Extend the Survival of Human Sperm In Vitro in Non-Freezing Conditions: Damage Mechanisms, Preservation Technologies, and Clinical Applications
Abstract
1. Introduction
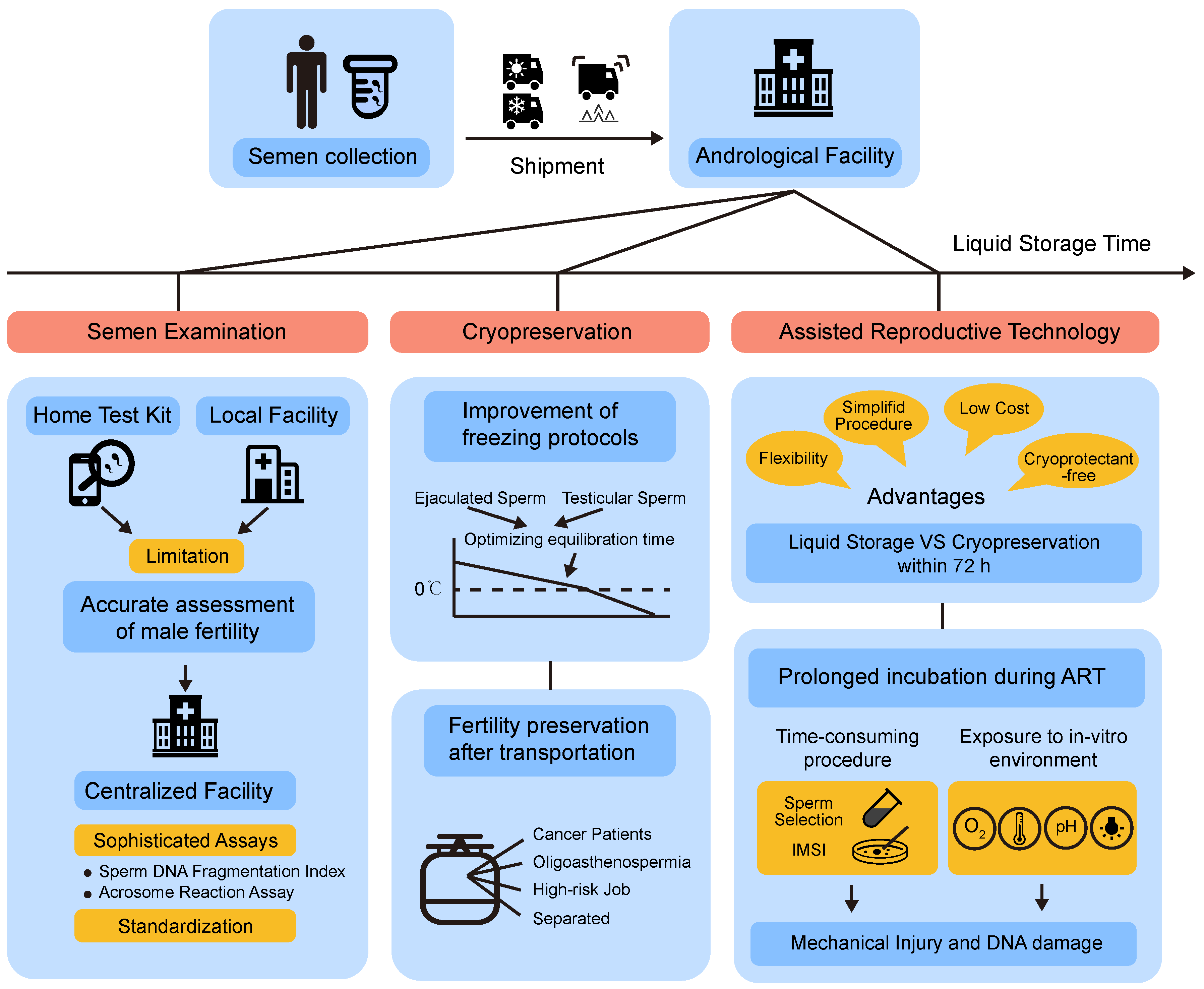
2. Aims of Prolonged Non-Freezing Storage
2.1. Laboratorial Test
2.2. Assisted Reproductive Technology (ART)
2.3. Cryopreservation
2.4. Shipment of Semen Samples
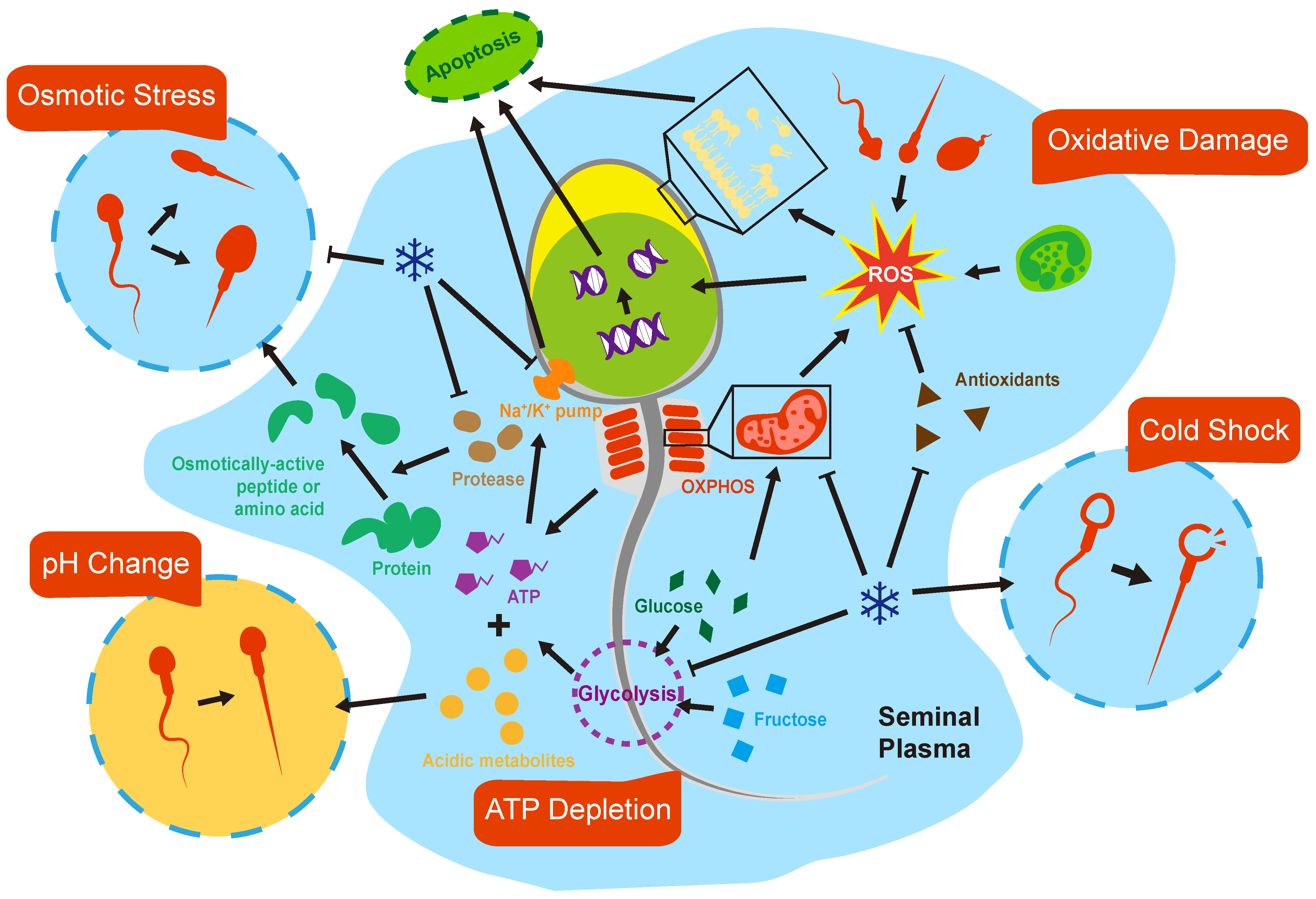
3. Sperm Injuries during Prolonged Non-Freezing Storage
3.1. pH and Osmolarity
3.2. Metabolism and ATP Depletion
3.3. Oxidative Stress
3.4. Cold Shock
4. Effects of Extended Non-Freezing Storage of Human Spermatozoa on Semen Examination and Pregnancy Outcomes
4.1. Sperm Parameters
4.2. Pregnancy Outcome
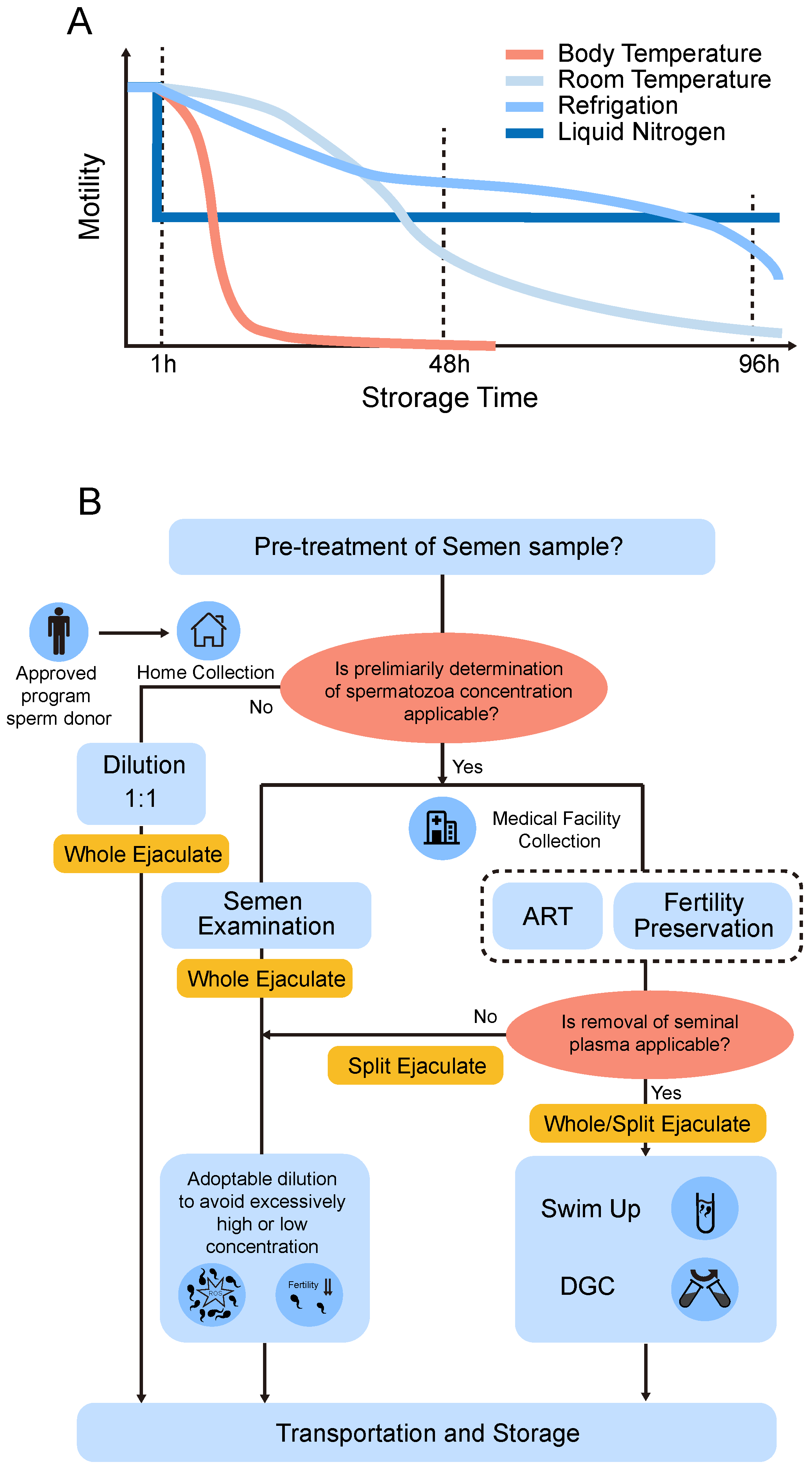
5. Storage Time and Temperature
6. Pre-Treatment of Semen
6.1. Semen Processing and the Effect of Seminal Plasma
6.2. The Split Ejaculate Collection for Non-Freezing Storage of Human Sperm
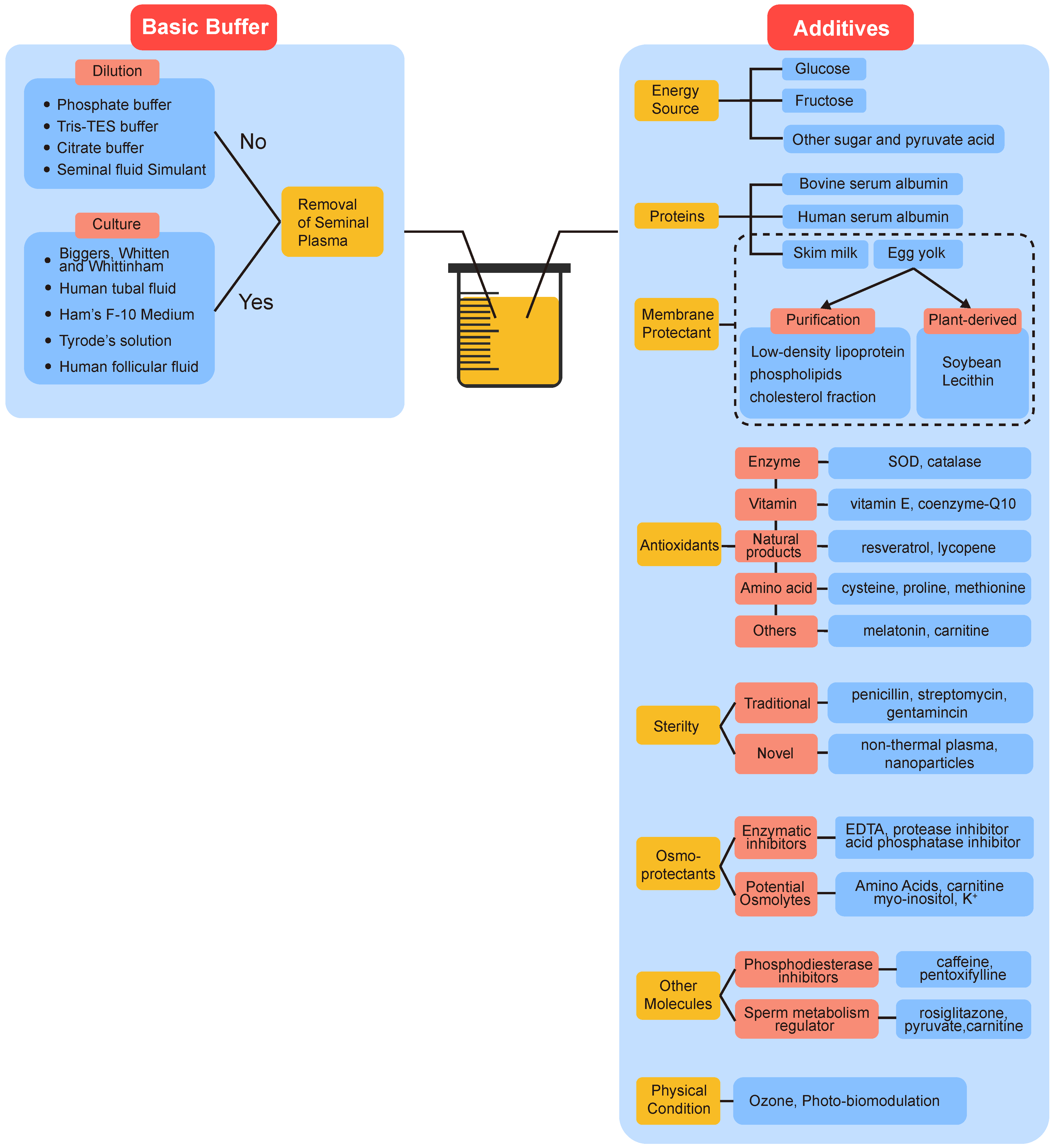
7. Semen Extenders Used in Non-Freezing Storage of Spermatozoa
7.1. Basic Buffer and Dilution Ratio
7.2. Additives
7.2.1. Energy Sources, Proteins, and Antibiotics
7.2.2. Membrane Protectant
7.2.3. Antioxidants
7.2.4. Osmoprotectants
7.2.5. Other Molecules
8. Typical Extenders Used to Store Human Spermatozoa in Liquid State
8.1. Refrigeration Medium-TEST-Yolk Buffer with Gentamicin (Commercial Agents)
8.2. Citrate TEST-Yolk Buffer (CYB)
8.3. Electrolyte-Free Solution (EFM)
8.4. Artificial Seminal Fluid
9. Future Perspective
9.1. Nanotechnology and Drug Delivery System
9.2. Omics Technologies
Author Contributions
Funding
Conflicts of Interest
References
- Makler, A.; Zaidise, I.; Paldi, E.; Brandes, J.M. Factors affecting sperm motility. I. In vitro change in motility with time after ejaculation. Fertil. Steril. 1979, 31, 147–154. [Google Scholar] [CrossRef]
- Makler, A.; Urbach, Y.; Lefler, E.; Merzbach, D. Factors affecting sperm motility. Vi. Sperm viability under the influence of bacterial growth in human ejaculates. Fertil. Steril. 1981, 35, 666–670. [Google Scholar] [CrossRef]
- Makler, A.; David, R.; Blumenfeld, Z.; Better, O.S. Factors affecting sperm motility. Vii. Sperm viability as affected by change of ph and osmolarity of semen and urine specimens. Fertil. Steril. 1981, 36, 507–511. [Google Scholar] [CrossRef]
- Makler, A.; Deutch, M.; Vilensky, A.; Palti, Y. Factors affecting sperm motility. Viii. Velocity and survival of human spermatozoa as related to temperatures above zero. Int. J. Androl. 1981, 4, 559–569. [Google Scholar] [CrossRef] [PubMed]
- Cicaré, J.; Caille, A.; Zumoffen, C.; Ghersevich, S.; Bahamondes, L.; Munuce, M.J. In vitro incubation of human spermatozoa promotes reactive oxygen species generation and DNA fragmentation. Andrologia 2015, 47, 861–866. [Google Scholar] [PubMed]
- Holmes, E.; Björndahl, L.; Kvist, U. Post-ejaculatory increase in human semen osmolality in vitro. Andrologia 2019, 51, e13311. [Google Scholar] [CrossRef] [PubMed]
- Pini, T.; Leahy, T.; de Graaf, S.P. Sublethal sperm freezing damage: Manifestations and solutions. Theriogenology 2018, 118, 172–181. [Google Scholar] [CrossRef]
- Sacha, C.R.; Vagios, S.; Hammer, K.; Fitz, V.; Souter, I.; Bormann, C.L. The effect of semen collection location and time to processing on sperm parameters and early ivf/icsi outcomes. J. Assist. Reprod. Genet. 2021, 38, 1449–1457. [Google Scholar] [CrossRef]
- Stimpfel, M.; Jancar, N.; Vrtacnik-Bokal, E. Collecting semen samples at home for ivf/icsi does not negatively affect the outcome of the fresh cycle. Reprod. Biomed. Online 2021, 42, 391–399. [Google Scholar] [CrossRef]
- Isaev, D.A.; Kapralova, I.V.; Zakharova, E.V.; Kartavenko, T.V.; Krivokharchenko, I.S.; Zharskaya, O.O.; Zaletova, V.V. Short-term hypothermic storage of human spermatozoa in electrolyte free medium (efm): Outcomes of 96 ivf cycles. Hum. Reprod. 2015, 30, 154–155. [Google Scholar]
- Riel, J.M.; Huang, T.T.F.; Ward, M.A. Freezing-free preservation of human spermatozoa–a pilot study. Arch. Androl. 2007, 53, 275–284. [Google Scholar] [CrossRef][Green Version]
- Torra-Massana, M.; Quintana-Vehí, A.; Barragán, M.; Bellido, R.; Rodríguez, A.; Vassena, R. How long can the sperm wait? Effect of incubation time on icsi outcomes. Mol. Reprod Dev. 2022, 89, 133–145. [Google Scholar] [CrossRef]
- Nabi, A.; Khalili, M.A.; Halvaei, I.; Roodbari, F. Prolonged incubation of processed human spermatozoa will increase DNA fragmentation. Andrologia 2014, 46, 374–379. [Google Scholar] [CrossRef]
- Hosseini, A.; Khalili, M.A. Improvement of motility after culture of testicular spermatozoa: The effects of incubation timing and temperature. Transl. Androl. Urol. 2017, 6, 271–276. [Google Scholar] [CrossRef]
- Uribe, P.; Rojas, C.; Merino, J.; Zambrano, F.; Villegas, J.V.; Treulen, F.; Boguen, R.; Isachenko, V.; Isachenko, E.; Sanchez, R. Effect of incubation temperature after devitrification on quality parameters in human sperm cells. Cryobiology 2017, 79, 78–81. [Google Scholar] [CrossRef]
- Gibb, Z.; Aitken, R.J. Recent developments in stallion semen preservation. J. Equine Vet. Sci. 2016, 43, S29–S36. [Google Scholar] [CrossRef]
- Baldi, E.; Muratori, M.; Marchiani, S.; Tamburrino, L.; Degl’Innocenti, S. Main effects of in vitro manipulation of human spermatozoa. In Proceedings of the XIIIth International Symposium on Spermatology, Stockholm, Sweden, 9–13 May 2021; pp. 263–272. [Google Scholar]
- Phillips, P.H. Preservation of bull semen. J. Biol. Chem. 1939, 130, 415. [Google Scholar] [CrossRef]
- Phillips, P.H.; Lardy, H.A. A yolk-buffer pabulum for the preservation of bull semen. J. Dairy Sci. 1940, 23, 399–404. [Google Scholar] [CrossRef]
- Phillips, P.H.; Spitzer, R.R. A synthetic pabulum for the preservation of bull semen. J. Dairy Sci. 1946, 29, 407–414. [Google Scholar] [CrossRef]
- Jaskey, D.G.; Cohen, M.R. Twenty-four to ninety-six-hour storage of human spermatozoa in test-yolk buffer. Fertil Steril 1981, 35, 205–208. [Google Scholar] [CrossRef]
- Aitken, R.J.; Allan, I.W.; Irvine, D.S.; Macnamee, M. Studies on the development of diluents for the transportation and storage of human semen at ambient temperature. Hum. Reprod. 1996, 11, 2186–2196. [Google Scholar] [CrossRef]
- Allan, I.W.; Irvine, D.S.; Macnamee, M.; Aitken, R.J. Field trial of a diluent for the transportation of human semen at ambient temperatures. Fertil. Steril. 1997, 67, 348–354. [Google Scholar] [CrossRef]
- Agarwal, A.; Sharma, R.; Gupta, S.; Sharma, R. Nextgen® home sperm banking kit: Outcomes of offsite vs onsite collection—preliminary findings. Urology 2015, 85, 1339–1346. [Google Scholar] [CrossRef]
- Gualtieri, R.; Kalthur, G.; Barbato, V.; Longobardi, S.; Di Rella, F.; Adiga, S.K.; Talevi, R. Sperm oxidative stress during in vitro manipulation and its effects on sperm function and embryo development. Antioxidants 2021, 10, 1025. [Google Scholar] [CrossRef]
- Kobori, Y. Home testing for male factor infertility: A review of current options. Fertil. Steril. 2019, 111, 864–870. [Google Scholar] [CrossRef]
- Huszar, G.; Celik-Ozenci, C.; Cayli, S.; Kovacs, T.; Vigue, L.; Kovanci, E. Semen characteristics after overnight shipping: Preservation of sperm concentrations, hspa2 ratios, ck activity, cytoplasmic retention, chromatin maturity, DNA integrity, and sperm shape. J. Androl. 2004, 25, 593–604. [Google Scholar] [CrossRef]
- Sati, L.; Bennett, D.; Janes, M.; Huszar, G. Next day determination of ejaculatory sperm motility after overnight shipment of semen to remote locations. J. Assist. Reprod. Genet. 2015, 32, 117–125. [Google Scholar] [CrossRef]
- McEvoy, A.; Roberts, P.; Yap, K.; Matson, P. Development of a simplified method of human semen storage for the testing of sperm DNA fragmentation using the halosperm g2 test kit. Fertil. Steril. 2014, 102, 981–988. [Google Scholar] [CrossRef]
- Wiebke, M.; Hensel, B.; Nitsche-Melkus, E.; Jung, M.; Schulze, M. Cooled storage of semen from livestock animals (part i): Boar, bull, and stallion. Anim. Reprod. Sci. 2021, 106822. [Google Scholar] [CrossRef] [PubMed]
- Zavos, P.M.; Correa, J.B.; Sofikitis, N.; Kofinas, G.D.; Zarmakoupis, P.N. A method of short-term cryostorage and selection of viable sperm for use in the various assisted reproductive techniques. Tohoku J. Exp. Med. 1995, 176, 75–81. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Zavos, P.M.; Goodpasture, J.C.; Zaneveld, L.J.; Cohen, M.R. Motility and enzyme activity of human spermatozoa stored for 24 hours at +5 degrees c and -196 degrees c. Fertil. Steril. 1980, 34, 607–609. [Google Scholar] [CrossRef]
- Kofinas, G.D.; Zavos, P.M. Short term cryostorage technique for human spermatozoa: Its possible application in an artificial insemination program. Infertility 1992, 15, 44–54. [Google Scholar]
- Riel, J.M.; Yamauchi, Y.; Huang, T.T.; Grove, J.; Ward, M.A. Short-term storage of human spermatozoa in electrolyte-free medium without freezing maintains sperm chromatin integrity better than cryopreservation. Biol. Reprod. 2011, 85, 536–547. [Google Scholar] [CrossRef]
- Kofinas, G.D.; Zavos, P.M. Selection of viable spermatozoa via sperm filtration following 24 h cryostorage at 5 °C in test-yolk buffer. Mol. Androl. 1992, 4, 113–119. [Google Scholar]
- Zollner, U.; Martin, S.; Liebermann, J.; Steck, T. Evaluation of a cut-off value for sperm motility after different hours of incubation to select the suitable reproductive technology (ivf or icsi). Acta Obs. Gynecol. Scand. 1999, 78, 326–331. [Google Scholar]
- Hossain, A.M.; Osuamkpe, C.O.; Nagamani, M. Extended culture of human spermatozoa in the laboratory may have practical value in the assisted reproductive procedures. Fertil. Steril. 2008, 89, 237–239. [Google Scholar] [CrossRef]
- World Health Organization (WHO). Who Laboratory Manual for the Examination and Processing of Human Semen; WHO: Geneva, Switzerland, 2021. [Google Scholar]
- Balasuriya, A.; Serhal, P.; Doshi, A.; Harper, J.C. Processes involved in assisted reproduction technologies significantly increase sperm DNA fragmentation and phosphatidylserine translocation. Andrologia 2014, 46, 86–97. [Google Scholar] [CrossRef]
- Peer, S.; Eltes, F.; Berkovitz, A.; Yehuda, R.; Itsykson, P.; Bartoov, B. Is fine morphology of the human sperm nuclei affected by in vitro incubation at 37 degrees c? Fertil Steril 2007, 88, 1589–1594. [Google Scholar] [CrossRef]
- Voigt, A.L.; Thiageswaran, S.; de Lima, E.M.L.N.; Dobrinski, I. Metabolic requirements for spermatogonial stem cell establishment and maintenance in vivo and in vitro. Int. J. Mol. Sci. 2021, 22, 1988. [Google Scholar] [CrossRef]
- Ibtisham, F.; Honaramooz, A. Spermatogonial stem cells for in vitro spermatogenesis and in vivo restoration of fertility. Cells 2020, 9, 745. [Google Scholar] [CrossRef]
- Donnez, J.; Dolmans, M.M. Fertility preservation in men and women: Where are we in 2021? Are we rising to the challenge? Fertil. Steril. 2021, 115, 1089–1090. [Google Scholar] [CrossRef]
- Brannigan, R.E.; Fantus, R.J.; Halpern, J.A. Fertility preservation in men: A contemporary overview and a look toward emerging technologies. Fertil. Steril. 2021, 115, 1126–1139. [Google Scholar] [CrossRef]
- Agarwal, A.; Sharma, R.; Singh, A.; Gupta, S.; Sharma, R. Standardisation of a novel sperm banking kit–nextgen®–to preserve sperm parameters during shipment. Andrologia 2016, 48, 662–669. [Google Scholar] [CrossRef]
- Li, Y.X.; Zhou, L.; Lv, M.Q.; Ge, P.; Liu, Y.C.; Zhou, D.X. Vitrification and conventional freezing methods in sperm cryopreservation: A systematic review and meta-analysis. Eur. J. Obs. Gynecol. Reprod. Biol. 2019, 233, 84–92. [Google Scholar] [CrossRef]
- Le, M.T.; Nguyen, T.T.T.; Nguyen, T.T.; Nguyen, V.T.; Nguyen, T.T.A.; Nguyen, V.Q.H.; Cao, N.T. Cryopreservation of human spermatozoa by vitrification versus conventional rapid freezing: Effects on motility, viability, morphology and cellular defects. Eur. J. Obs. Gynecol. Reprod. Biol. 2019, 234, 14–20. [Google Scholar] [CrossRef]
- Sieme, H.; Oldenhof, H.; Wolkers, W.F. Sperm membrane behaviour during cooling and cryopreservation. Reprod. Domest. Anim. 2015, 50 (Suppl. 3), 20–26. [Google Scholar] [CrossRef]
- Paul, R.K.; Balaganur, K.; Kumar, D.; Singh, R. Pre-freezing equilibration for 22 h improves post-thaw sperm functions in cryopreserved ram semen by reducing cholesterol efflux. Cryobiology 2020, 96, 76–84. [Google Scholar] [CrossRef]
- Torres, M.A.; Pedrosa, A.C.; Novais, F.J.; Alkmin, D.V.; Cooper, B.R.; Yasui, G.S.; Fukumasu, H.; Machaty, Z.; de Andrade, A.F.C. Metabolomic signature of spermatozoa established during holding time is responsible for differences in boar sperm freezability. Biol. Reprod. 2021, 106, 213–226. [Google Scholar] [CrossRef]
- Zavos, P.M.; Zavos, P.N.; Kaskar, K.; Correa-Perez, J.R.; Koundouros, S. Viability and fecundity of human semen specimens cryostored and transported at 5 degrees c using the bio-tranz shipping. Arch. Androl. 2006, 52, 149–154. [Google Scholar] [CrossRef]
- Kirilenko, I.A.; Vinokurov, A.A.; Danilov, V.P.; Barchukov, V.G.; Efimenko, I.A. Use of glass-forming water–salt systems for preparing human sperm to hypothermic semen preservation. Russ. J. Inorg. Chem. 2020, 65, 989–991. [Google Scholar] [CrossRef]
- Ma, Y.; Gao, L.; Tian, Y.; Chen, P.; Yang, J.; Zhang, L. Advanced biomaterials in cell preservation: Hypothermic preservation and cryopreservation. Acta Biomater. 2021, 131, 97–116. [Google Scholar] [CrossRef] [PubMed]
- Hamamah, S.; Magnoux, E.; Royere, D.; Barthelemy, C.; Dacheux, J.L.; Gatti, J.L. Internal ph of human spermatozoa: Effect of ions, human follicular fluid and progesterone. Mol. Hum. Reprod. 1996, 2, 219–224. [Google Scholar] [CrossRef] [PubMed]
- Swain, J.E. Optimizing the culture environment in the ivf laboratory: Impact of ph and buffer capacity on gamete and embryo quality. Reprod. Biomed. Online 2010, 21, 6–16. [Google Scholar] [CrossRef] [PubMed]
- Owen, D.H.; Katz, D.F. A review of the physical and chemical properties of human semen and the formulation of a semen simulant. J. Androl. 2005, 26, 459–469. [Google Scholar] [CrossRef]
- Dhumal, S.S.; Naik, P.; Dakshinamurthy, S.; Sullia, K. Semen ph and its correlation with motility and count—A study in subfertile men. JBRA Assist. Reprod. 2021, 25, 172–175. [Google Scholar] [CrossRef]
- Liu, C.H.; Dong, H.B.; Ma, D.L.; Li, Y.W.; Han, D.; Luo, M.J.; Chang, Z.L.; Tan, J.H. Effects of ph during liquid storage of goat semen on sperm viability and fertilizing potential. Anim. Reprod. Sci. 2016, 164, 47–56. [Google Scholar] [CrossRef]
- Saito, K.; Kinoshita, Y.; Kanno, H.; Iwasaki, A. The role of potassium ion and extracellular alkalization in reinitiation of human spermatozoa preserved in electrolyte-free solution at 4 degrees c. Fertil. Steril. 1996, 65, 1214–1218. [Google Scholar] [CrossRef]
- Mandal, A.; Bhattacharyya, A.K. Differences in osmolality, ph, buffering capacity, superoxide dismutase and maintenance of sperm motility in human ejaculates according to the degree of coagulation. Int. J. Androl. 1988, 11, 45–51. [Google Scholar] [CrossRef]
- Cooper, T.G.; Barfield, J.P.; Yeung, C.H. Changes in osmolality during liquefaction of human semen. Int. J. Androl. 2005, 28, 58–60. [Google Scholar] [CrossRef]
- Gopalkrishman, K.; Hinduja, I.N.; Kumar, T.C. Determining the osmolality of seminal fluid aids in the rapid diagnosis of the fertilizing potential of spermatozoa. J. Vitr. Fert. Embryo. Transf. 1989, 6, 119–121. [Google Scholar] [CrossRef]
- Velázquez, A.; Pedrón, N.; Delgado, N.M.; Rosado, A. Osmolality and conductance of normal and abnormal human seminal plasma. Int. J. Fertil. 1977, 22, 92–97. [Google Scholar]
- Rossato, M.; Balercia, G.; Lucarelli, G.; Foresta, C.; Mantero, F. Role of seminal osmolarity in the reduction of human sperm motility. Int. J. Androl. 2002, 25, 230–235. [Google Scholar] [CrossRef]
- Holmes, E.; Björndahl, L.; Kvist, U. Possible factors influencing post-ejaculatory changes of the osmolality of human semen in vitro. Andrologia 2019, 51, e13443. [Google Scholar] [CrossRef]
- Yeung, C.H.; Barfield, J.P.; Cooper, T.G. Physiological volume regulation by spermatozoa. Mol. Cell. Endocrinol. 2006, 250, 98–105. [Google Scholar] [CrossRef]
- Boatman, D.E. Responses of gametes to the oviductal environment. Hum. Reprod. 1997, 12, 133–149. [Google Scholar]
- Cooper, T.G.; Yeung, C.H. Acquisition of volume regulatory response of sperm upon maturation in the epididymis and the role of the cytoplasmic droplet. Microsc. Res. Tech. 2003, 61, 28–38. [Google Scholar] [CrossRef]
- Barati, F.; Papahn, A.A.; Afrough, M.; Barati, M. Effects of tyrode’s solution osmolarities and milk on bull sperm storage above zero temperatures. Iran J. Reprod. Med. 2011, 9, 25–30. [Google Scholar]
- Holmes, E.; Björndahl, L.; Kvist, U. Hypotonic challenge reduces human sperm motility through coiling and folding of the tail. Andrologia 2020, 52, e13859. [Google Scholar] [CrossRef]
- du Plessis, S.S.; Agarwal, A.; Mohanty, G.; van der Linde, M. Oxidative phosphorylation versus glycolysis: What fuel do spermatozoa use? Asian J. Androl. 2015, 17, 230–235. [Google Scholar] [CrossRef]
- Storey, B.T. Mammalian sperm metabolism: Oxygen and sugar, friend and foe. Int. J. Dev. Biol. 2008, 52, 427–437. [Google Scholar] [CrossRef]
- Piomboni, P.; Focarelli, R.; Stendardi, A.; Ferramosca, A.; Zara, V. The role of mitochondria in energy production for human sperm motility. Int. J. Androl. 2012, 35, 109–124. [Google Scholar] [CrossRef]
- Castellini, C.; D’Andrea, S.; Cordeschi, G.; Totaro, M.; Parisi, A.; Di Emidio, G.; Tatone, C.; Francavilla, S.; Barbonetti, A. Pathophysiology of mitochondrial dysfunction in human spermatozoa: Focus on energetic metabolism, oxidative stress and apoptosis. Antioxidants 2021, 10, 695. [Google Scholar] [CrossRef]
- Peterson, R.N.; Freund, M. Factors affecting fructose utilization and lactic acid formation by human semen. The role of glucose and pyruvic acid. Fertil. Steril. 1971, 22, 639–644. [Google Scholar] [CrossRef]
- Suominen, J. Seminal fructose and glucose in asthenozoospermia. Int. J. Androl. 2001, 24, 253–254. [Google Scholar] [CrossRef]
- Calvert, S.J.; Reynolds, S.; Paley, M.N.; Walters, S.J.; Pacey, A.A. Probing human sperm metabolism using 13c-magnetic resonance spectroscopy. Mol. Hum. Reprod. 2019, 25, 30–41. [Google Scholar] [CrossRef]
- Reynolds, S.; Ismail, N.F.B.; Calvert, S.J.; Pacey, A.A.; Paley, M.N.J. Evidence for rapid oxidative phosphorylation and lactate fermentation in motile human sperm by hyperpolarized (13)c magnetic resonance spectroscopy. Sci. Rep. 2017, 7, 4322. [Google Scholar] [CrossRef]
- Chianese, R.; Pierantoni, R. Mitochondrial reactive oxygen species (ros) production alters sperm quality. Antioxidants 2021, 10, 92. [Google Scholar] [CrossRef]
- Gibb, Z.; Aitken, R.J. The impact of sperm metabolism during in vitro storage: The stallion as a model. Biomed Res. Int. 2016, 2016, 9380609. [Google Scholar] [CrossRef] [PubMed]
- Walczak-Jedrzejowska, R.; Wolski, J.K.; Slowikowska-Hilczer, J. The role of oxidative stress and antioxidants in male fertility. Cent. Eur. J. Urol. 2013, 66, 60–67. [Google Scholar] [CrossRef]
- Evans, E.P.P.; Scholten, J.T.M.; Mzyk, A.; Reyes-San-Martin, C.; Llumbet, A.E.; Hamoh, T.; Arts, E.; Schirhagl, R.; Cantineau, A.E.P. Male subfertility and oxidative stress. Redox Biol. 2021, 46, 102071. [Google Scholar] [CrossRef]
- Bisht, S.; Faiq, M.; Tolahunase, M.; Dada, R. Oxidative stress and male infertility. Nat. Rev. Urol. 2017, 14, 470–485. [Google Scholar] [CrossRef] [PubMed]
- Silvestre, M.A.; Yániz, J.L.; Peña, F.J.; Santolaria, P.; Castelló-Ruiz, M. Role of antioxidants in cooled liquid storage of mammal spermatozoa. Antioxidants 2021, 10, 1096. [Google Scholar] [CrossRef] [PubMed]
- Koppers, A.J.; De Iuliis, G.N.; Finnie, J.M.; McLaughlin, E.A.; Aitken, R.J. Significance of mitochondrial reactive oxygen species in the generation of oxidative stress in spermatozoa. J. Clin. Endocrinol. Metab. 2008, 93, 3199–3207. [Google Scholar] [CrossRef]
- Dada, R.; Bisht, S. Oxidative stress and male infertility. In Male Infertility: Understanding, Causes and Treatment; Springer: Berlin/Heidelberg, Germany, 2017; pp. 151–165. [Google Scholar]
- Aitken, R.J. Reactive oxygen species as mediators of sperm capacitation and pathological damage. Mol. Reprod. Dev. 2017, 84, 1039–1052. [Google Scholar] [CrossRef]
- Esfandiari, N.; Saleh, R.A.; Blaut, A.P.; Sharma, R.K.; Nelson, D.R.; Thomas, A.J.; Falcone, T.; Agarwal, A. Effects of temperature on sperm motion characteristics and reactive oxygen species. Int. J. Fertil. Womens Med. 2002, 47, 227–235. [Google Scholar]
- Gupta, S.; Finelli, R.; Agarwal, A.; Henkel, R. Total antioxidant capacity-relevance, methods and clinical implications. Andrologia 2021, 53, e13624. [Google Scholar] [CrossRef]
- Benedetti, S.; Tagliamonte, M.C.; Catalani, S.; Primiterra, M.; Canestrari, F.; De Stefani, S.; Palini, S.; Bulletti, C. Differences in blood and semen oxidative status in fertile and infertile men, and their relationship with sperm quality. Reprod. Biomed. Online 2012, 25, 300–306. [Google Scholar] [CrossRef]
- Barranco, I.; Rubio, C.P.; Tvarijonaviciute, A.; Rodriguez-Martinez, H.; Roca, J. Measurement of oxidative stress index in seminal plasma can predict in vivo fertility of liquid-stored porcine artificial insemination semen doses. Antioxidants 2021, 10, 1203. [Google Scholar] [CrossRef]
- White, I.G. Lipids and calcium uptake of sperm in relation to cold shock and preservation: A review. Reprod. Fertil. Dev. 1993, 5, 639–658. [Google Scholar] [CrossRef]
- Wales, R.G.; White, I.G. The susceptibility of spermatozoa to temperature shock. J. Endocrinol. 1959, 19, 211–220. [Google Scholar] [CrossRef]
- Drobnis, E.Z.; Crowe, L.M.; Berger, T.; Anchordoguy, T.J.; Overstreet, J.W.; Crowe, J.H. Cold shock damage is due to lipid phase transitions in cell membranes: A demonstration using sperm as a model. J. Exp. Zool. 1993, 265, 432–437. [Google Scholar] [CrossRef] [PubMed]
- Darin-Bennett, A.; White, I.G. Influence of the cholesterol content of mammalian spermatozoa on susceptibility to cold-shock. Cryobiology 1977, 14, 466–470. [Google Scholar] [CrossRef]
- Aurich, C. Factors affecting the plasma membrane function of cooled-stored stallion spermatozoa. Anim. Reprod. Sci. 2005, 89, 65–75. [Google Scholar] [CrossRef]
- Moran, D.M.; Jasko, D.J.; Squires, E.L.; Amann, R.P. Determination of temperature and cooling rate which induce cold shock in stallion spermatozoa. Theriogenology 1992, 38, 999–1012. [Google Scholar] [CrossRef]
- Akhter, S.; Ansari, M.S.; Rakha, B.A.; Ullah, N.; Andrabi, S.M.; Khalid, M. In vitro evaluation of liquid-stored buffalo semen at 5 °C diluted in soya lecithin based extender (bioxcell®), tris-citric egg yolk, skim milk and egg yolk-citrate extenders. Reprod. Domest. Anim. 2011, 46, 45–49. [Google Scholar] [CrossRef]
- Xu, C.L.; Zhou, J.B.; Zhao, B.T.; Lan, G.C.; Luo, M.J.; Chang, Z.L.; Sui, H.S.; Tan, J.H. Liquid storage of goat semen in chemically defined extenders. Reprod. Domest. Anim. 2009, 44, 771–778. [Google Scholar] [CrossRef]
- Singh, A.K.; Singh, V.K.; Narwade, B.M.; Mohanty, T.K.; Atreja, S.K. Comparative quality assessment of buffalo (bubalus bubalis) semen chilled (5 °C) in egg yolk- and soya milk-based extenders. Reprod. Domest. Anim. 2012, 47, 596–600. [Google Scholar] [CrossRef]
- de Paz, P.; Esteso, M.C.; Alvarez, M.; Mata, M.; Chamorro, C.A.; Anel, L. Development of extender based on soybean lecithin for its application in liquid ram semen. Theriogenology 2010, 74, 663–671. [Google Scholar] [CrossRef]
- Vishwanath, R.; Shannon, P. Do sperm cells age? A review of the physiological changes in sperm during storage at ambient temperature. Reprod. Fertil. Dev. 1997, 9, 321–331. [Google Scholar] [CrossRef]
- Ahmed, I.; Abdelateef, S.; Laqqan, M.; Amor, H.; Abdel-Lah, M.A.; Hammadeh, M.E. Influence of extended incubation time on human sperm chromatin condensation, sperm DNA strand breaks and their effect on fertilisation rate. Andrologia 2018, 50, 8. [Google Scholar] [CrossRef]
- Pereira, R.; Sá, R.; Barros, A.; Sousa, M. Major regulatory mechanisms involved in sperm motility. Asian J. Androl. 2017, 19, 5–14. [Google Scholar] [CrossRef] [PubMed]
- Appell, R.A.; Evans, P.R. The effect of temperature on sperm motility and viability. Fertil. Steril. 1977, 28, 1329–1332. [Google Scholar] [CrossRef]
- Dondero, F.; Rossi, T.; Delfino, M.; Imbrogno, N.; Cannistrà, S.; Mazzilli, F. Human semen refrigeration at + 4 °c: Bio-kinetic characteristics. Cell Tissue Bank. 2006, 7, 61–64. [Google Scholar] [CrossRef]
- Iranpour, F.G.; Nateghian, Z.; Henkel, R.; Dashti, G.R. Effects of temperature and storage time on the motility, viability, DNA integrity and apoptosis of processed human spermatozoa. Andrologia 2020, 52, 8. [Google Scholar]
- Dashti, G.R.; Nateghian, Z.; Golshan Iranpour, F.; Golshan Iranpour, F.; Nateghian, Z.; Henkel, R.; Dashti, G.R.; Hosseini, A.; Khalili, M.A. Effect of preservation of human semen sample at 4–6 and 25 °C on sperm motility. Cell Tissue Bank. 2018, 19, 653–658. [Google Scholar] [CrossRef]
- Aitken, R.J.; Gibb, Z.; Mitchell, L.A.; Lambourne, S.R.; Connaughton, H.S.; De Iuliis, G.N. Sperm motility is lost in vitro as a consequence of mitochondrial free radical production and the generation of electrophilic aldehydes but can be significantly rescued by the presence of nucleophilic thiols. Biol. Reprod. 2012, 87, 110. [Google Scholar] [CrossRef]
- Bolanos, J.R.; Overstreet, J.W.; Katz, D.F. Human sperm penetration of zona-free hamster eggs after storage of the semen for 48 hours at 2 c to 5 c. Fertil. Steril. 1983, 39, 536–541. [Google Scholar] [CrossRef]
- Chao, H.T.; Ng, H.T.; Tsai, K.L.; Hong, C.Y.; Wei, Y.H. Human follicular fluid stimulates motility and velocity of washed human sperm in vitro. Andrologia 1992, 24, 47–51. [Google Scholar] [CrossRef]
- Karow, A.M.; Gilbert, W.B.; Black, J.B. Effects of temperature, potassium concentration, and sugar on human spermatozoa motility: A cell preservation model from reproductive medicine. Cryobiology 1992, 29, 250–254. [Google Scholar] [CrossRef]
- You, Y.-A.; Mohamed, E.A.; Oh, S.; Pang, M.-G. Optimized methods to maintain motility and viability in normozoospermic males. Clin. Exp. Reprod. Med. 2009, 36, 45–53. [Google Scholar]
- You, Y.-A.; Kwon, W.-S.; Saidur Rahman, M.; Park, Y.-J.; Kim, Y.-J.; Pang, M.-G. Sex chromosome-dependent differential viability of human spermatozoa during prolonged incubation. Hum. Reprod. 2017, 32, 1183–1191. [Google Scholar] [CrossRef] [PubMed]
- Petrella, C.; Hsieh, J.; Thrift, K.; Jarow, J.P.; Zacur, H.; Zhao, Y. Optimizing incubation conditions for the preservation of sperm motility in processed semen samples. Fertil. Steril. 2005, 84, 513–515. [Google Scholar] [CrossRef] [PubMed]
- Yenilmez, E.; Yildirmis, S.; Yulug, E.; Aydin, S.; Tekelioglu, Y.; Erdem, E.; Topbas, M.; Arvas, H. Ham’s f-10 medium and ham’s f-10 medium plus vitamin e have protective effect against oxidative stress in human semen. Urology 2006, 67, 384–387. [Google Scholar] [CrossRef] [PubMed]
- Karimi Zarchi, M.; Maleki, B.; Dehghani Ashkezari, M.; Motamed Zadeh, L.; Agha-Rahimi, A. The effects of in vitro incubation of asthenoteratozoospermic semen after density gradient centrifugation at room temperature and 37°c on sperm parameters, chromatin quality and DNA fragmentation in a short time period. J. Reprod. Infertil. 2020, 21, 275–282. [Google Scholar] [CrossRef]
- Matsuura, R.; Takeuchi, T.; Yoshida, A. Preparation and incubation conditions affect the DNA integrity of ejaculated human spermatozoa. Asian J. Androl. 2010, 12, 753–759. [Google Scholar] [CrossRef]
- Gholizadeh, L.; Agha-Rahimi, A.; Ghasemi-Esmailabad, S.; Maleki, B.; Ahamed, A.M.S.; Khalili, M.A. Impact of biological and artificial seminal fluids on sperm parameters and DNA status in asthenozoospermic ejaculates. Andrologia 2021, 53, e14018. [Google Scholar] [CrossRef]
- Amaral, A.; Paiva, C.; Baptista, M.; Sousa, A.P.; Ramalho-Santos, J. Exogenous glucose improves long-standing human sperm motility, viability, and mitochondrial function. Fertil. Steril. 2011, 96, 848–850. [Google Scholar] [CrossRef]
- Thijssen, A.; Klerkx, E.; Huyser, C.; Bosmans, E.; Campo, R.; Ombelet, W. Influence of temperature and sperm preparation on the quality of spermatozoa. Reprod. Biomed. Online 2014, 28, 436–442. [Google Scholar] [CrossRef]
- Aboulmaouahib, S.; Madkour, A.; Kaarouch, I.; Saadani, B.; Sefrioui, O.; Louanjli, N.; Copin, H.; Cadi, R.; Benkhalifa, M. Effect of semen preparation technique and its incubation on sperm quality in the moroccan population. Andrologia 2017, 49, 7. [Google Scholar] [CrossRef]
- Alamo, A.; De Luca, C.; Mongioi, L.M.; Barbagallo, F.; Cannarella, R.; La Vignera, S.; Calogero, A.E.; Condorelli, R.A. Mitochondrial membrane potential predicts 4-hour sperm motility. Biomedicines 2020, 8, 196. [Google Scholar] [CrossRef]
- Kanno, H.; Saito, K.; Ogawa, T.; Takeda, M.; Iwasaki, A.; Kinoshita, Y. Viability and function of human sperm in electrolyte-free cold preservation. Fertil. Steril. 1998, 69, 127–131. [Google Scholar] [CrossRef]
- Schuffner, A.; Morshedi, M.; Vaamonde, D.; Duran, E.H.; Oehninger, S. Effect of different incubation conditions on phosphatidylserine externalization and motion parameters of purified fractions of highly motile human spermatozoa. J. Androl. 2002, 23, 194–201. [Google Scholar]
- Jackson, R.E.; Bormann, C.L.; Hassun, P.A.; Rocha, A.M.; Motta, E.L.A.; Serafini, P.C.; Smith, G.D. Effects of semen storage and separation techniques on sperm DNA fragmentation. Fertil. Steril. 2010, 94, 2626–2630. [Google Scholar] [CrossRef]
- Tvrdá, E.; Arroyo, F.; Gosálvez, J. Dynamic assessment of human sperm DNA damage i: The effect of seminal plasma-sperm co-incubation after ejaculation. Int. Urol. Nephrol. 2018, 50, 1381–1388. [Google Scholar] [CrossRef]
- Dorado-Silva, M.; Bartolomé-Nebreda, J.; Sánchez-Martín, P.; Johnston, S.; Gosálvez, J. Co-incubation of spermatozoa with human follicular fluid reduces sperm DNA fragmentation by mitigating dnase activity in the seminal plasma. J. Assist. Reprod. Genet. 2020, 37, 63–69. [Google Scholar] [CrossRef]
- Nowicka-Bauer, K.; Nixon, B. Molecular changes induced by oxidative stress that impair human sperm motility. Antioxidants 2020, 9, 134. [Google Scholar] [CrossRef]
- Jumintono, J.; Alkubaisy, S.; Yánez Silva, D.; Singh, K.; Turki Jalil, A.; Mutia Syarifah, S.; Fakri Mustafa, Y.; Mikolaychik, I.; Morozova, L.; Derkho, M. The effect of cystamine on sperm and antioxidant parameters of ram semen stored at 4 °C for 50 hours. Arch. Razi Inst. 2021, 76, 115–1123. [Google Scholar]
- Khoi, H.X.; Shimizu, K.; Yoneda, Y.; Minagawa, I.; Abe, Y.; Kuwabara, Y.; Sasanami, T.; Kohsaka, T. Monitoring the reactive oxygen species in spermatozoa during liquid storage of boar semen and its correlation with sperm motility, free thiol content and seasonality. Andrologia 2021, 53, e14237. [Google Scholar] [CrossRef]
- Netherton, J.K.; Hetherington, L.; Ogle, R.A.; Gavgani, M.M.; Velkov, T.; Villaverde, A.I.B.; Tanphaichitr, N.; Baker, M.A. Mass spectrometry reveals new insights into the production of superoxide anions and 4-hydroxynonenal adducted proteins in human sperm. Proteomics 2020, 20, e1900205. [Google Scholar] [CrossRef]
- Baker, M.A.; Weinberg, A.; Hetherington, L.; Villaverde, A.I.; Velkov, T.; Baell, J.; Gordon, C.P. Defining the mechanisms by which the reactive oxygen species by-product, 4-hydroxynonenal, affects human sperm cell function. Biol. Reprod. 2015, 92, 108. [Google Scholar] [CrossRef]
- Fatima, S.; Alwaznah, R.; Aljuraiban, G.S.; Wasi, S.; Abudawood, M.; Abulmeaty, M.; Berika, M.Y.; Aljaser, F.S. Effect of seminal redox status on lipid peroxidation, apoptosis and DNA fragmentation in spermatozoa of infertile saudi males. Saudi Med. J. 2020, 41, 238–246. [Google Scholar] [CrossRef]
- Zhao, F.; Whiting, S.; Lambourne, S.; Aitken, R.J.; Sun, Y.P. Melatonin alleviates heat stress-induced oxidative stress and apoptosis in human spermatozoa. Free Radic. Biol. Med. 2021, 164, 410–416. [Google Scholar] [CrossRef]
- Liu, T.; Han, Y.; Zhou, T.; Zhang, R.; Chen, H.; Chen, S.; Zhao, H. Mechanisms of ros-induced mitochondria-dependent apoptosis underlying liquid storage of goat spermatozoa. Aging 2019, 11, 7880. [Google Scholar] [CrossRef]
- Trzcinska, M.; Bryla, M.; Smorag, Z. Effect of liquid storage on membrane integrity and mitochondrial activity: A new diagnostic method of evaluating boar sperm quality. J. Anim. Feed Sci. 2008, 17, 372–380. [Google Scholar] [CrossRef]
- Swegen, A.; Lambourne, S.R.; Aitken, R.J.; Gibb, Z. Rosiglitazone improves stallion sperm motility, atp content, and mitochondrial function. Biol. Reprod. 2016, 95, 107. [Google Scholar] [CrossRef] [PubMed]
- Agarwal, A.; Majzoub, A.; Esteves, S.C.; Ko, E.; Ramasamy, R.; Zini, A. Clinical utility of sperm DNA fragmentation testing: Practice recommendations based on clinical scenarios. Transl. Androl. Urol. 2016, 5, 935–950. [Google Scholar] [CrossRef] [PubMed]
- de la Torre, J.; Crespo, F.; Arroyo, F.; Zabal-Aguirre, M.; Abdoon, A.S.; Gosalvez, J. Effect of sperm dosage transportation in stallions: Effect on sperm DNA fragmentation. Anim. Reprod. Sci. 2019, 206, 38–45. [Google Scholar] [CrossRef] [PubMed]
- Uppangala, S.; Mathai, S.E.; Salian, S.R.; Kumar, D.; Singh, V.J.; D’Souza, F.; Kalthur, G.; Kamath, A.; Adiga, S.K. Sperm chromatin immaturity observed in short abstinence ejaculates affects DNA integrity and longevity in vitro. PLoS ONE 2016, 11, e0152942. [Google Scholar]
- Sáez-Espinosa, P.; Huerta-Retamal, N.; Robles-Gómez, L.; Avilés, M.; Aizpurua, J.; Velasco, I.; Romero, A.; Gómez-Torres, M.J. Influence of in vitro capacitation time on structural and functional human sperm parameters. Asian J. Androl. 2020, 22, 447–453. [Google Scholar]
- Mirsky, S.K.; Barnea, I.; Levi, M.; Greenspan, H.; Shaked, N.T. Automated analysis of individual sperm cells using stain-free interferometric phase microscopy and machine learning. Cytom. A 2017, 91, 893–900. [Google Scholar] [CrossRef]
- Boniewska-Bernacka, E.; Pańczyszyn, A.; Cybulska, N. Telomeres as a molecular marker of male infertility. Hum. Fertil. 2019, 22, 78–87. [Google Scholar] [CrossRef]
- He, W.; Sun, Υ.; Zhang, S.; Feng, X.; Xu, M.; Dai, J.; Ni, X.; Wang, X.; Wu, Q. Profiling the DNA methylation patterns of imprinted genes in abnormal semen samples by next-generation bisulfite sequencing. J. Assist. Reprod. Genet. 2020, 37, 2211–2221. [Google Scholar] [CrossRef]
- Nematollahi, S.; Mehdizadeh, M.; Hosseini, S.; Kashanian, M.; Amjadi, F.S.; Salehi, M. DNA integrity and methylation changes of mouse spermatozoa following prolonged incubation. Andrologia 2019, 51, e13276. [Google Scholar] [CrossRef]
- Keravnou, A.; Ioannides, M.; Tsangaras, K.; Loizides, C.; Hadjidaniel, M.D.; Papageorgiou, E.A.; Kyriakou, S.; Antoniou, P.; Mina, P.; Achilleos, A.; et al. Whole-genome fetal and maternal DNA methylation analysis using medip-ngs for the identification of differentially methylated regions. Genet. Res. 2016, 98, e15. [Google Scholar] [CrossRef]
- Çok, T.; Çağlar Aytaç, P.; Şimşek, E.; Haydardedeoğlu, B.; Kalaycı, H.; Özdemir, H.; Bulgan Kılıçdağ, E. The effect of preserving prepared sperm samples at room temperature or at 37 °C before intrauterine insemination (iui) on clinical pregnancy rate. Turk. J. Obs. Gynecol. 2015, 12, 6–10. [Google Scholar] [CrossRef]
- Jansen, C.; Elisen, M.; Leenstra, C.W.; Kaaijk, E.M.; van Stralen, K.J.; Verhoeve, H.R. Longer time interval between semen processing and intrauterine insemination does not affect pregnancy outcome. Fertil. Steril. 2017, 108, 764–769. [Google Scholar] [CrossRef][Green Version]
- Kuru Pekcan, M.; Kokanalı, D.; Kokanalı, K.; Taşçı, Y. Effect of time intervals from the end of sperm collection to intrauterine insemination on the pregnancy rates in controlled ovarian hyperstimulation-intrauterine insemination cycles. J. Gynecol Obs. Hum. Reprod 2018, 47, 561–564. [Google Scholar] [CrossRef]
- Yavas, Y.; Selub, M.R. Intrauterine insemination (iui) pregnancy outcome is enhanced by shorter intervals from semen collection to sperm wash, from sperm wash to iui time, and from semen collection to iui time. Fertil. Steril. 2004, 82, 1638–1647. [Google Scholar] [CrossRef]
- Fauque, P.; Lehert, P.; Lamotte, M.; Bettahar-Lebugle, K.; Bailly, A.; Diligent, C.; Clédat, M.; Pierrot, P.; Guénédal, M.L.; Sagot, P. Clinical success of intrauterine insemination cycles is affected by the sperm preparation time. Fertil. Steril. 2014, 101, 1618–1623.e3. [Google Scholar] [CrossRef]
- Punjabi, U.; Van Mulders, H.; Van de Velde, L.; Goovaerts, I.; Peeters, K.; Cassauwers, W.; Lyubetska, T.; Clasen, K.; Janssens, P.; Zemtsova, O.; et al. Time intervals between semen production, initiation of analysis, and iui significantly influence clinical pregnancies and live births. J. Assist. Reprod. Genet. 2021, 38, 421–428. [Google Scholar] [CrossRef]
- Ragni, G.; De Lauretis, L.; D’Ambrogio, G.; Pellegrini, S.; Maggioni, P.; Vegetti, W.; Perotti, L.; Dalla Serra, A.; Parazzini, F.; Crosignani, P.G. Semen preparation by standard swim-up versus swim-up with test yolk buffer incubation in intrauterine insemination: A randomized study. Hum. Reprod. 1998, 13, 1859–1863. [Google Scholar] [CrossRef]
- Barak, Y.; Amit, A.; Lessing, J.B.; Paz, G.; Homonnai, Z.T.; Yogev, L. Improved fertilization rate in an in vitro fertilization program by egg yolk-treated sperm. Fertil. Steril. 1992, 58, 197–198. [Google Scholar] [CrossRef]
- Jensen, J.R.; Walker, J.H.; Milki, A.A.; Westphal, L.; Behr, B. The effect of a two-hour, room temperature incubation of human spermatozoa in test-yolk buffer on the rate of fertilization in vitro. J. Assist. Reprod. Genet. 2004, 21, 169–173. [Google Scholar] [CrossRef]
- Paulson, R.J.; Sauer, M.V.; Francis, M.M.; Macaso, T.M.; Lobo, R.A. A prospective controlled evaluation of test-yolk buffer in the preparation of sperm for human in vitro fertilization in suspected cases of male infertility. Fertil. Steril. 1992, 58, 551–555. [Google Scholar] [CrossRef]
- Rogers, B.J. The use of test-yolk buffer in intrauterine inseminations (iui). In Proceedings of the 38the Annual Meeting of Pacific Coast Fertility Society, Program (Suppl.). Scottsdale, AZ, USA, 25–29 April 1990. abstract no. P-15. [Google Scholar]
- Barisic, D.; Suchanek, E.; Jeyendran, R.S. Milk versus test-yolk—Preincubated sperm—In-vitro fertilization outcome. Eur. J. Obstet. Gynecol. Reprod. Biol. 1995, 62, 231–233. [Google Scholar] [CrossRef]
- Jeyendran, R.S.; Gunawardana, V.K.; Barisic, D.; Wentz, A.C. Test-yolk media and sperm quality. Hum. Reprod. Update 1995, 1, 73–79. [Google Scholar] [CrossRef] [PubMed]
- Zavos, P.M.; Goodpasture, J.C.; Zaneveld, L.J.D. The effect of temperature on sperm motility and viability. Fertil. Steril. 1981, 36, 254–257. [Google Scholar] [CrossRef]
- Mohamed, J.; Ismail, M.N.; Chou, T.Y.; Louis, S.R.; Budin, S.B.J.I.J.o.C.R.o.I.M.; Health, P. A study of sperm quality characteristics changes in different storage temperatures above freezing point. J. Collab. Res. Intern. Med. Public Health 2012, 4, 736–743. [Google Scholar]
- Zhang, X.G.; Yan, G.J.; Hong, J.Y.; Su, Z.Z.; Yang, G.S.; Li, Q.W.; Hu, J.H. Effects of bovine serum albumin on boar sperm quality during liquid storage at 17 degrees C. Reprod. Domest. Anim. 2015, 50, 263–269. [Google Scholar] [CrossRef]
- Feng, T.Y.; Ren, F.; Fang, Q.; Dai, G.C.; Li, Y.; Li, Q.; Xi, H.M.; Li, H.; Hao, Y.Y.; Hu, J.H. Effects of sulfanilamide on boar sperm quality, bacterial composition, and fertility during liquid storage at 17 degrees C. Anim. Sci. J. 2019, 90, 1161–1169. [Google Scholar] [CrossRef]
- Ren, F.; Feng, T.Y.; Niu, T.J.; Yuan, Y.T.; Liu, Q.; Xiao, J.H.; Xu, G.X.; Hu, J.H. Notoginsenoside r1 protects boar sperm during liquid storage at 17 degrees C. Reprod. Domest. Anim. 2020, 55, 1072–1079. [Google Scholar] [CrossRef]
- Acharya, M.; Burke, J.M.; Rorie, R.W. Effect of semen extender and storage temperature on motility of ram spermatozoa. Adv. Reprod. Sci. 2019, 8, 14. [Google Scholar]
- Baek, K.; Feliciano, M.; Wang, A.; Rosenwaks, Z.; Palermo, G.D. P-1006: Can men produce and store semen at home? Fertil. Steril. 2006, 86, S507. [Google Scholar] [CrossRef]
- Lee, J.; Yoo, J.H.; Lee, J.H.; Ahn, H.S.; Hwang, K.J.; Kim, M. Effect of in vitro testicular spermatozoa culture on pregnancy outcomes: An experience at a single university hospital. Yeungnam Univ. J. Med. 2021, 38, 53–59. [Google Scholar] [CrossRef]
- Liu, J.; Tsai, Y.L.; Katz, E.; Compton, G.; Garcia, J.E.; Baramki, T.A. Outcome of in-vitro culture of fresh and frozen-thawed human testicular spermatozoa. Hum. Reprod. 1997, 12, 1667–1672. [Google Scholar] [CrossRef]
- Stuhtmann, G.; Oldenhof, H.; Peters, P.; Klewitz, J.; Martinsson, G.; Sieme, H. Iodixanol density gradient centrifugation for selecting stallion sperm for cold storage and cryopreservation. Anim. Reprod. Sci. 2012, 133, 184–190. [Google Scholar] [CrossRef]
- Heutelbeck, A.; Oldenhof, H.; Rohn, K.; Martinsson, G.; Morrell, J.M.; Sieme, H. Use of density centrifugation for delayed cryopreservation of stallion sperm: Perform sperm selection directly after collection or after storage? Reprod. Domest. Anim. 2015, 50, 76–83. [Google Scholar] [CrossRef]
- Takeshima, T.; Yumura, Y.; Kuroda, S.; Kawahara, T.; Uemura, H.; Iwasaki, A. Effect of density gradient centrifugation on reactive oxygen species in human semen. Syst. Biol. Reprod. Med. 2017, 63, 192–198. [Google Scholar] [CrossRef]
- Makler, A.; Jakobi, P. Factors affecting sperm motility. V. Washing and resuspension of human spermatozoa in various artificial media. Fertil. Steril. 1981, 35, 442–446. [Google Scholar] [CrossRef]
- Rigby, S.L.; Brinsko, S.P.; Cochran, M.; Blanchard, T.L.; Love, C.C.; Varner, D.D. Advances in cooled semen technologies: Seminal plasma and semen extender. Anim. Reprod. Sci. 2001, 68, 171–180. [Google Scholar] [CrossRef]
- Höfner, L.; Luther, A.M.; Waberski, D. The role of seminal plasma in the liquid storage of spermatozoa. Anim. Reprod. Sci. 2020, 220, 106290. [Google Scholar] [CrossRef]
- Akcay, E.; Reilas, T.; Andersson, M.; Katila, T. Effect of seminal plasma fractions on stallion sperm survival after cooled storage. J. Vet. Med. A Physiol. Pathol. Clin. Med. 2006, 53, 481–485. [Google Scholar] [CrossRef] [PubMed]
- Araujo, M.S.; de Oliveira Henriques Paulo, O.L.; Paulini, F.; de Souza Ramos Angrimani, D.; Tsunemi, M.H.; de Paula Freitas Dell’Aqua, C.; Papa, F.O.; de Souza, F.F. Seminal plasma does not influence canine semen stored at 5 degrees c for long-term conservation. Biopreserv. Biobank. 2021, 20, 149–162. [Google Scholar] [CrossRef] [PubMed]
- Brinsko, S.P.; Blanchard, T.L.; Rigby, S.L.; Love, C.C.; Varner, D.D. Effects of dead spermatozoa on motion characteristics and membrane integrity of live spermatozoa in fresh and cooled-stored equine semen. Theriogenology 2003, 59, 735–742. [Google Scholar] [CrossRef]
- Eini, F.; Kutenaei, M.A.; Shirzeyli, M.H.; Dastjerdi, Z.S.; Omidi, M.; Novin, M.G. Normal seminal plasma could preserve human spermatozoa against cryopreservation damages in oligozoospermic patients. BMC Mol. Cell Biol. 2021, 22, 50. [Google Scholar] [CrossRef]
- Fu, L.; Yuen, K.C.J.; Tint, A.N.; Hoffman, A.R.; Bongso, A.T.; Lee, K.O. Association of decreased sperm motility and increased seminal plasma igf-i, igf-ii, igfbp-2, and psa levels in infertile men. Endocrine 2021, 74, 698–706. [Google Scholar] [CrossRef]
- Turunen, T.; Magris, M.; Malinen, M.; Kekäläinen, J. Seminal-plasma-mediated effects on sperm performance in humans. Cells 2022, 11, 2147. [Google Scholar] [CrossRef]
- Seminal plasma as a diagnostic fluid for male reproductive system disorders. Nat. Rev. Urol. 2014, 11, 278–288. [CrossRef]
- Sanada, S.; Arai, E.; Ueda, M.; Ogura, K.; Yoshida, O. Long-term effects of human seminal plasma from whole semen and from different fractions of split ejaculate on motility of human ejaculated spermatozoa. Hinyokika Kiyo. 1988, 34, 1965–1972. [Google Scholar]
- Björndahl, L.; Kvist, U. Sequence of ejaculation affects the spermatozoon as a carrier and its message. Reprod. Biomed. Online 2003, 7, 440–448. [Google Scholar] [CrossRef]
- Amelar, R.D.; Hotchkiss, R.S. The split ejaculate: Its use in the management of male infertility. Fertil. Steril. 1965, 16, 46–60. [Google Scholar] [CrossRef]
- Santolaria, P.; Soler, C.; Recreo, P.; Carretero, T.; Bono, A.; Berné, J.M.; Yániz, J.L. Morphometric and kinematic sperm subpopulations in split ejaculates of normozoospermic men. Asian J. Androl. 2016, 18, 831–834. [Google Scholar] [CrossRef]
- Valsa, J.; Skandhan, K.P.; Khan, P.S.; Sumangala, B.; Gondalia, M. Split ejaculation study: Semen parameters and calcium and magnesium in seminal plasma. Cent. Eur. J. Urol. 2012, 65, 216–218. [Google Scholar] [CrossRef]
- Marmar, J.L.; Praiss, D.E.; Debenedictis, T.J. Statisical comparison of the parameters of semen analysis of whole semen versus the fractions of the split ejaculate. Fertil. Steril. 1978, 30, 439–443. [Google Scholar] [CrossRef]
- Sokol, R.Z.; Madding, C.I.; Handelsman, D.J.; Swerdloff, R.S. The split ejaculate: Assessment of fertility potential using two in vitro test systems. Andrologia 1986, 18, 380–386. [Google Scholar] [CrossRef]
- Propping, D.; Katzorke, T.; Tauber, P.F. Further evaluation of the split ejaculate for artificial insemination. Eur. J. Obs. Gynecol. Reprod. Biol. 1981, 11, 385–394. [Google Scholar] [CrossRef]
- Lindholmer, C. Survival of human spermatozoa in different fractions of split ejaculate. Fertil. Steril. 1973, 24, 521–526. [Google Scholar] [CrossRef]
- Salisbury, G.W.; Fuller, H.K.; Willett, E.L. Preservation of bovine spermatozoa in yolk-citrate diluent and field results from its use. J. Dairy Sci. 1941, 24, 905–910. [Google Scholar] [CrossRef]
- VanDemark, N.L.; Sharma, U.D. Preliminary fertility results from the preservation of bovine semen at room temperatures. J. Dairy Sci. 1957, 40, 438–439. [Google Scholar] [CrossRef]
- Yassen, A.M.; Foote, R.H. Freezability of bovine spermatozoa in tris-buffered yolk extenders containing different levels of tris, sodium, potassium and calcium ions. J. Dairy Sci. 1967, 50, 887–892. [Google Scholar] [CrossRef]
- Graham, E.F.; Crabo, B.G.; Brown, K.I. Effect of some zwitter ion buffers on the freezing and storage of spermatozoa. I. Bull. J. Dairy Sci. 1972, 55, 372–378. [Google Scholar] [CrossRef]
- Veeck, L.L. Tes and tris (test)-yolk buffer systems, sperm function testing, and in vitro fertilization. Fertil. Steril. 1992, 58, 484–486. [Google Scholar] [CrossRef]
- Jacobs, B.R.; Caulfield, J.; Boldt, J. Analysis of test (tes and tris) yolk buffer effects on human sperm. Fertil. Steril. 1995, 63, 1064–1070. [Google Scholar] [CrossRef]
- Jeyendran, R.S.; Graham, E.F. An evaluation of cryoprotective compounds on bovine spermatozoa. Cryobiology 1980, 17, 458–464. [Google Scholar] [CrossRef]
- Tay, J.I.; Rutherford, A.J.; Killick, S.R.; Maguiness, S.D.; Partridge, R.J.; Leese, H.J. Human tubal fluid: Production, nutrient composition and response to adrenergic agents. Hum. Reprod. 1997, 12, 2451–2456. [Google Scholar] [CrossRef]
- Quinn, P. Enhanced results in mouse and human embryo culture using a modified human tubal fluid medium lacking glucose and phosphate. J. Assist. Reprod. Genet. 1995, 12, 97–105. [Google Scholar] [CrossRef] [PubMed]
- Quinn, P.; Kerin, J.F.; Warnes, G.M. Improved pregnancy rate in human in vitro fertilization with the use of a medium based on the composition of human tubal fluid. Fertil. Steril. 1985, 44, 493–498. [Google Scholar] [CrossRef]
- Biggers, J.D.; Whitten, W.K.; Whittingham, D.G.; Daniel, J.D. The culture of mouse embryos in vitro. In Methods of Mammalian Embryology; Daniel, J.C., Ed.; Freeman: San Francisco, CA, USA, 1971; p. 86. [Google Scholar]
- Chi, H.J.; Kim, J.H.; Ryu, C.S.; Lee, J.Y.; Park, J.S.; Chung, D.Y.; Choi, S.Y.; Kim, M.H.; Chun, E.K.; Roh, S.I. Protective effect of antioxidant supplementation in sperm-preparation medium against oxidative stress in human spermatozoa. Hum. Reprod. 2008, 23, 1023–1028. [Google Scholar] [CrossRef]
- Gomez, E.; Aitken, J. Impact of in vitro fertilization culture media on peroxidative damage to human spermatozoa. Fertil. Steril. 1996, 65, 880–882. [Google Scholar] [CrossRef]
- Cheng, C.Y.; Boettcher, B. The effect of steroids on the in vitro migration of washed human spermatozoa in modified tyrode’s solution or in fasting human blood serum. Fertil. Steril. 1979, 32, 566–570. [Google Scholar] [CrossRef]
- Kenney, R.; Bergman, R.; Cooper, W.; Morse, G. Minimal contamination techniques for breeding mares: Technique and preliminary findings. Proc. Am. Ass. Equine Pract. 1975, 323–336. [Google Scholar]
- Padilla, A.W.; Foote, R.H. Extender and centrifugation effects on the motility patterns of slow-cooled stallion spermatozoa. J. Anim. Sci. 1991, 69, 3308–3313. [Google Scholar] [CrossRef]
- Makler, A.; Blumenfeld, Z.; Brandes, J.M.; Paldi, E. Factors affecting sperm motility. Ii. Human sperm velocity and percentage of motility as influenced by semen dilution. Fertil. Steril. 1979, 32, 443–449. [Google Scholar] [CrossRef]
- Gundogan, M.; Yeni, D.; Avdatek, F.; Fidan, A.F. Influence of sperm concentration on the motility, morphology, membrane and DNA integrity along with oxidative stress parameters of ram sperm during liquid storage. Anim. Reprod. Sci. 2010, 122, 200–207. [Google Scholar] [CrossRef]
- Alvarez, M.; Tamayo-Canul, J.; Anel, E.; Boixo, J.C.; Mata-Campuzano, M.; Martinez-Pastor, F.; Anel, L.; de Paz, P. Sperm concentration at freezing affects post-thaw quality and fertility of ram semen. Theriogenology 2012, 77, 1111–1118. [Google Scholar] [CrossRef]
- Murphy, C.; Fahey, A.G.; Shafat, A.; Fair, S. Reducing sperm concentration is critical to limiting the oxidative stress challenge in liquid bull semen. J. Dairy Sci. 2013, 96, 4447–4454. [Google Scholar] [CrossRef]
- Murphy, C.; Holden, S.A.; Murphy, E.M.; Cromie, A.R.; Lonergan, P.; Fair, S. The impact of storage temperature and sperm number on the fertility of liquid-stored bull semen. Reprod. Fertil. Dev. 2016, 28, 1349–1359. [Google Scholar] [CrossRef]
- Sadeghi, S.; Del Gallego, R.; García-Colomer, B.; Gómez, E.A.; Yániz, J.L.; Gosálvez, J.; López-Fernández, C.; Silvestre, M.A. Effect of sperm concentration and storage temperature on goat spermatozoa during liquid storage. Biology 2020, 9, 300. [Google Scholar] [CrossRef]
- Maxwell, W.M.; Johnson, L.A. Physiology of spermatozoa at high dilution rates: The influence of seminal plasma. Theriogenology 1999, 52, 1353–1362. [Google Scholar] [CrossRef]
- Hernández-Avilés, C.; Love, C.C.; Serafini, R.; Ramírez-Agámez, L.; Friedrich, M.; Ghosh, S.; Teague, S.R.; LaCaze, K.A.; Brinsko, S.P.; Varner, D.D. Effects of glucose concentration in semen extender and storage temperature on stallion sperm quality following long-term cooled storage. Theriogenology 2020, 147, 1–9. [Google Scholar] [CrossRef]
- Rungruangsak, J.; Suwimonteerabutr, J.; Buranaamnuay, K.; Asawakarn, S.; Chantavisoote, N.; Pisitkun, T.; Chaweewan, K.; Tummaruk, P. Difference of seminal plasma and sperm proteins in good and poor freezability boar ejaculates. Vet. Stanica 2021, 53, 113–126. [Google Scholar] [CrossRef]
- Barrios, B.; Pérez-Pé, R.; Gallego, M.; Tato, A.; Osada, J.; Muiño-Blanco, T.; Cebrián-Pérez, J.A. Seminal plasma proteins revert the cold-shock damage on ram sperm membrane. Biol. Reprod. 2000, 63, 1531–1537. [Google Scholar] [CrossRef] [PubMed]
- Daskalova, D.; Kukov, A.; Kirilova, I.; Ivanova-Kicheva, M. Protein analysis of boar seminal plasma proteins with protective effect during low-temperature storage of spermatozoa. Biotechnol. Biotechnol. Equip. 2014, 28, 716–720. [Google Scholar] [CrossRef] [PubMed]
- Foote, R.H.; Bratton, R.W. The fertility of bovine semen in extenders containing sulfanilamide, penicillin, streptomycin and polymyxin. J. Dairy Sci. 1950, 33, 544–547. [Google Scholar] [CrossRef]
- Tvrda, E.; Lovisek, D.; Kyzek, S.; Kovacik, D.; Galova, E. The effect of non-thermal plasma on the structural and functional characteristics of human spermatozoa. Int. J. Mol. Sci. 2021, 22, 4979. [Google Scholar] [CrossRef]
- Jasko, D.J.; Hathaway, J.A.; Schaltenbrand, V.L.; Simper, W.D.; Squires, E.L. Effect of seminal plasma and egg-yolk on motion characteristics of cooled stallion spermatozoa. Theriogenology 1992, 37, 1241–1252. [Google Scholar] [CrossRef]
- Pace, M.M.; Graham, E.F. Components in egg yolk which protect bovine spermatozoa during freezing. J. Anim. Sci. 1974, 39, 1144–1149. [Google Scholar] [CrossRef]
- Amirat, L.; Tainturier, D.; Jeanneau, L.; Thorin, C.; Gérard, O.; Courtens, J.L.; Anton, M. Bull semen in vitro fertility after cryopreservation using egg yolk ldl: A comparison with optidyl, a commercial egg yolk extender. Theriogenology 2004, 61, 895–907. [Google Scholar] [CrossRef]
- Moussa, M.; Marinet, V.; Trimeche, A.; Tainturier, D.; Anton, M. Low density lipoproteins extracted from hen egg yolk by an easy method: Cryoprotective effect on frozen-thawed bull semen. Theriogenology 2002, 57, 1695–1706. [Google Scholar] [CrossRef]
- Bencharif, D.; Amirat, L.; Anton, M.; Schmitt, E.; Desherces, S.; Delhomme, G.; Langlois, M.L.; Barrière, P.; Larrat, M.; Tainturier, D. The advantages of ldl (low density lipoproteins) in the cryopreservation of canine semen. Theriogenology 2008, 70, 1478–1488. [Google Scholar] [CrossRef]
- Emamverdi, M.; Zhandi, M.; Zare Shahneh, A.; Sharafi, M.; Akbari-Sharif, A. Optimization of ram semen cryopreservation using a chemically defined soybean lecithin-based extender. Reprod. Domest. Anim. 2013, 48, 899–904. [Google Scholar] [CrossRef]
- Pillet, E.; Duchamp, G.; Batellier, F.; Beaumal, V.; Anton, M.; Desherces, S.; Schmitt, E.; Magistrini, M. Egg yolk plasma can replace egg yolk in stallion freezing extenders. Theriogenology 2011, 75, 105–114. [Google Scholar] [CrossRef]
- Ashrafi, I.; Kia, H.D.; Parrish, J. Egg yolk saline soluble fraction was as efficient as ammonium sulfate insoluble yolk fraction in cryopreservation of bull semen in comparison with whole egg yolk. Rev. Med. Vet. 2019, 170, 104–109. [Google Scholar]
- Konyali, C.; Tomas, C.; Blanch, E.; Gomez, E.A.; Graham, J.K.; Moce, E. Optimizing conditions for treating goat semen with cholesterol-loaded cyclodextrins prior to freezing to improve cryosurvival. Cryobiology 2013, 67, 124–131. [Google Scholar] [CrossRef]
- Moce, E.; Blanch, E.; Tomas, C.; Graham, J.K. Use of cholesterol in sperm cryopreservation: Present moment and perspectives to future. Reprod. Domest. Anim. 2010, 45 (Suppl. 2), 57–66. [Google Scholar] [CrossRef]
- Layek, S.S.; Mohanty, T.K.; Kumaresan, A.; Parks, J.E. Cryopreservation of bull semen: Evolution from egg yolk based to soybean based extenders. Anim. Reprod. Sci. 2016, 172, 1–9. [Google Scholar] [CrossRef]
- Salmani, H.; Towhidi, A.; Zhandi, M.; Bahreini, M.; Sharafi, M. In vitro assessment of soybean lecithin and egg yolk based diluents for cryopreservation of goat semen. Cryobiology 2014, 68, 276–280. [Google Scholar] [CrossRef]
- Singh, A.K.; Kumar, A.; Honparkhe, M.; Kaur, S.; Kaur, H.; Ghuman, S.; Brar, P.S. Comparison of in vitro and in vivo fertilizing potential of buffalo bull semen frozen in egg yolk-, soya bean lecithin- and liposome-based extenders. Reprod. Domest. Anim. 2018, 53, 195–202. [Google Scholar] [CrossRef]
- Sun, L.; He, M.; Wu, C.; Zhang, S.; Dai, J.; Zhang, D. Beneficial influence of soybean lecithin nanoparticles on rooster frozen-thawed semen quality and fertility. Animals 2021, 11, 1769. [Google Scholar] [CrossRef]
- Khalifa, T.; Lymberopoulos, A. Changeability of sperm chromatin structure during liquid storage of ovine semen in milk-egg yolk- and soybean lecithin-based extenders and their relationships to field-fertility. Cell Tissue Bank. 2013, 14, 687–698. [Google Scholar] [CrossRef]
- Sun, L.; Fan, W.; Wu, C.; Zhang, S.; Dai, J.; Zhang, D. Effect of substituting different concentrations of soybean lecithin and egg yolk in tris-based extender on goat semen cryopreservation. Cryobiology 2020, 92, 146–150. [Google Scholar] [CrossRef]
- Jeyendran, R.S.; Acosta, V.C.; Land, S.; Coulam, C.B. Cryopreservation of human sperm in a lecithin-supplemented freezing medium. Fertil. Steril. 2008, 90, 1263–1265. [Google Scholar] [CrossRef]
- Reed, M.L.; Ezeh, P.C.; Hamic, A.; Thompson, D.J.; Caperton, C.L. Soy lecithin replaces egg yolk for cryopreservation of human sperm without adversely affecting postthaw motility, morphology, sperm DNA integrity, or sperm binding to hyaluronate. Fertil. Steril. 2009, 92, 1787–1790. [Google Scholar] [CrossRef]
- Sicchieri, F.; Silva, A.B.; Santana, V.P.; Vasconcelos, M.A.C.; Ferriani, R.A.; Vireque, A.A.; Dos Reis, R.M. Phosphatidylcholine and l-acetyl-carnitine-based freezing medium can replace egg yolk and preserves human sperm function. Transl. Androl. Urol. 2021, 10, 397–407. [Google Scholar] [CrossRef]
- Mutalik, S.; Salian, S.R.; Avadhani, K.; Menon, J.; Joshi, H.; Hegde, A.R.; Kumar, P.; Kalthur, G.; Adiga, S.K. Liposome encapsulated soy lecithin and cholesterol can efficiently replace chicken egg yolk in human semen cryopreservation medium. Syst. Biol. Reprod. Med. 2014, 60, 183–188. [Google Scholar] [CrossRef]
- Sarıözkan, S.; Ozdamar, S.; Türk, G.; Cantürk, F.; Yay, A. In vitro effects of l-carnitine and glutamine on motility, acrosomal abnormality, and plasma membrane integrity of rabbit sperm during liquid-storage. Cryobiology 2014, 68, 349–353. [Google Scholar] [CrossRef]
- Gibb, Z.; Lambourne, S.R.; Quadrelli, J.; Smith, N.D.; Aitken, R.J. L-carnitine and pyruvate are prosurvival factors during the storage of stallion spermatozoa at room temperature. Biol. Reprod. 2015, 93, 104. [Google Scholar] [CrossRef]
- Lafuente, R.; González-Comadrán, M.; Solà, I.; López, G.; Brassesco, M.; Carreras, R.; Checa, M.A. Coenzyme q10 and male infertility: A meta-analysis. J. Assist. Reprod. Genet. 2013, 30, 1147–1156. [Google Scholar] [CrossRef]
- Lewin, A.; Lavon, H. The effect of coenzyme q10 on sperm motility and function. Mol. Asp. Med. 1997, 18, S213–S219. [Google Scholar] [CrossRef]
- Liu, L.; Wang, M.J.; Yu, T.H.; Cheng, Z.; Li, M.; Guo, Q.W. mitochondria-targeted antioxidant mitoquinone protects post-thaw human sperm against oxidative stress injury. Zhonghua Nan Ke Xue 2016, 22, 205–211. [Google Scholar]
- Minucci, S.; Venditti, M. New insight on the in vitro effects of melatonin in preserving human sperm quality. Int. J. Mol. Sci. 2022, 23, 5128. [Google Scholar] [CrossRef]
- Mousavi, S.M.; Towhidi, A.; Zhandi, M.; Amoabediny, G.; Mohammadi-Sangcheshmeh, A.; Sharafi, M.; Hussaini, S.M.H. Comparison of two different antioxidants in a nano lecithin-based t extender for bull sperm cryopreservation. Anim. Reprod. Sci. 2019, 209, 106171. [Google Scholar] [CrossRef] [PubMed]
- Sánchez-Rubio, F.; Soria-Meneses, P.J.; Jurado-Campos, A.; Bartolomé-García, J.; Gómez-Rubio, V.; Soler, A.J.; Arroyo-Jimenez, M.M.; Santander-Ortega, M.J.; Plaza-Oliver, M.; Lozano, M.V.; et al. Nanotechnology in reproduction: Vitamin e nanoemulsions for reducing t oxidative stress in sperm cells. Free Radic. Biol. Med. 2020, 160, 47–56. [Google Scholar] [CrossRef] [PubMed]
- Moradi, M.; Moradi, B.; Hashemian, A.H.; Bakhtiari, M.; Khazaei, M.; Esmaeili, F.; Aghaz, F.; Faramarzi, A. Beneficial effect of l-proline supplementation on the quality of human spermatozoa. Andrologia 2022, 54, e14486. [Google Scholar] [CrossRef]
- Boatman, D.E.; Schaffer, S.; Takahashi, K.; Azuma, J.; Yeung, C.H.; Anapolski, M.; Depenbusch, M.; Zitzmann, M.; Cooper, T.G.; Petrunkina, A.M.; et al. Human sperm volume regulation. Response to physiological changes in osmolality, channel blockers and potential sperm osmolytes. Hum. Reprod. 1997, 12, 133–149. [Google Scholar] [PubMed]
- Barfield, J.P.; Yeung, C.H.; Cooper, T.G. Characterization of potassium channels involved in volume regulation of human spermatozoa. Mol. Hum. Reprod. 2005, 11, 891–897. [Google Scholar] [CrossRef] [PubMed]
- Yeung, C.H.; Barfield, J.P.; Cooper, T.G. Chloride channels in physiological volume regulation of human spermatozoa. Biol. Reprod. 2005, 73, 1057–1063. [Google Scholar] [CrossRef] [PubMed]
- Makler, A.; Makler, E.; Itzkovitz, J.; Brandes, J.M. Factors affecting sperm motility. Iv. Incubation of human semen with caffeine, kallikrein, and other metabolically active compounds. Fertil. Steril. 1980, 33, 624–630. [Google Scholar] [CrossRef]
- Slanina, T.; Miškeje, M.; Tirpák, F.; Błaszczyk, M.; Formicki, G.; Massányi, P. Caffeine strongly improves motility parameters of turkey spermatozoa with no effect on cell viability. Acta Vet. Hung. 2018, 66, 137–150. [Google Scholar] [CrossRef]
- Mahaldashtian, M.; Khalili, M.A.; Nottola, S.A.; Woodward, B.; Macchiarelli, G.; Miglietta, S. Does in vitro application of pentoxifylline have beneficial effects in assisted male reproduction? Andrologia 2021, 53, e13722. [Google Scholar] [CrossRef]
- Brennan, A.P.; Holden, C.A. Pentoxifylline-supplemented cryoprotectant improves human sperm motility after cryopreservation. Hum. Reprod. 1995, 10, 2308–2312. [Google Scholar] [CrossRef]
- Xian, Y.; Jiang, M.; Liu, B.; Zhao, W.; Zhou, B.; Liu, X.; Liu, S.; Li, F. A cryoprotectant supplemented with pentoxifylline can improve the effect of freezing on the motility of human testicular sperm. Zygote 2021, 30, 92–97. [Google Scholar] [CrossRef] [PubMed]
- Pariz, J.R.; Ranéa, C.; Monteiro, R.A.C.; Evenson, D.P.; Drevet, J.R.; Hallak, J. Melatonin and caffeine supplementation used, respectively, as protective and stimulating agents in the cryopreservation of human sperm improves survival, viability, and motility after thawing compared to traditional test-yolk buffer. Oxidative Med. Cell. Longev. 2019, 2019, 6472945. [Google Scholar] [CrossRef] [PubMed]
- Abd El-Hamid, I.S. Effect of adding different levels of caffeine in the extender on some biochemical constituents, enzymatic activities and physical characteristics of chilled and frozen ram semen. Reprod. Domest. Anim. 2019, 54, 225–233. [Google Scholar] [CrossRef] [PubMed]
- Sharma, R.K.; Tolentino, M.V., Jr.; Thomas, A.J., Jr.; Agarwal, A. Optimal dose and duration of exposure to artificial stimulants in cryopreserved human spermatozoa. J. Urol. 1996, 155, 568–573. [Google Scholar] [CrossRef]
- Pariz, J.R.; Hallak, J. Effects of caffeine supplementation in post-thaw human semen over different incubation periods. Andrologia 2016, 48, 961–966. [Google Scholar] [CrossRef]
- Bourdon, G.; Estienne, A.; Chevaleyre, C.; Ramé, C.; Guérif, F.; Brun, J.S.; Vasseur, C.; Fromont, G.; Plotton, I.; Dufour-Rainfray, D.; et al. The hepatokine fgf21 increases the human spermatozoa motility. Front. Endocrinol. 2022, 13, 775650. [Google Scholar] [CrossRef]
- Pereira, R.R.; Nogueira, B.G.; Milan, B.; Acácio, B.R.; Freitas- Dell’Aqua, C.P.; Souza, M.I.L.; Sampaio, B.F.B. Use low ozone dosages to have a positive effect in the cooling and cryopreservation of equine semen. J. Equine Vet. Sci. 2021, 108, 103800. [Google Scholar] [CrossRef]
- Safian, F.; Ghaffari Novin, M.; Nazarian, H.; Shams Mofarahe, Z.; Abdollahifar, M.A.; Jajarmi, V.; Karimi, S.; Kazemi, M.; Chien, S.; Bayat, M. Photobiomodulation preconditioned human semen protects sperm cells against detrimental effects of cryopreservation. Cryobiology 2021, 98, 239–244. [Google Scholar] [CrossRef]
- Hezavehei, M.; Mirzaei, M.; Sharafi, M.; Wu, Y.; Gupta, V.; Fitzhenry, M.; Kouchesfahani, H.M.; Eftekhari-Yazdi, P.; Baharvand, H.; Dalman, A.; et al. Proteomics study reveals the molecular mechanisms underlying cryotolerance induced by mild sublethal stress in human sperm. Cell Tissue Res. 2021, 387, 143–157. [Google Scholar] [CrossRef]
- Saito, K.; Kinoshita, Y.; Kanno, H.; Iwasaki, A.; Hosaka, M. A new method of the electrolyte-free long-term preservation of human sperm at 4 °C. Fertil. Steril. 1996, 65, 1210–1213. [Google Scholar] [CrossRef]
- Quan, S.; Zhou, H.K.; Shuji, Y.; Hisayo, N.; Toshihiro, A. Fertilizing capacity of human sperm preserved in cold electrolyte-free solution. Di Yi Jun Yi Da Xue Xue Bao 2002, 22, 928–930. [Google Scholar]
- Rastogi, R.; Su, J.; Mahalingam, A.; Clark, J.; Sung, S.; Hope, T.; Kiser, P.F. Engineering and characterization of simplified vaginal and seminal fluid simulants. Contraception 2016, 93, 337–346. [Google Scholar] [CrossRef][Green Version]
- Agha-Rahimi, A.; Khalili, M.A.; Nottola, S.A.; Miglietta, S.; Moradi, A. Cryoprotectant-free vitrification of human spermatozoa in new artificial seminal fluid. Andrology 2016, 4, 1037–1044. [Google Scholar] [CrossRef]
- Khan, I.M.; Cao, Z.; Liu, H.; Khan, A.; Rahman, S.U.; Khan, M.Z.; Sathanawongs, A.; Zhang, Y. Impact of cryopreservation on spermatozoa freeze-thawed traits and relevance omics to assess sperm cryo-tolerance in farm animals. Front. Vet. Sci. 2021, 8, 609180. [Google Scholar] [CrossRef]
- Saadeldin, I.M.; Khalil, W.A.; Alharbi, M.G.; Lee, S.H. The current trends in using nanoparticles, liposomes, and exosomes for semen cryopreservation. Animals 2020, 10, 2281. [Google Scholar] [CrossRef]
- Comizzoli, P.; Loi, P.; Patrizio, P.; Hubel, A. Long-term storage of gametes and gonadal tissues at room temperatures: The end of the ice age? J. Assist. Reprod. Genet. 2022, 39, 321–325. [Google Scholar] [CrossRef]
- Shahin, M.A.; Khalil, W.A.; Saadeldin, I.M.; Swelum, A.A.; El-Harairy, M.A. Comparison between the effects of adding vitamins, trace elements, and nanoparticles to shotor extender on the cryopreservation of dromedary camel epididymal spermatozoa. Animals 2020, 10, 78. [Google Scholar] [CrossRef]
- Préaubert, L.; Tassistro, V.; Auffan, M.; Sari-Minodier, I.; Rose, J.; Courbiere, B.; Perrin, J. Very low concentration of cerium dioxide nanoparticles induce DNA damage, but no loss of vitality, in human spermatozoa. Toxicol. Vitr. 2018, 50, 236–241. [Google Scholar] [CrossRef]
- Santonastaso, M.; Mottola, F.; Colacurci, N.; Iovine, C.; Pacifico, S.; Cammarota, M.; Cesaroni, F.; Rocco, L. In vitro genotoxic effects of titanium dioxide nanoparticles (n-tio(2) ) in human sperm cells. Mol. Reprod. Dev. 2019, 86, 1369–1377. [Google Scholar] [CrossRef]
- Moretti, E.; Terzuoli, G.; Renieri, T.; Iacoponi, F.; Castellini, C.; Giordano, C.; Collodel, G. In vitro effect of gold and silver nanoparticles on human spermatozoa. Andrologia 2013, 45, 392–396. [Google Scholar]
- Jurado-Campos, A.; Soria-Meneses, P.J.; Sánchez-Rubio, F.; Niza, E.; Bravo, I.; Alonso-Moreno, C.; Arenas-Moreira, M.; García-Álvarez, O.; Soler, A.J.; Garde, J.J.; et al. Vitamin e delivery systems increase resistance to oxidative stress in red deer sperm cells: Hydrogel and nanoemulsion carriers. Antioxidants 2021, 10, 1780. [Google Scholar] [CrossRef]
- Abdelnour, S.A.; Hassan, M.A.E.; Mohammed, A.K.; Alhimaidi, A.R.; Al-Gabri, N.; Al-Khaldi, K.O.; Swelum, A.A. The effect of adding different levels of curcumin and its nanoparticles to extender on post-thaw quality of cryopreserved rabbit sperm. Animals 2020, 10, 1508. [Google Scholar] [CrossRef]
- Ismail, A.A.; Abdel-Khalek, A.E.; Khalil, W.A.; Yousif, A.I.; Saadeldin, I.M.; Abomughaid, M.M.; El-Harairy, M.A. Effects of mint, thyme, and curcumin extract nanoformulations on the sperm quality, apoptosis, chromatin decondensation, enzyme activity, and oxidative status of cryopreserved goat semen. Cryobiology 2020, 97, 144–152. [Google Scholar] [CrossRef]
- Safa, S.; Moghaddam, G.; Jozani, R.J.; Daghigh Kia, H.; Janmohammadi, H. Effect of vitamin e and selenium nanoparticles on post-thaw variables and oxidative status of rooster semen. Anim. Reprod. Sci. 2016, 174, 100–106. [Google Scholar] [CrossRef]
- Mortazavi, S.H.; Eslami, M.; Farrokhi-Ardabili, F. Comparison of different carrier-compounds and varying concentrations of oleic acid on freezing tolerance of ram spermatozoa in tris-citric acid-egg yolk plasma semen diluent. Anim. Reprod. Sci. 2020, 219, 106533. [Google Scholar] [CrossRef]
- Leao, A.P.A.; Souza, A.V.; Mesquita, N.F.; Pereira, L.J.; Zangeronimo, M.G. Antioxidant enrichment of rooster semen extenders—A systematic review. Res. Vet. Sci. 2021, 136, 111–118. [Google Scholar] [CrossRef]
- Ferreira, G.; Costa, C.; Bassaizteguy, V.; Santos, M.; Cardozo, R.; Montes, J.; Settineri, R.; Nicolson, G.L. Incubation of human sperm with micelles made from glycerophospholipid mixtures increases sperm motility and resistance to oxidative stress. PLoS ONE 2018, 13, e0197897. [Google Scholar] [CrossRef]
- Roca, J.; Rodriguez-Martinez, H.; Padilla, L.; Lucas, X.; Barranco, I. Extracellular vesicles in seminal fluid and effects on male reproduction. An overview in farm animals and pets. Anim. Reprod. Sci. 2021, 106853. [Google Scholar] [CrossRef]
- Du, J.; Shen, J.; Wang, Y.; Pan, C.; Pang, W.; Diao, H.; Dong, W. Boar seminal plasma exosomes maintain sperm function by infiltrating into the sperm membrane. Oncotarget 2016, 7, 58832–58847. [Google Scholar] [CrossRef]
- Mahiddine, F.Y.; Kim, J.W.; Qamar, A.Y.; Ra, J.C.; Kim, S.H.; Jung, E.J.; Kim, M.J. Conditioned medium from canine amniotic membrane-derived mesenchymal stem cells improved dog sperm post-thaw quality-related parameters. Animals 2020, 10, 1899. [Google Scholar] [CrossRef]
- Gosálvez, J.; López-Fernández, C.; Fernández, J.L.; Johnston, S. Microencapsulation of human spermatozoa increases membrane stability and DNA longevity. Andrologia 2021, 53, e13924. [Google Scholar] [CrossRef] [PubMed]
- Rickard, J.P.; Schmidt, R.E.; Maddison, J.W.; Bathgate, R.; Lynch, G.W.; Druart, X.; de Graaf, S.P. Variation in seminal plasma alters the ability of ram spermatozoa to survive cryopreservation. Reprod. Fertil. Dev. 2016, 28, 516–523. [Google Scholar] [CrossRef] [PubMed]
- Salinas, M.B.; Chuammitri, P.; Sringarm, K.; Boonyayatra, S.; Sathanawongs, A. Current perspectives on ruminant sperm freezability: Harnessing molecular changes related to semen quality through omics technologies. Vet. Integr. Sci. 2021, 19, 487–511. [Google Scholar] [CrossRef]
- Chen, X.; Wang, Y.; Zhu, H.; Hao, H.; Zhao, X.; Qin, T.; Wang, D. Comparative transcript profiling of gene expression of fresh and frozen-thawed bull sperm. Theriogenology 2015, 83, 504–511. [Google Scholar] [CrossRef]
- Dietrich, M.A.; Irnazarow, I.; Ciereszko, A. Proteomic identification of seminal plasma proteins related to the freezability of carp semen. J. Proteom. 2017, 162, 52–61. [Google Scholar] [CrossRef]
- Rickard, J.P.; Leahy, T.; Soleilhavoup, C.; Tsikis, G.; Labas, V.; Harichaux, G.; Lynch, G.W.; Druart, X.; de Graaf, S.P. The identification of proteomic markers of sperm freezing resilience in ram seminal plasma. J. Proteom. 2015, 126, 303–311. [Google Scholar] [CrossRef]
- Wang, J.; Wang, J.; Wang, M.; Hong, R.; Tang, S.; Xu, Y.; Zhao, X.; Zhou, T.; Wang, Z.; Huang, S. Quantitative phosphoproteomics reveals gsk3a substrate network is involved in the cryodamage of sperm motility. Biosci. Rep. 2021, 41, BSR20211326. [Google Scholar] [CrossRef]
- Soleilhavoup, C.; Tsikis, G.; Labas, V.; Harichaux, G.; Kohnke, P.L.; Dacheux, J.L.; Guerin, Y.; Gatti, J.L.; de Graaf, S.P.; Druart, X. Ram seminal plasma proteome and its impact on liquid preservation of spermatozoa. J. Proteom. 2014, 109, 245–260. [Google Scholar] [CrossRef]
- Lombo, M.; Ruiz-Diaz, S.; Gutierrez-Adan, A.; Sanchez-Calabuig, M.J. Sperm metabolomics through nuclear magnetic resonance spectroscopy. Animals 2021, 11, 1669. [Google Scholar] [CrossRef]
- Zhang, Y.; Liang, H.; Liu, Y.; Zhao, M.; Xu, Q.; Liu, Z.; Weng, X. Metabolomic analysis and identification of sperm freezability-related metabolites in boar seminal plasma. Animals 2021, 11, 1939. [Google Scholar] [CrossRef]
- Mateo-Otero, Y.; Fernandez-Lopez, P.; Ribas-Maynou, J.; Roca, J.; Miro, J.; Yeste, M.; Barranco, I. Metabolite profiling of pig seminal plasma identifies potential biomarkers for sperm resilience to liquid preservation. Front. Cell Dev. Biol. 2021, 9, 669974. [Google Scholar] [CrossRef]
| Parameters/Assays | Extender/Pre-treatment | Storage Temperature | Storage Duration | Main Results | Reference |
|---|---|---|---|---|---|
| Viability, | SP | 4, 20 or 37 °C | 18 h | Semen should be kept at 20 °C if any delay happens | [105] |
| Motility | 4, 25 or 37 °C | 1 h | Semen should be stored at 37 °C after collection | [88] | |
| and motion parameters | TEST-yolk buffer | 5 °C | 96 h | 50% motility recovery after 96 h | [21] |
| 2–5 °C | 48 h | 70% motility recovery after 96 h | [110] | ||
| 4 °C | 72 h | 52% viability and 14% PR recovery after 72 h | [106] | ||
| 24, 37 or 40 °C | 24 h | Motility recovery was best preserved at 24 °C | [45] | ||
| Human follicular fluid, PBS | 37 °C | 12 h | 30% and nearly 0% motility and PR recovery after 12 h in human follicular fluid and in PBS | [111] | |
| Modified Tyrode’s solution | 3 or 37 °C | 24 h | 50% and 1% motility recovery after 24 h at 3 °C and 37 °C | [112] | |
| 4, 22 or 37 °C | 5 days | Viability and Motility recovery (85% and 50%) was best preserved at 22 °C after 5 days | [113] | ||
| 4, 22 or 37 °C | 5 days | Viability and Motility recovery (80% and 50%) was best preserved at 22 °C after 5 days | [114] | ||
| Citrate TEST-yolk buffer | RT | 25–30 h | Motility recovery are nearly 100% after 24 h at RT, VCL, VAP and PR are sustained | [22] | |
| EFM | 4 °C | Record till 0 | Viability and motility maintained for up to 42 and 28 days | [11] | |
| Modified Human tubal fluid | 4, 23 or 37 °C | Record till 0 | >50% Viability and Motility recovery was best preserved at 22 °C after 21 and 10 days | [115] | |
| 4 °C | Record till 0 | Viability and motility maintained <2 weeks and <4 weeks | [11] | ||
| 24 °C | 7 days | Viability and Motility recovery (53% and 10%) after 5 days | [37] | ||
| 24, 37 or 40 °C | 24 h | Motility recovery was best preserved at 24 °C | [45] | ||
| Modified Ham’s F-10 media | 37 °C | 24 h | Viability and Motility recovery (62% and 1%) after 24 h | [116] | |
| RT, 37 °C | 72 h | Testicular sperm motility was optimized at RT after 24 h of incubation | [14] | ||
| 4–6 or 25 °C | 12 days | Motility recovery (8% at 4–6 °C and 2% at 25 °C) after 7 days | [108] | ||
| RT, 37 °C | 4 h | Viability and Motility recovery (73% and 67%) of asthenoteratozoospermia and no difference between RT and 37 °C | [117] | ||
| PBS/Human follicular fluids | 37 °C | 12 h | Sperm motility and VCL were significantly stimulated | [111] | |
| SP, G-IVFTM | RT, 37 °C | 24 h | 25% motility recovery at RT with no treatment. Motility was totally lost at 37 °C in SP or G- IVFTM | [118] | |
| SP, Ham’s F10 media, ASF | RT | 24 h | Viability and Motility in Ham’s F10 and ASF are comparable and better than no SP | [119] | |
| PBS plus glucose | 4, 22 and 37 °C | 10 days | Motility recovery was 20% at 22 °C | [120] | |
| BWW medium | 37 °C | 3 days | Motility and PR recovery was 12% and 0% at 37 °C, significant increase in VSL, VCL, and VAP after penicillamine supplementation | [109] | |
| Earle’s balanced salt solution | RT, 35 °C | 24 h | Viability and Motility recovery (95% and 90%) was best at RT | [121] | |
| BM1 | RT, 35 °C | 24 h | Motility recovery was 76% at RT | [122] | |
| G-IVFTM | 4–6 or 25 °C | 12 days | Motility recovery was 8% at 4–6 °C and 1.5% and 25 °C after 7 days | [108] | |
| Morphology | Ringer Glucose Phosphate buffer | 21 or 37 °C | 22 h | Prolonged (>2 h) sperm manipulations for ART should be performed at 21 °C rather than 37 °C | [40] |
| Modified Human tubal fluid | 24 °C | 7 days | Deterioration in morphology is slower during 7-day incubation | [37] | |
| BM1 | RT, 35 °C | 24 h | Morphologically normal spermatozoa were significantly higher in RT compared with 35 °C | [122] | |
| Not clear | 37 °C | 5 h | Morphologically normal spermatozoa increased after incubation | [103] | |
| Hamster egg | TEST-yolk buffer | 2–5 °C | 48 h | Increased significantly following storage | [110] |
| fusion | Citrate TEST-yolk buffer | RT | 25–30 h | Remained unchanged | [22] |
| Acrosome | |||||
| Function | |||||
| FITC-PNA stained | Citrate TEST-yolk buffer | RT | 25–30 h | Remained unchanged | [22] |
| Ham’s F10 medium | 37 °C | 22 h | No effect | [5] | |
| Oxdative Stress | |||||
| ROS level | Ham’s F10 medium | 37 °C | 22 h | Significant increase after 22 h | [5] |
| SP | 4, 25 or 37 °C | 1 h | Semen should be stored at 37 °C after collection | [88] | |
| Membrane damage | |||||
| MMP(JC-1) | SP | Not clear | 4 h | Worse MMP is associated with a significant decline in motility over time | [123] |
| HOS | Modified human tubal fluid | 24 °C | 7 days | Membrane integrity dropped down to 80% | [37] |
| EFM | 4 °C | 4 weeks | Membrane integrity were significantly better than TEST-yolk buffer | [124] | |
| SYBR14/PI and JC-1 | PBS plus glucose | 4, 22 and 37 °C | 10 days | Viable sperm with functional mitochondria are >40% after 10 days | [120] |
| SYBR14/PI | EFM | 4 °C | Record till 0 | Sperm with intact cell membrane maintained for up to 7 weeks | [34] |
| Annexin V/PI | G-IVFTM | 4–6 or 25 °C | 12 days | The mean percentage of late apoptotic sperm cells increased significantly from day 0 to day 12 | [107] |
| Human tubal fluid | 37 °C | 24 h | The mean percentage of apoptotic sperm cells increase nearly 50% after 24 h | [125] | |
| LPO | |||||
| TBA assay | Citrate TEST-yolk buffer | RT | 25–30 h | Preserve motility but not suppress lipid peroxidation | [22] |
| Ham’s F10 medium | 37 °C | 22 h | Significant increase after 22 h | [5] | |
| DNA damage | |||||
| EFM | 4 °C | 2 weeks | SDF was slightly higher after than before storage | [10] | |
| SCSA | SP, G-IVFTM | RT, 37 °C | 24 h | SDF increased, but at RT was significantly lower than 37 °C | [118] |
| SCD | SP, human tubal fluid | On ice, RT | 24 h | RT for 24 h significantly increased SDF | [126] |
| SP, PBS | 37 °C | 24 h | SP increase SDF after 2 h so that it must be removed | [127] | |
| Ham’s F10 media | 37 °C | 3 h | SDF was significantly higher after 2 h | [13] | |
| 37 °C | 22 h | SDF was significant increased after 22 h | [5] | ||
| Human follicular fluid | 37 °C | 24 h | Co-incubation with HFF could reduce SDF | [128] | |
| SP, Ham’s F10 media, ASF | RT | 24 h | SDF preserved in ASF are similar with normal SP | [119] | |
| TUNEL | BM1 | RT, 35 °C | 24 h | Increase of SDF was significantly lower at RT than at 35 °C | [122] |
| Modified Tyrode’s medium | 37 °C | 3 days | Results in significant DNA damage | [114] | |
| Comat assay | EFM | 4 °C | 1 week | DNA integrity was significantly higher than cryopreservation | [34] |
| Acridine orange | G-IVFTM | 4–6 or 25 °C | 12 days | Sperm DNA remained unchanged after 12 days at both temperatures | [107] |
| ART | Time Interval of Incubation | Extender/Pre-Treatment | Temperature | Main Results | Reference |
|---|---|---|---|---|---|
| ICSI | 3.66 ± 2.26 h | Swim-up/Commercial Culture Medium | 37 °C | No adverse effects on the pregnancy and live birth rates | [12] |
| IVF/ICSI | Home collection to processing: within 1.5 h | DGC/Commercial Culture Medium | Close to 37 °C | No significant difference in oocytes, embryos, pregnancy rates | [9] |
| ICSI | 2 weeks | Electrolyte-free medium (EFM) | 4 °C | High pregnancy rate and high physical status scores in the resulting newborns | [10] |
| IVF/ICSI | Home collection to processing: up to 2 h | DGC/HTF | 37 °C | Does not negatively impact sperm parameters, fertilization rate, pregnancy rates | [8] |
| ICSI | 5 h | DGC/Commercial Culture Medium | 37 °C | No influence on fertilization rates | [103] |
| IUI | Within 24 h | DGC/Commercial Culture Medium | 21–37 °C | No negative effect on pregnancy rate | [149] |
| IUI | Not clear | DGC/Commercial Culture Medium | RT or 37 °C | No negative effect on clinical pregnancy rate when preserving at RT or at 37 °C | [148] |
| IUI | Semen collection at clinic or at home. Delaying semen processing: 0.5 h up to 1 h; Delaying IUI: 1.5 h up to 2 h | DGC/Commercial Culture Medium | 21–40 °C or 37 °C | Delaying semen collection, processing and IUI compromise the pregnancy outcome | [151] |
| IUI | 40–80 min | DGC or Swim-up/Commercial Culture Medium | RT or 37 °C | Sperm preparation for 40–80 min has a potential positive effect on pregnancy rate. | [152] |
| IUI | 107 min between semen production and IUI | DGC/HTF | Not clear | Time intervals between semen production and analysis and IUI significantly influenced clinical pregnancies and live births and should be kept low. | [153] |
| IUI | Semen collection to washing: 40 min Semen washing to IUI: 60 min Semen collection to IUI: 90 min | Swim-up/Commercial Culture Medium | RT | Shorter time interval between semen collection, washing and IUI resulted in higher pregnancy rate. | [150] |
| IUI | 24 h | TEST-Yolk Buffer/Commercial Culture Medium | 5 °C | Similar pregnancy rates were obtained between the immediately IUI group and 24 h storage group | [51] |
| IUI | 2 h | Swim-up/TEST-Yolk Buffer/Earle’s medium | 4 °C | TYB significantly increased pregnancy rate in patients with unexplained infertility but not in those with male factor | [154] |
| IVF | 2 h | Double-step washing technique/TEST-Yolk Buffer/Ham’s F-10 medium | RT | Significantly improved fertilization in couples who had low fertilization rates | [155] |
| IVF | 2 h | DGC/TEST-Yolk Buffer/Commercial Culture Medium | RT | Incubation of TEST-Yolk Buffer produces significantly higher fertilization rates than standard IVF preparation | [156] |
| IVF | 24 h | TEST-Yolk Buffer-treated: DGC/Ham’s F10 medium Fresh semen: swim-up/Ham’s F10 medium | 4 °C | TEST-yolk buffer pretreatment of sperm for 24 h results in higher fertilization rates during IVF among suspected male factor patients. | [157] |
| IUI | Not clear | TEST-Yolk Buffer/BWW | Not clear | Higher pregnancy rates with TEST-Yolk than BWW | [158] |
| IVF | 2 h | Percoll processed/TEST-Yolk Buffer/Milk/Ham’s F10 medium | 5 °C | Preincubation of spermatozoa in milk or TEST-Yolk yields a similar IVF outcome | [159] |
| Names | RM | CYB | EFM | Artificial Seminal Fluid | Human Tubal Fluid |
|---|---|---|---|---|---|
| Components | |||||
| Buffers | Tris (80 mM) | Tris (46 mM) | - | NaCl (2.69 g/L); KCl (0.43 g/L); K2HPO4 (1.91 g/L); MgSO4 (0.54 g/L); | NaCl (5.93 g/L); KCl (0.35 g/L); KH2PO4 (0.05 g/L); |
| TES (176 mM) | TES (108 mM) | - | ZnSO4·7H2O (0.5 g/L); CaCl2·7H2O (0.73 g/L); NaHCO3 (2.1 g/L); Sodium | NaCl (5.93 g/L); KCl (0.35 g/L); KH2PO4 (0.05 g/L); | |
| Sodium citrate (131 mM) | - | Citrate·2H2O (8.13 g/L); Sodium Pyruvate (0.374 g/L); Sodium Lactate (0.779 g/L) | CaCl2 ·2H2O (0.3 g/L); MgSO4·7H2O (0.05 g/L); Pyruvate (0.036 g/L); Lactate (4 g/L) | ||
| pH | 7.0 | 7.3–7.5 | 7.0 | 7.4–7.7 | 7.4 |
| Osmolarity (mOs/kg)) | 325 ± 2 | 290–320 | 325–354 | 280–295 | |
| Energy sources | Glucose (9 mM) | Fructose (95 mM) | Glucose (0.33 M) | Glucose·1H2O (1.12 g/L) and fructose (2.72 g/L) | Glucose (0.5 g/L) |
| Membrane protectant (v/v) | Egg yolk (20%) | Egg yolk (20%) | BSA (3%) | BSA (5.04 g/100 mL) | HSA (optional) |
| Antioxidants | - | α-tocopherol (1 mM) | - | Urate (0.07 g/L); urea (0.72 g/L) | |
| Antibiotics | Gentamicin (10 mg/L) | P-S (10000 U) | - | Gentamycin (40 mg/L) | P-S (10000 U) |
| Pre-treatment | 1:1 dilution | 1:1 dilution | Removal of SP | Removal of SP | Removal of SP |
| Storage Temperature | RT, 2–5 °C | RT | 2–5 °C | RT | RT, 2–5 °C, 37 °C |
| Storage Duration | Up to 4 days | 25–30 h | Up to 7 weeks | 24 h | 2–24 h (most of experiments) |
| Application | ART; Cryopreservation | Laboratorial test | ART | Pharmacy; ART | ART; Cryopreservation |
| Commercialization | Yes | No | No | No | Yes |
| Reference | [21,106] | [22,23] | [10,11,266] | [56,119] | [37,115,201] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cheng, Q.; Li, L.; Jiang, M.; Liu, B.; Xian, Y.; Liu, S.; Liu, X.; Zhao, W.; Li, F. Extend the Survival of Human Sperm In Vitro in Non-Freezing Conditions: Damage Mechanisms, Preservation Technologies, and Clinical Applications. Cells 2022, 11, 2845. https://doi.org/10.3390/cells11182845
Cheng Q, Li L, Jiang M, Liu B, Xian Y, Liu S, Liu X, Zhao W, Li F. Extend the Survival of Human Sperm In Vitro in Non-Freezing Conditions: Damage Mechanisms, Preservation Technologies, and Clinical Applications. Cells. 2022; 11(18):2845. https://doi.org/10.3390/cells11182845
Chicago/Turabian StyleCheng, Qingyuan, Liman Li, Min Jiang, Bo Liu, Yang Xian, Shasha Liu, Xiao Liu, Wenrui Zhao, and Fuping Li. 2022. "Extend the Survival of Human Sperm In Vitro in Non-Freezing Conditions: Damage Mechanisms, Preservation Technologies, and Clinical Applications" Cells 11, no. 18: 2845. https://doi.org/10.3390/cells11182845
APA StyleCheng, Q., Li, L., Jiang, M., Liu, B., Xian, Y., Liu, S., Liu, X., Zhao, W., & Li, F. (2022). Extend the Survival of Human Sperm In Vitro in Non-Freezing Conditions: Damage Mechanisms, Preservation Technologies, and Clinical Applications. Cells, 11(18), 2845. https://doi.org/10.3390/cells11182845






