef] [Green Version]
- Rajkowska, G.; Miguel-Hidalgo, J.J.; Wei, J.; Dilley, G.; Pittman, S.D.; Meltzer, H.Y.; Overholser, J.C.; Roth, B.L.; Stockmeier, C.A. Morphometric Evidence for Neuronal and Glial Prefrontal Cell Pathology in Major Depression. Biol. Psychiatry 1999, 45, 1085–1098. [Google Scholar] [CrossRef]
- Gos, T.; Schroeter, M.L.; Lessel, W.; Bernstein, H.G.; Dobrowolny, H.; Schiltz, K.; Bogerts, B.; Steiner, J. S100B-Immunopositive Astrocytes and Oligodendrocytes in the Hippocampus Are Differentially Afflicted in Unipolar and Bipolar Depression: A Postmortem Study. J. Psychiatr. Res. 2013, 47, 1694–1699. [Google Scholar] [CrossRef] [PubMed]
- Chana, G.; Landau, S.; Beasley, C.; Everall, I.P.; Cotter, D. Two-Dimensional Assessment of Cytoarchitecture in the Anterior Cingulate Cortex in Major Depressive Disorder, Bipolar Disorder, and Schizophrenia: Evidence for Decreased Neuronal Somal Size and Increased Neuronal Density. Biol. Psychiatry 2003, 53, 1086–1098. [Google Scholar] [CrossRef]
- Rajkowska, G.; Halaris, A.; Selemon, L.D. Reductions in Neuronal and Glial Density Characterize the Dorsolateral Prefrontal Cortex in Bipolar Disorder. Biol. Psychiatry 2001, 49, 741–752. [Google Scholar] [CrossRef]
- Machado-Santos, A.R.; Alves, N.D.; Araújo, B.; Correia, J.S.; Patrício, P.; Mateus-Pinheiro, A.; Loureiro-Campos, E.; Bessa, J.M.; Sousa, N.; Pinto, L. Astrocytic Plasticity at the Dorsal Dentate Gyrus on an Animal Model of Recurrent Depression. Neuroscience 2019, 454, 94–104. [Google Scholar] [CrossRef] [PubMed]
- Selemon, L.D.; Rajkowska, G.; Goldman-Rakic, P.S. Elevated Neuronal Density in Prefrontal Area 46 in Brains from Schizophrenic Patients: Application of a Three-dimensional, Stereologic Counting Method. J. Comp. Neurol. 1998, 392, 402–412. [Google Scholar] [CrossRef]
- Oliveira, J.F.; Sardinha, V.M.; Guerra-Gomes, S.; Araque, A.; Sousa, N. Do Stars Govern Our Actions? Astrocyte Involvement in Rodent Behavior. Trends Neurosci. 2015, 38, 535–549. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Guerra-Gomes, S.; Sousa, N.; Pinto, L.; Oliveira, J.F. Functional Roles of Astrocyte Calcium Elevations: From Synapses to Behavior. Front. Cell. Neurosci. 2018, 11, 427. [Google Scholar] [CrossRef] [Green Version]
- Steiner, B.; Kronenberg, G.; Jessberger, S.; Brandt, M.D.; Reuter, K.; Kempermann, G. Differential Regulation of Gliogenesis in the Context of Adult Hippocampal Neurogenesis in Mice. Glia 2004, 46, 41–52. [Google Scholar] [CrossRef]
- Ninkovic, J.; Götz, M. Fate Specification in the Adult Brain - Lessons for Eliciting Neurogenesis from Glial Cells. BioEssays 2013, 35, 242–252. [Google Scholar] [CrossRef]
- Von Bohlen Und Halbach, O. Immunohistological Markers for Proliferative Events, Gliogenesis, and Neurogenesis within the Adult Hippocampus. Cell Tissue Res. 2011, 345, 1–19. [Google Scholar] [CrossRef] [PubMed]
- Kraus-Ruppert, R.; Laissue, J.; Bürki, H.; Odartchenko, N. Kinetic Studies on Glial, Schwann and Capsular Cells Labelled with [3H]Thymidine in Cerebrospinal Tissue of Young Mice. J. Neurol. Sci. 1975, 26, 555–563. [Google Scholar] [CrossRef]
- Kornack, D.R.; Rakic, P. Cell Proliferation without Neurogenesis in Adult Primate Neocortex. Science 2001, 294, 2127–2130. [Google Scholar] [CrossRef] [Green Version]
- Gensert, J.M.; Goldman, J.E. Heterogeneity of Cycling Glial Progenitors in the Adult Mammalian Cortex and White Matter. J. Neurobiol. 2001, 48, 75–86. [Google Scholar] [CrossRef]
- Eriksson, P.S.; Perfilieva, E.; Björk-Eriksson, T.; Alborn, A.M.; Nordborg, C.; Peterson, D.A.; Gage, F.H. Neurogenesis in the Adult Human Hippocampus. Nat. Med. 1998, 4, 1313–1317. [Google Scholar] [CrossRef]
- Bhardwaj, R.D.; Curtis, M.A.; Spalding, K.L.; Buchholz, B.A.; Fink, D.; Björk-Eriksson, T.; Nordborg, C.; Gage, F.H.; Druid, H.; Eriksson, P.S.; et al. Neocortical Neurogenesis in Humans Is Restricted to Development. Proc. Natl. Acad. Sci. USA 2006, 103, 12564–12568. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Alonso, G. Prolonged Corticosterone Treatment of Adult Rats Inhibits the Proliferation of Oligodendrocyte Progenitors Present throughout White and Gray Matter Regions of the Brain. Glia 2000, 31, 219–231. [Google Scholar] [CrossRef]
- Wennström, M.; Hellsten, J.; Ekstrand, J.; Lindgren, H.; Tingström, A. Corticosterone-Induced Inhibition of Gliogenesis in Rat Hippocampus Is Counteracted by Electroconvulsive Seizures. Biol. Psychiatry 2006, 59, 178–186. [Google Scholar] [CrossRef] [Green Version]
- Patrício, P.; Mateus-Pinheiro, A.; Irmler, M.; Alves, N.D.; Machado-Santos, A.R.; Morais, M.; Correia, J.S.; Korostynski, M.; Piechota, M.; Stoffel, R.; et al. Differential and Converging Molecular Mechanisms of Antidepressants’ Action in the Hippocampal Dentate Gyrus. Neuropsychopharmacology 2015, 40, 338–349. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vogel-Ciernia, A.; Wood, M.A. Examining Object Location and Object Recognition Memory in Mice. Curr. Protoc. Neurosci. 2014, 69, 8–31. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Alves, N.D.; Correia, J.S.; Patrício, P.; Mateus-Pinheiro, A.; Machado-Santos, A.R.; Loureiro-Campos, E.; Morais, M.; Bessa, J.M.; Sousa, N.; Pinto, L. Adult Hippocampal Neuroplasticity Triggers Susceptibility to Recurrent Depression. Transl. Psychiatry 2017, 7, 1058. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Patrício, P.; Mateus-Pinheiro, A.; Alves, N.D.; Morais, M.; Rodrigues, A.J.; Bessa, J.M.; Sousa, N.; Pinto, L. MiR-409 and MiR-411 Modulation in the Adult Brain of a Rat Model of Depression and After Fluoxetine Treatment. Front. Behav. Neurosci. 2020, 14, 00136. [Google Scholar] [CrossRef] [PubMed]
- Tavares, G.; Martins, M.; Correia, J.S.; Sardinha, V.M.; Guerra-Gomes, S.; das Neves, S.P.; Marques, F.; Sousa, N.; Oliveira, J.F. Employing an Open-Source Tool to Assess Astrocyte Tridimensional Structure. Brain Struct. Funct. 2017, 222, 1989–1999. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cavanagh, B.L.; Walker, T.; Norazit, A.; Meedeniya, A.C.B. Thymidine Analogues for Tracking DNA Synthesis. Molecules 2011, 16, 7980–7993. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tynan, R.J.; Beynon, S.B.; Hinwood, M.; Johnson, S.J.; Nilsson, M.; Woods, J.J.; Walker, F.R. Chronic Stress-Induced Disruption of the Astrocyte Network Is Driven by Structural Atrophy and Not Loss of Astrocytes. Acta Neuropathol. 2013, 126, 75–91. [Google Scholar] [CrossRef]
- Raponi, E.; Agenes, F.; Delphin, C.; Assard, N.; Baudier, J.; Legraverend, C.; Deloulme, J.C. S100B Expression Defines a State in Which GFAP-Expressing Cells Lose Their Neural Stem Cell Potential and Acquire a More Mature Developmental Stage. GLIA 2007, 55, 165–177. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kim, R.; Healey, K.L.; Sepulveda-Orengo, M.T.; Reissner, K.J. Astroglial Correlates of Neuropsychiatric Disease: From Astrocytopathy to Astrogliosis. Prog. Neuro-Psychopharmacol. Biol. Psychiatry 2018, 87, 126–146. [Google Scholar] [CrossRef]
- Saur, L.; Baptista, P.P.A.; Bagatini, P.B.; Neves, L.T.; de Oliveira, R.M.; Vaz, S.P.; Ferreira, K.; Machado, S.A.; Mestriner, R.G.; Xavier, L.L. Experimental Post-Traumatic Stress Disorder Decreases Astrocyte Density and Changes Astrocytic Polarity in the CA1 Hippocampus of Male Rats. Neurochem. Res. 2016, 41, 892–904. [Google Scholar] [CrossRef] [PubMed]
- Torres-Platas, S.G.; Hercher, C.; Davoli, M.A.; Maussion, G.; Labonté, B.; Turecki, G.; Mechawar, N. Astrocytic Hypertrophy in Anterior Cingulate White Matter of Depressed Suicides. Neuropsychopharmacology 2011, 36, 2650–2658. [Google Scholar] [CrossRef] [Green Version]
- Herman, J.P.; McKlveen, J.M.; Ghosal, S.; Kopp, B.; Wulsin, A.; Makinson, R.; Scheimann, J.; Myers, B. Regulation of the Hypothalamic-Pituitary- Adrenocortical Stress Response. Compr. Physiol. 2016, 6, 603–621. [Google Scholar] [CrossRef] [Green Version]
- Sawangjit, A.; Oyanedel, C.N.; Niethard, N.; Salazar, C.; Born, J.; Inostroza, M. The Hippocampus Is Crucial for Forming Non-Hippocampal Long-Term Memory during Sleep. Nature 2018, 564, 109–113. [Google Scholar] [CrossRef]
- Takano, K.; Yamasaki, H.; Kawabe, K.; Moriyama, M.; Nakamura, Y. Imipramine Induces Brain-Derived Neurotrophic Factor MRNA Expression in Cultured Astrocytes. J. Pharmacol. Sci. 2012, 120, 176–186. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kedracka-Krok, S.; Swiderska, B.; Bielecka-Wajdman, A.M.; Prus, G.; Skupien-Rabian, B.; Jankowska, U.; Obuchowicz, E. Impact of Imipramine on Proteome of Rat Primary Glial Cells. J. Neuroimmunol. 2018, 320, 25–37. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.; Kim, S.H.; Kim, Y.S.; Lee, Y.H.; Ha, K.; Shin, S.Y. Imipramine Activates Glial Cell Line-Derived Neurotrophic Factor via Early Growth Response Gene 1 in Astrocytes. Prog. Neuro-Psychopharmacol. Biol. Psychiatry 2011, 35, 1026–1032. [Google Scholar] [CrossRef] [PubMed]
- Jhaveri, D.J.; Mackay, E.W.; Hamlin, A.S.; Marathe, S.V.; Nandam, L.S.; Vaidya, V.A.; Bartlett, P.F. Norepinephrine Directly Activates Adult Hippocampal Precursors via Β3-Adrenergic Receptors. J. Neurosci. 2010, 30, 2795–2806. [Google Scholar] [CrossRef] [PubMed]
- Liu, B.; Teschemacher, A.G.; Kasparov, S. Neuroprotective Potential of Astroglia. J. Neurosci. Res. 2017, 95, 2126–2139. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cheng, X.; Wang, J.; Sun, X.; Shao, L.; Guo, Z.; Li, Y. Morphological and Functional Alterations of Astrocytes Responding to Traumatic Brain Injury. J. Integr. Neurosci. 2019, 18, 203–215. [Google Scholar] [CrossRef] [PubMed]
- Encinas, J.M.; Michurina, T.V.; Peunova, N.; Park, J.-H.; Tordo, J.; Peterson, D.A.; Fishell, G.; Koulakov, A.; Enikolopov, G. Division-Coupled Astrocytic Differentiation and Age-Related Depletion of Neural Stem Cells in the Adult Hippocampus. Cell Stem Cell 2011, 8, 566–579. [Google Scholar] [CrossRef] [Green Version]
- Rosenblat, J.D.; Kakar, R.; McIntyre, R.S. The Cognitive Effects of Antidepressants in Major Depressive Disorder: A Systematic Review and Meta-Analysis of Randomized Clinical Trials. Int. J. Neuropsychopharmacol. 2015, 19, pyv082. [Google Scholar] [CrossRef] [Green Version]
- Leiser, S.C.; Pehrson, A.L.; Robichaud, P.J.; Sanchez, C. Multimodal Antidepressant Vortioxetine Increases Frontal Cortical Oscillations Unlike Escitalopram and Duloxetine – a Quantitative EEG Study in Rats. Br. J. Pharmacol. 2014, 171, 4255–4272. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pehrson, A.L.; Sanchez, C. Serotonergic Modulation of Glutamate Neurotransmission as a Strategy for Treating Depression and Cognitive Dysfunction. CNS Spectr. 2014, 19, 121–133. [Google Scholar] [CrossRef] [PubMed]
- Katona, C.; Hansen, T.; Olsen, C.K. A Randomized, Double-Blind, Placebo-Controlled, Duloxetine-Referenced, Fixed-Dose Study Comparing the Efficacy and Safety of Lu AA21004 in Elderly Patients with Major Depressive Disorder. Int. Clin. Psychopharmacol. 2012, 27, 215–223. [Google Scholar] [CrossRef] [PubMed]
- Pan, L.A.; Martin, P.; Zimmer, T.; Segreti, A.M.; Kassiff, S.; McKain, B.W.; Baca, C.A.; Rengasamy, M.; Hyland, K.; Walano, N.; et al. Neurometabolic Disorders: Potentially Treatable Abnormalities in Patients With Treatment-Refractory Depression and Suicidal Behavior. Am. J. Psychiatry 2017, 174, 42–50. [Google Scholar] [CrossRef] [PubMed]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Machado-Santos, A.R.; Loureiro-Campos, E.; Patrício, P.; Araújo, B.; Alves, N.D.; Mateus-Pinheiro, A.; Correia, J.S.; Morais, M.; Bessa, J.M.; Sousa, N.; et al. Beyond New Neurons in the Adult Hippocampus: Imipramine Acts as a Pro-Astrogliogenic Factor and Rescues Cognitive Impairments Induced by Stress Exposure. Cells 2022, 11, 390. https://doi.org/10.3390/cells11030390
Machado-Santos AR, Loureiro-Campos E, Patrício P, Araújo B, Alves ND, Mateus-Pinheiro A, Correia JS, Morais M, Bessa JM, Sousa N, et al. Beyond New Neurons in the Adult Hippocampus: Imipramine Acts as a Pro-Astrogliogenic Factor and Rescues Cognitive Impairments Induced by Stress Exposure. Cells. 2022; 11(3):390. https://doi.org/10.3390/cells11030390
Chicago/Turabian StyleMachado-Santos, Ana R., Eduardo Loureiro-Campos, Patrícia Patrício, Bruna Araújo, Nuno Dinis Alves, António Mateus-Pinheiro, Joana Sofia Correia, Mónica Morais, João M. Bessa, Nuno Sousa, and et al. 2022. "Beyond New Neurons in the Adult Hippocampus: Imipramine Acts as a Pro-Astrogliogenic Factor and Rescues Cognitive Impairments Induced by Stress Exposure" Cells 11, no. 3: 390. https://doi.org/10.3390/cells11030390
APA StyleMachado-Santos, A. R., Loureiro-Campos, E., Patrício, P., Araújo, B., Alves, N. D., Mateus-Pinheiro, A., Correia, J. S., Morais, M., Bessa, J. M., Sousa, N., Rodrigues, A. J., Oliveira, J. F., & Pinto, L. (2022). Beyond New Neurons in the Adult Hippocampus: Imipramine Acts as a Pro-Astrogliogenic Factor and Rescues Cognitive Impairments Induced by Stress Exposure. Cells, 11(3), 390. https://doi.org/10.3390/cells11030390







