Human Basal and Suprabasal Keratinocytes Are Both Able to Generate and Maintain Dermo–Epidermal Skin Substitutes in Long-Term In Vivo Experiments
Abstract
1. Introduction
2. Materials and Methods
2.1. Histological and Immunofluorescence Staining
2.2. Cell Isolation and Culture
2.3. Ki67 Quantification
2.4. Fluorescence Activated Cell Sorting (FACS) and Analysis
2.5. Proteomics Analysis of Sorted Basal and Suprabasal Keratinocytes
2.6. Colony Formation Assay
2.7. Raman Microscopic Measurements
2.8. Organotypic Cultures
2.9. In Vivo Transplantation
3. Results
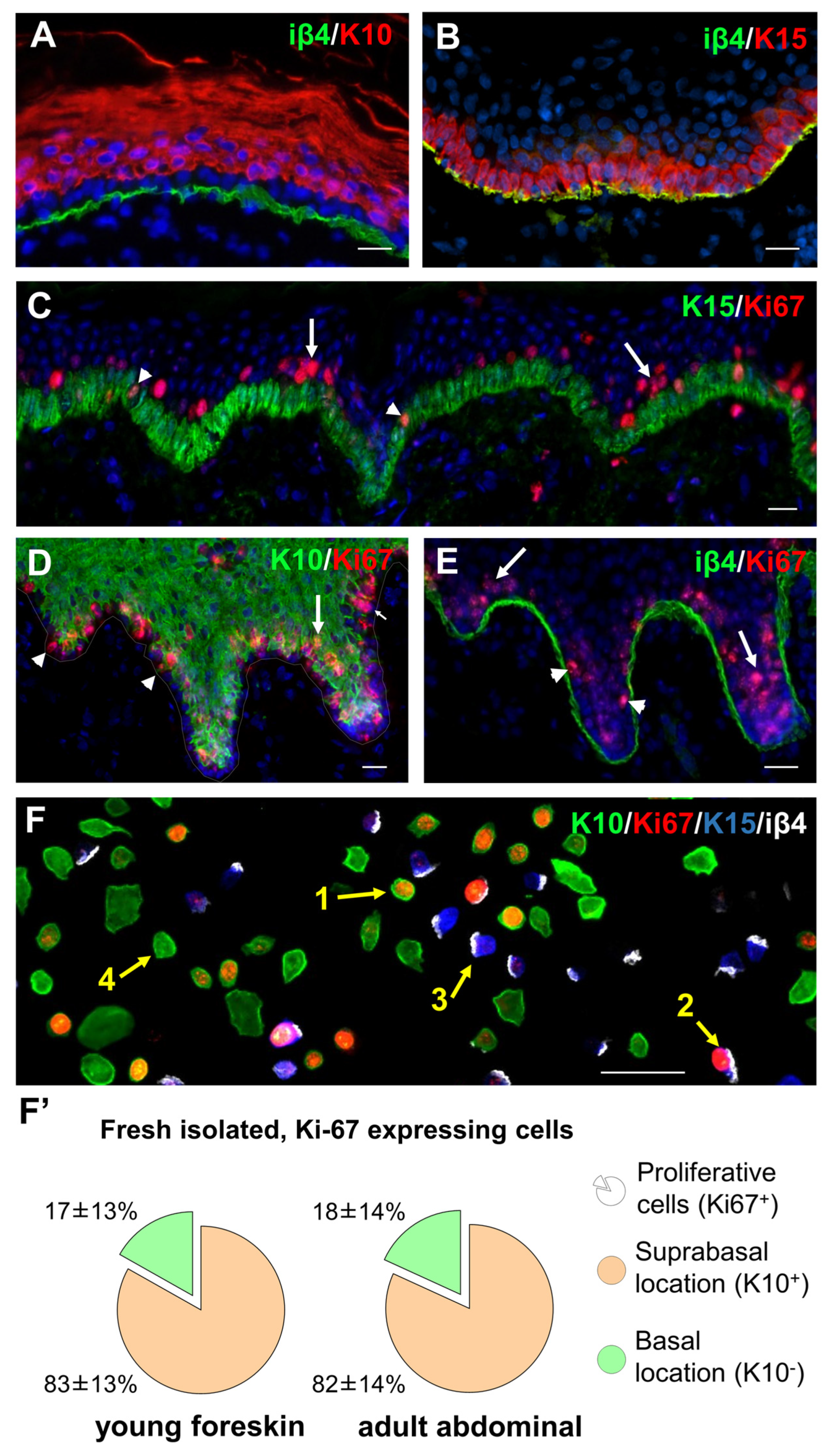
3.1. Suprabasal Keratinocytes Proliferate in the Human Interfollicular Epidermis under Physiological Conditions
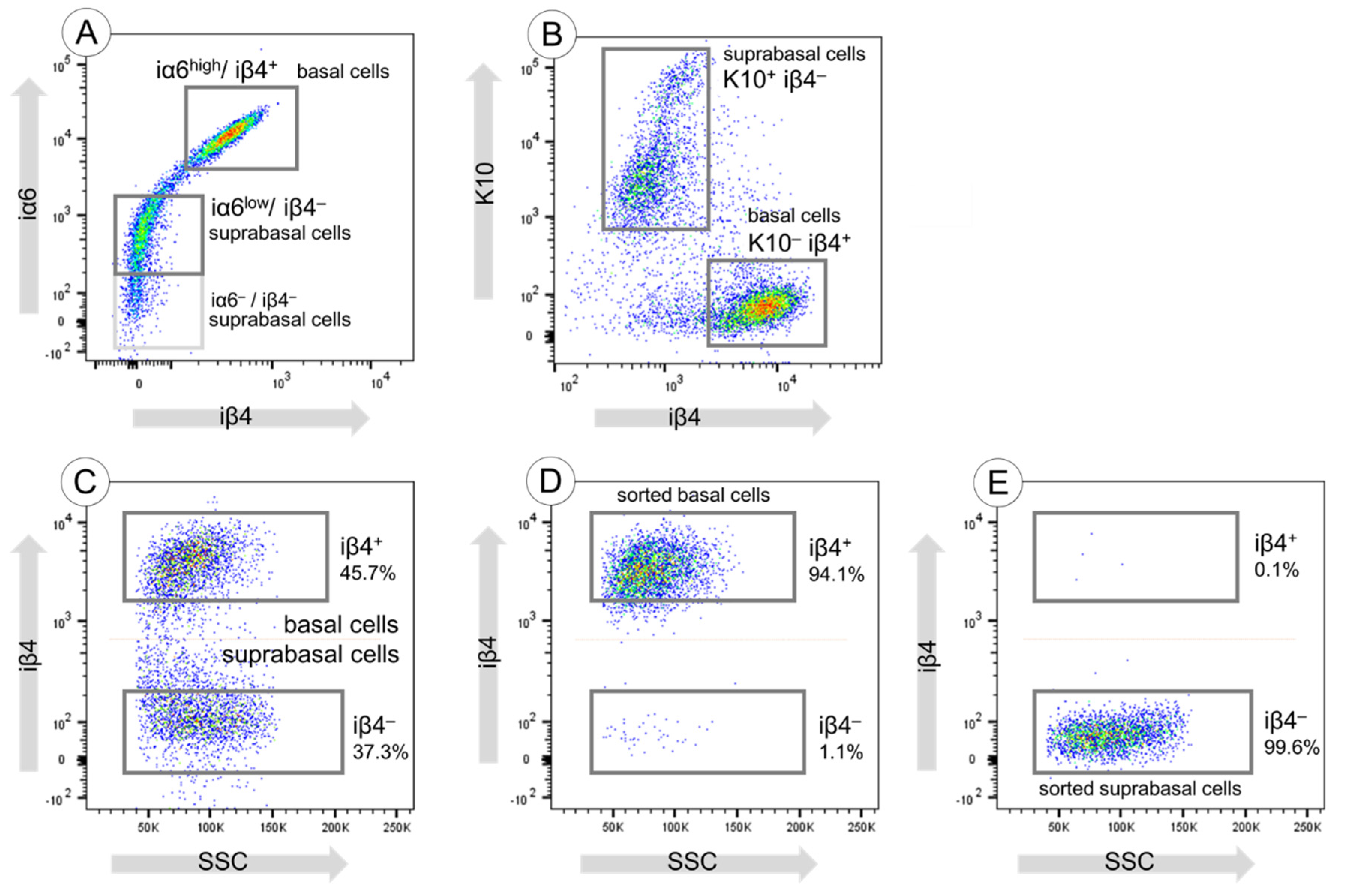
3.2. Sorting of Basal and Suprabasal Keratinocytes from Human Skin
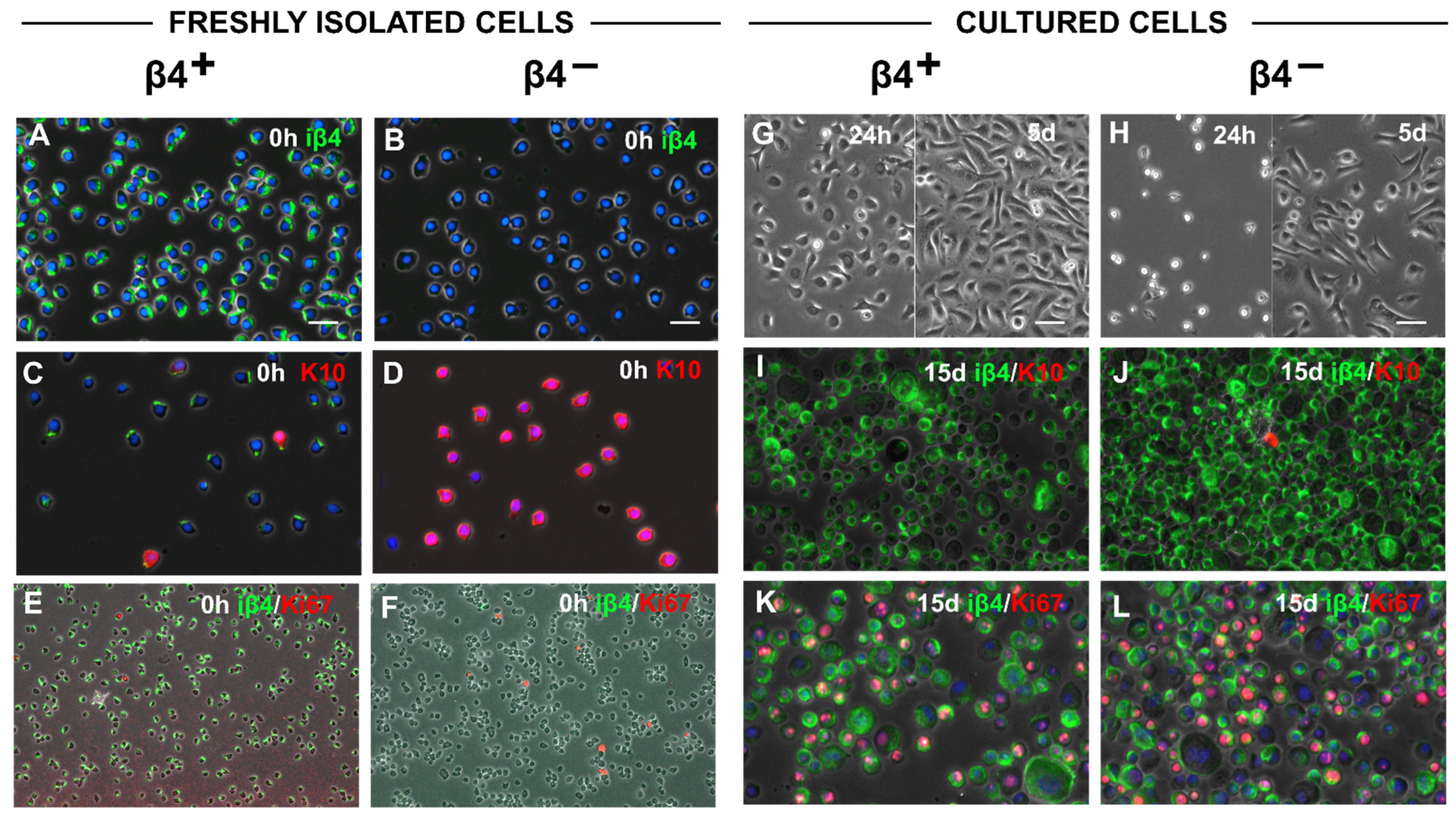
3.3. Slow Adhering iβ4− Cells Regain the Ability to Adhere and Proliferate in Culture
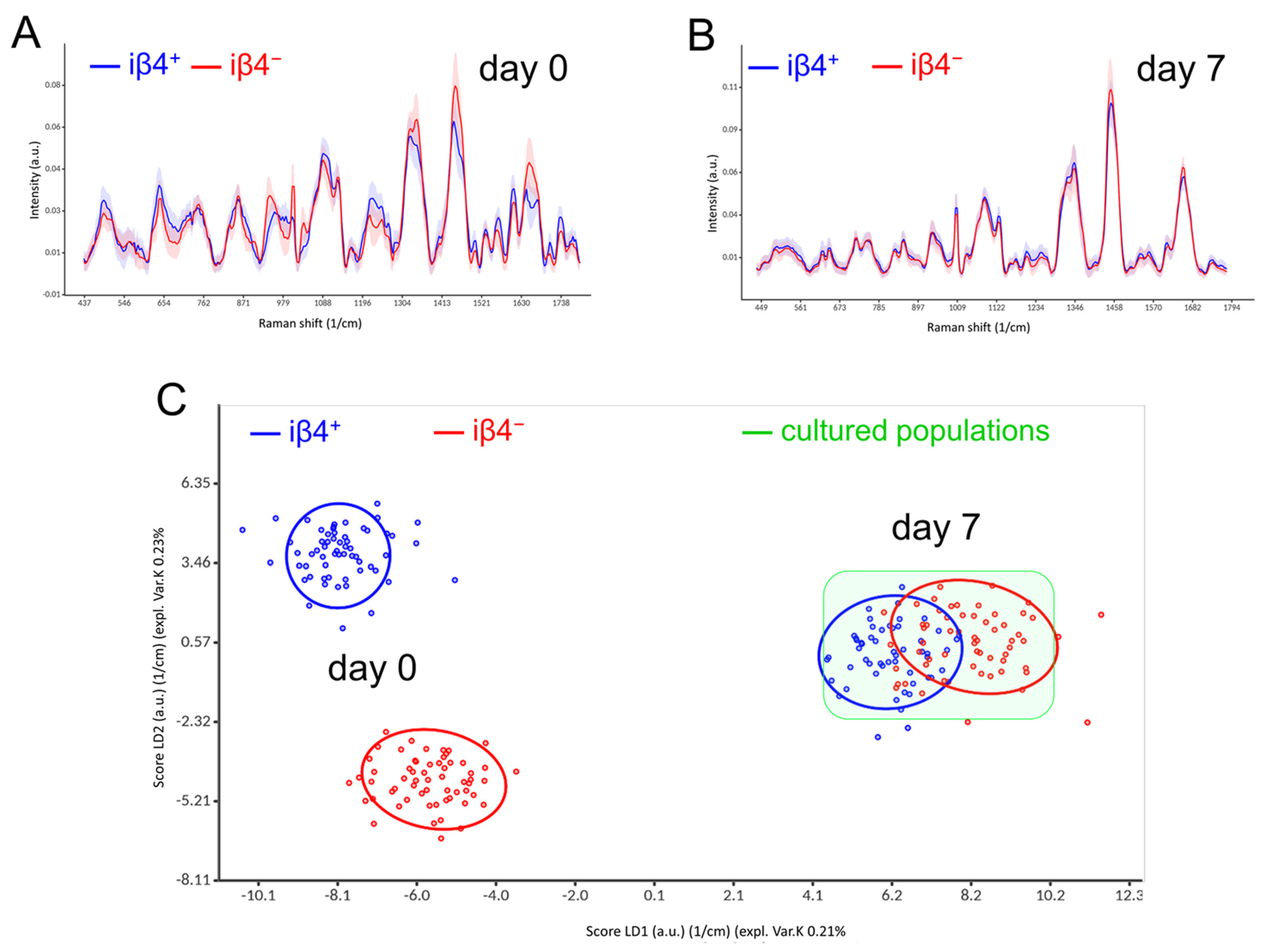
3.4. Basal and Suprabasal Keratinocytes Lose Their Distinct Raman Spectra In Vitro
3.5. Slow Adhering iβ4− Cells Regain the Ability to form Colonies in Culture
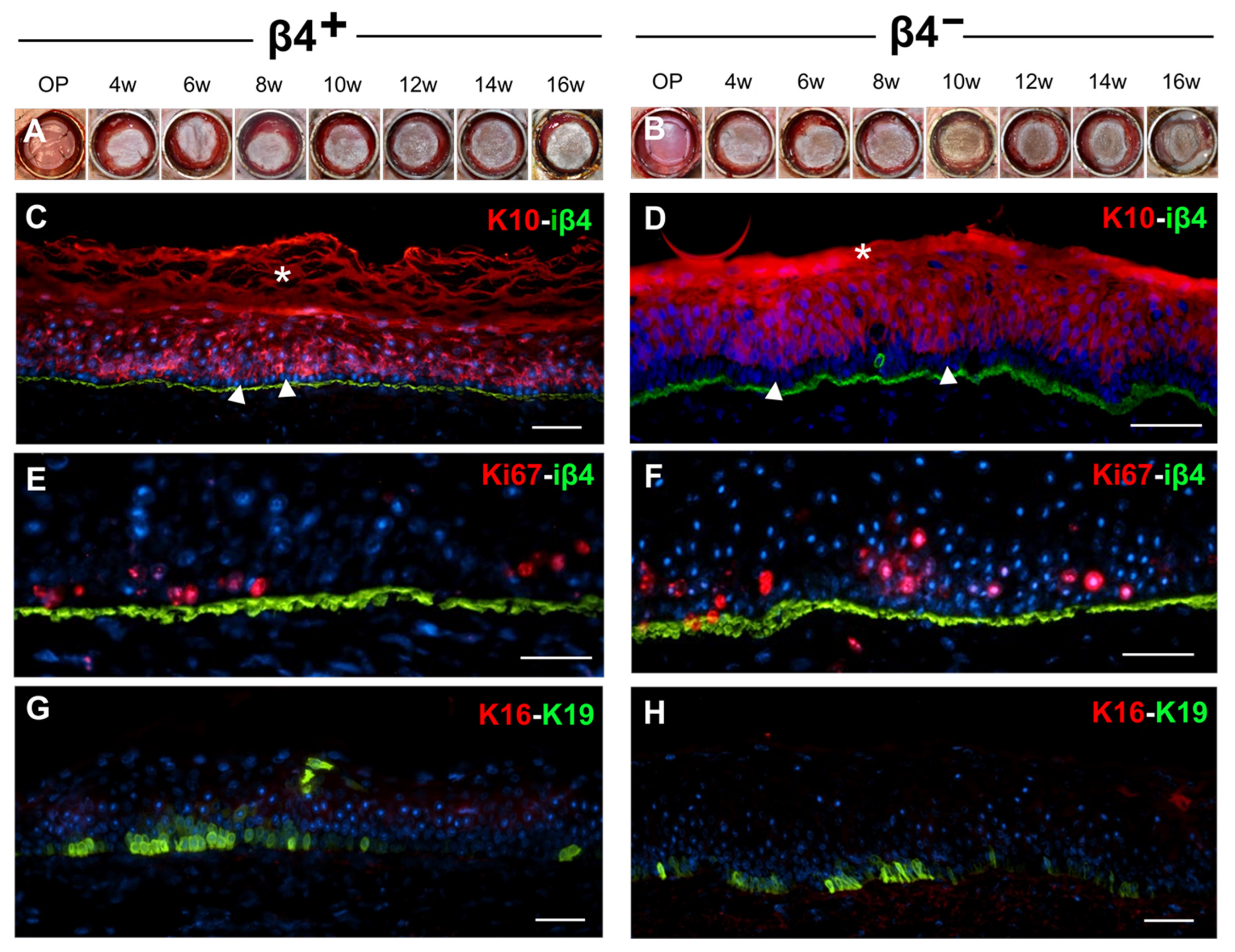
3.6. Basal and Suprabasal Keratinocytes Are Competent for Ensuring the Long-Term Survival of Human Dermo–Epidermal Skin Substitutes In Vivo
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Hoath, S.B.; Leahy, D.G. The organization of human epidermis: Functional epidermal units and phi proportionality. J. Investig. Dermatol. 2003, 121, 1440–1446. [Google Scholar] [CrossRef] [PubMed]
- Watt, F.M. Epidermal stem cells: Markers, patterning and the control of stem cell fate. Philos. Trans. R. Soc. Lond. B Biol. Sci. 1998, 353, 831–837. [Google Scholar] [CrossRef] [PubMed]
- Ghazizadeh, S.; Taichman, L.B. Organization of stem cells and their progeny in human epidermis. J. Investig. Dermatol. 2005, 124, 367–372. [Google Scholar] [CrossRef]
- Legg, J.; Jensen, U.B.; Broad, S.; Leigh, I.; Watt, F.M. Role of melanoma chondroitin sulphate proteoglycan in patterning stem cells in human interfollicular epidermis. Development 2003, 130, 6049–6063. [Google Scholar] [CrossRef] [PubMed]
- Muffler, S.; Stark, H.J.; Amoros, M.; Falkowska-Hansen, B.; Boehnke, K.; Buhring, H.J.; Marme, A.; Bickenbach, J.R.; Boukamp, P. A Stable Niche Supports Long-Term Maintenance of Human Epidermal Stem Cells in Organotypic Cultures. Stem Cells 2008, 26, 2506–2515. [Google Scholar] [CrossRef]
- Sotiropoulou, P.A.; Blanpain, C. Development and homeostasis of the skin epidermis. Cold Spring Harb. Perspect. Biol. 2012, 4, a008383. [Google Scholar] [CrossRef]
- Webb, A.; Li, A.; Kaur, P. Location and phenotype of human adult keratinocyte stem cells of the skin. Differentiation 2004, 72, 387–395. [Google Scholar] [CrossRef]
- Boehnke, K.; Falkowska-Hansen, B.; Stark, H.J.; Boukamp, P. Stem cells of the human epidermis and their niche: Composition and function in epidermal regeneration and carcinogenesis. Carcinogenesis 2012, 33, 1247–1258. [Google Scholar] [CrossRef]
- Lavker, R.M.; Sun, T.T. Heterogeneity in epidermal basal keratinocytes: Morphological and functional correlations. Science 1982, 215, 1239–1241. [Google Scholar] [CrossRef]
- Penneys, N.S.; Fulton, J.E., Jr.; Weinstein, G.D.; Frost, P. Location of proliferating cells in human epidermis. Arch. Dermatol. 1970, 101, 323–327. [Google Scholar] [CrossRef]
- Thuringer, J.M. Regeneration of stratified squamous epithelium. Anat. Rec. 1924, 28, 31–43. [Google Scholar] [CrossRef]
- Alonso, L.; Fuchs, E. Stem cells of the skin epithelium. Proc. Natl. Acad. Sci. USA 2003, 100 (Suppl. S1), 11830–11835. [Google Scholar] [CrossRef]
- Ito, M.; Liu, Y.; Yang, Z.; Nguyen, J.; Liang, F.; Morris, R.J.; Cotsarelis, G. Stem cells in the hair follicle bulge contribute to wound repair but not to homeostasis of the epidermis. Nat. Med. 2005, 11, 1351–1354. [Google Scholar] [CrossRef] [PubMed]
- Terskikh, V.V.; Vasiliev, A.V.; Vorotelyak, E.A. Label retaining cells and cutaneous stem cells. Stem Cell Rev. Rep. 2012, 8, 414–425. [Google Scholar] [CrossRef] [PubMed]
- Fuchs, E. Skin stem cells: Rising to the surface. J. Cell Biol. 2008, 180, 273–284. [Google Scholar] [CrossRef]
- Clayton, E.; Doupé, D.P.; Klein, A.M.; Winton, D.J.; Simons, B.D.; Jones, P.H. A single type of progenitor cell maintains normal epidermis. Nature 2007, 446, 185–189. [Google Scholar] [CrossRef]
- Lechler, T.; Fuchs, E. Asymmetric cell divisions promote stratification and differentiation of mammalian skin. Nature 2005, 437, 275–280. [Google Scholar] [CrossRef]
- Aragona, M.; Dekoninck, S.; Rulands, S.; Lenglez, S.; Mascré, G.; Simons, B.D.; Blanpain, C. Defining stem cell dynamics and migration during wound healing in mouse skin epidermis. Nat. Commun. 2017, 8, 14684. [Google Scholar] [CrossRef]
- Jensen, K.B.; Watt, F.M. Single-cell expression profiling of human epidermal stem and transit-amplifying cells: Lrig1 is a regulator of stem cell quiescence. Proc. Natl. Acad. Sci. USA 2006, 103, 11958–11963. [Google Scholar] [CrossRef]
- Li, A.; Pouliot, N.; Redvers, R.; Kaur, P. Extensive tissue-regenerative capacity of neonatal human keratinocyte stem cells and their progeny. J. Clin. Investig. 2004, 113, 390–400. [Google Scholar] [CrossRef]
- Pellegrini, G.; Dellambra, E.; Golisano, O.; Martinelli, E.; Fantozzi, I.; Bondanza, S.; Ponzin, D.; McKeon, F.; De Luca, M. p63 identifies keratinocyte stem cells. Proc. Natl. Acad. Sci. USA 2001, 98, 3156–3161. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Clevers, H. Coexistence of quiescent and active adult stem cells in mammals. Science 2010, 327, 542–545. [Google Scholar] [CrossRef] [PubMed]
- Mascré, G.; Dekoninck, S.; Drogat, B.; Youssef, K.K.; Broheé, S.; Sotiropoulou, P.A.; Simons, B.D.; Blanpain, C. Distinct contribution of stem and progenitor cells to epidermal maintenance. Nature 2012, 489, 257–262. [Google Scholar] [CrossRef]
- Senoo, M. Epidermal Stem Cells in Homeostasis and Wound Repair of the Skin. Adv. Wound Care 2013, 2, 273–282. [Google Scholar] [CrossRef] [PubMed]
- Raman, C.V.; Krishnan, K.S. A new type of secondary radiation. Nature 1928, 121, 501–502. [Google Scholar] [CrossRef]
- Steinke, M.; Gross, R.; Walles, H.; Gangnus, R.; Schutze, K.; Walles, T. An engineered 3D human airway mucosa model based on an SIS scaffold. Biomaterials 2014, 35, 7355–7362. [Google Scholar] [CrossRef]
- Buchacher, T.; Wiesinger-Mayr, H.; Vierlinger, K.; Ruger, B.M.; Stanek, G.; Fischer, M.B.; Weber, V. Human blood monocytes support persistence, but not replication of the intracellular pathogen C. pneumoniae. BMC Immunol. 2014, 15, 60. [Google Scholar] [CrossRef]
- Aljakouch, K.; Lechtonen, T.; Yosef, H.K.; Hammoud, M.K.; Alsaidi, W.; Kötting, C.; Mügge, C.; Kourist, R.; El-Mashtoly, S.F.; Gerwert, K. Raman Microspectroscopic Evidence for the Metabolism of a Tyrosine Kinase Inhibitor, Neratinib, in Cancer Cells. Angew. Chem. Int. Ed. 2018, 57, 7250–7254. [Google Scholar] [CrossRef]
- Biedermann, T.; Pontiggia, L.; Bottcher-Haberzeth, S.; Tharakan, S.; Braziulis, E.; Schiestl, C.; Meuli, M.; Reichmann, E. Human Eccrine Sweat Gland Cells Can Reconstitute a Stratified Epidermis. J. Investig. Dermatol. 2010, 130, 1996–2009. [Google Scholar] [CrossRef]
- Pontiggia, L.; Biedermann, T.; Meuli, M.; Widmer, D.; Bottcher-Haberzeth, S.; Schiestl, C.; Schneider, J.; Braziulis, E.; Montano, I.; Meuli-Simmen, C.; et al. Markers to evaluate the quality and self-renewing potential of engineered human skin substitutes in vitro and after transplantation. J. Investig. Dermatol. 2009, 129, 480–490. [Google Scholar] [CrossRef]
- Wiśniewski, J.R.; Zougman, A.; Nagaraj, N.; Mann, M. Universal sample preparation method for proteome analysis. Nat. Methods 2009, 6, 359–362. [Google Scholar] [CrossRef] [PubMed]
- Benjamini, Y.; Hochberg, Y. Controlling the False Discovery Rate: A Practical and Powerful Approach to Multiple Testing. J. R. Stat. Soc. Ser. B Methodol. 1995, 57, 289–300. [Google Scholar] [CrossRef]
- Pontiggia, L.; Biedermann, T.; Bottcher-Haberzeth, S.; Oliveira, C.; Braziulis, E.; Klar, A.S.; Meuli-Simmen, C.; Meuli, M.; Reichmann, E. De novo epidermal regeneration using human eccrine sweat gland cells: Higher competence of secretory over absorptive cells. J. Investig. Dermatol. 2014, 134, 1735–1742. [Google Scholar] [CrossRef] [PubMed]
- Schindelin, J.; Arganda-Carreras, I.; Frise, E.; Kaynig, V.; Longair, M.; Pietzsch, T.; Preibisch, S.; Rueden, C.; Saalfeld, S.; Schmid, B.; et al. Fiji: An open-source platform for biological-image analysis. Nat. Methods 2012, 9, 676–682. [Google Scholar] [CrossRef]
- Notingher, I.; Hench, L.L. Raman microspectroscopy: A noninvasive tool for studies of individual living cells in vitro. Expert Rev. Med. Devices 2006, 3, 215–234. [Google Scholar] [CrossRef] [PubMed]
- Costea, D.E.; Loro, L.L.; Dimba, E.A.; Vintermyr, O.K.; Johannessen, A.C. Crucial effects of fibroblasts and keratinocyte growth factor on morphogenesis of reconstituted human oral epithelium. J. Investig. Dermatol. 2003, 121, 1479–1486. [Google Scholar]
- Braziulis, E.; Diezi, M.; Biedermann, T.; Pontiggia, L.; Schmucki, M.; Hartmann-Fritsch, F.; Luginbuhl, J.; Schiestl, C.; Meuli, M.; Reichmann, E. Modified plastic compression of collagen hydrogels provides an ideal matrix for clinically applicable skin substitutes. Tissue Eng. Part C Methods 2012, 18, 464–474. [Google Scholar] [CrossRef]
- Michalczyk, T.; Biedermann, T.; Bottcher-Haberzeth, S.; Klar, A.S.; Meuli, M.; Reichmann, E. UVB exposure of a humanized skin model reveals unexpected dynamic of keratinocyte proliferation and Wnt inhibitor balancing. J. Tissue Eng. Regen. Med. 2018, 12, 505–515. [Google Scholar] [CrossRef]
- Schneider, J.; Biedermann, T.; Widmer, D.; Montano, I.; Meuli, M.; Reichmann, E.; Schiestl, C. Matriderm versus Integra: A comparative experimental study. Burns 2009, 35, 51–57. [Google Scholar] [CrossRef]
- Krafft, C.; Neudert, L.; Simat, T.; Salzer, R. Near infrared Raman spectra of human brain lipids. Spectrochim. Acta A Mol. Biomol. Spectrosc. 2005, 61, 1529–1535. [Google Scholar] [CrossRef]
- Talari, A.C.S.; Movasaghi, Z.; Rehman, S.; Rehman, I.u. Raman Spectroscopy of Biological Tissues. Appl. Spectrosc. Rev. 2015, 50, 46–111. [Google Scholar] [CrossRef]
- Czamara, K.; Majzner, K.; Pacia, M.Z.; Kochan, K.; Kaczor, A.; Baranska, M. Raman spectroscopy of lipids: A review. J. Raman Spectrosc. 2015, 46, 4–20. [Google Scholar] [CrossRef]
- Noske, K.; Stark, H.J.; Nevaril, L.; Berning, M.; Langbein, L.; Goyal, A.; Diederichs, S.; Boukamp, P. Mitotic Diversity in Homeostatic Human Interfollicular Epidermis. Int. J. Mol. Sci. 2016, 17, 167. [Google Scholar] [CrossRef] [PubMed]
- Dunaway, S.; Rothaus, A.; Zhang, Y.; Luisa Kadekaro, A.; Andl, T.; Andl, C.D. Divide and conquer: Two stem cell populations in squamous epithelia, reserves and the active duty forces. Int. J. Oral Sci. 2019, 11, 26. [Google Scholar] [CrossRef]
- Kaur, P.; Li, A. Adhesive properties of human basal epidermal cells: An analysis of keratinocyte stem cells, transit amplifying cells, and postmitotic differentiating cells. J. Investig. Dermatol. 2000, 114, 413–420. [Google Scholar] [CrossRef]
- Levy, V.; Lindon, C.; Zheng, Y.; Harfe, B.D.; Morgan, B.A. Epidermal stem cells arise from the hair follicle after wounding. FASEB J. 2007, 21, 1358–1366. [Google Scholar] [CrossRef]
- Krebsbach, P.H.; Villa-Diaz, L.G. The Role of Integrin α6 (CD49f) in Stem Cells: More than a Conserved Biomarker. Stem Cells Dev. 2017, 26, 1090–1099. [Google Scholar] [CrossRef]
- Cariati, M.; Naderi, A.; Brown, J.P.; Smalley, M.J.; Pinder, S.E.; Caldas, C.; Purushotham, A.D. Alpha-6 integrin is necessary for the tumourigenicity of a stem cell-like subpopulation within the MCF7 breast cancer cell line. Int. J. Cancer 2008, 122, 298–304. [Google Scholar] [CrossRef]
- Lathia, J.D.; Gallagher, J.; Heddleston, J.M.; Wang, J.; Eyler, C.E.; Macswords, J.; Wu, Q.; Vasanji, A.; McLendon, R.E.; Hjelmeland, A.B.; et al. Integrin alpha 6 regulates glioblastoma stem cells. Cell Stem Cell 2010, 6, 421–432. [Google Scholar] [CrossRef]
- Pontier, S.M.; Muller, W.J. Integrins in mammary-stem-cell biology and breast-cancer progression—A role in cancer stem cells? J. Cell Sci. 2009, 122, 207–214. [Google Scholar] [CrossRef][Green Version]
- Wilschut, K.J.; van Tol, H.T.; Arkesteijn, G.J.; Haagsman, H.P.; Roelen, B.A. Alpha 6 integrin is important for myogenic stem cell differentiation. Stem Cell Res. 2011, 7, 112–123. [Google Scholar] [CrossRef] [PubMed]
- Li, A.; Simmons, P.J.; Kaur, P. Identification and isolation of candidate human keratinocyte stem cells based on cell surface phenotype. Proc. Natl. Acad. Sci. USA 1998, 95, 3902–3907. [Google Scholar] [CrossRef] [PubMed]
- Nanba, D.; Toki, F.; Tate, S.; Imai, M.; Matsushita, N.; Shiraishi, K.; Sayama, K.; Toki, H.; Higashiyama, S.; Barrandon, Y. Cell motion predicts human epidermal stemness. J. Cell Biol. 2015, 209, 305–315. [Google Scholar] [CrossRef]
- Schluter, H.; Paquet-Fifield, S.; Gangatirkar, P.; Li, J.; Kaur, P. Functional characterization of quiescent keratinocyte stem cells and their progeny reveals a hierarchical organization in human skin epidermis. Stem Cells 2011, 29, 1256–1268. [Google Scholar] [CrossRef]
- Wang, S.; Drummond, M.L.; Guerrero-Juarez, C.F.; Tarapore, E.; MacLean, A.L.; Stabell, A.R.; Wu, S.C.; Gutierrez, G.; That, B.T.; Benavente, C.A.; et al. Single cell transcriptomics of human epidermis identifies basal stem cell transition states. Nat. Commun. 2020, 11, 4239. [Google Scholar] [CrossRef] [PubMed]
- Fu, X.; Sun, X.; Li, X.; Sheng, Z. Dedifferentiation of epidermal cells to stem cells in vivo. Lancet 2001, 358, 1067–1068. [Google Scholar] [CrossRef]
- Mannik, J.; Alzayady, K.; Ghazizadeh, S. Regeneration of multilineage skin epithelia by differentiated keratinocytes. J. Investig. Dermatol. 2010, 130, 388–397. [Google Scholar] [CrossRef]
- Rompolas, P.; Mesa, K.R.; Greco, V. Spatial organization within a niche as a determinant of stem-cell fate. Nature 2013, 502, 513–518. [Google Scholar] [CrossRef]
- Chacón-Martínez, C.A.; Klose, M.; Niemann, C.; Glauche, I.; Wickström, S.A. Hair follicle stem cell cultures reveal self-organizing plasticity of stem cells and their progeny. EMBO J. 2017, 36, 151–164. [Google Scholar] [CrossRef]
- Haensel, D.; Jin, S.; Sun, P.; Cinco, R.; Dragan, M.; Nguyen, Q.; Cang, Z.; Gong, Y.; Vu, R.; MacLean, A.L.; et al. Defining Epidermal Basal Cell States during Skin Homeostasis and Wound Healing Using Single-Cell Transcriptomics. Cell Rep. 2020, 30, 3932–3947.e3936. [Google Scholar] [CrossRef]
- Wang, Y.Z.; Plane, J.M.; Jiang, P.; Zhou, C.J.; Deng, W. Concise review: Quiescent and active states of endogenous adult neural stem cells: Identification and characterization. Stem Cells 2011, 29, 907–912. [Google Scholar] [CrossRef] [PubMed]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pontiggia, L.; Ahuja, A.K.; Yosef, H.K.; Rütsche, D.; Reichmann, E.; Moehrlen, U.; Biedermann, T. Human Basal and Suprabasal Keratinocytes Are Both Able to Generate and Maintain Dermo–Epidermal Skin Substitutes in Long-Term In Vivo Experiments. Cells 2022, 11, 2156. https://doi.org/10.3390/cells11142156
Pontiggia L, Ahuja AK, Yosef HK, Rütsche D, Reichmann E, Moehrlen U, Biedermann T. Human Basal and Suprabasal Keratinocytes Are Both Able to Generate and Maintain Dermo–Epidermal Skin Substitutes in Long-Term In Vivo Experiments. Cells. 2022; 11(14):2156. https://doi.org/10.3390/cells11142156
Chicago/Turabian StylePontiggia, Luca, Akshay Kumar Ahuja, Hesham Kamaleldin Yosef, Dominic Rütsche, Ernst Reichmann, Ueli Moehrlen, and Thomas Biedermann. 2022. "Human Basal and Suprabasal Keratinocytes Are Both Able to Generate and Maintain Dermo–Epidermal Skin Substitutes in Long-Term In Vivo Experiments" Cells 11, no. 14: 2156. https://doi.org/10.3390/cells11142156
APA StylePontiggia, L., Ahuja, A. K., Yosef, H. K., Rütsche, D., Reichmann, E., Moehrlen, U., & Biedermann, T. (2022). Human Basal and Suprabasal Keratinocytes Are Both Able to Generate and Maintain Dermo–Epidermal Skin Substitutes in Long-Term In Vivo Experiments. Cells, 11(14), 2156. https://doi.org/10.3390/cells11142156






