TDP-43 Cytoplasmic Translocation in the Skin Fibroblasts of ALS Patients
Abstract
:1. Introduction
2. Materials and Methods
2.1. Participants and Samples
2.2. Skin Biopsy and Processing
2.3. Confocal Imaging and Measurements
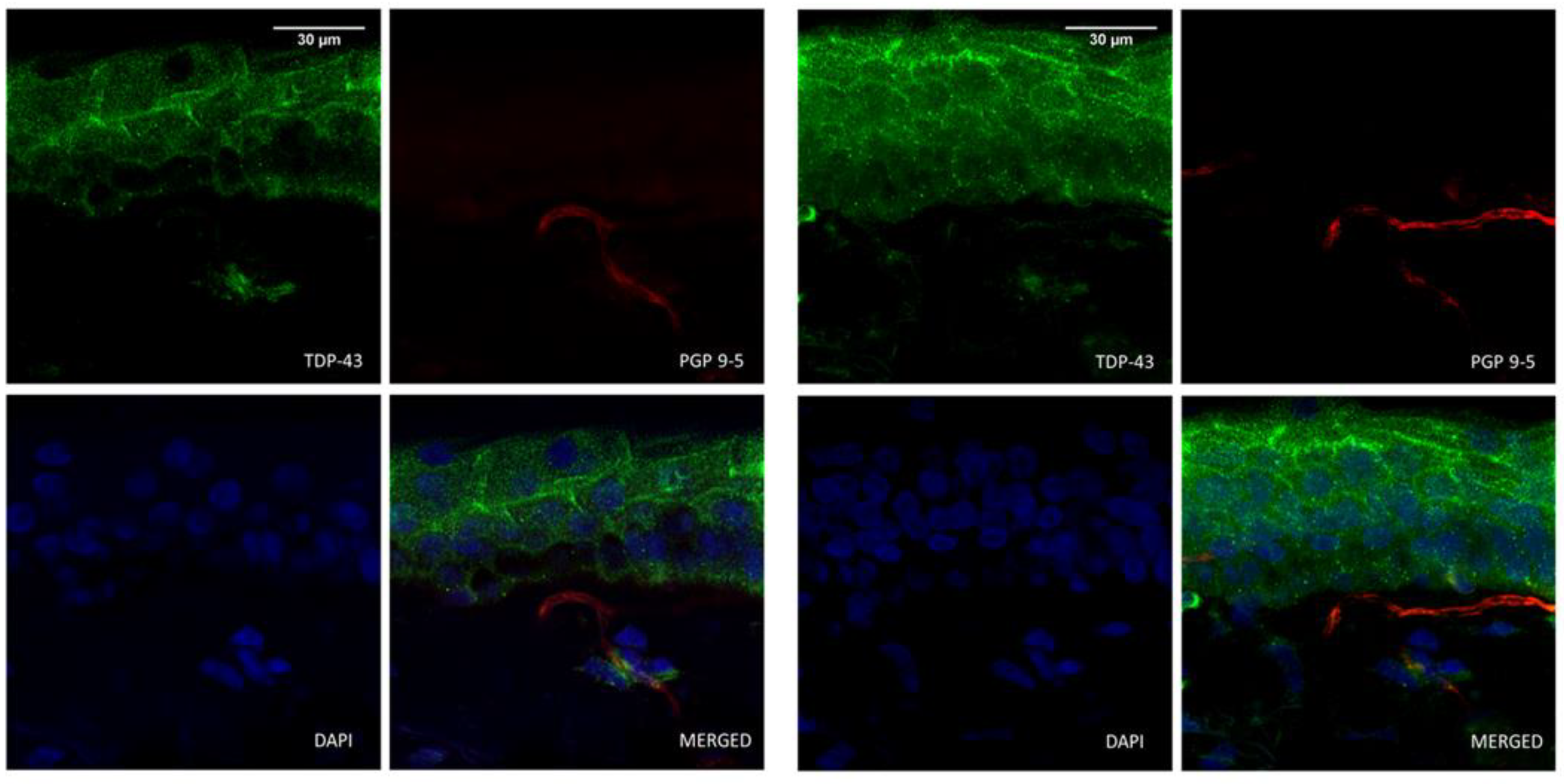
2.4. Intraepidermal Innervation and TDP-43 Nerve Colocalization
2.5. Data Analysis
3. Results
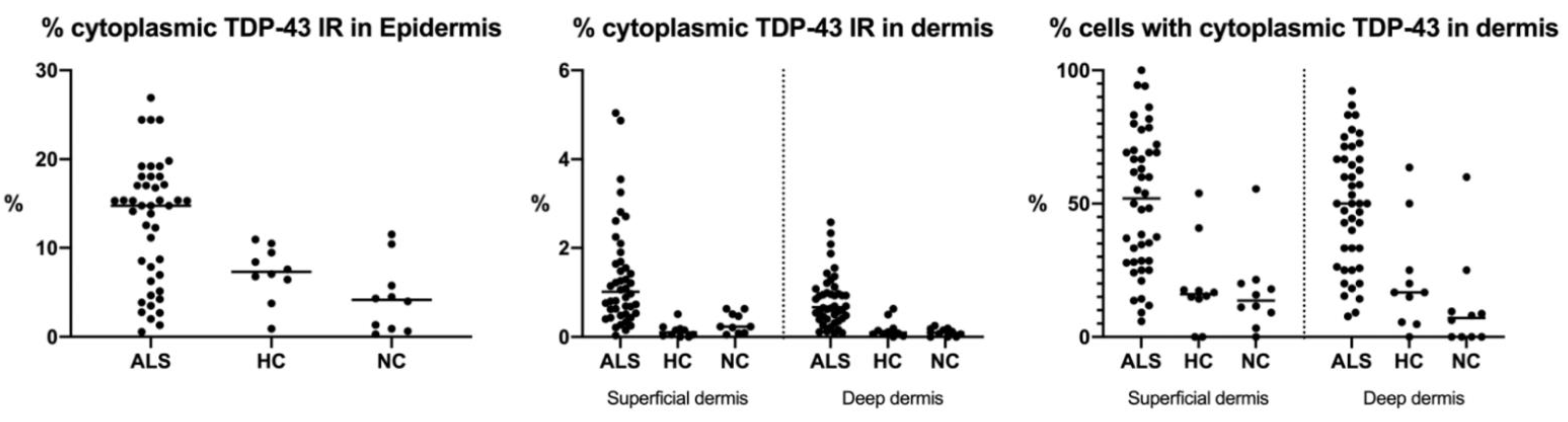
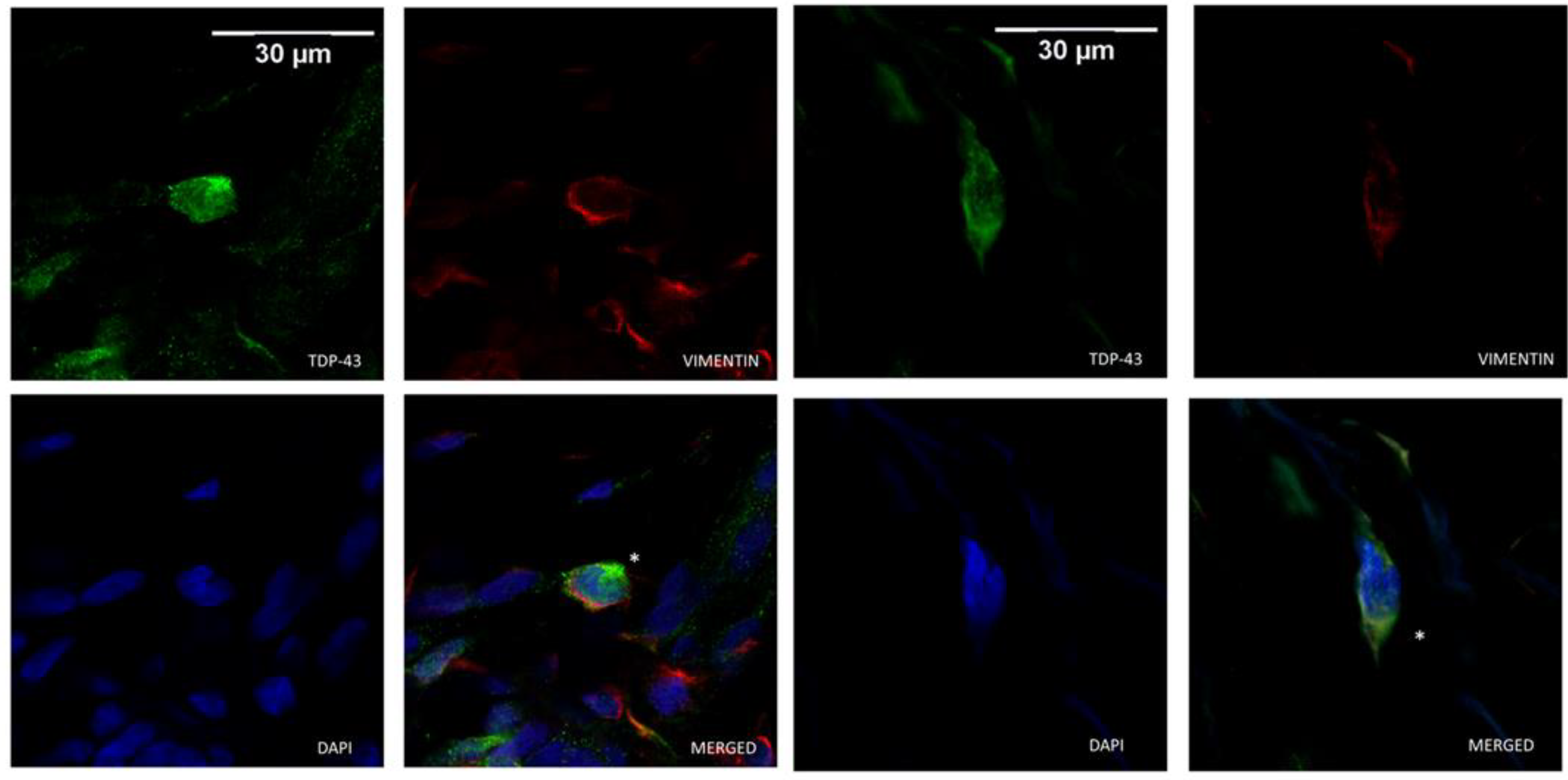
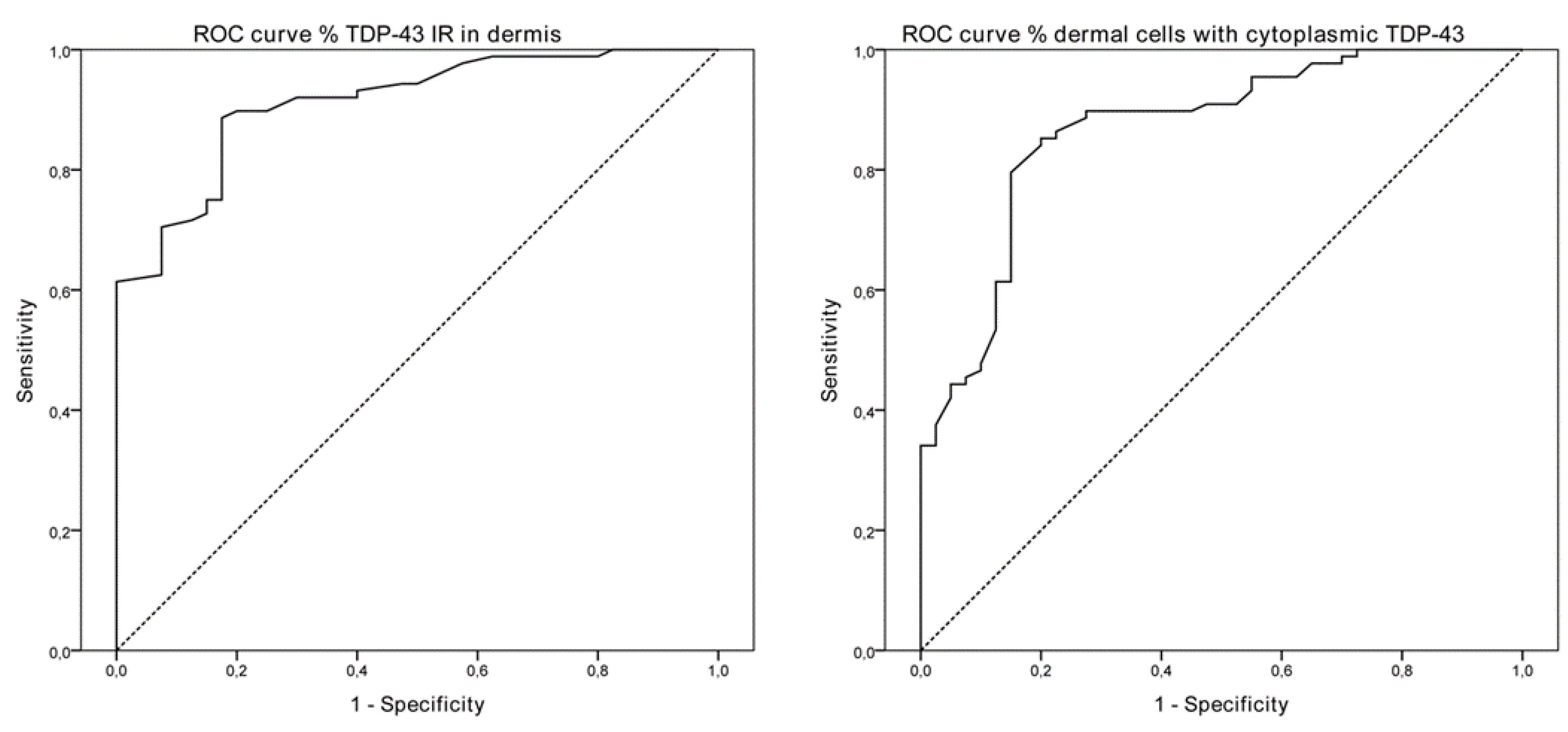
3.1. TDP-43 Cytoplasmic Localization in ALS Patients Skin Biopsies
3.2. TDP-43 Changes over Time
3.3. TDP-43 and Epidermal Nerve Fibers
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Hardiman, O.; Al-Chalabi, A.; Chio, A.; Corr, E.M.; Logroscino, G.; Robberecht, W.; Van Den Berg, L.H. Amyotrophic lateral sclerosis. Nat. Rev. Dis. Primers 2017, 3, 1–19. [Google Scholar] [CrossRef]
- Mancuso, R.; Navarro, X. Amyotrophic lateral sclerosis: Current perspectives from basic research to the clinic. Prog. Neurobiol. 2015, 133, 1–26. [Google Scholar] [CrossRef]
- McCombe, P.A.; Wray, N.R.; Henderson, R.D. Extra-motor abnormalities in amyotrophic lateral sclerosis: Another layer of heterogeneity. Expert Rev. Neurother. 2017, 17, 561–577. [Google Scholar] [CrossRef]
- McCluskey, L.; Vandriel, S.; Elman, L.; Van Deerlin, V.M.; Powers, J.; Boller, A.; Wood, E.M.; Woo, J.; McMillan, C.T.; Rascovsky, K.; et al. ALS-Plus syndrome: Non-pyramidal features in a large ALS cohort. J. Neurol. Sci. 2014, 345, 118–124. [Google Scholar] [CrossRef] [Green Version]
- Strong, M.J. The syndromes of frontotemporal dysfunction in amyotrophic lateral sclerosis. Amyotroph. Lateral Scler. 2008, 9, 323–338. [Google Scholar] [CrossRef] [PubMed]
- Phukan, J.; Pender, N.; Hardiman, O. Cognitive impairment in amyotrophic lateral sclerosis. Lancet Neurol. 2007, 6, 994–1003. [Google Scholar] [CrossRef]
- Kwong, L.K.; Neumann, M.; Sampathu, D.M.; Lee, V.M.-Y.; Trojanowski, J.Q. TDP-43 proteinopathy: The neuropathology underlying major forms of sporadic and familial frontotemporal lobar degeneration and motor neuron disease. Acta Neuropathol. 2007, 114, 63–70. [Google Scholar] [CrossRef]
- Neumann, M.; Sampathu, D.M.; Kwong, L.K.; Truax, A.C.; Micsenyi, M.C.; Chou, T.T.; Bruce, J.; Schuck, T.; Grossman, M.; Clark, C.M.; et al. Ubiquitinated TDP-43 in Frontotemporal Lobar Degeneration and Amyotrophic Lateral Sclerosis. Science 2006, 314, 130–133. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chia, R.; Chiò, A.; Traynor, B.J. Novel genes associated with amyotrophic lateral sclerosis: Diagnostic and clinical implications. Lancet Neurol. 2018, 17, 94–102. [Google Scholar] [CrossRef]
- Brettschneider, J.; Del Tredici, K.; Toledo, J.B.; Robinson, J.L.; Irwin, D.J.; Grossman, M.; Suh, E.R.; Van Deerlin, V.M.; Wood, E.M.; Baek, Y.; et al. Stages of pTDP-43 pathology in amyotrophic lateral sclerosis. Ann. Neurol. 2013, 74, 20–38. [Google Scholar] [CrossRef]
- Neumann, M. Molecular Neuropathology of TDP-43 Proteinopathies. Int. J. Mol. Sci. 2009, 10, 232–246. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Braak, H.; Ludolph, A.C.; Neumann, M.; Ravits, J.; Del Tredici, K. Pathological TDP-43 changes in Betz cells differ from those in bulbar and spinal α-motoneurons in sporadic amyotrophic lateral sclerosis. Acta Neuropathol. 2017, 133, 79–90. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Clos, A.L.; Kayed, R.; Lasagna-Reeves, C.A. Association of Skin with the Pathogenesis and Treatment of Neurodegenerative Amyloidosis. Front. Neurol. 2012, 3, 5. [Google Scholar] [CrossRef] [Green Version]
- Suzuki, M.; Mikami, H.; Watanabe, T.; Yamano, T.; Yamazaki, T.; Nomura, M.; Yasui, K.; Ishikawa, H.; Ono, S. Increased expression of TDP-43 in the skin of amyotrophic lateral sclerosis. Acta Neurol. Scand. 2010, 122, 367–372. [Google Scholar] [CrossRef]
- Wang, X.; Zhou, S.; Ding, X.; Ma, M.; Zhang, J.; Zhou, Y.; Wu, E.; Teng, J. Activation of ER Stress and Autophagy Induced by TDP-43 A315T as Pathogenic Mechanism and the Corresponding Histological Changes in Skin as Potential Biomarker for ALS with the Mutation. Int. J. Biol. Sci. 2015, 11, 1140–1149. [Google Scholar] [CrossRef] [Green Version]
- Paré, B.; Touzel-Deschênes, L.; Lamontagne, R.; Lamarre, M.-S.; Scott, F.-D.; Khuong, H.T.; Dion, P.A.; Bouchard, J.-P.; Gould, P.; Rouleau, G.A.; et al. Early detection of structural abnormalities and cytoplasmic accumulation of TDP-43 in tissue-engineered skins derived from ALS patients. Acta Neuropathol. Commun. 2015, 3, 1–12. [Google Scholar] [CrossRef] [Green Version]
- Sabatelli, M.; Zollino, M.; Conte, A.; Del Grande, A.; Marangi, G.; Lucchini, M.; Mirabella, M.; Romano, A.; Piacentini, R.; Bisogni, G.; et al. Primary fibroblasts cultures reveal TDP-43 abnormalities in amyotrophic lateral sclerosis patients with and without SOD1 mutations. Neurobiol. Aging 2015, 36, 2005-e5–2005-e13. [Google Scholar] [CrossRef]
- Yang, S.; Zhang, K.Y.; Kariawasam, R.; Bax, M.; Fifita, J.A.; Ooi, L.; Yerbury, J.J.; Nicholson, G.A.; Blair, I.P. Evaluation of Skin Fibroblasts from Amyotrophic Lateral Sclerosis Patients for the Rapid Study of Pathological Features. Neurotox. Res. 2015, 28, 138–146. [Google Scholar] [CrossRef]
- Abe, K.; Ohkubo, T.; Yokota, T. TDP-43 in the skin of amyotrophic lateral sclerosis patients. J. Med. Dent. Sci. 2017, 64, 9–17. [Google Scholar] [PubMed]
- Riancho, J.; Castanedo-Vázquez, D.; Gil-Bea, F.; Tapia, O.; Arozamena, J.; Durán-Vían, C.; Sedano, M.J.; Berciano, M.T.; De Munain, A.L.; Lafarga, M. ALS-derived fibroblasts exhibit reduced proliferation rate, cytoplasmic TDP-43 aggregation and a higher susceptibility to DNA damage. J. Neurol. 2020, 267, 1291–1299. [Google Scholar] [CrossRef]
- Romano, N.; Catalani, A.; Lattante, S.; Belardo, A.; Proietti, S.; Bertini, L.; Ceci, M. ALS skin fibroblasts reveal oxidative stress and ERK1/2-mediated cytoplasmic localization of TDP-43. Cell. Signal. 2020, 70, 109591. [Google Scholar] [CrossRef] [PubMed]
- Uhlen, M.; Oksvold, P.; Fagerberg, L.; Lundberg, E.; Jonasson, K.; Forsberg, M.; Zwahlen, M.; Kampf, C.; Wester, K.; Hober, S.; et al. Towards a knowledge-based Human Protein Atlas. Nat. Biotechnol. 2010, 28, 1248–1250. [Google Scholar] [CrossRef]
- Thul, P.J.; Åkesson, L.; Wiking, M.; Mahdessian, D.; Geladaki, A.; Blal, H.A.; Alm, T.; Asplund, A.; Björk, L.; Breckels, L.M.; et al. A subcellular map of the human proteome. Science 2017, 356, eaal3321. [Google Scholar] [CrossRef] [PubMed]
- Brooks, B.R.; Miller, R.G.; Swash, M.; Munsat, T.L. El Escorial revisited: Revised criteria for the diagnosis of amyotrophic lateral sclerosis. Amyotroph. Lateral Scler. Other Mot. Neuron. Disord. 2000, 1, 293–299. [Google Scholar] [CrossRef]
- Williams, S.M.; Khan, G.; Harris, B.T.; Ravits, J.; Sierks, M.R. TDP-43 protein variants as biomarkers in amyotrophic lateral sclerosis. BMC Neurosci. 2017, 18, 1–12. [Google Scholar] [CrossRef] [Green Version]
- Codron, P.; Cassereau, J.; Vourc’H, P.; Veyrat-Durebex, C.; Blasco, H.; Kane, S.; Procaccio, V.; Letournel, F.; Verny, C.; Lenaers, G.; et al. Primary fibroblasts derived from sporadic amyotrophic lateral sclerosis patients do not show ALS cytological lesions. Amyotroph. Lateral Scler. Front. Degener. 2018, 19, 446–456. [Google Scholar] [CrossRef]
- Ren, Y.; Liu, W.; Li, Y.; Sun, B.; Li, Y.; Yang, F.; Wang, H.; Li, M.; Cui, F.; Huang, X. Cutaneous somatic and autonomic nerve TDP-43 deposition in amyotrophic lateral sclerosis. J. Neurol. 2018, 265, 1753–1763. [Google Scholar] [CrossRef]
- Ono, S. The skin in amyotrophic lateral sclerosis. Amyotroph. Lateral Scler. Other Mot. Neuron Disord. 2000, 1, 191–199. [Google Scholar] [CrossRef]
- Zhang, H.; Tan, C.-F.; Mori, F.; Tanji, K.; Kakita, A.; Takahashi, H.; Wakabayashi, K. TDP-43-immunoreactive neuronal and glial inclusions in the neostriatum in amyotrophic lateral sclerosis with and without dementia. Acta Neuropathol. 2007, 115, 115–122. [Google Scholar] [CrossRef]
- Paré, B.; Gros-Louis, F. Potential skin involvement in ALS: Revisiting Charcot’s observation–a review of skin abnormalities in ALS. Rev. Neurosci. 2017, 28, 551–572. [Google Scholar] [CrossRef] [PubMed]
- Neumann, M.; Kwong, L.K.; Lee, E.B.; Kremmer, E.; Flatley, A.; Xu, Y.; Lee, V.M.Y. Phosphorylation of S409/410 of TDP-43 is a consistent feature in all sporadic and familial forms of TDP-43 proteinopathies. Acta Neuropathol. 2009, 117, 137–149. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hasegawa, M.; Arai, T.; Nonaka, T.; Kametani, F.; Yoshida, M.; Hashizume, Y.; Beach, T.G.; Buratti, E.; Baralle, F.; Morita, M.; et al. Phosphorylated TDP-43 in frontotemporal lobar degeneration and amyotrophic lateral sclerosis. Ann. Neurol. 2008, 64, 60–70. [Google Scholar] [CrossRef] [Green Version]
- Miki, Y.; Mori, F.; Nunomura, J.; Ookawa, K.; Yajima, N.; Yagihashi, S.; Wakabayashi, K. Sporadic amyotrophic lateral sclerosis with pallido-nigro-luysian degeneration: A TDP-43 immunohistochemical study. Neuropathology 2010, 30, 149–153. [Google Scholar] [CrossRef]
- Igaz, L.M.; Kwong, L.K.; Xu, Y.; Truax, A.C.; Uryu, K.; Neumann, M.; Clark, C.M.; Elman, L.B.; Miller, B.L.; Grossman, M.; et al. Enrichment of C-Terminal Fragments in TAR DNA-Binding Protein-43 Cytoplasmic Inclusions in Brain but not in Spinal Cord of Frontotemporal Lobar Degeneration and Amyotrophic Lateral Sclerosis. Am. J. Pathol. 2008, 173, 182–194. [Google Scholar] [CrossRef] [Green Version]
- Nishihira, Y.; Tan, C.-F.; Onodera, O.; Toyoshima, Y.; Yamada, M.; Morita, T.; Nishizawa, M.; Kakita, A.; Takahashi, H. Sporadic amyotrophic lateral sclerosis: Two pathological patterns shown by analysis of distribution of TDP-43-immunoreactive neuronal and glial cytoplasmic inclusions. Acta Neuropathol. 2008, 116, 169–182. [Google Scholar] [CrossRef]
- Weskamp, K.; Tank, E.M.; Miguez, R.; McBride, J.P.; Gómez, N.B.; White, M.; Lin, Z.; Gonzalez, C.M.; Serio, A.; Sreedharan, J.; et al. Shortened TDP43 isoforms upregulated by neuronal hyperactivity drive TDP43 pathology in ALS. J. Clin. Investig. 2020, 130, 1139–1155. [Google Scholar] [CrossRef] [Green Version]
- Weis, J.; Katona, I.; Muller-Newen, G.; Sommer, C.; Necula, G.; Hendrich, C.; Ludolph, A.C.; Sperfeld, A.-D. Small-fiber neuropathy in patients with ALS. Neurology 2011, 76, 2024–2029. [Google Scholar] [CrossRef] [PubMed]
- de Carvalho, M.; Dengler, R.; Eisen, A.; England, J.D.; Kaji, R.; Kimura, J.; Swash, M. Electrodiagnostic criteria for diagnosis of ALS. Clin. Neurophysiol. 2008, 119, 497–503. [Google Scholar] [CrossRef]
- Shefner, J.M.; Al-Chalabi, A.; Baker, M.R.; Cui, L.-Y.; de Carvalho, M.; Eisen, A.; Grosskreutz, J.; Hardiman, O.; Henderson, R.; Matamala, J.M.; et al. A proposal for new diagnostic criteria for ALS. Clin. Neurophysiol. 2020, 131, 1975–1978. [Google Scholar] [CrossRef]
- Palese, F.; Sartori, A.; Logroscino, G.; Pisa, F.E. Predictors of diagnostic delay in amyotrophic lateral sclerosis: A cohort study based on administrative and electronic medical records data. Amyotroph. Lateral Scler. Front. Degener. 2019, 20, 176–185. [Google Scholar] [CrossRef] [PubMed]
- Paganoni, S.; Macklin, E.; Lee, A.; Murphy, A.; Chang, J.; Zipf, A.; Cudkowicz, M.; Atassi, N. Diagnostic timelines and delays in diagnosing amyotrophic lateral sclerosis (ALS). Amyotroph. Lateral Scler. Front. Degener. 2014, 15, 453–456. [Google Scholar] [CrossRef] [Green Version]
- Feneberg, E.; Gray, E.; Ansorge, O.; Talbot, K.; Turner, M. Towards a TDP-43-Based Biomarker for ALS and FTLD. Mol. Neurobiol. 2018, 55, 7789–7801. [Google Scholar] [CrossRef] [Green Version]
- Verstraete, E.; Kuiperij, H.B.; Van Blitterswijk, M.M.; Veldink, J.H.; Schelhaas, H.J.; Van Den Berg, L.H.; Verbeek, M.M. TDP-43 plasma levels are higher in amyotrophic lateral sclerosis. Amyotroph. Lateral Scler. 2012, 13, 446–451. [Google Scholar] [CrossRef]
- De Marco, G.; Lomartire, A.; Calvo, A.; Risso, A.; De Luca, E.; Mostert, M.; Chiò, A. Monocytes of patients with amyotrophic lateral sclerosis linked to gene mutations display altered TDP-43 subcellular distribution. Neuropathol. Appl. Neurobiol. 2017, 43, 133–153. [Google Scholar] [CrossRef]
- De Marco, G.; Lupino, E.; Calvo, A.; Moglia, C.; Buccinna, B.; Grifoni, S.; Chio, A. Cytoplasmic accumulation of TDP-43 in circulating lymphomonocytes of ALS patients with and without TARDBP mutations. Acta Neuropathol. 2011, 121, 611–622. [Google Scholar] [CrossRef] [PubMed]
- Junttila, A.; Kuvaja, M.; Hartikainen, P.; Siloaho, M.; Helisalmi, S.; Moilanen, V.; Kiviharju, A.; Jansson, L.; Tienari, P.J.; Remes, A.M.; et al. Cerebrospinal Fluid TDP-43 in Frontotemporal Lobar Degeneration and Amyotrophic Lateral Sclerosis Patients with and without the C9ORF72 Hexanucleotide Expansion. Dement. Geriatr. Cogn. Disord. Extra 2016, 6, 142–149. [Google Scholar] [CrossRef] [PubMed]
- Noto, Y.-I.; Shibuya, K.; Sato, Y.; Kanai, K.; Misawa, S.; Sawai, S.; Mori, M.; Uchiyama, T.; Isose, S.; Nasu, S.; et al. Elevated CSF TDP-43 levels in amyotrophic lateral sclerosis: Specificity, sensitivity, and a possible prognostic value. Amyotroph. Lateral Scler. 2010, 12, 140–143. [Google Scholar] [CrossRef]
- Kasai, T.; Tokuda, T.; Ishigami, N.; Sasayama, H.; Foulds, P.; Mitchell, D.J.; Mann, D.M.A.; Allsop, D.; Nakagawa, M. Increased TDP-43 protein in cerebrospinal fluid of patients with amyotrophic lateral sclerosis. Acta Neuropathol. 2009, 117, 55–62. [Google Scholar] [CrossRef]
- Steinacker, P.; Hendrich, C.; Sperfeld, A.D.; Jesse, S.; von Arnim, C.A.F.; Lehnert, S.; Pabst, A.; Uttner, I.; Tumani, H.; Lee, V.M.-Y.; et al. TDP-43 in Cerebrospinal Fluid of Patients with Frontotemporal Lobar Degeneration and Amyotrophic Lateral Sclerosis. Arch. Neurol. 2008, 65, 1481–1487. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Olsson, B.; Lautner, R.; Andreasson, U.; Öhrfelt, A.; Portelius, E.; Bjerke, M.; Hölttä, M.; Rosén, C.; Olsson, C.; Strobel, G.; et al. CSF and blood biomarkers for the diagnosis of Alzheimer’s disease: A systematic review and meta-analysis. Lancet Neurol. 2016, 15, 673–684. [Google Scholar] [CrossRef]
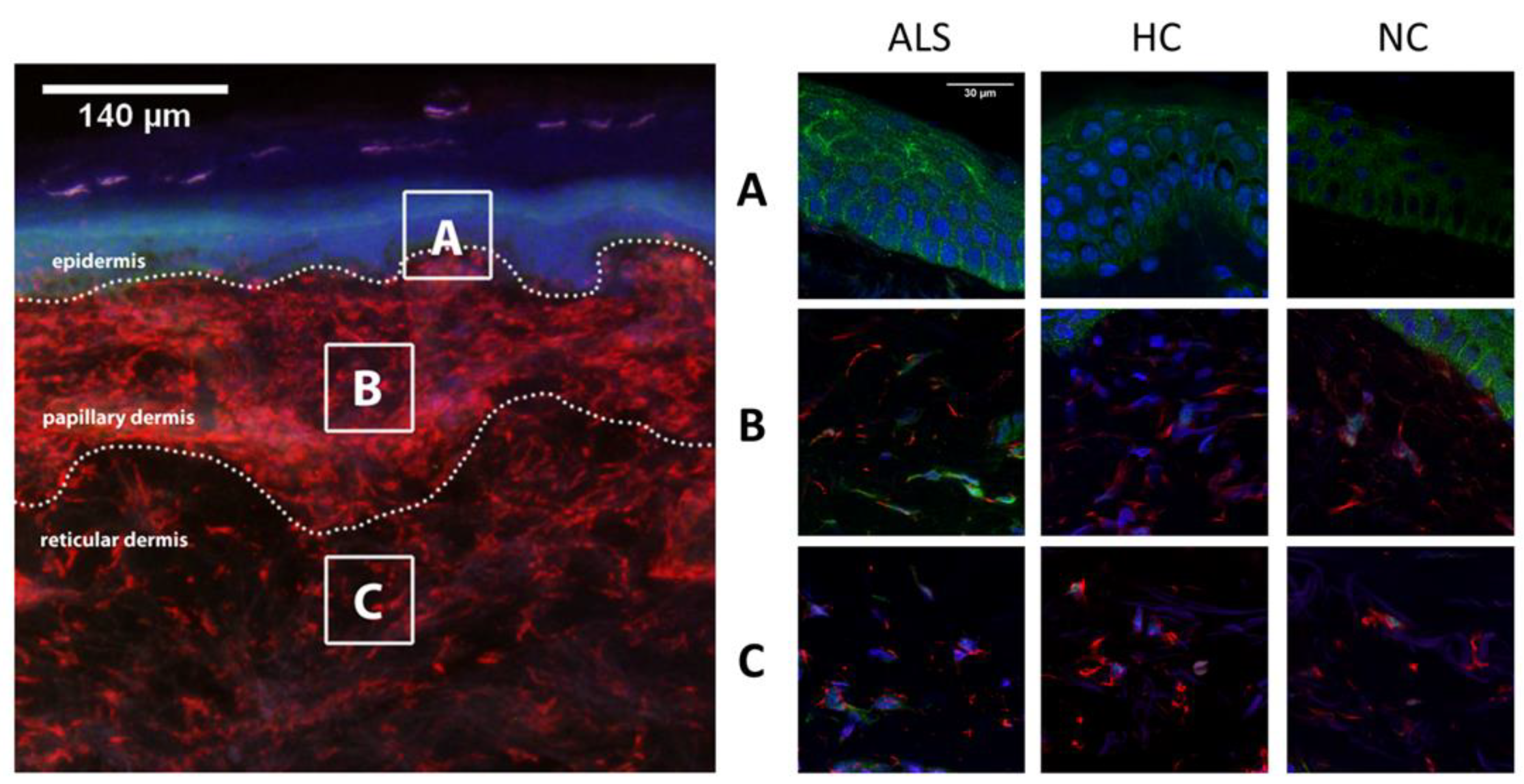
| ALS (N = 44) | HC (N = 10) | NC (N = 10) | p Value | |
|---|---|---|---|---|
| Epidermis | ||||
| % TDP-43 IR | 12.94 ± 1.05 | 7.19 ± 0.97 | 4.37 ± 1.25 | <0.001 |
| Papillary Dermis | ||||
| % TDP-43 IR | 1.33 ± 0.17 | 0.13 ± 0.05 | 0.31 ± 0.07 | <0.001 |
| % Cytoplasmic TDP-43+ cells | 50.88 ± 3.91 | 19.11 ± 5.2 | 16.58 ± 4.85 | <0.001 |
| Reticular Dermis | ||||
| % TDP-43 IR | 0.82 ± 0.09 | 0.18 ± 0.07 | 0.10 ± 0.03 | <0.001 |
| % Cytoplasmic TDP-43+ cells | 50.36 ± 3.64 | 21.73 ± 6.40 | 11.75 ± 5.88 | <0.001 |
| Epidermis | Superficial Dermis | Deep Dermis | |||
|---|---|---|---|---|---|
| % TDP-43 IR | % TDP-43 IR | % Cytoplasmic TDP-43+ Cells | % TDP-43 IR | % Cytoplasmic TDP-43+ Cells | |
| Gender | |||||
| male female | 14.66 ± 1.33 | 1.24 ± 0.19 | 47.37 ± 5.60 | 0.85 ± 0.12 | 50.96 ± 5.34 |
| 11.21 ± 1.57 | 1.43 ± 0.29 | 54.39 ± 5.49 | 0.81 ± 0.13 | 47.95 ± 4.50 | |
| p = 0.102 | p = 0.601 | p = 0.376 | p = 0.832 | p = 0.669 | |
| Site of onset | |||||
| spinal bulbar | 14.31 ± 1.21 | 1.33 ± 0.20 | 46.57 ± 5.30 | 0.98 ± 0.12 | 49.02 ± 4.40 |
| 10.52 ± 1.86 | 1.34 ± 0.34 | 58.43 ± 5.12 | 0.55 ± 0.08 | 50.21 ± 5.76 | |
| p = 0.083 | p = 0.991 | p = 0.147 | p = 0.017 | p = 0.871 | |
| Age | R = −0.050 | R = −0.180 | R = −0.199 | R = 0.043 | R = −0.114 |
| p = 0.747 | p = 0.243 | p = 0.194 | p = 0.780 | p = 0.460 | |
| Time from onset to biopsy | R = 0.071 | R = 0.032 | R = −0.115 | R = 0.053 | R = −0.119 |
| p = 0.646 | p = 0.836 | p = 0.457 | p = 0.731 | p = 0.440 | |
| ALSFRS-R slope | R = −0.169 | R = −0.205 | R = −0.046 | R = −0.086 | R = 0.057 |
| p = 0.277 | p = 0.186 | p = 0.772 | p = 0.585 | p = 0.717 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rubio, M.A.; Herrando-Grabulosa, M.; Velasco, R.; Blasco, I.; Povedano, M.; Navarro, X. TDP-43 Cytoplasmic Translocation in the Skin Fibroblasts of ALS Patients. Cells 2022, 11, 209. https://doi.org/10.3390/cells11020209
Rubio MA, Herrando-Grabulosa M, Velasco R, Blasco I, Povedano M, Navarro X. TDP-43 Cytoplasmic Translocation in the Skin Fibroblasts of ALS Patients. Cells. 2022; 11(2):209. https://doi.org/10.3390/cells11020209
Chicago/Turabian StyleRubio, Miguel A., Mireia Herrando-Grabulosa, Roser Velasco, Israel Blasco, Monica Povedano, and Xavier Navarro. 2022. "TDP-43 Cytoplasmic Translocation in the Skin Fibroblasts of ALS Patients" Cells 11, no. 2: 209. https://doi.org/10.3390/cells11020209
APA StyleRubio, M. A., Herrando-Grabulosa, M., Velasco, R., Blasco, I., Povedano, M., & Navarro, X. (2022). TDP-43 Cytoplasmic Translocation in the Skin Fibroblasts of ALS Patients. Cells, 11(2), 209. https://doi.org/10.3390/cells11020209







