Recurrent Implication of Striatal Cholinergic Interneurons in a Range of Neurodevelopmental, Neurodegenerative, and Neuropsychiatric Disorders
Abstract
1. Introduction
2. Development of Striatal Cholinergic Interneurons
3. Striatal Cholinergic Interneuron Anatomy
4. Electrophysiological Properties of Striatal Cholinergic Interneurons
4.1. Unique Electrophysiological Profile
4.2. Cholinergic Interneuron “Pauses”
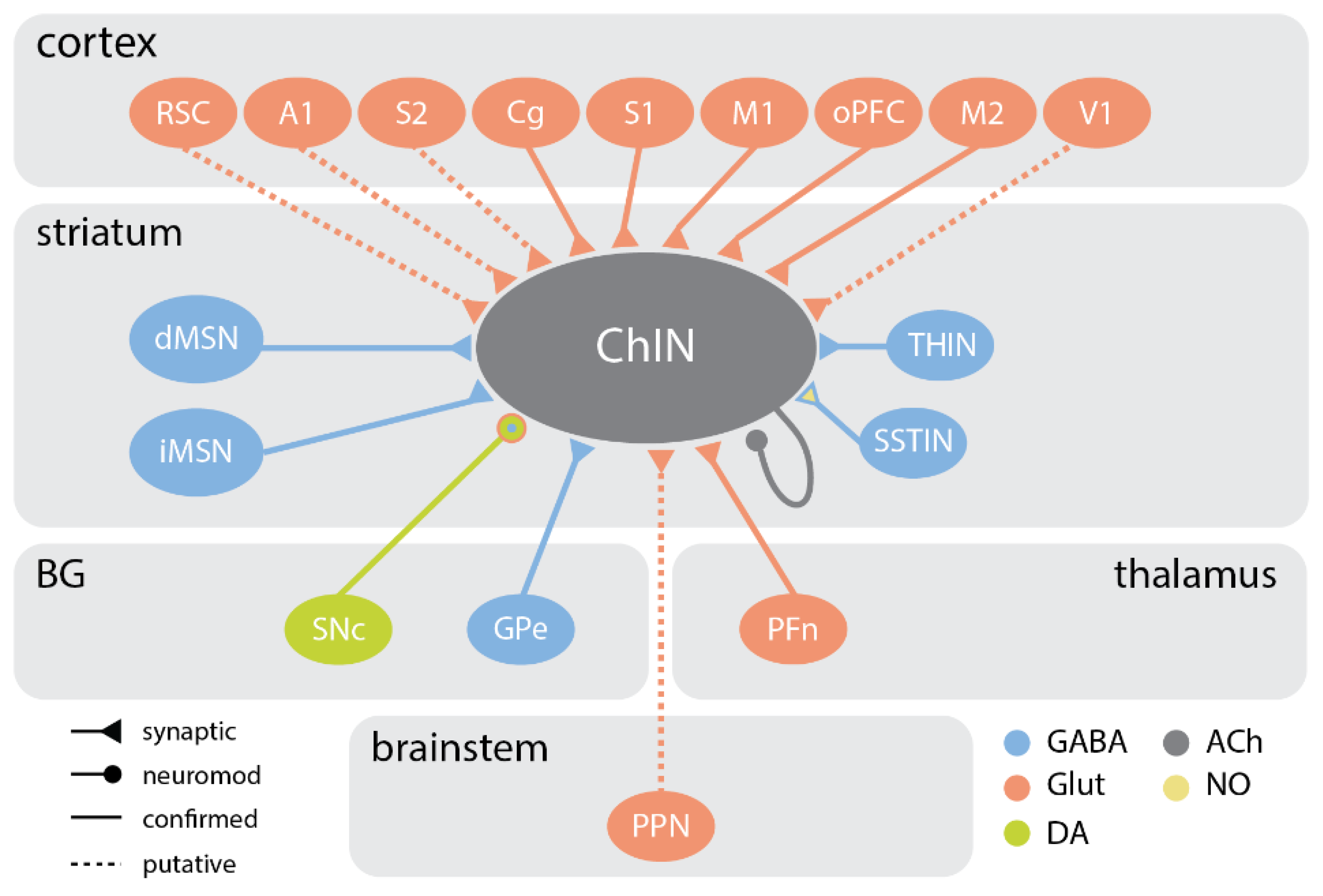
5. Synaptic Inputs and Neuromodulation
5.1. Excitatory Connections
5.1.1. Cortical Inputs
5.1.2. Thalamic Inputs
5.1.3. Brainstem Inputs
5.2. Inhibitory Connections
5.3. Neuromodulation
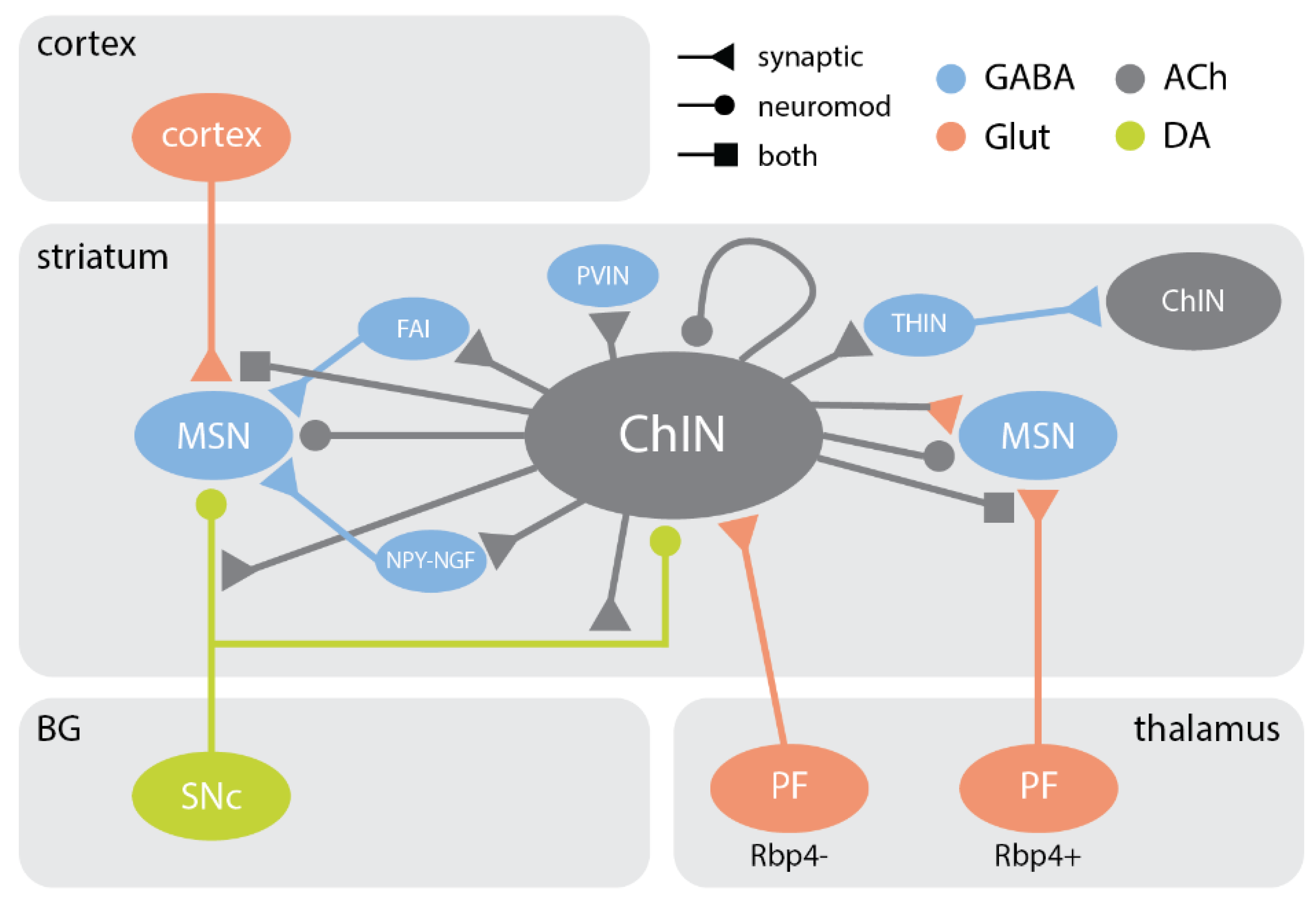
6. Synaptic Outputs and Neuromodulation
6.1. The Medium Spiny Neuron
6.2. Local Inhibitory Interneurons
6.3. The Midbrain Dopaminergic Axon
6.4. Automodulation
7. Cholinergic Interneuron Dysfunction
7.1. Tourette Disorder (TD)
7.2. Attention-Deficit Hyperactivity Disorder (ADHD)
7.3. Eating Disorders
7.4. Obsessive-Compulsive Disorder (OCD)
7.5. Parkinson’s Disease (PD)
7.6. Dystonia
7.7. Huntington’s Disease (HD)
8. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Tepper, J.M.; Koos, T.; Ibanez-Sandoval, O.; Tecuapetla, F.; Faust, T.W.; Assous, M. Heterogeneity and Diversity of Striatal GABAergic Interneurons: Update 2018. Front. Neuroanat. 2018, 12, 91. [Google Scholar] [CrossRef] [PubMed]
- Graveland, G.A.; DiFiglia, M. The frequency and distribution of medium-sized neurons with indented nuclei in the primate and rodent neostriatum. Brain Res. 1985, 327, 307–311. [Google Scholar] [CrossRef]
- Kawaguchi, Y.; Wilson, C.J.; Augood, S.J.; Emson, P.C. Striatal interneurones: Chemical, physiological and morphological characterization. Trends Neurosci. 1995, 18, 527–535. [Google Scholar] [CrossRef]
- Kreitzer, A.C. Physiology and pharmacology of striatal neurons. Annu. Rev. Neurosci. 2009, 32, 127–147. [Google Scholar] [CrossRef] [PubMed]
- Tepper, J.M.; Tecuapetla, F.; Koos, T.; Ibanez-Sandoval, O. Heterogeneity and diversity of striatal GABAergic interneurons. Front. Neuroanat. 2010, 4, 150. [Google Scholar] [CrossRef] [PubMed]
- Gittis, A.H.; Kreitzer, A.C. Striatal microcircuitry and movement disorders. Trends Neurosci. 2012, 35, 557–564. [Google Scholar] [CrossRef] [PubMed]
- Koós, T.; Tepper, J.M. Inhibitory control of neostriatal projection neurons by GABAergic interneurons. Nat. Neurosci. 1999, 2, 467–472. [Google Scholar] [CrossRef]
- Anderson, M.E. Discharge patterns of basal ganglia neurons during active maintenance of postural stability and adjustment to chair tilt. Brain Res. 1978, 143, 325–338. [Google Scholar] [CrossRef]
- Kimura, M.; Rajkowski, J.; Evarts, E. Tonically discharging putamen neurons exhibit set-dependent responses. Proc. Natl. Acad. Sci. USA 1984, 81, 4998–5001. [Google Scholar] [CrossRef]
- Kawaguchi, Y. Physiological, morphological, and histochemical characterization of three classes of interneurons in rat neostriatum. J. Neurosci. 1993, 13, 4908–4923. [Google Scholar] [CrossRef]
- Oorschot, D.E. The percentage of interneurons in the dorsal striatum of the rat, cat, monkey and human: A critique of the evidence. Basal Ganglia 2013, 3, 19–24. [Google Scholar] [CrossRef]
- Aosaki, T.; Kimura, M.; Graybiel, A.M. Temporal and spatial characteristics of tonically active neurons of the primate’s striatum. J. Neurophysiol. 1995, 73, 1234–1252. [Google Scholar] [CrossRef] [PubMed]
- Contant, C.; Umbriaco, D.; Garcia, S.; Watkins, K.C.; Descarries, L. Ultrastructural characterization of the acetylcholine innervation in adult rat neostriatum. Neuroscience 1996, 71, 937–947. [Google Scholar] [CrossRef]
- Mesulam, M.M.; Mash, D.; Hersh, L.; Bothwell, M.; Geula, C. Cholinergic innervation of the human striatum, globus pallidus, subthalamic nucleus, substantia nigra, and red nucleus. J. Comp. Neurol. 1992, 323, 252–268. [Google Scholar] [CrossRef] [PubMed]
- Woolf, N.J.; Butcher, L.L. Cholinergic neurons in the caudate-putamen complex proper are intrinsically organized: A combined Evans blue and acetylcholinesterase analysis. Brain Res. Bull. 1981, 7, 487–507. [Google Scholar] [CrossRef]
- Cheney, D.L.; LeFevere, H.F.; Racagni, G. Choline acetyltransferase activity and mass fragmentographic measurement of acetylcholine in specific nuclei and tracts of rat brain. Neuropharmacology 1975, 14, 801–809. [Google Scholar] [CrossRef]
- Goldberg, J.A.; Ding, J.B.; Surmeier, D.J. Muscarinic modulation of striatal function and circuitry. Handb. Exp. Pharm. 2012. [Google Scholar] [CrossRef]
- Pisani, A.; Bernardi, G.; Ding, J.; Surmeier, D.J. Re-emergence of striatal cholinergic interneurons in movement disorders. Trends Neurosci. 2007, 30, 545–553. [Google Scholar] [CrossRef]
- Gonzales, K.K.; Smith, Y. Cholinergic interneurons in the dorsal and ventral striatum: Anatomical and functional considerations in normal and diseased conditions. Ann. N. Y. Acad. Sci. 2015, 1349, 1–45. [Google Scholar] [CrossRef]
- Mallet, N.; Leblois, A.; Maurice, N.; Beurrier, C. Striatal Cholinergic Interneurons: How to Elucidate Their Function in Health and Disease. Front. Pharm. 2019, 10, 1488. [Google Scholar] [CrossRef] [PubMed]
- Tanimura, A.; Pancani, T.; Lim, S.A.O.; Tubert, C.; Melendez, A.E.; Shen, W.; Surmeier, D.J. Striatal cholinergic interneurons and Parkinson’s disease. Eur. J. Neurosci. 2018, 47, 1148–1158. [Google Scholar] [CrossRef] [PubMed]
- Abudukeyoumu, N.; Hernandez-Flores, T.; Garcia-Munoz, M.; Arbuthnott, G.W. Cholinergic modulation of striatal microcircuits. Eur. J. Neurosci. 2019, 49, 604–622. [Google Scholar] [CrossRef] [PubMed]
- Apicella, P. The role of the intrinsic cholinergic system of the striatum: What have we learned from TAN recordings in behaving animals? Neuroscience 2017, 360, 81–94. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, N.Y.; Knowles, R.; Dehorter, N. New Insights Into Cholinergic Neuron Diversity. Front. Mol. Neurosci. 2019, 12, 204. [Google Scholar] [CrossRef] [PubMed]
- Hamasaki, T.; Goto, S.; Nishikawa, S.; Ushio, Y. Neuronal cell migration for the developmental formation of the mammalian striatum. Brain Res. Rev. 2003, 41, 1–12. [Google Scholar] [CrossRef]
- Kang, H.J.; Kawasawa, Y.I.; Cheng, F.; Zhu, Y.; Xu, X.; Li, M.; Sousa, A.M.; Pletikos, M.; Meyer, K.A.; Sedmak, G.; et al. Spatio-temporal transcriptome of the human brain. Nature 2011, 478, 483–489. [Google Scholar] [CrossRef] [PubMed]
- Marín, O.; Anderson, S.A.; Rubenstein, J.L. Origin and molecular specification of striatal interneurons. J. Neurosci. 2000, 20, 6063–6076. [Google Scholar] [CrossRef]
- Allaway, K.C.; Machold, R. Developmental specification of forebrain cholinergic neurons. Dev. Biol. 2017, 421, 1–7. [Google Scholar] [CrossRef]
- Marín, O.; Yaron, A.; Bagri, A.; Tessier-Lavigne, M.; Rubenstein, J.L. Sorting of striatal and cortical interneurons regulated by semaphorin-neuropilin interactions. Science 2001, 293, 872–875. [Google Scholar] [CrossRef] [PubMed]
- Nóbrega-Pereira, S.; Kessaris, N.; Du, T.; Kimura, S.; Anderson, S.A.; Marin, O. Postmitotic Nkx2-1 controls the migration of telencephalic interneurons by direct repression of guidance receptors. Neuron 2008, 59, 733–745. [Google Scholar] [CrossRef] [PubMed]
- Semba, K.; Fibiger, H.C. Time of origin of cholinergic neurons in the rat basal forebrain. J. Comp. Neurol. 1988, 269, 87–95. [Google Scholar] [CrossRef]
- Phelps, P.E.; Brady, D.R.; Vaughn, J.E. The generation and differentiation of cholinergic neurons in rat caudate-putamen. Dev. Brain Res. 1989, 46, 47–60. [Google Scholar] [CrossRef]
- Chen, L.; Chatterjee, M.; Li, J.Y. The mouse homeobox gene Gbx2 is required for the development of cholinergic interneurons in the striatum. J. Neurosci. 2010, 30, 14824–14834. [Google Scholar] [CrossRef]
- Olsson, M.; Campbell, K.; Wictorin, K.; Bjorklund, A. Projection neurons in fetal striatal transplants are predominantly derived from the lateral ganglionic eminence. Neuroscience 1995, 69, 1169–1182. [Google Scholar] [CrossRef]
- Magno, L.; Barry, C.; Schmidt-Hieber, C.; Theodotou, P.; Hausser, M.; Kessaris, N. NKX2-1 Is Required in the Embryonic Septum for Cholinergic System Development, Learning, and Memory. Cell Rep. 2017, 20, 1572–1584. [Google Scholar] [CrossRef] [PubMed]
- Bulfone, A.; Puelles, L.; Porteus, M.H.; Frohman, M.A.; Martin, G.R.; Rubenstein, J.L. Spatially restricted expression of Dlx-1, Dlx-2 (Tes-1), Gbx-2, and Wnt-3 in the embryonic day 12.5 mouse forebrain defines potential transverse and longitudinal segmental boundaries. J. Neurosci. 1993, 13, 3155–3172. [Google Scholar] [CrossRef] [PubMed]
- Waters, S.T.; Wilson, C.P.; Lewandoski, M. Cloning and embryonic expression analysis of the mouse Gbx1 gene. Gene Expr. Patterns 2003, 3, 313–317. [Google Scholar] [CrossRef]
- Fragkouli, A.; van Wijk, N.V.; Lopes, R.; Kessaris, N.; Pachnis, V. LIM homeodomain transcription factor-dependent specification of bipotential MGE progenitors into cholinergic and GABAergic striatal interneurons. Development 2009, 136, 3841–3851. [Google Scholar] [CrossRef]
- Bachy, I.; Rétaux, S. GABAergic specification in the basal forebrain is controlled by the LIM-hd factor Lhx7. Dev. Biol. 2006, 291, 218–226. [Google Scholar] [CrossRef]
- Liodis, P.; Denaxa, M.; Grigoriou, M.; Akufo-Addo, C.; Yanagawa, Y.; Pachnis, V. Lhx6 activity is required for the normal migration and specification of cortical interneuron subtypes. J. Neurosci. 2007, 27, 3078–3089. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Marin, O.; Hermesz, E.; Powell, A.; Flames, N.; Palkovits, M.; Rubenstein, J.L.; Westphal, H. The LIM-homeobox gene Lhx8 is required for the development of many cholinergic neurons in the mouse forebrain. Proc. Natl. Acad. Sci. USA 2003, 100, 9005–9010. [Google Scholar] [CrossRef] [PubMed]
- Cho, H.H.; Cargnin, F.; Kim, Y.; Lee, B.; Kwon, R.J.; Nam, H.; Shen, R.; Barnes, A.P.; Lee, J.W.; Lee, S.; et al. Isl1 directly controls a cholinergic neuronal identity in the developing forebrain and spinal cord by forming cell type-specific complexes. PLoS Genet 2014, 10, e1004280. [Google Scholar] [CrossRef] [PubMed]
- Lopes, R.; Verhey van Wijk, N.; Neves, G.; Pachnis, V. Transcription factor LIM homeobox 7 (Lhx7) maintains subtype identity of cholinergic interneurons in the mammalian striatum. Proc. Natl. Acad. Sci. USA 2012, 109, 3119–3124. [Google Scholar] [CrossRef]
- Lozovaya, N.; Eftekhari, S.; Cloarec, R.; Gouty-Colomer, L.A.; Dufour, A.; Riffault, B.; Billon-Grand, M.; Pons-Bennaceur, A.; Oumar, N.; Burnashev, N.; et al. GABAergic inhibition in dual-transmission cholinergic and GABAergic striatal interneurons is abolished in Parkinson disease. Nat. Commun. 2018, 9, 1422. [Google Scholar] [CrossRef]
- Ranjbar-Slamloo, Y.; Ahmed, N.Y.; Al Abed, A.S.; Gao, L.; Sontani, Y.; Gauthier, A.R.C.; Arabzadeh, E.; Dehorter, N. Loss of the Er81 transcription factor in cholinergic cells alters striatal activity and habit formation. bioRxiv 2020. [Google Scholar] [CrossRef]
- Flames, N.; Pla, R.; Gelman, D.M.; Rubenstein, J.L.; Puelles, L.; Marín, O. Delineation of multiple subpallial progenitor domains by the combinatorial expression of transcriptional codes. J. Neurosci. 2007, 27, 9682–9695. [Google Scholar] [CrossRef]
- Mancini, M.; Bassani, S.; Passafaro, M. Right Place at the Right Time: How Changes in Protocadherins Affect Synaptic Connections Contributing to the Etiology of Neurodevelopmental Disorders. Cells 2020, 9, 2711. [Google Scholar] [CrossRef] [PubMed]
- Prem, S.; Millonig, J.H.; DiCicco-Bloom, E. Dysregulation of Neurite Outgrowth and Cell Migration in Autism and Other Neurodevelopmental Disorders. In Neurodevelopmental Disorders. Advances in Neurobiology; Di-Cicco-Bloom, E., Millonig, J., Eds.; Springer: Cham, Switzerland, 2020. [Google Scholar]
- Hawi, Z.; Tong, J.; Dark, C.; Yates, H.; Johnson, B.; Bellgrove, M.A. The role of cadherin genes in five major psychiatric disorders: A literature update. Am. J. Med. Genet. B Neuropsychiatr. Genet. 2018, 177, 168–180. [Google Scholar] [CrossRef] [PubMed]
- Kölliker, A. Handbuch der Gewbelehre des Menchen; Kengleman: Leipzig, Germany, 1896; Volume 2. [Google Scholar]
- McGeer, P.L.; McGeer, E.G.; Peng, J.H. Choline acetyltransferase: Purification and immunohistochemical localization. Life Sci. 1984, 34, 2319–2338. [Google Scholar] [CrossRef]
- Kimura, H.; McGeer, P.L.; Peng, F.; McGeer, E.G. Choline acetyltransferase-containing neurons in rodent brain demonstrated by immunohistochemistry. Science 1980, 208, 1057–1059. [Google Scholar] [CrossRef]
- Kimura, H.; McGeer, P.L.; Peng, J.H.; McGeer, E.G. The central cholinergic system studied by choline acetyltransferase immunohistochemistry in the cat. J. Comp. Neurol. 1981, 200, 151–201. [Google Scholar] [CrossRef]
- Mesulam, M.M.; Mufson, E.J.; Levey, A.I.; Wainer, B.H. Atlas of cholinergic neurons in the forebrain and upper brainstem of the macaque based on monoclonal choline acetyltransferase immunohistochemistry and acetylcholinesterase histochemistry. Neuroscience 1984, 12, 669–686. [Google Scholar] [CrossRef]
- Bolam, J.P.; Wainer, B.H.; Smith, A.D. Characterization of cholinergic neurons in the rat neostriatum. A combination of choline acetyltransferase immunocytochemistry, Golgi-impregnation and electron microscopy. Neuroscience 1984, 12, 711–718. [Google Scholar] [CrossRef]
- Lee, K.; Dixon, A.K.; Freeman, T.C.; Richardson, P.J. Identification of an ATP-sensitive potassium channel current in rat striatal cholinergic interneurones. J. Physiol. 1998, 510 Pt 2, 441–453. [Google Scholar] [CrossRef] [PubMed]
- Kawaguchi, Y. Large aspiny cells in the matrix of the rat neostriatum in vitro: Physiological identification, relation to the compartments and excitatory postsynaptic currents. J. Neurophysiol. 1992, 67, 1669–1682. [Google Scholar] [CrossRef] [PubMed]
- Phelps, P.E.; Houser, C.R.; Vaughn, J.E. Immunocytochemical localization of choline acetyltransferase within the rat neostriatum: A correlated light and electron microscopic study of cholinergic neurons and synapses. J. Comp. Neurol. 1985, 238, 286–307. [Google Scholar] [CrossRef]
- Doig, N.M.; Magill, P.J.; Apicella, P.; Bolam, J.P.; Sharott, A. Cortical and thalamic excitation mediate the multiphasic responses of striatal cholinergic interneurons to motivationally salient stimuli. J. Neurosci. 2014, 34, 3101–3117. [Google Scholar] [CrossRef]
- Wilson, C.J.; Chang, H.T.; Kitai, S.T. Firing patterns and synaptic potentials of identified giant aspiny interneurons in the rat neostriatum. J. Neurosci. 1990, 10, 508–519. [Google Scholar] [CrossRef]
- Goldberg, J.A.; Reynolds, J.N. Spontaneous firing and evoked pauses in the tonically active cholinergic interneurons of the striatum. Neuroscience 2011, 198, 27–43. [Google Scholar] [CrossRef]
- Lapper, S.R.; Bolam, J.P. Input from the frontal cortex and the parafascicular nucleus to cholinergic interneurons in the dorsal striatum of the rat. Neuroscience 1992, 51, 533–545. [Google Scholar] [CrossRef]
- Wouterlood, F.G.; Hartig, W.; Groenewegen, H.J.; Voorn, P. Density gradients of vesicular glutamate- and GABA transporter-immunoreactive boutons in calbindinand mu-opioid receptor-defined compartments in the rat striatum. J. Comp. Neurol. 2012, 520, 2123–2142. [Google Scholar] [CrossRef] [PubMed]
- Bennett, B.D.; Wilson, C.J. Spontaneous activity of neostriatal cholinergic interneurons in vitro. J. Neurosci. 1999, 19, 5586–5596. [Google Scholar] [CrossRef] [PubMed]
- Descarries, L.; Gisiger, V.; Steriade, M. Diffuse transmission by acetylcholine in the CNS. Prog. Neurobiol. 1997, 53, 603–625. [Google Scholar] [CrossRef]
- Threlfell, S.; Lalic, T.; Platt, N.J.; Jennings, K.A.; Deisseroth, K.; Cragg, S.J. Striatal dopamine release is triggered by synchronized activity in cholinergic interneurons. Neuron 2012, 75, 58–64. [Google Scholar] [CrossRef] [PubMed]
- Cachope, R.; Mateo, Y.; Mathur, B.N.; Irving, J.; Wang, H.L.; Morales, M.; Lovinger, D.M.; Cheer, J.F. Selective activation of cholinergic interneurons enhances accumbal phasic dopamine release: Setting the tone for reward processing. Cell Rep. 2012, 2, 33–41. [Google Scholar] [CrossRef]
- Higley, M.J.; Gittis, A.H.; Oldenburg, I.A.; Balthasar, N.; Seal, R.P.; Edwards, R.H.; Lowell, B.B.; Kreitzer, A.C.; Sabatini, B.L. Cholinergic interneurons mediate fast VGluT3-dependent glutamatergic transmission in the striatum. PLoS ONE 2011, 6, e19155. [Google Scholar] [CrossRef] [PubMed]
- Gras, C.; Amilhon, B.; Lepicard, E.M.; Poirel, O.; Vinatier, J.; Herbin, M.; Dumas, S.; Tzavara, E.T.; Wade, M.R.; Nomikos, G.G.; et al. The vesicular glutamate transporter VGLUT3 synergizes striatal acetylcholine tone. Nat. Neurosci. 2008, 11, 292–300. [Google Scholar] [CrossRef] [PubMed]
- Surmeier, D.J.; Mercer, J.N.; Chan, C.S. Autonomous pacemakers in the basal ganglia: Who needs excitatory synapses anyway? Curr. Opin. Neurobiol. 2005, 15, 312–318. [Google Scholar] [CrossRef]
- Jiang, Z.G.; North, R.A. Membrane properties and synaptic responses of rat striatal neurones in vitro. J. Physiol. 1991, 443, 533–553. [Google Scholar] [CrossRef]
- Wilson, C.J. The mechanism of intrinsic amplification of hyperpolarizations and spontaneous bursting in striatal cholinergic interneurons. Neuron 2005, 45, 575–585. [Google Scholar] [CrossRef]
- Apicella, P.; Deffains, M.; Ravel, S.; Legallet, E. Tonically active neurons in the striatum differentiate between delivery and omission of expected reward in a probabilistic task context. Eur. J. Neurosci. 2009, 30, 515–526. [Google Scholar] [CrossRef]
- Goldberg, J.A.; Wilson, C.J. Control of spontaneous firing patterns by the selective coupling of calcium currents to calcium-activated potassium currents in striatal cholinergic interneurons. J. Neurosci. 2005, 25, 10230–10238. [Google Scholar] [CrossRef] [PubMed]
- Aosaki, T.; Tsubokawa, H.; Ishida, A.; Watanabe, K.; Graybiel, A.M.; Kimura, M. Responses of tonically active neurons in the primate’s striatum undergo systematic changes during behavioral sensorimotor conditioning. J. Neurosci. 1994, 14, 3969–3984. [Google Scholar] [CrossRef] [PubMed]
- Apicella, P. Tonically active neurons in the primate striatum and their role in the processing of information about motivationally relevant events. Eur. J. Neurosci. 2002, 16, 2017–2026. [Google Scholar] [CrossRef] [PubMed]
- Ravel, S.; Legallet, E.; Apicella, P. Responses of tonically active neurons in the monkey striatum discriminate between motivationally opposing stimuli. J. Neurosci. 2003, 23, 8489–8497. [Google Scholar] [CrossRef] [PubMed]
- Joshua, M.; Adler, A.; Mitelman, R.; Vaadia, E.; Bergman, H. Midbrain dopaminergic neurons and striatal cholinergic interneurons encode the difference between reward and aversive events at different epochs of probabilistic classical conditioning trials. J. Neurosci. 2008, 28, 11673–11684. [Google Scholar] [CrossRef]
- Matsumoto, N.; Minamimoto, T.; Graybiel, A.M.; Kimura, M. Neurons in the thalamic CM-Pf complex supply striatal neurons with information about behaviorally significant sensory events. J. Neurophysiol. 2001, 85, 960–976. [Google Scholar] [CrossRef]
- Zhang, Y.F.; Cragg, S.J. Pauses in Striatal Cholinergic Interneurons: What is Revealed by Their Common Themes and Variations? Front. Syst. Neurosci. 2017, 11, 80. [Google Scholar] [CrossRef] [PubMed]
- Morris, G.; Arkadir, D.; Nevet, A.; Vaadia, E.; Bergman, H. Coincident but distinct messages of midbrain dopamine and striatal tonically active neurons. Neuron 2004, 43, 133–143. [Google Scholar] [CrossRef]
- Straub, C.; Tritsch, N.X.; Hagan, N.A.; Gu, C.; Sabatini, B.L. Multiphasic modulation of cholinergic interneurons by nigrostriatal afferents. J. Neurosci. 2014, 34, 8557–8569. [Google Scholar] [CrossRef]
- Wieland, S.; Du, D.; Oswald, M.J.; Parlato, R.; Kohr, G.; Kelsch, W. Phasic dopaminergic activity exerts fast control of cholinergic interneuron firing via sequential NMDA, D2, and D1 receptor activation. J. Neurosci. 2014, 34, 11549–11559. [Google Scholar] [CrossRef]
- Brown, M.T.; Tan, K.R.; O’Connor, E.C.; Nikonenko, I.; Muller, D.; Luscher, C. Ventral tegmental area GABA projections pause accumbal cholinergic interneurons to enhance associative learning. Nature 2012, 492, 452–456. [Google Scholar] [CrossRef] [PubMed]
- Ding, J.B.; Guzman, J.N.; Peterson, J.D.; Goldberg, J.A.; Surmeier, D.J. Thalamic gating of corticostriatal signaling by cholinergic interneurons. Neuron 2010, 67, 294–307. [Google Scholar] [CrossRef] [PubMed]
- Oswald, M.J.; Oorschot, D.E.; Schulz, J.M.; Lipski, J.; Reynolds, J.N. IH current generates the afterhyperpolarisation following activation of subthreshold cortical synaptic inputs to striatal cholinergic interneurons. J. Physiol. 2009, 587, 5879–5897. [Google Scholar] [CrossRef] [PubMed]
- Reynolds, J.N.; Hyland, B.I.; Wickens, J.R. Modulation of an afterhyperpolarization by the substantia nigra induces pauses in the tonic firing of striatal cholinergic interneurons. J. Neurosci. 2004, 24, 9870–9877. [Google Scholar] [CrossRef]
- Zhang, Y.F.; Reynolds, J.N.J.; Cragg, S.J. Pauses in Cholinergic Interneuron Activity Are Driven by Excitatory Input and Delayed Rectification, with Dopamine Modulation. Neuron 2018, 98, 918–925.e913. [Google Scholar] [CrossRef]
- Guo, Q.; Wang, D.; He, X.; Feng, Q.; Lin, R.; Xu, F.; Fu, L.; Luo, M. Whole-brain mapping of inputs to projection neurons and cholinergic interneurons in the dorsal striatum. PLoS ONE 2015, 10, e0123381. [Google Scholar] [CrossRef]
- Cai, Y.; Ford, C.P. Dopamine Cells Differentially Regulate Striatal Cholinergic Transmission across Regions through Corelease of Dopamine and Glutamate. Cell Rep. 2018, 25, 3148–3157.e3143. [Google Scholar] [CrossRef]
- Mamaligas, A.A.; Barcomb, K.; Ford, C.P. Cholinergic Transmission at Muscarinic Synapses in the Striatum Is Driven Equally by Cortical and Thalamic Inputs. Cell Rep. 2019, 28, 1003–1014.e1003. [Google Scholar] [CrossRef]
- English, D.F.; Ibanez-Sandoval, O.; Stark, E.; Tecuapetla, F.; Buzsaki, G.; Deisseroth, K.; Tepper, J.M.; Koos, T. GABAergic circuits mediate the reinforcement-related signals of striatal cholinergic interneurons. Nat. Neurosci. 2011, 15, 123–130. [Google Scholar] [CrossRef] [PubMed]
- Higley, M.J.; Soler-Llavina, G.J.; Sabatini, B.L. Cholinergic modulation of multivesicular release regulates striatal synaptic potency and integration. Nat. Neurosci. 2009, 12, 1121–1128. [Google Scholar] [CrossRef] [PubMed]
- Mamaligas, A.A.; Ford, C.P. Spontaneous Synaptic Activation of Muscarinic Receptors by Striatal Cholinergic Neuron Firing. Neuron 2016, 91, 574–586. [Google Scholar] [CrossRef] [PubMed]
- Klug, J.R.; Engelhardt, M.D.; Cadman, C.N.; Li, H.; Smith, J.B.; Ayala, S.; Williams, E.W.; Hoffman, H.; Jin, X. Differential inputs to striatal cholinergic and parvalbumin interneurons imply functional distinctions. eLife 2018, 7. [Google Scholar] [CrossRef]
- Reynolds, J.N.; Wickens, J.R. The corticostriatal input to giant aspiny interneurons in the rat: A candidate pathway for synchronising the response to reward-related cues. Brain Res. 2004, 1011, 115–128. [Google Scholar] [CrossRef]
- Johansson, Y.; Silberberg, G. The Functional Organization of Cortical and Thalamic Inputs onto Five Types of Striatal Neurons Is Determined by Source and Target Cell Identities. Cell Rep. 2020, 30, 1178–1194.e1173. [Google Scholar] [CrossRef] [PubMed]
- Fujiyama, F.; Kuramoto, E.; Okamoto, K.; Hioki, H.; Furuta, T.; Zhou, L.; Nomura, S.; Kaneko, T. Presynaptic localization of an AMPA-type glutamate receptor in corticostriatal and thalamostriatal axon terminals. Eur. J. Neurosci. 2004, 20, 3322–3330. [Google Scholar] [CrossRef]
- Fremeau, R.T., Jr.; Voglmaier, S.; Seal, R.P.; Edwards, R.H. VGLUTs define subsets of excitatory neurons and suggest novel roles for glutamate. Trends Neurosci. 2004, 27, 98–103. [Google Scholar] [CrossRef]
- Thomas, T.M.; Smith, Y.; Levey, A.I.; Hersch, S.M. Cortical inputs to m2-immunoreactive striatal interneurons in rat and monkey. Synapse 2000, 37, 252–261. [Google Scholar] [CrossRef]
- Schilman, E.A.; Uylings, H.B.; Galis-de Graaf, Y.; Joel, D.; Groenewegen, H.J. The orbital cortex in rats topographically projects to central parts of the caudate-putamen complex. Neurosci. Lett. 2008, 432, 40–45. [Google Scholar] [CrossRef]
- McAlonan, K.; Brown, V.J. Orbital prefrontal cortex mediates reversal learning and not attentional set shifting in the rat. Behav. Brain Res. 2003, 146, 97–103. [Google Scholar] [CrossRef]
- Ragozzino, M.E.; Kim, J.; Hassert, D.; Minniti, N.; Kiang, C. The contribution of the rat prelimbic-infralimbic areas to different forms of task switching. Behav. Neurosci. 2003, 117, 1054–1065. [Google Scholar] [CrossRef]
- Boulougouris, V.; Dalley, J.W.; Robbins, T.W. Effects of orbitofrontal, infralimbic and prelimbic cortical lesions on serial spatial reversal learning in the rat. Behav. Brain Res. 2007, 179, 219–228. [Google Scholar] [CrossRef]
- Norman, L.J.; Carlisi, C.O.; Christakou, A.; Murphy, C.M.; Chantiluke, K.; Giampietro, V.; Simmons, A.; Brammer, M.; Mataix-Cols, D.; Rubia, K. Frontostriatal Dysfunction During Decision Making in Attention-Deficit/Hyperactivity Disorder and Obsessive-Compulsive Disorder. Biol. Psychiatry Cogn. Neurosci. Neuroimaging 2018, 3, 694–703. [Google Scholar] [CrossRef] [PubMed]
- Lange, F.; Seer, C.; Muller-Vahl, K.; Kopp, B. Cognitive flexibility and its electrophysiological correlates in Gilles de la Tourette syndrome. Dev. Cogn. Neurosci. 2017, 27, 78–90. [Google Scholar] [CrossRef] [PubMed]
- Aoki, S.; Liu, A.W.; Zucca, A.; Zucca, S.; Wickens, J.R. Role of Striatal Cholinergic Interneurons in Set-Shifting in the Rat. J. Neurosci. 2015, 35, 9424–9431. [Google Scholar] [CrossRef]
- Apicella, P. Leading tonically active neurons of the striatum from reward detection to context recognition. Trends Neurosci. 2007, 30, 299–306. [Google Scholar] [CrossRef] [PubMed]
- Ragozzino, M.E.; Mohler, E.G.; Prior, M.; Palencia, C.A.; Rozman, S. Acetylcholine activity in selective striatal regions supports behavioral flexibility. Neurobiol. Learn. Mem. 2009, 91, 13–22. [Google Scholar] [CrossRef] [PubMed]
- Okada, K.; Nishizawa, K.; Fukabori, R.; Kai, N.; Shiota, A.; Ueda, M.; Tsutsui, Y.; Sakata, S.; Matsushita, N.; Kobayashi, K. Enhanced flexibility of place discrimination learning by targeting striatal cholinergic interneurons. Nat. Commun. 2014, 5, 3778. [Google Scholar] [CrossRef]
- Stalnaker, T.A.; Berg, B.; Aujla, N.; Schoenbaum, G. Cholinergic Interneurons Use Orbitofrontal Input to Track Beliefs about Current State. J. Neurosci. 2016, 36, 6242–6257. [Google Scholar] [CrossRef]
- Walton, M.E.; Croxson, P.L.; Behrens, T.E.; Kennerley, S.W.; Rushworth, M.F. Adaptive decision making and value in the anterior cingulate cortex. Neuroimage 2007, 36 (Suppl. 2), T142–T154. [Google Scholar] [CrossRef]
- Melendez-Zaidi, A.E.; Lakshminarasimhah, H.; Surmeier, D.J. Cholinergic modulation of striatal nitric oxide-producing interneurons. Eur. J. Neurosci. 2019, 50, 3713–3731. [Google Scholar] [CrossRef]
- Rafalovich, I.V.; Melendez, A.E.; Plotkin, J.L.; Tanimura, A.; Zhai, S.; Surmeier, D.J. Interneuronal Nitric Oxide Signaling Mediates Post-synaptic Long-Term Depression of Striatal Glutamatergic Synapses. Cell Rep. 2015, 13, 1336–1342. [Google Scholar] [CrossRef]
- Calabresi, P.; Gubellini, P.; Centonze, D.; Sancesario, G.; Morello, M.; Giorgi, M.; Pisani, A.; Bernardi, G. A critical role of the nitric oxide/cGMP pathway in corticostriatal long-term depression. J. Neurosci. 1999, 19, 2489–2499. [Google Scholar] [CrossRef]
- Yamanaka, K.; Hori, Y.; Minamimoto, T.; Yamada, H.; Matsumoto, N.; Enomoto, K.; Aosaki, T.; Graybiel, A.M.; Kimura, M. Roles of centromedian parafascicular nuclei of thalamus and cholinergic interneurons in the dorsal striatum in associative learning of environmental events. J. Neural Transm. 2018, 125, 501–513. [Google Scholar] [CrossRef] [PubMed]
- Minamimoto, T.; Hori, Y.; Kimura, M. Roles of the thalamic CM-PF complex-Basal ganglia circuit in externally driven rebias of action. Brain Res. Bull. 2009, 78, 75–79. [Google Scholar] [CrossRef]
- Brown, H.D.; Baker, P.M.; Ragozzino, M.E. The parafascicular thalamic nucleus concomitantly influences behavioral flexibility and dorsomedial striatal acetylcholine output in rats. J. Neurosci. 2010, 30, 14390–14398. [Google Scholar] [CrossRef]
- Tanimura, A.; Du, Y.; Kondapalli, J.; Wokosin, D.L.; Surmeier, D.J. Cholinergic Interneurons Amplify Thalamostriatal Excitation of Striatal Indirect Pathway Neurons in Parkinson’s Disease Models. Neuron 2019, 101, 444–458.e446. [Google Scholar] [CrossRef]
- Dautan, D.; Huerta-Ocampo, I.; Witten, I.B.; Deisseroth, K.; Bolam, J.P.; Gerdjikov, T.; Mena-Segovia, J. A major external source of cholinergic innervation of the striatum and nucleus accumbens originates in the brainstem. J. Neurosci. 2014, 34, 4509–4518. [Google Scholar] [CrossRef] [PubMed]
- Mena-Segovia, J.; Bolam, J.P. Rethinking the Pedunculopontine Nucleus: From Cellular Organization to Function. Neuron 2017, 94, 7–18. [Google Scholar] [CrossRef] [PubMed]
- Gonzales, K.K.; Pare, J.F.; Wichmann, T.; Smith, Y. GABAergic inputs from direct and indirect striatal projection neurons onto cholinergic interneurons in the primate putamen. J. Comp. Neurol. 2013, 521, 2502–2522. [Google Scholar] [CrossRef]
- DeBoer, P.; Westerink, B.H. GABAergic modulation of striatal cholinergic interneurons: An in vivo microdialysis study. J. Neurochem. 1994, 62, 70–75. [Google Scholar] [CrossRef]
- Sato, A.; Sasaoka, T.; Nishijo, T.; Momiyama, T. GABAergic synaptic transmission onto striatal cholinergic interneurons in dopamine D2 receptor knock-out mice. Neuroscience 2014, 263, 138–147. [Google Scholar] [CrossRef]
- Suzuki, T.; Miura, M.; Nishimura, K.; Aosaki, T. Dopamine-dependent synaptic plasticity in the striatal cholinergic interneurons. J. Neurosci. 2001, 21, 6492–6501. [Google Scholar] [CrossRef]
- Bonsi, P.; De Persis, C.; Calabresi, P.; Bernardi, G.; Pisani, A. Coordinate high-frequency pattern of stimulation and calcium levels control the induction of LTP in striatal cholinergic interneurons. Learn. Mem. 2004, 11, 755–760. [Google Scholar] [CrossRef][Green Version]
- Chuhma, N.; Tanaka, K.F.; Hen, R.; Rayport, S. Functional connectome of the striatal medium spiny neuron. J. Neurosci. 2011, 31, 1183–1192. [Google Scholar] [CrossRef]
- Dorst, M.C.; Tokarska, A.; Zhou, M.; Lee, K.; Stagkourakis, S.; Broberger, C.; Masmanidis, S.; Silberberg, G. Polysynaptic inhibition between striatal cholinergic interneurons shapes their network activity patterns in a dopamine-dependent manner. Nat. Commun. 2020, 11, 5113. [Google Scholar] [CrossRef]
- Straub, C.; Saulnier, J.L.; Begue, A.; Feng, D.D.; Huang, K.W.; Sabatini, B.L. Principles of Synaptic Organization of GABAergic Interneurons in the Striatum. Neuron 2016, 92, 84–92. [Google Scholar] [CrossRef]
- Zhou, F.M.; Liang, Y.; Dani, J.A. Endogenous nicotinic cholinergic activity regulates dopamine release in the striatum. Nat. Neurosci. 2001, 4, 1224–1229. [Google Scholar] [CrossRef] [PubMed]
- Sullivan, M.A.; Chen, H.; Morikawa, H. Recurrent inhibitory network among striatal cholinergic interneurons. J. Neurosci. 2008, 28, 8682–8690. [Google Scholar] [CrossRef]
- Martone, M.E.; Armstrong, D.M.; Young, S.J.; Groves, P.M. Ultrastructural examination of enkephalin and substance P input to cholinergic neurons within the rat neostriatum. Brain Res. 1992, 594, 253–262. [Google Scholar] [CrossRef]
- Cui, G.; Jun, S.B.; Jin, X.; Pham, M.D.; Vogel, S.S.; Lovinger, D.M.; Costa, R.M. Concurrent activation of striatal direct and indirect pathways during action initiation. Nature 2013, 494, 238–242. [Google Scholar] [CrossRef]
- Tecuapetla, F.; Jin, X.; Lima, S.Q.; Costa, R.M. Complementary Contributions of Striatal Projection Pathways to Action Initiation and Execution. Cell 2016, 166, 703–715. [Google Scholar] [CrossRef] [PubMed]
- Lim, S.A.; Kang, U.J.; McGehee, D.S. Striatal cholinergic interneuron regulation and circuit effects. Front. Synaptic Neurosci. 2014, 6, 22. [Google Scholar] [CrossRef]
- Francis, T.C.; Yano, H.; Demarest, T.G.; Shen, H.; Bonci, A. High-Frequency Activation of Nucleus Accumbens D1-MSNs Drives Excitatory Potentiation on D2-MSNs. Neuron 2019, 103, 432–444.e433. [Google Scholar] [CrossRef] [PubMed]
- Chuhma, N.; Mingote, S.; Moore, H.; Rayport, S. Dopamine neurons control striatal cholinergic neurons via regionally heterogeneous dopamine and glutamate signaling. Neuron 2014, 81, 901–912. [Google Scholar] [CrossRef]
- Chuhma, N.; Mingote, S.; Yetnikoff, L.; Kalmbach, A.; Ma, T.; Ztaou, S.; Sienna, A.C.; Tepler, S.; Poulin, J.F.; Ansorge, M.; et al. Dopamine neuron glutamate cotransmission evokes a delayed excitation in lateral dorsal striatal cholinergic interneurons. eLife 2018, 7, e39786. [Google Scholar] [CrossRef] [PubMed]
- Dautan, D.; Huerta-Ocampo, I.; Gut, N.K.; Valencia, M.; Kondabolu, K.; Kim, Y.; Gerdjikov, T.V.; Mena-Segovia, J. Cholinergic midbrain afferents modulate striatal circuits and shape encoding of action strategies. Nat. Commun. 2020, 11, 1739. [Google Scholar] [CrossRef]
- Threlfell, S.; Cragg, S.J. Dopamine signaling in dorsal versus ventral striatum: The dynamic role of cholinergic interneurons. Front. Syst. Neurosci. 2011, 5, 11. [Google Scholar] [CrossRef]
- Granger, A.J.; Mulder, N.; Saunders, A.; Sabatini, B.L. Cotransmission of acetylcholine and GABA. Neuropharmacology 2016, 100, 40–46. [Google Scholar] [CrossRef]
- Gras, C.; Herzog, E.; Bellenchi, G.C.; Bernard, V.; Ravassard, P.; Pohl, M.; Gasnier, B.; Giros, B.; El Mestikawy, S. A third vesicular glutamate transporter expressed by cholinergic and serotoninergic neurons. J. Neurosci. 2002, 22, 5442–5451. [Google Scholar] [CrossRef] [PubMed]
- Calabresi, P.; Pisani, A.; Rothwell, J.; Ghiglieri, V.; Obeso, J.A.; Picconi, B. Hyperkinetic disorders and loss of synaptic downscaling. Nat. Neurosci. 2016, 19, 868–875. [Google Scholar] [CrossRef]
- Ueno, T.; Yamada, J.; Nishijima, H.; Arai, A.; Migita, K.; Baba, M.; Ueno, S.; Tomiyama, M. Morphological and electrophysiological changes in intratelencephalic-type pyramidal neurons in the motor cortex of a rat model of levodopa-induced dyskinesia. Neurobiol. Dis. 2014, 64, 142–149. [Google Scholar] [CrossRef] [PubMed]
- Gritton, H.J.; Howe, W.M.; Romano, M.F.; DiFeliceantonio, A.G.; Kramer, M.A.; Saligrama, V.; Bucklin, M.E.; Zemel, D.; Han, X. Unique contributions of parvalbumin and cholinergic interneurons in organizing striatal networks during movement. Nat. Neurosci. 2019, 22, 586–597. [Google Scholar] [CrossRef] [PubMed]
- Witten, I.B.; Lin, S.C.; Brodsky, M.; Prakash, R.; Diester, I.; Anikeeva, P.; Gradinaru, V.; Ramakrishnan, C.; Deisseroth, K. Cholinergic interneurons control local circuit activity and cocaine conditioning. Science 2010, 330, 1677–1681. [Google Scholar] [CrossRef] [PubMed]
- Calabresi, P.; Picconi, B.; Tozzi, A.; Di Filippo, M. Dopamine-mediated regulation of corticostriatal synaptic plasticity. Trends Neurosci. 2007, 30, 211–219. [Google Scholar] [CrossRef] [PubMed]
- Shen, W.; Plotkin, J.L.; Francardo, V.; Ko, W.K.; Xie, Z.; Li, Q.; Fieblinger, T.; Wess, J.; Neubig, R.R.; Lindsley, C.W.; et al. M4 Muscarinic Receptor Signaling Ameliorates Striatal Plasticity Deficits in Models of L-DOPA-Induced Dyskinesia. Neuron 2015, 88, 762–773. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Kai, L.; Day, M.; Ronesi, J.; Yin, H.H.; Ding, J.; Tkatch, T.; Lovinger, D.M.; Surmeier, D.J. Dopaminergic control of corticostriatal long-term synaptic depression in medium spiny neurons is mediated by cholinergic interneurons. Neuron 2006, 50, 443–452. [Google Scholar] [CrossRef]
- Gerdeman, G.L.; Ronesi, J.; Lovinger, D.M. Postsynaptic endocannabinoid release is critical to long-term depression in the striatum. Nat. Neurosci. 2002, 5, 446–451. [Google Scholar] [CrossRef]
- Kreitzer, A.C.; Malenka, R.C. Dopamine modulation of state-dependent endocannabinoid release and long-term depression in the striatum. J. Neurosci. 2005, 25, 10537–10545. [Google Scholar] [CrossRef]
- Calabresi, P.; Centonze, D.; Gubellini, P.; Pisani, A.; Bernardi, G. Blockade of M2-like muscarinic receptors enhances long-term potentiation at corticostriatal synapses. Eur. J. Neurosci. 1998, 10, 3020–3023. [Google Scholar] [CrossRef] [PubMed]
- Alcantara, A.A.; Mrzljak, L.; Jakab, R.L.; Levey, A.I.; Hersch, S.M.; Goldman-Rakic, P.S. Muscarinic m1 and m2 receptor proteins in local circuit and projection neurons of the primate striatum: Anatomical evidence for cholinergic modulation of glutamatergic prefronto-striatal pathways. J. Comp. Neurol. 2001, 434, 445–460. [Google Scholar] [CrossRef] [PubMed]
- Pakhotin, P.; Bracci, E. Cholinergic interneurons control the excitatory input to the striatum. J. Neurosci. 2007, 27, 391–400. [Google Scholar] [CrossRef]
- Hersch, S.M.; Gutekunst, C.A.; Rees, H.D.; Heilman, C.J.; Levey, A.I. Distribution of m1-m4 muscarinic receptor proteins in the rat striatum: Light and electron microscopic immunocytochemistry using subtype-specific antibodies. J. Neurosci. 1994, 14, 3351–3363. [Google Scholar] [CrossRef]
- Yan, Z.; Flores-Hernandez, J.; Surmeier, D.J. Coordinated expression of muscarinic receptor messenger RNAs in striatal medium spiny neurons. Neuroscience 2001, 103, 1017–1024. [Google Scholar] [CrossRef]
- Bernard, V.; Normand, E.; Bloch, B. Phenotypical characterization of the rat striatal neurons expressing muscarinic receptor genes. J. Neurosci. 1992, 12, 3591–3600. [Google Scholar] [CrossRef]
- Tozzi, A.; de Iure, A.; Di Filippo, M.; Tantucci, M.; Costa, C.; Borsini, F.; Ghiglieri, V.; Giampa, C.; Fusco, F.R.; Picconi, B.; et al. The distinct role of medium spiny neurons and cholinergic interneurons in the D(2)/A(2)A receptor interaction in the striatum: Implications for Parkinson’s disease. J. Neurosci. 2011, 31, 1850–1862. [Google Scholar] [CrossRef]
- Faust, T.W.; Assous, M.; Shah, F.; Tepper, J.M.; Koos, T. Novel fast adapting interneurons mediate cholinergic-induced fast GABAA inhibitory postsynaptic currents in striatal spiny neurons. Eur. J. Neurosci. 2015, 42, 1764–1774. [Google Scholar] [CrossRef]
- Faust, T.W.; Assous, M.; Tepper, J.M.; Koos, T. Neostriatal GABAergic Interneurons Mediate Cholinergic Inhibition of Spiny Projection Neurons. J. Neurosci. 2016, 36, 9505–9511. [Google Scholar] [CrossRef]
- Muñoz-Manchado, A.B.; Foldi, C.; Szydlowski, S.; Sjulson, L.; Farries, M.; Wilson, C.; Silberberg, G.; Hjerling-Leffler, J. Novel Striatal GABAergic Interneuron Populations Labeled in the 5HT3a(EGFP) Mouse. Cereb. Cortex 2016, 26, 96–105. [Google Scholar] [CrossRef] [PubMed]
- Chang, H.T.; Kita, H. Interneurons in the rat striatum: Relationships between parvalbumin neurons and cholinergic neurons. Brain Res. 1992, 574, 307–311. [Google Scholar] [CrossRef]
- Koós, T.; Tepper, J.M. Dual cholinergic control of fast-spiking interneurons in the neostriatum. J. Neurosci. 2002, 22, 529–535. [Google Scholar] [CrossRef]
- Kosillo, P.; Zhang, Y.F.; Threlfell, S.; Cragg, S.J. Cortical Control of Striatal Dopamine Transmission via Striatal Cholinergic Interneurons. Cereb. Cortex 2016. [Google Scholar] [CrossRef] [PubMed]
- Nelson, A.B.; Hammack, N.; Yang, C.F.; Shah, N.M.; Seal, R.P.; Kreitzer, A.C. Striatal cholinergic interneurons Drive GABA release from dopamine terminals. Neuron 2014, 82, 63–70. [Google Scholar] [CrossRef]
- Calabresi, P.; Centonze, D.; Pisani, A.; Sancesario, G.; North, R.A.; Bernardi, G. Muscarinic IPSPs in rat striatal cholinergic interneurones. J. Physiol. 1998, 510 Pt 2, 421–427. [Google Scholar] [CrossRef] [PubMed]
- Yan, Z.; Surmeier, D.J. Muscarinic (m2/m4) receptors reduce N- and P-type Ca2+ currents in rat neostriatal cholinergic interneurons through a fast, membrane-delimited, G-protein pathway. J. Neurosci. 1996, 16, 2592–2604. [Google Scholar] [CrossRef]
- Zhao, Z.; Zhang, K.; Liu, X.; Yan, H.; Ma, X.; Zhang, S.; Zheng, J.; Wang, L.; Wei, X. Involvement of HCN Channel in Muscarinic Inhibitory Action on Tonic Firing of Dorsolateral Striatal Cholinergic Interneurons. Front. Cell. Neurosci. 2016, 10, 71. [Google Scholar] [CrossRef]
- Aliane, V.; Perez, S.; Bohren, Y.; Deniau, J.M.; Kemel, M.L. Key role of striatal cholinergic interneurons in processes leading to arrest of motor stereotypies. Brain 2011, 134, 110–118. [Google Scholar] [CrossRef] [PubMed]
- Crittenden, J.R.; Lacey, C.J.; Weng, F.J.; Garrison, C.E.; Gibson, D.J.; Lin, Y.; Graybiel, A.M. Striatal Cholinergic Interneurons Modulate Spike-Timing in Striosomes and Matrix by an Amphetamine-Sensitive Mechanism. Front. Neuroanat. 2017, 11, 20. [Google Scholar] [CrossRef] [PubMed]
- Cavanna, A.E.; Black, K.J.; Hallett, M.; Voon, V. Neurobiology of the Premonitory Urge in Tourette’s Syndrome: Pathophysiology and Treatment Implications. J. Neuropsychiatry Clin. Neurosci. 2017, 29, 95–104. [Google Scholar] [CrossRef]
- Willsey, A.J.; Fernandez, T.V.; Yu, D.; King, R.A.; Dietrich, A.; Xing, J.; Sanders, S.J.; Mandell, J.D.; Huang, A.Y.; Richer, P.; et al. De Novo Coding Variants Are Strongly Associated with Tourette Disorder. Neuron 2017, 94, 486–499.e489. [Google Scholar] [CrossRef]
- Leckman, J.F.; King, R.A.; Bloch, M.H. Clinical Features of Tourette Syndrome and Tic Disorders. J. Obs. Compuls. Relat. Disord. 2014, 3, 372–379. [Google Scholar] [CrossRef]
- 174. Cross-Disorder Group of the Psychiatric Genomics Consortium. Genomic Relationships, Novel Loci, and Pleiotropic Mechanisms across Eight Psychiatric Disorders. Cell 2019, 179, 1469–1482.e1411. [CrossRef]
- Leckman, J.F.; Peterson, B.S.; Pauls, D.L.; Cohen, D.J. Tic disorders. Psychiatr. Clin. N. Am. 1997, 20, 839–861. [Google Scholar] [CrossRef]
- Peterson, B.; Riddle, M.A.; Cohen, D.J.; Katz, L.D.; Smith, J.C.; Hardin, M.T.; Leckman, J.F. Reduced basal ganglia volumes in Tourette’s syndrome using three-dimensional reconstruction techniques from magnetic resonance images. Neurology 1993, 43, 941–949. [Google Scholar] [CrossRef] [PubMed]
- Peterson, B.S.; Thomas, P.; Kane, M.J.; Scahill, L.; Zhang, H.; Bronen, R.; King, R.A.; Leckman, J.F.; Staib, L. Basal Ganglia volumes in patients with Gilles de la Tourette syndrome. Arch. Gen. Psychiatry 2003, 60, 415–424. [Google Scholar] [CrossRef] [PubMed]
- Worbe, Y.; Malherbe, C.; Hartmann, A.; Pelegrini-Issac, M.; Messe, A.; Vidailhet, M.; Lehericy, S.; Benali, H. Functional immaturity of cortico-basal ganglia networks in Gilles de la Tourette syndrome. Brain 2012, 135, 1937–1946. [Google Scholar] [CrossRef] [PubMed]
- Atkinson-Clement, C.; Porte, C.A.; de Liege, A.; Klein, Y.; Delorme, C.; Beranger, B.; Valabregue, R.; Gallea, C.; Robbins, T.W.; Hartmann, A.; et al. Impulsive prepotent actions and tics in Tourette disorder underpinned by a common neural network. Mol. Psychiatry 2020. [Google Scholar] [CrossRef] [PubMed]
- Alcantara, A.A.; Chen, V.; Herring, B.E.; Mendenhall, J.M.; Berlanga, M.L. Localization of dopamine D2 receptors on cholinergic interneurons of the dorsal striatum and nucleus accumbens of the rat. Brain Res. 2003, 986, 22–29. [Google Scholar] [CrossRef]
- Scholl, C.; Baladron, J.; Vitay, J.; Hamker, F.H. Enhanced habit formation in Tourette syndrome: Dopamine release and striatal disonhibition modulate shortcut connections in a hierarchical model of cortico-basal ganglia loops. bioRxiv 2021. [Google Scholar] [CrossRef]
- Aoki, S.; Liu, A.W.; Akamine, Y.; Zucca, A.; Zucca, S.; Wickens, J.R. Cholinergic interneurons in the rat striatum modulate substitution of habits. Eur. J. Neurosci. 2018, 47, 1194–1205. [Google Scholar] [CrossRef]
- Mink, J.W. Basal ganglia dysfunction in Tourette’s syndrome: A new hypothesis. Pediatr. Neurol. 2001, 25, 190–198. [Google Scholar] [CrossRef]
- Barbeau, A. The pathogenesis of Parkinson’s disease: A new hypothesis. Can. Med. Assoc. J. 1962, 87, 802–807. [Google Scholar]
- Tanner, C.M.; Goetz, C.G.; Klawans, H.L. Cholinergic mechanisms in Tourette syndrome. Neurology 1982, 32, 1315–1317. [Google Scholar] [CrossRef]
- Kataoka, Y.; Kalanithi, P.S.; Grantz, H.; Schwartz, M.L.; Saper, C.; Leckman, J.F.; Vaccarino, F.M. Decreased number of parvalbumin and cholinergic interneurons in the striatum of individuals with Tourette syndrome. J. Comp. Neurol. 2010, 518, 277–291. [Google Scholar] [CrossRef]
- Bronfeld, M.; Yael, D.; Belelovsky, K.; Bar-Gad, I. Motor tics evoked by striatal disinhibition in the rat. Front. Syst. Neurosci. 2013, 7, 50. [Google Scholar] [CrossRef] [PubMed]
- McCairn, K.W.; Nagai, Y.; Hori, Y.; Ninomiya, T.; Kikuchi, E.; Lee, J.Y.; Suhara, T.; Iriki, A.; Minamimoto, T.; Takada, M.; et al. A Primary Role for Nucleus Accumbens and Related Limbic Network in Vocal Tics. Neuron 2016, 89, 300–307. [Google Scholar] [CrossRef]
- Xu, M.; Kobets, A.; Du, J.C.; Lennington, J.; Li, L.; Banasr, M.; Duman, R.S.; Vaccarino, F.M.; DiLeone, R.J.; Pittenger, C. Targeted ablation of cholinergic interneurons in the dorsolateral striatum produces behavioral manifestations of Tourette syndrome. Proc. Natl. Acad. Sci. USA 2015, 112, 893–898. [Google Scholar] [CrossRef] [PubMed]
- Martos, Y.V.; Braz, B.Y.; Beccaria, J.P.; Murer, M.G.; Belforte, J.E. Compulsive Social Behavior Emerges after Selective Ablation of Striatal Cholinergic Interneurons. J. Neurosci. 2017, 37, 2849–2858. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Maia, T.V.; Marsh, R.; Colibazzi, T.; Gerber, A.; Peterson, B.S. The neural circuits that generate tics in Tourette’s syndrome. Am. J. Psychiatry 2011, 168, 1326–1337. [Google Scholar] [CrossRef] [PubMed]
- Rapanelli, M.; Frick, L.R.; Xu, M.; Groman, S.M.; Jindachomthong, K.; Tamamaki, N.; Tanahira, C.; Taylor, J.R.; Pittenger, C. Targeted Interneuron Depletion in the Dorsal Striatum Produces Autism-like Behavioral Abnormalities in Male but Not Female Mice. Biol. Psychiatry 2017, 82, 194–203. [Google Scholar] [CrossRef]
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders: DSM-5, 5th ed.; American Psychiatric Publishing: Arlington, VA, USA, 2013. [Google Scholar]
- Mega, M.S.; Cummings, J.L. Frontal-subcortical circuits and neuropsychiatric disorders. J. Neuropsychiatry Clin. Neurosci. 1994, 6, 358–370. [Google Scholar] [CrossRef] [PubMed]
- Castellanos, F.X. Toward a pathophysiology of attention-deficit/hyperactivity disorder. Clin. Pediatr. 1997, 36, 381–393. [Google Scholar] [CrossRef] [PubMed]
- Hoogman, M.; Bralten, J.; Hibar, D.P.; Mennes, M.; Zwiers, M.P.; Schweren, L.S.J.; van Hulzen, K.J.E.; Medland, S.E.; Shumskaya, E.; Jahanshad, N.; et al. Subcortical brain volume differences in participants with attention deficit hyperactivity disorder in children and adults: A cross-sectional mega-analysis. Lancet Psychiatry 2017, 4, 310–319. [Google Scholar] [CrossRef]
- Dickstein, S.G.; Bannon, K.; Castellanos, F.X.; Milham, M.P. The neural correlates of attention deficit hyperactivity disorder: An ALE meta-analysis. J. Child Psychol. Psychiatry 2006, 47, 1051–1062. [Google Scholar] [CrossRef] [PubMed]
- Hart, H.; Radua, J.; Nakao, T.; Mataix-Cols, D.; Rubia, K. Meta-analysis of functional magnetic resonance imaging studies of inhibition and attention in attention-deficit/hyperactivity disorder: Exploring task-specific, stimulant medication, and age effects. Jama Psychiatry 2013, 70, 185–198. [Google Scholar] [CrossRef] [PubMed]
- Scheres, A.; Milham, M.P.; Knutson, B.; Castellanos, F.X. Ventral striatal hyporesponsiveness during reward anticipation in attention-deficit/hyperactivity disorder. Biol. Psychiatry 2007, 61, 720–724. [Google Scholar] [CrossRef]
- Dark, C.; Homman-Ludiye, J.; Bryson-Richardson, R.J. The role of ADHD associated genes in neurodevelopment. Dev. Biol. 2018, 438, 69–83. [Google Scholar] [CrossRef]
- Kent, L.; Middle, F.; Hawi, Z.; Fitzgerald, M.; Gill, M.; Feehan, C.; Craddock, N. Nicotinic acetylcholine receptor alpha4 subunit gene polymorphism and attention deficit hyperactivity disorder. Psychiatr. Genet. 2001, 11, 37–40. [Google Scholar] [CrossRef]
- Todd, R.D.; Lobos, E.A.; Sun, L.W.; Neuman, R.J. Mutational analysis of the nicotinic acetylcholine receptor alpha 4 subunit gene in attention deficit/hyperactivity disorder: Evidence for association of an intronic polymorphism with attention problems. Mol. Psychiatry 2003, 8, 103–108. [Google Scholar] [CrossRef]
- English, B.A.; Hahn, M.K.; Gizer, I.R.; Mazei-Robison, M.; Steele, A.; Kurnik, D.M.; Stein, M.A.; Waldman, I.D.; Blakely, R.D. Choline transporter gene variation is associated with attention-deficit hyperactivity disorder. J. Neurodev. Disord. 2009, 1, 252–263. [Google Scholar] [CrossRef]
- Apparsundaram, S.; Martinez, V.; Parikh, V.; Kozak, R.; Sarter, M. Increased capacity and density of choline transporters situated in synaptic membranes of the right medial prefrontal cortex of attentional task-performing rats. J. Neurosci. 2005, 25, 3851–3856. [Google Scholar] [CrossRef]
- Grottick, A.J.; Higgins, G.A. Effect of subtype selective nicotinic compounds on attention as assessed by the five-choice serial reaction time task. Behav. Brain Res. 2000, 117, 197–208. [Google Scholar] [CrossRef]
- Young, J.W.; Meves, J.M.; Geyer, M.A. Nicotinic agonist-induced improvement of vigilance in mice in the 5-choice continuous performance test. Behav. Brain Res. 2013, 240, 119–133. [Google Scholar] [CrossRef]
- Hoyle, E.; Genn, R.F.; Fernandes, C.; Stolerman, I.P. Impaired performance of alpha7 nicotinic receptor knockout mice in the five-choice serial reaction time task. Psychopharmacology 2006, 189, 211–223. [Google Scholar] [CrossRef] [PubMed]
- Young, J.W.; Crawford, N.; Kelly, J.S.; Kerr, L.E.; Marston, H.M.; Spratt, C.; Finlayson, K.; Sharkey, J. Impaired attention is central to the cognitive deficits observed in alpha 7 deficient mice. Eur. Neuropsychopharmacol. 2007, 17, 145–155. [Google Scholar] [CrossRef] [PubMed]
- Stergiakouli, E.; Hamshere, M.; Holmans, P.; Langley, K.; Zaharieva, I.; deCODE Genetics, Psychiatric GWAS Consortium: ADHD Subgroup; Hawi, Z.; Kent, L.; Gill, M.; Williams, N. Investigating the contribution of common genetic variants to the risk and pathogenesis of ADHD. Am. J. Psychiatry 2012, 169, 186–194. [Google Scholar] [CrossRef]
- Favier, M.; Janickova, H.; Justo, D.; Kljakic, O.; Runtz, L.; Natsheh, J.Y.; Pascoal, T.A.; Germann, J.; Gallino, D.; Kang, J.I.; et al. Cholinergic dysfunction in the dorsal striatum promotes habit formation and maladaptive eating. J. Clin. Investig. 2020, 130, 6616–6630. [Google Scholar] [CrossRef]
- Breiter, H.C.; Rauch, S.L.; Kwong, K.K.; Baker, J.R.; Weisskoff, R.M.; Kennedy, D.N.; Kendrick, A.D.; Davis, T.L.; Jiang, A.; Cohen, M.S.; et al. Functional magnetic resonance imaging of symptom provocation in obsessive-compulsive disorder. Arch. Gen. Psychiatry 1996, 53, 595–606. [Google Scholar] [CrossRef]
- Remijnse, P.L.; Nielen, M.M.; van Balkom, A.J.; Cath, D.C.; van Oppen, P.; Uylings, H.B.; Veltman, D.J. Reduced orbitofrontal-striatal activity on a reversal learning task in obsessive-compulsive disorder. Arch. Gen. Psychiatry 2006, 63, 1225–1236. [Google Scholar] [CrossRef] [PubMed]
- Frick, L.R.; Rapanelli, M.; Jindachomthong, K.; Grant, P.; Leckman, J.F.; Swedo, S.; Williams, K.; Pittenger, C. Differential binding of antibodies in PANDAS patients to cholinergic interneurons in the striatum. Brain Behav. Immun. 2018, 69, 304–311. [Google Scholar] [CrossRef]
- Xu, J.; Liu, R.J.; Fahey, S.; Frick, L.; Leckman, J.; Vaccarino, F.; Duman, R.S.; Williams, K.; Swedo, S.; Pittenger, C. Antibodies From Children With PANDAS Bind Specifically to Striatal Cholinergic Interneurons and Alter Their Activity. Am. J. Psychiatry 2021, 178, 48–64. [Google Scholar] [CrossRef]
- Jankovic, J. Parkinson’s disease: Clinical features and diagnosis. J. Neurol. Neurosurg. Psychiatry 2008, 79, 368–376. [Google Scholar] [CrossRef] [PubMed]
- Albin, R.L.; Young, A.B.; Penney, J.B. The functional anatomy of basal ganglia disorders. Trends Neurosci. 1989, 12, 366–375. [Google Scholar] [CrossRef]
- Gerfen, C.R.; Surmeier, D.J. Modulation of striatal projection systems by dopamine. Annu. Rev. Neurosci. 2011, 34, 441–466. [Google Scholar] [CrossRef]
- Lewis, S.J.; Dove, A.; Robbins, T.W.; Barker, R.A.; Owen, A.M. Cognitive impairments in early Parkinson’s disease are accompanied by reductions in activity in frontostriatal neural circuitry. J. Neurosci. 2003, 23, 6351–6356. [Google Scholar] [CrossRef] [PubMed]
- Sanjari Moghaddam, H.; Zare-Shahabadi, A.; Rahmani, F.; Rezaei, N. Neurotransmission systems in Parkinson’s disease. Rev. Neurosci. 2017, 28, 509–536. [Google Scholar] [CrossRef] [PubMed]
- Aosaki, T.; Miura, M.; Suzuki, T.; Nishimura, K.; Masuda, M. Acetylcholine-dopamine balance hypothesis in the striatum: An update. Geriatr. Gerontol. Int. 2010, 10 (Suppl. 1), S148–S157. [Google Scholar] [CrossRef]
- Ancelin, M.L.; Artero, S.; Portet, F.; Dupuy, A.M.; Touchon, J.; Ritchie, K. Non-degenerative mild cognitive impairment in elderly people and use of anticholinergic drugs: Longitudinal cohort study. BMJ 2006, 332, 455–459. [Google Scholar] [CrossRef] [PubMed]
- Tubert, C.; Murer, M.G. What’s wrong with the striatal cholinergic interneurons in Parkinson’s disease? Focus on intrinsic excitability. Eur. J. Neurosci. 2020. [Google Scholar] [CrossRef] [PubMed]
- Maurice, N.; Liberge, M.; Jaouen, F.; Ztaou, S.; Hanini, M.; Camon, J.; Deisseroth, K.; Amalric, M.; Kerkerian-Le Goff, L.; Beurrier, C. Striatal Cholinergic Interneurons Control Motor Behavior and Basal Ganglia Function in Experimental Parkinsonism. Cell Rep. 2015, 13, 657–666. [Google Scholar] [CrossRef]
- Fino, E.; Glowinski, J.; Venance, L. Effects of acute dopamine depletion on the electrophysiological properties of striatal neurons. Neurosci. Res. 2007, 58, 305–316. [Google Scholar] [CrossRef] [PubMed]
- Choi, S.J.; Ma, T.C.; Ding, Y.; Cheung, T.; Joshi, N.; Sulzer, D.; Mosharov, E.V.; Kang, U.J. Alterations in the intrinsic properties of striatal cholinergic interneurons after dopamine lesion and chronic L-DOPA. eLife 2020, 9, e56920. [Google Scholar] [CrossRef]
- Ding, J.; Guzman, J.N.; Tkatch, T.; Chen, S.; Goldberg, J.A.; Ebert, P.J.; Levitt, P.; Wilson, C.J.; Hamm, H.E.; Surmeier, D.J. RGS4-dependent attenuation of M4 autoreceptor function in striatal cholinergic interneurons following dopamine depletion. Nat. Neurosci. 2006, 9, 832–842. [Google Scholar] [CrossRef] [PubMed]
- Henderson, J.M.; Carpenter, K.; Cartwright, H.; Halliday, G.M. Degeneration of the centre median-parafascicular complex in Parkinson’s disease. Ann. Neurol. 2000, 47, 345–352. [Google Scholar] [CrossRef]
- Villalba, R.M.; Wichmann, T.; Smith, Y. Neuronal loss in the caudal intralaminar thalamic nuclei in a primate model of Parkinson’s disease. Brain Struct. Funct. 2014, 219, 381–394. [Google Scholar] [CrossRef] [PubMed]
- Howe, M.; Ridouh, I.; Allegra Mascaro, A.L.; Larios, A.; Azcorra, M.; Dombeck, D.A. Coordination of rapid cholinergic and dopaminergic signaling in striatum during spontaneous movement. eLife 2019, 8, e44903. [Google Scholar] [CrossRef] [PubMed]
- Smith, Y.; Surmeier, D.J.; Redgrave, P.; Kimura, M. Thalamic contributions to Basal Ganglia-related behavioral switching and reinforcement. J. Neurosci. 2011, 31, 16102–16106. [Google Scholar] [CrossRef]
- Aceves Buendia, J.J.; Tiroshi, L.; Chiu, W.H.; Goldberg, J.A. Selective remodeling of glutamatergic transmission to striatal cholinergic interneurons after dopamine depletion. Eur. J. Neurosci. 2019, 49, 824–833. [Google Scholar] [CrossRef] [PubMed]
- Tozzi, A.; de Iure, A.; Bagetta, V.; Tantucci, M.; Durante, V.; Quiroga-Varela, A.; Costa, C.; Di Filippo, M.; Ghiglieri, V.; Latagliata, E.C.; et al. Alpha-Synuclein Produces Early Behavioral Alterations via Striatal Cholinergic Synaptic Dysfunction by Interacting With GluN2D N-Methyl-D-Aspartate Receptor Subunit. Biol. Psychiatry 2016, 79, 402–414. [Google Scholar] [CrossRef]
- McKinley, J.W.; Shi, Z.; Kawikova, I.; Hur, M.; Bamford, I.J.; Sudarsana Devi, S.P.; Vahedipour, A.; Darvas, M.; Bamford, N.S. Dopamine Deficiency Reduces Striatal Cholinergic Interneuron Function in Models of Parkinson’s Disease. Neuron 2019, 103, 1056–1072. [Google Scholar] [CrossRef] [PubMed]
- Cai, Y.; Nielsen, B.E.; Boxer, E.E.; Aoto, J.; Ford, C.P. Loss of nigral excitation of cholinergic interneurons contributes to parkinsonian motor impairments. Neuron 2021, 109, 1–13. [Google Scholar] [CrossRef]
- Bordia, T.; Perez, X.A.; Heiss, J.; Zhang, D.; Quik, M. Optogenetic activation of striatal cholinergic interneurons regulates L-dopa-induced dyskinesias. Neurobiol. Dis. 2016, 91, 47–58. [Google Scholar] [CrossRef] [PubMed]
- Paz, R.M.; Tubert, C.; Stahl, A.M.; Amarillo, Y.; Rela, L.; Murer, M.G. Levodopa Causes Striatal Cholinergic Interneuron Burst-Pause Activity in Parkinsonian Mice. Mov. Disord. 2021. [Google Scholar] [CrossRef] [PubMed]
- Calabresi, P.; Standaert, D.G. Dystonia and levodopa-induced dyskinesias in Parkinson’s disease: Is there a connection? Neurobiol. Dis. 2019, 132, 104579. [Google Scholar] [CrossRef] [PubMed]
- Mazere, J.; Dilharreguy, B.; Catheline, G.; Vidailhet, M.; Deffains, M.; Vimont, D.; Ribot, B.; Barse, E.; Cif, L.; Mazoyer, B.; et al. Striatal and cerebellar vesicular acetylcholine transporter expression is disrupted in human DYT1 dystonia. Brain 2021. [Google Scholar] [CrossRef]
- Balint, B.; Mencacci, N.E.; Valente, E.M.; Pisani, A.; Rothwell, J.; Jankovic, J.; Vidailhet, M.; Bhatia, K.P. Dystonia. Nat. Rev. Dis. Primers 2018, 4, 25. [Google Scholar] [CrossRef]
- Eskow Jaunarajs, K.L.; Bonsi, P.; Chesselet, M.F.; Standaert, D.G.; Pisani, A. Striatal cholinergic dysfunction as a unifying theme in the pathophysiology of dystonia. Prog. Neurobiol. 2015, 127–128, 91–107. [Google Scholar] [CrossRef]
- Maltese, M.; Martella, G.; Madeo, G.; Fagiolo, I.; Tassone, A.; Ponterio, G.; Sciamanna, G.; Burbaud, P.; Conn, P.J.; Bonsi, P.; et al. Anticholinergic drugs rescue synaptic plasticity in DYT1 dystonia: Role of M1 muscarinic receptors. Mov. Disord. 2014, 29, 1655–1665. [Google Scholar] [CrossRef]
- Yu-Taeger, L.; Ott, T.; Bonsi, P.; Tomczak, C.; Wassouf, Z.; Martella, G.; Sciamanna, G.; Imbriani, P.; Ponterio, G.; Tassone, A.; et al. Impaired dopamine- and adenosine-mediated signaling and plasticity in a novel rodent model for DYT25 dystonia. Neurobiol. Dis. 2020, 134, 104634. [Google Scholar] [CrossRef]
- Pappas, S.S.; Darr, K.; Holley, S.M.; Cepeda, C.; Mabrouk, O.S.; Wong, J.M.; LeWitt, T.M.; Paudel, R.; Houlden, H.; Kennedy, R.T.; et al. Forebrain deletion of the dystonia protein torsinA causes dystonic-like movements and loss of striatal cholinergic neurons. eLife 2015, 4, e08352. [Google Scholar] [CrossRef]
- Imbriani, P.; Ponterio, G.; Tassone, A.; Sciamanna, G.; El Atiallah, I.; Bonsi, P.; Pisani, A. Models of dystonia: An update. J. Neurosci. Methods 2020, 339, 108728. [Google Scholar] [CrossRef] [PubMed]
- Eskow Jaunarajs, K.L.; Scarduzio, M.; Ehrlich, M.E.; McMahon, L.L.; Standaert, D.G. Diverse Mechanisms Lead to Common Dysfunction of Striatal Cholinergic Interneurons in Distinct Genetic Mouse Models of Dystonia. J. Neurosci. 2019, 39, 7195–7205. [Google Scholar] [CrossRef]
- Scarduzio, M.; Zimmerman, C.N.; Jaunarajs, K.L.; Wang, Q.; Standaert, D.G.; McMahon, L.L. Strength of cholinergic tone dictates the polarity of dopamine D2 receptor modulation of striatal cholinergic interneuron excitability in DYT1 dystonia. Exp. Neurol. 2017, 295, 162–175. [Google Scholar] [CrossRef] [PubMed]
- The Huntington’s Disease Collaborative Research Group. A novel gene containing a trinucleotide repeat that is expanded and unstable on Huntington’s disease chromosomes. Cell 1993, 72, 971–983. [Google Scholar] [CrossRef]
- Ferrante, R.J.; Beal, M.F.; Kowall, N.W.; Richardson, E.P., Jr.; Martin, J.B. Sparing of acetylcholinesterase-containing striatal neurons in Huntington’s disease. Brain Res. 1987, 411, 162–166. [Google Scholar] [CrossRef]
- Lallani, S.B.; Villalba, R.M.; Chen, Y.; Smith, Y.; Chan, A.W.S. Striatal Interneurons in Transgenic Nonhuman Primate Model of Huntington’s Disease. Sci. Rep. 2019, 9, 3528. [Google Scholar] [CrossRef] [PubMed]
- Calabresi, P.; Centonze, D.; Pisani, A.; Sancesario, G.; Gubellini, P.; Marfia, G.A.; Bernardi, G. Striatal spiny neurons and cholinergic interneurons express differential ionotropic glutamatergic responses and vulnerability: Implications for ischemia and Huntington’s disease. Ann. Neurol. 1998, 43, 586–597. [Google Scholar] [CrossRef]
- Calabresi, P.; Saulle, E.; Centonze, D.; Pisani, A.; Marfia, G.A.; Bernardi, G. Post-ischaemic long-term synaptic potentiation in the striatum: A putative mechanism for cell type-specific vulnerability. Brain 2002, 125, 844–860. [Google Scholar] [CrossRef]
- Deng, Y.P.; Wong, T.; Bricker-Anthony, C.; Deng, B.; Reiner, A. Loss of corticostriatal and thalamostriatal synaptic terminals precedes striatal projection neuron pathology in heterozygous Q140 Huntington’s disease mice. Neurobiol. Dis. 2013, 60, 89–107. [Google Scholar] [CrossRef]
- Hiley, C.R.; Bird, E.D. Decreased muscarinic receptor concentration in post-mortem brain in Huntington’s chorea. Brain Res. 1974, 80, 355–358. [Google Scholar] [CrossRef]
- Wastek, G.J.; Stern, L.Z.; Johnson, P.C.; Yamamura, H.I. Huntington’s disease: Regional alteration in muscarinic cholinergic receptor binding in human brain. Life Sci. 1976, 19, 1033–1039. [Google Scholar] [CrossRef]
- Ferrante, R.J.; Kowall, N.W.; Beal, M.F.; Martin, J.B.; Bird, E.D.; Richardson, E.P., Jr. Morphologic and histochemical characteristics of a spared subset of striatal neurons in Huntington’s disease. J. Neuropathol. Exp. Neurol. 1987, 46, 12–27. [Google Scholar] [CrossRef] [PubMed]
- Vetter, J.M.; Jehle, T.; Heinemeyer, J.; Franz, P.; Behrens, P.F.; Jackisch, R.; Landwehrmeyer, G.B.; Feuerstein, T.J. Mice transgenic for exon 1 of Huntington’s disease: Properties of cholinergic and dopaminergic pre-synaptic function in the striatum. J. Neurochem. 2003, 85, 1054–1063. [Google Scholar] [CrossRef] [PubMed]
- Smith, R.; Chung, H.; Rundquist, S.; Maat-Schieman, M.L.; Colgan, L.; Englund, E.; Liu, Y.J.; Roos, R.A.; Faull, R.L.; Brundin, P.; et al. Cholinergic neuronal defect without cell loss in Huntington’s disease. Hum. Mol. Genet. 2006, 15, 3119–3131. [Google Scholar] [CrossRef]
- Mangiarini, L.; Sathasivam, K.; Seller, M.; Cozens, B.; Harper, A.; Hetherington, C.; Lawton, M.; Trottier, Y.; Lehrach, H.; Davies, S.W.; et al. Exon 1 of the HD gene with an expanded CAG repeat is sufficient to cause a progressive neurological phenotype in transgenic mice. Cell 1996, 87, 493–506. [Google Scholar] [CrossRef]
- Cepeda, C.; Levine, M.S. Synaptic Dysfunction in Huntington’s Disease: Lessons from Genetic Animal Models. Neuroscientist 2020, 1073858420972662. [Google Scholar] [CrossRef]
- Picconi, B.; Passino, E.; Sgobio, C.; Bonsi, P.; Barone, I.; Ghiglieri, V.; Pisani, A.; Bernardi, G.; Ammassari-Teule, M.; Calabresi, P. Plastic and behavioral abnormalities in experimental Huntington’s disease: A crucial role for cholinergic interneurons. Neurobiol. Dis. 2006, 22, 143–152. [Google Scholar] [CrossRef]
- Lawrence, A.D.; Sahakian, B.J.; Hodges, J.R.; Rosser, A.E.; Lange, K.W.; Robbins, T.W. Executive and mnemonic functions in early Huntington’s disease. Brain 1996, 119 Pt 5, 1633–1645. [Google Scholar] [CrossRef]
- Lawrence, A.D.; Hodges, J.R.; Rosser, A.E.; Kershaw, A.; Ffrench-Constant, C.; Rubinsztein, D.C.; Robbins, T.W.; Sahakian, B.J. Evidence for specific cognitive deficits in preclinical Huntington’s disease. Brain 1998, 121 Pt 7, 1329–1341. [Google Scholar] [CrossRef]
- Deng, Y.P.; Reiner, A. Cholinergic interneurons in the Q140 knockin mouse model of Huntington’s disease: Reductions in dendritic branching and thalamostriatal input. J. Comp. Neurol. 2016, 524, 3518–3529. [Google Scholar] [CrossRef]
- Heinsen, H.; Rub, U.; Gangnus, D.; Jungkunz, G.; Bauer, M.; Ulmar, G.; Bethke, B.; Schuler, M.; Bocker, F.; Eisenmenger, W.; et al. Nerve cell loss in the thalamic centromedian-parafascicular complex in patients with Huntington’s disease. Acta Neuropathol. 1996, 91, 161–168. [Google Scholar] [CrossRef] [PubMed]
- Tanimura, A.; Lim, S.A.; Aceves Buendia, J.J.; Goldberg, J.A.; Surmeier, D.J. Cholinergic Interneurons Amplify Corticostriatal Synaptic Responses in the Q175 Model of Huntington’s Disease. Front. Syst. Neurosci. 2016, 10, 102. [Google Scholar] [CrossRef] [PubMed]
- Holley, S.M.; Joshi, P.R.; Parievsky, A.; Galvan, L.; Chen, J.Y.; Fisher, Y.E.; Huynh, M.N.; Cepeda, C.; Levine, M.S. Enhanced GABAergic Inputs Contribute to Functional Alterations of Cholinergic Interneurons in the R6/2 Mouse Model of Huntington’s Disease. eNeuro 2015, 2. [Google Scholar] [CrossRef]
- Mehler, M.F.; Petronglo, J.R.; Arteaga-Bracho, E.E.; Gulinello, M.E.; Winchester, M.L.; Pichamoorthy, N.; Young, S.K.; DeJesus, C.D.; Ishtiaq, H.; Gokhan, S.; et al. Loss-of-Huntingtin in Medial and Lateral Ganglionic Lineages Differentially Disrupts Regional Interneuron and Projection Neuron Subtypes and Promotes Huntington’s Disease-Associated Behavioral, Cellular, and Pathological Hallmarks. J. Neurosci. 2019, 39, 1892–1909. [Google Scholar] [CrossRef] [PubMed]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Poppi, L.A.; Ho-Nguyen, K.T.; Shi, A.; Daut, C.T.; Tischfield, M.A. Recurrent Implication of Striatal Cholinergic Interneurons in a Range of Neurodevelopmental, Neurodegenerative, and Neuropsychiatric Disorders. Cells 2021, 10, 907. https://doi.org/10.3390/cells10040907
Poppi LA, Ho-Nguyen KT, Shi A, Daut CT, Tischfield MA. Recurrent Implication of Striatal Cholinergic Interneurons in a Range of Neurodevelopmental, Neurodegenerative, and Neuropsychiatric Disorders. Cells. 2021; 10(4):907. https://doi.org/10.3390/cells10040907
Chicago/Turabian StylePoppi, Lauren A., Khue Tu Ho-Nguyen, Anna Shi, Cynthia T. Daut, and Max A. Tischfield. 2021. "Recurrent Implication of Striatal Cholinergic Interneurons in a Range of Neurodevelopmental, Neurodegenerative, and Neuropsychiatric Disorders" Cells 10, no. 4: 907. https://doi.org/10.3390/cells10040907
APA StylePoppi, L. A., Ho-Nguyen, K. T., Shi, A., Daut, C. T., & Tischfield, M. A. (2021). Recurrent Implication of Striatal Cholinergic Interneurons in a Range of Neurodevelopmental, Neurodegenerative, and Neuropsychiatric Disorders. Cells, 10(4), 907. https://doi.org/10.3390/cells10040907





