Perspective: Why and How Ubiquitously Distributed, Vascular-Associated, Pluripotent Stem Cells in the Adult Body (vaPS Cells) Are the Next Generation of Medicine
Abstract
1. Introduction
1.1. The Current State of Confusion about the Term Stem Cells in the Literature
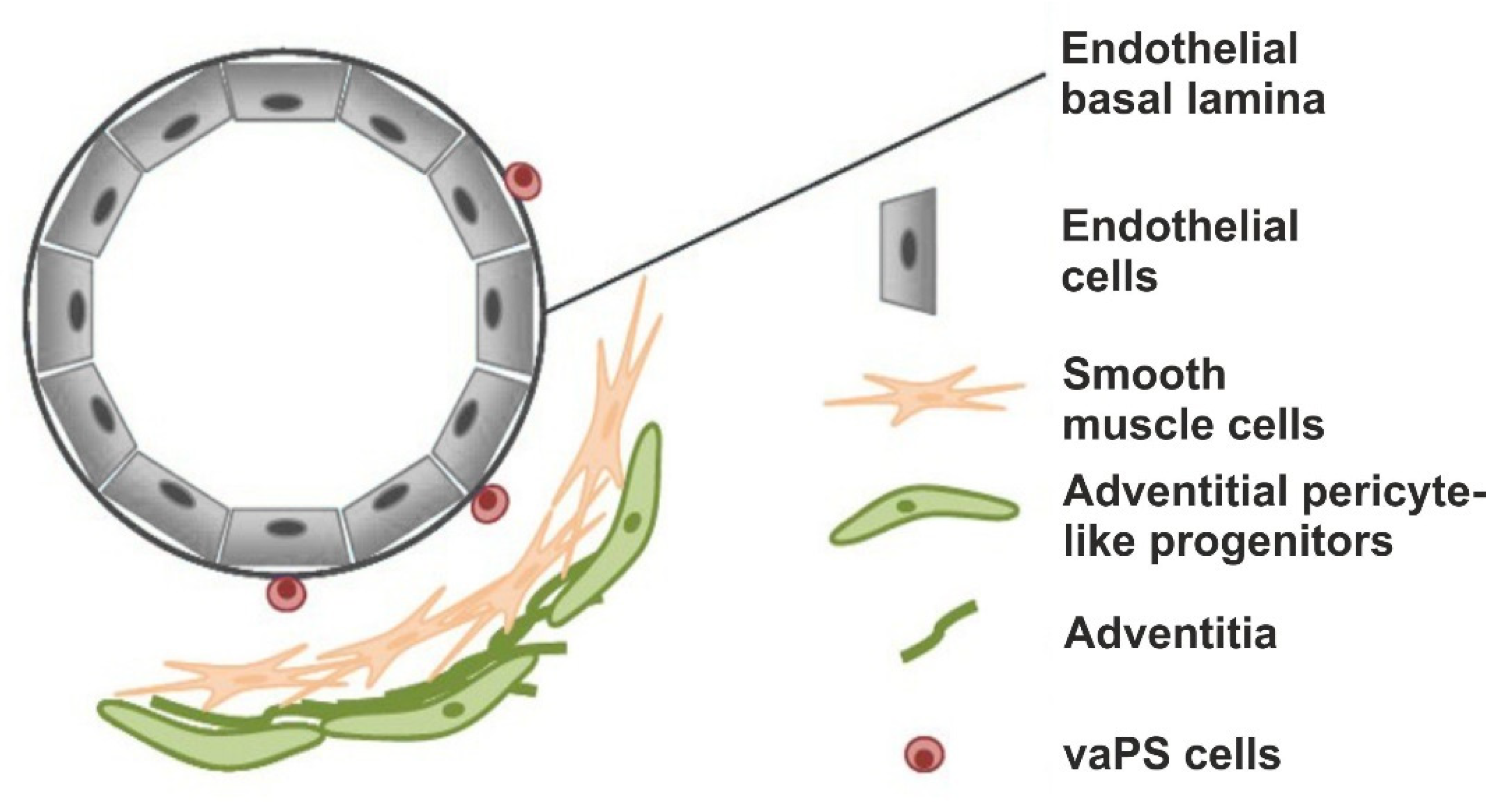
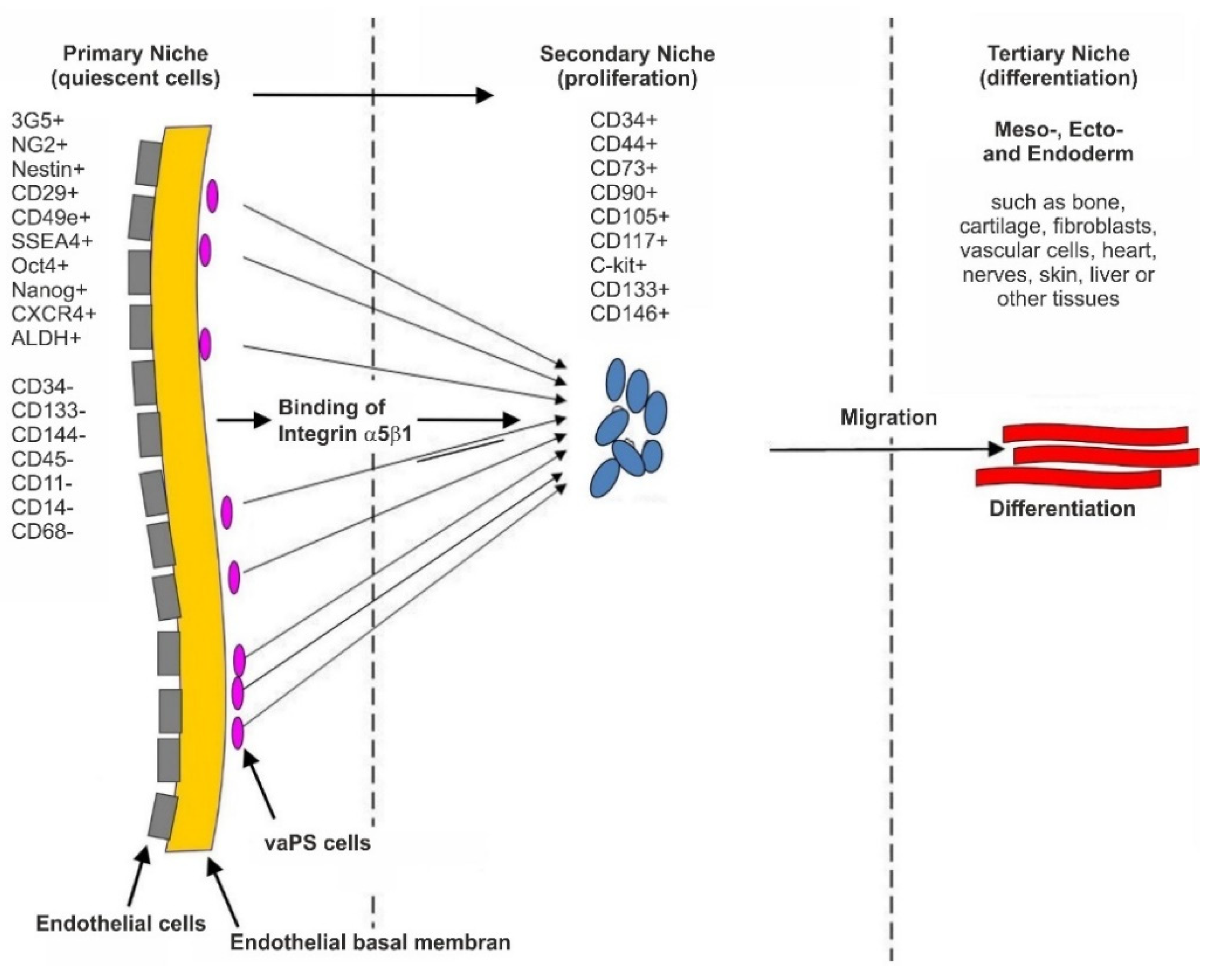
1.2. Introduction of the Term Vascular-Associated, Pluripotent Stem Cells in the Adult Body (vaPS Cells)
1.3. Need for Introducing the Term vaPS Cells
2. The Ubiquitous Occurrence of Stem Cells in Tissue That Contains Blood Vessels
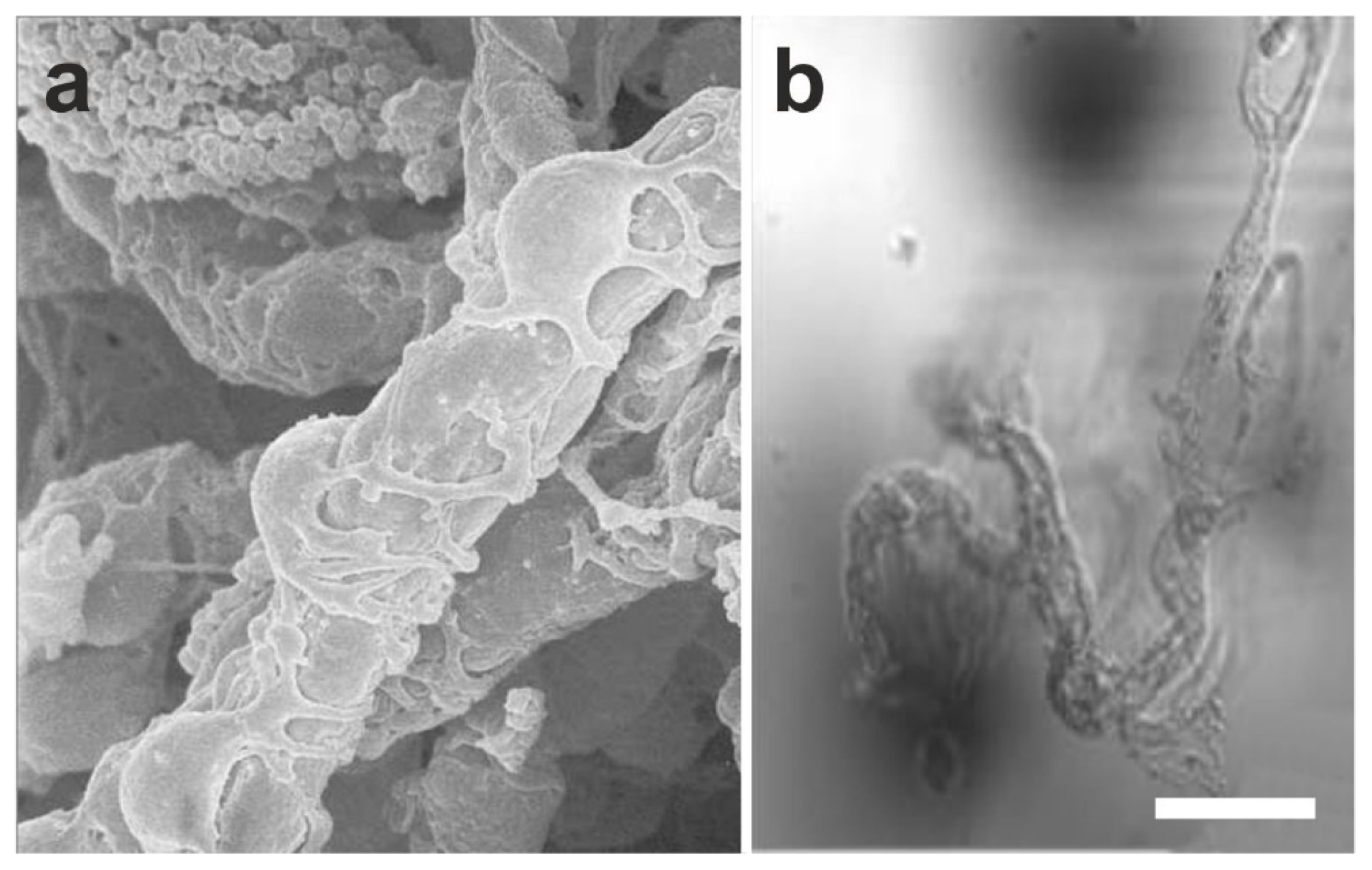
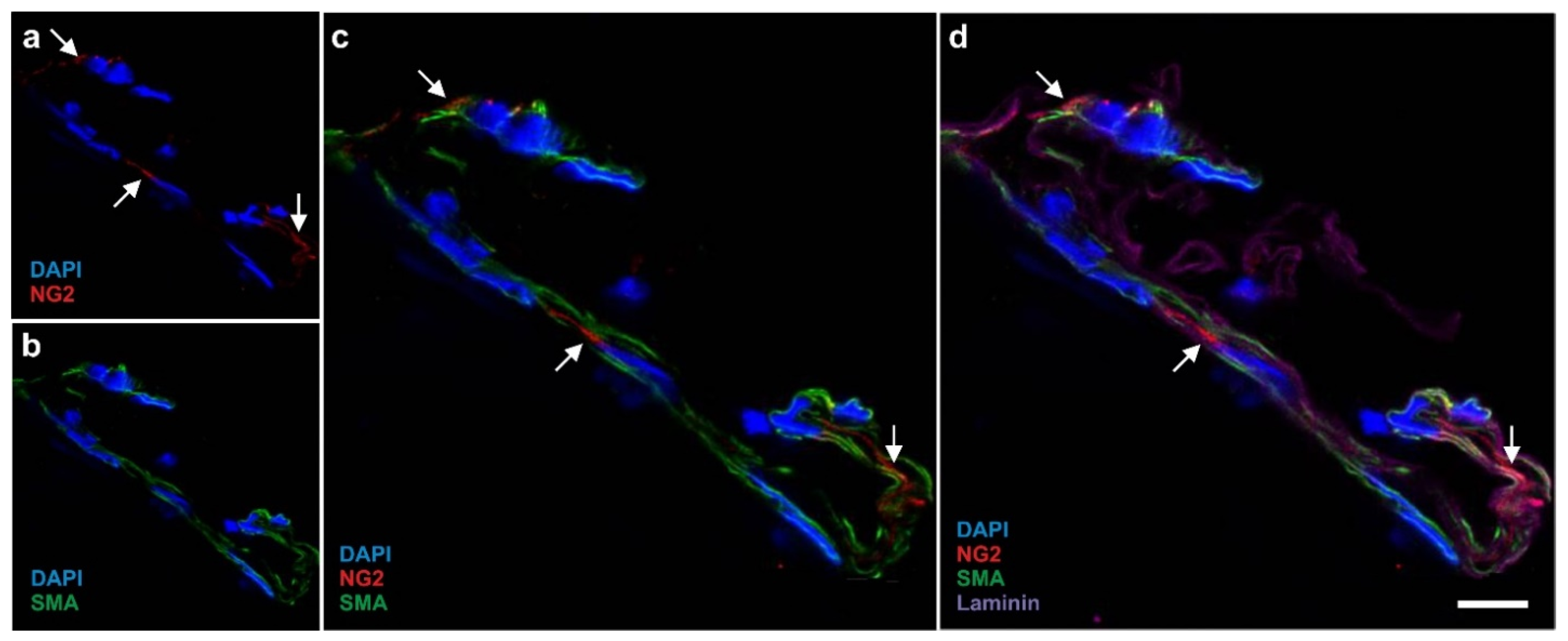
2.1. Existence of Mesenchymal Stem Cells in Blood Vessels

2.2. The Hypothesis of a Universal, Vascular-Associated Stem Cell
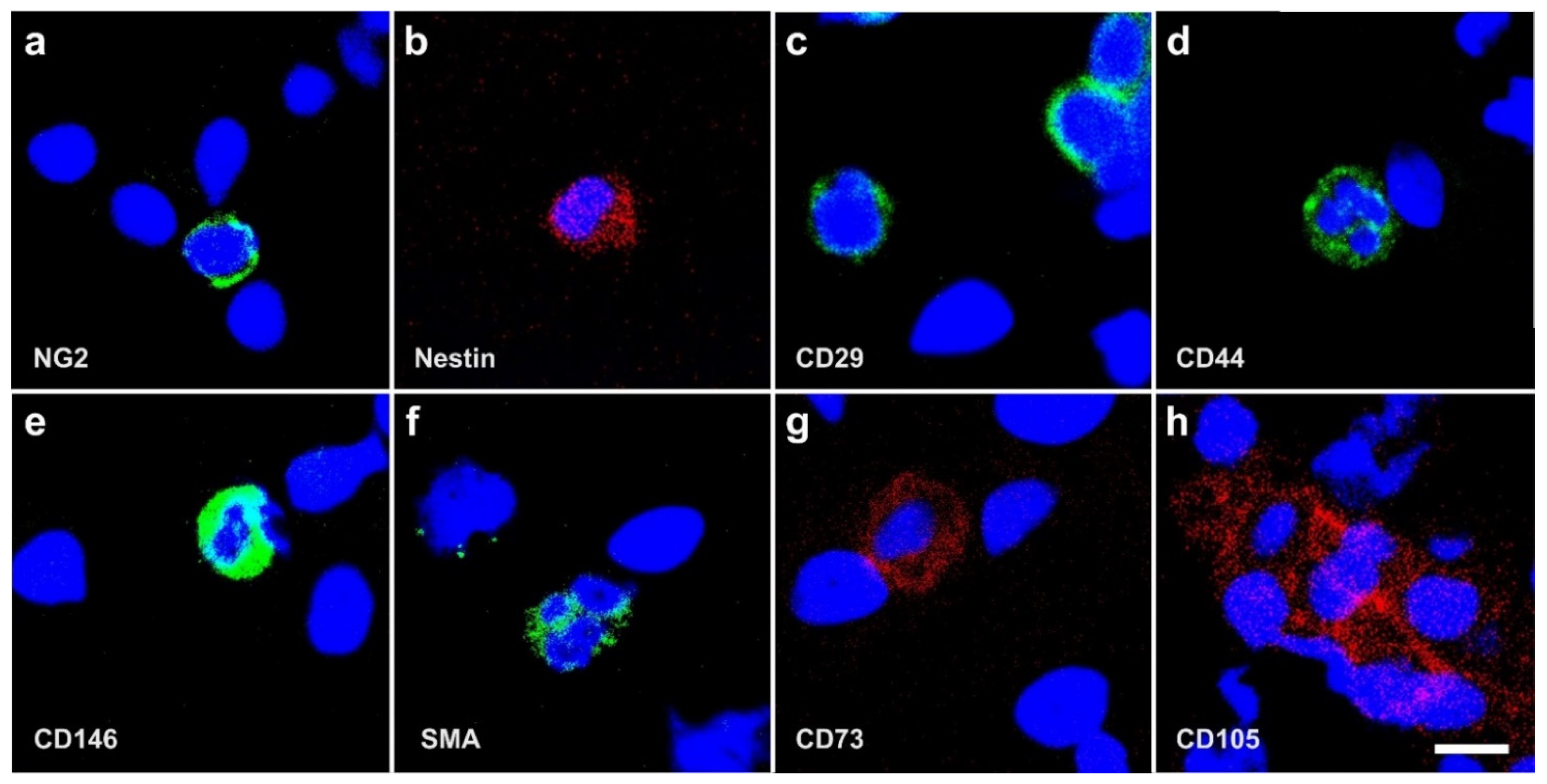
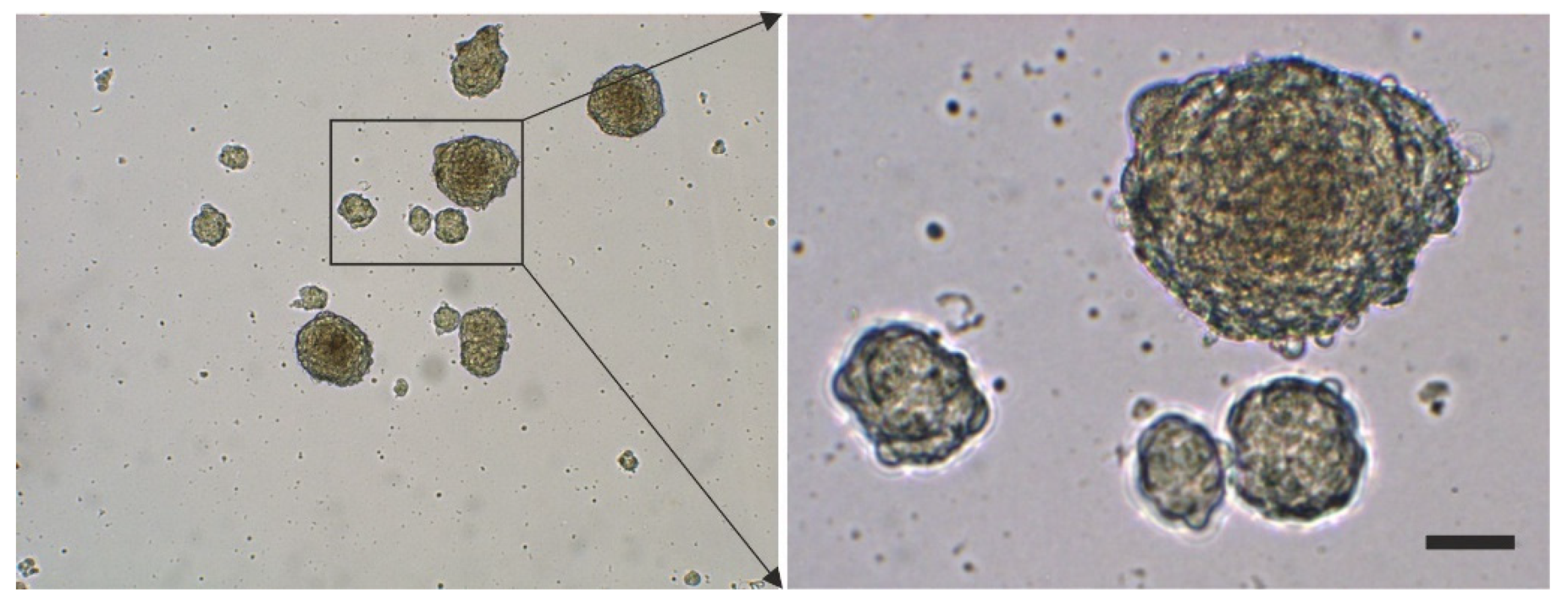
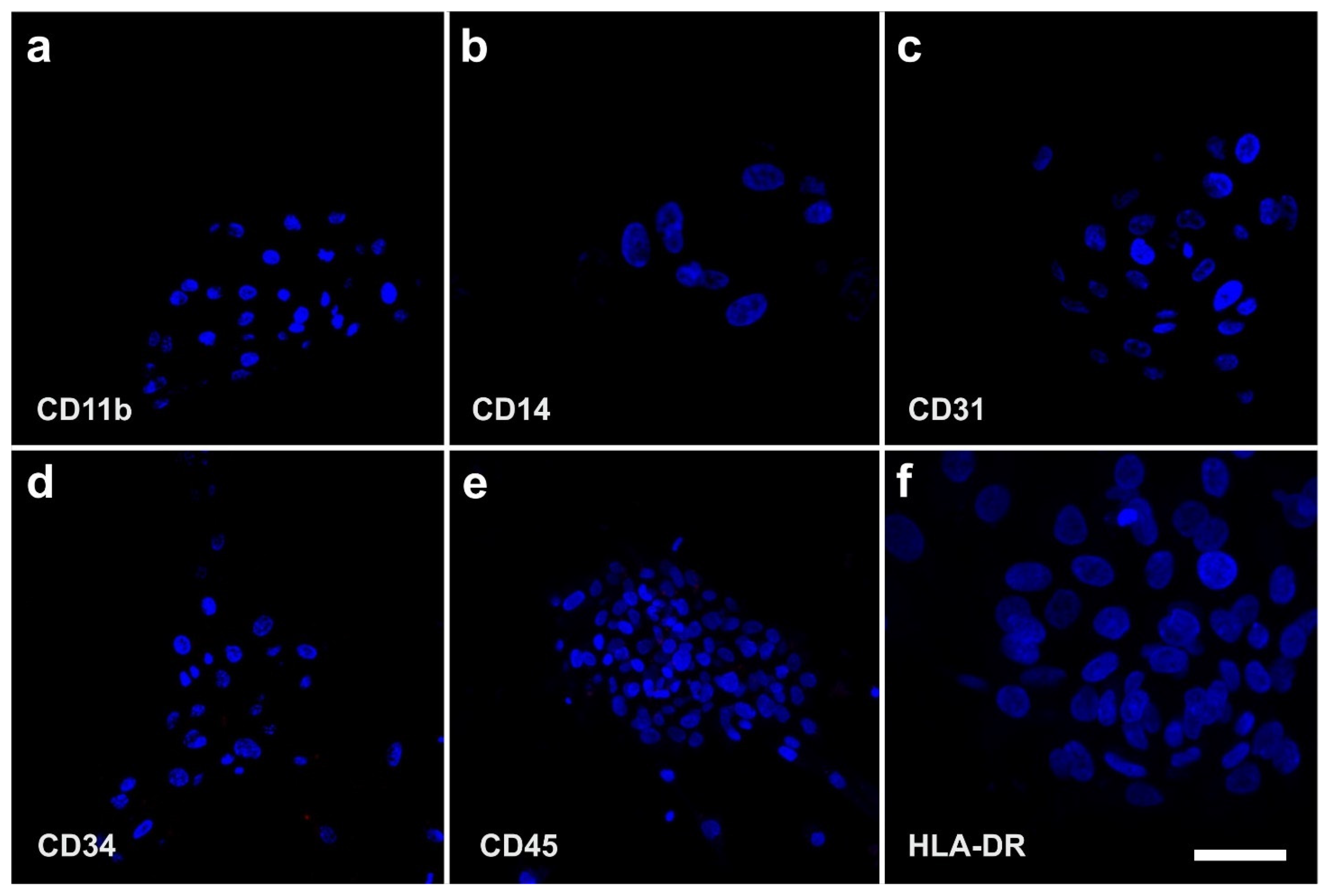
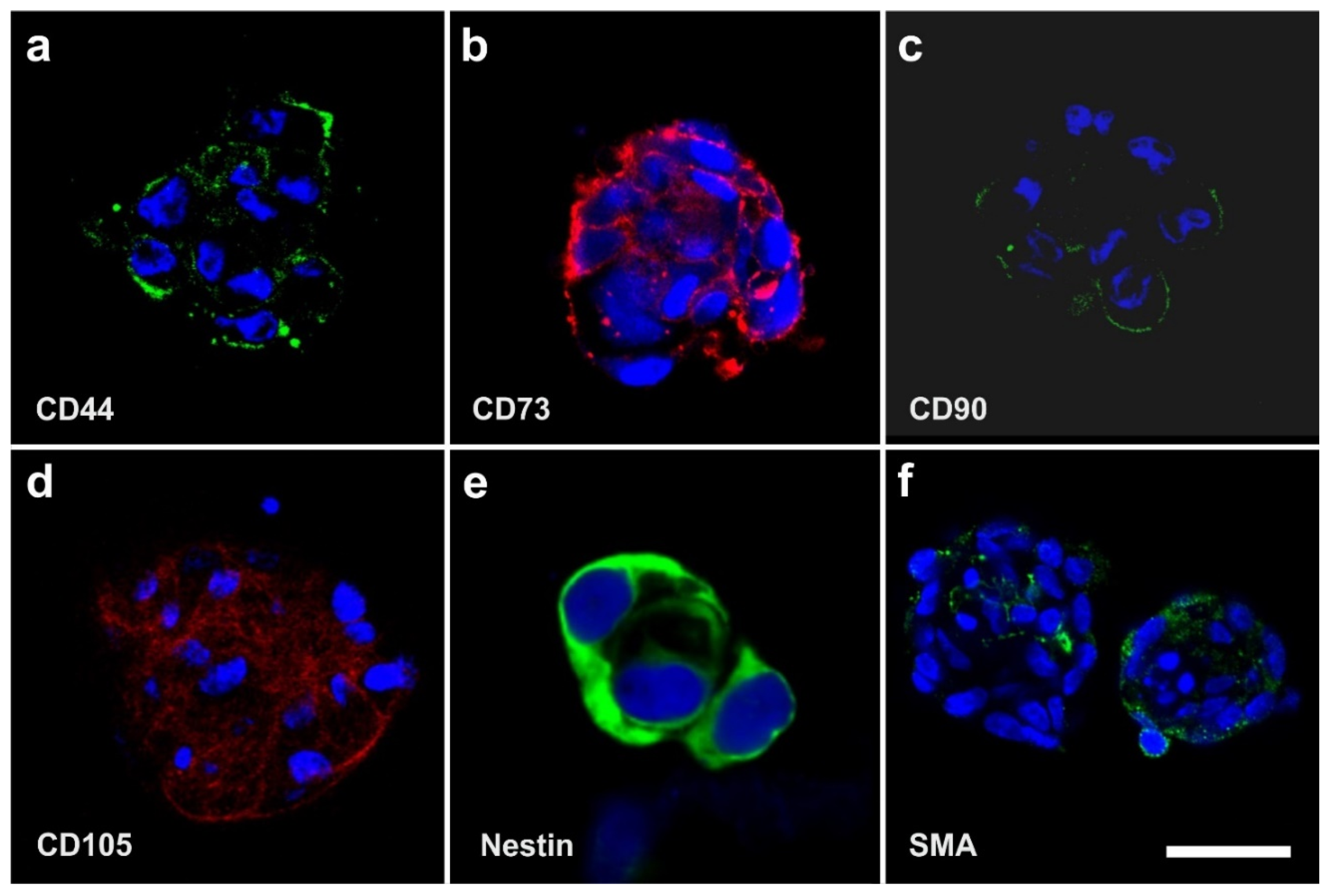
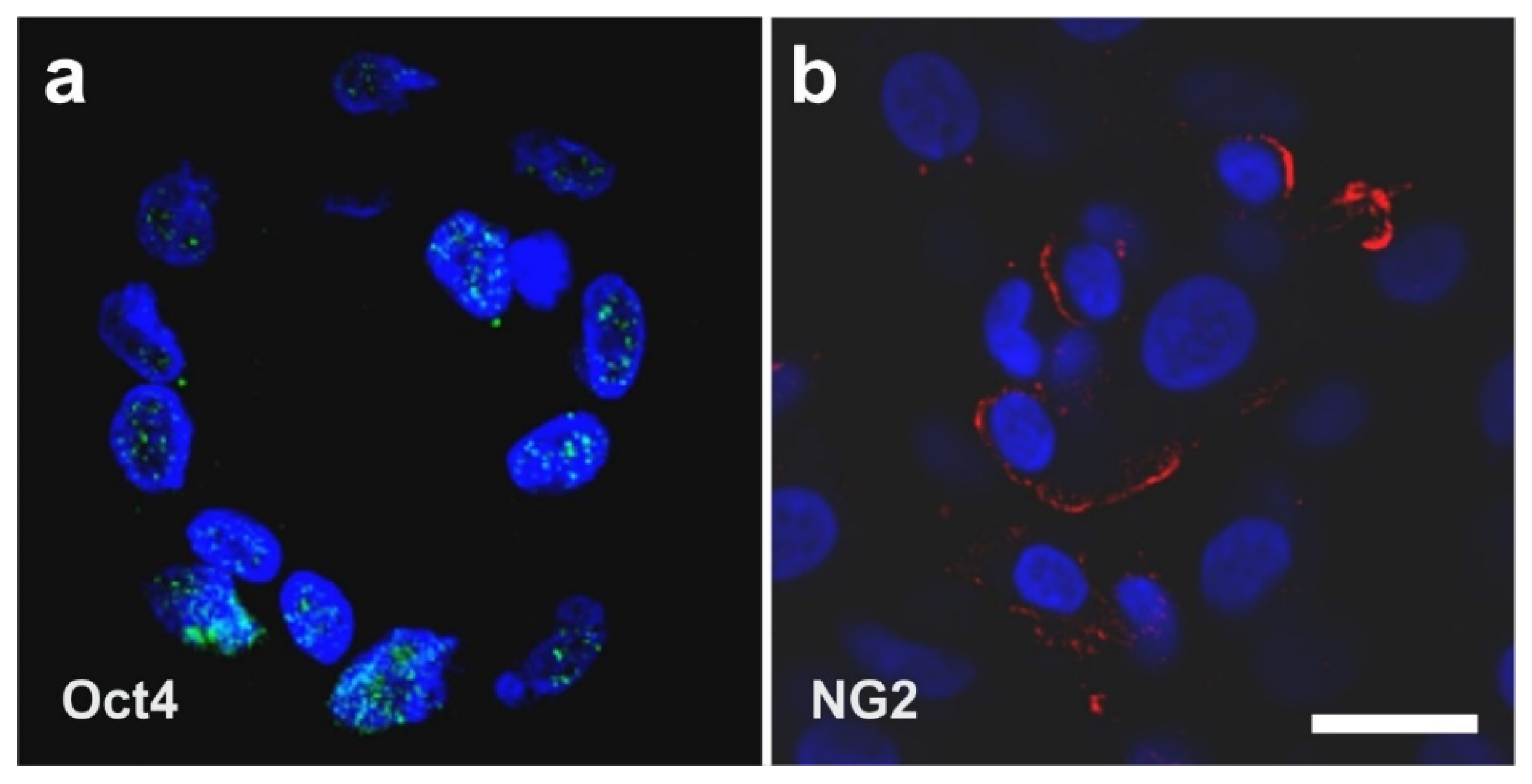
2.3. Surface Markers of vaPS Cells Cultured in Serum-Free Media
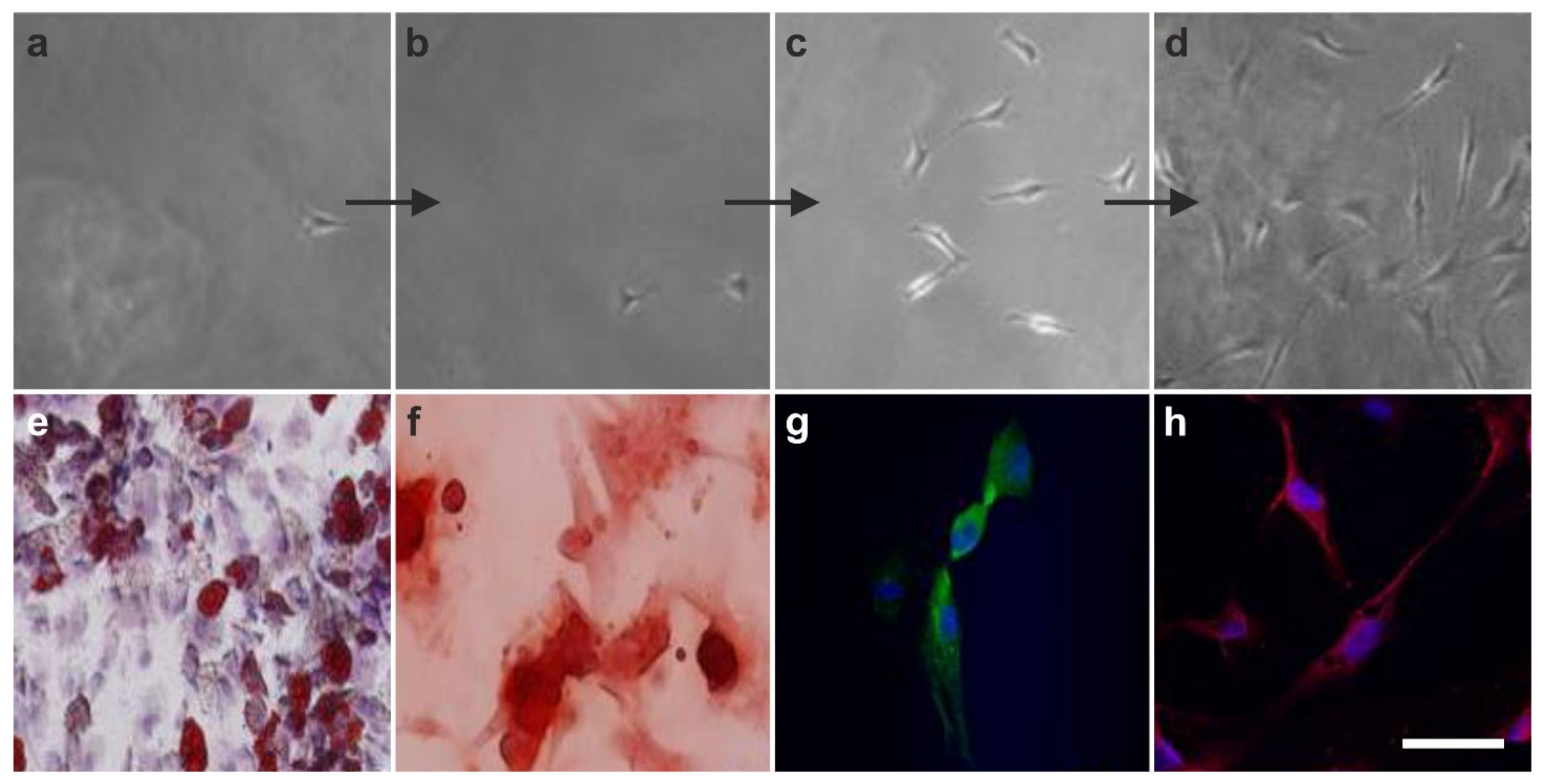
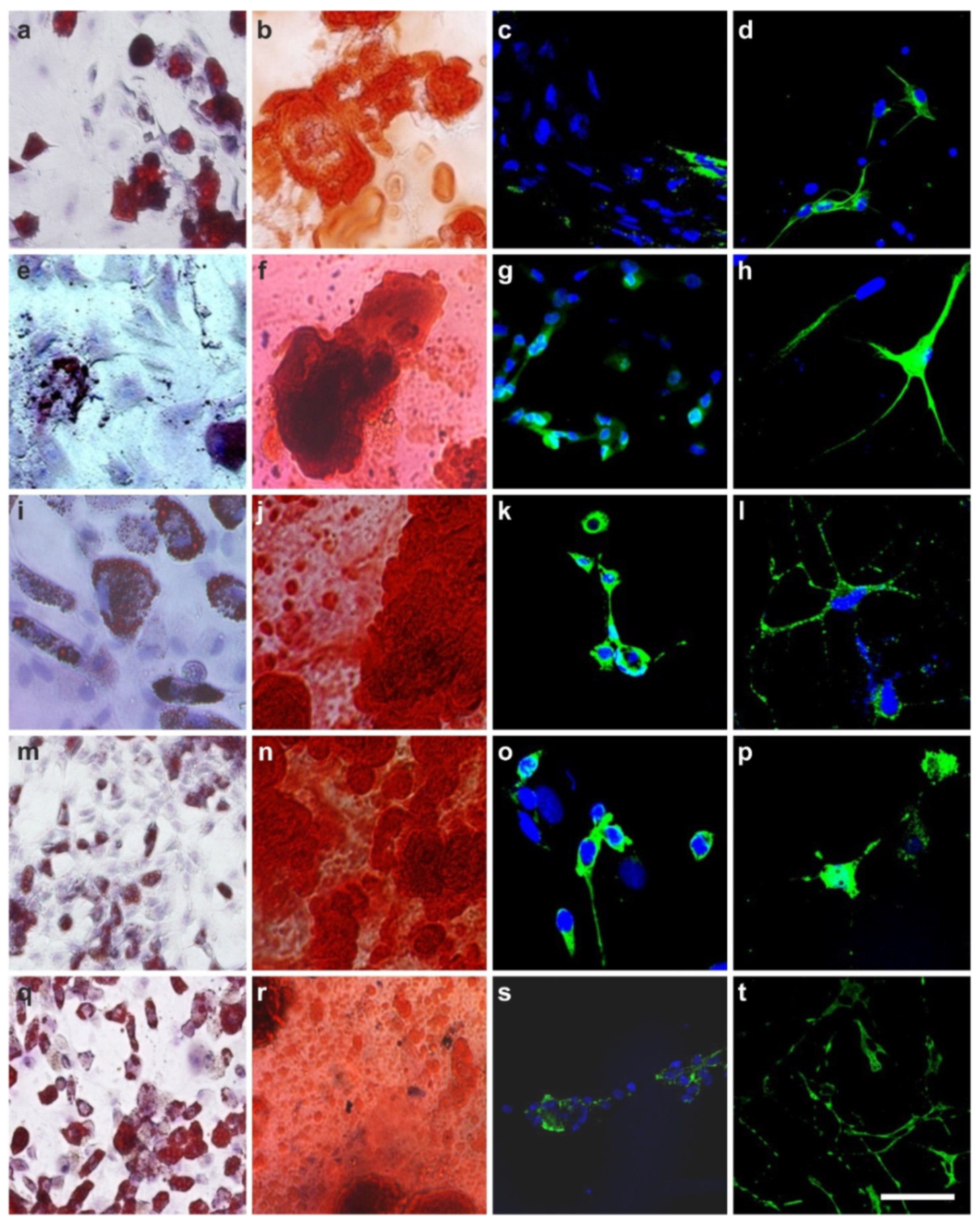
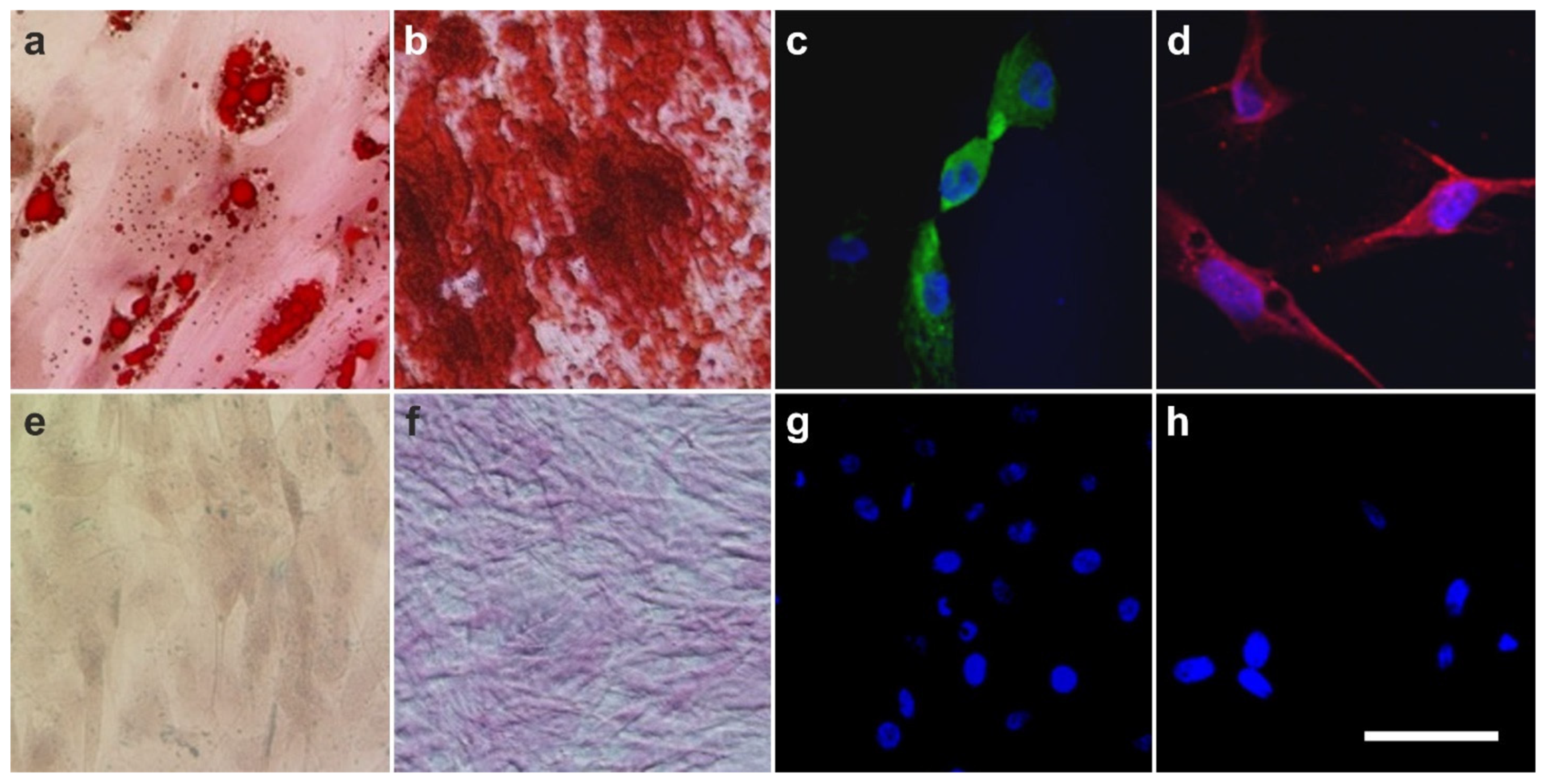
2.4. The Pluripotency of vaPS Cells
2.5. The Three Germ Layer Differentiation Potential of Human Adipose-Derived Stem Cells
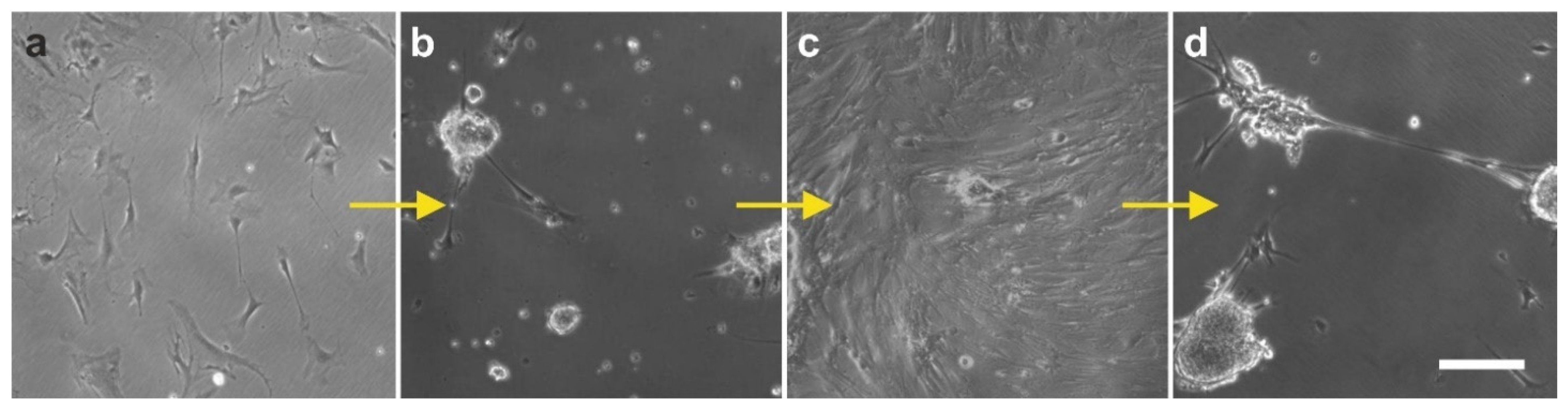
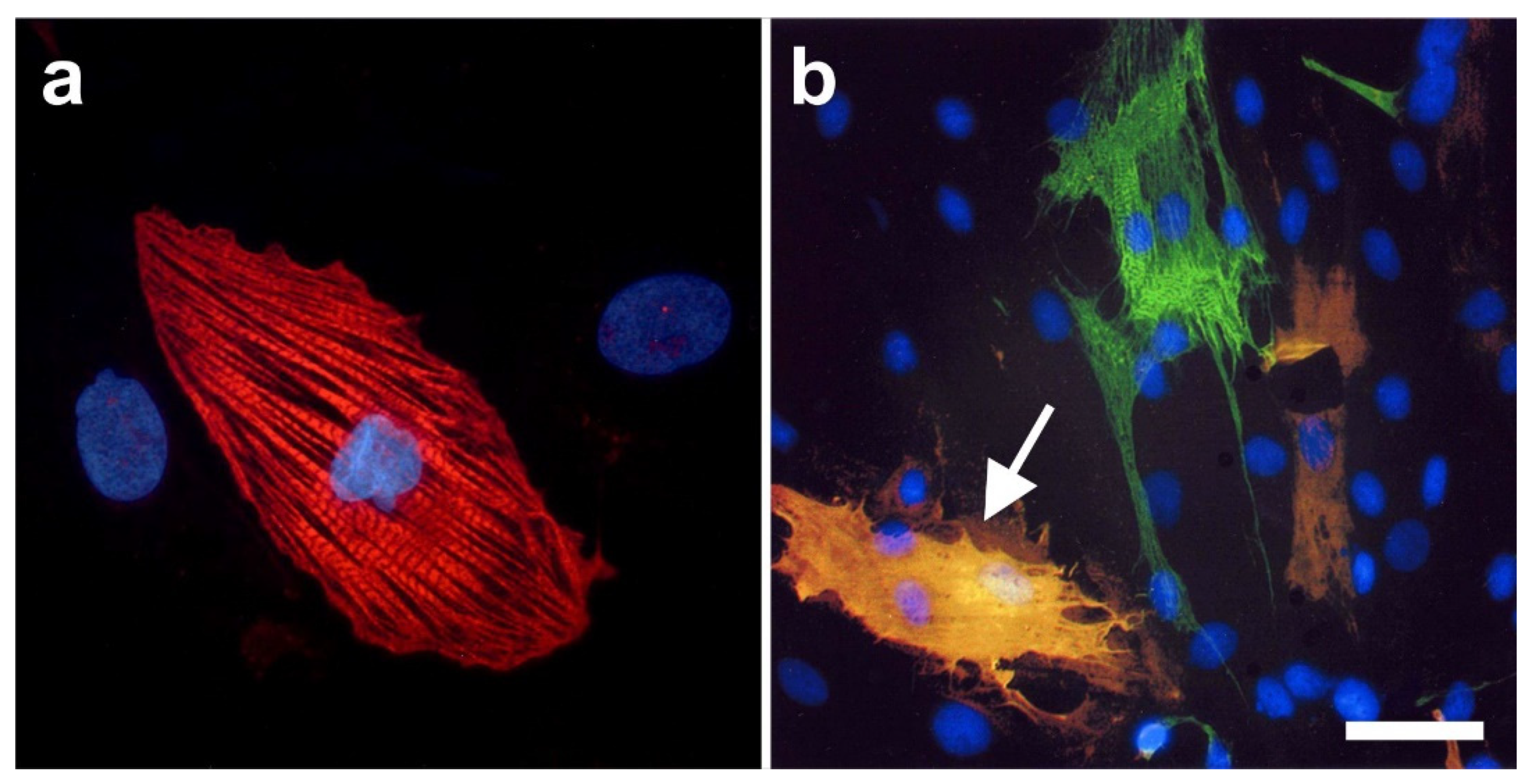
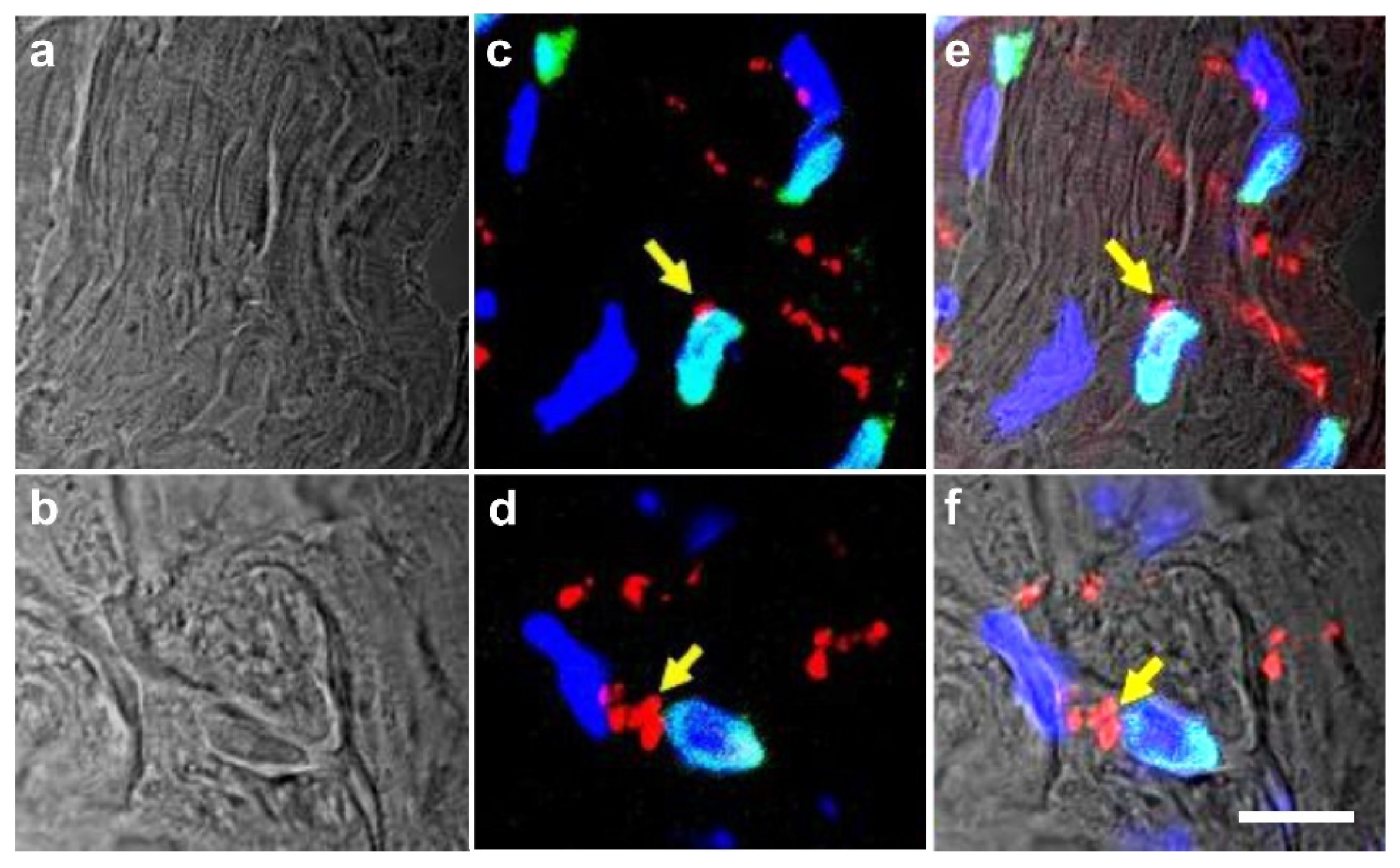
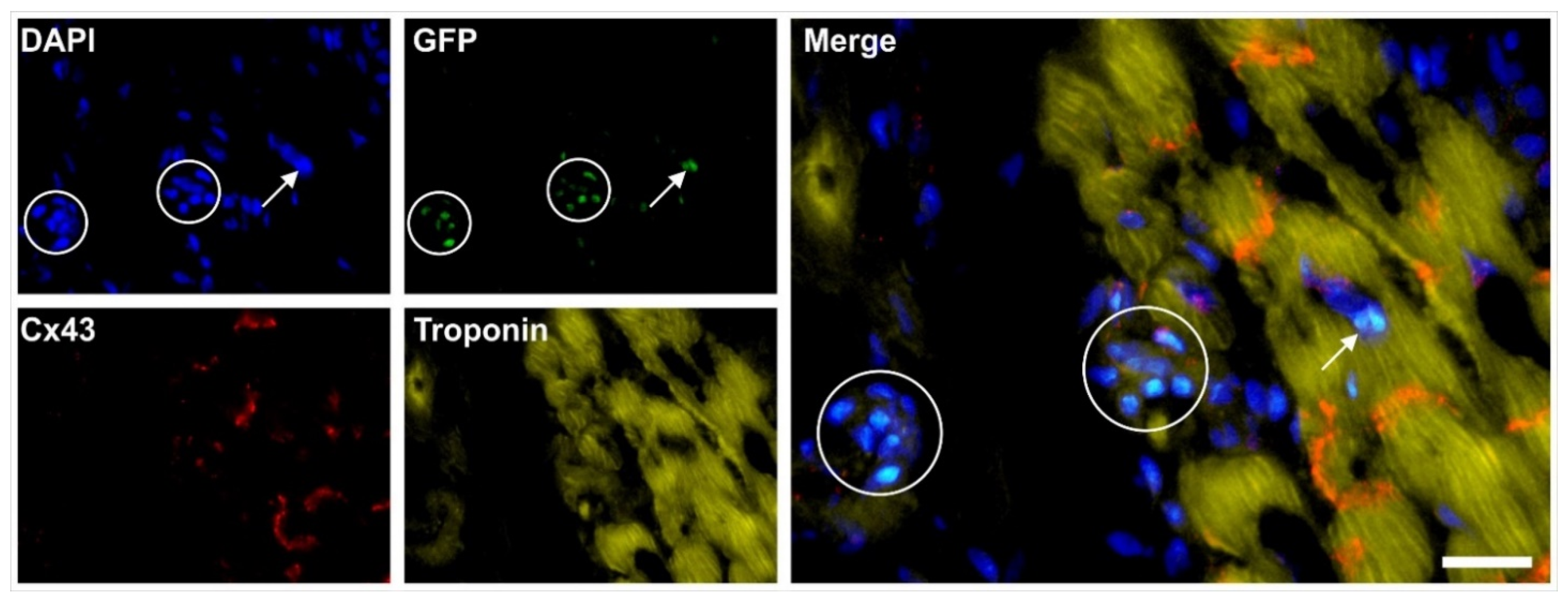
2.6. Integration of vaPS Cells into Host Tissue upon Activation
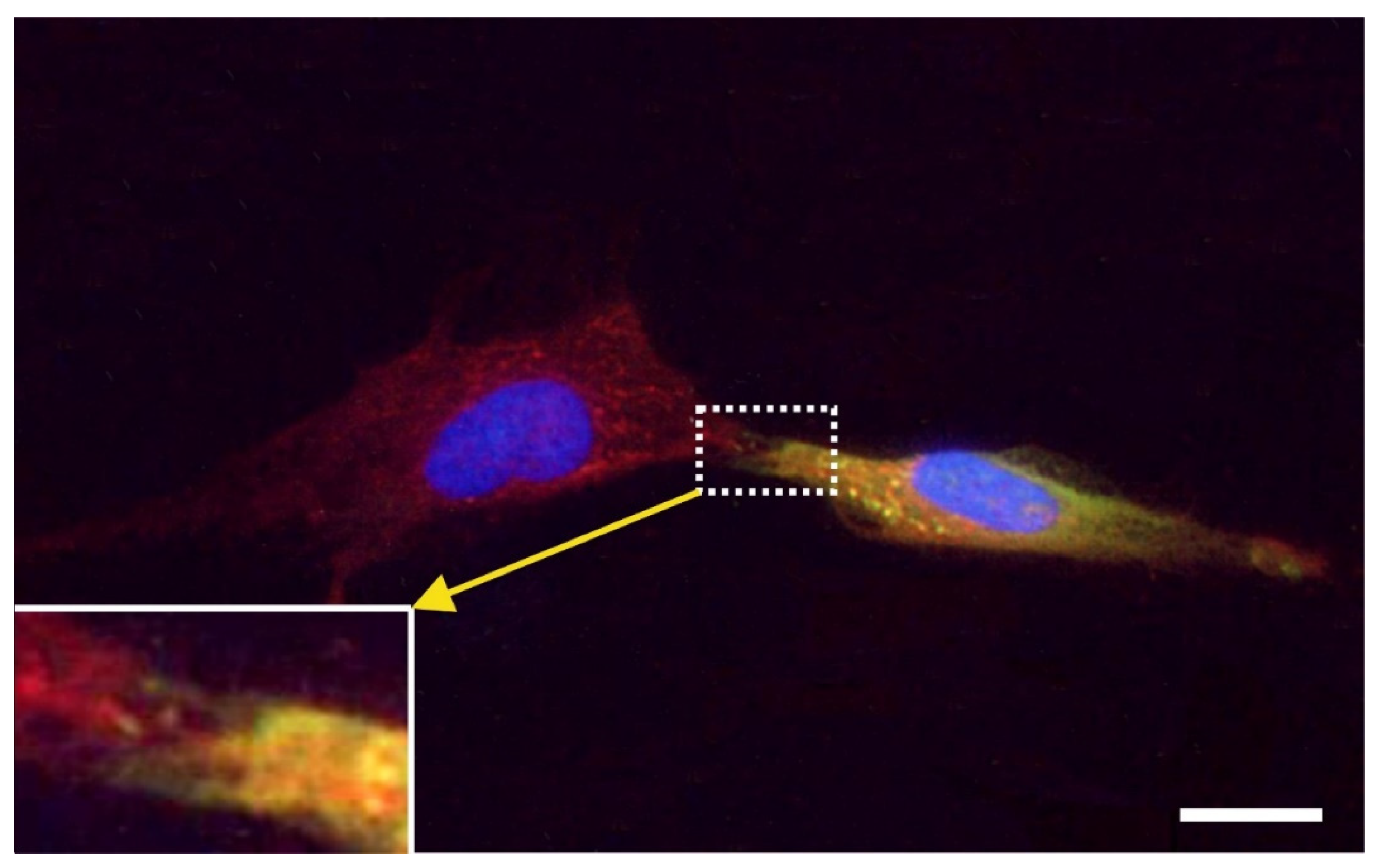
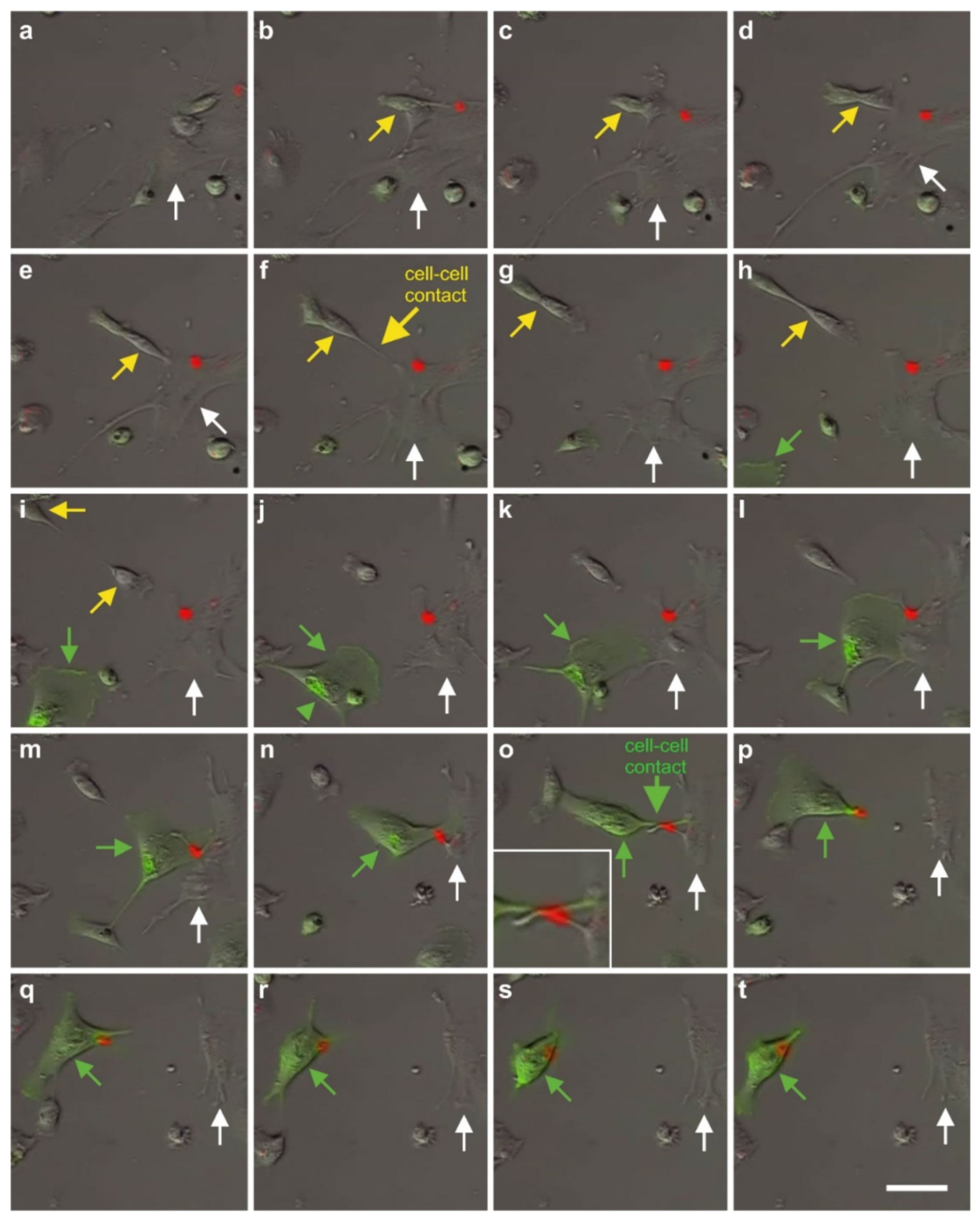
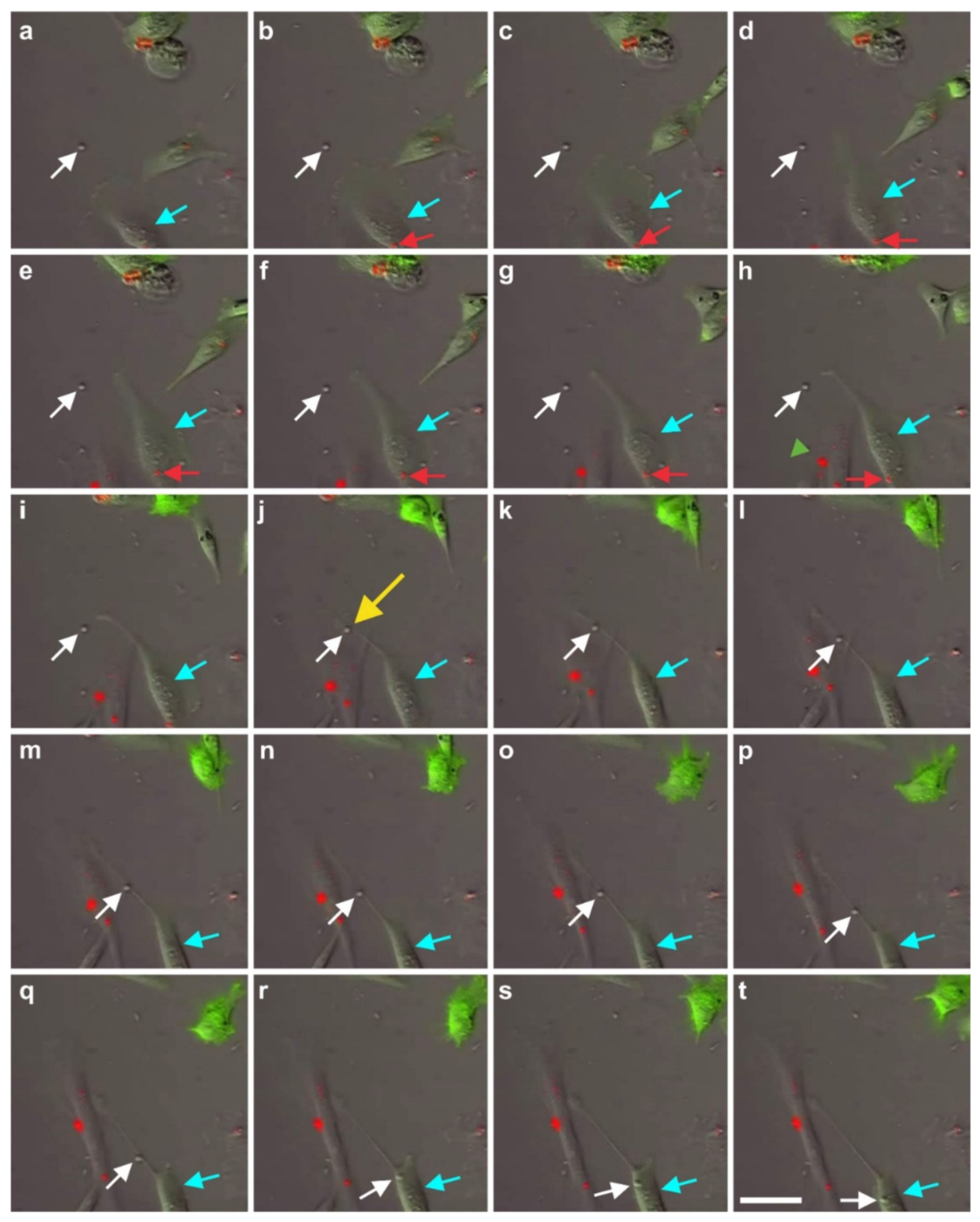
2.7. Exchange of Information between vaPS Cells and Other Cells in Cell Culture
2.8. Immunosuppressive and Anti-Inflammatory Activities of ADSCs
3. A Key Role of Stem Cell Therapy in Re-Establishing Tissue Homeostasis between Dying and Replacing Cells
4. The Significance of vaPS Cells and iPS Cells for the Practice of Medicine
5. Key Advantages of Adipose-Derived Regenerative Cells and Adipose-Derived Stem Cells for Cell Based Therapies
5.1. Comparison of Bone Marrow Derived Stem Cells with Adipose-Derived Regenerative Cells and Adipose-Derived Stem Cells
5.2. Differences in the Effectiveness of Various Systems and Methods That Are Available for Isolating Adipose-Derived Regenerative Cells
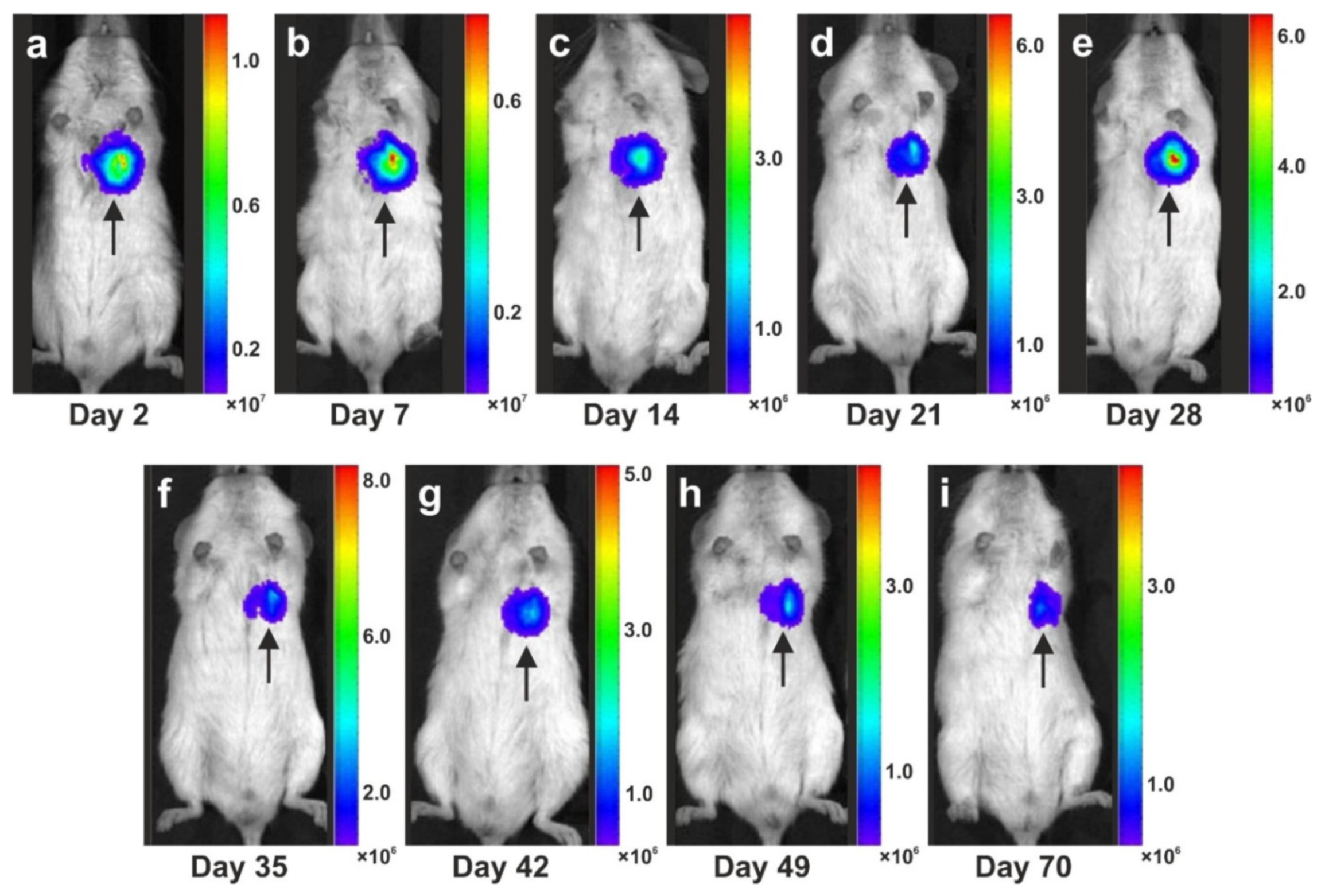
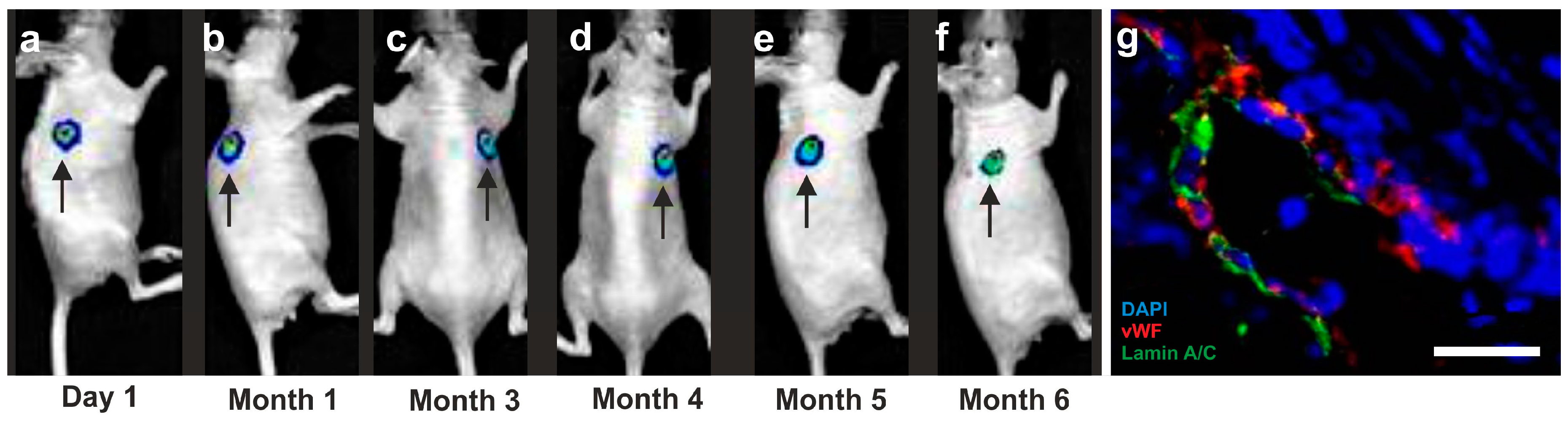
5.3. Long-Term Survival of Adipose-Derived Stem Cells after Transplantation in Animal Models into the Heart and Subcutaneous Locations
5.4. Specific Therapeutic Benefits of Adipose-Derived Regenerative Cells
5.5. Local vs. Systemic Application of UA-ADRCs
6. Examples of Application of Unmodified, Autologous, Adipose-Derived Regenerative Cells (UA-ADRCs) in Regenerative Cell Therapy
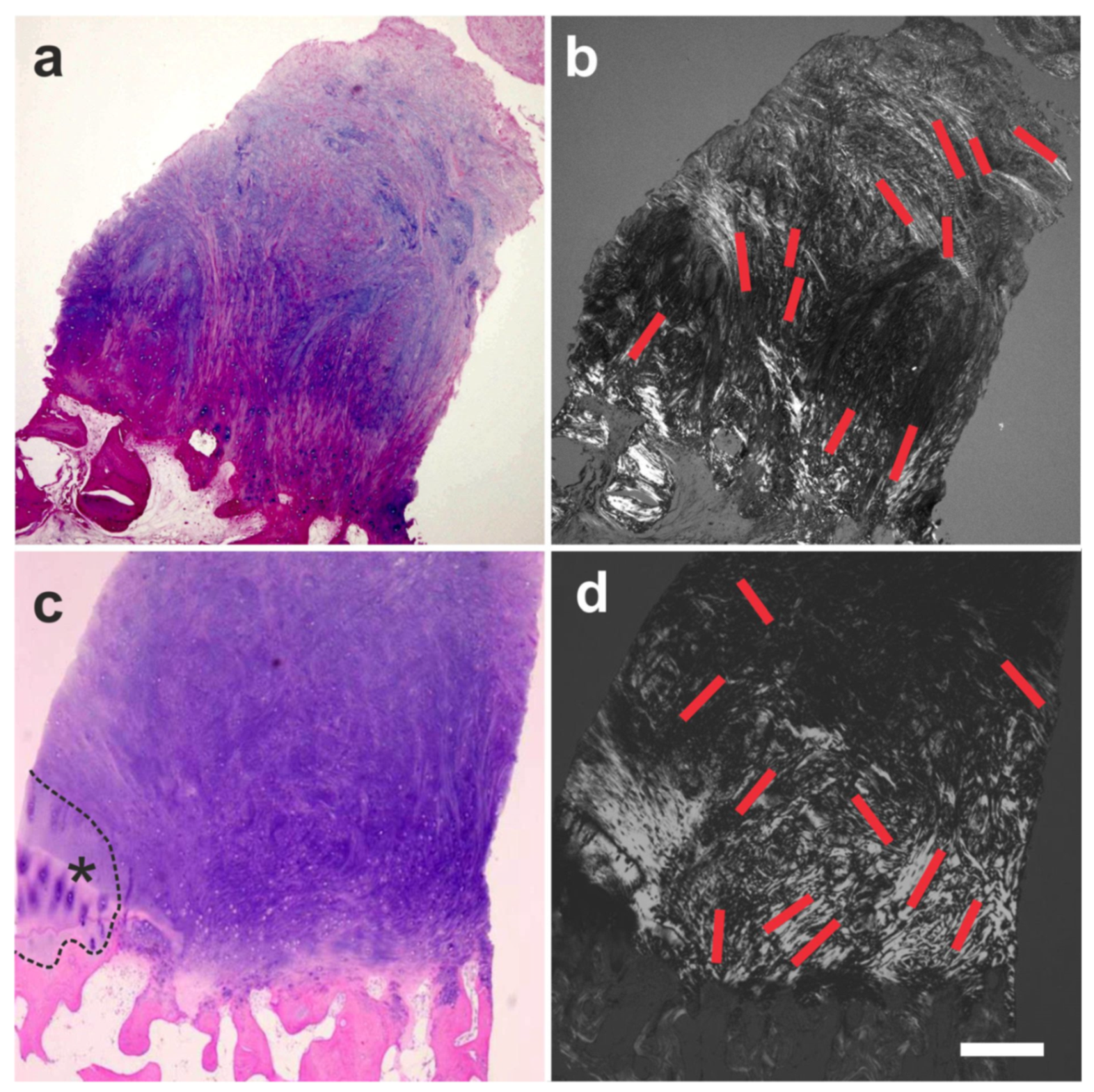
6.1. Tendon Defects
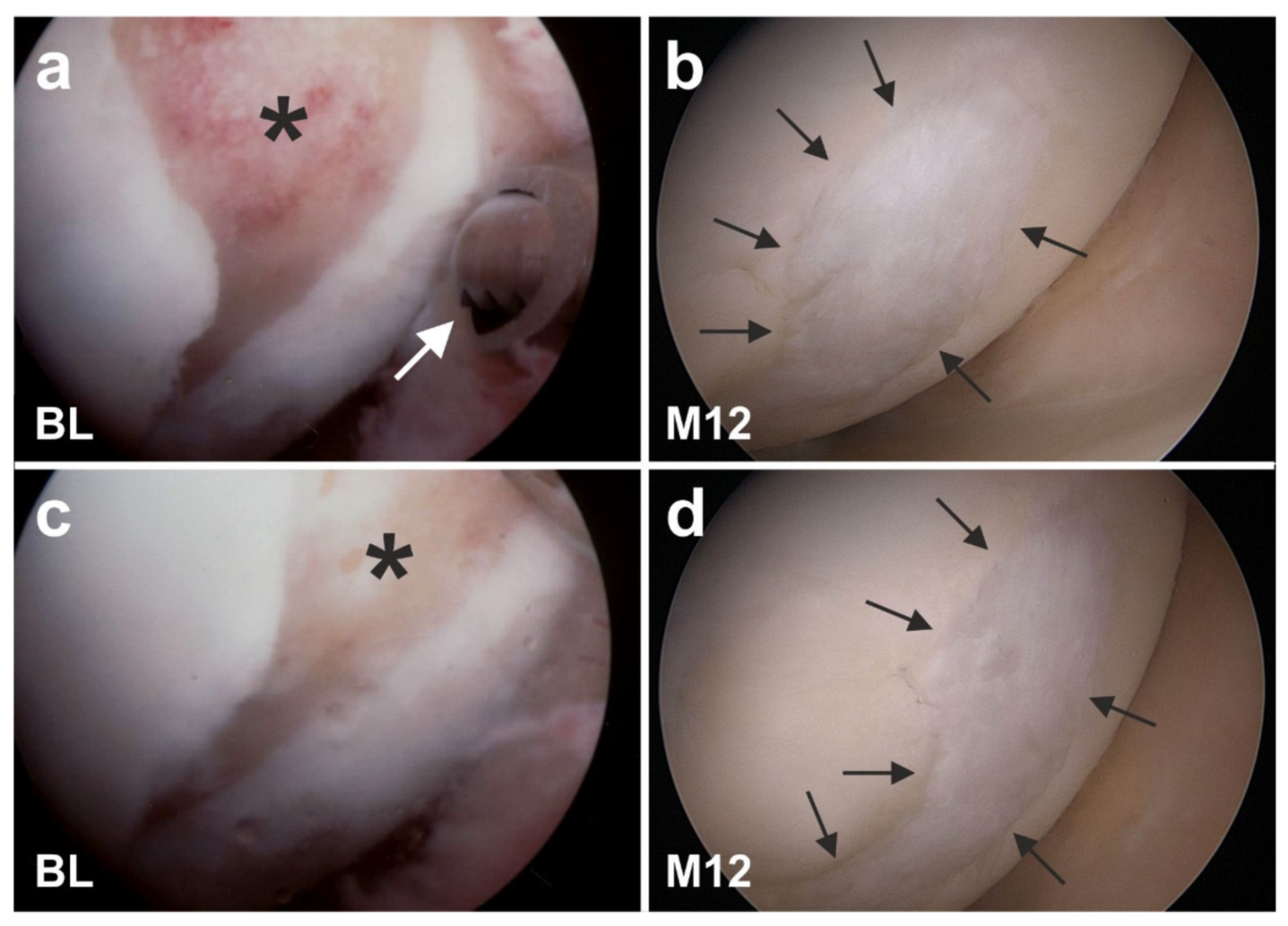
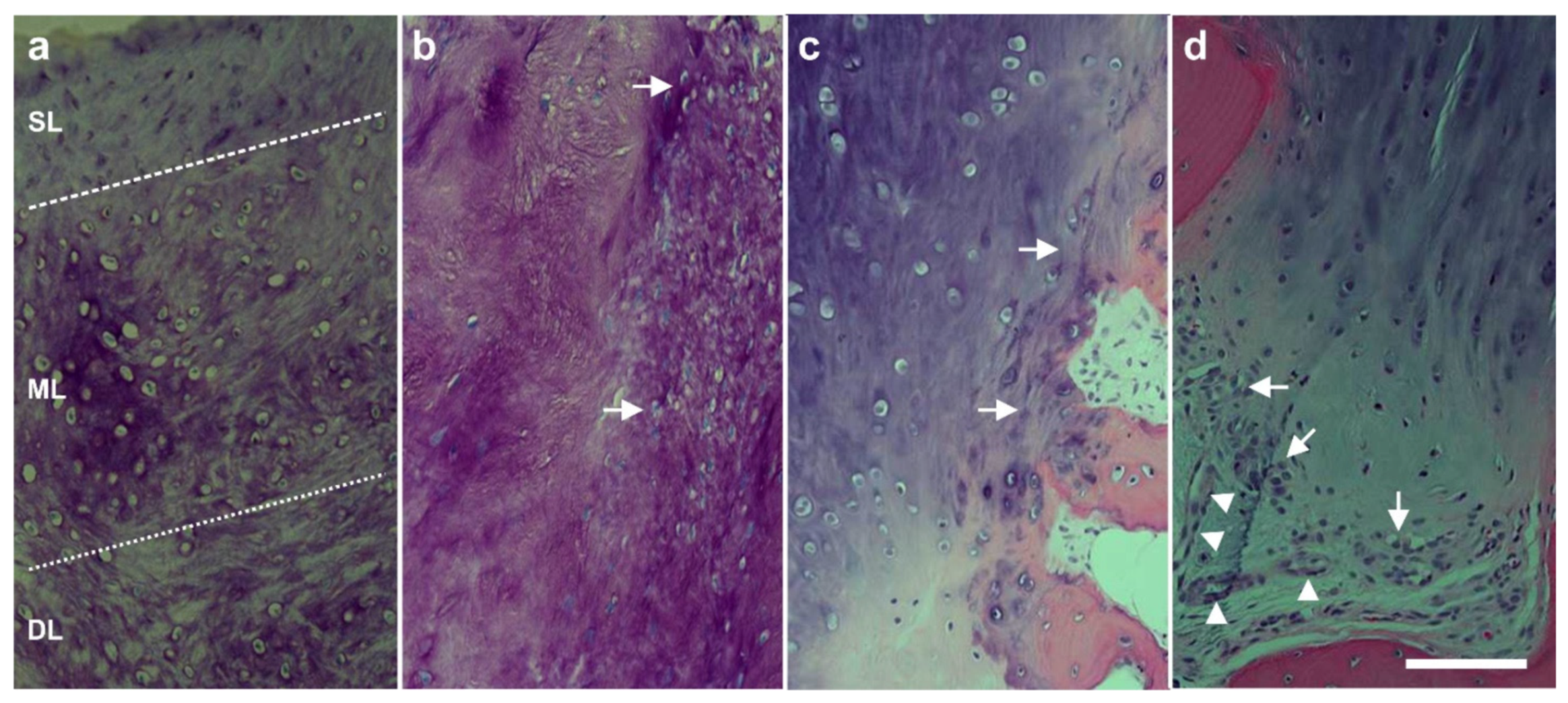
6.2. Cartilage Defects
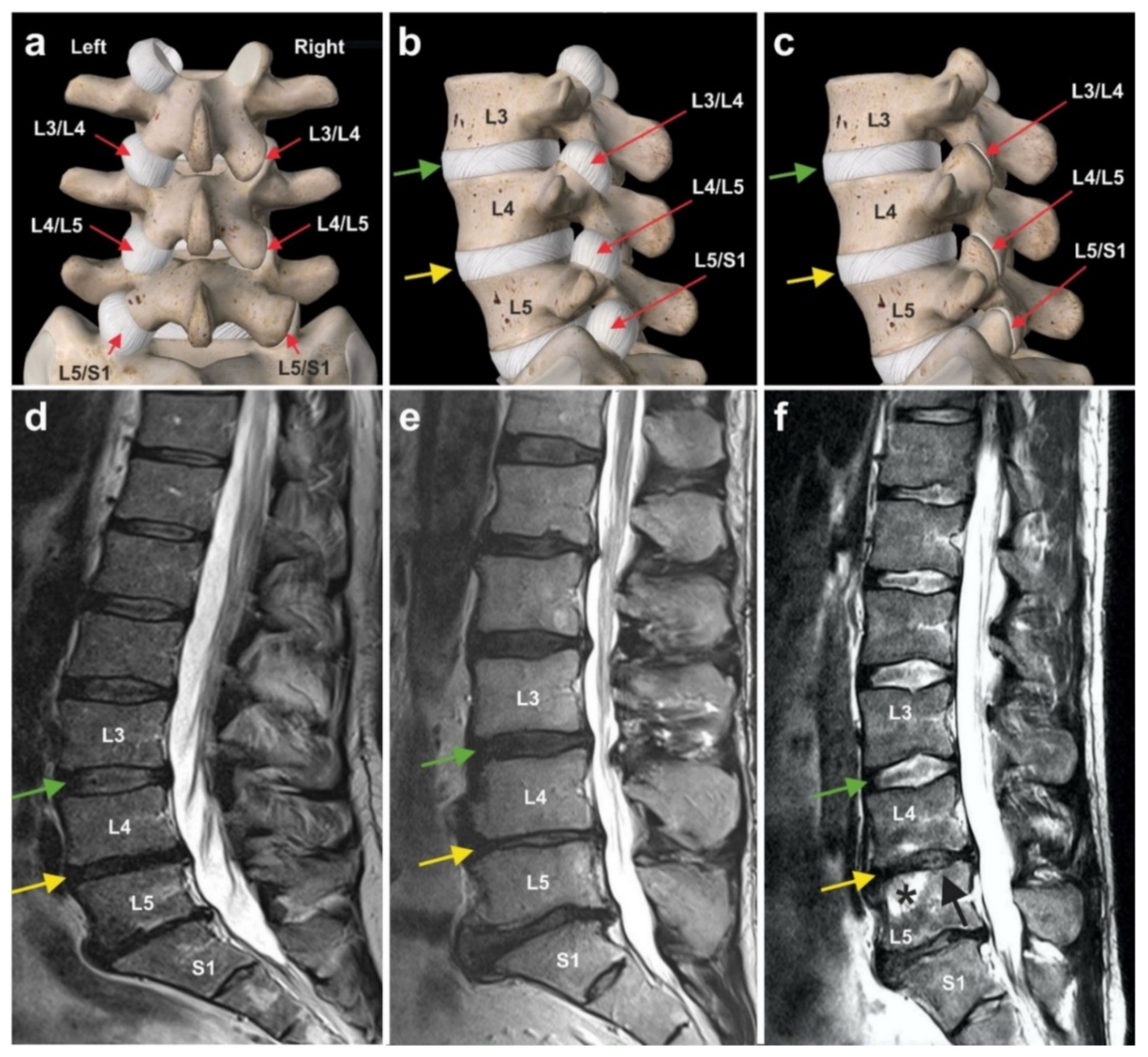
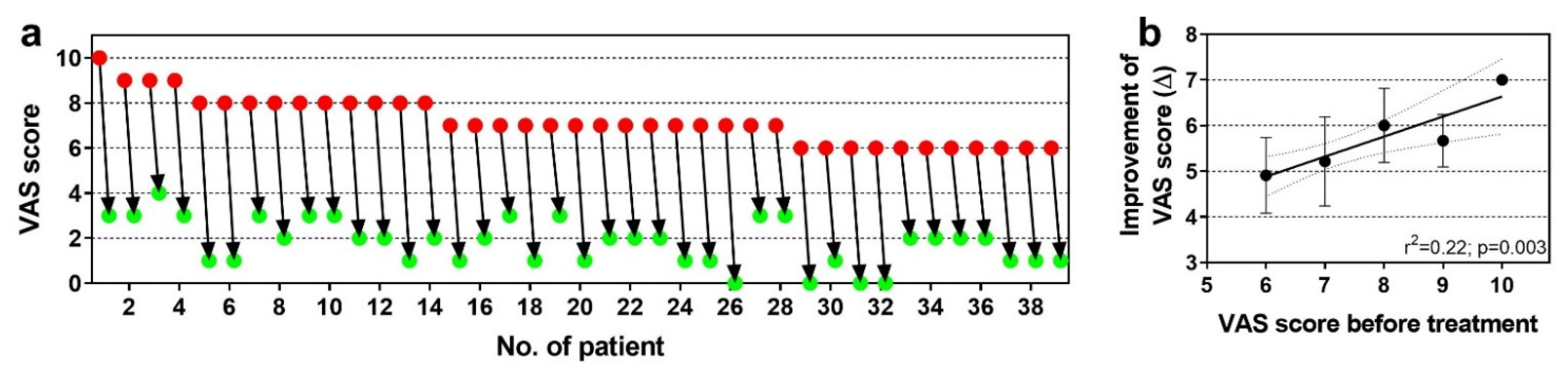
6.3. Chronic, Recalcitrant Low Back Pain Caused by Lumbosacral Facet Syndrome
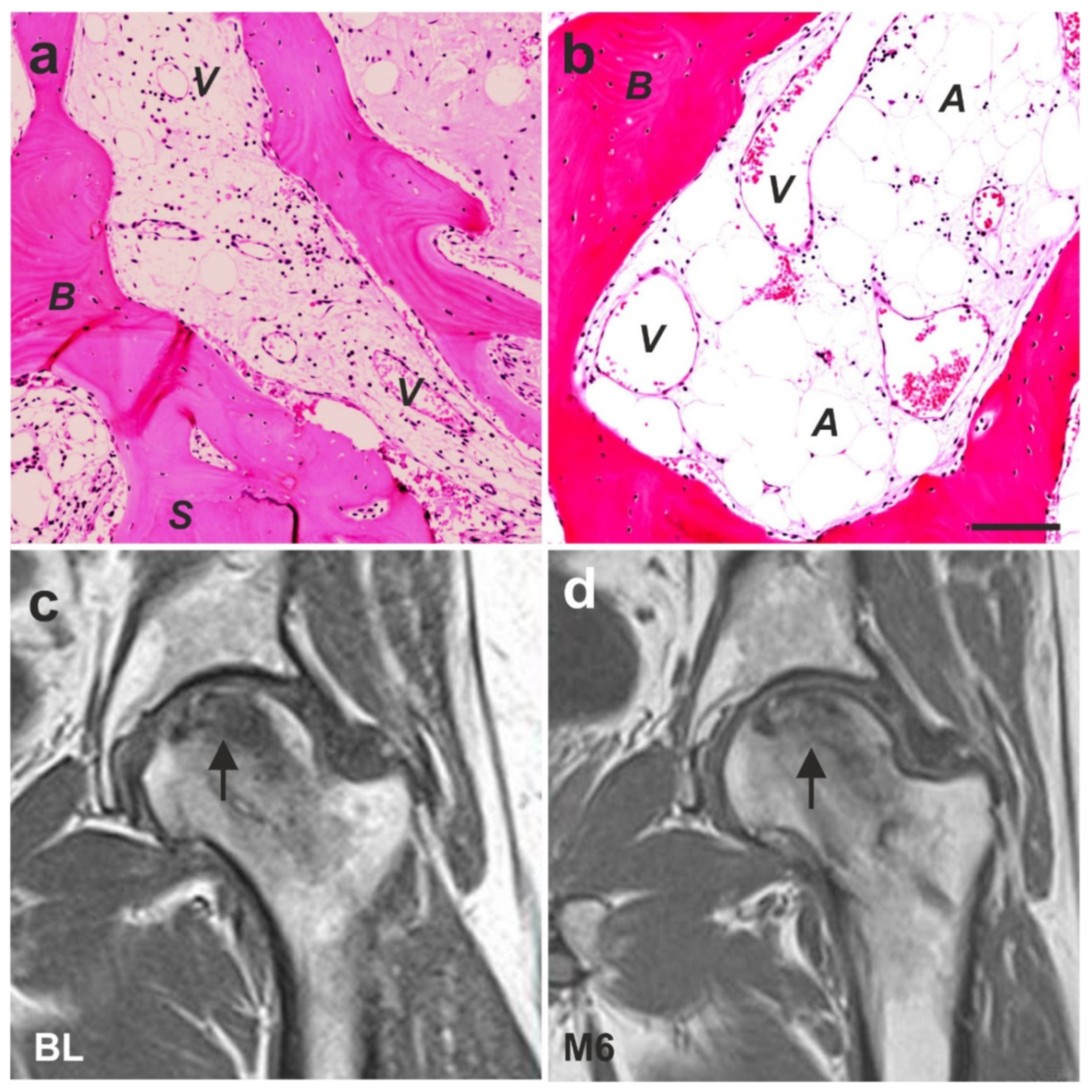
6.4. Guided Bone Regeneration (Individual Case Reports)
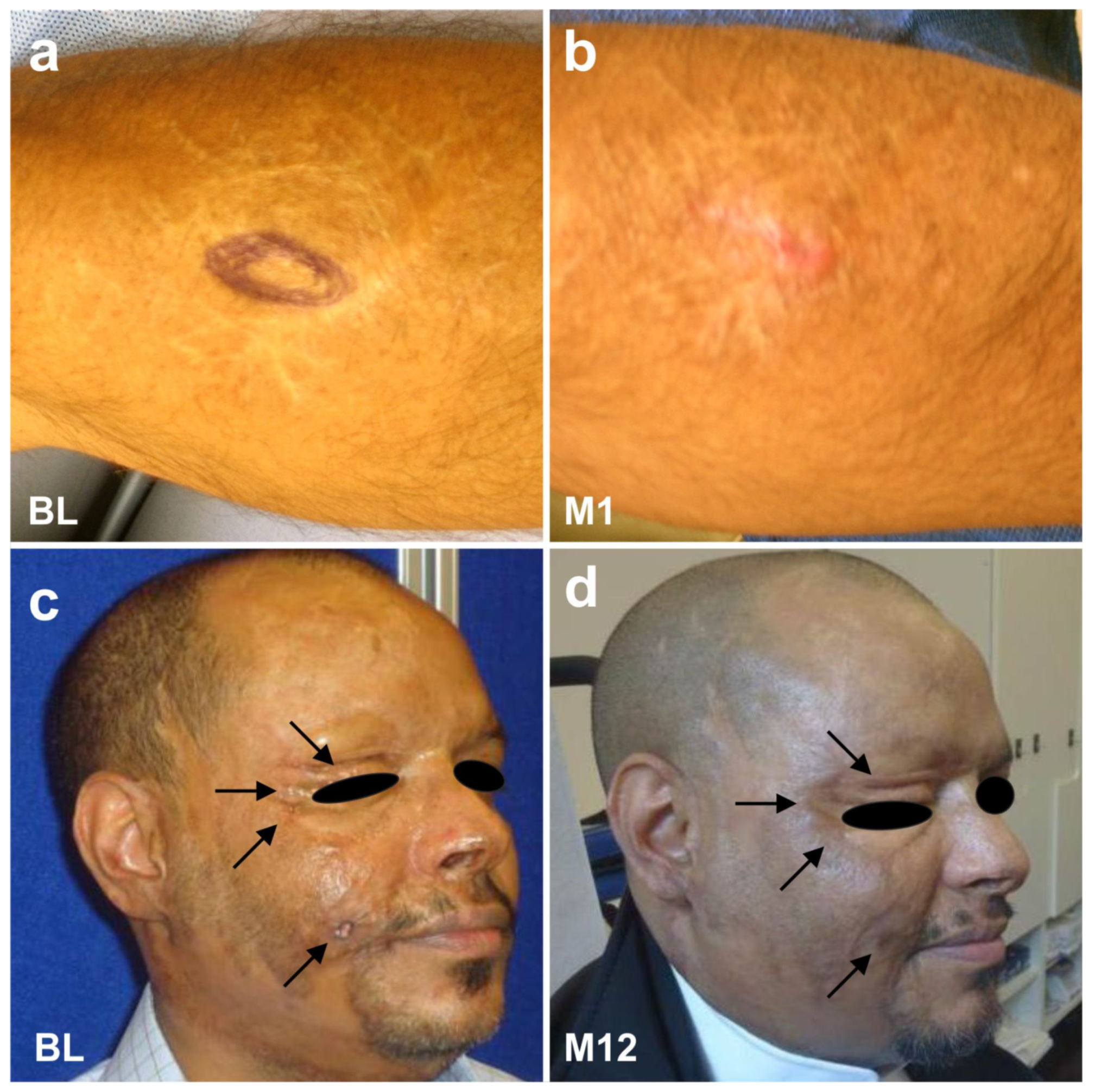
6.5. Examples of Other Treatments with UA-ADRCs (Individual Case Reports)
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Friedenstein, A.J.; Petrakova, K.V.; Kurolesova, A.I.; Frolova, G.P. Heterotopic of bone marrow. Analysis of precursor cells for osteogenic and hematopoietic tissues. Transplantation 1968, 6, 230–247. [Google Scholar] [CrossRef]
- Friedenstein, A.J.; Deriglasova, U.F.; Kulagina, N.N.; Panasuk, A.F.; Rudakowa, S.F.; Luriá, E.A.; Ruadkow, I.A. Precursors for fibroblasts in different populations of hematopoietic cells as detected by the in vitro colony assay method. Exp. Hematol. 1974, 2, 83–92. [Google Scholar]
- Nature Portfolio Stem Cells. Available online: https://www.nature.com/subjects/stem-cells (accessed on 6 August 2021).
- Stem Cell Key Terms. Available online: https://www.cirm.ca.gov/patients/stem-cell-key-terms#2 (accessed on 6 August 2021).
- Types of Stem Cells. Available online: https://www.closerlookatstemcells.org/learn-about-stem-cells/types-of-stem-cells/#mesenchymal (accessed on 6 August 2021).
- Boland, M.J.; Hazen, J.L.; Nazor, K.L.; Rodriguez, A.R.; Gifford, W.; Martin, G.; Kupriyanov, S.; Baldwin, K.K. Adult mice generated from induced pluripotent stem cells. Nature 2009, 461, 91–94. [Google Scholar] [CrossRef]
- Zhao, X.; Li, W.; Lv, Z.; Liu, L.; Tong, M.; Hai, T.; Hao, J.; Guo, C.; Ma, Q.; Wang, L.; et al. IPS cells produce viable mice through tetraploid complementation. Nature 2009, 461, 86–90. [Google Scholar] [CrossRef] [PubMed]
- Dominici, M.; Le Blanc, K.; Mueller, I.; Slaper-Cortenbach, I.; Marini, F.C.; Krause, D.S.; Deans, R.J.; Keating, A.; Prockop, D.J.; Horwitz, E.M. Minimal criteria for defining multipotent mesenchymal stromal cells. The International Society for Cellular Therapy position statement. Cytotherapy 2006, 8, 315–317. [Google Scholar] [CrossRef] [PubMed]
- Bai, X.; Yan, Y.; Song, Y.-H.; Seidensticker, M.; Rabinovich, B.; Metzele, R.; Bankson, J.A.; Vykoukal, D.; Alt, E. Both cultured and freshly isolated adipose tissue-derived stem cells enhance cardiac function after acute myocardial infarction. Eur. Heart J. 2010, 31, 489–501. [Google Scholar] [CrossRef] [PubMed]
- Winnier, G.E.; Valenzuela, N.; Peters-Hall, J.; Kellner, J.; Alt, C.; Alt, E.U. Isolation of adipose tissue derived regenerative cells from human subcutaneous tissue with or without the use of an enzymatic reagent. PLoS ONE 2019, 14, e0221457. [Google Scholar] [CrossRef] [PubMed]
- Davidoff, M.S. The pluripotent microvascular pericytes are the adult stem cells even in the testis. Adv. Exp. Med. Biol. 2019, 1122, 235–267. [Google Scholar] [PubMed]
- Lv, F.-J.; Tuan, R.S.; Cheung, K.M.C.; Leung, V.Y.L. Concise review: The surface markers and identity of human mesenchymal stem cells: Markers and identity of MSCs. Stem Cells 2014, 32, 1408–1419. [Google Scholar] [CrossRef]
- Crisan, M.; Corselli, M.; Chen, W.C.W.; Péault, B. Perivascular cells for regenerative medicine. J. Cell. Mol. Med. 2012, 16, 2851–2860. [Google Scholar] [CrossRef]
- Sá da Bandeira, D.; Casamitjana, J.; Crisan, M. Pericytes, integral components of adult hematopoietic stem cell niches. Pharmacol. Ther. 2017, 171, 104–113. [Google Scholar] [CrossRef] [PubMed]
- Sims, D.E. The pericyte-A review. Tissue Cell 1986, 18, 153–174. [Google Scholar] [CrossRef]
- Mannino, G.; Gennuso, F.; Giurdanella, G.; Conti, F.; Drago, F.; Salomone, S.; Furno, D.L.; Bucolo, C.; Giuffrida, R. Pericyte-like differentiation of human adipose-derived mesenchymal stem cells: An in vitro study. World J. Stem Cells 2020, 12, 1152–1170. [Google Scholar] [CrossRef]
- Binamé, F. Transduction of extracellular cues into cell polarity: The role of the transmembrane proteoglycan NG2. Mol. Neurobiol. 2014, 50, 482–493. [Google Scholar] [CrossRef]
- Barilani, M.; Peli, V.; Cherubini, A.; Dossena, M.; Dolo, V.; Lazzari, L. NG2 as an identity and quality marker of mesenchymal stem cell extracellular vesicles. Cells 2019, 8, 1524. [Google Scholar] [CrossRef] [PubMed]
- Loder, S.J.; Agarwal, S.; Chung, M.T.; Cholok, D.; Hwang, C.; Visser, N.; Vasquez, K.; Sorkin, M.; Habbouche, J.; Sung, H.H.; et al. Characterizing the circulating cell populations in traumatic heterotopic ossification. Am. J. Pathol. 2018, 188, 2464–2473. [Google Scholar] [CrossRef] [PubMed]
- Naaijkens, B.A.; Krijnen, P.A.; Meinster, E.; ter Horst, E.N.; Vo, K.; Musters, R.J.; Kamp, O.; Niessen, H.W.; Juffermans, L.J.; van Dijk, A. Acute myocardial infarction does not affect functional characteristics of adipose-derived stem cells in rats, but reduces the number of stem cells in adipose tissue. Cell Tissue Res. 2005, 362, 623–632. [Google Scholar] [CrossRef]
- Díaz-Flores, L.; Gutiérrez, R.; Varela, H.; Rancel, N.; Valladares, F. Microvascular pericytes: A review of their morphological and functional characteristics. Histol. Histopathol. 1991, 6, 269–286. [Google Scholar]
- Guillemin, G.J.; Brew, B.J. Microglia, macrophages, perivascular macrophages, and pericytes: A review of function and identification. J. Leukoc. Biol. 2004, 75, 388–397. [Google Scholar] [CrossRef] [PubMed]
- Fernandez-Klett, F.; Priller, J. Diverse functions of pericytes in cerebral blood flow regulation and ischemia. J. Cereb. Blood Flow Metab. 2015, 35, 883–88760. [Google Scholar] [CrossRef]
- Tillet, E.; Ruggiero, F.; Nishiyama, A.; Stallcup, W.B. The membrane-spanning proteoglycan NG2 binds to collagens V and VI through the central nonglobular domain of its core protein. J. Biol. Chem. 1997, 272, 10769–10776. [Google Scholar] [CrossRef] [PubMed]
- Kuo, H.-J.; Maslen, C.L.; Keene, D.R.; Glanville, R.W. Type VI collagen anchors endothelial basement membranes by interacting with Type IV collagen. J. Biol. Chem. 1997, 272, 26522–26529. [Google Scholar] [CrossRef]
- Ratajczak, M.Z.; Ratajczak, J.; Suszynska, M.; Miller, D.M.; Kucia, M.; Shin, D.M. A novel view of the adult stem cell compartment from the perspective of a quiescent population of very small embryonic-like stem cells. Circ. Res. 2017, 120, 166–178. [Google Scholar] [CrossRef] [PubMed]
- Ratajczak, M.Z.; Ratajczak, J.; Kucia, M. Very small embryonic-like stem cells (VSELs). Circ. Res. 2019, 124, 208–210. [Google Scholar] [CrossRef] [PubMed]
- Song, Y.H.; Prantl, L.; Alt, E. Differentiation and plasticity of stem cells for tissue engineering. In Tissue Engineering; Pallua, N., Suscheck, C.V., Eds.; Springer: Berlin, Germany, 2011; pp. 113–130. [Google Scholar]
- Bruno, I.; Martinez, R.; Sanchez, A.; Friddle, C.; McClure, S.R. Characterization of nucleated cells from equine adipose tissue and bone marrow aspirate processed for point-of-care use. J. Equine Vet. Sci. 2014, 34, 1118–1127. [Google Scholar] [CrossRef]
- Velten, L.; Haas, S.F.; Raffel, S.; Blaszkiewicz, S.; Islam, S.; Hennig, B.P.; Hirche, C.; Lutz, C.; Buss, E.C.; Nowak, D.; et al. Human haematopoietic stem cell lineage commitment is a continuous process. Nat. Cell Biol. 2017, 19, 271–281. [Google Scholar] [CrossRef]
- Schmitz, C.; Alt, E. Stammzellentherapie. Erstmalige Erfolgreiche Behandlung von Teilrupturen der Rotatorenmanschette Mit Frischen, Adulten, Körpereigenen Regenerativen Zellen. Available online: https://sportaerztezeitung.com/rubriken/therapie/1227/stammzellentherapie/ (accessed on 30 August 2021).
- Di Russo, J.; Hannocks, M.J.; Luik, A.L.; Song, J.; Zhang, X.; Yousif, L.; Aspite, G.; Hallmann, R.; Sorokin, L. Vascular laminins in physiology and pathology. Matrix Biol. 2017, 57–58, 140–148. [Google Scholar] [CrossRef]
- Suzuki, S.; Namiki, J.; Shibata, S.; Mastuzaki, Y.; Okano, H. The neural stem/progenitor cell marker nestin is expressed in proliferative endothelial cells, but not in mature vasculature. J. Histochem. Cytochem. 2010, 58, 721–730. [Google Scholar] [CrossRef] [PubMed]
- Clark, R.A.F. Fibronectin matrix deposition and fibronectin receptor expression in healing and normal skin. J. Investig. Dermatol. 1990, 94, 128S–134S. [Google Scholar] [CrossRef]
- Serini, G.; Valdembri, D.; Bussolino, F. Integrins and angiogenesis: A sticky business. Exp. Cell Res. 2006, 312, 651–658. [Google Scholar] [CrossRef]
- Vega, M.E.; Schwarzbauer, J.E. Collaboration of fibronectin matrix with other extracellular signals in morphogenesis and differentiation. Curr. Opin. Cell Biol. 2016, 42, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Lane, T.F.; Sage, E.H. The biology of SPARC, a protein that modulates cell-matrix interactions. FASEB J. 1994, 8, 163–173. [Google Scholar] [CrossRef]
- Murphy-Ullrich, J.E.; Sage, E.H. Revisiting the matricellular concept. Matrix Biol. 2014, 37, 1–14. [Google Scholar] [CrossRef]
- Nie, J.; Chang, B.; Traktuev, D.O.; Sun, J.; March, K.; Chan, L.; Sage, E.H.; Pasqualini, R.; Arap, W.; Kolonin, M.G. IFATS collection: Combinatorial peptides identify alpha5beta1 integrin as a receptor for the matricellular protein SPARC on adipose stromal cells. Stem Cells 2008, 26, 2735–2745. [Google Scholar] [CrossRef] [PubMed]
- Tseng, C.; Kolonin, M.G. Proteolytic isoforms of SPARC induce adipose stromal cell mobilization in obesity: Mobilization of adipose stromal cells by SPARC. Stem Cells 2016, 34, 174–190. [Google Scholar] [CrossRef] [PubMed]
- Holland, P.W.; Harper, S.J.; McVey, J.H.; Hogan, B.L. In vivo expression of mRNA for the Ca++-binding protein SPARC (osteonectin) revealed by in situ hybridization. J. Cell Biol. 1987, 105, 473–482. [Google Scholar] [CrossRef]
- Brekken, R.A.; Sage, E.H. SPARC, a matricellular protein: At the crossroads of cell–matrix communication. Matrix Biol. 2001, 19, 815–827. [Google Scholar] [CrossRef]
- Chiodoni, C.; Colombo, M.P.; Sangaletti, S. Matricellular proteins: From homeostasis to inflammation, cancer, and metastasis. Cancer Metastasis Rev. 2010, 29, 295–307. [Google Scholar] [CrossRef]
- Bradshaw, A.D. The role of secreted protein acidic and rich in cysteine (SPARC) in cardiac repair and fibrosis: Does expression of SPARC by macrophages influence outcomes? J. Mol. Cell. Cardiol. 2016, 93, 156–161. [Google Scholar] [CrossRef]
- Le Bail, B.; Faouzi, S.; Boussarie, L.; Guirouilh, J.; Blanc, J.F.; Carles, J.; Bioulac-Sage, P.; Balabaud, C.; Rosenbaum, J. Osteonectin/SPARC is overexpressed in human hepatocellular carcinoma. J. Pathol. 1999, 189, 46–52. [Google Scholar] [CrossRef]
- Shaik, S.; Wu, X.; Gimble, J.; Devireddy, R. Effects of decade long freezing storage on adipose derived stem cells functionality. Sci. Rep. 2018, 8, 8162. [Google Scholar] [CrossRef]
- Choi, Y.A.; Lim, J.; Kim, K.M.; Acharya, B.; Cho, J.Y.; Bae, Y.C.; Shin, H.I.; Kim, S.Y.; Park, E.K. Secretome analysis of human BMSCs and identification of SMOC1 as an important ECM protein in osteoblast differentiation. J. Proteome Res. 2010, 9, 2946–2956. [Google Scholar] [CrossRef]
- Kusuma, G.D.; Carthew, J.; Lim, R.; Frith, J.E. Effect of the microenvironment on mesenchymal stem cell paracrine signaling: Opportunities to engineer the therapeutic effect. Stem Cells Dev. 2017, 26, 617–631. [Google Scholar] [CrossRef] [PubMed]
- Alt, E.; Yan, Y.; Gehmert, S.; Song, Y.H.; Altman, A.; Gehmert, S.; Vykoukal, D.; Bai, X. Fibroblasts share mesenchymal phenotypes with stem cells, but lack their differentiation and colony-forming potential. Biol. Cell 2011, 103, 197–208. [Google Scholar] [CrossRef] [PubMed]
- Holmes, C.; Stanford, W.L. Concise review: Stem cell antigen-1: Expression, function, and enigma. Stem Cells 2007, 25, 1339–1347. [Google Scholar] [CrossRef] [PubMed]
- D’Ippolito, G.; Howard, G.A.; Roos, B.A.; Schillerc, P.C. Sustained stromal stem cell self-renewal and osteoblastic differentiation during aging. Rejuvenation Res. 2006, 9, 10–19. [Google Scholar] [CrossRef] [PubMed]
- Corselli, M.; Chen, C.-W.; Sun, B.; Yap, S.; Rubin, J.P.; Péault, B. The tunica adventitia of human arteries and veins as a source of mesenchymal stem cells. Stem Cells Dev. 2012, 21, 1299–1308. [Google Scholar] [CrossRef]
- de Souza, L.E.B.; Malta, T.M.; Kashima Haddad, S.; Covas, D.T. Mesenchymal stem cells and pericytes: To what extent are they related? Stem Cells Dev. 2016, 25, 1843–1852. [Google Scholar] [CrossRef] [PubMed]
- Ilmer, M.; Vykoukal, J.; Boiles, A.R.; Coleman, M.; Alt, E. Two sides of the same coin: Stem cells in cancer and regenerative medicine. FASEB J. 2014, 28, 2748–2761. [Google Scholar] [CrossRef]
- Alt, E.U.; Winnier, G.; Haenel, A.; Rothoerl, R.; Solakoglu, O.; Alt, C.; Schmitz, C. Towards a comprehensive understanding of UA-ADRCs (uncultured, autologous, fresh, unmodified, adipose derived regenerative cells, isolated at point of care) in regenerative medicine. Cells 2020, 9, 1097. [Google Scholar] [CrossRef]
- Willert, K.; Brown, J.D.; Danenberg, E.; Duncan, A.W.; Weissman, I.L.; Reya, T.; Yates, J.R.; Nusse, R. Wnt proteins are lipid-modified and can act as stem cell growth factors. Nature 2003, 423, 448–452. [Google Scholar] [CrossRef] [PubMed]
- Coudreuse, D.; Korswagen, H.C. The making of Wnt: New insights into Wnt maturation, sorting and secretion. Development 2007, 134, 3–12. [Google Scholar] [CrossRef] [PubMed]
- Neth, P.; Ries, C.; Karow, M.; Egea, V.; Ilmer, M.; Jochum, M. The Wnt signal transduction pathway in stem cells and cancer cells: Influence on cellular invasion. Stem Cell Rev. 2007, 3, 18–29. [Google Scholar] [CrossRef] [PubMed]
- Bourin, P.; Bunnell, B.A.; Casteilla, L.; Dominici, M.; Katz, A.J.; March, K.L.; Redl, H.; Rubin, J.P.; Yoshimura, K.; Gimble, J.M. Stromal cells from the adipose tissue-derived stromal vascular fraction and culture expanded adipose tissue-derived stromal/stem cells: A joint statement of the International Federation for Adipose Therapeutics and Science (IFATS) and the International Society for Cellular Therapy (ISCT). Cytotherapy 2013, 15, 641–648. [Google Scholar] [PubMed]
- Rezza, A.; Sennett, R.; Rendl, M. Adult stem cell niches: Cellular and molecular components. Curr. Top. Dev. Biol. 2014, 107, 333–372. [Google Scholar]
- Dong, L.; Hao, H.; Han, W.; Fu, X. The Role of the microenvironment on the fate of adult stem cells. Sci. China Life Sci. 2015, 58, 639–648. [Google Scholar] [CrossRef]
- Wabik, A.; Jones, P.H. Switching roles: The functional plasticity of adult tissue stem cells. EMBO J. 2015, 34, 1164–1179. [Google Scholar] [CrossRef]
- Ader, M.; Tanaka, E.M. Modeling human development in 3D culture. Curr. Opin. Cell Biol. 2014, 31, 23–28. [Google Scholar] [CrossRef]
- Izadpanah, R.; Trygg, C.; Patel, B.; Kriedt, C.; Dufour, J.; Gimble, J.M.; Bunnell, B.A. Biologic properties of mesenchymal stem cells derived from bone marrow and adipose tissue. J. Cell. Biochem. 2006, 99, 1285–1297. [Google Scholar] [CrossRef]
- Mitchell, J.B.; McIntosh, K.; Zvonic, S.; Garrett, S.; Floyd, Z.E.; Kloster, A.; Di Halvorsen, Y.; Storms, R.W.; Goh, B.; Kilroy, G.; et al. Immunophenotype of human adipose-derived cells: Temporal changes in stromal-associated and stem cell-associated markers. Stem Cells 2006, 24, 376–385. [Google Scholar] [CrossRef]
- Valente, S.; Alviano, F.; Ciavarella, C.; Buzzi, M.; Ricci, F.; Tazzari, P.; Pagliaro, P.; Pasquinelli, G. Human cadaver multipotent stromal/stem cells isolated from arteries stored in liquid nitrogen for 5 years. Stem Cell Res. Ther. 2014, 5, 8. [Google Scholar] [CrossRef]
- Bai, X.; Sadat, S.; Gehmert, S.; Alt, E.; Song, Y.H. VEGF Receptor Flk-1 plays an important role in c-Kit expression in adipose tissue derived stem cells. FEBS Lett. 2007, 581, 4681–4684. [Google Scholar] [CrossRef] [PubMed]
- Metzele, R.; Alt, C.; Bai, X.; Yan, Y.; Zhang, Z.; Pan, Z.; Coleman, M.; Vykoukal, J.; Song, Y.; Alt, E. Human adipose tissue-derived stem cells exhibit proliferation potential and spontaneous rhythmic contraction after fusion with neonatal rat cardiomyocytes. FASEB J. 2011, 25, 830–839. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Belema Bedada, F.; Technau, A.; Ebelt, H.; Schulze, M.; Braun, T. Activation of myogenic differentiation pathways in adult bone marrow-derived stem cells. Mol. Cell. Biol. 2005, 25, 9509–9519. [Google Scholar] [CrossRef]
- Haenel, A.; Ghosn, M.; Karimi, T.; Vykoukal, J.; Shah, D.; Valderrabano, M.; Schulz, D.G.; Raizner, A.; Schmitz, C.; Alt, E.U. Unmodified autologous stem cells at point of care for chronic myocardial infarction. World J. Stem Cells 2019, 11, 831–858. [Google Scholar] [CrossRef]
- Yukawa, H.; Mizufune, S.; Mamori, C.; Kagami, Y.; Oishi, K.; Kaji, N.; Okamoto, Y.; Takeshi, M.; Noguchi, H.; Baba, Y.; et al. Quantum dots for labeling adipose tissue-derived stem cells. Cell Transpl. 2009, 18, 591–600. [Google Scholar] [CrossRef]
- Brinkley, B.R.; Beall, P.T.; Wible, L.J.; Mace, M.L.; Turner, D.S.; Cailleau, R.M. Variations in cell form and cytoskeleton in human breast carcinoma cells in vitro. Cancer Res. 1980, 40, 3118–3129. [Google Scholar] [PubMed]
- Koyanagi, M.; Brandes, R.P.; Haendeler, J.; Zeiher, A.M.; Dimmeler, S. Cell-to-cell connection of endothelial progenitor cells with cardiac myocytes by nanotubes: A novel mechanism for cell fate changes? Circ. Res. 2005, 96, 1039–1041. [Google Scholar] [CrossRef]
- Valadi, H.; Ekström, K.; Bossios, A.; Sjöstrand, M.; Lee, J.J.; Lötvall, J.O. Exosome-mediated transfer of mRNAs and microRNAs is a novel mechanism of genetic exchange between cells. Nat. Cell Biol. 2007, 9, 654–659. [Google Scholar] [CrossRef] [PubMed]
- Xin, H.; Li, Y.; Buller, B.; Katakowski, M.; Zhang, Y.; Wang, X.; Shang, X.; Zhang, Z.G.; Chopp, M. Exosome-mediated transfer of miR-133b from multipotent mesenchymal stromal cells to neural cells contributes to neurite outgrowth. Stem Cells 2012, 30, 1556–1564. [Google Scholar] [CrossRef] [PubMed]
- Ismail, N.; Wang, Y.; Dakhlallah, D.; Moldovan, L.; Agarwal, K.; Batte, K.; Shah, P.; Wisler, J.; Eubank, T.D.; Tridandapani, S.; et al. Macrophage microvesicles induce macrophage differentiation and miR-223 transfer. Blood 2013, 121, 984–995. [Google Scholar] [CrossRef] [PubMed]
- Kruger, S.; Elmageed, Z.Y.A.; Hawke, D.H.; Wörner, P.M.; Jansen, D.A.; Abdel-Mageed, A.B.; Alt, E.U.; Izadpanah, R. Molecular characterization of exosome-like vesicles from breast cancer cells. BMC Cancer 2014, 14, 44. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.Y.; Chen, G.H.; Yang, Y.J. Exosomes: A rising star in failing hearts. Front. Physiol. 2017, 8, 494. [Google Scholar] [CrossRef] [PubMed]
- González, M.A.; Gonzalez–Rey, E.; Rico, L.; Büscher, D.; Delgado, M. Adipose-derived mesenchymal stem cells alleviate experimental colitis by inhibiting inflammatory and autoimmune responses. Gastroenterology 2009, 136, 978–989. [Google Scholar] [CrossRef]
- Leto Barone, A.A.; Khalifian, S.; Lee, W.P.A.; Brandacher, G. Immunomodulatory effects of adipose-derived stem cells: Fact or fiction? BioMed Res. Int. 2013, 2013, 383685. [Google Scholar] [CrossRef] [PubMed]
- Caruso, S.; Poon, I.K.H. Apoptotic cell-derived extracellular vesicles: More than just debris. Front. Immunol. 2018, 9, 1486. [Google Scholar] [CrossRef]
- Galleu, A.; Riffo-Vasquez, Y.; Trento, C.; Lomas, C.; Dolcetti, L.; Cheung, T.S.; von Bonin, M.; Barbieri, L.; Halai, K.; Ward, S.; et al. Apoptosis in mesenchymal stromal cells induces in vivo recipient-mediated immunomodulation. Sci. Transl. Med. 2017, 9, eaam7828. [Google Scholar] [CrossRef]
- Hochreiter-Hufford, A.; Ravichandran, K.S. Clearing the dead: Apoptotic cell sensing, recognition, engulfment, and digestion. Cold Spring Harb. Perspect. Biol. 2013, 5, a008748. [Google Scholar] [CrossRef]
- Lauber, K.; Bohn, E.; Kröber, S.M.; Xiao, Y.; Blumenthal, S.G.; Lindemann, R.K.; Marini, P.; Wiedig, C.; Zobywalski, A.; Baksh, S.; et al. Apoptotic cells induce migration of phagocytes via caspase-3-mediated release of a lipid attraction signal. Cell 2003, 113, 717–730. [Google Scholar] [CrossRef]
- Fuchs, B.; Schiller, J.; Cross, M.A. Apoptosis-associated changes in the glycerophospholipid composition of hematopoietic progenitor cells monitored by 31P NMR spectroscopy and MALDI-TOF mass spectrometry. Chem. Phys. Lipids 2007, 150, 229–238. [Google Scholar] [CrossRef]
- Rakic, P. DNA Synthesis and cell division in the adult primate brain. Ann. N. Y. Acad. Sci. 1985, 457, 193–211. [Google Scholar] [CrossRef]
- Breunig, J.J.; Arellano, J.I.; Macklis, J.D.; Rakic, P. Everything that glitters isn’t gold: A critical review of postnatal neural precursor analyses. Cell Stem Cell 2007, 1, 612–627. [Google Scholar] [CrossRef] [PubMed]
- Jellinger, K.A.; Attems, J. Neuropathological approaches to cerebral aging and neuroplasticity. Dialogues Clin. Neurosci. 2013, 15, 29–43. [Google Scholar]
- Demidova-Rice, T.N.; Hamblin, M.R.; Herman, I.M. Acute and impaired wound healing: Pathophysiology and current methods for drug delivery, part 1: Normal and chronic wounds: Biology, causes, and approaches to care. Adv. Skin Wound Care 2012, 25, 304–314. [Google Scholar] [CrossRef] [PubMed]
- McCusker, C.; Gardiner, D.M. The axolotl model for regeneration and aging research: A mini-review. Gerontology 2011, 57, 565–571. [Google Scholar] [CrossRef] [PubMed]
- Farah, Z.; Fan, H.; Liu, Z.; He, J.Q. A concise review of common animal models for the study of limb regeneration. Organogenesis 2016, 12, 109–118. [Google Scholar] [CrossRef] [PubMed]
- Grillo, M.; Konstantinides, N.; Averof, M. Old questions, new models: Unraveling complex organ regeneration with new experimental approaches. Curr. Opin. Genet. Dev. 2016, 40, 23–31. [Google Scholar] [CrossRef]
- Zielins, E.R.; Ransom, R.C.; Leavitt, T.E.; Longaker, M.T.; Wan, D.C. The role of stem cells in limb regeneration. Organogenesis 2016, 12, 16–27. [Google Scholar] [CrossRef] [PubMed]
- Satoh, A.; Mitogawa, K.; Makanae, A. Regeneration inducers in limb regeneration. Dev. Growth Differ. 2015, 57, 421–429. [Google Scholar] [CrossRef] [PubMed]
- Haas, B.J.; Whited, J.L. Advances in decoding axolotl limb regeneration. Trends Genet. 2017, 33, 553–565. [Google Scholar] [CrossRef]
- Jullien, J.; Pasque, V.; Halley-Stott, R.P.; Miyamoto, K.; Gurdon, J.B. Mechanisms of nuclear reprogramming by eggs and oocytes: A deterministic process? Nat. Rev. Mol. Cell Biol. 2011, 12, 453–459. [Google Scholar] [CrossRef]
- Heffernan, C.; Sumer, H.; Verma, P.J. Generation of clinically relevant "induced pluripotent stem" (iPS) cells. J. Stem Cells 2011, 6, 109–127. [Google Scholar] [PubMed]
- Shi, Y.; Inoue, H.; Wu, J.C.; Yamanaka, S. Induced pluripotent stem cell technology: A decade of progress. Nat. Rev. Drug Discov. 2017, 16, 115–130. [Google Scholar] [CrossRef]
- Takahashi, K.; Tanabe, K.; Ohnuki, M.; Narita, M.; Ichisaka, T.; Tomoda, K.; Yamanaka, S. Induction of pluripotent stem cells from adult human fibroblasts by defined factors. Cell 2007, 131, 861–872. [Google Scholar] [CrossRef]
- Yu, J.; Vodyanik, M.A.; Smuga-Otto, K.; Antosiewicz-Bourget, J.; Frane, J.L.; Tian, S.; Nie, J.; Jonsdottir, G.A.; Ruotti, V.; Stewart, R.; et al. Induced pluripotent stem cell lines derived from human somatic cells. Science 2007, 318, 1917–1920. [Google Scholar] [CrossRef] [PubMed]
- Lee, A.S.; Tang, C.; Rao, M.S.; Weissman, I.L.; Wu, J.C. Tumorigenicity as a clinical hurdle for pluripotent stem cell therapies. Nat. Med. 2013, 19, 998–1004. [Google Scholar] [CrossRef] [PubMed]
- Riggs, J.W.; Barrilleaux, B.L.; Varlakhanova, N.; Bush, K.M.; Chan, V.; Knoepfler, P.S. Induced pluripotency and oncogenic transformation are related processes. Stem Cells Dev. 2013, 22, 37–50. [Google Scholar] [CrossRef] [PubMed]
- Steinemann, D.; Göhring, G.; Schlegelberger, B. Genetic instability of modified stem cells-a first step towards malignant transformation? Am. J. Stem Cells 2013, 2, 39–51. [Google Scholar] [PubMed]
- Tung, P.-Y.; Knoepfler, P.S. Epigenetic mechanisms of tumorigenicity manifesting in stem cells. Oncogene 2015, 34, 2288–2296. [Google Scholar] [CrossRef]
- Lee, A.S.; Tang, C.; Hong, W.X.; Park, S.; Bazalova-Carter, M.; Nelson, G.; Sanchez-Freire, V.; Bakerman, I.; Zhang, W.; Neofytou, E.; et al. Brief report: External beam radiation therapy for the treatment of human pluripotent stem cell-derived teratomas. Stem Cells 2017, 35, 1994–2000. [Google Scholar] [CrossRef]
- Marsoner, F.; Koch, P.; Ladewig, J. Cortical organoids: Why all this hype? Curr. Opin. Genet. Dev. 2018, 52, 22–28. [Google Scholar] [CrossRef]
- Murphy, M.B.; Moncivais, K.; Caplan, A.I. Mesenchymal stem cells: Environmentally responsive therapeutics for regenerative medicine. Exp. Mol. Med. 2013, 45, e54. [Google Scholar] [CrossRef]
- Al-Drees, M.A.; Yeo, J.H.; Boumelhem, B.B.; Antas, V.I.; Brigden, K.W.L.; Colonne, C.K.; Fraser, S.T. Making blood: The haematopoietic niche throughout ontogeny. Stem Cells Int. 2015, 2015, 571893. [Google Scholar] [CrossRef] [PubMed]
- Yoshimura, K.; Shigeura, T.; Matsumoto, D.; Sato, T.; Takaki, Y.; Aiba-Kojima, E.; Sato, K.; Inoue, K.; Nagase, T.; Koshima, I.; et al. Characterization of freshly isolated and cultured cells derived from the fatty and fluid portions of liposuction aspirates. J. Cell. Physiol. 2006, 208, 64–76. [Google Scholar] [CrossRef] [PubMed]
- Schäffler, A.; Büchler, C. Concise review: Adipose tissue-derived stromal cells-basic and clinical implications for novel cell-based therapies. Stem Cells 2007, 25, 818–827. [Google Scholar] [CrossRef] [PubMed]
- Gimble, J.M.; Guilak, F.; Bunnell, B.A. Clinical and preclinical translation of cell-based therapies using adipose tissue-derived cells. Stem Cell Res. Ther. 2010, 1, 19. [Google Scholar] [CrossRef]
- Altman, A.M.; Khalek, F.J.A.; Alt, E.U.; Butler, C.E. Adipose tissue–derived stem cells enhance bioprosthetic mesh repair of ventral hernias. Plast. Reconstr. Surg. 2010, 126, 845–854. [Google Scholar] [CrossRef]
- Gir, P.; Oni, G.; Brown, S.A.; Mojallal, A.; Rohrich, R.J. Human adipose stem cells: Current clinical applications. Plast. Reconstr. Surg. 2012, 129, 1277–1290. [Google Scholar] [CrossRef]
- Klein, S.M.; Vykoukal, J.; Li, D.P.; Pan, H.L.; Zeitler, K.; Alt, E.; Geis, S.; Felthaus, O.; Prantl, L. Peripheral motor and sensory nerve conduction following transplantation of undifferentiated autologous adipose tissue-derived stem cells in a biodegradable U.S. Food and Drug Administration-approved nerve conduit. Plast. Reconstr. Surg. 2016, 138, 132–139. [Google Scholar] [CrossRef] [PubMed]
- Oberbauer, E.; Steffenhagen, C.; Wurzer, C.; Gabriel, C.; Redl, H.; Wolbank, S. Enzymatic and non-enzymatic isolation systems for adipose tissue-derived cells: Current state of the art. Cell Regen. 2015, 4, 7. [Google Scholar] [CrossRef]
- Condé-Green, A.; Kotamarti, V.S.; Sherman, L.S.; Keith, J.D.; Lee, E.S.; Granick, M.S.; Rameshwar, P. Shift toward mechanical isolation of adipose-derived stromal vascular fraction: Review of upcoming techniques. Plast. Reconstr. Surg. Glob. Open 2016, 4, e1017. [Google Scholar] [CrossRef]
- van Dongen, J.A.; Tuin, A.J.; Spiekman, M.; Jansma, J.; van der Lei, B.; Harmsen, M.C. Comparison of intraoperative procedures for isolation of clinical grade stromal vascular fraction for regenerative purposes: A systematic review: Intraoperative procedures for stromal vascular fraction isolation. J. Tissue Eng. Regen. Med. 2018, 12, e261–e274. [Google Scholar] [CrossRef] [PubMed]
- Metcalf, G.L.; McClure, S.R.; Hostetter, J.M.; Martinez, R.F.; Wang, C. Evaluation of adipose-derived stromal vascular fraction from the lateral tailhead, inguinal region, and mesentery of horses. Can. J. Vet. Res. 2016, 80, 294–301. [Google Scholar]
- Aronowitz, J.A.; Ellenhorn, J.D.I. Adipose stromal vascular fraction isolation: A head-to-head comparison of four commercial cell separation systems. Plast. Reconstr. Surg. 2013, 132, 932e–939e. [Google Scholar] [CrossRef] [PubMed]
- Aronowitz, J.A.; Lockhart, R.A.; Hakakian, C.S.; Birnbaum, Z.E. Adipose stromal vascular fraction isolation: A head-to-head comparison of 4 cell separation systems #2. Ann. Plast. Surg. 2016, 77, 354–362. [Google Scholar] [PubMed]
- de Almeida, P.E.; van Rappard, J.R.M.; Wu, J.C. In vivo bioluminescence for tracking cell fate and function. Am. J. Physiol. Heart Circ. Physiol. 2011, 301, H663–H671. [Google Scholar] [CrossRef] [PubMed]
- Scarfe, L.; Brillant, N.; Kumar, J.D.; Ali, N.; Alrumayh, A.; Amali, M.; Barbellion, S.; Jones, V.; Niemeijer, M.; Potdevin, S.; et al. Preclinical imaging methods for assessing the safety and efficacy of regenerative medicine therapies. NPJ Regen. Med. 2017, 2, 28. [Google Scholar] [CrossRef]
- Bai, X.; Yan, Y.; Coleman, M.; Wu, G.; Rabinovich, B.; Seidensticker, M.; Alt, E. Tracking long-term survival of intramyocardially delivered human adipose tissue-derived stem cells using bioluminescence imaging. Mol. Imaging Biol. 2011, 13, 633–645. [Google Scholar] [CrossRef]
- Sadat, S.; Gehmert, S.; Song, Y.-H.; Yen, Y.; Bai, X.; Gaiser, S.; Klein, H.; Alt, E. The cardioprotective effect of mesenchymal stem cells is mediated by IGF-I and VEGF. Biochem. Biophys. Res. Commun. 2007, 363, 674–679. [Google Scholar] [CrossRef]
- Rothoerl, R.; Alt, C.; Preuss, A.; Mueller, C.; Lackermeier, P.; Alt, E. Clinical results of adipose derived stem cell injection for facet joint syndrome. In Proceedings of the IFATS San Diego 2016 Conference Proceedings, San Diego, CA, USA, 17–20 November 2016; p. 79. Available online: https://www.ifats.org/assets/docs/IFATS-Program-Book-2016-FINAL-11.23.16.pdf (accessed on 6 August 2021).
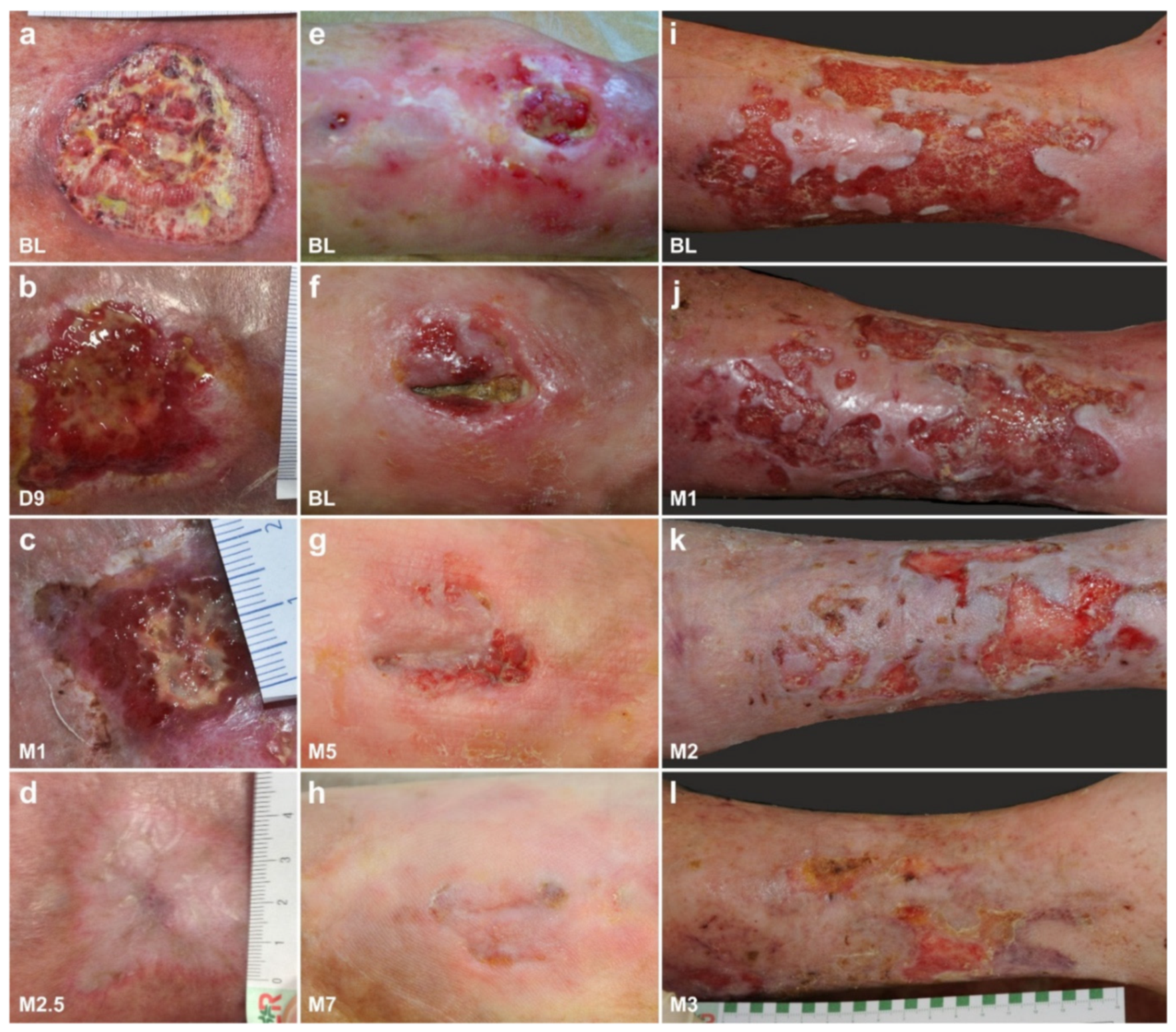
- Konstantinow, A.; Arnold, A.; Djabali, K.; Kempf, W.; Gutermuth, J.; Fischer, T.; Biedermann, T. Therapy of ulcus cruris of venous and mixed venous arterial origin with autologous, adult, native progenitor cells from subcutaneous adipose tissue: A prospective clinical pilot study. J. Eur. Acad. Dermatol. Venereol. 2017, 31, 2104–2118. [Google Scholar] [CrossRef]
- Hurd, J.L.; Facile, T.R.; Weiss, J.; Hayes, M.; Hayes, M.; Furia, J.P.; Maffulli, N.; Winnier, G.E.; Alt, C.; Schmitz, C.; et al. Safety and efficacy of treating symptomatic, partial-thickness rotator cuff tear with fresh, uncultured, unmodified, autologous adipose derived regenerative cells (UA-ADRCs) isolated at the point of care: A prospective, randomized, controlled first-in-human pilot study. J. Orthop. Surg. Res. 2020, 15, 122. [Google Scholar]
- Flier, J.S.; Underhill, L.H.; Dvorak, H.F. Tumors: Wounds that do not heal. N. Engl. J. Med. 1986, 315, 1650–1659. [Google Scholar] [CrossRef]
- Dvorak, H.F. Tumors: Wounds that do not heal—Redux. Cancer Immunol. Res. 2015, 3, 1–11. [Google Scholar] [CrossRef]
- Gehmert, S.; Gehmert, S.; Prantl, L.; Vykoukal, J.; Alt, E.; Song, Y.-H. Breast cancer cells attract the migration of adipose tissue-derived stem cells via the PDGF-BB/PDGFR-β signaling pathway. Biochem. Biophys. Res. Commun. 2010, 398, 601–605. [Google Scholar] [CrossRef]
- Altman, A.M.; Yan, Y.; Matthias, N.; Bai, X.; Rios, C.; Mathur, A.B.; Song, Y.H.; Alt, E.U. IFATS collection: Human adipose-derived stem cells seeded on a silk fibroin-chitosan scaffold enhance wound repair in a murine soft tissue injury model. Stem Cells 2009, 27, 250–258. [Google Scholar] [CrossRef] [PubMed]
- Angst, F.; Schwyzer, H.K.; Aeschlimann, A.; Simmen, B.R.; Goldhahn, J. Measures of adult shoulder function: Disabilities of the Arm, Shoulder, and Hand Questionnaire (DASH) and its short version (QuickDASH), Shoulder Pain and Disability Index (SPADI), American Shoulder and Elbow Surgeons (ASES) Society standardized shoulder assessment form, Constant (Murley) Score (CS), Simple Shoulder Test (SST), Oxford Shoulder Score (OSS), Shoulder Disability Questionnaire (SDQ), and Western Ontario Shoulder Instability Index (WOSI). Arthr. Care Res. 2011, 63 (Suppl. 11), S174–S188. [Google Scholar]
- Hurd, J. Autologous Adult Adipose-Derived Regenerative Cell Injection into Chronic Partial-Thickness Rotator Cuff Tears. Available online: https://www.clinicaltrials.gov/ct2/show/NCT03752827 (accessed on 6 August 2021).
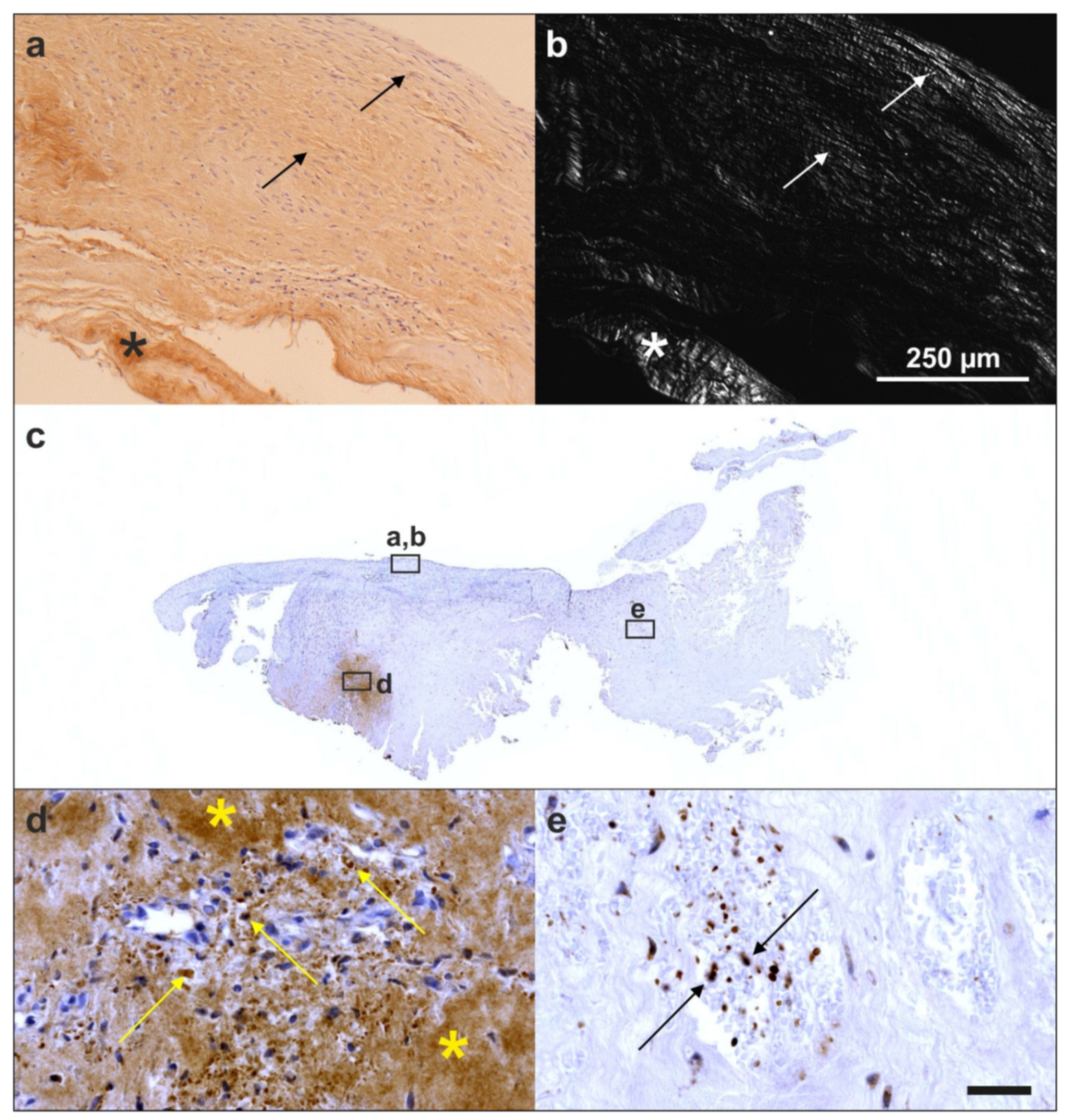
- Alt, E.U.; Rothoerl, R.; Hoppert, M.; Frank, H.G.; Wuerfel, T.; Alt, C.; Schmitz, C. First immunohistochemical evidence of human tendon repair following stem cell injection: A case report and review of literature. World J. Stem Cells 2021, 13, 944–970. [Google Scholar] [CrossRef] [PubMed]
- Shukunami, C.; Oshima, Y.; Hiraki, Y. Molecular Cloning of Tenomodulin, a Novel Chondromodulin-I Related Gene. Biochem. Biophys. Res. Commun. 2001, 280, 1323–1327. [Google Scholar] [CrossRef]
- Brandau, O.; Meindl, A.; Fässler, R.; Aszódi, A. A novel gene, Tendin, is strongly expressed in tendons and ligaments and shows high homology with Chondromodulin-I: Tendin and Chondromodulin-I. Dev. Dyn. 2001, 221, 72–80. [Google Scholar] [CrossRef] [PubMed]
- Dex, S.; Lin, D.; Shukunami, C.; Docheva, D. TENOgenic MODULating INsider factor: Systematic assessment on the functions of tenomodulin gene. Gene 2016, 587, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Jung, C.K. Articular cartilage: Histology and physiology. In Techniques in Cartilage Repair Surgery; Shetty, A.A., Kim, S.J., Nakamura, N., Brittberg, M., Eds.; Springer: Berlin, Germany, 2014; pp. 17–21. [Google Scholar]
- Rieppo, J.; Hallikainen, J.; Jurvelin, J.S.; Kiviranta, I.; Helminen, H.J.; Hyttinen, M.M. Practical considerations in the use of polarized light microscopy in the analysis of the collagen network in articular cartilage. Microsc. Res. Tech. 2008, 71, 279–287. [Google Scholar] [CrossRef] [PubMed]
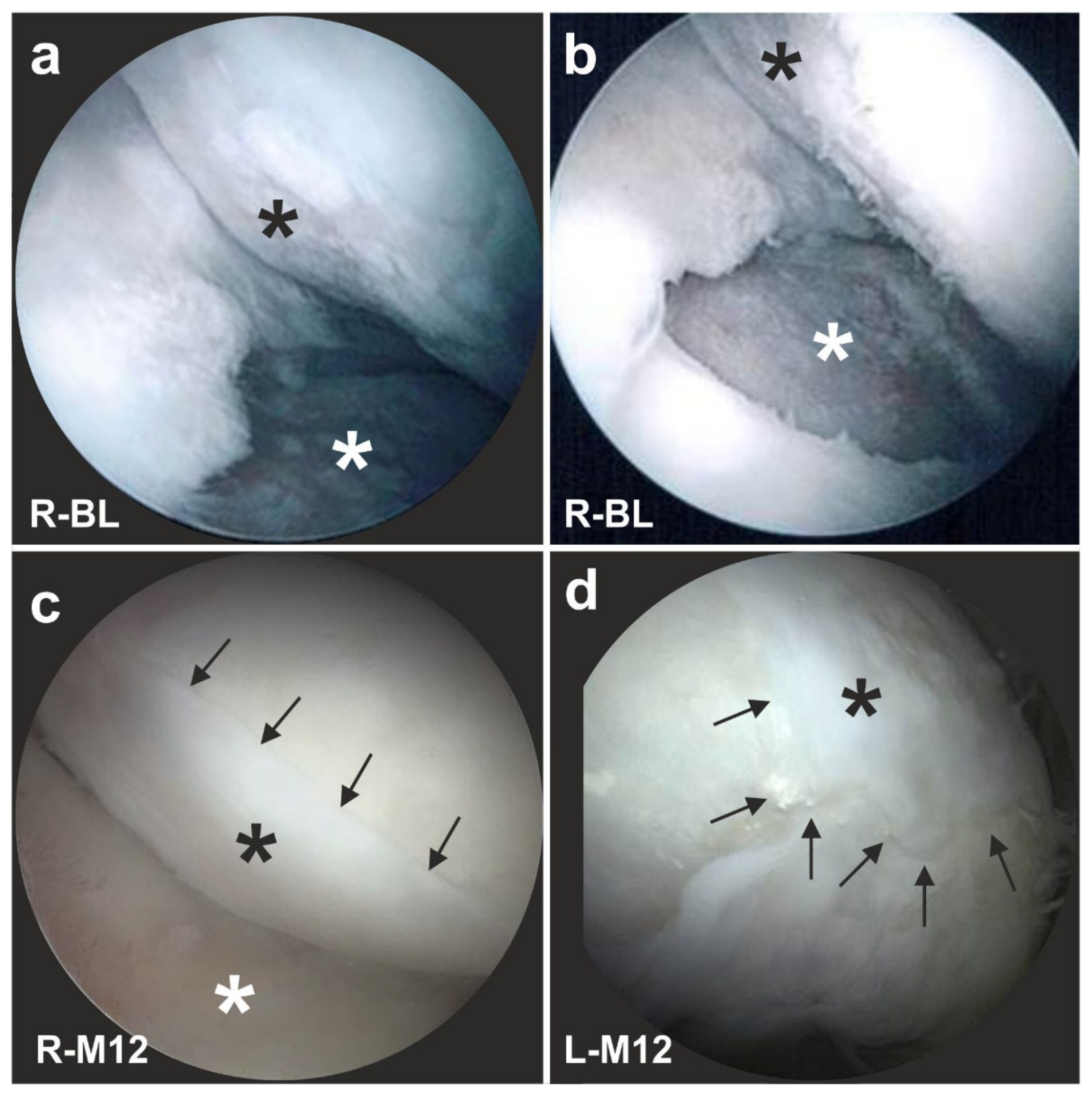
- Damia, E.; Chicharro, D.; Lopez, S.; Cuervo, B.; Rubio, M.; Sopena, J.; Vilar, J.; Carrillo, J. Adipose-derived mesenchymal stem cells: Are they a good therapeutic strategy for osteoarthritis? Int. J. Mol. Sci. 2018, 19, 1926. [Google Scholar] [CrossRef] [PubMed]
- Jo, C.H.; Lee, Y.G.; Shin, W.H.; Kim, H.; Chai, J.W.; Jeong, E.C.; Kim, J.E.; Shim, H.; Shin, J.S.; Shin, I.S.; et al. Intra-articular injection of mesenchymal stem cells for the treatment of osteoarthritis of the knee: A proof-of-concept clinical trial: Ia injection of MSCs for knee osteoarthritis. Stem Cells 2014, 32, 1254–1266. [Google Scholar] [CrossRef]
- Pers, Y.-M.; Rackwitz, L.; Ferreira, R.; Pullig, O.; Delfour, C.; Barry, F.; Sensebe, L.; Casteilla, L.; Fleury, S.; Bourin, P.; et al. Adipose mesenchymal stromal cell-based therapy for severe osteoarthritis of the knee: A phase I dose-escalation trial: ASCs for severe OA of the knee. Stem Cells Transl. Med. 2016, 5, 847–856. [Google Scholar] [CrossRef]
- Freitag, J.; Li, D.; Wickham, J.; Shah, K.; Tenen, A. Effect of autologous adipose-derived mesenchymal stem cell therapy in the treatment of a post-traumatic chondral defect of the knee. BMJ Case Rep. 2017, 2017, bcr2017220852. [Google Scholar] [CrossRef]
- Alexander, C.E.; Sandean, D.P.; Varacallo, M. Lumbosacral facet syndrome. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2021. [Google Scholar]
- Boetel, T. Safety of Adipose-Derived Regenerative Cells Injection for Treatment of Osteoarthritis of the Facet Joint. Available online: https://www.clinicaltrials.gov/ct2/show/NCT03513731 (accessed on 26 July 2021).
- Solakoglu, Ö.; Götz, W.; Kiessling, M.C.; Alt, C.; Schmitz, C.; Alt, E.U. Improved guided bone regeneration by combined application of unmodified, fresh autologous adipose derived regenerative cells and plasma rich in growth factors: A first-in-human case report and literature review. World J. Stem Cells 2019, 11, 124–146. [Google Scholar] [CrossRef] [PubMed]
- Larson, E.; Jones, L.C.; Goodman, S.B.; Koo, K.-H.; Cui, Q. Early-stage osteonecrosis of the femoral head: Where are we and where are we going in year 2018? Int. Orthop. 2018, 42, 1723–1728. [Google Scholar] [CrossRef]
- Andriolo, L.; Merli, G.; Tobar, C.; Altamura, S.A.; Kon, E.; Filardo, G. Regenerative therapies increase survivorship of avascular necrosis of the femoral head: A systematic review and meta-analysis. Int. Orthop. 2018, 42, 1689–1704. [Google Scholar] [CrossRef]
- Pak, J.; Lee, J.H.; Jeon, J.H.; Lee, S.H. Complete resolution of avascular necrosis of the human femoral head treated with adipose tissue-derived stem cells and platelet-rich plasma. J. Int. Med. Res. 2014, 42, 1353–1362. [Google Scholar] [CrossRef]
- Sorice, S.; Rustad, K.C.; Li, A.Y.; Gurtner, G.C. The role of stem cell therapeutics in wound healing: Current understanding and future directions. Plast. Reconstr. Surg. 2016, 138, 31S–41S. [Google Scholar] [CrossRef] [PubMed]
- Altman, A.M.; Abdul Khalek, F.J.; Seidensticker, M.; Pinilla, S.; Yan, Y.; Coleman, M.; Song, Y.-H.; Butler, C.E.; Alt, E.U. Human tissue-resident stem cells combined with hyaluronic acid gel provide fibrovascular-integrated soft-tissue augmentation in a murine photoaged skin model. Plast. Reconstr. Surg. 2010, 125, 63–73. [Google Scholar] [CrossRef] [PubMed]
- Pourjafar, M.; Saidijam, M.; Etemadi, K.; Najafi, R. All-trans retinoic acid enhances in vitro mesenchymal stem cells migration by targeting matrix metalloproteinases 2 and 9. Biotechnol. Lett. 2017, 39, 1263–1268. [Google Scholar] [CrossRef] [PubMed]
- Master, Z.; Matthews, K.R.W.; Abou-el-Enein, M. Unproven stem cell interventions: A global public health problem requiring global deliberation. Stem Cell Rep. 2021, 16, 1435–1445. [Google Scholar] [CrossRef] [PubMed]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Alt, E.U.; Schmitz, C.; Bai, X. Perspective: Why and How Ubiquitously Distributed, Vascular-Associated, Pluripotent Stem Cells in the Adult Body (vaPS Cells) Are the Next Generation of Medicine. Cells 2021, 10, 2303. https://doi.org/10.3390/cells10092303
Alt EU, Schmitz C, Bai X. Perspective: Why and How Ubiquitously Distributed, Vascular-Associated, Pluripotent Stem Cells in the Adult Body (vaPS Cells) Are the Next Generation of Medicine. Cells. 2021; 10(9):2303. https://doi.org/10.3390/cells10092303
Chicago/Turabian StyleAlt, Eckhard U., Christoph Schmitz, and Xiaowen Bai. 2021. "Perspective: Why and How Ubiquitously Distributed, Vascular-Associated, Pluripotent Stem Cells in the Adult Body (vaPS Cells) Are the Next Generation of Medicine" Cells 10, no. 9: 2303. https://doi.org/10.3390/cells10092303
APA StyleAlt, E. U., Schmitz, C., & Bai, X. (2021). Perspective: Why and How Ubiquitously Distributed, Vascular-Associated, Pluripotent Stem Cells in the Adult Body (vaPS Cells) Are the Next Generation of Medicine. Cells, 10(9), 2303. https://doi.org/10.3390/cells10092303






