Modeling Hepatotropic Viral Infections: Cells vs. Animals
Abstract
1. Introduction
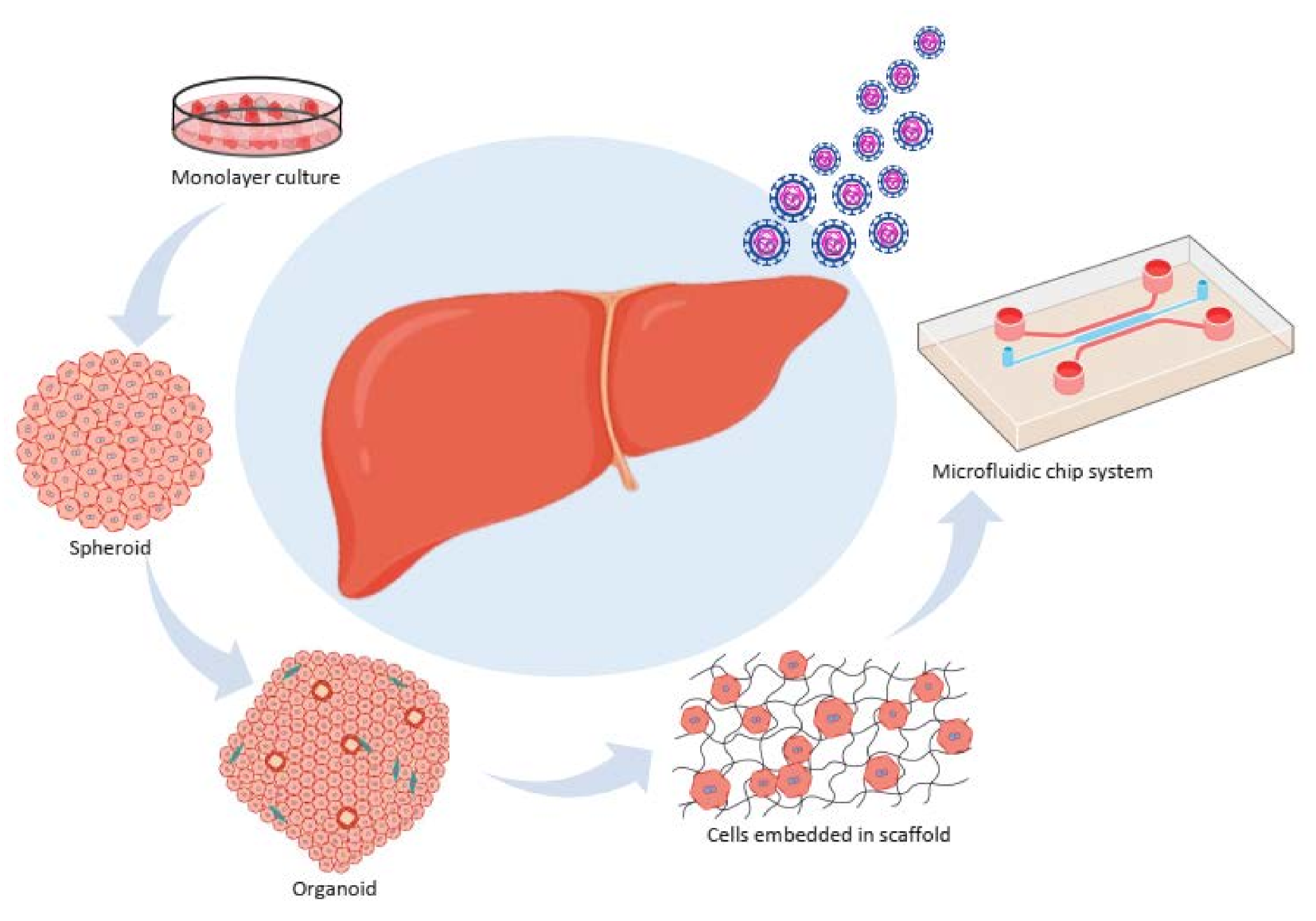
2. In Vitro Models
2.1. A monolayer Culture (Primary Cells, Cell Lines, and Coculture System)
2.2. Three Dimensional (3D) Culture Systems
2.2.1. Spheroid Culture Models
2.2.2. Organoid Systems
2.2.3. Cells Embedded in a Scaffold
2.2.4. A Microfluidic Chip System
3. Various In Vivo Approaches in Hepatotropic Virus Modeling
3.1. Transgenic Animals
3.2. Humanized Animals
3.3. Viral Adaptation
3.4. Transfected Models
Hydrodynamic Injection and Adenovirus (AdV)
4. Conclusions
| Culture Model | Cell Type | HAV | HBV | HCV | HDV | HEV |
|---|---|---|---|---|---|---|
| 2D | Primary cells | Adult PHH [15,21] | Adult PHH [17] | Adult PHH [18,19,20,21] | Adult PHH [18] primary human intestinal cells [23] primary human endometrial cells [24] | |
| Cancer cell lines | HepAD38 [34] HepDE19 [35] Hepa RG [44,46] HLCZ01 [36] (HepNB2.7) [38] HrpaRG [39,40] HepG2 +Huh-7 [49,55] HepG2 [51] HepG2-NTCP [52] HLC [24,26,64] | HLCZ01 [36] HepG2 [51] Huh-7 [55] | RG [45,46] HepG2, Huh-7 [50] | HepaRG cell line, PICM19 [43] Hepa RG [44] HepG2 and Huh-7 [53] HepG2 and HepG2/C3A [54] | ||
| PSCs | HLC [61,62,63,66,67,68,69] | HLC [70,71,72] | ||||
| Co-culture | Adult PHH + Murine fibroblast cells [28], Fetal PHH + nonparenchymal cells [29], Adult PHH + Murine stromal cells [30] | Adult PHH + Murine fibroblast cells [26], Adult PHH + Murine fibroblast cells [28] | PHH + 3T3-J2 [31] | peripheral blood mononuclear cells + bone marrow-derived macrophages [32] primary proximal tubular epithelial cells + immune cells [33] | ||
| 3D | Spheroid | HLCs [91] | HCC cell lines + fibroblast cells + HUVEC + stellate cells [83] | Adult PHH [75] Immortalized PHH [76] Huh-7 [77,78,79] Huh-7.5 + PHH [81] | ||
| Organoid | ||||||
| Scafffold based | PSC-endoderm+ mesenchymal stem cells +HUVEC [87] HCC-derived PHH/Healthy PHH [176] HepLPCs [82] | Huh-7.5 [89,90] | ||||
| Microfluid chip | Rat hepatocyte, PHH [93] PHH+endothelila cells [94] PHH with/without murin fibroblast cells [95] | Huh-7 + calcium alginate scaffold [92] |
| Animal | N | Infecting Agent | Therapeutic Agent | Outcomes | Ref. |
|---|---|---|---|---|---|
| Chimpanzee | 4 | HCV | Recombinant HCV-like particles and AS01B adjuvant | HCV-specific cellular immune response induced by immunization | [96] |
| Woodchuck | 27 | WHBV | RG7834 in combination with ETV and IFN-α | Efficacy of the treatment No sustained antiviral response | [102] |
| Transgenic HBV mice | 113 | HBV | ETV | Minimal effective daily doses for male and female mice revealed | [116] |
| Transgenic HCV mice | 21 | HCV | PRI-724 | Significant antifibrotic effect | [118] |
| uPA/RAG-2 mice | 25 | WMHBV | HBVpreS/2-48stearoyl HBVpreS/2-39myr | Inhibition of virus entry by peptides | [127] |
| uPA/SCID mice | 6 | HBV and HDV | Myrcludex-B | Inhibition of HDV infection In vivo kinetics of HDV spreading revealed No detectable HDV and HBV serological markers in treated mice | [129] |
| uPA/SCID mice | 24 | HEV | PEGylated IFN-α | Rapid viral clearance Confirmation of model applicability [132] | |
| FRG mice | 12 | HCV | PEGylated IFN-α 2a, ribavirin, Debio 025, adefovir dipivoxil | Same efficacy of the treatment with different agents | [137] |
| FRG mice | 9 | HEV | - | Model susceptibility [138] | |
| FRGS mice | 6 | HBV | Myrcludex B and ETV | Drug synergism Reduced HBV RNA and DNA production | [141] |
| TK-NOG mice | 8 | HBV | NM23TC | Sustained anti-HBV response | [146] |
| AFC8 mice | 6 | HCV | - | Infiltration with human T-cells, macrophages, and dendritic and NK cells observed Human HCV-specific T-cell response Poor B-cell response | [147] |
| BALB/c Rag2−/− IL-2Rγc−/− NOD.sirpa uPAtg/tg mice | 4 | HBV | ETV | Immune cells infiltration observed Synthesis of HBV-specific IGs Virus progression affected by immune system | [150] |
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| 2D | Two dimensional |
| 3D | Three dimensional |
| AdV | Adenovirus |
| c ccDNA | Covalently closed circular DNA |
| FRG | Fumaryl acetoacetate hydrolase (Fah)/RAG2/interleukin (IL) 2-gammaC (FRG) triple mutant mice |
| HAV | Hepatitis A virus |
| HBV | Hepatitis B virus |
| HCC | hepatocellular carcinoma |
| HCV | Hepatitis C virus |
| HDV | Hepatitis D virus |
| HepG2 | Hepatoma G2 |
| HLCs | Hepatic like cells |
| HUH7 | human hepatocellular carcinoma cell line 7 |
| HUVECs | Human umbilical vein endothelial cells |
| IL | Interleukin |
| NK | Natural killer |
| NTCP | Sodium taurocholate cotransporting polypeptide |
| PHH | Primary human hepatocyte |
| pgRNA | Pregenomic RNA |
| PSCs | Pluripotent stem cells |
| SCID | Severe combined immunodeficiency |
| shRNA | Short hairpin RNA |
| siRNA | Small interfering RNA |
| TK-NOG | NOG mouse expressing a thymidine kinase transgene (NOG-Tg(Alb-UL23)7-2/ShiJic) |
| uPA/RAG-2 | urokinase-type plasminogen activator (uPA)/recombinant activation gene-2 (RAG-2) |
| WHBV | Woodchuck hepatitis B virus |
References
- Burwitz, B.J.; Zhou, Z.; Li, W. Animal models for the study of human hepatitis B and D virus infection: New insights and progress. Antivir. Res. 2020, 182, 104898. [Google Scholar] [CrossRef]
- Ortega-Prieto, A.M.; Cherry, C.; Gunn, H.; Dorner, M. In Vivo Model Systems for Hepatitis B Virus Research. Acs Infect. Dis. 2019, 5, 688–702. [Google Scholar] [CrossRef] [PubMed]
- Inoue, T.; Tanaka, Y. Hepatitis B virus and its sexually transmitted infection—An update. Microb. Cell 2016, 3, 419–436. [Google Scholar] [CrossRef] [PubMed]
- Kushner, T.; Sperling, R.S.; Dieterich, D. Family Counseling for Hepatitis B and Hepatitis C. Clin. Liver Dis. A Multimed. Rev. J. 2019, 13, 93–97. [Google Scholar] [CrossRef] [PubMed]
- Farci, P.; Niro, G.A.; Zamboni, F.; Diaz, G. Hepatitis D and hepatocellular carcinoma. Viruses 2021, 13, 538. [Google Scholar] [CrossRef]
- Usman, Z.; Velkov, S.; Protzer, U.; Roggendorf, M.; Frishman, D.; Karimzadeh, H. HDVdb: A comprehensive hepatitis d virus database. Viruses 2020, 12, 538. [Google Scholar] [CrossRef] [PubMed]
- Ringehan, M.; Mckeating, J.A.; Protzer, U. Viral hepatitis and liver cancer. Philos. Trans. R. Soc. L. B Biol. Sci. 2017, 372, 20160274. [Google Scholar] [CrossRef]
- Wang, J.; Qu, B.; Zhang, F.; Zhang, C.; Deng, W.; Thi, V.L.D.; Xia, Y. Stem cell-derived hepatocyte-like cells as model for viral hepatitis research. Stem Cells Int. 2019, 2019. [Google Scholar] [CrossRef]
- Yu, J.M.; Li, L.L.; Zhang, C.Y.; Lu, S.; Ao, Y.Y.; Gao, H.C.; Xie, Z.; Xie, G.C.; Sun, X.M.; Pang, L.L.; et al. A novel hepatovirus identified in wild woodchuck Marmota himalayana. Sci. Rep. 2016, 6, 22361. [Google Scholar] [CrossRef]
- Debing, Y.; Moradpour, D.; Neyts, J.; Gouttenoire, J. Update on hepatitis e virology: Implications for clinical practice. J. Hepatol. 2016, 65, 200–212. [Google Scholar] [CrossRef]
- Lamontagne, J.; Mell, J.C.; Bouchard, M.J. Transcriptome-Wide Analysis of Hepatitis B Virus-Mediated Changes to Normal Hepatocyte Gene Expression. PLoS Pathog. 2016, 12, e1005438. [Google Scholar] [CrossRef]
- Catanese, M.T.; Dorner, M. Advances in experimental systems to study hepatitis C virus in vitro and in vivo. Virology 2015, 479–480, 221–233. [Google Scholar] [CrossRef]
- Ortega-Prieto, A.M.; Skelton, J.K.; Wai, S.N.; Large, E.; Lussignol, M.; Vizcay-Barrena, G.; Hughes, D.; Fleck, R.A.; Thursz, M.; Catanese, M.T.; et al. 3D microfluidic liver cultures as a physiological preclinical tool for hepatitis B virus infection. Nat. Commun. 2018, 9, 1–15. [Google Scholar] [CrossRef]
- Xiang, C.; Du, Y.; Meng, G.; Yi, L.S.; Sun, S.; Song, N.; Zhang, X.; Xiao, Y.; Wang, J.; Yi, Z.; et al. Long-term functional maintenance of primary human hepatocytes in vitro. Science 2019, 364, 399–402. [Google Scholar] [CrossRef] [PubMed]
- Zahmatkesh, E.; Vosough, M. A Quick update from the Past to Current Status of Human Pluripotent Stem Cell-derived Hepatocyte culture systems. Mod. Med. Lab. J. 2018, 2, 110–112. [Google Scholar] [CrossRef][Green Version]
- Galle, P.R.; Hagelstein, J.; Kommerell, B.; Volkmann, M.; Schranz, P.; Zentgraf, H. In vitro experimental infection of primary human hepatocytes with hepatitis B virus. Gastroenterology 1994, 106, 664–673. [Google Scholar] [CrossRef]
- Fournier, C.; Pageaux, G.; Maurel, P.; Coste, J.; Larrey, D.; Ducos, J.; Sureau, C.; Domergue, J. In vitro infection of adult normal human hepatocytes in primary culture by hepatitis C virus. J. Gen. Virol. 1998, 79, 2367–2374. [Google Scholar] [CrossRef]
- Yin, X.; Li, X.; Ambardekar, C.; Hu, Z.; Lhomme, S.; Feng, Z. Hepatitis E virus persists in the presence of a type III interferon response. PLoS Pathog. 2017, 13, e1006417. [Google Scholar] [CrossRef] [PubMed]
- Gudima, S.; He, Y.; Meier, A.; Chang, J.; Chen, R.; Jarnik, M.; Nicolas, E.; Bruss, V.; Taylor, J. Assembly of Hepatitis Delta Virus: Particle Characterization, Including the Ability To Infect Primary Human Hepatocytes. J. Virol. 2007, 81, 3608–3617. [Google Scholar] [CrossRef] [PubMed]
- Gudima, S.; He, Y.; Chai, N.; Bruss, V.; Urban, S.; Mason, W.; Taylor, J. Primary Human Hepatocytes Are Susceptible to Infection by Hepatitis Delta Virus Assembled with Envelope Proteins of Woodchuck Hepatitis Virus. J. Virol. 2008, 82, 7276–7283. [Google Scholar] [CrossRef]
- Zhang, Z.; Filzmayer, C.; Ni, Y.; Sültmann, H.; Mutz, P.; Hiet, M.S.; Vondran, F.W.R.; Bartenschlager, R.; Urban, S. Hepatitis D virus replication is sensed by MDA5 and induces IFN-β/λ responses in hepatocytes. J. Hepatol. 2018, 69, 25–35. [Google Scholar] [CrossRef] [PubMed]
- Taylor, J.M.; Han, Z. Purinergic receptor functionality is necessary for infection of human hepatocytes by hepatitis delta virus and hepatitis b virus. PLoS ONE 2010, 5, e15784. [Google Scholar] [CrossRef]
- Marion, O.; Lhomme, S.; Nayrac, M.; Dubois, M.; Pucelle, M.; Requena, M.; Migueres, M.; Abravanel, F.; Peron, J.M.; Carrere, N.; et al. Hepatitis E virus replication in human intestinal cells. Gut 2020, 69, 901–910. [Google Scholar] [CrossRef]
- El-mokhtar, M.A.; Othman, E.R.; Khashbah, M.Y.; Ismael, A.; Ghaliony, M.A.A.; Seddik, M.I.; Sayed, I.M. Evidence of the Extrahepatic Replication of Hepatitis. Pathogens 2020, 9, 295. [Google Scholar] [CrossRef] [PubMed]
- Xia, Y.; Carpentier, A.; Cheng, X.; Block, P.D.; Zhao, Y.; Zhang, Z.; Protzer, U.; Liang, T.J. Human stem cell-derived hepatocytes as a model for hepatitis B virus infection, spreading and virus-host interactions. Physiol. Behav. 2018, 176, 139–148. [Google Scholar] [CrossRef]
- PLoSs, A.; Khetani, S.R.; Jones, C.T.; Syder, A.J.; Trehan, K.; Gaysinskaya, V.A.; Mu, K.; Ritola, K.; Rice, C.M.; Bhatia, S.N. Persistent hepatitis C virus infection in microscale primary human hepatocyte cultures. Proc. Natl. Acad. Sci. USA 2010, 107, 3141–3145. [Google Scholar] [CrossRef] [PubMed]
- Shlomai, A.; Schwartz, R.E.; Ramanan, V.; Bhatta, A.; De Jong, Y.P.; Bhatia, S.N.; Rice, C.M. Modeling host interactions with hepatitis B virus using primary and induced pluripotent stem cell-derived hepatocellular systems. Proc. Natl. Acad. Sci. USA 2014, 111, 12193–12198. [Google Scholar] [CrossRef]
- March, S.; Ramanan, V.; Trehan, K.; Ng, S.; Galstian, A.; Gural, N.; Scull, M.A.; Shlomai, A.; Mota, M.M.; Fleming, H.E.; et al. Micropatterned coculture of primary human hepatocytes and supportive cells for the study of hepatotropic pathogens. Nat. Protoc. 2015, 10, 2027–2053. [Google Scholar] [CrossRef]
- Zhou, M.; Zhao, F.; Li, J.; Cheng, Z.; Tian, X.; Zhi, X.; Huang, Y.; Hu, K. Long-term maintenance of human fetal hepatocytes and prolonged susceptibility to HBV infection by co-culture with non-parenchymal cells. J. Virol. Methods 2014, 195, 185–193. [Google Scholar] [CrossRef]
- Winer, B.Y.; Huang, T.S.; Pludwinski, E.; Heller, B.; Wojcik, F.; Lipkowitz, G.E.; Parekh, A.; Cho, C.; Shrirao, A.; Muir, T.W.; et al. Long-term hepatitis B infection in a scalable hepatic co-culture system. Nat. Commun. 2017, 8, 1–11. [Google Scholar] [CrossRef]
- Winer, B.Y.; Gaska, J.M.; Lipkowitz, G.; Bram, Y.; Parekh, A.; Parsons, L.; Leach, R.; Jindal, R.; Cho, C.H.; Shrirao, A.; et al. Analysis of Host Responses to Hepatitis B and Delta Viral Infections in a Micro-scalable Hepatic Co-culture System. Hepatology 2020, 71, 14–30. [Google Scholar] [CrossRef]
- Sayed, I.M.; Seddik, M.I.; Gaber, M.A.; Saber, S.H.; Mandour, S.A.; El-Mokhtar, M.A. Replication of hepatitis e virus (HEV) in primary human-derived monocytes and macrophages in vitro. Vaccines 2020, 8, 239. [Google Scholar] [CrossRef] [PubMed]
- El-Mokhtar, M.A.; Seddik, M.I.; Osman, A.; Adel, S.; Abdel Aziz, E.M.; Mandour, S.A.; Mohammed, N.; Zarzour, M.A.; Abdel-Wahid, L.; Radwan, E.; et al. Hepatitis e virus mediates renal injury via the interaction between the immune cells and renal epithelium. Vaccines 2020, 8, 454. [Google Scholar] [CrossRef]
- Ladner, S.K.; Otto, M.J.; Barker, C.S.; Zaifert, K.; Wang, G.H.; Guo, J.U.T.; Seeger, C.; King, R.W. Inducible Expression of Human Hepatitis B Virus (HBV) in Stably Transfected Hepatoblastoma Cells: A Novel System for Screening Potential Inhibitors of HBV Replication. Antimicrob. Agents Chemother. 1997, 41, 1715–1720. [Google Scholar] [CrossRef] [PubMed]
- Guo, H.; Jiang, D.; Zhou, T.; Cuconati, A.; Block, T.M.; Guo, J.-T. Characterization of the Intracellular Deproteinized Relaxed Circular DNA of Hepatitis B Virus: An Intermediate of Covalently Closed Circular DNA Formation. J. Virol. 2007, 81, 12472–12484. [Google Scholar] [CrossRef]
- Yang, D.; Zuo, C.; Wang, X.; Meng, X.; Xue, B.; Liu, N.; Yu, R.; Qin, Y.; Gao, Y.; Wang, Q.; et al. Complete replication of hepatitis B virus and hepatitis C virus in a newly developed hepatoma cell line. Proc. Natl. Acad. Sci. USA 2014, 111, E1264–E1273. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Cai, W.; Sun, X.; Bi, Y.; Zeng, C.; Zhao, X.Y.; Zhou, Q.; Xu, T.; Xie, Q.; Sun, P.; et al. Defined host factors support HBV infection in non-hepatic 293T cells. J. Cell. Mol. Med. 2020, 24, 2507–2518. [Google Scholar] [CrossRef]
- Lempp, F.A.; Schlund, F.; Rieble, L.; Nussbaum, L.; Link, C.; Zhang, Z.; Ni, Y.; Urban, S. Recapitulation of HDV infection in a fully permissive hepatoma cell line allows efficient drug evaluation. Nat. Commun. 2019, 10, 1–11. [Google Scholar] [CrossRef]
- So, C.W.; Randall, G. Three-Dimensional Cell Culture Systems for Studying Hepatitis C Virus. Viruses 2021, 13, 211. [Google Scholar] [CrossRef]
- Hantz, O.; Parent, R.; Durantel, D.; Gripon, P.; Guguen-Guillouzo, C.; Zoulim, F. Persistence of the hepatitis B virus covalently closed circular DNA in HepaRG human hepatocyte-like cells. J. Gen. Virol. 2009, 90, 127–135. [Google Scholar] [CrossRef]
- Yuan, L.; Liu, X.; Zhang, L.; Zhang, Y.; Chen, Y.; Li, X.; Wu, K.; Cao, J.; Hou, W.; Que, Y.; et al. Optimized HepaRG is a suitable cell source to generate the human liver chimeric mouse model for the chronic hepatitis B virus infection. Emerg. Microbes Infect. 2018, 7, 1–17. [Google Scholar] [CrossRef]
- Shen, F.; Li, Y.; Wang, Y.; Sozzi, V.; Revill, P.A.; Liu, J.; Gao, L.; Yang, G.; Lu, M.; Sutter, K.; et al. Hepatitis B Virus Sensitivity to interferon-α in Hepatocytes Is More Associated With Cellular Interferon Response Than with Viral Genotype. Hepatology 2018, 67, 1237–1252. [Google Scholar] [CrossRef]
- Rogée, S.; Talbot, N.; Caperna, T.; Bouquet, J.; Barnaud, E.; Pavio, N. New models of hepatitis E virus replication in human and porcine hepatocyte cell lines. J. Gen. Virol. 2013, 94, 549–558. [Google Scholar] [CrossRef] [PubMed]
- Pellerin, M.; Hirchaud, E.; Blanchard, Y.; Pavio, N.; Doceul, V. Characterization of a cell culture system of persistent hepatitis e virus infection in the human heparg hepatic cell line. Viruses 2021, 13, 406. [Google Scholar] [CrossRef] [PubMed]
- Lucifora, J.; Michelet, M.; Salvetti, A.; Durantel, D. Fast Differentiation of HepaRG Cells Allowing Hepatitis B and Delta Virus Infections. Cells 2020, 9, 2288. [Google Scholar] [CrossRef]
- Alfaiate, D.; Lucifora, J.; Abeywickrama-Samarakoon, N.; Michelet, M.; Testoni, B.; Cortay, J.C.; Sureau, C.; Zoulim, F.; Dény, P.; Durantel, D. HDV RNA replication is associated with HBV repression and interferon-stimulated genes induction in super-infected hepatocytes. Antivir. Res. 2016, 136, 19–31. [Google Scholar] [CrossRef]
- Ni, Y.; Lempp, F.A.; Mehrle, S.; Nkongolo, S.; Kaufman, C.; Fälth, M.; Stindt, J.; Königer, C.; Nassal, M.; Kubitz, R.; et al. Hepatitis B and D viruses exploit sodium taurocholate co-transporting polypeptide for species-specific entry into hepatocytes. Gastroenterology 2014, 146, 1070–1083.e6. [Google Scholar] [CrossRef] [PubMed]
- Verrier, E.R.; Colpitts, C.C.; Schuster, C.; Zeisel, M.B.; Baumert, T.F. Cell culture models for the investigation of Hepatitis B and D Virus infection. Viruses 2016, 8, 261. [Google Scholar] [CrossRef] [PubMed]
- Farag, M.M.S.; Mansour, M.T.M. Characterization of Subviral Particles of Hepatitis B Virus Produced by HepG2.2.15 Cell Line—In vitro Study. Int. J. Virol. Mol. Biol. 2016, 5, 1–7. [Google Scholar] [CrossRef]
- Yan, H.; Zhong, G.; Xu, G.; He, W.; Jing, Z.; Gao, Z.; Huang, Y.; Qi, Y.; Peng, B.; Wang, H.; et al. Sodium taurocholate cotransporting polypeptide is a functional receptor for human hepatitis B and D virus. Elife 2012, 2012. [Google Scholar] [CrossRef]
- Otsuka, M.; Aizaki, H.; Kato, N.; Suzuki, T.; Miyamura, T.; Omata, M.; Seki, N. Differential cellular gene expression induced by hepatitis B and C viruses. Biochem. Biophys. Res. Commun. 2003, 300, 443–447. [Google Scholar] [CrossRef]
- König, A.; Yang, J.; Jo, E.; Park, K.H.P.; Kim, H.; Than, T.T.; Song, X.; Qi, X.; Dai, X.; Park, S.; et al. Efficient long-term amplification of hepatitis B virus isolates after infection of slow proliferating HepG2-NTCP cells. J. Hepatol. 2019, 71, 289–300. [Google Scholar] [CrossRef] [PubMed]
- Shukla, P.; Nguyen, H.T.; Torian, U.; Engle, R.E.; Faulk, K.; Dalton, H.R.; Bendall, R.P.; Keane, F.E.; Purcell, R.H.; Emerson, S.U. Cross-species infections of cultured cells by hepatitis E virus and discovery of an infectious virus-host recombinant. Proc. Natl. Acad. Sci. USA 2011, 108, 2438–2443. [Google Scholar] [CrossRef]
- Todt, D.; Friesland, M.; Moeller, N.; Praditya, D.; Kinast, V.; Brüggemann, Y.; Knegendorf, L.; Burkard, T.; Steinmann, J.; Burm, R.; et al. Robust hepatitis e virus infection and transcriptional response in human hepatocytes. Proc. Natl. Acad. Sci. USA 2020, 117, 1731–1741. [Google Scholar] [CrossRef]
- Jiang, W. A visualizable hepatitis A virus and hepatitis C virus coinfection model in vitro: Coexistence of two hepatic viruses under limited competition in viral RNA synthesis. bioRxivxiv 2019, 1–22. [Google Scholar] [CrossRef]
- Sun, D.; Nassal, M. Stable HepG2- and Huh7-based human hepatoma cell lines for efficient regulated expression of infectious hepatitis B virus. J. Hepatol. 2006, 45, 636–645. [Google Scholar] [CrossRef] [PubMed]
- Scarselli, E.; Ansuini, H.; Cerino, R.; Roccasecca, R.M.; Acali, S.; Filocamo, G.; Traboni, C.; Nicosia, A.; Cortese, R.; Vitelli, A. The human scavenger receptor class B type I is a novel candidate receptor for the hepatitis C virus. Embo J. 2002, 21, 5017–5025. [Google Scholar] [CrossRef]
- Zhang, J.; Randall, G.; Higginbottom, A.; Monk, P.; Rice, C.M.; McKeating, J.A. CD81 Is Required for Hepatitis C Virus Glycoprotein-Mediated Viral Infection. J. Virol. 2004, 78, 1448–1455. [Google Scholar] [CrossRef]
- Vosough, M.; Omidinia, E.; Kadivar, M.; Shokrgozar, M.A.; Pournasr, B.; Aghdami, N.; Baharvand, H. Generation of functional hepatocyte-like cells from human pluripotent stem cells in a scalable suspension culture. Stem Cells Dev. 2013, 22, 2693–2705. [Google Scholar] [CrossRef]
- Arez, F.; Rodrigues, A.F.; Brito, C.; Alves, P.M. bioengineered liver cell models of hepatotropic infections. Viruses 2021, 13, 773. [Google Scholar] [CrossRef]
- Schwartz, R.E.; Bram, Y.; Frankel, A. Pluripotent Stem Cell-Derived Hepatocyte-like Cells: A Tool to Study Infectious Disease. Curr. Pathobiol. Rep. 2016, 4, 147–156. [Google Scholar] [CrossRef][Green Version]
- Roelandt, P.; Obeid, S.; Paeshuyse, J.; Vanhove, J.; Van Lommel, A.; Nahmias, Y.; Nevens, F.; Neyts, J.; Verfaillie, C.M. Human pluripotent stem cell-derived hepatocytes support complete replication of hepatitis C virus. J. Hepatol. 2012, 57, 246–251. [Google Scholar] [CrossRef] [PubMed]
- Ng, S.; Schwartz, R.E.; March, S.; Galstian, A.; Gural, N.; Shan, J.; Prabhu, M.; Mota, M.M.; Bhatia, S.N. Human iPSC-derived hepatocyte-like cells support plasmodium liver-stage infection in vitro. Stem Cell Rep. 2015, 4, 348–359. [Google Scholar] [CrossRef] [PubMed]
- Yan, F.; Wang, Y.; Zhang, W.; Chang, M.; He, Z.; Xu, J.; Shang, C.; Chen, T.; Liu, J.; Wang, X.; et al. Human ES Cell-derived Hepatoblasts are an Optimal Lineage Stage for HCV Infection Fang. Hepatology 2017, 66, 717–735. [Google Scholar] [CrossRef] [PubMed]
- Kaneko, S.; Kakinuma, S.; Asahina, Y.; Kamiya, A.; Miyoshi, M.; Tsunoda, T.; Nitta, S.; Asano, Y.; Nagata, H.; Otani, S.; et al. Human induced pluripotent stem cell-derived hepatic cell lines as a new model for host interaction with hepatitis B virus. Sci. Rep. 2016, 6, 29358. [Google Scholar] [CrossRef] [PubMed]
- Sakurai, F.; Mitani, S.; Yamamoto, T.; Takayama, K.; Tachibana, M.; Watashi, K.; Wakita, T.; Iijima, S.; Tanaka, Y.; Mizuguchi, H. Human induced-pluripotent stem cell-derived hepatocyte-like cells as an in vitro model of human hepatitis B virus infection. Sci. Rep. 2017, 7, 45698. [Google Scholar] [CrossRef]
- Schwartz, R.E.; Trehan, K.; Andrus, L.; Sheahan, T.P.; PLoSs, A.; Duncan, S.A.; Rice, C.M.; Bhatia, S.N. Modeling hepatitis C virus infection using human induced pluripotent stem cells. Proc. Natl. Acad. Sci. USA 2012, 109, 2544–2548. [Google Scholar] [CrossRef]
- Wu, X.; Robotham, J.M.; Lee, E.; Dalton, S.; Kneteman, N.M.; Gilbert, D.M.; Tang, H. Productive hepatitis C virus infection of stem cell-derived hepatocytes reveals a critical transition to viral permissiveness during differentiation. PLoS Pathog. 2012, 8, e1002617. [Google Scholar] [CrossRef]
- Zhou, X.; Sun, P.; Lucendo-Villarin, B.; Angus, A.G.N.; Szkolnicka, D.; Cameron, K.; Farnworth, S.L.; Patel, A.H.; Hay, D.C. Modulating innate immunity improves hepatitis C virus infection and replication in stem cell-derived hepatocytes. Stem Cell Rep. 2014, 3, 204–214. [Google Scholar] [CrossRef][Green Version]
- Dao Thi, V.L.; Debing, Y.; Wu, X.; Rice, C.M.; Neyts, J.; Moradpour, D.; Gouttenoire, J. Sofosbuvir Inhibits Hepatitis e Virus Replication in Vitro and Results in an Additive Effect When Combined with Ribavirin. Gastroenterology 2016, 150, 82–85.e4. [Google Scholar] [CrossRef]
- Helsen, N.; Debing, Y.; Paeshuyse, J.; Dallmeier, K.; Boon, R.; Coll, M.; Sancho-Bru, P.; Claes, C.; Neyts, J.; Verfaillie, C.M. Stem cell-derived hepatocytes: A novel model for hepatitis e virus replication. J. Hepatol. 2016, 64, 565–573. [Google Scholar] [CrossRef] [PubMed]
- Wu, X.; Thi, V.L.D.; Liu, P.; Takacs, C.N.; Xiang, K.; Andrus, L.; Gouttenoire, J.; Moradpour, D.; Rice, C.M. Pan-Genotype Hepatitis E Virus Replication in Stem Cell-derived Hepatocellular Systems. Gastroenterology. 2018, 154, 663–674.e7. [Google Scholar] [CrossRef] [PubMed]
- Shinozawa, T.; Kimura, M.; Cai, Y.; Saiki, N.; Yoneyama, Y.; Ouchi, R.; Koike, H.; Maezawa, M.; Zhang, R.-R.; Dunn, A.; et al. High-Fidelity Drug-Induced Liver Injury Screen Using Human Pluripotent Stem Cell–Derived Organoids. Gastroenterology 2021, 160, 831–846.e10. [Google Scholar] [CrossRef]
- Shpichka, A.; Bikmulina, P.; Peshkova, M.; Kosheleva, N.; Zurina, I.; Zahmatkesh, E.; Khoshdel-Rad, N.; Lipina, M.; Golubeva, E.; Butnaru, D.; et al. Engineering a model to study viral infections: Bioprinting, microfluidics, and organoids to defeat coronavirus disease 2019 (COVID-19). Int. J. Bioprinting 2020, 6, 1–20. [Google Scholar] [CrossRef]
- Chong, T.W.; Smith, R.L.; Hughes, M.G.; Camden, J.; Rudy, C.K.; Evans, H.L.; Sawyer, R.G.; Pruett, T.L. Primary human hepatocytes in spheroid formation to study hepatitis C infection. J. Surg. Res. 2006, 130, 52–57. [Google Scholar] [CrossRef] [PubMed]
- Aly, H.H.; Shimotohno, K.; Hijikata, M. 3D cultured immortalized human hepatocytes useful to develop drugs for blood-borne HCV. Biochem. Biophys. Res. Commun. 2009, 379, 330–334. [Google Scholar] [CrossRef] [PubMed]
- Sainz, B.; Tencate, V.; Uprichard, S.L. Three-dimensional Huh7 cell culture system for the study of Hepatitis C virus infection. Virol. J. 2009, 6, 103. [Google Scholar] [CrossRef]
- Murakami, K.; Ishii, K.; Ishihara, Y.; Yoshizaki, S.; Tanaka, K.; Gotoh, Y.; Aizaki, H.; Kohara, M.; Yoshioka, H.; Mori, Y.; et al. Production of infectious hepatitis C virus particles in three-dimensional cultures of the cell line carrying the genome-length dicistronic viral RNA of genotype 1b. Virology 2006, 351, 381–392. [Google Scholar] [CrossRef]
- Molina-Jimenez, F.; Benedicto, I.; Dao Thi, V.L.; Gondar, V.; Lavillette, D.; Marin, J.J.; Briz, O.; Moreno-Otero, R.; Aldabe, R.; Baumert, T.F.; et al. Matrigel-embedded 3D culture of Huh-7 cells as a hepatocyte-like polarized system to study hepatitis C virus cycle. Virology 2012, 425, 31–39. [Google Scholar] [CrossRef] [PubMed]
- Durantel, D.; Zoulim, F. Going towards more relevant cell culture models to study the in vitro replication of serum-derived hepatitis C virus and virus/host cell interactions? J. Hepatol. 2007, 46, 1–5. [Google Scholar] [CrossRef]
- Ananthanarayanan, A.; Nugraha, B.; Triyatni, M.; Hart, S.; Sankuratri, S.; Yu, H. Scalable spheroid model of human hepatocytes for hepatitis C infection and replication. Mol. Pharm. 2014, 11, 2106–2114. [Google Scholar] [CrossRef]
- Fu, G.B.; Huang, W.J.; Zeng, M.; Zhou, X.; Wu, H.P.; Liu, C.C.; Wu, H.; Weng, J.; Zhang, H.D.; Cai, Y.C.; et al. Expansion and differentiation of human hepatocyte-derived liver progenitor-like cells and their use for the study of hepatotropic pathogens. Cell Res. 2019, 29, 8–22. [Google Scholar] [CrossRef]
- Song, Y.; Kim, J.S.; Kim, S.H.; Park, Y.K.; Yu, E.; Kim, K.H.; Seo, E.J.; Oh, H.B.; Lee, H.C.; Kim, K.M.; et al. Patient-derived multicellular tumor spheroids towards optimized treatment for patients with hepatocellular carcinoma. J. Exp. Clin. Cancer Res. 2018, 37, 1–13. [Google Scholar] [CrossRef]
- Dutta, D.; Heo, I.; Clevers, H. Disease Modeling in Stem Cell-Derived 3D Organoid Systems. Trends Mol. Med. 2017, 23, 393–410. [Google Scholar] [CrossRef]
- Zahmatkesh, E.; Ghanian, M.H.; Zarkesh, I.; Farzaneh, Z.; Halvaei, M.; Heydari, Z.; Moeinvaziri, F.; Othman, A.; Ruoß, M.; Piryaei, A.; et al. Tissue-Specific Microparticles Improve Organoid Microenvironment for Efficient Maturation of Pluripotent Stem-Cell-Derived Hepatocytes. Cells 2021, 10, 1274. [Google Scholar] [CrossRef]
- Zahmatkesh, E.; Khoshdel-Rad, N.; Mirzaei, H.; Shpichka, A.; Timashev, P.; Mahmoudi, T.; Vosough, M. Evolution of organoid technology: Lessons learnt in Co-Culture systems from developmental biology. Dev. Biol. 2021, 475, 37–53. [Google Scholar] [CrossRef]
- Nie, Y.Z.; Zheng, Y.W.; Miyakawa, K.; Murata, S.; Zhang, R.R.; Sekine, K.; Ueno, Y.; Takebe, T.; Wakita, T.; Ryo, A.; et al. Recapitulation of hepatitis B virus–host interactions in liver organoids from human induced pluripotent stem cells. EBioMedicine 2018, 35, 114–123. [Google Scholar] [CrossRef]
- Crignis, E.D.; Carofiglio, F.; Moulos, P.; Verstegen, M.M.A.; Romal, S.; Khalid, M.M.; Pourfarzad, F.; Koutsothanassis, C.; Gehart, H.; Kan, T.W.; et al. Human liver organoids; a patient-derived primary model for HBV Infection and Related Hepatocellular Carcinoma. bioRxiv 2019, 31, 1–40. [Google Scholar]
- Baktash, Y.; Madhav, A.; Coller, K.E.; Randall, G. Single Particle Imaging of Polarized Hepatoma Organoids upon Hepatitis C Virus Infection Reveals an Ordered and Sequential Entry Process. Cell Host Microbe 2018, 23, 382–394.e5. [Google Scholar] [CrossRef] [PubMed]
- Brown, R.J.P.; Tegtmeyer, B.; Sheldon, J.; Khera, T.; Anggakusuma; Todt, D.; Vieyres, G.; Weller, R.; Joecks, S.; Zhang, Y.; et al. Liver-expressed Cd302 and Cr1l limit hepatitis C virus cross-species transmission to mice. Sci. Adv. 2020, 6, eabd3233. [Google Scholar] [CrossRef] [PubMed]
- Kulsuptrakul, J.; Wang, R.; Meyers, N.L.; Ott, M.; Puschnik, A.S. A genome-wide CRISPR screen identifies UFMylation and TRAMP-like complexes as host factors required for hepatitis A virus infection. Cell Rep. 2021, 34, 108859. [Google Scholar] [CrossRef]
- Tran, N.M.; Dufresne, M.; Duverlie, G.; Castelain, S.; Défarge, C.; Paullier, P.; Legallais, C. An appropriate selection of a 3d alginate culture model for hepatic Huh-7 cell line encapsulation intended for viral studies. Tissue Eng. Part. A 2013, 19, 103–113. [Google Scholar] [CrossRef] [PubMed]
- Sodunke, T.R.; Bouchard, M.J.; Noh, H. Microfluidic platform for hepatitis B viral replication study. Biomed. Microdevices 2008, 10, 393–402. [Google Scholar] [CrossRef] [PubMed]
- Kang, Y.B.A.; Sodunke, T.R.; Lamontagne, J.; Cirillo, J.; Rajiv, C.; Bouchard, M.J.; Noh, M. Liver sinusoid on a chip: Long-term layered co-culture of primary rat hepatocytes and endothelial cells in microfluidic platforms. Biotechnol. Bioeng. 2015, 112, 2571–2582. [Google Scholar] [CrossRef] [PubMed]
- Ortega-Prieto, A.M.; Skelton, J.K.; Cherry, C.; Briones-Orta, M.A.; Hateley, C.A.; Dorner, M. “Liver-on-a-Chip” Cultures of Primary Hepatocytes and Kupffer Cells for Hepatitis B Virus Infection. J. Vis. Exp. 2019, e58333. [Google Scholar] [CrossRef] [PubMed]
- Elmowalid, G.A.; Qiao, M.; Jeong, S.H.; Borg, B.B.; Baumert, T.F.; Sapp, R.K.; Hu, Z.; Murthy, K.; Liang, T.J. Immunization with hepatitis C virus-like particles results in control of hepatitis C virus infection in chimpanzees. Proc. Natl. Acad. Sci. USA 2007, 104, 8427–8432. [Google Scholar] [CrossRef] [PubMed]
- Forns, X.; Payette, P.J.; Ma, X.; Satterfield, W.; Eder, G.; Mushahwar, I.K.; Govindarajan, S.; Davis, H.L.; Emerson, S.U.; Purcell, R.H.; et al. Vaccination of chimpanzees with plasmid DNA encoding the Hepatitis C virus (HCV) envelope E2 protein modified the infection after challenge with homologous monoclonal HCV. Hepatology 2000, 32, 618–625. [Google Scholar] [CrossRef]
- Lanford, R.E.; Guerra, B.; Chavez, D.; Giavedoni, L.; Hodara, V.L.; Brasky, K.M.; Fosdick, A.; Frey, C.R.; Zheng, J.; Wolfgang, G.; et al. GS-9620, an oral agonist of toll-like receptor-7, induces prolonged suppression of hepatitis B virus in chronically infected chimpanzees. Gastroenterology 2013, 144, 1508–1517.e10. [Google Scholar] [CrossRef] [PubMed]
- Walter, E.; Keist, R.; Niederöst, B.; Pult, I.; Blum, H.E. Hepatitis B virus infection of tupaia hepatocytes in vitro and in vivo. Hepatology 1996, 24, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Xie, Z.C.; Riezu-Boj, J.I.; Lasarte, J.J.; Guillen, J.; Su, J.H.; Civeira, M.P.; Prieto, J. Transmission of hepatitis C virus infection to tree shrews. J. Hepatol. 1998, 28, 102. [Google Scholar] [CrossRef][Green Version]
- Yang, C.; Ruan, P.; Ou, C.; Su, J.; Cao, J.; Luo, C.; Tang, Y.; Wang, Q.; Qin, H.; Sun, W.; et al. Chronic hepatitis B virus infection and occurrence of hepatocellular carcinoma in tree shrews (Tupaia belangeri chinensis). Virol. J. 2015, 12, 26. [Google Scholar] [CrossRef]
- Menne, S.; Wildum, S.; Steiner, G.; Suresh, M.; Korolowicz, K.; Balarezo, M.; Yon, C.; Murreddu, M.; Hong, X.; Kallakury, B.V.; et al. Efficacy of an Inhibitor of Hepatitis B Virus Expression in Combination With Entecavir and Interferon-α in Woodchucks Chronically Infected With Woodchuck Hepatitis Virus. Hepatol. Commun. 2020, 4, 916–931. [Google Scholar] [CrossRef]
- Guo, W.N.; Zhu, B.; Ai, L.; Yang, D.L.; Wang, B.J. Animal models for the study of hepatitis B virus infection. Zool. Res. 2018, 39, 25–31. [Google Scholar] [CrossRef] [PubMed]
- Vandegrift, K.J.; Critchlow, J.T.; Kapoor, A.; Friedman, D.A.; Hudson, P.J. Peromyscus as a model system for human hepatitis C: An opportunity to advance our understanding of a complex host parasite system. Semin. Cell Dev. Biol. 2017, 61, 123–130. [Google Scholar] [CrossRef] [PubMed]
- Chisari, F.V.; Pinkert, C.A.; Milich, D.R.; Filippi, P.; Mclachlan, A.; Palmiter, R.D.; Brinster, R.L. A transgenic mouse model of the chronic hepatitis B surface antigen carrier state. Science 1985, 230, 1157–1160. [Google Scholar] [CrossRef] [PubMed]
- Guidotti, L.G.; Martinez, V.; Loh, Y.T.; Rogler, C.E.; Chisari, F. V Hepatitis B virus nucleocapsid particles do not cross the hepatocyte nuclear membrane in transgenic mice. J. Virol. 1994, 68, 5469–5475. [Google Scholar] [CrossRef] [PubMed]
- Milich, D.R.; Jones, J.E.; Hughes, J.L.; Price, J.; Raney, A.K.; Mclachlan, A. Is a function of the secreted hepatitis B e antigen to induce immunologic tolerance in utero? Proc. Natl. Acad. Sci. USA 1990, 87, 6599–6603. [Google Scholar] [CrossRef] [PubMed]
- Kim, C.M.; Koike, K.; Saito, I.; Miyamura, T.; Jay, G. HBx gene of hepatitis B virus induces liver cancer in transgenic mice. Nature 1991, 351, 317–320. [Google Scholar] [CrossRef] [PubMed]
- Koike, K.; Moriya, K.; Ishibashi, K.; Matsuura, Y.; Suzuki, T.; Saito, I.; Iino, S.; Kurokawa, K.; Miyamura, T. Expression of hepatitis C virus envelope proteins in transgenic mice. J. Gen. Virol. 1995, 76, 3031–3038. [Google Scholar] [CrossRef]
- Pasquinelli, C.; Shoenberger, J.M.; Chung, J.; Chang, K.M.I.; Guidotti, L.G.; Selby, M.; Berger, K.; Lesniewski, R.; Houghton, M.; Chisari, F.V. Hepatitis C virus core and E2 protein expression in transgenic mice. Hepatology 1997, 25, 719–727. [Google Scholar] [CrossRef] [PubMed]
- Araki, K.; Miyazaki, J.; Hino, O.; Tomita, N.; Chisaka, O.; Matsubara, K.; Yamamura, K. Expression and replication of hepatitis B virus genome in transgenic mice. Proc. Natl. Acad. Sci. USA 1989, 86, 207–211. [Google Scholar] [CrossRef] [PubMed]
- Guidotti, L.G.; Matzke, B.; Schaller, H.; Chisari, F.V. High-level hepatitis B virus replication in transgenic mice. J. Virol. 1995, 69, 6158–6169. [Google Scholar] [CrossRef]
- Hu, Y.P.; Hu, W.J.; Zheng, W.C.; Li, J.X.; Dai, D.S.; Wang, X.M.; Zhang, S.Z.; Yu, H.Y.; Sun, W.; Hao, G.R. Establishment of transgenic mouse harboring hepatitis B virus (adr subtype) genomes. World J. Gastroenterol. 2001, 7, 111–114. [Google Scholar] [CrossRef]
- Matsuda, J.I.; Suzuki, M.; Nozaki, C.; Shinya, N.; Tashiro, K.; Mizuno, K.; Uchinuno, Y.; Yamamura, K.I. Transgenic mouse expressing a full-length hepatitis C virus cDNA. Jpn. J. Cancer Res. 1998, 89, 150–158. [Google Scholar] [CrossRef]
- Julander, J.G.; Sidwell, R.W.; Morrey, J.D. Characterizing antiviral activity of adefovir dipivoxil in transgenic mice expressing hepatitis B virus. Antivir. Res. 2002, 55, 27–40. [Google Scholar] [CrossRef]
- Julander, J.G.; Colonno, R.J.; Sidwell, R.W.; Morrey, J.D. Characterization of antiviral activity of entecavir in transgenic mice expressing hepatitis B virus. Antivir. Res. 2003, 59, 155–161. [Google Scholar] [CrossRef]
- Li, Q.; Zhang, H.; Qi, Y.; Wang, J.; Li, J.; Niu, J. Antiviral effects of a niobium-substituted heteropolytungstate on hepatitis B virus-transgenic mice. Drug Dev. Res. 2019, 80, 1062–1070. [Google Scholar] [CrossRef]
- Tokunaga, Y.; Osawa, Y.; Ohtsuki, T.; Hayashi, Y.; Yamaji, K.; Yamane, D.; Hara, M.; Munekata, K.; Tsukiyama-Kohara, K.; Hishima, T.; et al. Selective inhibitor of Wnt/β-catenin/CBP signaling ameliorates hepatitis C virus-induced liver fibrosis in mouse model. Sci. Rep. 2017, 7, 325. [Google Scholar] [CrossRef]
- Satoh, K.; Takahashi, H.; Matsuda, C.; Tanaka, T.; Miyasaka, M.; Zeniya, M.; Kohara, M. Natural killer cells target HCV core proteins during the innate immune response in HCV transgenic mice. J. Med. Virol. 2010, 82, 1545–1553. [Google Scholar] [CrossRef]
- Chouteau, P.; Defer, N.; Florimond, A.; Caldéraro, J.; Higgs, M.; Gaudin, A.; Mérour, E.; Dhumeaux, D.; Lerat, H.; Pawlotsky, J.M. Hepatitis C virus (HCV) protein expression enhances hepatic fibrosis in HCV transgenic mice exposed to a fibrogenic agent. J. Hepatol. 2012, 57, 499–507. [Google Scholar] [CrossRef]
- Lu, J.W.; Yang, W.Y.; Lin, Y.M.; Jin, S.L.C.; Yuh, C.H. Hepatitis B virus X antigen and aflatoxin B1 synergistically cause hepatitis, steatosis and liver hyperplasia in transgenic zebrafish. Acta Histochem. 2013, 115, 728–739. [Google Scholar] [CrossRef]
- Ohashi, K.; Marion, P.L.; Nakai, H.; Meuse, L.; Cullen, J.M.; Bordier, B.B.; Schwali, R.; Greenberg, H.B.; Glenn, J.S.; Kay, M.A. Sustained survival of human hepatocytes in mice: A model for in vivo infection with human hepatitis B and hepatitis delta viruses. Nat. Med. 2000, 6, 327–331. [Google Scholar] [CrossRef] [PubMed]
- Maeda, N.; Watanabe, M.; Okamoto, S.; Kanai, T.; Yamada, T.; Hata, J.; Hozumi, N.; Katsume, A.; Nuriya, H.; Sandhu, J.; et al. Hepatitis C virus infection in human liver tissue engrafted in mice with an infectious molecular clone. Liver Int. 2004, 24, 259–267. [Google Scholar] [CrossRef] [PubMed]
- Dandri, M.; Burda, M.R.; Török, E.; Polok, J.M.; Iwanska, A.; Sommer, G.; Rogiers, X.; Rogler, C.E.; Gupta, S.; Will, H.; et al. Repopulation of mouse liver with human hepatocytes and in vivo infection with hepatitis B virus. Hepatology 2001, 33, 981–988. [Google Scholar] [CrossRef] [PubMed]
- Dandri, M.; Burda, M.R.; Zuckerman, D.M.; Wursthorn, K.; Matschl, U.; Pollok, J.M.; Rogiers, X.; Gocht, A.; Köck, J.; Blum, H.E.; et al. Chronic infection with hepatitis B viruses and antiviral drug evaluation in uPA mice after liver repopulation with tupaia hepatocytes. J. Hepatol. 2005, 42, 54–60. [Google Scholar] [CrossRef]
- Hasegawa, M.; Kawai, K.; Mitsui, T.; Taniguchi, K.; Monnai, M.; Wakui, M.; Ito, M.; Suematsu, M.; Peltz, G.; Nakamura, M.; et al. The reconstituted “humanized liver” in TK-NOG mice is mature and functional. Biochem. Biophys. Res. Commun. 2011, 405, 405–410. [Google Scholar] [CrossRef]
- Petersen, J.; Dandri, M.; Mier, W.; Lütgehetmann, M.; Volz, T.; Von Weizsäcker, F.; Haberkorn, U.; Fischer, L.; Pollok, J.M.; Erbes, B.; et al. Prevention of hepatitis B virus infection in vivo by entry inhibitors derived from the large envelope protein. Nat. Biotechnol. 2008, 26, 335–341. [Google Scholar] [CrossRef]
- Meuleman, P.; Libbrecht, L.; De Vos, R.; De Hemptinne, B.; Gevaert, K.; Vandekerckhove, J.; Roskams, T.; Leroux-Roels, G. Morphological and biochemical characterization of a human liver in a uPA-SCID mouse chimera. Hepatology 2005, 41, 847–856. [Google Scholar] [CrossRef]
- Lütgehetmann, M.; Mancke, L.V.; Volz, T.; Helbig, M.; Allweiss, L.; Bornscheuer, T.; Pollok, J.M.; Lohse, A.W.; Petersen, J.; Urban, S.; et al. Humanized chimeric uPA mouse model for the study of hepatitis B and D virus interactions and preclinical drug evaluation. Hepatology 2012, 55, 685–694. [Google Scholar] [CrossRef]
- Sayed, I.M.; Verhoye, L.; Montpellier, C.; Abravanel, F.; Izopet, J.; Cocquerel, L.; Meuleman, P. Study of hepatitis E virus ORF2 antigen kinetics in human-liver chimeric mice and its impact on HEV diagnosis. J. Infect. Dis. 2019, 220, 811–819. [Google Scholar] [CrossRef]
- Allweiss, L.; Gass, S.; Giersch, K.; Groth, A.; Kah, J.; Volz, T.; Rapp, G.; Schöbel, A.; Lohse, A.W.; Polywka, S.; et al. Human liver chimeric mice as a new model of chronic hepatitis e virus infection and preclinical drug evaluation. J. Hepatol. 2016, 64, 1033–1040. [Google Scholar] [CrossRef] [PubMed]
- Van De Garde, M.D.B.; Pas, S.D.; Van Oord, G.W.; Gama, L.; Choi, Y.; De Man, R.A.; Boonstra, A.; Vanwolleghem, T. Interferon-alpha treatment rapidly clears Hepatitis e virus infection in humanized mice. Sci. Rep. 2017, 7, 1–9. [Google Scholar] [CrossRef]
- van de Garde, M.D.B.; Pas, S.D.; van der Net, G.; de Man, R.A.; Osterhaus, A.D.M.E.; Haagmans, B.L.; Boonstra, A.; Vanwolleghem, T. Hepatitis E Virus (HEV) Genotype 3 Infection of Human Liver Chimeric Mice as a Model for Chronic HEV Infection. J. Virol. 2016, 9, 4394–4401. [Google Scholar] [CrossRef]
- Sayed, I.M.; Meuleman, P. Updates in Hepatitis E virus (HEV) field; lessons learned from human liver chimeric mice. Rev. Med. Virol. 2020, 30, e2086. [Google Scholar] [CrossRef] [PubMed]
- Azuma, H.; Paulk, N.; Ranade, A.; Dorrell, C.; Al-Dhalimy, M.; Ellis, E.; Strom, S.; Kay, M.A.; Finegold, M.; Grompe, M. Robust expansion of human hepatocytes in Fah-/-/Rag2 -/-/Il2rg-/- mice. Nat. Biotechnol. 2007, 25, 903–910. [Google Scholar] [CrossRef]
- Hopkins, S.; Gallay, P. Cyclophilin inhibitors: An emerging class of therapeutics for the treatment of chronic hepatitis C infection. Viruses 2012, 4, 2558–2577. [Google Scholar] [CrossRef]
- Bissig, K.D.; Wieland, S.F.; Tran, P.; Isogawa, M.; Le, T.T.; Chisari, F.V.; Verma, I.M. Human liver chimeric mice provide a model for hepatitis B and C virus infection and treatment. J. Clin. Invest. 2010, 120, 924–930. [Google Scholar] [CrossRef]
- Sayed, I.M.; Foquet, L.; Verhoye, L.; Abravanel, F.; Farhoudi, A.; Leroux-Roels, G.; Izopet, J.; Meuleman, P. Transmission of hepatitis E virus infection to human-liver chimeric FRG mice using patient plasma. Antivir. Res. 2017, 141, 150–154. [Google Scholar] [CrossRef]
- Vaughan, A.M.; Kappe, S.H.I.; PLoSs, A.; Mikolajczak, S.A. Development of humanized mouse models to study human malaria parasite infection. Future Microbiol. 2012, 7, 657–665. [Google Scholar] [CrossRef] [PubMed]
- Kruse, R.L.; Shum, T.; Tashiro, H.; Barzi, M.; Yi, Z.; Whitten-Bauer, C.; Legras, X.; Bissig-Choisat, B.; Garaigorta, U.; Gottschalk, S.; et al. HBsAg-redirected T cells exhibit antiviral activity in HBV-infected human liver chimeric mice. Cytotherapy 2018, 20, 697–705. [Google Scholar] [CrossRef]
- Yuan, L.; Liu, X.; Zhang, L.; Li, X.; Zhang, Y.; Wu, K.; Chen, Y.; Cao, J.; Hou, W.; Zhang, J.; et al. A chimeric humanized mouse model by engrafting the human induced pluripotent stem cell-derived hepatocyte-like cell for the chronic hepatitis B virus infection. Front. Microbiol. 2018, 9, 908. [Google Scholar] [CrossRef]
- Kosaka, K.; Hiraga, N.; Imamura, M.; Yoshimi, S.; Murakami, E.; Nakahara, T.; Honda, Y.; Ono, A.; Kawaoka, T.; Tsuge, M.; et al. A novel TK-NOG based humanized mouse model for the study of HBV and HCV infections. Biochem. Biophys. Res. Commun. 2013, 441, 230–235. [Google Scholar] [CrossRef]
- Murai, K.; Hikita, H.; Kai, Y.; Kondo, Y.; Fukuoka, M.; Fukutomi, K.; Doi, A.; Yamai, T.; Nakabori, T.; Fukuda, R.; et al. Hepatitis C virus infection suppresses hepatitis B virus replication via the RIG-I-like helicase pathway. Sci. Rep. 2020, 10, 941. [Google Scholar] [CrossRef]
- Nakabori, T.; Hikita, H.; Murai, K.; Nozaki, Y.; Kai, Y.; Makino, Y.; Saito, Y.; Tanaka, S.; Wada, H.; Eguchi, H.; et al. Sodium taurocholate cotransporting polypeptide inhibition efficiently blocks hepatitis B virus spread in mice with a humanized liver. Sci. Rep. 2016, 6, 27782. [Google Scholar] [CrossRef] [PubMed]
- Kanbe, A.; Ishikawa, T.; Hara, A.; Suemizu, H.; Nanizawa, E.; Tamaki, Y.; Ito, H. Novel hepatitis B virus infection mouse model using herpes simplex virus type 1 thymidine kinase transgenic mice. J. Gastroenterol. Hepatol. 2020, 36, 782–789. [Google Scholar] [CrossRef]
- Wang, W.; Smith, N.; Makarov, E.; Sun, Y.; Gebhart, C.L.; Ganesan, M.; Osna, N.A.; Gendelman, H.E.; Edagwa, B.J.; Poluektova, L.Y. A long-acting 3TC ProTide nanoformulation suppresses HBV replication in humanized mice. Nanomed. Nanotechnol. Biol. Med. 2020, 28, 102185. [Google Scholar] [CrossRef] [PubMed]
- Washburn, M.L.; Bility, M.T.; Zhang, L.; Kovalev, G.I.; Buntzman, A.; Frelinger, J.A.; Barry, W.; PLoSs, A.; Rice, C.M.; Su, L. A humanized mouse model to study hepatitis C virus infection, immune response, and liver disease. Gastroenterology 2011, 140, 1334–1344. [Google Scholar] [CrossRef]
- Robinet, E.; Baumert, T.F. A first step towards a mouse model for hepatitis C virus infection containing a human immune system. J. Hepatol. 2011, 55, 718–720. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Yuan, L.; Jiang, J.; Liu, X.; Zhang, Y.; Zhang, L.; Xin, J.; Wu, K.; Li, X.; Cao, J.; Guo, X.; et al. HBV infection-induced liver cirrhosis development in dual-humanised mice with human bone mesenchymal stem cell transplantation. Gut 2019, 68, 2044–2056. [Google Scholar] [CrossRef]
- Dusséaux, M.; Masse-Ranson, G.; Darche, S.; Ahodantin, J.; Li, Y.; Fiquet, O.; Beaumont, E.; Moreau, P.; Rivière, L.; Neuveut, C.; et al. Viral Load Affects the Immune Response to HBV in Mice With Humanized Immune System and Liver. Gastroenterology 2017, 153, 1647–1661. [Google Scholar] [CrossRef] [PubMed]
- Pileri, P.; Uematsu, Y.; Campagnoli, S.; Galli, G.; Falugi, F.; Petracca, R.; Weiner, A.J.; Houghton, M.; Rosa, D.; Grandi, G.; et al. Binding of hepatitis C virus to CD81. Science 1998, 282, 938–941. [Google Scholar] [CrossRef] [PubMed]
- Bitzegeio, J.; Bankwitz, D.; Hueging, K.; Haid, S.; Brohm, C.; Zeisel, M.B.; Herrmann, E.; Iken, M.; Ott, M.; Baumert, T.F.; et al. Adaptation of Hepatitis C Virus to Mouse CD81 Permits Infection of Mouse Cells in the Absence of Human Entry Factors. PLoS Pathog. 2010, 6, e1000978. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Q.; Guo, J.-T.; Seeger, C. Replication of Hepatitis C Virus Subgenomes in Nonhepatic Epithelial and Mouse Hepatoma Cells. J. Virol. 2003, 77, 9204–9210. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Uprichard, S.L.; Chung, J.; Chisari, F.V.; Wakita, T. Replication of a hepatitis C virus replicon clone in mouse cells. Virol. J. 2006, 3, 89. [Google Scholar] [CrossRef][Green Version]
- Liu, S.; Yang, W.; Shen, L.; Turner, J.R.; Coyne, C.B.; Wang, T. Tight Junction Proteins Claudin-1 and Occludin Control Hepatitis C Virus Entry and Are Downregulated during Infection To Prevent Superinfection. J. Virol. 2009, 83, 2011–2014. [Google Scholar] [CrossRef]
- von Schaewen, M.; Dorner, M.; Hueging, K.; Foquet, L.; Gerges, S.; Hrebikova, G.; Heller, B.; Bitzegeio, J.; Doerrbecker, J.; Horwitz, J.A.; et al. Expanding the host range of hepatitis C virus through viral adaptation. MBio 2016, 7, e01915-16. [Google Scholar] [CrossRef]
- Scull, M.A.; Shi, C.; de Jong, Y.P.; Gerold, G.; Ries, M.; von Schaewen, M.; Donovan, B.M.; Labitt, R.N.; Horwitz, J.A.; Gaska, J.M.; et al. Hepatitis C Virus Infects Rhesus Macaque Hepatocytes and Simianized Mice. Hepatology 2015, 62, 57–67. [Google Scholar] [CrossRef]
- Huang, M.; Sun, R.; Huang, Q.; Tian, Z. Technical improvement and application of hydrodynamic gene delivery in study of liver diseases. Front. Pharmacol. 2017, 8, 591. [Google Scholar] [CrossRef]
- Sendra, L.; Herrero, M.J.; Aliño, S.F. Translational advances of hydrofection by hydrodynamic injection. Genes 2018, 9, 136. [Google Scholar] [CrossRef]
- Zhang, Z.H.; Wu, C.C.; Chen, X.W.; Li, X.; Li, J.; Lu, M.J. Genetic variation of hepatitis B virus and its significance for pathogenesis. World J. Gastroenterol. 2016, 22, 126–144. [Google Scholar] [CrossRef]
- Hartman, Z.C.; Kiang, A.; Everett, R.S.; Serra, D.; Yang, X.Y.; Clay, T.M.; Amalfitano, A. Adenovirus Infection Triggers a Rapid, MyD88-Regulated Transcriptome Response Critical to Acute-Phase and Adaptive Immune Responses In Vivo. J. Virol. 2007, 81, 1796–1812. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Zhu, J.; Zhang, Y.; Li, Y.; Ma, T.; Li, Q.; Xu, J.; Xu, L. The doses of plasmid backbone plays a major role in determining the HBV clearance in hydrodynamic injection mouse model. Virol. J. 2018, 15, 89. [Google Scholar] [CrossRef] [PubMed]
- Gao, X.; Chi, X.; Wang, X.; Wu, R.; Xu, H.; Zhan, M.; Li, D.; Ding, Y.; Xu, D.; Niu, J. IL-33 Inhibits Hepatitis B Virus through Its Receptor ST2 in Hydrodynamic HBV Mouse Model. Mediat. Inflamm. 2020, 2020. [Google Scholar] [CrossRef] [PubMed]
- Yang, P.L.; Althage, A.; Chung, J.; Chisari, F.V. Hydrodynamic injection of viral DNA: A mouse model of acute hepatitis B virus infection. Proc. Natl. Acad. Sci. USA 2002, 99, 13825–13830. [Google Scholar] [CrossRef]
- Sprinzl, M.F.; Oberwinkler, H.; Schaller, H.; Protzer, U. Transfer of Hepatitis B Virus Genome by Adenovirus Vectors into Cultured Cells and Mice: Crossing the Species Barrier. J. Virol. 2001, 75, 5108–5118. [Google Scholar] [CrossRef]
- Ma, Z.; Liu, J.; Wu, W.; Zhang, E.; Zhang, X.; Li, Q.; Zelinskyy, G.; Buer, J.; Dittmer, U.; Kirschning, C.J.; et al. The IL-1R/TLR signaling pathway is essential for efficient CD8 + T-cell responses against hepatitis B virus in the hydrodynamic injection mouse model. Cell. Mol. Immunol. 2017, 14, 997–1008. [Google Scholar] [CrossRef]
- Reherman, B. Pathogenesis of chronic viral hepatitis: Differential roles of T cells and NK cells. Nat. Med. 2013, 19, 859–868. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Yang, J.; Zhao, Y.; Song, Y.; Yin, S.; Guo, J.; Zhang, H.; Wang, K.; Wei, L.; Li, S.; et al. MCPIP1 inhibits Hepatitis B virus replication by destabilizing viral RNA and negatively regulates the virus-induced innate inflammatory responses. Antivir. Res. 2020, 174, 104705. [Google Scholar] [CrossRef] [PubMed]
- Chuai, X.; Wang, W.; Chen, H.; Deng, Y.; Wen, B.; Tan, W. Lentiviral backbone-based hepatitis B virus replicon-mediated transfer favours the establishment of persistent hepatitis B virus infection in mice after hydrodynamic injection. Antivir. Res. 2014, 101, 68–74. [Google Scholar] [CrossRef]
- Huang, L.; Wu, H.; Chen, P.; Chen, D. An immunocompetent mouse model for the tolerance of human chronic hepatitis B virus infection. J. Clin. Virol. 2006, 36, S74. [Google Scholar] [CrossRef]
- Peng, X.H.; Ren, X.N.; Chen, L.X.; Shi, B.S.; Xu, C.H.; Fang, Z.; Liu, X.; Chen, J.L.; Zhang, X.N.; Hu, Y.W.; et al. High persistence rate of hepatitis B virus in a hydrodynamic injection-based transfection model in C3H/HeN mice. World J. Gastroenterol. 2015, 21, 3527–3536. [Google Scholar] [CrossRef] [PubMed]
- Chou, H.; Chien, W.; Wu, L.; Cheng, C.; Chung, C.; Horng, J. Age-related immune clearance of hepatitis B virus infection requires the establishment of gut microbiota. Proc. Natl. Acad. Sci. USA 2015, 112, 2175–2180. [Google Scholar] [CrossRef]
- Dion, S.; Bourgine, M.; Godon, O.; Levillayer, F.; Michel, M.-L. Adeno-Associated Virus-Mediated Gene Transfer Leads to Persistent Hepatitis B Virus Replication in Mice Expressing HLA-A2 and HLA-DR1 Molecules. J. Virol. 2013, 87, 5554–5563. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Dong, Q.; Li, Q.; Li, Y.; Zhao, D.; Sun, J.; Fu, J.; Meng, F.; Lin, H.; Luan, J.; et al. Dysregulated Response of Follicular Helper T Cells to Hepatitis B Surface Antigen Promotes HBV Persistence in Mice and Associates With Outcomes of Patients. Gastroenterology 2018, 154, 2222–2236. [Google Scholar] [CrossRef]
- McCaffrey, A.; Nakai, H.; Pandey, K.; Haung, Z.; Salazar, F.; Xu, H.; Wieland, S.; Marion, P.; Kay, M. Inhibition of hepatitis B virus replication by RNA interference. Nat. Biotechnol. 2003, 21, 639–644. [Google Scholar] [CrossRef]
- Wu, H.L.; Huang, L.R.; Huang, C.C.; Lai, H.L.; Liu, C.J.; Huang, Y.T.; Hsu, Y.W.; Lu, C.Y.; Chen, D.S.; Chen, P.J. RNA interference-mediated control of hepatitis B virus and emergence of resistant mutant. Gastroenterology 2005, 128, 708–716. [Google Scholar] [CrossRef] [PubMed]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Khoshdel-Rad, N.; Zahmatkesh, E.; Bikmulina, P.; Peshkova, M.; Kosheleva, N.; Bezrukov, E.A.; Sukhanov, R.B.; Solovieva, A.; Shpichka, A.; Timashev, P.; et al. Modeling Hepatotropic Viral Infections: Cells vs. Animals. Cells 2021, 10, 1726. https://doi.org/10.3390/cells10071726
Khoshdel-Rad N, Zahmatkesh E, Bikmulina P, Peshkova M, Kosheleva N, Bezrukov EA, Sukhanov RB, Solovieva A, Shpichka A, Timashev P, et al. Modeling Hepatotropic Viral Infections: Cells vs. Animals. Cells. 2021; 10(7):1726. https://doi.org/10.3390/cells10071726
Chicago/Turabian StyleKhoshdel-Rad, Niloofar, Ensieh Zahmatkesh, Polina Bikmulina, Maria Peshkova, Nastasia Kosheleva, Evgeny A. Bezrukov, Roman B. Sukhanov, Anna Solovieva, Anastasia Shpichka, Peter Timashev, and et al. 2021. "Modeling Hepatotropic Viral Infections: Cells vs. Animals" Cells 10, no. 7: 1726. https://doi.org/10.3390/cells10071726
APA StyleKhoshdel-Rad, N., Zahmatkesh, E., Bikmulina, P., Peshkova, M., Kosheleva, N., Bezrukov, E. A., Sukhanov, R. B., Solovieva, A., Shpichka, A., Timashev, P., & Vosough, M. (2021). Modeling Hepatotropic Viral Infections: Cells vs. Animals. Cells, 10(7), 1726. https://doi.org/10.3390/cells10071726







