AMPK Modulates the Metabolic Adaptation of C6 Glioma Cells in Glucose-Deprived Conditions without Affecting Glutamate Transport
Abstract
:1. Introduction
2. Materials and Methods
2.1. Generation of the Inducible AMPK-DN System
2.2. Cell Transfection and Selection
2.3. Glucose Deprivation Protocol
2.4. Total RNA Extraction and Real-Time Quantitative PCR (RT-qPCR)
2.5. Western Blot
2.6. d-[3H]-Aspartate Uptake
2.7. MTT Assay
2.8. ATP Assay
2.9. XF Real-Time ATP Rate Assay
2.10. Migration Assay
2.11. Statistics
3. Results
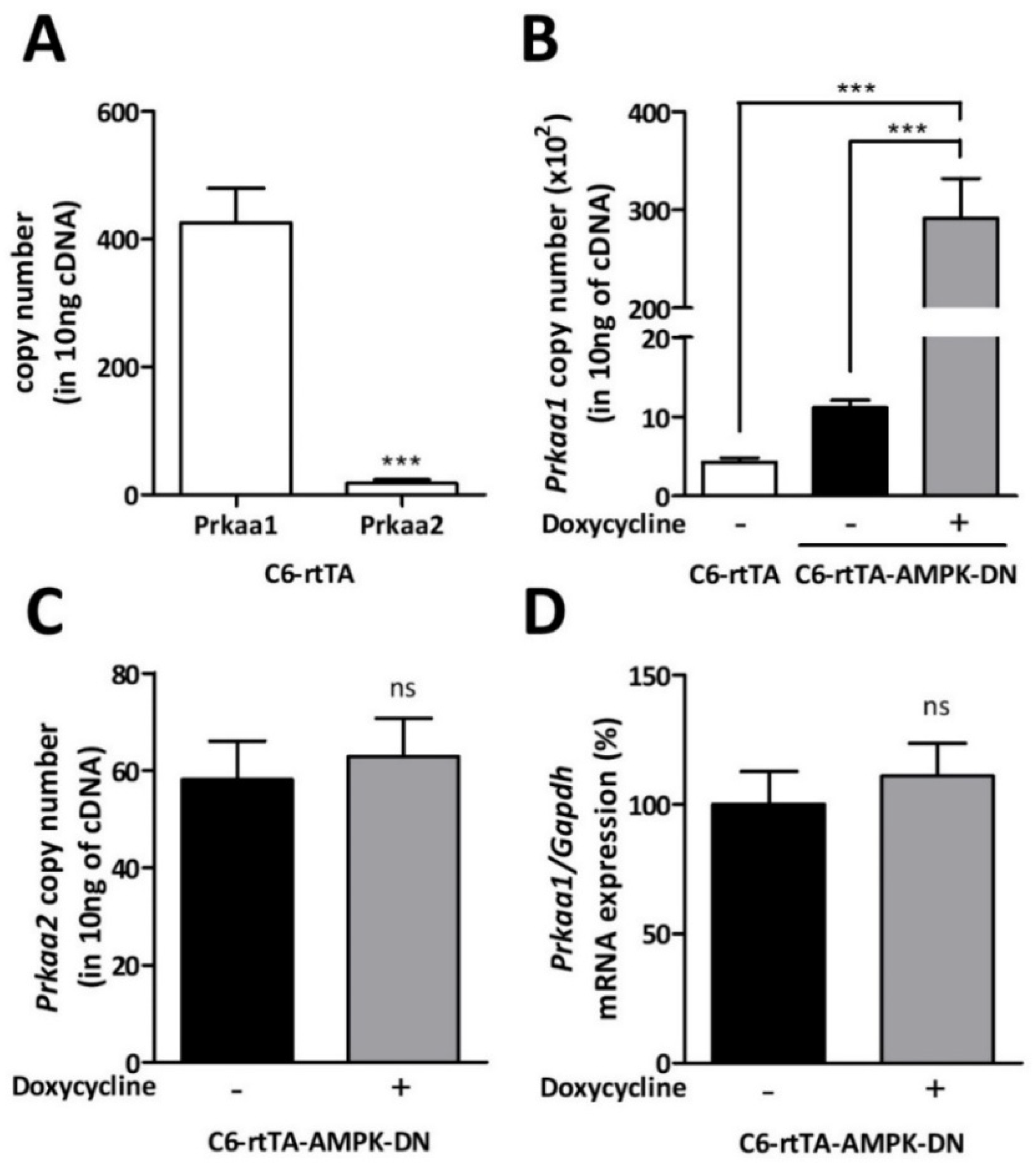
3.1. Expression of an AMPK Dominant Negative Mutant in C6 Cells
3.2. Validation of the Doxycycline-Inducible AMPK-DN System
3.3. AMPK Silencing Limits the Metabolic Adaptation to Glucose Deprivation in C6 Cells
3.4. AMPK Silencing Impairs Cell Migration
3.5. AMPK Silencing Does Not Alter Glutamate Transport
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Bélanger, M.; Allaman, I.; Magistretti, P.J. Brain Energy Metabolism: Focus on Astrocyte-Neuron Metabolic Cooperation. Cell Metab. 2011, 14, 724–738. [Google Scholar] [CrossRef] [Green Version]
- Stobart, J.L.; Anderson, C.M. Multifunctional Role of Astrocytes as Gatekeepers of Neuronal Energy Supply. Front. Cell. Neurosci. 2013, 7, 1–21. [Google Scholar] [CrossRef] [Green Version]
- Almeida, A.; Moncada, S.; Bolaños, J.P. Nitric Oxide Switches on Glycolysis through the AMP Protein Kinase and 6-Phosphofructo-2-Kinase Pathway. Nat. Cell. Biol. 2004, 6, 45–51. [Google Scholar] [CrossRef]
- Sang, W.S.; Bergher, J.P.; Anderson, C.M.; Treadway, J.L.; Fosgerau, K.; Swanson, R.A. Astrocyte Glycogen Sustains Neuronal Activity during Hypoglycemia: Studies with the Glycogen Phosphorylase Inhibitor CP-316,819 ([R-R*,S*]-5- Chloro-N-[2-Hydroxy-3-(Methoxymethylamino)-3-Oxo-1-(Phenylmethyl)Propyl] -1H-Indole-2-Carboxamide). J. Pharmacol. Exp. Ther. 2007, 321, 45–50. [Google Scholar] [CrossRef] [Green Version]
- Auestad, N.; Korsak, R.A.; Morrow, J.W.; Edmond, J. Fatty Acid Oxidation and Ketogenesis by Astrocytes in Primary Culture. J. Neurochem. 1991, 56, 1376–1386. [Google Scholar] [CrossRef]
- Gorina, Y.V.; Salmina, A.B.; Erofeev, A.I.; Can, Z.; Bolshakova, A.V.; Balaban, P.M.; Bezprozvanny, I.B.; Vlasova, O.L. Metabolic Plasticity of Astrocytes. J. Evol. Biochem. Physiol. 2021, 57, 1207–1224. [Google Scholar] [CrossRef]
- Anderson, C.M.; Swanson, R.A. Astrocyte Glutamate Transport: Review of Properties, Regulation, and Physiological Functions. Glia 2000, 32, 1–14. [Google Scholar] [CrossRef]
- Danbolt, N.C. Glutamate Uptake. Prog. Neurobiol. 2001, 65, 1–105. [Google Scholar] [CrossRef]
- Magistretti, P.J.; Chatton, J.Y. Relationship between L-Glutamate-Regulated Intracellular Na+ Dynamics and ATP Hydrolysis in Astrocytes. J. Neural Transm. 2005, 112, 77–85. [Google Scholar] [CrossRef]
- Bak, L.K.; Schousboe, A.; Waagepetersen, H.S. The Glutamate/GABA-Glutamine Cycle: Aspects of Transport, Neurotransmitter Homeostasis and Ammonia Transfer. J. Neurochem. 2006, 98, 641–653. [Google Scholar] [CrossRef]
- McKenna, M.C. The Glutamate-Glutamine Cycle Is Not Stoichiometric: Fates of Glutamate in Brain Mary. J. Neurosci. Res. 2007, 85, 3347–3358. [Google Scholar] [CrossRef]
- Andersen, J.V.; Markussen, K.H.; Jakobsen, E.; Schousboe, A.; Waagepetersen, H.S.; Rosenberg, P.A.; Aldana, B.I. Glutamate Metabolism and Recycling at the Excitatory Synapse in Health and Neurodegeneration. Neuropharmacology 2021, 196, 108719. [Google Scholar] [CrossRef]
- McKenna, M.C. Glutamate Pays Its Own Way in Astrocytes. Front. Endocrinol. 2013, 4, 1–6. [Google Scholar] [CrossRef] [Green Version]
- Pellerin, L.; Magistretti, P.J. Glutamate Uptake into Astrocytes Stimulates Aerobic Glycolysis: A Mechanism Coupling Neuronal Activity to Glucose Utilization. Proc. Natl. Acad. Sci. USA 1994, 91, 10625–10629. [Google Scholar] [CrossRef] [Green Version]
- Pellerin, L.; Magistretti, P.J. Sweet Sixteen for ANLS. J. Cereb. Blood Flow Metab. 2012, 32, 1152–1166. [Google Scholar] [CrossRef]
- Robinson, M.B.; Jackson, J.G. Astroglial Glutamate Transporters Coordinate Excitatory Signaling and Brain Energetics. Neurochem. Int. 2016, 98, 56–71. [Google Scholar] [CrossRef] [Green Version]
- Davies, S.P.; Hawley, S.A.; Woods, A.; Carling, D.; Haystead, T.A.J.; Hardie, D.G. Purification of the AMP-activated Protein Kinase on ATP-γ-Sepharose and Analysis of Its Subunit Structure. Eur. J. Biochem. 1994, 223, 351–357. [Google Scholar] [CrossRef]
- Hardie, D.G.; Carling, D. The AMP-Activated Protein Kinase. Fuel Gauge of the Mammalian Cell? Eur. J. Biochem. 1997, 246, 259–273. [Google Scholar] [CrossRef]
- Stapleton, D.; Woollatt, E.; Mitchelhill, K.I.; Nicholl, J.K.; Fernandez, C.S.; Michell, B.J.; Witters, L.A.; Power, D.A.; Sutherland, G.R.; Kemp, B.E. AMP-Activated Protein Kinase Isoenzyme Family: Subunit Structure and Chromosomal Location. FEBS Lett. 1997, 409, 452–456. [Google Scholar] [CrossRef] [Green Version]
- Thornton, C.; Snowden, M.A.; Carling, D. Identification of a Novel AMP-Activated Protein Kinase β Subunit Isoform That Is Highly Expressed in Skeletal Muscle. J. Biol. Chem. 1998, 273, 12443–12450. [Google Scholar] [CrossRef] [Green Version]
- Cheung, P.C.F.; Salt, I.P.; Davies, S.P.; Hardie, D.G.; Carling, D. Characterization of AMP-Activated Protein Kinase γ-Subunit Isoforms and Their Role in AMP Binding. Biochem. J. 2000, 346, 659–669. [Google Scholar] [CrossRef]
- Ross, F.A.; MacKintosh, C.; Hardie, D.G. AMP-Activated Protein Kinase: A Cellular Energy Sensor That Comes in 12 Flavours. FEBS J. 2016, 283, 2987–3001. [Google Scholar] [CrossRef]
- Hardie, D.G. AMP-Activated Protein Kinase-an Energy Sensor That Regulates All Aspects of Cell Function. Genes Dev. 2011, 25, 1895–1908. [Google Scholar] [CrossRef] [Green Version]
- Hardie, D.G.; Ross, F.A.; Hawley, S.A. AMPK: A Nutrient and Energy Sensor That Maintains Energy Homeostasis. Nat. Rev. Mol. Cell Biol. 2012, 13, 251–262. [Google Scholar] [CrossRef] [Green Version]
- Vara-Ciruelos, D.; Dandapani, M.; Hardie, D.G. AMP-Activated Protein Kinase: Friend or Foe in Cancer? Annu. Rev. Cancer Biol. 2020, 4, 1–16. [Google Scholar] [CrossRef] [Green Version]
- Cox, S.E.; Pearce, B.; Michael, R. AMP-Activated Protein Kinase in Astrocytes. Biochem. Soc. Trans. 1997, 25, S583. [Google Scholar] [CrossRef] [Green Version]
- Turnley, A.M.; Stapleton, D.; Mann, R.J.; Witters, L.A.; Kemp, B.E.; Bartlett, P.F. Cellular Distribution and Developmental Expression of AMP-Activated Protein Kinase Isoforms in Mouse Central Nervous System. J. Neurochem. 1999, 72, 1707–1716. [Google Scholar] [CrossRef]
- Gabryel, B.; Kost, A.; Kasprowska, D.; Liber, S.; Machnik, G.; Wiaderkiewicz, R.; Labuzek, K. AMP-Activated Protein Kinase Is Involved in Induction of Protective Autophagy in Astrocytes Exposed to Oxygen-Glucose Deprivation. Cell Biol. Int. 2014, 38, 1086–1097. [Google Scholar] [CrossRef]
- Voss, C.M.; Andersen, J.V.; Jakobsen, E.; Siamka, O.; Karaca, M.; Maechler, P.; Waagepetersen, H.S. AMP-Activated Protein Kinase (AMPK) Regulates Astrocyte Oxidative Metabolism by Balancing TCA Cycle Dynamics. Glia 2020, 68, 1824–1839. [Google Scholar] [CrossRef]
- Jakobsen, E.; Andersen, J.V.; Christensen, S.K.; Siamka, O.; Larsen, M.R.; Waagepetersen, H.S.; Aldana, B.I.; Bak, L.K. Pharmacological Inhibition of Mitochondrial Soluble Adenylyl Cyclase in Astrocytes Causes Activation of AMP-Activated Protein Kinase and Induces Breakdown of Glycogen. Glia 2021, 69, 2828–2844. [Google Scholar] [CrossRef]
- Peixoto, C.A.; de Oliveira, W.H.; da Racho Araújo, S.M.; Nunes, A.K.S. AMPK Activation: Role in the Signaling Pathways of Neuroinflammation and Neurodegeneration. Exp. Neurol. 2017, 298, 31–41. [Google Scholar] [CrossRef]
- Domise, M. Chapter 7: AMPK in Neurodegenerative Diseases. In AMP-activated Protein Kinase; Springer: Cham, Switzerland, 2016; pp. 153–177. [Google Scholar] [CrossRef] [Green Version]
- Muraleedharan, R.; Gawali, M.V.; Tiwari, D.; Sukumaran, A.; Oatman, N.; Anderson, J.; Nardini, D.; Bhuiyan, M.A.N.; Tkáč, I.; Ward, A.L.; et al. AMPK-Regulated Astrocytic Lactate Shuttle Plays a Non-Cell-Autonomous Role in Neuronal Survival. Cell Rep. 2020, 32, 108092. [Google Scholar] [CrossRef]
- Muraleedharan, R.; Dasgupta, B. AMPK in the Brain: Its Roles in Glucose and Neural Metabolism. FEBS J. 2021, 289, 1–16. [Google Scholar] [CrossRef]
- Benda, P.; Lighbody, J.; Sato, G.; Levine, L.; Sweet, W. Differentiated Rat Glial Cell Strain in Tissue Culture. Science 1968, 161, 370–371. [Google Scholar] [CrossRef]
- Takanaga, H.; Yoshitake, T.; Hara, S.; Yamasaki, C.; Kunimoto, M. CAMP-Induced Astrocytic Differentiation of C6 Glioma Cells Is Mediated by Autocrine Interleukin-6. J. Biol. Chem. 2004, 279, 15441–15447. [Google Scholar] [CrossRef] [Green Version]
- Park, S.H.; Lee, J.Y.; Jhee, K.H.; Yang, S.A. Amyloid-ß Peptides Inhibit the Expression of AQP4 and Glutamate Transporter EAAC1 in Insulin-Treated C6 Glioma Cells. Toxicol. Rep. 2020, 7, 1083–1089. [Google Scholar] [CrossRef]
- Regan, J.T.; Mirczuk, S.M.; Scudder, C.J.; Stacey, E.; Khan, S.; Worwood, M.; Powles, T.; Dennis-Beron, J.S.; Ginley-Hidinger, M.; McGonnell, I.M.; et al. Sensitivity of the Natriuretic Peptide/Cgmp System to Hyperammonaemia in Rat C6 Glioma Cells and Gpnt Brain Endothelial Cells. Cells 2021, 10, 398. [Google Scholar] [CrossRef]
- Stein, S.C.; Woods, A.; Jones, N.A.; Davison, M.D.; Cabling, D. The Regulation of AMP-Activated Protein Kinase by Phosphorylation. Biochem. J. 2000, 345, 437–443. [Google Scholar] [CrossRef]
- Vanhoutte, N.; Hermans, E. Glutamate-Induced Glioma Cell Proliferation Is Prevented by Functional Expression of the Glutamate Transporter GLT-1. FEBS Lett. 2008, 582, 1847–1852. [Google Scholar] [CrossRef] [Green Version]
- Graham, F.L.; Van der Eb, A. A New Technique for the Assay Adenovirus of Infectivity of Human Adenovirus 5 DNA. Virology 1973, 467, 456–467. [Google Scholar] [CrossRef]
- Jonkman, J.E.N.; Cathcart, J.A.; Xu, F.; Bartolini, M.E.; Amon, J.E.; Stevens, K.M.; Colarusso, P. Cell Adhesion & Migration An Introduction to the Wound Healing Assay Using Livecell Microscopy. Cell Adhes. Migr. 2014, 8, 440–451. [Google Scholar] [CrossRef] [Green Version]
- Gao, M.; Liang, J.; Lu, Y.; Guo, H.; German, P.; Bai, S.; Jonasch, E.; Yang, X.; Mills, G.B.; Ding, Z. Site-Specific Activation of AKT Protects Cells from Death Induced by Glucose Deprivation. Oncogene 2014, 33, 745–755. [Google Scholar] [CrossRef] [Green Version]
- Huet, C.; Boudaba, N.; Guigas, B.; Viollet, B.; Foretz, M.; Pessin, J.E. Glucose Availability but Not Changes in Pancreatic Hormones Sensitizes Hepatic AMPK Activity during Nutritional Transition in Rodents. J. Biol. Chem. 2020, 295, 5836–5849. [Google Scholar] [CrossRef] [Green Version]
- Stockert, J.C.; Horobin, R.W.; Colombo, L.L.; Blázquez-Castro, A. Tetrazolium Salts and Formazan Products in Cell Biology: Viability Assessment, Fluorescence Imaging, and Labeling Perspectives. Acta Histochem. 2018, 120, 159–167. [Google Scholar] [CrossRef] [Green Version]
- Bernardinelli, Y.; Magistretti, P.J.; Chatton, J.Y. Astrocytes Generate Na+-Mediated Metabolic Waves. Proc. Natl. Acad. Sci. USA 2004, 101, 14937–14942. [Google Scholar] [CrossRef] [Green Version]
- Dasgupta, B.; Chhipa, R.R. Evolving Lessons on the Complex Role of AMPK in Normal Physiology and Cancer. Trends Pharmacol. Sci. 2016, 37, 192–206. [Google Scholar] [CrossRef] [Green Version]
- Woods, A.; Azzout-Marniche, D.; Foretz, M.; Stein, S.C.; Lemarchand, P.; Ferré, P.; Foufelle, F.; Carling, D. Characterization of the Role of AMP-Activated Protein Kinase in the Regulation of Glucose-Activated Gene Expression Using Constitutively Active and Dominant Negative Forms of the Kinase. Mol. Cell. Biol. 2000, 20, 6704–6711. [Google Scholar] [CrossRef] [Green Version]
- Galland, F.; Seady, M.; Taday, J.; Smaili, S.S.; Gonçalves, C.A.; Leite, M.C. Astrocyte Culture Models: Molecular and Function Characterization of Primary Culture, Immortalized Astrocytes and C6 Glioma Cells. Neurochem. Int. 2019, 131, 104538. [Google Scholar] [CrossRef]
- Turner, D.A.; Adamson, D.C. Neuronal-Astrocyte Metabolic Interactions: Understanding the Transition into Abnormal Astrocytoma Metabolism. J. Neuropathol. Exp. Neurol. 2011, 70, 167–176. [Google Scholar] [CrossRef]
- Goldberg, M.P.; Choi, D.W. Combined Oxygen and Glucose Deprivation Calcium-Dependent and Calcium-Independent Neuronal Injury in Cortical Cell Culture: Calcium-Dependent and Calcium-Independent Mechanisms of Neuronal Injury. J. Neurosci. 1993, 13, 3510–3524. [Google Scholar] [CrossRef]
- Herrero-Mendez, A.; Almeida, A.; Fernández, E.; Maestre, C.; Moncada, S.; Bolaños, J.P. The Bioenergetic and Antioxidant Status of Neurons Is Controlled by Continuous Degradation of a Key Glycolytic Enzyme by APC/C-Cdh1. Nat. Cell. Biol. 2009, 11, 747–752. [Google Scholar] [CrossRef]
- Bittner, C.X. High Resolution Measurement of the Glycolytic Rate. Front. Neuroenergetics 2010, 2, 1–11. [Google Scholar] [CrossRef] [Green Version]
- Chhipa, R.R.; Fan, Q.; Anderson, J.; Muraleedharan, R.; Ciraolo, G.; Chen, X.; Waclaw, R.; Chow, L.M.; Kofron, M.; Weirauch, M.T.; et al. AMP Kinase Promotes Glioblastoma Bioenergetics and Tumour Growth. Nat. Cell Biol. 2018, 20, 823–835. [Google Scholar] [CrossRef]
- Meares, G.P.; Qin, H.; Liu, Y.; Holdbrooks, A.T.; Benveniste, E.N. AMP-Activated Protein Kinase Restricts IFN-γ Signaling. J. Immunol. 2013, 190, 372–380. [Google Scholar] [CrossRef]
- Ou-Yang, L.; Liu, Y.; Wang, B.Y.; Cao, P.; Zhang, J.J.; Huang, Y.Y.; Shen, Y.; Lyu, J.X. Carnosine Suppresses Oxygen-Glucose Deprivation/ Recovery-Induced Proliferation and Migration of Reactive Astrocytes of Rats in Vitro. Acta Pharmacol. Sin. 2018, 39, 24–34. [Google Scholar] [CrossRef]
- Zhang, Y.; Miao, J.M. Ginkgolide K Promotes Astrocyte Proliferation and Migration after Oxygen-Glucose Deprivation via Inducing Protective Autophagy through the AMPK/MTOR/ULK1 Signaling Pathway. Eur. J. Pharmacol. 2018, 832, 96–103. [Google Scholar] [CrossRef]
- Lang, F.; Föller, M. Regulation of Ion Channels and Transporters by AMP-Activated Kinase (AMPK). Channels 2014, 8, 20–28. [Google Scholar] [CrossRef]
- Sopjani, M.; Alesutan, I.; Dërmaku-Sopjani, M.; Fraser, S.; Kemp, B.E.; Föller, M.; Lang, F. Down-Regulation of Na+-Coupled Glutamate Transporter EAAT3 and EAAT4 by AMP-Activated Protein Kinase. J. Neurochem. 2010, 113, 1426–1435. [Google Scholar] [CrossRef]
- Najimi, M.; Maloteaux, J.M.; Hermans, E. Pertussis Toxin-Sensitive Modulation of Glutamate Transport by Endothelin-1 Type A Receptors in Glioma Cells. Biochim. Biophys. Acta Biomembr. 2005, 1668, 195–202. [Google Scholar] [CrossRef] [Green Version]
- Voss, C.M.; Pajęcka, K.; Stridh, M.H.; Nissen, J.D.; Schousboe, A.; Waagepetersen, H.S. AMPK Activation Affects Glutamate Metabolism in Astrocytes. Neurochem. Res. 2015, 40, 2431–2442. [Google Scholar] [CrossRef]







| Gene | Forward Sequence (5′–3′) | Reverse Sequence (5′–3′) |
|---|---|---|
| Prkaa1 | TTAAACCCACAGAAATCCAAACAC | CTTCGCACACGCAAATAATAGG |
| Endogenous Prkaa1 | ATGCGCAGACTCAGTTCCTG | GTCCAGTCAACTCGTGCTTG |
| Prkaa2 | GTGGTGTTATCCTGTATGCCCTTCT | CTGTTTAAACCATTCATGCTCTCGT |
| Gapdh | GTCTCCTGTGACTTCAACAG | AGTTGTCATTGAGAGCAATGC |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Belo do Nascimento, I.; Verfaillie, M.; Ates, G.; Beckers, P.; Joris, V.; Desmet, N.; Massie, A.; Hermans, E. AMPK Modulates the Metabolic Adaptation of C6 Glioma Cells in Glucose-Deprived Conditions without Affecting Glutamate Transport. Cells 2022, 11, 1800. https://doi.org/10.3390/cells11111800
Belo do Nascimento I, Verfaillie M, Ates G, Beckers P, Joris V, Desmet N, Massie A, Hermans E. AMPK Modulates the Metabolic Adaptation of C6 Glioma Cells in Glucose-Deprived Conditions without Affecting Glutamate Transport. Cells. 2022; 11(11):1800. https://doi.org/10.3390/cells11111800
Chicago/Turabian StyleBelo do Nascimento, Inês, Marie Verfaillie, Gamze Ates, Pauline Beckers, Virginie Joris, Nathalie Desmet, Ann Massie, and Emmanuel Hermans. 2022. "AMPK Modulates the Metabolic Adaptation of C6 Glioma Cells in Glucose-Deprived Conditions without Affecting Glutamate Transport" Cells 11, no. 11: 1800. https://doi.org/10.3390/cells11111800
APA StyleBelo do Nascimento, I., Verfaillie, M., Ates, G., Beckers, P., Joris, V., Desmet, N., Massie, A., & Hermans, E. (2022). AMPK Modulates the Metabolic Adaptation of C6 Glioma Cells in Glucose-Deprived Conditions without Affecting Glutamate Transport. Cells, 11(11), 1800. https://doi.org/10.3390/cells11111800






