MiRNA Profiling in Plasma and Placenta of SARS-CoV-2-Infected Pregnant Women
Abstract
:1. Introduction
2. Methods
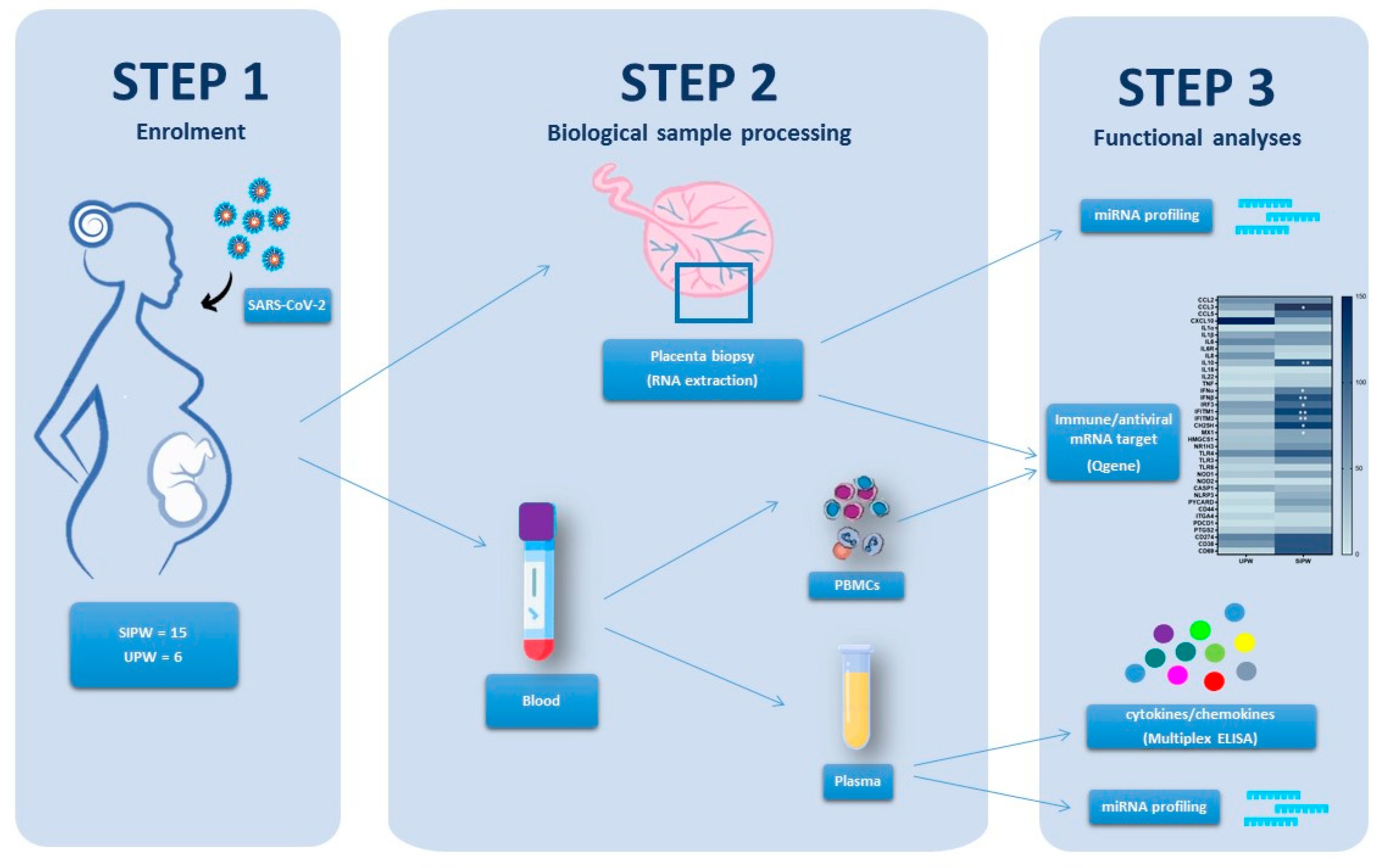
2.1. Study Population
2.2. Plasma, Peripheral Blood Mononuclear Cells (PBMCs) and Placenta Biopsy Collection
2.3. Total RNA and miRNA Extraction from Placenta Biopsies, PBMCs and Plasma
2.4. MicroRNA Reverse Transcription and Real-Time PCR Array Analysis
2.5. Quantigene Plex Gene Expression Assay
2.6. Cytokine and Chemokine Measurement by Multiplex Assay
2.7. Statistical Analyses
3. Results
3.1. Population
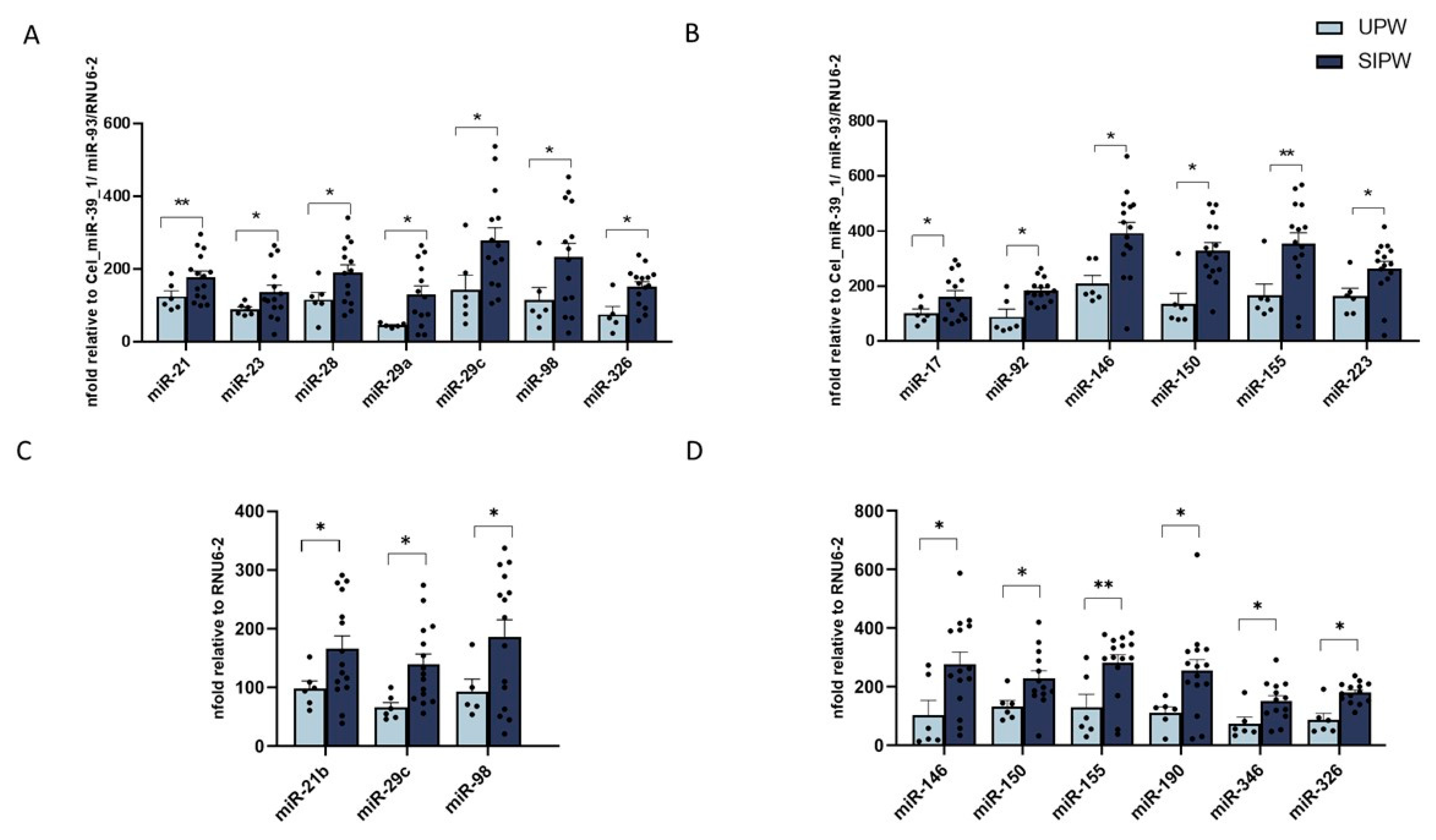
3.2. MiRNA Expression in Human Plasma from SIPW and UPW
3.3. MiRNA Expression in Human Placenta Biopsies from SIPW and UPW
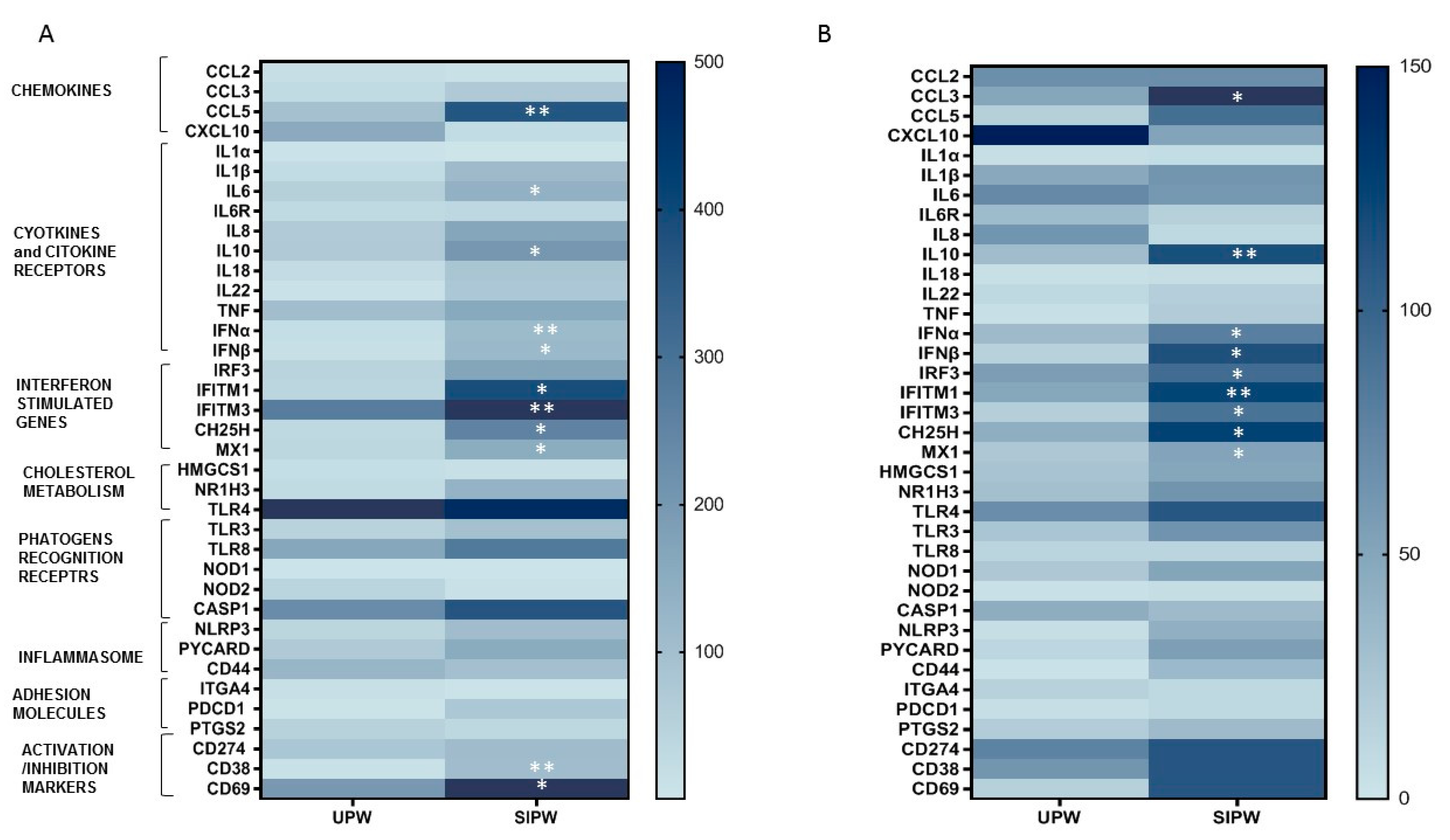
3.4. Gene Expression of Immune/Antiviral Selected Effectors in PBMCs and Placenta Biopsies from SIPW and UPW
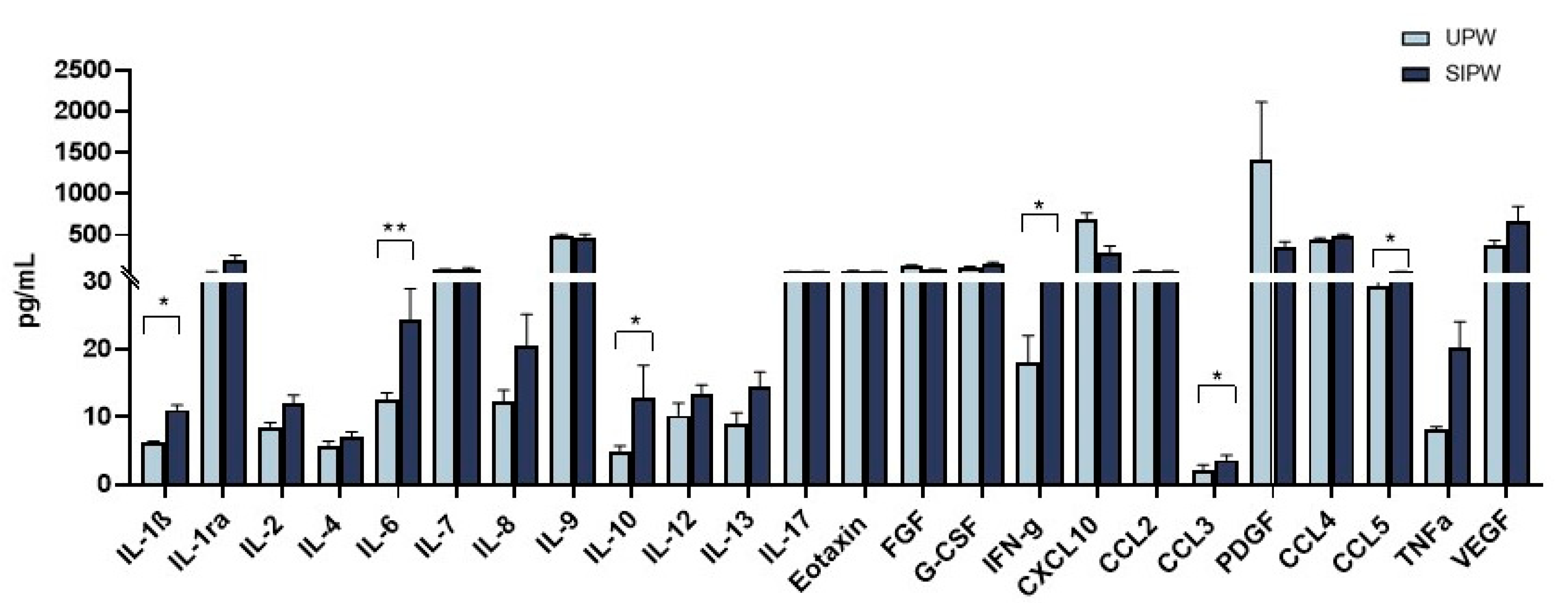
3.5. Modulation of Cytokine and Chemokine Production in Plasma from SIPW and UPW
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Robbins, J.R.; Bakardjiev, A.I. Pathogens and the Placental Fortress. Curr. Opin. Microbiol. 2012, 15, 36–43. [Google Scholar] [CrossRef] [Green Version]
- Heerema-McKenney, A. Defense and Infection of the Human Placenta. APMIS 2018, 126, 570–588. [Google Scholar] [CrossRef] [Green Version]
- Ander, S.E.; Diamond, M.S.; Coyne, C.B. Immune Responses at the Maternal-Fetal Interface. Sci. Immunol. 2019, 4. [Google Scholar] [CrossRef]
- Stegmann, B.J.; Carey, J.C. TORCH Infections. Toxoplasmosis, Other (Syphilis, Varicella-Zoster, Parvovirus B19), Rubella, Cytomegalovirus (CMV), and Herpes Infections. Curr. Womens Health Rep. 2002, 2, 253–258. [Google Scholar] [PubMed]
- Racicot, K.; Mor, G. Risks Associated with Viral Infections during Pregnancy. J. Clin. Investig. 2017, 127, 1591–1599. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fenizia, C.; Biasin, M.; Cetin, I.; Vergani, P.; Mileto, D.; Spinillo, A.; Gismondo, M.R.; Perotti, F.; Callegari, C.; Mancon, A.; et al. Analysis of SARS-CoV-2 Vertical Transmission during Pregnancy. Nat. Commun. 2020, 11, 5128. [Google Scholar] [CrossRef] [PubMed]
- Vivanti, A.J.; Vauloup-Fellous, C.; Prevot, S.; Zupan, V.; Suffee, C.; Do Cao, J.; Benachi, A.; De Luca, D. Transplacental Transmission of SARS-CoV-2 Infection. Nat. Commun. 2020, 11, 3572. [Google Scholar] [CrossRef]
- Kreis, N.-N.; Ritter, A.; Louwen, F.; Yuan, J. A Message from the Human Placenta: Structural and Immunomodulatory Defense against SARS-CoV-2. Cells 2020, 9, 1777. [Google Scholar] [CrossRef]
- He, L.; Hannon, G.J. MicroRNAs: Small RNAs with a Big Role in Gene Regulation. Nat. Rev. Genet. 2004, 5, 522–531. [Google Scholar] [CrossRef] [PubMed]
- Gottwein, E.; Cullen, B.R. Viral and Cellular MicroRNAs as Determinants of Viral Pathogenesis and Immunity. Cell Host Microbe 2008, 3, 375–387. [Google Scholar] [CrossRef] [Green Version]
- Chandan, K.; Gupta, M.; Sarwat, M. Role of Host and Pathogen-Derived MicroRNAs in Immune Regulation During Infectious and Inflammatory Diseases. Front. Immunol. 2019, 10, 3081. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yahyaei, S.; Biasin, M.; Saulle, I.; Gnudi, F.; De Luca, M.; Tasca, K.I.; Trabattoni, D.; Lo Caputo, S.; Mazzotta, F.; Clerici, M. Identification of a Specific MiRNA Profile in HIV-Exposed Seronegative Individuals. J. Acquir. Immune Defic. Syndr. 2016, 73, 11–19. [Google Scholar] [CrossRef] [Green Version]
- Marchi, R.; Sugita, B.; Centa, A.; Fonseca, A.S.; Bortoletto, S.; Fiorentin, K.; Ferreira, S.; Cavalli, L.R. The Role of MicroRNAs in Modulating SARS-CoV-2 Infection in Human Cells: A Systematic Review. Infect. Genet. Evol. 2021, 91, 104832. [Google Scholar] [CrossRef]
- Tang, H.; Gao, Y.; Li, Z.; Miao, Y.; Huang, Z.; Liu, X.; Xie, L.; Li, H.; Wen, W.; Zheng, Y.; et al. The Noncoding and Coding Transcriptional Landscape of the Peripheral Immune Response in Patients with COVID-19. Clin. Transl. Med. 2020, 10. [Google Scholar] [CrossRef] [PubMed]
- Testa, U.; Pelosi, E.; Castelli, G.; Labbaye, C. MiR-146 and MiR-155: Two Key Modulators of Immune Response and Tumor Development. Non-Coding RNA 2017, 3, 22. [Google Scholar] [CrossRef] [PubMed]
- Nejad, C.; Stunden, H.J.; Gantier, M.P. A Guide to MiRNAs in Inflammation and Innate Immune Responses. FEBS J. 2018, 285, 3695–3716. [Google Scholar] [CrossRef] [PubMed]
- Clerici, M.; Seminari, E.; Suter, F.; Castelli, F.; Pan, A.; Biasin, M.; Colombo, F.; Trabattoni, D.; Maggiolo, F.; Carosi, G.; et al. Different Immunologic Profiles Characterize HIV Infection in Highly Active Antiretroviral Therapy-Treated and Antiretroviral-Naïve Patients with Undetectable Viraemia. The Master Group. AIDS 2000, 14, 109–116. [Google Scholar] [CrossRef]
- De Gonzalo-Calvo, D.; Benítez, I.D.; Pinilla, L.; Carratalá, A.; Moncusí-Moix, A.; Gort-Paniello, C.; Molinero, M.; González, J.; Torres, G.; Bernal, M.; et al. Circulating MicroRNA Profiles Predict the Severity of COVID-19 in Hospitalized Patients. Transl. Res. 2021. [Google Scholar] [CrossRef]
- Donyavi, T.; Bokharaei-Salim, F.; Baghi, H.B.; Khanaliha, K.; Alaei Janat-Makan, M.; Karimi, B.; Sadri Nahand, J.; Mirzaei, H.; Khatami, A.; Garshasbi, S.; et al. Acute and Post-Acute Phase of COVID-19: Analyzing Expression Patterns of MiRNA-29a-3p, 146a-3p, 155-5p, and Let-7b-3p in PBMC. Int. Immunopharmacol. 2021, 97, 107641. [Google Scholar] [CrossRef]
- Jafarinejad-Farsangi, S.; Jazi, M.M.; Rostamzadeh, F.; Hadizadeh, M. High Affinity of Host Human MicroRNAs to SARS-CoV-2 Genome: An in Silico Analysis. Non-Coding RNA Res. 2020, 5, 222–231. [Google Scholar] [CrossRef]
- Nersisyan, S.; Engibaryan, N.; Gorbonos, A.; Kirdey, K.; Makhonin, A.; Tonevitsky, A. The Potential Role of MiR-21-3p in Coronavirus-Host Interplay. BioRxiv 2020. [Google Scholar] [CrossRef]
- Chen, Y.; Chen, J.; Wang, H.; Shi, J.; Wu, K.; Liu, S.; Liu, Y.; Wu, J. HCV-Induced MiR-21 Contributes to Evasion of Host Immune System by Targeting MyD88 and IRAK1. PLoS Pathog. 2013, 9, e1003248. [Google Scholar] [CrossRef] [PubMed]
- Ortega, P.A.S.; Saulle, I.; Mercurio, V.; Ibba, S.V.; Lori, E.M.; Fenizia, C.; Masetti, M.; Trabattoni, D.; Caputo, S.L.; Vichi, F.; et al. Interleukin 21 (IL-21)/MicroRNA-29 (MiR-29) Axis Is Associated with Natural Resistance to HIV-1 Infection. AIDS 2018, 32, 2453–2461. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yang, X.; Liang, Y.; Bamunuarachchi, G.; Xu, Y.; Vaddadi, K.; Pushparaj, S.; Xu, D.; Zhu, Z.; Blaha, R.; Huang, C.; et al. MiR-29a Is a Negative Regulator of Influenza Virus Infection through Targeting of the Frizzled 5 Receptor. Arch. Virol. 2021, 166, 363–373. [Google Scholar] [CrossRef]
- Xia, B.; Lu, J.; Wang, R.; Yang, Z.; Zhou, X.; Huang, P. MiR-21-3p Regulates Influenza A Virus Replication by Targeting Histone Deacetylase-8. Front. Cell. Infect. Microbiol. 2018, 8. [Google Scholar] [CrossRef] [Green Version]
- Wang, G.; Dong, F.; Xu, Z.; Sharma, S.; Hu, X.; Chen, D.; Zhang, L.; Zhang, J.; Dong, Q. MicroRNA Profile in HBV-Induced Infection and Hepatocellular Carcinoma. BMC Cancer 2017, 17, 805. [Google Scholar] [CrossRef] [Green Version]
- Leon-Icaza, S.A.; Zeng, M.; Rosas-Taraco, A.G. MicroRNAs in Viral Acute Respiratory Infections: Immune Regulation, Biomarkers, Therapy, and Vaccines. ExRNA 2019, 1, 1. [Google Scholar] [CrossRef] [Green Version]
- Matarese, A.; Gambardella, J.; Sardu, C.; Santulli, G. MiR-98 Regulates TMPRSS2 Expression in Human Endothelial Cells: Key Implications for COVID-19. Biomedicines 2020, 8, 462. [Google Scholar] [CrossRef]
- Taganov, K.D.; Boldin, M.P.; Chang, K.-J.; Baltimore, D. NF-KappaB-Dependent Induction of MicroRNA MiR-146, an Inhibitor Targeted to Signaling Proteins of Innate Immune Responses. Proc. Natl. Acad. Sci. USA 2006, 103, 12481–12486. [Google Scholar] [CrossRef] [Green Version]
- Rydyznski Moderbacher, C.; Ramirez, S.I.; Dan, J.M.; Grifoni, A.; Hastie, K.M.; Weiskopf, D.; Belanger, S.; Abbott, R.K.; Kim, C.; Choi, J.; et al. Antigen-Specific Adaptive Immunity to SARS-CoV-2 in Acute COVID-19 and Associations with Age and Disease Severity. Cell 2020, 183, 996–1012.e19. [Google Scholar] [CrossRef]
- Coperchini, F.; Chiovato, L.; Croce, L.; Magri, F.; Rotondi, M. The Cytokine Storm in COVID-19: An Overview of the Involvement of the Chemokine/Chemokine-Receptor System. Cytokine Growth Factor Rev. 2020, 53, 25–32. [Google Scholar] [CrossRef]
- Wang, H.; Zhang, Y.; Wu, X.; Wang, Y.; Cui, H.; Li, X.; Zhang, J.; Tun, N.; Peng, Y.; Yu, J. Regulation of Human Natural Killer Cell IFN-γ Production by MicroRNA-146a via Targeting the NF-ΚB Signaling Pathway. Front. Immunol. 2018, 9. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, T.; Ma, Y.; Gao, L.; Mao, C.; Zeng, H.; Wang, X.; Sun, Y.; Gu, J.; Wang, Y.; Chen, K.; et al. MicroRNA-146a Protects against Myocardial Ischaemia Reperfusion Injury by Targeting Med1. Cell. Mol. Biol. Lett. 2019, 24, 62. [Google Scholar] [CrossRef]
- Chen, B.-B.; Li, Z.-H.; Gao, S. Circulating MiR-146a/b Correlates with Inflammatory Cytokines in COPD and Could Predict the Risk of Acute Exacerbation COPD. Medicine 2018, 97, e9820. [Google Scholar] [CrossRef]
- Rebane, A.; Runnel, T.; Aab, A.; Maslovskaja, J.; Rückert, B.; Zimmermann, M.; Plaas, M.; Kärner, J.; Treis, A.; Pihlap, M.; et al. MicroRNA-146a Alleviates Chronic Skin Inflammation in Atopic Dermatitis through Suppression of Innate Immune Responses in Keratinocytes. J. Allergy Clin. Immunol. 2014, 134, 836–847.e11. [Google Scholar] [CrossRef]
- Yu, T.; Ju, Z.; Luo, M.; Hu, R.; Teng, Y.; Xie, L.; Zhong, C.; Chen, L.; Hou, W.; Xiong, Y.; et al. Elevated Expression of MiR-146a Correlates with High Levels of Immune Cell Exhaustion Markers and Suppresses Cellular Immune Function in Chronic HIV-1-Infected Patients. Sci. Rep. 2019, 9, 18829. [Google Scholar] [CrossRef]
- Chen, R.-F.; Yang, K.D.; Lee, I.-K.; Liu, J.-W.; Huang, C.-H.; Lin, C.-Y.; Chen, Y.-H.; Chen, C.-L.; Wang, L. Augmented MiR-150 Expression Associated with Depressed SOCS1 Expression Involved in Dengue Haemorrhagic Fever. J. Infect. 2014, 69, 366–374. [Google Scholar] [CrossRef] [Green Version]
- Badry, A.; Jaspers, V.L.B.; Waugh, C.A. Environmental Pollutants Modulate RNA and DNA Virus-Activated MiRNA-155 Expression and Innate Immune System Responses: Insights into New Immunomodulative Mechanisms. J. Immunotoxicol. 2020, 17, 86–93. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dickey, L.L.; Hanley, T.M.; Huffaker, T.B.; Ramstead, A.G.; O’Connell, R.M.; Lane, T.E. MicroRNA 155 and Viral-Induced Neuroinflammation. J. Neuroimmunol. 2017, 308, 17–24. [Google Scholar] [CrossRef] [PubMed]
- Zheng, X.; Huang, H.; Liu, J.; Li, M.; Liu, M.; Luo, T. Propofol Attenuates Inflammatory Response in LPS-Activated Microglia by Regulating the MiR-155/SOCS1 Pathway. Inflammation 2018, 41, 11–19. [Google Scholar] [CrossRef]
- Wyler, E.; Mösbauer, K.; Franke, V.; Diag, A.; Gottula, L.T.; Arsiè, R.; Klironomos, F.; Koppstein, D.; Hönzke, K.; Ayoub, S.; et al. Transcriptomic Profiling of SARS-CoV-2 Infected Human Cell Lines Identifies HSP90 as Target for COVID-19 Therapy. IScience 2021, 24, 102151. [Google Scholar] [CrossRef] [PubMed]
- Goncalves-Alves, E.; Saferding, V.; Schliehe, C.; Benson, R.; Kurowska-Stolarska, M.; Brunner, J.S.; Puchner, A.; Podesser, B.K.; Smolen, J.S.; Redlich, K.; et al. MicroRNA-155 Controls T Helper Cell Activation During Viral Infection. Front. Immunol. 2019, 10. [Google Scholar] [CrossRef] [Green Version]
- Dudda, J.C.; Salaun, B.; Ji, Y.; Palmer, D.C.; Monnot, G.C.; Merck, E.; Boudousquie, C.; Utzschneider, D.T.; Escobar, T.M.; Perret, R.; et al. MicroRNA-155 Is Required for Effector CD8+ T Cell Responses to Virus Infection and Cancer. Immunity 2013, 38, 742–753. [Google Scholar] [CrossRef] [Green Version]
- Yang, X.; Zhang, J.; Ding, Y. Association of MicroRNA-155, Interleukin 17A, and Proteinuria in Preeclampsia. Medicine (Baltimore) 2017, 96, e6509. [Google Scholar] [CrossRef]
- Zhou, F.; Sun, Y.; Gao, Q.; Wang, H. MicroRNA-21 Regulates the Proliferation of Placental Cells via FOXM1 in Preeclampsia. Exp. Ther. Med. 2020, 20, 1871–1878. [Google Scholar] [CrossRef] [PubMed]
- Akgör, U.; Ayaz, L.; Çayan, F. Expression Levels of Maternal Plasma MicroRNAs in Preeclamptic Pregnancies. J. Obstet. Gynaecol. 2020. [Google Scholar] [CrossRef]
- Zhang, J.-T.; Cai, Q.-Y.; Ji, S.-S.; Zhang, H.-X.; Wang, Y.-H.; Yan, H.-T.; Yang, X.-J. Decreased MiR-143 and Increased MiR-21 Placental Expression Levels Are Associated with Macrosomia. Mol. Med. Rep. 2016, 13, 3273–3280. [Google Scholar] [CrossRef] [PubMed]
- Maccani, M.A.; Padbury, J.F.; Marsit, C.J. MiR-16 and MiR-21 Expression in the Placenta Is Associated with Fetal Growth. PLoS ONE 2011, 6, e21210. [Google Scholar] [CrossRef] [PubMed]
- Whitehead, C.L.; Teh, W.T.; Walker, S.P.; Leung, C.; Larmour, L.; Tong, S. Circulating MicroRNAs in Maternal Blood as Potential Biomarkers for Fetal Hypoxia In-Utero. PLoS ONE 2013, 8, e78487. [Google Scholar] [CrossRef] [Green Version]
- Gillet, V.; Ouellet, A.; Stepanov, Y.; Rodosthenous, R.S.; Croft, E.K.; Brennan, K.; Abdelouahab, N.; Baccarelli, A.; Takser, L. MiRNA Profiles in Extracellular Vesicles From Serum Early in Pregnancies Complicated by Gestational Diabetes Mellitus. J. Clin. Endocrinol. Metab. 2019, 104, 5157–5169. [Google Scholar] [CrossRef] [PubMed]
- Cao, Y.; Li, L.; Feng, Z.; Wan, S.; Huang, P.; Sun, X.; Wen, F.; Huang, X.; Ning, G.; Wang, W. Comparative Genetic Analysis of the Novel Coronavirus (2019-NCoV/SARS-CoV-2) Receptor ACE2 in Different Populations. Cell Discov. 2020, 6, 11. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Buchrieser, J.; Dufloo, J.; Hubert, M.; Monel, B.; Planas, D.; Rajah, M.M.; Planchais, C.; Porrot, F.; Guivel-Benhassine, F.; Van der Werf, S.; et al. Syncytia Formation by SARS-CoV-2-Infected Cells. EMBO J. 2020, 39, e106267. [Google Scholar] [CrossRef] [PubMed]
- Sironi, M.; Biasin, M.; Cagliani, R.; Gnudi, F.; Saulle, I.; Ibba, S.; Filippi, G.; Yahyaei, S.; Tresoldi, C.; Riva, S.; et al. Evolutionary Analysis Identifies an MX2 Haplotype Associated with Natural Resistance to HIV-1 Infection. Mol. Biol. Evol. 2014, 31, 2402–2414. [Google Scholar] [CrossRef] [Green Version]
- Ivaska, L.; Niemelä, J.; Lempainen, J.; Österback, R.; Waris, M.; Vuorinen, T.; Hytönen, J.; Rantakokko-Jalava, K.; Peltola, V. Aetiology of Febrile Pharyngitis in Children: Potential of Myxovirus Resistance Protein A (MxA) as a Biomarker of Viral Infection. J. Infect. 2017, 74, 385–392. [Google Scholar] [CrossRef] [PubMed]
- Haller, O.; Kochs, G. Human MxA Protein: An Interferon-Induced Dynamin-like GTPase with Broad Antiviral Activity. J. Interf. Cytokine Res. 2011, 31, 79–87. [Google Scholar] [CrossRef]
- Chandrashekar, A.; Liu, J.; Martinot, A.J.; McMahan, K.; Mercado, N.B.; Peter, L.; Tostanoski, L.H.; Yu, J.; Maliga, Z.; Nekorchuk, M.; et al. SARS-CoV-2 Infection Protects against Rechallenge in Rhesus Macaques. Science 2020, 369, 812–817. [Google Scholar] [CrossRef]
- Chen, J.; Liang, Y.; Yi, P.; Xu, L.; Hawkins, H.K.; Rossi, S.L.; Soong, L.; Cai, J.; Menon, R.; Sun, J. Outcomes of Congenital Zika Disease Depend on Timing of Infection and Maternal-Fetal Interferon Action. Cell Rep. 2017, 21, 1588–1599. [Google Scholar] [CrossRef] [Green Version]
- Aye, I.L.; Waddell, B.J.; Mark, P.J.; Keelan, J. Oxysterols exert proinflammatory effects in placental trophoblasts via TLR4-dependent, cholesterol-sensitive activation of NF-κB. Mol. Hum. Reprod. 2012, 18, 341–353. [Google Scholar] [CrossRef] [Green Version]
- Biasin, M.; De Luca, M.; Gnudi, F.; Clerici, M. The Genetic Basis of Resistance to HIV Infection and Disease Progression. Expert Rev. Clin. Immunol. 2013, 9, 319–334. [Google Scholar] [CrossRef] [PubMed]
- Dicks, M.D.J.; Betancor, G.; Jimenez-Guardeño, J.M.; Pessel-Vivares, L.; Apolonia, L.; Goujon, C.; Malim, M.H. Multiple Components of the Nuclear Pore Complex Interact with the Amino-Terminus of MX2 to Facilitate HIV-1 Restriction. PLoS Pathog. 2018, 14, e1007408. [Google Scholar] [CrossRef]
- Tan, L.; Wang, Q.; Zhang, D.; Ding, J.; Huang, Q.; Tang, Y.; Wang, Q.; Miao, H. Lymphopenia predicts disease severity of COVID-19: A descriptive and predictive study. Signal. Transduct. Target. Ther. 2020, 5, 33. [Google Scholar] [CrossRef] [PubMed]
| SIPW (n = 15) | UPW (n = 6) | |
|---|---|---|
| Maternal baseline characteristics | ||
| Maternal age, years, median (range) | 32 (21–39) | 33.5 (28–40) |
| Third trimester of pregnancy, n (%) | 15 (100) | 6 (100) |
| Gestational age at admission, median (range) | 39 (36–41) | 40 (38–41) |
| RT-PCR assay of a maternal nasopharyngeal swab | ||
| Positive, n (%) | 15 (100) | 0 (0) |
| Prepregnancy BMI, kg/m2, median (range) | 23.4 (17.1–31.1) | 22.9 (18.2–33.1) |
| Smoking habit, n (%) | 0 (0) | 0 (0) |
| Ethnicity, Caucasian, n (%) | 9 (60) | 6 (100) |
| Chronic comorbidity, n (%) | 4 (26.6) | 0 (0) |
| Nulliparous n (%) | 6 (40) | 1 (16.6) |
| Oxygen support without ICU admission, n (%) | 2 (13.3) | 0 (0) |
| Positive chest X-ray, n (%) | 5 (33.3) | 0 (0) |
| Severe case, n (%) | 2 (13.3) | 0 (0) |
| Admission to ICU, n (%) | 1 (6.7) | 0 (0) |
| SIPW (n = 15) | UPW (n = 6) | |
|---|---|---|
| Total of deliveries, n (%) | 15 (100) | 6 (100) |
| Delivery mode | ||
| Vaginal, n (%) | 10 (66.7) | 3 (50) |
| Caesarean section, n (%) | 5 (33.3) | 3 (50) |
| GA at delivery, weeks median (range) | 38 (36–40) | 40 (38–41) |
| Caesarean section for severe maternal illness related to COVID-19, n (%) | 2 (13.3) | 0 (0) |
| Birth weight, g, median (range) | 3160 (2665–3775) | 2955 (2715–3500) |
| Umbilical artery pH, median (range) | 7.34 (7.24–7.53) | 7.31 (7.19–7.36) |
| APGAR score 5′ < 7, n (%) | 1 (6.7) | 1 (16.7) |
| Infected neonates, positive, n (%) | 0 (0) | NA |
| NICU admission, n (%) | 0 (0) | NA |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Saulle, I.; Garziano, M.; Fenizia, C.; Cappelletti, G.; Parisi, F.; Clerici, M.; Cetin, I.; Savasi, V.; Biasin, M. MiRNA Profiling in Plasma and Placenta of SARS-CoV-2-Infected Pregnant Women. Cells 2021, 10, 1788. https://doi.org/10.3390/cells10071788
Saulle I, Garziano M, Fenizia C, Cappelletti G, Parisi F, Clerici M, Cetin I, Savasi V, Biasin M. MiRNA Profiling in Plasma and Placenta of SARS-CoV-2-Infected Pregnant Women. Cells. 2021; 10(7):1788. https://doi.org/10.3390/cells10071788
Chicago/Turabian StyleSaulle, Irma, Micaela Garziano, Claudio Fenizia, Gioia Cappelletti, Francesca Parisi, Mario Clerici, Irene Cetin, Valeria Savasi, and Mara Biasin. 2021. "MiRNA Profiling in Plasma and Placenta of SARS-CoV-2-Infected Pregnant Women" Cells 10, no. 7: 1788. https://doi.org/10.3390/cells10071788
APA StyleSaulle, I., Garziano, M., Fenizia, C., Cappelletti, G., Parisi, F., Clerici, M., Cetin, I., Savasi, V., & Biasin, M. (2021). MiRNA Profiling in Plasma and Placenta of SARS-CoV-2-Infected Pregnant Women. Cells, 10(7), 1788. https://doi.org/10.3390/cells10071788








