miRNA Changes in Retinal Ganglion Cells after Optic Nerve Crush and Glaucomatous Damage
Abstract
1. Introduction
2. Materials and Methods
2.1. Animals
2.2. Materials
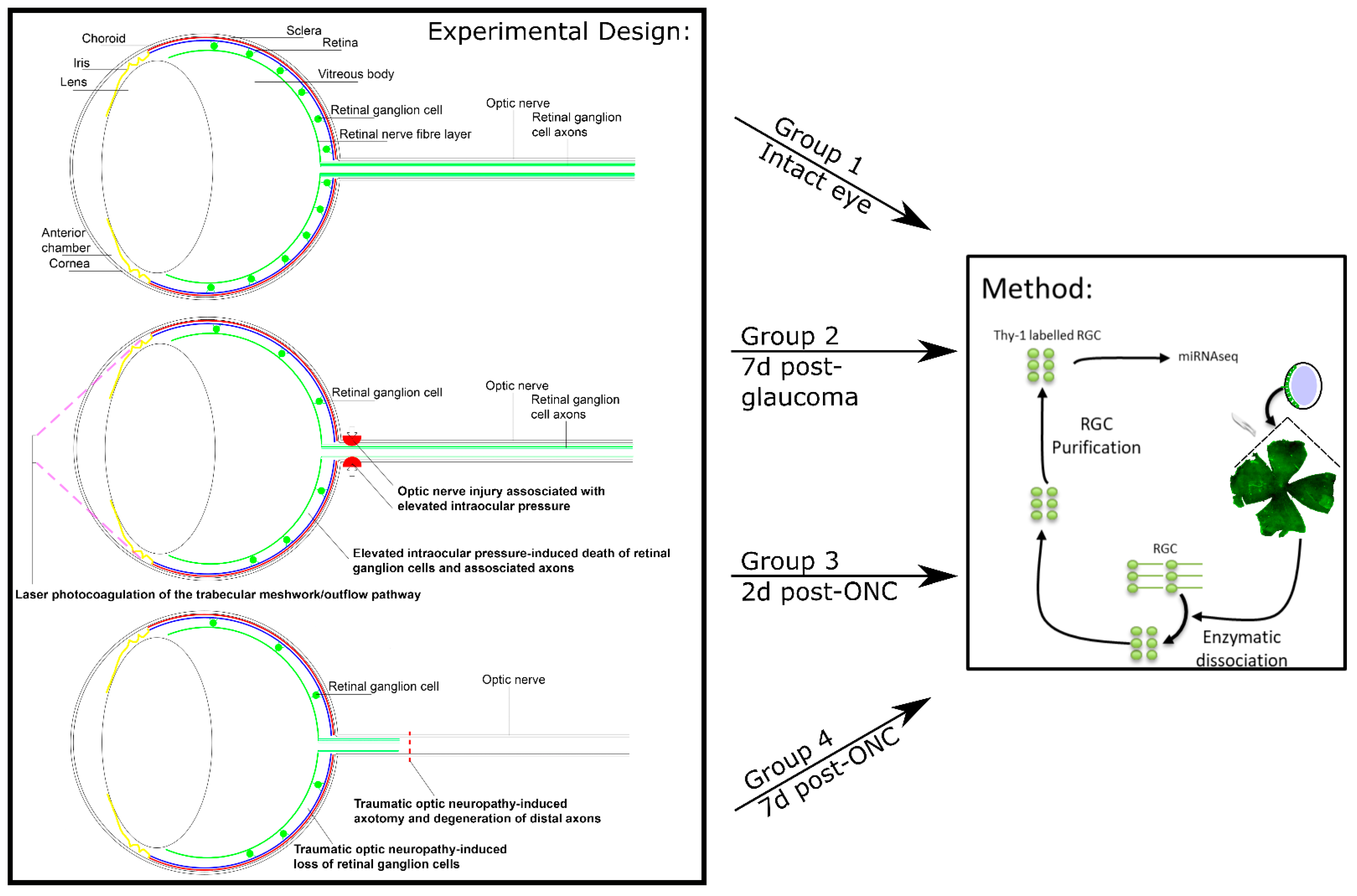
2.3. In Vivo Experimental Design
2.4. Induction of Ocular Hypertension with Laser Photocoagulation (Rats)
2.5. Induction of Ocular Hypertension with Silicon Oil Injection (Mice)
2.6. Intraocular Pressure Recording
2.7. Optic Nerve Crush
2.8. Isolation, Purification, and/or Culture of Retinal Ganglion Cells
2.9. RNA Sequencing
2.10. In Situ Hybridization-Mediated Localization of miRNA in Mouse Retina
2.11. miRNA Modulation Treatment of Heterogeneous Retinal Cultures
2.12. Immunocytochemistry and Microscopy
2.13. Statistics
3. Results
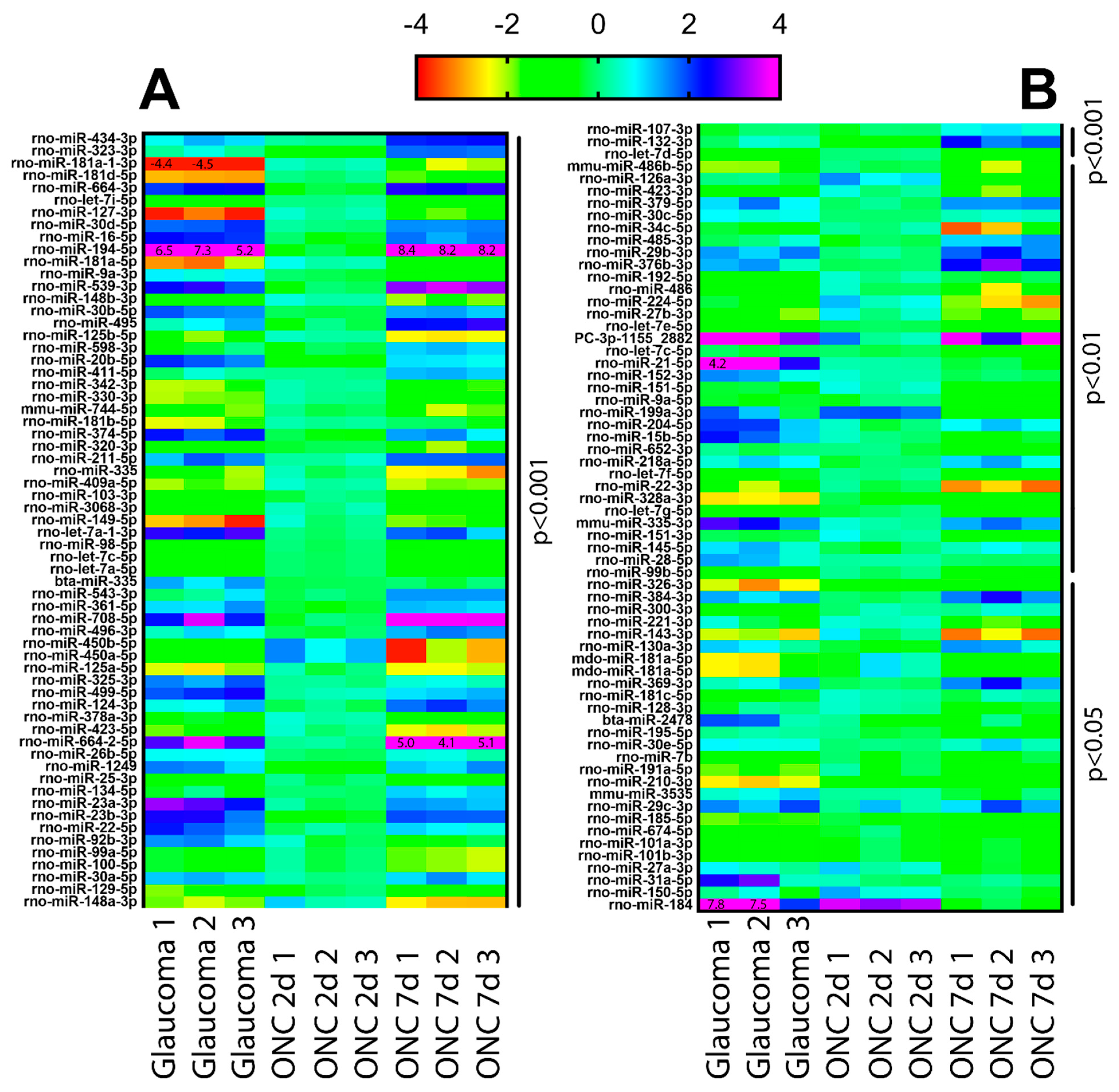
3.1. Ocular Hypertension and TON Induce Significant Changes in RGC miRNA
3.2. Changes in miR-181d-5p Level in the Retina after IOP Elevation
3.3. Modulation of Candidate miRNA in RGC Promotes Neuroprotection and Neuritogenesis
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Berry, M.; Ahmed, Z.; Lorber, B.; Douglas, M.; Logan, A. Regeneration of axons in the visual system. Restor. Neurol. Neurosci. 2008, 26, 147–174. [Google Scholar]
- Berkelaar, M.; Clarke, D.B.; Wang, Y.C.; Bray, G.M.; Aguayo, A.J. Axotomy results in delayed death and apoptosis of retinal ganglion cells in adult rats. J. Neurosci. Off. J. Soc. Neurosci. 1994, 14, 4368–4374. [Google Scholar] [CrossRef]
- Liddelow, S.A.; Guttenplan, K.A.; Clarke, L.E.; Bennett, F.C.; Bohlen, C.J.; Schirmer, L.; Bennett, M.L.; Münch, A.E.; Chung, W.-S.; Peterson, T.C.; et al. Neurotoxic reactive astrocytes are induced by activated microglia. Nature 2017, 541, 481. [Google Scholar] [CrossRef]
- Tham, Y.-C.; Li, X.; Wong, T.Y.; Quigley, H.A.; Aung, T.; Cheng, C.-Y. Global prevalence of glaucoma and projections of glaucoma burden through 2040. Ophthalmology 2014, 121, 2081–2090. [Google Scholar] [CrossRef]
- Almasieh, M.; Wilson, A.M.; Morquette, B.; Cueva Vargas, J.L.; Di Polo, A. The molecular basis of retinal ganglion cell death in glaucoma. Prog. Retin. Eye Res. 2012, 31, 152–181. [Google Scholar] [CrossRef] [PubMed]
- Calkins, D.J. Adaptive responses to neurodegenerative stress in glaucoma. Prog. Retin. Eye Res. 2021, 100953. [Google Scholar] [CrossRef] [PubMed]
- Mélik Parsadaniantz, S.; Réaux-le Goazigo, A.; Sapienza, A.; Habas, C.; Baudouin, C. Glaucoma: A degenerative optic neuropathy related to neuroinflammation? Cells 2020, 9, 535. [Google Scholar] [CrossRef] [PubMed]
- Donahue, R.J.; Maes, M.E.; Grosser, J.A.; Nickells, R.W. Bax-depleted retinal ganglion cells survive and become quiescent following optic nerve damage. Mol. Neurobiol. 2020, 57, 1070–1084. [Google Scholar] [CrossRef]
- Libby, R.T.; Li, Y.; Savinova, O.V.; Barter, J.; Smith, R.S.; Nickells, R.W.; John, S.W.M. Susceptibility to neurodegeneration in a glaucoma is modified by bax gene dosage. PLoS Genet. 2005, 1, e4. [Google Scholar] [CrossRef]
- Mead, B.; Berry, M.; Logan, A.; Scott, R.A.H.; Leadbeater, W.; Scheven, B.A. Stem cell treatment of degenerative eye disease. Stem Cell Res. 2015, 14, 243–257. [Google Scholar] [CrossRef]
- Mead, B.; Tomarev, S. Extracellular vesicle therapy for retinal diseases. Prog. Retin. Eye Res. 2020, 79, 100849. [Google Scholar] [CrossRef]
- Park, Y.H.; Snook, J.D.; Ostrin, E.J.; Kim, S.; Chen, R.; Frankfort, B.J. Transcriptomic profiles of retinal ganglion cells are defined by the magnitude of intraocular pressure elevation in adult mice. Sci. Rep. 2019, 9, 2594. [Google Scholar] [CrossRef] [PubMed]
- Rheaume, B.A.; Jereen, A.; Bolisetty, M.; Sajid, M.S.; Yang, Y.; Renna, K.; Sun, L.; Robson, P.; Trakhtenberg, E.F. Single cell transcriptome profiling of retinal ganglion cells identifies cellular subtypes. Nat. Commun. 2018, 9, 2759. [Google Scholar] [CrossRef] [PubMed]
- Jayaram, H.; Cepurna, W.O.; Johnson, E.C.; Morrison, J.C. Microrna expression in the glaucomatous retina. Investig. Ophthalmol. Vis. Sci. 2015, 56, 7971–7982. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Chen, Y.; Wang, Y.; Zhang, X.; Gao, K.; Chen, S.; Zhang, X. Microrna profiling in glaucoma eyes with varying degrees of optic neuropathy by using next-generation sequencing. Investig. Ophthalmol. Vis. Sci. 2018, 59, 2955–2966. [Google Scholar] [CrossRef]
- Zuzic, M.; Rojo Arias, J.E.; Wohl, S.G.; Busskamp, V. Retinal mirna functions in health and disease. Genes 2019, 10, 377. [Google Scholar] [CrossRef]
- Brennecke, J.; Stark, A.; Russell, R.B.; Cohen, S.M. Principles of microrna–target recognition. PLoS Biol. 2005, 3, e85. [Google Scholar] [CrossRef] [PubMed]
- Emde, A.; Hornstein, E. Mirnas at the interface of cellular stress and disease. EMBO J. 2014, 33, 1428–1437. [Google Scholar] [CrossRef] [PubMed]
- Leung, A.K.; Sharp, P.A. Microrna functions in stress responses. Mol. Cell 2010, 40, 205–215. [Google Scholar] [CrossRef]
- DeVeale, B.; Swindlehurst-Chan, J.; Blelloch, R. The roles of micrornas in mouse development. Nat. Rev. Genet. 2021, 22, 307–323. [Google Scholar] [CrossRef] [PubMed]
- Levkovitch-Verbin, H.; Quigley, H.A.; Martin, K.R.; Valenta, D.; Baumrind, L.A.; Pease, M.E. Translimbal laser photocoagulation to the trabecular meshwork as a model of glaucoma in rats. Investig. Ophthalmol. Vis. Sci 2002, 43, 402–410. [Google Scholar]
- Mead, B.; Amaral, J.; Tomarev, S. Mesenchymal stem cell–derived small extracellular vesicles promote neuroprotection in rodent models of glaucoma. Investig. Ophthalmol. Vis. Sci. 2018, 59, 702–714. [Google Scholar] [CrossRef]
- Zhang, J.; Li, L.; Huang, H.; Fang, F.; Webber, H.C.; Zhuang, P.; Liu, L.; Dalal, R.; Tang, P.H.; Mahajan, V.B.; et al. Silicone oil-induced ocular hypertension and glaucomatous neurodegeneration in mouse. eLife 2019, 8, e45881. [Google Scholar] [CrossRef] [PubMed]
- Berry, M.; Carlile, J.; Hunter, A. Peripheral nerve explants grafted into the vitreous body of the eye promote the regeneration of retinal ganglion cell axons severed in the optic nerve. J. Neurocytol. 1996, 25, 147–170. [Google Scholar] [CrossRef] [PubMed]
- Wohl, S.G.; Reh, T.A. The microrna expression profile of mouse müller glia in vivo and in vitro. Sci. Rep. 2016, 6, 35423. [Google Scholar] [CrossRef] [PubMed]
- Shinohara, Y.; Yahagi, K.; Kawano, M.; Nishiyori, H.; Kawazu, C.; Suzuki, N.; Manabe, R.; Hirase, H. Mirna profiling of bilateral rat hippocampal ca3 by deep sequencing. Biochem. Biophys. Res. Commun. 2011, 409, 293–298. [Google Scholar] [CrossRef]
- Yao, M.-j.; Chen, G.; Zhao, P.-p.; Lu, M.-h.; Jian, J.; Liu, M.-f.; Yuan, X.-b. Transcriptome analysis of micrornas in developing cerebral cortex of rat. BMC Genom. 2012, 13, 232. [Google Scholar] [CrossRef]
- Jeon, C.-J.; Strettoi, E.; Masland, R.H. The major cell populations of the mouse retina. J. Neurosci. 1998, 18, 8936. [Google Scholar] [CrossRef] [PubMed]
- Carrella, S.; Barbato, S.; D’Agostino, Y.; Salierno, F.G.; Manfredi, A.; Banfi, S.; Conte, I. Tgf-β controls mir-181/erk regulatory network during retinal axon specification and growth. PLoS ONE 2015, 10, e0144129. [Google Scholar] [CrossRef] [PubMed]
- Suggate, E.L.; Ahmed, Z.; Read, M.L.; Eaton-Charnock, K.; Douglas, M.R.; Gonzalez, A.M.; Berry, M.; Logan, A. Optimisation of sirna-mediated rhoa silencing in neuronal cultures. Mol. Cell. Neurosci. 2009, 40, 451–462. [Google Scholar] [CrossRef]
- Indrieri, A.; Carrella, S.; Carotenuto, P.; Banfi, S.; Franco, B. The pervasive role of the mir-181 family in development, neurodegeneration, and cancer. Int. J. Mol. Sci. 2020, 21, 2092. [Google Scholar] [CrossRef] [PubMed]
- Xu, L.-J.; Ouyang, Y.-B.; Xiong, X.; Stary, C.M.; Giffard, R.G. Post-stroke treatment with mir-181 antagomir reduces injury and improves long-term behavioral recovery in mice after focal cerebral ischemia. Exp. Neurol. 2015, 264, 1–7. [Google Scholar] [CrossRef]
- Indrieri, A.; Carrella, S.; Romano, A.; Spaziano, A.; Marrocco, E.; Fernandez-Vizarra, E.; Barbato, S.; Pizzo, M.; Ezhova, Y.; Golia, F.M.; et al. Mir-181a/b downregulation exerts a protective action on mitochondrial disease models. EMBO Mol. Med. 2019, 11, e8734. [Google Scholar] [CrossRef]
- Wang, X.; You, Z.; Zhao, G.; Wang, T. Microrna-194-5p levels decrease during deep hypothermic circulatory arrest. Sci. Rep. 2018, 8, 14044. [Google Scholar] [CrossRef]
- Monteleone, N.J.; Lutz, C.S. Mir-708-5p: A microrna with emerging roles in cancer. Oncotarget 2017, 8, 71292–71316. [Google Scholar] [CrossRef] [PubMed]
- Berry, M.; Ahmed, Z.; Morgan-Warren, P.; Fulton, D.; Logan, A. Prospects for mtor-mediated functional repair after central nervous system trauma. Neurobiol. Dis. 2016, 85, 99–110. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, K.; Yamaji, R.; Ohtsuki, T. Microrna-664a-5p promotes neuronal differentiation of sh-sy5y cells. Genes Cells 2018, 23, 225–233. [Google Scholar] [CrossRef] [PubMed]
- Cai, R.; Qimuge, N.; Ma, M.; Wang, Y.; Tang, G.; Zhang, Q.; Sun, Y.; Chen, X.; Yu, T.; Dong, W.; et al. Microrna-664-5p promotes myoblast proliferation and inhibits myoblast differentiation by targeting serum response factor and wnt1. J. Biol. Chem. 2018, 293, 19177–19190. [Google Scholar] [CrossRef] [PubMed]
- Nie, X.-G.; Fan, D.-S.; Huang, Y.-X.; He, Y.-Y.; Dong, B.-L.; Gao, F. Downregulation of microrna-149 in retinal ganglion cells suppresses apoptosis through activation of the pi3k/akt signaling pathway in mouse with glaucoma. Am. J. Physiol. Cell Physiol. 2018, 15, C839–C849. [Google Scholar] [CrossRef] [PubMed]
- Li, R.; Jin, Y.; Li, Q.; Sun, X.; Zhu, H.; Cui, H. Mir-93-5p targeting pten regulates the nmda-induced autophagy of retinal ganglion cells via akt/mtor pathway in glaucoma. Biomed. Pharmacother. 2018, 100, 1–7. [Google Scholar] [CrossRef]
- Wang, X.; Archibald, M.L.; Stevens, K.; Baldridge, W.H.; Chauhan, B.C. Cyan fluorescent protein (cfp) expressing cells in the retina of thy1-cfp transgenic mice before and after optic nerve injury. Neurosci. Lett. 2010, 468, 110–114. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez, A.R.; de Sevilla Müller, L.P.; Brecha, N.C. The rna binding protein rbpms is a selective marker of ganglion cells in the mammalian retina. J. Comp. Neurol. 2014, 522, 1411–1443. [Google Scholar] [CrossRef] [PubMed]
- Mead, B.; Cullather, E.; Nakaya, N.; Niu, Y.; Kole, C.; Ahmed, Z.; Tomarev, S. Viral delivery of multiple mirnas promotes retinal ganglion cell survival and functional preservation after optic nerve crush injury. Exp. Eye Res. 2020, 197, 108071. [Google Scholar] [CrossRef] [PubMed]
- Xu, M.; Yang, Q.; Sun, X.; Wang, Y. Recent advancements in the loading and modification of therapeutic exosomes. Front. Bioeng. Biotechnol. 2020, 8, 586130. [Google Scholar] [CrossRef] [PubMed]



| Retinal Ganglion Cells | Muller Glia Wohl and Reh (2016) | Neural Retina without Muller Glia Wohl and Reh (2016) | CA3 Hippocampus Shinohara et al. (2011) | Cortex Yao et al. (2012) |
|---|---|---|---|---|
| let-7g | mir-204 | mir-124 | let-7c | mir-128 |
| mir-182 | mir-125b-5p | mir-183 | let-7a | let-7c |
| mir-125-5p | mir-9 | mir-96 | let-7f | let-7a |
| mir-181a-5p | mir-181a | mir-181a | let-7b | let-7f |
| let-7f | let-7c | let-7g | mir-9 | let-7d |
| mir-29a-3p | mir-720 | mir-1944 | mir-138 | mir-29a |
| mir-127-3p | mir-99a | mir-16 | mir-30e | let-7b |
| let-7d | let-7b | let-7d | mir-126 | mir-124 |
| mir-24a-5p | let-7g | mir-30c | mir-24 | mir-103 |
| mir-9a-5p | mic-30c | mir-29c | mir-143 | let-7e |
| mir-125a-5p | let-7d | mir-720 | mir-21 | mir-107 |
| let-7c-5p | mir-135a | let-7b | mir-127 | mir-99a |
| let-7a | mir-29a | mir-204 | mir-30a | let-7i |
| mir-99a-5p | let-7a | let-7a | mir-26a | mir-30d |
| mir-100-5p | mir-16 | let-7c | mir-9 * | mir-9 |
| mir-99b-5p | mir-125a-5p | mir-211 | mir-29b | mir-30a |
| let-7i | mir-100 | mir-125b-5p | mir-103 | mir-101a |
| mir-183-5p | mir-1944 | mir-129-3p | mir-27b | mir-219-2-3p |
| mir-7a-5p | mir-22 | mir-9 | mir-101a | mir-185 |
| mir-96-5p | mir-30d | mir-25 | mir-30d | |
| mir-30d-5p | let-7e | mir-22 | mir-181b | |
| mir-30c-5p | mir-335-5p | mir-342-3p | mir-379 | |
| mir-181b-5p | mir-23a | mir-125a-5p | mir-101b | |
| mir-204-5p | let-7f | mir-132 | ||
| let-7c-5p | mir-151-5p | mir-125b-5p | ||
| let-7b-5p | mir-1186 | mir-218 | ||
| mir-191a-5p | let-7e | mir-411 | ||
| mir-126a-3p | mir-182 | mir-378 | ||
| mir-143-3p | mir-451 | let-7i | ||
| mir-151-5p | mir-29a | mir-124 | ||
| let-7e-5p | mir-30d | mir-146b | ||
| mir-124-3p | mir-210 | let-7d | ||
| mir-26b-5p | let-7i | |||
| mir-9a-3p | mir-500 | |||
| mir-30e-5p | mir-15b | |||
| mir-30a-5p | mir-1224 | |||
| mir-30b-5p | mir-301a | |||
| mir-7b | mir-1937a + b | |||
| mir-129-2-3p | mir-15a |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mead, B.; Kerr, A.; Nakaya, N.; Tomarev, S.I. miRNA Changes in Retinal Ganglion Cells after Optic Nerve Crush and Glaucomatous Damage. Cells 2021, 10, 1564. https://doi.org/10.3390/cells10071564
Mead B, Kerr A, Nakaya N, Tomarev SI. miRNA Changes in Retinal Ganglion Cells after Optic Nerve Crush and Glaucomatous Damage. Cells. 2021; 10(7):1564. https://doi.org/10.3390/cells10071564
Chicago/Turabian StyleMead, Ben, Alicia Kerr, Naoki Nakaya, and Stanislav I. Tomarev. 2021. "miRNA Changes in Retinal Ganglion Cells after Optic Nerve Crush and Glaucomatous Damage" Cells 10, no. 7: 1564. https://doi.org/10.3390/cells10071564
APA StyleMead, B., Kerr, A., Nakaya, N., & Tomarev, S. I. (2021). miRNA Changes in Retinal Ganglion Cells after Optic Nerve Crush and Glaucomatous Damage. Cells, 10(7), 1564. https://doi.org/10.3390/cells10071564







