Understanding Metabolic Alterations in Cancer Cachexia through the Lens of Exercise Physiology
Abstract
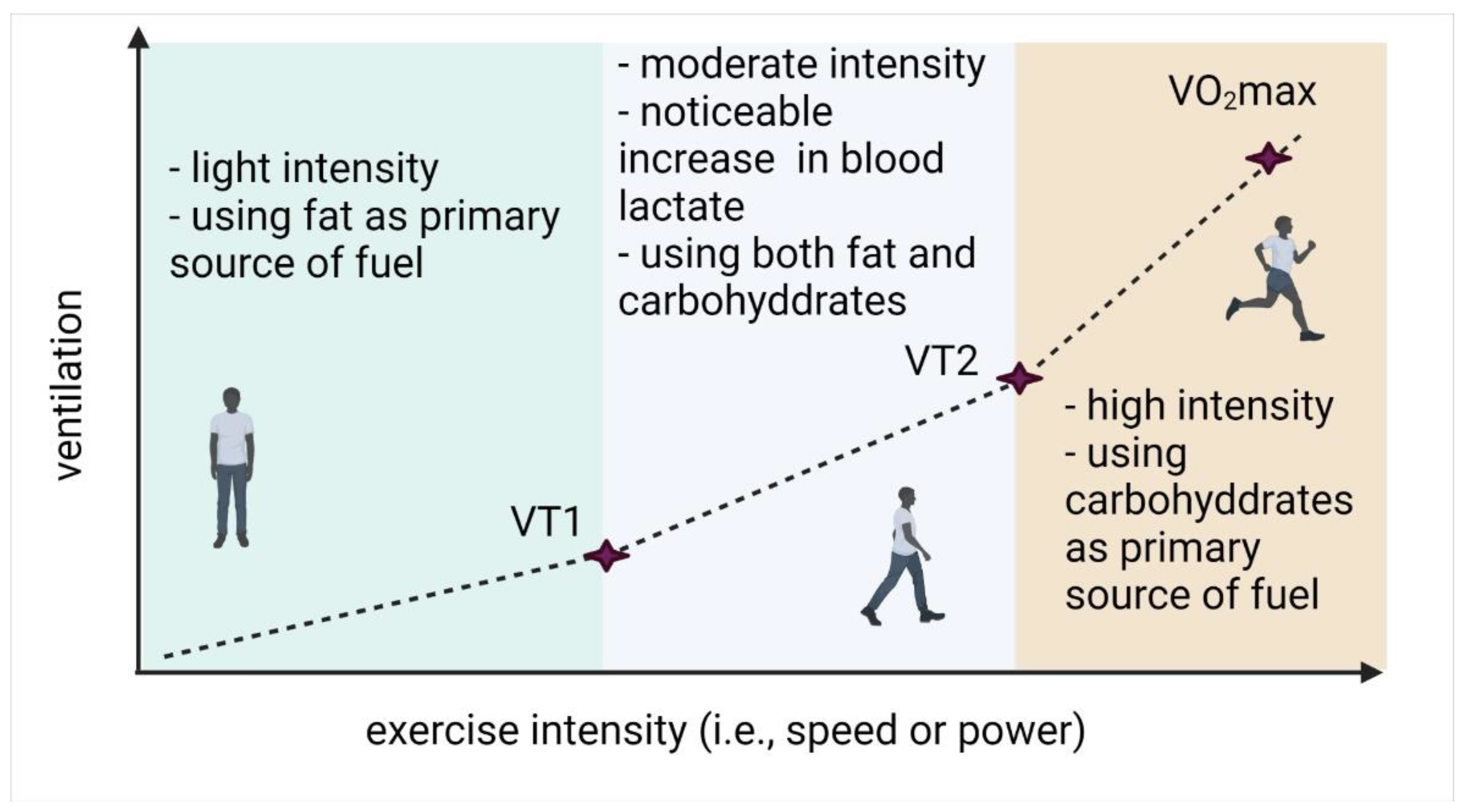
:1. Introduction
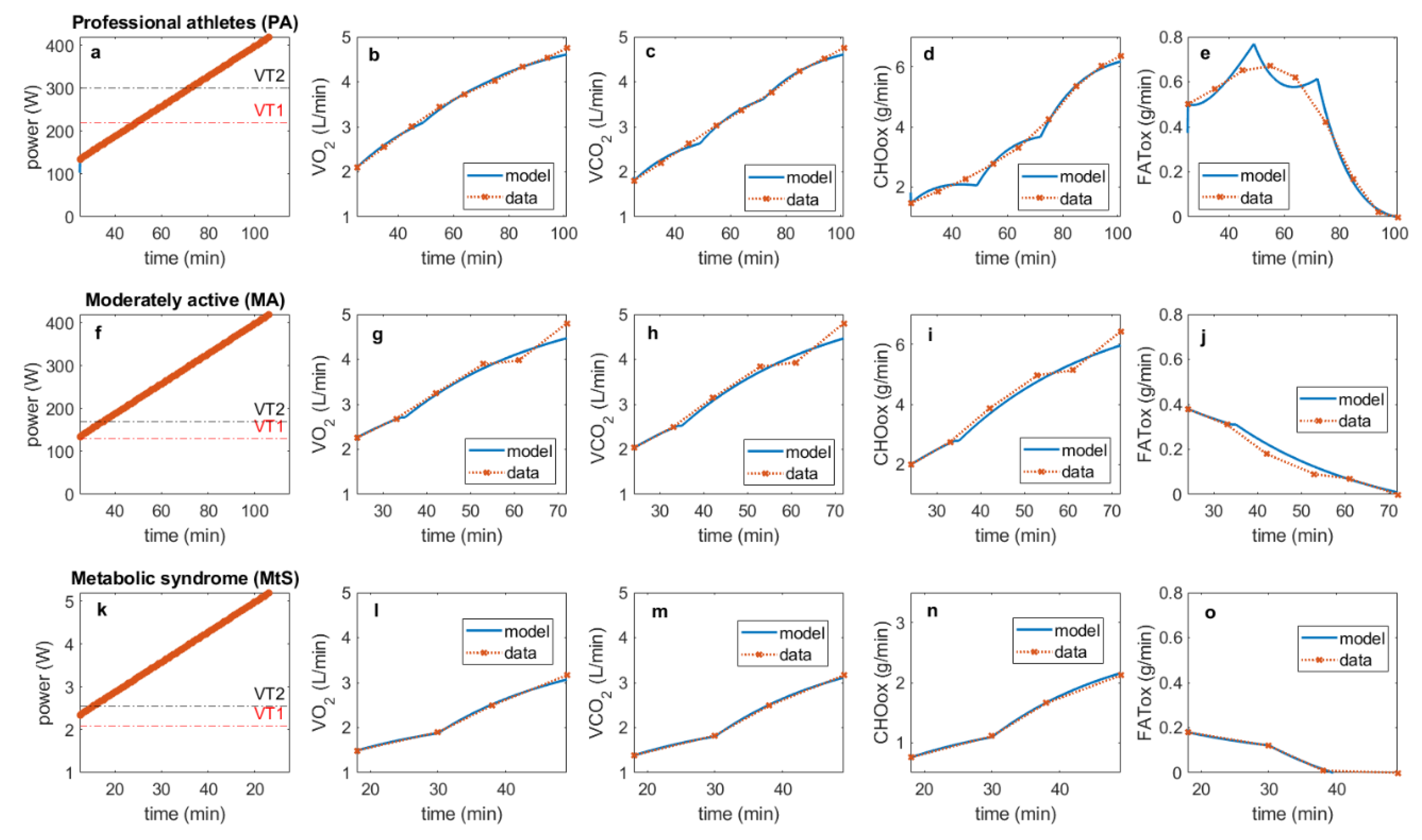
2. Materials and Methods
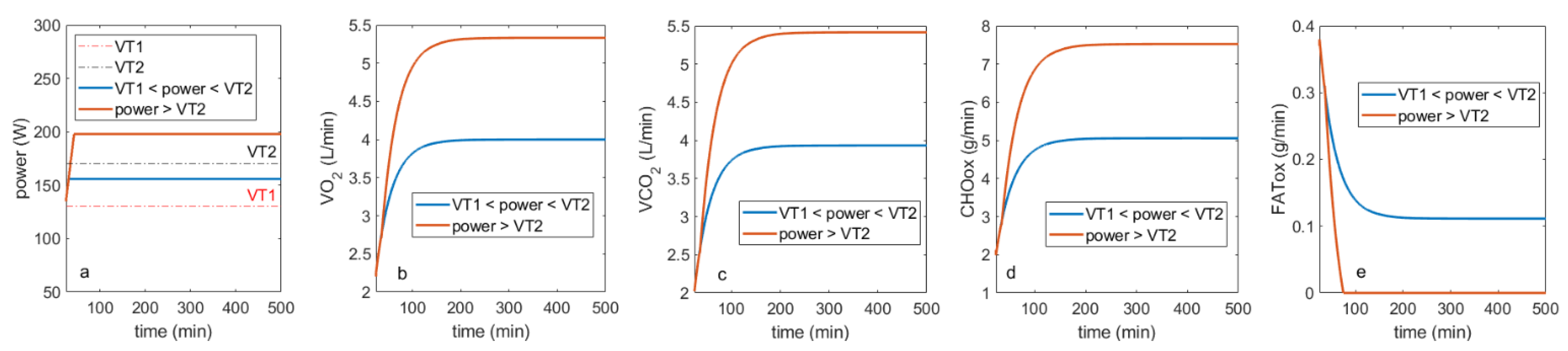
Simulation of “Tumor-like” Effort
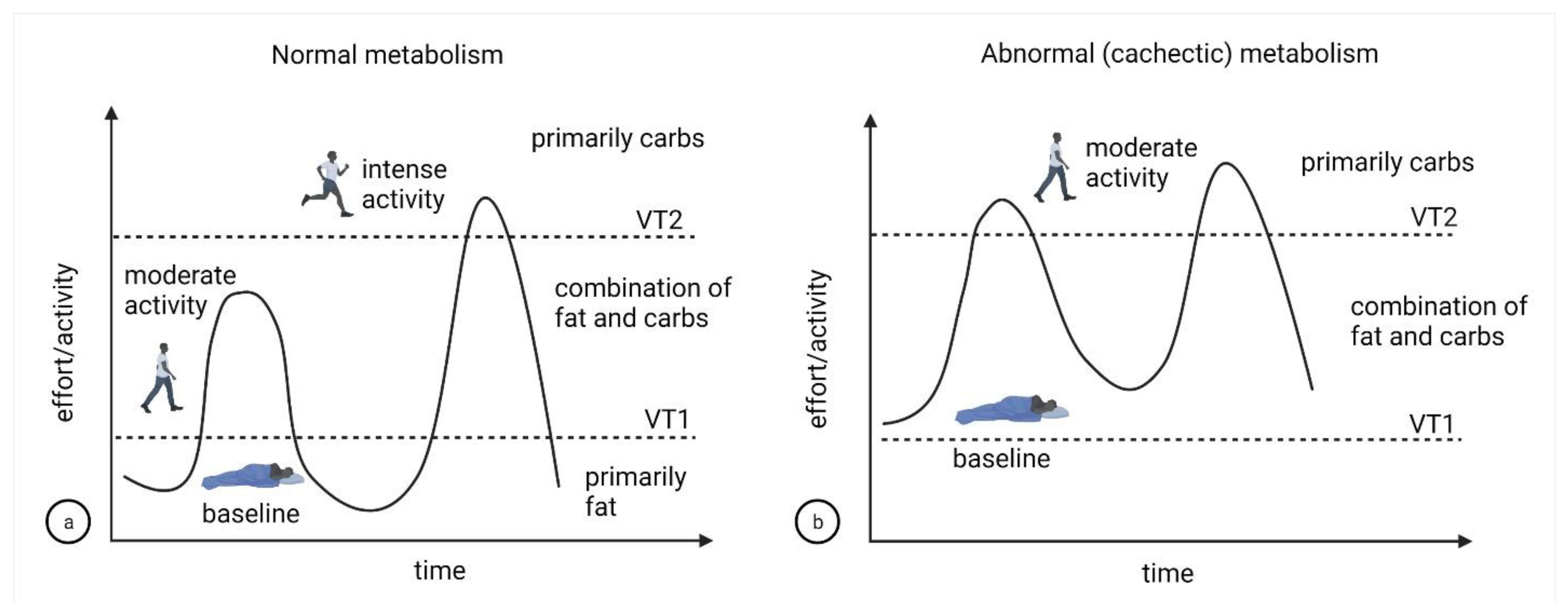
3. Discussion
“Worsley’s access to near-instantaneous help … allowed him to push much closer to the margins—to empty his tank day after day, after struggling through the snow for 12, 14, or 16 h; to ignore his increasing weakness and 50-pound weight loss…On January 21, his seventieth day of travel, he made the call…The next day, he was picked up for the six-hour flight back to Union Glacier, where logistical support for Antarctic expeditions is based, and then airlifted to the hospital in Punta Aernas, Chile, to be treated for exhaustion and dehydration… In the hospital, though, the situation took an unexpected turn: Worsley was diagnosed with bacterial peritonitis, an infection of the abdominal linings, and rushed into surgery. On January 24th, at the age of fifty-five, Henry Worsley died of widespread organ failure”.
4. The Obesity Paradox
5. Validation and Interventions
Supplementary Materials
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Sadeghi, M.; Keshavarz-Fathi, M.; Baracos, V.; Arends, J.; Mahmoudi, M.; Rezaei, N. Cancer cachexia: Diagnosis, assessment, and treatment. Crit. Rev. Oncol./Hematol. 2018, 127, 91–104. [Google Scholar] [CrossRef] [PubMed]
- Roeland, E.J.; Bohlke, K.; Baracos, V.E.; Bruera, E.; Del Fabbro, E.; Dixon, S.; Fallon, M.; Herrstedt, J.; Lau, H.; Platek, M.; et al. Management of cancer cachexia: ASCO guideline. Am. Soc. Clin. Oncol. 2020, 38, 2438–2453. [Google Scholar] [CrossRef] [PubMed]
- Argilés, J.M.; Busquets, S.; López-Soriano, F.J. Cancer cachexia, a clinical challenge. Curr. Opin. Oncol. LWW 2019, 31, 286–290. [Google Scholar] [CrossRef] [PubMed]
- Kelley, D.E.; He, J.; Menshikova, E.V.; Ritov, V.B. Dysfunction of mitochondria in human skeletal muscle in type 2 diabetes. Diabetes. Am. Diabetes Assoc. 2002, 51, 2944–2950. [Google Scholar] [CrossRef] [Green Version]
- Segal, K.R. Comparison of indirect calorimetric measurements of resting energy expenditure with a ventilated hood, face mask, and mouthpiece. Am. J. Clin. Nutr. 1987, 45, 1420–1423. [Google Scholar] [CrossRef] [PubMed]
- Ferrannini, E. The theoretical bases of indirect calorimetry: A review. Metabolism 1988, 37, 287–301. [Google Scholar] [CrossRef]
- Frayn, K. Calculation of substrate oxidation rates in vivo from gaseous exchange. J. Appl. Physiol. 1983, 55, 628–634. [Google Scholar] [CrossRef] [Green Version]
- San-Millán, I.; Brooks, G.A. Assessment of metabolic flexibility by means of measuring blood lactate, fat, and carbohydrate oxidation responses to exercise in professional endurance athletes and less-fit individuals. Sports Med. 2018, 48, 467–479. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Huang, P.L. A comprehensive definition for metabolic syndrome. Dis. Models Mech. 2009, 2, 231–237. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Esposito, K.; Chiodini, P.; Colao, A.; Lenzi, A.; Giugliano, D. Metabolic syndrome and risk of cancer: A systematic review and meta-analysis. Diabetes Care. Am. Diabetes Assoc. 2012, 35, 2402–2411. [Google Scholar] [CrossRef] [Green Version]
- Gouw, A.H.; Van Guilder, G.P.; Larusson, A.; Laredo, G.; Weatherwax, R.M.; Byrd, B.R.; Dalleck, L.C. Ventilation can exclusively be used to predict ventilatory thresholds: A retrospective analysis. Int. J. Res. Ex. Phys. 2021, 16, 1–18. [Google Scholar]
- Anselmi, F.; Cavigli, L.; Pagliaro, A.; Valente, S.; Valentini, F.; Cameli, M.; Focardi, M.; Mochi, N.; Dendale, P.; Hansen, D.; et al. The importance of ventilatory thresholds to define aerobic exercise intensity in cardiac patients and healthy subjects. Scand. J. Med. Sci. Sports 2021, 31, 1796–1808. [Google Scholar] [CrossRef] [PubMed]
- Bhambhani, Y.; Singh, M. Ventilatory Thresholds during a Graded Exercise Test. Respiration 1985, 47, 120–128. [Google Scholar] [CrossRef] [PubMed]
- Neder, J.A.; Stein, R.A. Simplified strategy for the estimation of the exercise ventilatory thresholds. Med. Sci. Sports Exerc. 2006, 38, 1007. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Goodwin, M.L.; Harris, J.E.; Hernández, A.; Gladden, L.B. Blood lactate measurements and analysis during exercise: A guide for clinicians. J. Diabetes Sci. Technol. 2007, 1, 558–569. [Google Scholar] [CrossRef] [Green Version]
- Ramos-Jiménez, A.; Hernández-Torres, R.P.; Torres-Durán, P.V.; Romero-Gonzalez, J.; Mascher, D.; Posadas-Romero, C.; Juárez-Oropeza, M.A. The respiratory exchange ratio is associated with fitness indicators both in trained and untrained men: A possible application for people with reduced exercise tolerance. Clin. Med. Circ. Respir. Pulm. Med. 2008, 2, CCRPM-S449. [Google Scholar] [CrossRef]
- Olson, B.; Marks, D.L.; Grossberg, A.J. Diverging metabolic programmes and behaviours during states of starvation, protein malnutrition, and cachexia. J. Cachexia Sarcopenia Muscle 2020, 11, 1429–1446. [Google Scholar] [CrossRef] [PubMed]
- Michaelis, K.A.; Zhu, X.; Burfeind, K.G.; Krasnow, S.M.; Levasseur, P.R.; Morgan, T.K.; Marks, D.L. Establishment and characterization of a novel murine model of pancreatic cancer cachexia. J. Cachexia Sarcopenia Muscle 2017, 8, 824–838. [Google Scholar] [CrossRef]
- Kareva, I.; Abou-Slaybi, A.; Dodd, O.; Dashevsky, O.; Klement, G.L. Normal wound healing and tumor angiogenesis as a game of competitive inhibition. PLoS ONE 2016, 11, e0166655. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dvorak, H.F. Tumors: Wounds that do not heal. N. Engl. J. Med. Mass Med. Soc. 1986, 315, 1650–1659. [Google Scholar] [CrossRef] [Green Version]
- Kareva, I. Cancer as a disease of homeostasis: An angiogenesis perspective. In Ecology and Evolution of Cancer; Elsevier: Amsterdam, The Netherlands, 2017; pp. 229–235. [Google Scholar]
- Holtan, S.G.; Creedon, D.J.; Haluska, P.; Markovic, S.N. Cancer and pregnancy: Parallels in growth, invasion, and immune modulation and implications for cancer therapeutic agents. In Mayo Clinic Proceedings; Elsevier: Amsterdam, The Netherlands, 2009; pp. 985–1000. [Google Scholar]
- Kareva, I. Immune suppression in pregnancy and cancer: Parallels and insights. Transl. Oncol. 2020, 13, 10In0759. [Google Scholar] [CrossRef] [PubMed]
- Marshall, S.; Agarwal, E. Comparing characteristics of malnutrition, starvation, sarcopenia and cachexia in older adults. In Handbook of Famine, Starvation, and Nutrient Deprivation: From Biology to Policy; Springer: Berlin/Heidelberg, Germany, 2017; pp. 1–23. [Google Scholar]
- Thomas, D.R. Distinguishing starvation from cachexia. Clin. Geriatr. Med. 2002, 18, 883–891. [Google Scholar] [CrossRef]
- Kimmelman, A.C.; White, E. Autophagy and tumor metabolism. Cell Metab. 2017, 25, 1037–1043. [Google Scholar] [CrossRef]
- Poillet-Perez, L.; White, E. Role of tumor and host autophagy in cancer metabolism. Genes Dev. 2019, 33, 610–619. [Google Scholar] [CrossRef] [Green Version]
- Storlien, L.; Oakes, N.D.; Kelley, D.E. Metabolic flexibility. Proc. Nutr. Soc. 2004, 63, 363–368. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kelley, D.E.; Mandarino, L.J. Fuel selection in human skeletal muscle in insulin resistance: A reexamination. Diabetes 2000, 49, 677–683. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Koutnik, A.P.; Poff, A.M.; Ward, N.P.; DeBlasi, J.M.; Soliven, M.A.; Romero, M.A.; Roberson, P.A.; Fox, C.D.; Roberts, M.D.; D’Agostino, D.P. Ketone bodies attenuate wasting in models of atrophy. J. Cachexia Sarcopenia Muscle 2020, 11, 973–996. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hutchinson, A.; Gladwell, M. Endure: Mind, Body, and the Curiously Elastic Limits of Human Performance; HarperCollins: New York, NY, USA, 2018. [Google Scholar]
- Mokdad, A.H.; Ford, E.S.; Bowman, B.A.; Dietz, W.H.; Vinicor, F.; Bales, V.S.; Marks, J.S. Prevalence of obesity, diabetes, and obesity-related health risk factors, 2001. JAMA Am. Med. Assoc. 2003, 289, 76–79. [Google Scholar] [CrossRef]
- Ahima, R.S.; Lazar, M.A. The health risk of obesity—Better metrics imperative. Science 2013, 341, 856–858. [Google Scholar] [CrossRef] [PubMed]
- Pauling, L. The relation between longevity and obesity in human beings. Proc. Natl. Acad. Sci. USA 1958, 44, 619–622. [Google Scholar] [CrossRef] [Green Version]
- Weiss, E.P.; Jordan, R.C.; Frese, E.M.; Albert, S.G.; Villareal, D.T. Effects of weight loss on lean mass, strength, bone, and aerobic capacity. Med. Sci. Sports Exerc. 2017, 49, 206. [Google Scholar] [CrossRef] [PubMed]
- Nonaka, Y.; Terada, S. Differential effects of rapid or slow body weight loss on muscle weight and protein degradation pathways in rat skeletal muscle. J. Int. Soc. Sports Nutr. 2015, 12 (Suppl. S1), 58. [Google Scholar] [CrossRef] [Green Version]
- Gonzalez, M.C.; Pastore, C.A.; Orlandi, S.P.; Heymsfield, S.B. Obesity paradox in cancer: New insights provided by body composition. Am. J. Clin. Nutr. 2014, 99, 999–1005. [Google Scholar] [CrossRef]
- Daly, L.E.; Ni Bhuachalla, E.B.; Power, D.G.; Cushen, S.J.; James, K.; Ryan, A.M. Loss of skeletal muscle during systemic chemotherapy is prognostic of poor survival in patients with foregut cancer. J. Cachexia Sarcopenia Muscle 2018, 9, 315–325. [Google Scholar] [CrossRef] [Green Version]
- Stokes, A. Using maximum weight to redefine body mass index categories in studies of the mortality risks of obesity. Popul. Health Metr. 2014, 12, 6. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Martini, A.D.; Dalleck, L.C.; Mejuto, G.; Larwood, T.; Weatherwax, R.M.; Ramos, J.S. Changes in the Second Ventilatory Threshold Following Individualised versus Standardised Exercise Prescription among Physically Inactive Adults: A Randomised Trial. Int. J. Environ. Res. Public Health 2022, 19, 3962. [Google Scholar] [CrossRef] [PubMed]
- Lorenz, K.A.; Yeshurun, S.; Aziz, R.; Ortiz-Delatorre, J.; Bagley, J.R.; Mor, M.; Kern, M. A handheld metabolic device (lumen) to measure fuel utilization in healthy young adults: Device validation study. Interact. J. Med. Res. 2021, 10, 25371. [Google Scholar] [CrossRef]
- Kliewer, K.L.; Ke, J.-Y.; Tian, M.; Cole, R.M.; Andridge, R.R.; Belury, M.A. Adipose tissue lipolysis and energy metabolism in early cancer cachexia in mice. Cancer Biol. Ther. 2015, 16, 886–897. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wolpern, A.E.; Burgos, D.J.; Janot, J.M.; Dalleck, L.C. Is a threshold-based model a superior method to the relative percent concept for establishing individual exercise intensity? A randomized controlled trial. BMC Sports Sci. Med. Rehabil. 2015, 7, 16. [Google Scholar] [CrossRef] [Green Version]
- San-Millan, I. The Use of Individualized Exercise Prescription to Target Oxidative Metabolism in A Stage Iv Colorectal, Metastatic Cancer Patient: 581 May 29 1: 20 PM–1: 40 PM. Med. Sci. Sports Exerc. LWW 2019, 51, 151. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kareva, I. Understanding Metabolic Alterations in Cancer Cachexia through the Lens of Exercise Physiology. Cells 2022, 11, 2317. https://doi.org/10.3390/cells11152317
Kareva I. Understanding Metabolic Alterations in Cancer Cachexia through the Lens of Exercise Physiology. Cells. 2022; 11(15):2317. https://doi.org/10.3390/cells11152317
Chicago/Turabian StyleKareva, Irina. 2022. "Understanding Metabolic Alterations in Cancer Cachexia through the Lens of Exercise Physiology" Cells 11, no. 15: 2317. https://doi.org/10.3390/cells11152317
APA StyleKareva, I. (2022). Understanding Metabolic Alterations in Cancer Cachexia through the Lens of Exercise Physiology. Cells, 11(15), 2317. https://doi.org/10.3390/cells11152317





