Depleting Tumor Cells Expressing Immune Checkpoint Ligands—A New Approach to Combat Cancer
Abstract
1. Introduction
2. Mechanisms Underlying the Overexpression of ICPMLs on Tumor Cells
3. The Consequences of the Expression of ICPMLs on the Biology of Tumor Cells
4. Why Non-Depleting Antibodies Have Been Used against Inhibitory ICPMLs
5. Does Target Cell Depletion Contribute to the Therapeutic Activity of Some of the Approved ICIs?
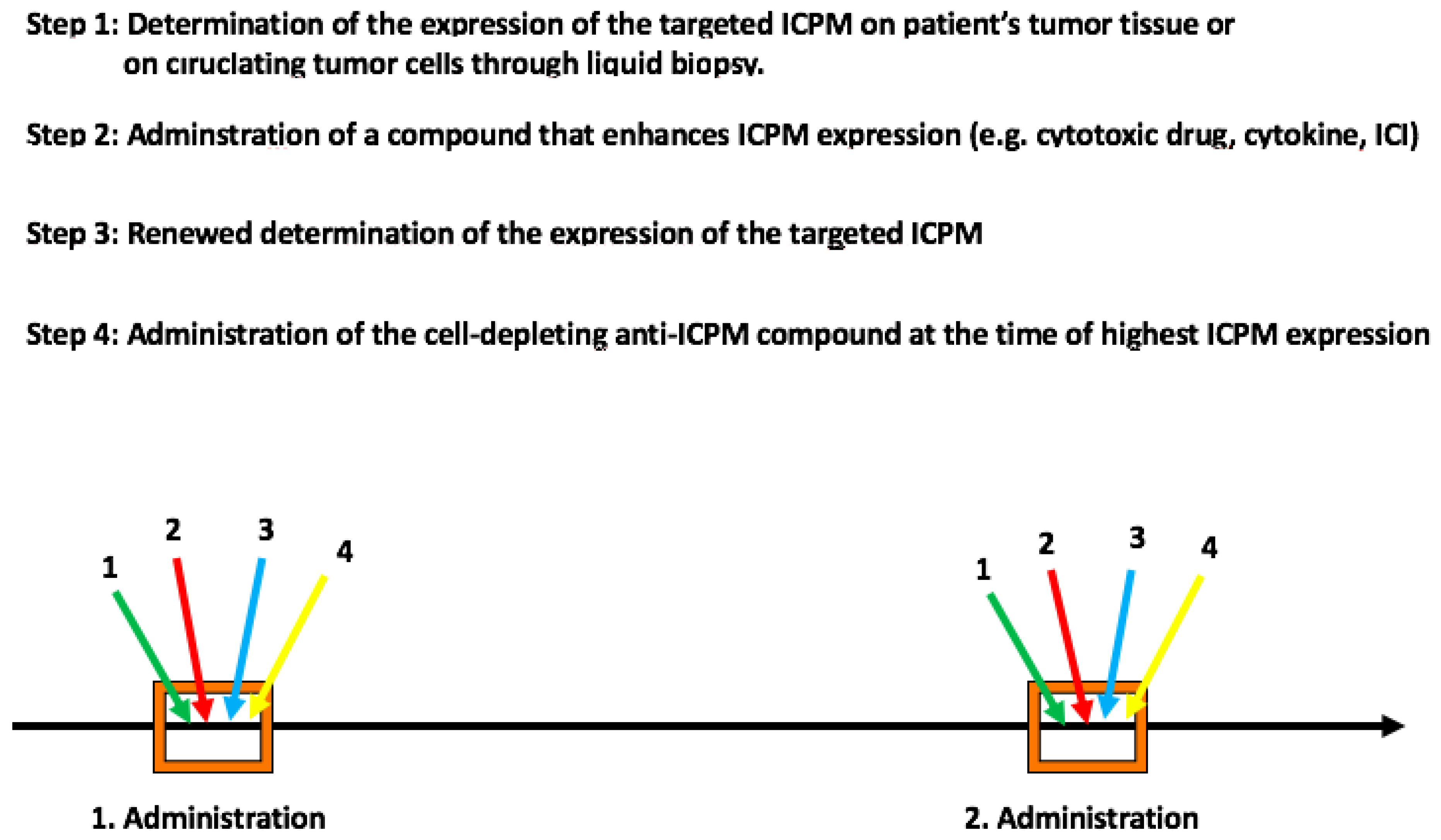
6. Improving the Efficacy of Depleting Compounds by Enhancing the Expression of Tumor Cell-Associated ICPMLs
7. Improving the Efficacy of Anti-ICPML Compounds by Increasing Their Cell-Depleting Potential
7.1. Antibodies with Increased Effector Functions or Direct Apoptotic Effects against ICPML+ Tumor Cells
7.2. Recruiting T-Cells for Depleting ICPML+ Tumor Cells
7.3. Non-Antibody-Based Approaches for the Depletion of ICPML+ Tumor Cells
8. Which Are the Most Promising ICPMLs as Targets for Cell-Depleting Compounds?
9. Conclusions and Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Sharpe, A.H.; Pauken, K.E. The diverse functions of the PD1 inhibitory pathway. Nat. Rev. Immunol. 2018, 18, 153–167. [Google Scholar] [CrossRef]
- Feng, M.; Jiang, W.; Kim, B.Y.S.; Zhang, C.C.; Fu, Y.X.; Weissman, I.L. Phagocytosis checkpoints as new targets for cancer immunotherapy. Nat. Rev. Cancer 2019, 19, 568–586. [Google Scholar] [CrossRef] [PubMed]
- Janakiram, M.; Shah, U.A.; Liu, W.; Zhao, A.; Schoenberg, M.P.; Zang, X. The third group of the B7-CD28 immune checkpoint family: HHLA2, TMIGD2, B7x, and B7-H3. Immunol. Rev. 2017, 276, 26–39. [Google Scholar] [CrossRef]
- Watts, T.H. TNF/TNFR family members in costimulation of T cell responses. Annu. Rev. Immunol. 2005, 23, 23–68. [Google Scholar] [CrossRef]
- Pardoll, D. The blockade of immune checkpoints in cancer immunotherapy. Nat. Rev. Cancer 2012, 12, 252–264. [Google Scholar] [CrossRef]
- Mayes, P.A.; Hance, K.W.; Hoos, A. The promise and challenges of immune agonist antibody development in cancer. Nat. Rev. Drug Discov. 2018, 17, 509–527. [Google Scholar] [CrossRef]
- Ribas, A.; Wolchok, J.D. Cancer immunotherapy using checkpoint blockade. Science 2018, 359, 1350–1355. [Google Scholar] [CrossRef] [PubMed]
- Andrews, L.P.; Yano, H.; Vignali, D.A.A. Inhibitory receptors and ligands beyond PD-1, PD-L1 and CTLA-4: Breakthroughs or backups. Nat. Immunol. 2019, 20, 1425–1434. [Google Scholar] [CrossRef]
- Marcucci, F.; Rumio, C.; Corti, A. Tumor cell-associated immune checkpoint molecules—Drivers of malignancy and stemness. Biochim. Biophys. Acta 2017, 1868, 571–583. [Google Scholar] [CrossRef] [PubMed]
- George, J.; Saito, M.; Tsuta, K.; Iwakawa, R.; Shiraishi, K.; Scheel, A.H.; Uchida, S.; Watanabe, S.I.; Nishikawa, R.; Noguchi, M.; et al. Genomic amplification of CD274 (PD-L1) in small-cell lung cancer. Clin. Cancer Res. 2017, 23, 1220–1226. [Google Scholar] [CrossRef] [PubMed]
- Ota, K.; Azuma, K.; Kawahara, A.; Hattori, S.; Iwama, E.; Tanizaki, J.; Harada, T.; Matsumoto, K.; Takayama, K.; Takamori, S.; et al. Induction of PD-L1 expression by the EML4-ALK oncoprotein and downstream signaling pathways in non-small cell lung cancer. Clin. Cancer Res. 2015, 21, 4014–4021. [Google Scholar] [CrossRef]
- Kataoka, K.; Shiraishi, Y.; Takeda, Y.; Sakata, S.; Matsumoto, M.; Nagano, S.; Maeda, T.; Nagata, Y.; Kitanaka, A.; Mizuno, S.; et al. Aberrant PD-L1 expression through 3’-UTR disruption in multiple cancers. Nature 2016, 534, 402–406. [Google Scholar] [CrossRef]
- Balan, M.; Mier y Teran, E.; Waaga-Gasser, A.M.; Gasser, M.; Choueiri, T.K.; Freeman, G.; Pal, S. Novel roles of c-Met in the survival of renal cancer cells through the regulation of HO-1 and PD-L1 expression. J. Biol. Chem. 2015, 290, 8110–8120. [Google Scholar] [CrossRef]
- Parsa, A.T.; Waldron, J.S.; Panner, A.; Crane, C.A.; Parney, I.F.; Barry, J.J.; Cachola, K.E.; Murray, J.C.; Tihan, T.; Jensen, M.C.; et al. Loss of tumor suppressor PTEN function increases B7-H1 expression and immunoresistance in glioma. Nat. Med. 2007, 13, 84–88. [Google Scholar] [CrossRef]
- Lastwika, K.J.; Wilson, W., 3rd; Li, Q.K.; Norris, J.; Xu, H.; Ghazarian, S.R.; Kitagawa, H.; Kawabata, S.; Taube, J.M.; Yao, S.; et al. Control of PD-L1 expression by oncogenic activation of the AKT–mTOR pathway in non–small cell lung cancer. Cancer Res. 2016, 76, 227–238. [Google Scholar] [CrossRef]
- Marzec, M.; Zhang, Q.; Goradia, A.; Raghunath, P.N.; Liu, X.; Paessler, M.; Wang, H.Y.; Wysocka, M.; Cheng, M.; Ruggeri, B.A.; et al. Oncogenic kinase NPM/ALK induces through STAT3 expression of immunosuppressive protein CD274 (PD-L1, B7-H1). Proc. Natl. Acad. Sci. USA 2008, 105, 20852–20857. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Yu, M.; Qu, M.; Ma, Y.; Zheng, D.; Yue, Y.; Guo, S.; Tang, L.; Li, G.; Zheng, W.; et al. Hepatitis B virus-triggered PTEN/β-catenin/c-Myc signaling enhances PD-L1 expression to promote immune evasion. Am. J. Physiol. Gastrointest. Liver Physiol. 2020, 318, G162–G173. [Google Scholar] [CrossRef] [PubMed]
- Noman, M.Z.; Desantis, G.; Janji, B.; Hasmim, M.; Karray, S.; Dessen, P.; Bronte, V.; Chouaib, S. PD-L1 is a novel direct target of HIF-1α, and its blockade under hypoxia enhanced MDSC-mediated T cell activation. J. Exp. Med. 2014, 211, 781–790. [Google Scholar] [CrossRef] [PubMed]
- Xiong, W.; Deng, H.; Huang, C.; Zen, C.; Jian, C.; Ye, K.; Zhong, Z.; Zhao, X.; Zhu, L. MLL3 enhances the transcription of PD-L1 and regulates anti-tumor immunity. Biochim. Biophys. Acta Mol. Basis Dis. 2019, 1865, 454–463. [Google Scholar] [CrossRef] [PubMed]
- Huang, K.C.; Chiang, S.F.; Chen, W.T.; Chen, T.W.; Hu, C.H.; Yang, P.C.; Ke, T.W.; Chao, K.S.C. Decitabine augments chemotherapy-induced PD-L1 upregulation for PD-L1 blockade in colorectal cancer. Cancers 2020, 12, 462. [Google Scholar] [CrossRef]
- Kondo, A.; Yamashita, T.; Tamura, H.; Zhao, W.; Tsuji, T.; Shimizu, M.; Shinya, E.; Takahashi, H.; Tamada, K.; Chen, L.; et al. Interferon-gamma and tumor necrosis factor-alpha induce an immunoinhibitory molecule, B7-H1, via nuclear factor-kappaB activation in blasts in myelodysplastic syndromes. Blood 2010, 116, 1124–1131. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.; Shin, J.H.; Longmire, M.; Wang, H.; Kohrt, H.E.; Chang, H.Y.; Sunwoo, J.B. CD44+ cells in head and neck squamous cell carcinoma suppress T-cell-mediated immunity by selective constitutive and inducible expression of PD-L1. Clin. Cancer Res. 2016, 22, 3571–3581. [Google Scholar] [CrossRef]
- Messai, Y.; Gad, S.; Noman, M.Z.; Le Teuff, G.; Couve, S.; Janji, B.; Kammerer, S.F.; Rioux-Leclerc, N.; Hasmim, M.; Ferlicot, S.; et al. Renal cell carcinoma programmed death-ligand 1, a new direct target of hypoxia-inducible factor-2α, is regulated by von Hippel-Lindau gene mutation status. Eur. Urol. 2016, 70, 623–632. [Google Scholar] [CrossRef] [PubMed]
- Peng, J.; Hamanishi, J.; Matsumura, N.; Abiko, K.; Murat, K.; Baba, T.; Yamaguchi, K.; Horikawa, N.; Hosoe, Y.; Murphy, S.K.; et al. Chemotherapy induces programmed cell death-ligand 1 overexpression via the nuclear factor-κB to foster an immunosuppressive tumor microenvironment in ovarian cancer. Cancer Res. 2015, 75, 5034–5045. [Google Scholar] [CrossRef] [PubMed]
- Feng, J.; Yang, H.; Zhang, Y.; Wei, H.; Zhu, Z.; Zhu, B.; Yang, M.; Cao, W.; Wang, L.; Wu, Z. Tumor cell-derived lactate induces TAZ-dependent upregulation of PD-L1 through GPR81 in human lung cancer cells. Oncogene 2017, 36, 5829–5839. [Google Scholar] [CrossRef]
- Jeon, Y.K.; Park, S.G.; Choi, I.W.; Lee, S.W.; Lee, S.M.; Choi, I. Cancer cell-associated cytoplasmic B7-H4 is induced by hypoxia through hypoxia-inducible factor-1α and promotes cancer cell proliferation. Biochem. Biophys. Res. Commun. 2015, 459, 277–283. [Google Scholar] [CrossRef]
- Ruf, M.; Mittmann, C.; Nowicka, A.M.; Hartmann, A.; Hermanns, T.; Poyet, C.; van den Broek, M.; Sulser, T.; Moch, H.; Schraml, P. pVHL/HIF-regulated CD70 expression is associated with infiltration of CD27+ lymphocytes and increased serum levels of soluble CD27 in clear cell renal cell carcinoma. Clin. Cancer Res. 2015, 21, 889–898. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Lu, H.; Xiang, L.; Bullen, J.W.; Zhang, C.; Samanta, D.; Gilkes, D.M.; He, J.; Semenza, G.L. HIF-1 regulates CD47 expression in breast cancer cells to promote evasion of phagocytosis and maintenance of cancer stem cells. Proc. Natl. Acad. Sci. USA 2015, 112, E6215–E6223. [Google Scholar] [CrossRef]
- Riether, C.; Schürch, C.M.; Flury, C.; Hinterbrandner, M.; Drück, L.; Huguenin, A.L.; Baerlocher, G.M.; Radpour, R.; Ochsenbein, A.F. Tyrosine kinase inhibitor-induced CD70 expression mediates drug resistance in leukemia stem cells by activating Wnt signaling. Sci. Transl. Med. 2015, 7, 298ra119. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Huang, C.; Zhang, Z.; Feng, Y.; Wang, Z.; Tang, X.; Zhong, K.; Hu, Y.; Guo, G.; Zhou, L.; et al. MEK inhibitor augments antitumor activity of B7-H3-redirected bispecific antibody. Front. Oncol. 2020, 10, 1527. [Google Scholar] [CrossRef]
- Glorieux, C.; Huang, P. Regulation of CD137 expression through K-Ras signaling in pancreatic cancer cells. Cancer Commun. 2019, 39, 41. [Google Scholar] [CrossRef] [PubMed]
- Kučan Brlić, P.; Lenac Roviš, T.; Cinamon, G.; Tsukerman, P.; Mandelboim, O.; Jonjić, S. Targeting PVR (CD155) and its receptors in anti-tumor therapy. Cell. Mol. Immunol. 2019, 16, 40–52. [Google Scholar] [CrossRef]
- Wang, J.; Wei, W.; Tang, Q.; Lu, L.; Luo, Z.; Li, W.; Lu, Y.; Pu, J. Oxysophocarpine suppresses hepatocellular carcinoma growth and sensitizes the therapeutic blockade of anti-Lag-3 via reducing FGL1 expression. Cancer Med. 2020, 9, 7125–7136. [Google Scholar] [CrossRef] [PubMed]
- Li, R.; Zatloukalova, P.; Muller, P.; Gil-Mir, M.; Kote, S.; Wilkinson, S.; Kemp, A.J.; Hernychova, L.; Wang, Y.; Ball, K.L.; et al. The MDM2 ligand Nutlin-3 differentially alters expression of the immune blockade receptors PD-L1 and CD276. Cell. Mol. Biol. Lett. 2020, 25, 41. [Google Scholar] [CrossRef] [PubMed]
- Keir, M.E.; Butte, M.J.; Freeman, G.J.; Sharpe, A.H. PD-1 and its ligands in tolerance and immunity. Annu. Rev. Immunol. 2008, 26, 677–704. [Google Scholar] [CrossRef]
- Sharma, P.; Allison, J.P. The future of immune checkpoint therapy. Science 2015, 348, 56–64. [Google Scholar] [CrossRef] [PubMed]
- Ni, L.; Dong, C. New B7 family checkpoints in human cancers. Mol. Cancer Ther. 2017, 16, 1203–1211. [Google Scholar] [CrossRef]
- Marcucci, F.; Rumio, C. Glycolysis-induced drug resistance in tumors-A response to danger signals? Neoplasia 2021, 23, 234–245. [Google Scholar] [CrossRef]
- Zhi, Y.; Mou, Z.; Chen, J.; He, Y.; Dong, H.; Fu, X.; Wu, Y. B7H1 expression and epithelial-to-mesenchymal transition phenotypes on colorectal cancer stem-like cells. PLoS ONE 2015, 10, e0135528. [Google Scholar] [CrossRef]
- Sanchez-Tillo, E.; Liu, Y.; De Barrios, O.; Siles, L.; Fanlo, L.; Cuatrecasas, M.; Darling, D.S.; Dean, D.C.; Castells, A.; Postigo, A. EMT-activating transcription factors in cancer: Beyond EMT and tumor invasiveness. Cell. Mol. Life Sci. 2012, 69, 3429–3456. [Google Scholar] [CrossRef]
- Dongre, A.; Weinberg, R.A. New insights into the mechanisms of epithelial-mesenchymal transition and implications for cancer. Nat. Rev. Mol. Cell Biol. 2019, 20, 69–84. [Google Scholar] [CrossRef]
- Marcucci, F.; Stassi, G.; De Maria, R. Epithelial–mesenchymal transition: A new target in anticancer drug discovery. Nat. Rev. Drug Discov. 2016, 15, 311–325. [Google Scholar] [CrossRef] [PubMed]
- Feng, Y.; Yang, Z.; Zhang, C.; Che, N.; Liu, X.; Xuan, Y. B7-H4 induces epithelial-mesenchymal transition and promotes colorectal cancer stemness. Pathol. Res. Pract. 2021, 218, 153323. [Google Scholar] [CrossRef] [PubMed]
- Zheng, Q.; Gao, J.; Yin, P.; Wang, W.; Wang, B.; Li, Y.; Zhao, C. CD155 contributes to the mesenchymal phenotype of triple-negative breast cancer. Cancer Sci. 2020, 111, 383–394. [Google Scholar] [CrossRef]
- Ge, Y.; Chen, W.; Zhang, X.; Wang, H.; Cui, J.; Liu, Y.; Ju, S.; Tian, X.; Ju, S. Nuclear-localized costimulatory molecule 4-1BBL promotes colon cancer cell proliferation and migration by regulating nuclear Gsk3β, and is linked to the poor outcomes associated with colon cancer. Cell Cycle 2020, 19, 577–591. [Google Scholar] [CrossRef]
- Zhang, Y.; Qiao, H.X.; Zhou, Y.T.; Hong, L.; Chen, J.H. Fibrinogen-like-protein 1 promotes the invasion and metastasis of gastric cancer and is associated with poor prognosis. Mol. Med. Rep. 2018, 18, 1465–1472. [Google Scholar] [CrossRef]
- Li, J.; Chen, L.; Xiong, Y.; Zheng, X.; Xie, Q.; Zhou, Q.; Shi, L.; Wu, C.; Jiang, J.; Wang, H. Knockdown of PD-L1 in human gastric cancer cells inhibits tumor progression and improves the cytotoxic sensitivity to CIK therapy. Cell. Physiol. Biochem. 2017, 41, 907–920. [Google Scholar] [CrossRef]
- Noman, M.Z.; Janji, B.; Abdou, A.; Hasmim, M.; Terry, S.; Tan, T.Z.; Mami-Chouaib, F.; Thiery, J.P.; Chouaib, S. The immune checkpoint ligand PD-L1 is upregulated in EMT-activated human breast cancer cells by a mechanism involving ZEB-1 and miR-200. Oncoimmunology 2017, 6, e1263412. [Google Scholar] [CrossRef] [PubMed]
- Jiang, B.; Zhang, T.; Liu, F.; Sun, Z.; Shi, H.; Hua, D.; Yang, C. The co-stimulatory molecule B7-H3 promotes the epithelial-mesenchymal transition in colorectal cancer. Oncotarget 2016, 7, 31755–31771. [Google Scholar] [CrossRef]
- Shan, B.; Man, H.; Liu, J.; Wang, L.; Zhu, T.; Ma, M.; Xv, Z.; Chen, X.; Yang, X.; Li, P. TIM-3 promotes the metastasis of esophageal squamous cell carcinoma by targeting epithelial-mesenchymal transition via the Akt/GSK-3β/Snail signaling pathway. Oncol. Rep. 2016, 36, 1551–1561. [Google Scholar] [CrossRef]
- Wang, Y.; Wang, H.; Zhao, Q.; Xia, Y.; Hu, X.; Guo, J. PD-L1 induces epithelial-to-mesenchymal transition via activating SREBP-1c in renal cell carcinoma. Med. Oncol. 2015, 32, 212. [Google Scholar] [CrossRef]
- Romeo, E.; Caserta, C.A.; Rumio, C.; Marcucci, F. The vicious cross-talk between tumor cells with an EMT phenotype and cells of the immune system. Cells 2019, 8, 460. [Google Scholar] [CrossRef] [PubMed]
- Shields, B.D.; Koss, B.; Taylor, E.M.; Storey, A.J.; West, K.L.; Byrum, S.D.; Mackintosh, S.G.; Edmondson, R.; Mahmoud, F.; Shalin, S.C.; et al. Loss of E-cadherin inhibits CD103 antitumor activity and reduces checkpoint blockade responsiveness in melanoma. Cancer Res. 2019, 79, 1113–1123. [Google Scholar] [CrossRef]
- Mak, M.P.; Tong, P.; Diao, L.; Cardnell, R.J.; Gibbons, D.L.; William, W.N.; Skoulidis, F.; Parra, E.R.; Rodriguez-Canales, J.; Wistuba, I.I.; et al. A patient-derived, pan-cancer EMT signature identifies global molecular alterations and immune target enrichment following epithelial-to-mesenchymal transition. Clin. Cancer Res. 2016, 22, 609–620. [Google Scholar] [CrossRef] [PubMed]
- Lou, Y.; Diao, L.; Cuentas, E.R.P.; Denning, W.L.; Chen, L.; Fan, Y.H.; Byers, L.A.; Wang, J.; Papadimitrakopoulou, V.A.; Behrens, C.; et al. Epithelial–mesenchymal transition is associated with a distinct tumor microenvironment including elevation of inflammatory signals and multiple immune checkpoints in lung adenocarcinoma. Clin. Cancer Res. 2016, 22, 3630–3642. [Google Scholar] [CrossRef]
- Emran, A.A.; Chatterjee, A.; Rodger, E.J.; Tiffen, J.C.; Gallagher, S.J.; Eccles, M.R.; Hersey, P. Reactive oxygen species modulate macrophage immunosuppressive phenotype through the up-regulation of PD-L1. Proc. Natl. Acad. Sci. USA 2019, 116, 4326–4335. [Google Scholar]
- Ock, C.Y.; Kim, S.; Keam, B.; Kim, M.; Kim, T.M.; Kim, J.H.; Jeon, Y.K.; Lee, J.S.; Kwon, S.K.; Hah, J.H.; et al. PD-L1 expression is associated with epithelial-mesenchymal transition in head and neck squamous cell carcinoma. Oncotarget 2016, 7, 15901–15914. [Google Scholar] [CrossRef]
- Xie, N.; Cai, J.B.; Zhang, L.; Zhang, P.F.; Shen, Y.H.; Yang, X.; Lu, J.C.; Gao, D.M.; Kang, Q.; Liu, L.X.; et al. Upregulation of B7-H4 promotes tumor progression of intrahepatic cholangiocarcinoma. Cell Death Dis. 2017, 8, 3205. [Google Scholar] [CrossRef]
- Pai, S.; Bamodu, O.A.; Lin, Y.K.; Lin, C.S.; Chu, P.Y.; Chien, M.H.; Wang, L.S.; Hsiao, M.; Yeh, C.T.; Tsai, J.T. CD47-SIRPα signaling induces epithelial-mesenchymal transition and cancer stemness and links to a poor prognosis in patients with oral squamous cell carcinoma. Cells 2019, 8, 1658. [Google Scholar] [CrossRef] [PubMed]
- Noman, M.Z.; Van Moer, K.; Marani, V.; Gemmill, R.M.; Tranchevent, L.C.; Azuaje, F.; Muller, A.; Chouaib, S.; Thiery, J.P.; Berchem, G.; et al. CD47 is a direct target of SNAI1 and ZEB1 and its blockade activates the phagocytosis of breast cancer cells undergoing EMT. Oncoimmunology 2018, 7, e1345415. [Google Scholar] [CrossRef]
- Zhao, H.; Wang, J.; Kong, X.; Li, E.; Liu, Y.; Du, X.; Kang, Z.; Tang, Y.; Kuang, Y.; Yang, Z.; et al. CD47 promotes tumor invasion and metastasis in non-small cell lung cancer. Sci. Rep. 2016, 6, 29719. [Google Scholar] [CrossRef]
- Grimmig, T.; Gasser, M.; Moench, R.; Zhu, L.J.; Nawalaniec, K.; Callies, S.; Wagner, M.; Polat, B.; Mothi, S.S.; Luo, Y.; et al. Expression of tumor-mediated CD137 ligand in human colon cancer indicates dual signaling effects. Oncoimmunology 2019, 8, e1651622. [Google Scholar] [CrossRef]
- Xiao, Y.; Qing, J.; Li, B.; Chen, L.; Nong, S.; Yang, W.; Tang, X.; Chen, Z. TIM-3 participates in the invasion and metastasis of nasopharyngeal carcinoma via SMAD7/SMAD2/SNAIL1 axis-mediated epithelial-mesenchymal transition. OncoTargets Ther. 2020, 13, 1993–2006. [Google Scholar] [CrossRef]
- Bertino, P.; Premeaux, T.A.; Fujita, T.; Haun, B.K.; Marciel, M.P.; Hoffmann, F.W.; Garcia, A.; Yiang, H.; Pastorino, S.; Carbone, M.; et al. Targeting the C-terminus of galectin-9 induces mesothelioma apoptosis and M2 macrophage depletion. Oncoimmunology 2019, 8, 1601482. [Google Scholar] [CrossRef]
- Ge, H.; Mu, L.; Jin, L.; Yang, C.; Chang, Y.E.; Long, Y.; DeLeon, G.; Deleyrolle, L.; Mitchell, D.A.; Kubilis, P.S.; et al. Tumor associated CD70 expression is involved in promoting tumor migration and macrophage infiltration in GBM. Int. J. Cancer 2017, 141, 1434–1444. [Google Scholar] [CrossRef] [PubMed]
- Pich, C.; Sarrabayrouse, G.; Teiti, I.; Mariamé, B.; Rochaix, P.; Lamant, L.; Favre, G.; Maisongrosse, V.; Tilkin-Mariamé, A.F. Melanoma-expressed CD70 is involved in invasion and metastasis. Br. J. Cancer 2016, 114, 63–70. [Google Scholar] [CrossRef] [PubMed]
- Aggarwal, S.; He, T.; Fitzhugh, W.; Rosenthal, K.; Field, B.; Heidbrink, J.; Mesmer, D.; Ruben, S.M.; Moore, P.A. Immune modulator CD70 as a potential cisplatin resistance predictive marker in ovarian cancer. Gynecol. Oncol. 2009, 115, 430–437. [Google Scholar] [CrossRef]
- Budczies, J.; Bockmayr, M.; Denkert, C.; Klauschen, F.; Gröschel, S.; Darb-Esfahani, S.; Pfarr, N.; Leichsenring, J.; Onozato, M.L.; Lennerz, J.K.; et al. Pan-cancer analysis of copy number changes in programmed death-ligand 1 (PD-L1, CD274)—Associations with gene expression, mutational load, and survival. Genes Chromosomes Cancer 2016, 55, 626–639. [Google Scholar] [CrossRef]
- Barrett, M.T.; Anderson, K.S.; Lenkiewicz, E.; Andreozzi, M.; Cunliffe, H.E.; Klassen, C.L.; Dueck, A.C.; McCullough, A.E.; Reddy, S.K.; Ramanathan, R.K.; et al. Genomic amplification of 9p24.1 targeting JAK2, PD-L1, and PD-L2 is enriched in high-risk triple negative breast cancer. Oncotarget 2015, 6, 26483–26493. [Google Scholar] [CrossRef] [PubMed]
- Pastushenko, I.; Brisebarre, A.; Sifrim, A.; Fioramonti, M.; Revenco, T.; Boumahdi, S.; Van Keymeulen, A.; Brown, D.; Moers, V.; Lemaire, S.; et al. Identification of the tumour transition states occurring during EMT. Nature 2018, 556, 463–468. [Google Scholar] [CrossRef]
- Calderaro, J.; Rousseau, B.; Amaddeo, G.; Mercey, M.; Charpy, C.; Costentin, C.; Luciani, A.; Zafrani, E.S.; Laurent, A.; Azoulay, D.; et al. Programmed death ligand 1 expression in hepatocellular carcinoma: Relationship with clinical and pathological features. Hepatology 2016, 64, 2038–2046. [Google Scholar] [CrossRef]
- Madore, J.; Vilain, R.E.; Menzies, A.M.; Kakavand, H.; Wilmott, J.S.; Hyman, J.; Yearley, J.H.; Kefford, R.F.; Thompson, J.F.; Long, G.V.; et al. PD-L1 expression in melanoma shows marked heterogeneity within and between patients: Implications for anti-PD-1/PD-L1 clinical trials. Pigment. Cell Melanoma Res. 2015, 28, 245–253. [Google Scholar] [CrossRef]
- Lepletier, A.; Madore, J.; O’Donnell, J.S.; Johnston, R.L.; Li, X.Y.; McDonald, E.; Ahern, E.; Kuchel, A.; Eastgate, M.; Pearson, S.A.; et al. Tumor CD155 expression is associated with resistance to anti-PD1 immunotherapy in metastatic melanoma. Clin. Cancer Res. 2020, 26, 3671–3681. [Google Scholar] [CrossRef]
- Song, X.; Zhou, Z.; Li, H.; Xue, Y.; Lu, X.; Bahar, I.; Kepp, O.; Hung, M.C.; Kroemer, G.; Wan, Y. Pharmacologic suppression of B7-H4 glycosylation restores antitumor immunity in immune-cold breast cancers. Cancer Discov. 2020, 10, 1872–1893. [Google Scholar] [CrossRef]
- Cheng, H.; Borczuk, A.; Janakiram, M.; Ren, X.; Lin, J.; Assal, A.; Halmos, B.; Perez-Soler, R.; Zang, X. Wide expression and significance of alternative immune checkpoint molecules, B7x and HHLA2, in PD-L1–negative human lung cancers. Clin. Cancer Res. 2018, 24, 1954–1964. [Google Scholar] [CrossRef]
- Guo, M.; Yuan, F.; Qi, F.; Sun, J.; Rao, Q.; Zhao, Z.; Huang, P.; Fang, T.; Yang, B.; Xia, J. Expression and clinical significance of LAG-3, FGL1, PD-L1 and CD8 + T cells in hepatocellular carcinoma using multiplex quantitative analysis. J. Transl. Med. 2020, 18, 306. [Google Scholar] [CrossRef]
- Malissen, N.; Macagno, N.; Granjeaud, S.; Granier, C.; Moutardier, V.; Gaudy-Marqueste, C.; Habel, N.; Mandavit, M.; Guillot, B.; Pasero, C.; et al. HVEM has a broader expression than PD-L1 and constitutes a negative prognostic marker and potential treatment target for melanoma. Oncoimmunology 2019, 8, e1665976. [Google Scholar] [CrossRef]
- Koyama, S.; Akbay, E.A.; Li, Y.Y.; Herter-Sprie, G.S.; Buczkowski, K.A.; Richards, W.G.; Gandhi, L.; Redig, A.J.; Rodig, S.J.; Asahina, H.; et al. Adaptive resistance to therapeutic PD-1 blockade is associated with upregulation of alternative immune checkpoints. Nat. Commun. 2016, 7, 10501. [Google Scholar] [CrossRef]
- Stewart, R.; Morrow, M.; Hammond, S.A.; Mulgrew, K.; Marcus, D.; Poon, E.; Watkins, A.; Mullins, S.; Chodorge, M.; Andrews, J.; et al. Identification and characterization of MEDI4736, an antagonistic anti-PD-L1 monoclonal antibody. Cancer Immunol. Res. 2015, 3, 1052–1062. [Google Scholar] [CrossRef]
- Casella, G.; Rasouli, J.; Thome, R.; Descamps, H.C.; Vattikonda, A.; Ishikawa, L.; Boehm, A.; Hwang, D.; Zhang, W.; Xiao, D.; et al. Interferon-γ/Interleukin-27 axis induces programmed death ligand 1 expression in monocyte-derived dendritic cells and restores immune tolerance in central nervous system autoimmunity. Front. Immunol. 2020, 11, 576752. [Google Scholar] [CrossRef]
- Zong, Z.; Zou, J.; Mao, R.; Ma, C.; Li, N.; Wang, J.; Wang, X.; Zhou, H.; Zhang, L.; Shi, Y. M1 macrophages induce PD-L1 expression in hepatocellular carcinoma cells through IL-1β signaling. Front. Immunol. 2019, 10, 1643. [Google Scholar] [CrossRef]
- Shklovskaya, E.; Rizos, H. spatial and temporal changes in PD-L1 expression in cancer: The role of genetic drivers, tumor microenvironment and resistance to therapy. Int. J. Mol. Sci. 2020, 21, 7139. [Google Scholar] [CrossRef]
- Lee, Y.; Moon, Y.; Hyung, K.; Yoo, J. Macrophage PD-L1 strikes back: PD-1/PDL1 interaction drives macrophages toward regulatory subsets. Adv. Biosci. 2013, 4, 19–29. [Google Scholar] [CrossRef]
- Herbst, R.S.; Soria, J.C.; Kowanetz, M.; Fine, G.D.; Hamid, O.; Gordon, M.S.; Sosman, J.A.; McDermott, D.F.; Powderly, J.D.; Gettinger, S.N.; et al. Predictive correlates of response to the anti-PD-L1 antibody MPDL3280A in cancer patients. Nature 2014, 515, 563–567. [Google Scholar] [CrossRef]
- Kryczek, I.; Zou, L.; Rodriguez, P.; Zhu, G.; Wei, S.; Mottram, P.; Brumlik, M.; Cheng, P.; Curiel, T.; Myers, L.; et al. B7-H4 expression identifies a novel suppressive macrophage population in human ovarian carcinoma. J. Exp. Med. 2006, 203, 871–881. [Google Scholar] [CrossRef]
- Hao, N.B.; Lü, M.H.; Fan, Y.H.; Cao, Y.L.; Zhang, Z.R.; Yang, S.M. Macrophages in tumor microenvironments and the progression of tumors. Clin. Dev. Immunol. 2012, 2012, 948098. [Google Scholar] [CrossRef]
- Pulko, V.; Harris, K.J.; Liu, X.; Gibbons, R.M.; Harrington, S.M.; Krco, C.J.; Kwon, E.D.; Dong, H. B7-h1 expressed by activated CD8 T cells is essential for their survival. J. Immunol. 2011, 187, 5606–5614. [Google Scholar] [CrossRef]
- Liu, X.; Wu, X.; Cao, S.; Harrington, S.M.; Yin, P.; Mansfield, A.S.; Dong, H. B7-H1 antibodies lose antitumor activity due to activation of p38 MAPK that leads to apoptosis of tumor-reactive CD8(+) T cells. Sci. Rep. 2016, 6, 36722. [Google Scholar] [CrossRef]
- Diskin, B.; Adam, S.; Cassini, M.F.; Sanchez, G.; Liria, M.; Aykut, B.; Buttar, C.; Li, E.; Sundberg, B.; Salas, R.D.; et al. PD-L1 engagement on T cells promotes self-tolerance and suppression of neighboring macrophages and effector T cells in cancer. Nat. Immunol. 2020, 21, 442–454. [Google Scholar] [CrossRef]
- Teramoto, K.; Igarashi, T.; Kataoka, Y.; Ishida, M.; Hanaoka, J.; Sumimoto, H.; Daigo, Y. Clinical significance of PD-L1-positive cancer-associated fibroblasts in pN0M0 non-small cell lung cancer. Lung Cancer 2019, 137, 56–63. [Google Scholar] [CrossRef]
- Li, X.Y.; Das, I.; Lepletier, A.; Addala, V.; Bald, T.; Stannard, K.; Barkauskas, D.; Liu, J.; Aguilera, A.R.; Takeda, K.; et al. CD155 loss enhances tumor suppression via combined host and tumor-intrinsic mechanisms. J. Clin. Investig. 2018, 128, 2613–2625. [Google Scholar] [CrossRef]
- Juneja, V.R.; McGuire, K.A.; Manguso, R.T.; LaFleur, M.W.; Collins, N.; Haining, W.N.; Freeman, G.J.; Sharpe, A.H. PD-L1 on tumor cells is sufficient for immune evasion in immunogenic tumors and inhibits CD8 T cell cytotoxicity. J. Exp. Med. 2017, 214, 895–904. [Google Scholar] [CrossRef]
- Kleinovink, J.W.; van Hall, T.; Ossendorp, F.; Fransen, M.F. PD-L1 immune suppression in cancer: Tumor cells or host cells? Oncoimmunology 2017, 6, e1294299. [Google Scholar] [CrossRef] [PubMed]
- Dong, W.; Wu, X.; Ma, S.; Wang, Y.; Nalin, A.P.; Zhu, Z.; Zhang, J.; Benson, D.M.; He, K.; Caligiuri, M.A.; et al. The mechanism of anti-PD-L1 antibody efficacy against PD-L1-negative tumors identifies NK cells expressing PD-L1 as a cytolytic effector. Cancer Discov. 2019, 9, 1422–1437. [Google Scholar] [CrossRef] [PubMed]
- Champiat, S.; Dercle, L.; Ammari, S.; Massard, C.; Hollebecque, A.; Postel-Vinay, S.; Chaput, N.; Eggermont, A.; Marabelle, A.; Soria, J.C.; et al. Hyperprogressive disease is a new pattern of progression in cancer patients treated by anti-PD-1/PDL1. Clin. Cancer Res. 2017, 23, 1920–1928. [Google Scholar] [CrossRef]
- Ferrara, R.; Mezquita, L.; Texier, M.; Lahmar, J.; Audigier-Valette, C.; Tessonnier, L.; Mazieres, J.; Zalcman, G.; Brosseau, S.; Le Moulec, S.; et al. Hyperprogressive disease in patients with advanced non-small cell lung cancer treated with PD-1/PD-L1 inhibitors or with single-agent chemotherapy. JAMA Oncol. 2018, 4, 1543–1552. [Google Scholar] [CrossRef] [PubMed]
- Marcucci, F.; Rumio, C. The tumor-promoting effects of the adaptive immune system: A cause of hyperprogressive disease in cancer? Cell. Mol. Life Sci. 2021, 78, s00018–s00020. [Google Scholar] [CrossRef]
- Daud, A.I.; Wolchok, J.D.; Robert, C.; Hwu, W.J.; Weber, J.S.; Ribas, A.; Hodi, F.S.; Joshua, A.M.; Kefford, R.; Hersey, P.; et al. Programmed death-ligand 1 expression and response to the anti-programmed death 1 antibody pembrolizumab in melanoma. J. Clin. Oncol. 2016, 34, 4102–4109. [Google Scholar] [CrossRef]
- Robert, C.; Long, G.V.; Brady, B.; Dutriaux, C.; Maio, M.; Mortier, L.; Hassel, J.C.; Rutkowski, P.; McNeil, C.; Kalinka-Warzocha, E.; et al. Nivolumab in previously untreated melanoma without BRAF mutation. N. Engl. J. Med. 2014, 372, 320–330. [Google Scholar] [CrossRef]
- Taube, J.M.; Klein, A.; Brahmer, J.R.; Xu, H.; Pan, X.; Kim, J.H.; Chen, L.; Pardoll, D.M.; Topalian, S.L.; Anders, R.A. Association of PD-1, PD-1 ligands, and other features of the tumor immune microenvironment with response to anti-PD-1 therapy. Clin. Cancer Res. 2014, 1, 5064–5075. [Google Scholar] [CrossRef]
- Dahan, R.; Sega, E.; Engelhardt, J.; Selby, M.; Korman, A.J.; Ravetch, J.V. FcγRs modulate the anti-tumor activity of antibodies targeting the PD-1/PD-L1 axis. Cancer Cell 2015, 28, 285–295. [Google Scholar] [CrossRef]
- Boyerinas, B.; Jochems, C.; Fantini, M.; Heery, C.R.; Gulley, J.L.; Tsang, K.; Schlom, J. Antibody-dependent cellular cytotoxicity activity of a novel anti-PD-L1 antibody avelumab (MSB0010718C) on human tumor cells. Cancer Immunol. Res. 2015, 3, 1148–1157. [Google Scholar] [CrossRef]
- Khanna, S.; Thomas, A.; Abate-Daga, D.; Zhang, J.; Morrow, B.; Steinberg, S.M.; Orlandi, A.; Ferroni, P.; Schlom, J.; Guadagni, F.; et al. Malignant mesothelioma effusions are infiltrated by CD3 + T Cells highly expressing PD-L1 and the PD-L1 + tumor cells within these effusions are susceptible to ADCC by the anti-PD-L1 antibody avelumab. J. Thorac. Oncol. 2016, 11, 1993–2005. [Google Scholar] [CrossRef]
- Giles, A.J.; Hao, S.; Padget, M.; Song, H.; Zhang, W.; Lynes, J.; Sanchez, V.; Liu, Y.; Jung, J.; Cao, X.; et al. Efficient ADCC killing of meningioma by avelumab and a high-affinity natural killer cell line, haNK. JCI Insight 2019, 4, e130688. [Google Scholar] [CrossRef]
- Hamilton, G.; Rath, B. Avelumab: Combining immune checkpoint inhibition and antibody-dependent cytotoxicity. Expert Opin. Biol. Ther. 2017, 17, 515–523. [Google Scholar] [CrossRef]
- Byrd, J.C.; Kipps, T.J.; Flinn, I.W.; Cooper, M.; Odenike, O.; Bendiske, J.; Rediske, J.; Bilic, S.; Dey, J.; Baeck, J.; et al. Phase I study of the anti-CD40 humanized monoclonal antibody lucatumumab (HCD122) in relapsed chronic lymphocytic leukemia. Leuk. Lymphoma 2012, 53, 2136–2142. [Google Scholar] [CrossRef]
- Kim, M.J.; Lee, J.C.; Lee, J.J.; Kim, S.; Lee, S.G.; Park, S.W.; Sung, M.W.; Heo, D.S. Association of CD47 with natural killer cell-mediated cytotoxicity of head-and-neck squamous cell carcinoma lines. Tumour Biol. 2008, 29, 28–34. [Google Scholar] [CrossRef]
- Gao, Y.; Zhang, D.; Yang, C.; Duan, X.; Li, X.; Zhong, D. Two validated liquid chromatography-mass spectrometry methods with different pretreatments for the quantification of an anti-CD47 monoclonal antibody in rat and cynomolgus monkey serum compared with an electrochemiluminescence method. J. Pharm. Biomed. Anal. 2019, 175, 112792. [Google Scholar] [CrossRef]
- Park, J.E.; Kim, S.E.; Keam, B.; Park, H.R.; Kim, S.; Kim, M.; Kim, T.M.; Doh, J.; Kim, D.W.; Heo, D.S. Anti-tumor effects of NK cells and anti-PD-L1 antibody with antibody-dependent cellular cytotoxicity in PD-L1-positive cancer cell lines. J. Immunother. Cancer 2020, 8, e000873. [Google Scholar] [CrossRef]
- McEarchern, J.A.; Smith, L.M.; McDonagh, C.F.; Klussman, K.; Gordon, K.A.; Morris-Tilden, C.A.; Duniho, S.; Ryan, M.; Boursalian, T.E.; Carter, P.J.; et al. Preclinical characterization of SGN-70, a humanized antibody directed against CD70. Clin. Cancer Res. 2008, 14, 7763–7772. [Google Scholar] [CrossRef]
- Jeon, H.; Vigdorovich, V.; Garrett-Thomson, S.C.; Janakiram, M.; Ramagopal, U.A.; Abadi, Y.M.; Lee, J.S.; Scandiuzzi, L.; Ohaegbulam, K.C.; Chinai, J.M.; et al. Structure and cancer immunotherapy of the B7 family member B7x. Cell Rep. 2014, 9, 1089–1098. [Google Scholar] [CrossRef]
- Dong, H.; Strome, S.E.; Salomao, D.R.; Tamura, H.; Hirano, F.; Flies, D.B.; Roche, P.C.; Lu, J.; Zhu, G.; Tamada, K.; et al. Tumor-associated B7-H1 promotes T-cell apoptosis: A potential mechanism of immune evasion. Nat. Med. 2002, 8, 793–800. [Google Scholar] [CrossRef]
- Ju, X.; Zhang, H.; Zhou, Z.; Chen, M.; Wang, Q. Tumor-associated macrophages induce PD-L1 expression in gastric cancer cells through IL-6 and TNF-α signaling. Exp. Cell Res. 2020, 396, 112315. [Google Scholar] [CrossRef]
- Zhou, X.; Mao, Y.; Zhu, J.; Meng, F.; Chen, Q.; Tao, L.; Li, R.; Fu, F.; Liu, C.; Hu, Y.; et al. TGF-β1 promotes colorectal cancer immune escape by elevating B7-H3 and B7-H4 via the miR-155/miR-143 axis. Oncotarget 2016, 7, 67196–67211. [Google Scholar] [CrossRef]
- Kryczek, I.; Wei, S.; Zou, L.; Zhu, G.; Mottram, P.; Xu, H.; Chen, L.; Zou, W. induction of B7-H4 on APCs through IL-10: Novel suppressive mode for regulatory T cells. J. Immunol. 2006, 177, 40–44. [Google Scholar] [CrossRef] [PubMed]
- Corti, A.; Curnis, F.; Rossoni, G.; Marcucci, F.; Gregorc, V. Peptide-mediated targeting of cytokines to tumor vasculature: The NGR-hTNF example. BioDrugs 2013, 27, 591–603. [Google Scholar] [CrossRef] [PubMed]
- Pasche, N.; Wulhfard, S.; Pretto, F.; Carugati, E.; Neri, D. The antibody-based delivery of interleukin-12 to the tumor neovasculature eradicates murine models of cancer in combination with paclitaxel. Clin. Cancer Res. 2012, 18, 4092–4103. [Google Scholar] [CrossRef]
- Vilain, R.E.; Menzies, A.M.; Wilmott, J.S.; Kakavand, H.; Madore, J.; Guminski, A.; Liniker, E.; Kong, B.Y.; Cooper, A.J.; Howle, J.R.; et al. Dynamic changes in PD-L1 expression and immune infiltrates early during treatment predict response to PD-1 blockade in melanoma. Clin. Cancer Res. 2017, 23, 5024–5033. [Google Scholar] [CrossRef]
- Yamazaki, T.; Akiba, H.; Koyanagi, A.; Azuma, M.; Yagita, H.; Okumura, K. Blockade of B7-H1 on macrophages suppresses CD4+ T cell proliferation by augmenting IFN-gamma-induced nitric oxide production. J. Immunol. 2005, 175, 1586–1592. [Google Scholar] [CrossRef] [PubMed]
- Marcucci, F.; Bellone, M.; Caserta, C.A.; Corti, A. Pushing tumor cells towards a malignant phenotype. Stimuli from the microenvironment, intercellular communications and alternative roads. Int. J. Cancer 2014, 135, 1265–1276. [Google Scholar] [CrossRef]
- Yang, M.; Liu, P.; Wang, K.; Glorieux, C.; Hu, Y.; Wen, S.; Jiang, W.; Huang, P. Chemotherapy induces tumor immune evasion by upregulation of programmed cell death ligand 1 expression in bone marrow stromal cells. Mol. Oncol. 2017, 11, 358–372. [Google Scholar] [CrossRef] [PubMed]
- Niu, C.; Jin, H.; Li, M.; Zhu, S.; Zhou, L.; Jin, F.; Zhou, Y.; Xu, D.; Xu, J.; Zhao, L.; et al. Low-dose bortezomib increases the expression of NKG2D and DNAM-1 ligands and enhances induced NK and γδ T cell-mediated lysis in multiple myeloma. Oncotarget 2017, 8, 5954–5964. [Google Scholar] [CrossRef] [PubMed]
- López-Cobo, S.; Pieper, N.; Campos-Silva, C.; García-Cuesta, E.M.; Reyburn, H.T.; Paschen, A.; Valés-Gómez, M. Impaired NK cell recognition of vemurafenib-treated melanoma cells is overcome by simultaneous application of histone deacetylase inhibitors. Oncoimmunology 2018, 7, e1392426. [Google Scholar] [CrossRef]
- Sheng, J.; Fang, W.; Yu, J.; Chen, N.; Zhan, J.; Ma, Y.; Yang, Y.; Yan, H.; Zhao, H.; Zhang, L. Expression of programmed death ligand-1 on tumor cells varies pre and post chemotherapy in non-small cell lung cancer. Sci. Rep. 2016, 6, 20090. [Google Scholar] [CrossRef]
- Zhang, J.; Bu, X.; Wang, H.; Zhu, Y.; Geng, Y.; Nihira, N.T.; Tan, Y.; Ci, Y.; Wu, F.; Dai, X.; et al. Cyclin D-CDK4 kinase destabilizes PD-L1 via cullin 3-SPOP to control cancer immune surveillance. Nature 2018, 553, 91–95. [Google Scholar] [CrossRef]
- Goel, S.; DeCristo, M.J.; Watt, A.C.; BrinJones, H.; Sceneay, J.; Li, B.B.; Khan, N.; Ubellacker, J.M.; Xie, S.; Metzger-Filho, O.; et al. CDK4/6 inhibition triggers anti-tumour immunity. Nature 2017, 548, 471–475. [Google Scholar] [CrossRef]
- Emran, A.A.; Chatterjee, A.; Rodger, E.J.; Tiffen, J.C.; Gallagher, S.J.; Eccles, M.R.; Hersey, P. Targeting DNA methylation and EZH2 Activity to Overcome Melanoma Resistance to Immunotherapy. Trends Immunol. 2019, 40, 328–344. [Google Scholar] [CrossRef] [PubMed]
- Iwasa, M.; Harada, T.; Oda, A.; Bat-Erdene, A.; Teramachi, J.; Tenshin, H.; Ashtar, M.; Oura, M.; Sogabe, K.; Udaka, K.; et al. PD-L1 upregulation in myeloma cells by panobinostat in combination with interferon-γ. Oncotarget 2019, 10, 1903–1917. [Google Scholar] [CrossRef]
- Zhou, S.; Zhao, X.; Yang, Z.; Yang, R.; Chen, C.; Zhao, K.; Wang, W.; Ma, Y.; Zhang, Q.; Wang, X. Neddylation inhibition upregulates PD-L1 expression and enhances the efficacy of immune checkpoint blockade in glioblastoma. Int. J. Cancer 2019, 145, 763–774. [Google Scholar] [CrossRef] [PubMed]
- Loo, D.; Alderson, R.F.; Chen, F.Z.; Huang, L.; Zhang, W.; Gorlatov, S.; Burke, S.; Ciccarone, V.; Li, H.; Yang, Y.; et al. Development of an Fc-enhanced anti-B7-H3 monoclonal antibody with potent antitumor activity. Clin. Cancer Res. 2012, 18, 3834–3845. [Google Scholar] [CrossRef] [PubMed]
- Seaman, S.; Zhu, Z.; Saha, S.; Zhang, X.M.; Yang, M.Y.; Hilton, M.B.; Morris, K.; Szot, C.; Morris, H.; Swing, D.A.; et al. Eradication of tumors through simultaneous ablation of CD276/B7-H3-positive tumor cells and tumor vasculature. Cancer Cell 2017, 31, 501–515. [Google Scholar] [CrossRef]
- Du, H.; Hirabayashi, K.; Ahn, S.; Kren, N.P.; Montgomery, S.A.; Wang, X.; Tiruthani, K.; Mirlekar, B.; Michaud, D.; Greene, K.; et al. Antitumor responses in the absence of toxicity in solid tumors by targeting B7-H3 via chimeric antigen receptor T cells. Cancer Cell 2019, 35, 221–237.e8. [Google Scholar] [CrossRef]
- Zhan, S.; Liu, Z.; Zhang, M.; Guo, T.; Quan, Q.; Huang, L.; Guo, L.; Cao, L.; Zhang, X. Overexpression of B7-H3 in α-SMA-positive fibroblasts is associated with cancer progression and survival in gastric adenocarcinomas. Front. Oncol. 2020, 9, 1466. [Google Scholar] [CrossRef]
- Dutsch-Wicherek, M.; Kazmierczak, W. Creation of a suppressive microenvironment by macrophages and cancer-associated fibroblasts. Front. Biosci. 2013, 18, 1003–1016. [Google Scholar] [CrossRef]
- Aftimos, P.; Rolfo, C.; Rottey, S.; Offner, F.; Bron, D.; Maerevoet, M.; Soria, J.C.; Moshir, M.; Dreier, T.; Van Rompaey, L.; et al. Phase I Dose-escalation study of the anti-CD70 antibody ARGX-110 in advanced malignancies. Clin. Cancer Res. 2017, 23, 6411–6420. [Google Scholar] [CrossRef]
- Hermans, C.; Rolfo, C.; Peeters, M.; De Wever, O.; Lardon, F.; Siozopoulou, V.; Smits, E.; Pauwels, P. Unveiling a CD70-positive subset of cancer-associated fibroblasts marked by pro-migratory activity and thriving regulatory T cell accumulation. Oncoimmunology 2018, 7, e1440167. [Google Scholar] [CrossRef]
- De Meulenaere, A.; Vermassen, T.; Aspeslagh, S.; Zwaenepoel, K.; Deron, P.; Duprez, F.; Ferdinande, L.; Rottey, S. CD70 expression and its correlation with clinicopathological variables in squamous cell carcinoma of the head and neck. Pathobiology 2016, 83, 327–333. [Google Scholar] [CrossRef]
- Escalante, N.K.; von Rossum, A.; Lee, M.; Choy, J.C. CD155 on human vascular endothelial cells attenuates the acquisition of effector functions in CD8 T cells. Arterioscler. Thromb. Vasc Biol. 2011, 31, 1177–1184. [Google Scholar] [CrossRef]
- Thomas, L.J.; Vitale, L.; O’Neill, T.; Dolnick, R.Y.; Wallace, P.K.; Minderman, H.; Gergel, L.E.; Forsberg, E.M.; Boyer, J.M.; Storey, J.R.; et al. Development of a novel antibody-drug conjugate for the potential treatment of ovarian, lung, and renal cell carcinoma expressing TIM-1. Mol. Cancer Ther. 2016, 15, 2946–2954. [Google Scholar] [CrossRef]
- Kaur, S.; Cicalese, K.V.; Bannerjee, R.; Roberts, D.D. Preclinical and clinical development of therapeutic antibodies targeting functions of CD47 in the tumor microenvironment. Antib. Ther. 2020, 3, 179–192. [Google Scholar] [CrossRef]
- Griffioen, A.W.; Thijssen, V.L. Galectins in tumor angiogenesis. Ann. Transl. Med. 2014, 2, 90. [Google Scholar] [CrossRef]
- Lewis, T.S.; McCormick, R.S.; Stone, I.J.; Emmerton, K.; Mbow, B.; Miyamoto, J.; Drachman, J.G.; Grewal, I.S.; Law, C.L. Proapoptotic signaling activity of the anti-CD40 monoclonal antibody dacetuzumab circumvents multiple oncogenic transformation events and chemosensitizes NHL cells. Leukemia 2011, 25, 1007–1016. [Google Scholar] [CrossRef][Green Version]
- Kluth, B.; Hess, S.; Engelmann, H.; Schafnitzel, S.; Riethmüller, G.; Feucht, H.E. Endothelial expression of CD40 in renal cell carcinoma. Cancer Res. 1997, 57, 891–899. [Google Scholar]
- Silence, K.; Dreier, T.; Moshir, M.; Ulrichts, P.; Gabriels, S.M.; Saunders, M.; Wajant, H.; Brouckaert, P.; Huyghe, L.; Van Hauwermeiren, T.; et al. ARGX-110, a highly potent antibody targeting CD70, eliminates tumors via both enhanced ADCC and immune checkpoint blockade. mAbs 2014, 6, 523–532. [Google Scholar] [CrossRef]
- Oflazoglu, E.; Stone, I.J.; Gordon, K.; Wood, C.G.; Repasky, E.A.; Grewal, I.S.; Law, C.-L.; Gerber, H.-P. Potent anticarcinoma activity of the humanized anti-CD70 antibody h1F6 conjugated to the tubulin inhibitor auristatin via an uncleavable linker. Clin. Cancer Res. 2008, 14, 6171–6180. [Google Scholar] [CrossRef]
- Tannir, N.M.; Forero-Torres, A.; Ramchandren, R.; Pal, S.K.; Ansell, S.M.; Infante, J.R.; de Vos, S.; Hamlin, P.A.; Kim, S.K.; Whiting, N.C.; et al. Phase I dose-escalation study of SGN-75 in patients with CD70-positive relapsed/refractory non-Hodgkin lymphoma or metastatic renal cell carcinoma. Investig. New Drugs 2014, 32, 1246–1257. [Google Scholar] [CrossRef]
- Jeffrey, S.C.; Burke, P.J.; Lyon, R.P.; Meyer, D.W.; Sussman, D.; Anderson, M.; Hunter, J.H.; Leiske, C.I.; Miyamoto, J.B.; Nicholas, N.D.; et al. A potent anti-CD70 antibody−drug conjugate combining a dimeric pyrrolobenzodiazepine drug with site-specific conjugation technology. Bioconjugate Chem. 2013, 24, 1256–1263. [Google Scholar] [CrossRef] [PubMed]
- Phillips, T.; Barr, P.M.; Park, S.I.; Kolibaba, K.; Caimi, P.F.; Chhabra, S.; Kingsley, E.C.; Boyd, T.; Chen, R.; Carret, A.S.; et al. A phase 1 trial of SGN-CD70A in patients with CD70-positive diffuse large B cell lymphoma and mantle cell lymphoma. Investig. New Drugs 2019, 37, 297–306. [Google Scholar] [CrossRef]
- Wang, H.; Rangan, V.S.; Sung, M.C.; Passmore, D.; Kempe, T.; Wang, X.; Thevanayagam, L.; Pan, C.; Rao, C.; Srinivasan, M.; et al. Pharmacokinetic characterization of BMS-936561, an anti-CD70 antibody-drug conjugate, in preclinical animal species and prediction of its pharmacokinetics in humans. Biopharm. Drug Dispos. 2016, 37, 93–106. [Google Scholar] [CrossRef]
- Owonikoko, T.K.; Hussain, A.; Stadler, W.M.; Smith, D.C.; Kluger, H.; Molina, A.M.; Gulati, P.; Shah, A.; Ahlers, C.M.; Cardarelli, P.M.; et al. First-in-human multicenter phase I study of BMS-936561 (MDX-1203), an antibody-drug conjugate targeting CD70. Cancer Chemother. Pharmacol. 2016, 77, 155–162. [Google Scholar] [CrossRef]
- Scribner, J.A.; Brown, J.G.; Son, T.; Chiechi, M.; Li, P.; Sharma, S.; Li, H.; De Costa, A.; Li, Y.; Chen, Y.; et al. Preclinical development of MGC018, a duocarmycin-based antibody-drug conjugate targeting B7-H3 for solid cancer. Mol. Cancer Ther. 2020, 19. [Google Scholar] [CrossRef] [PubMed]
- Zhu, L.; Liu, J.; Zhou, G.; Ng, H.M.; Ang, I.L.; Ma, G.; Liu, Y.; Yang, S.; Zhang, F.; Miao, K.; et al. Targeting immune checkpoint B7-H3 antibody-chlorin e6 bioconjugates for spectroscopic photoacoustic imaging and photodynamic therapy. Chem. Commun. 2019, 55, 14255–14258. [Google Scholar] [CrossRef]
- Leong, S.R.; Liang, W.C.; Wu, Y.; Crocker, L.; Cheng, E.; Sampath, D.; Ohri, R.; Raab, H.; Hass, P.E.; Pham, T.; et al. An anti-B7-H4 antibody-drug conjugate for the treatment of breast cancer. Mol. Pharm. 2015, 12, 1717–1729. [Google Scholar] [CrossRef]
- Li, C.W.; Lim, S.O.; Chung, E.M.; Kim, Y.S.; Park, A.H.; Yao, J.; Cha, J.H.; Xia, W.; Chan, L.C.; Kim, T.; et al. Eradication of triple-negative breast cancer cells by targeting glycosylated PD-L1. Cancer Cell 2018, 33, 187–201.e10. [Google Scholar] [CrossRef]
- Kalim, M.; Wang, S.; Liang, K.; Khan, M.S.I.; Zhan, J. Engineered scPDL1-DM1 drug conjugate with improved in vitro analysis to target PD-L1 positive cancer cells and intracellular trafficking studies in cancer therapy. Genet. Mol. Biol. 2020, 42, e20180391. [Google Scholar] [CrossRef] [PubMed]
- Sau, S.; Petrovici, A.; Alsaab, H.O.; Bhise, K.; Iyer, A.K. PDL-1 antibody drug conjugate for selective chemo-guided immune modulation of cancer. Cancers 2019, 11, 232. [Google Scholar] [CrossRef] [PubMed]
- Sun, X.; Yu, Y.; Ma, L.; Xue, X.; Gao, Z.; Ma, J.; Zhang, M. T cell cytotoxicity toward hematologic malignancy via B7-H3 targeting. Investig. New Drugs 2020, 38, 722–732. [Google Scholar] [CrossRef] [PubMed]
- Zheng, M.; Yu, L.; Hu, J.; Zhang, Z.; Wang, H.; Lu, D.; Tang, X.; Huang, J.; Zhong, K.; Wang, Z.; et al. Efficacy of B7-H3-redirected BiTE and CAR-T immunotherapies against extranodal nasal natural killer/T cell lymphoma. Transl. Oncol. 2020, 13, 100770. [Google Scholar] [CrossRef] [PubMed]
- Iizuka, A.; Nonomura, C.; Ashizawa, T.; Kondou, R.; Ohshima, K.; Sugino, T.; Mitsuya, K.; Hayashi, N.; Nakasu, Y.; Maruyama, K.; et al. A T-cell-engaging B7-H4/CD3-bispecific Fab-scFv antibody targets human breast cancer. Clin. Cancer Res. 2019, 25, 2925–2934. [Google Scholar] [CrossRef]
- Zhao, H.; Ma, J.; Lei, T.; Ma, W.; Zhang, M. The bispecific anti-CD3 × anti-CD155 antibody mediates T cell immunotherapy for human prostate cancer. Investig. New Drugs 2019, 37, 810–817. [Google Scholar] [CrossRef]
- Ma, W.; Ma, J.; Lei, T.; Zhao, M.; Zhang, M. Targeting immunotherapy for bladder cancer by using anti-CD3 × CD155 bispecific antibody. J. Cancer. 2019, 10, 5153–5161. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.Y.; Fan, M.H.; Miao, C.H.; Liao, Y.J.; Yuan, R.H.; Liu, C.L. Engineering chimeric antigen receptor T cells against immune checkpoint inhibitors PD-1/PD-L1 for treating pancreatic cancer. Mol. Ther. Oncolytics 2020, 17, 571–585. [Google Scholar] [CrossRef] [PubMed]
- Huang, B.; Luo, L.; Wang, J.; He, B.; Feng, R.; Xian, N.; Zhang, Q.; Chen, L.; Huang, G. B7-H3 specific T cells with chimeric antigen receptor and decoy PD-1 receptors eradicate established solid human tumors in mouse models. Oncoimmunology 2019, 9, 1684127. [Google Scholar] [CrossRef]
- Majzner, R.G.; Theruvath, J.L.; Nellan, A.; Heitzeneder, S.; Cui, Y.; Mount, C.W.; Rietberg, S.P.; Linde, M.H.; Xu, P.; Rota, C.; et al. CAR T cells targeting B7-H3, a pan-cancer antigen, demonstrate potent preclinical activity against pediatric solid tumors and brain tumors. Clin. Cancer Res. 2019, 25, 2560–2574. [Google Scholar] [CrossRef]
- Yang, S.; Cao, B.; Zhou, G.; Zhu, L.; Wang, L.; Zhang, L.; Kwok, H.F.; Zhang, Z.; Zhao, Q. Targeting B7-H3 immune checkpoint with chimeric antigen receptor-engineered natural killer cells exhibits potent cytotoxicity against non-small cell lung cancer. Front. Pharmacol. 2020, 11, 1089. [Google Scholar] [CrossRef]
- Smith, J.B.; Lanitis, E.; Dangaj, D.; Buza, E.; Poussin, M.; Stashwick, C.; Scholler, N.; Powell, D.J., Jr. Tumor regression and delayed onset toxicity following B7-H4 CAR T cell therapy. Mol. Ther. 2016, 24, 1987–1999. [Google Scholar] [CrossRef]
- Golubovskaya, V.; Berahovich, R.; Zhou, H.; Xu, S.; Harto, H.; Li, L.; Chao, C.C.; Mao, M.M.; Wu, L. CD47-CAR-T cells effectively kill target cancer cells and block pancreatic tumor growth. Cancers 2017, 9, 139. [Google Scholar] [CrossRef]
- Jin, L.; Ge, H.; Long, Y.; Yang, C.; Chang, Y.E.; Mu, L.; Sayour, E.J.; De Leon, G.; Wang, Q.J.; Yang, J.C.; et al. CD70, a novel target of CAR T-cell therapy for gliomas. Neuro Oncol. 2018, 20, 55–65. [Google Scholar] [CrossRef]
- Park, Y.P.; Jin, L.; Bennett, K.B.; Wang, D.; Fredenburg, K.M.; Tseng, J.E.; Chang, L.J.; Huang, J.; Chan, E.K.L. CD70 as a target for chimeric antigen receptor T cells in head and neck squamous cell carcinoma. Oral Oncol. 2018, 78, 145–150. [Google Scholar] [CrossRef] [PubMed]
- Shaffer, D.R.; Savoldo, B.; Yi, Z.; Chow, K.K.; Kakarla, S.; Spencer, D.M.; Dotti, G.; Wu, M.F.; Liu, H.; Kenney, S.; et al. T cells redirected against CD70 for the immunotherapy of CD70-positive malignancies. Blood 2011, 117, 4304–4314. [Google Scholar] [CrossRef]
- Puro, R.J.; Bouchlaka, M.N.; Hiebsch, R.R.; Capoccia, B.J.; Donio, M.J.; Manning, P.T.; Frazier, W.A.; Karr, R.W.; Pereira, D.S. Development of AO-176, a next-generation humanized anti-CD47 antibody with novel anticancer properties and negligible red blood cell binding. Mol. Cancer Ther. 2020, 19, 835–846. [Google Scholar] [CrossRef]
- Pettersen, R.D.; Hestdal, K.; Olafsen, M.K.; Lie, S.O.; Lindberg, F.P. CD47 signals T cell death. J. Immunol. 1999, 162, 7031–7040. [Google Scholar]
- Leclair, P.; Liu, C.C.; Monajemi, M.; Reid, G.S.; Sly, L.M.; Lim, C.J. CD47-ligation induced cell death in T-acute lymphoblastic leukemia. Cell Death Dis. 2018, 9, 544. [Google Scholar] [CrossRef]
- Cioffi, M.; Trabulo, S.; Hidalgo, M.; Costello, E.; Greenhalf, W.; Erkan, M.; Kleeff, J.; Sainz, B., Jr.; Heeschen, C. Inhibition of CD47 effectively targets pancreatic cancer stem cells via dual mechanisms. Clin. Cancer Res. 2015, 21, 2325–2337. [Google Scholar] [CrossRef] [PubMed]
- Wu, X.; Li, F.; Li, Y.; Yu, Y.; Liang, C.; Zhang, B.; Zhao, C.; Lu, A.; Zhang, G. A PD-L1 aptamer selected by loss-gain cell-SELEX conjugated with paclitaxel for treating triple-negative breast cancer. Med. Sci. Monit. 2020, 26, e925583. [Google Scholar] [CrossRef]
- Gromeier, M.; Alexander, L.; Wimmer, E. Internal ribosomal entry site substitution eliminates neurovirulence in intergeneric poliovirus recombinants. Proc. Natl Acad. Sci. USA 1996, 93, 2370–2375. [Google Scholar] [CrossRef] [PubMed]
- Brown, M.C.; Gromeier, M. Cytotoxic and immunogenic mechanisms of recombinant oncolytic poliovirus. Curr. Opin. Virol. 2015, 13, 81–85. [Google Scholar] [CrossRef]
- Desjardins, A.; Gromeier, M.; Herndon, J.E., 2nd; Beaubier, N.; Bolognesi, D.P.; Friedman, A.H.; Friedman, H.S.; McSherry, F.; Muscat, A.M.; Nair, S.; et al. Recurrent glioblastoma treated with recombinant poliovirus. N. Engl. J. Med. 2018, 379, 150–161. [Google Scholar] [CrossRef] [PubMed]
- Gerdes, C.A.; Nicolini, V.G.; Herter, S.; van Puijenbroek, E.; Lang, S.; Roemmele, M.; Moessner, E.; Freytag, O.; Friess, T.; Ries, C.H.; et al. GA201 (RG7160): A novel, humanized, glycoengineered anti-EGFR antibody with enhanced ADCC and superior in vivo efficacy compared with cetuximab. Clin. Cancer Res. 2013, 19, 1126–1138. [Google Scholar] [CrossRef] [PubMed]
- Ni, X.; Jorgensen, J.L.; Goswami, M.; Challagundla, P.; Decker, W.K.; Kim, Y.H.; Duvic, M.A. Reduction of regulatory T cells by Mogamulizumab, a defucosylated anti-CC chemokine receptor 4 antibody, in patients with aggressive/refractory mycosis fungoides and Sézary syndrome. Clin. Cancer Res. 2015, 21, 274–285. [Google Scholar] [CrossRef] [PubMed]
- Stavenhagen, J.B.; Gorlatov, S.; Tuaillon, N.; Rankin, C.T.; Li, H.; Burke, S.; Huang, L.; Vijh, S.; Johnson, S.; Bonvini, E.; et al. Fc optimization of therapeutic antibodies enhances their ability to kill tumor cells in vitro and controls tumor expansion in vivo via low-affinity activating Fcgamma receptors. Cancer Res. 2007, 67, 8882–8890. [Google Scholar] [CrossRef]
- Dan, N.; Setua, S.; Kashyap, V.K.; Khan, S.; Jaggi, M.; Yallapu, M.M.; Chauhan, S.C. Antibody-drug conjugates for cancer therapy: Chemistry to clinical implications. Pharmaceuticals 2018, 11, 32. [Google Scholar] [CrossRef]
- Diamantis, N.; Banerji, U. Antibody-drug conjugates—An emerging class of cancer treatment. Br. J. Cancer 2016, 114, 362–367. [Google Scholar] [CrossRef]
- Labrijn, A.F.; Janmaat, M.L.; Reichert, J.M.; Parren, P.W.H.I. Bispecific antibodies: A mechanistic review of the pipeline. Nat. Rev. Drug Discov. 2019, 18, 585–608. [Google Scholar] [CrossRef]
- Larson, R.C.; Maus, M.V. Recent advances and discoveries in the mechanism and function of CAR T cells. Nat. Rev. Cancer 2021, 22. [Google Scholar] [CrossRef]
- Wang, H.; Kaur, G.; Sankin, A.I.; Chen, F.; Guan, F.; Zang, X. Immune checkpoint blockade and CAR-T cell therapy in hematologic malignancies. J. Hematol. Oncol. 2019, 12, 59. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.; Rossi, J. Aptamers as targeted therapeutics: Current potential and challenges. Nat. Rev. Drug Discov. 2017, 16, 181–202. [Google Scholar] [CrossRef] [PubMed]
- Marcucci, F.; Corti, A. Improving drug penetration to curb tumor drug resistance. Drug Discov. Today 2012, 17, 1139–1147. [Google Scholar] [CrossRef]
- Marcucci, F.; Corti, A. How to improve exposure of tumor cells to drugs—Promoter drugs increase tumor uptake and penetration of effector drugs. Adv. Drug Deliv. Rev. 2012, 64, 53–68. [Google Scholar] [CrossRef]
- Marcucci, F.; Bellone, M.; Rumio, C.; Corti, A. Approaches to improve tumor accumulation and interactions between monoclonal antibodies and immune cells. mAbs 2013, 5, 34–46. [Google Scholar] [CrossRef] [PubMed]
- Perdigoto, A.L.; Kluger, H.; Herold, K.C. Adverse events induced by immune checkpoint inhibitors. Curr. Opin. Immunol. 2021, 69, 29–38. [Google Scholar] [CrossRef]
- Macek Jilkova, Z.; Aspord, C.; Kurma, K.; Granon, A.; Sengel, C.; Sturm, N.; Marche, P.N.; Decaens, T. Immunologic features of patients with advanced hepatocellular carcinoma before and during sorafenib or anti-programmed death-1/programmed death-L1 treatment. Clin. Transl. Gastroenterol. 2019, 10, e00058. [Google Scholar] [CrossRef]
- Finn, R.S.; Qin, S.; Ikeda, M.; Galle, P.R.; Ducreux, M.; Kim, T.Y.; Kudo, M.; Breder, V.; Merle, P.; Kaseb, A.O.; et al. Atezolizumab plus bevacizumab in unresectable hepatocellular carcinoma. N. Engl. J. Med. 2020, 382, 1894–1905. [Google Scholar] [CrossRef] [PubMed]
- Rini, B.I.; Powles, T.; Atkins, M.B.; Escudier, B.; McDermott, D.F.; Suarez, C.; Bracarda, S.; Stadler, W.M.; Donskov, F.; Lee, J.L.; et al. Atezolizumab plus bevacizumab versus sunitinib in patients with previously untreated metastatic renal cell carcinoma (IMmotion151): A multicentre, open-label, phase 3, randomised controlled trial. Lancet 2019, 393, 2404–2415. [Google Scholar] [CrossRef]
- Lan, Y.; Zhang, D.; Xu, C.; Hance, K.W.; Marelli, B.; Qi, J.; Yu, H.; Qin, G.; Sircar, A.; Hernández, V.M.; et al. Enhanced preclinical antitumor activity of M7824, a bifunctional fusion protein simultaneously targeting PD-L1 and TGF-β. Sci. Transl. Med. 2018, 10, eaan5488. [Google Scholar] [CrossRef] [PubMed]
- Mariathasan, S.; Turley, S.J.; Nickles, D.; Castiglioni, A.; Yuen, K.; Wang, Y.; Kadel, E.E., III; Koeppen, H.; Astarita, J.L.; Cubas, R.; et al. TGFβ attenuates tumour response to PD-L1 blockade by contributing to exclusion of T cells. Nature 2018, 554, 544–548. [Google Scholar] [CrossRef] [PubMed]
| ICPML | Tumor Type | Immune Cells | Other Cells of the TME | References |
|---|---|---|---|---|
| PD-L1 | On cells of many hematologic and solid tumor types | Inducible expression on T cells, macrophages, DCs in response to inflammatory cytokines | On tumor endothelial cells, CAFs | [22,80,81,82,87,90] |
| B7-H3 | On cells of many hematologic and solid tumor types | Inducible expression on T cells, NK cells, DCs and macrophages | On tumor endothelial cells, CAFs | [3,130,131,132,133] |
| B7-H4 | On cells of many tumor types | Inducible expression on monocytes, macrophages, and myeloid DCs. Constitutive expression on TAMs, tumor Tregs | On tumor endothelial cells, CAFs | [3,85,134] |
| CD70 | On cells of many hematologic and solid tumor types, very frequent on RCC | On small subset of antigen-stimulated B and T cells, and mature DCs | On tumor endothelial cells, CAFs | [27,29,65,66,135,136,137] |
| CD155 | On cells of many types of solid tumors | Low-level expression on immune cells, becomes up-regulated in response to inflammatory stimuli (LPS, cytokines). Expressed on tumor-infiltrating myeloid cells (macrophages, DCs). | On human vascular endothelial cells | [73,91,138] |
| TIM-1 | O cells of many solid tumor types, most notably RCC and ovarian clear cell carcinoma | On T cells (in particular Th2), Breg and DCs in mice. | [139] | |
| CD47 | On cells of many tumor types | Inducible (thrombospondin-1) expression on different types of immune cells (CD8+ T cells, macrophages, DCs, NK cells) | Ubiquitously expressed | [2,140] |
| Galectin-9 | On cells of several human tumors including melanoma, multiple myeloma, mesothelioma | Constitutive expression on Treg, on Th cells upon activation, macrophages. | [64,141] | |
| CD40 | On cells of hematologic tumors | On APCs (DCs), B cells, monocytes | [142,143] |
| Compound | Type of Construct | Preclinical or Clinical (ClinicalTrials.gov Identifier, Phase, Comments) Development | References |
|---|---|---|---|
| Antibodies with enhanced constitutive effector functions | |||
| Anti-B7-H3 (eroblituzumab/MGA271) | mAb with mutated Fc domain | Clinical: NCT01391143, phase I; NCT02381314, phase I, plus ipilimumab; NCT02982941, phase I in children; NCT02475213, phase I, plus pembrolizumab; NCT02923180, phase II, neoadjuvant in prostate cancer; NCT04129320, NCT04634825, phase II/III, plus anti-PD-1 mAb or bispecific anti-PD-1xLAG-3 mAb. | [130] |
| Anti-CD70 (cusatuzumab/ARGX-110) | Afucosylated mAb | Clinical: NCT03030612, NCT04264806, NCT04241549, NCT04150887, NCT04023526, phase I/II, plus AZA or venetoclax in MDS, AML, CML; NCT02759250, phase I in NPC; NCT01813539, phase I/II neoplasms. | [135,144] |
| ADCs | |||
| Anti-CD70 (SGN-75) | Humanized anti-CD70 mAb linked to tubulin inhibitor auristatin | Clinical: NCT01015911, phase I in NHL, RCC, modest single-agent activity; NCT01677390, phase Ib, plus everolimus in RCC. | [145,146] |
| Anti-CD70 (SGN-CD70A) | Anti-CD70 mAb linked to PBD dimer | Clinical: NCT02216890, phase I in NHL, RCC, showed modest single-agent activity and high frequency/severity of thrombocytopenia. | [147,148] |
| Anti-CD70 (BMS-936561/ MDX-1203) | Human anti-CD70 mAb linked to duocarmycin derivative | Clinical: NCT00944905, phase I in NHL, RCC. | [149,150] |
| Anti-B7-H3 (MGC018) | Humanized anti-B7-H3 mAb linked to duocarmycin | Clinical: NCT03729596, phase I/II, plus anti-PD-1 in several solid tumors. | [151] |
| Anti-B7-H3 (m276) | Human anti-B7-H3 mAb linked to PBD dimer | Preclinical: It depleted both B7-H3+ tumor cells as well as B7-H3+ tumor endothelial cells leading to the eradication of established tumors. Moderate expression of B7-H3 was detected also on normal tissues. | [131] |
| Anti-B7-H3 | Anti-B7-H3 mAb linked to chlorin e6 for photodynamic therapy | Preclinical | [152] |
| Anti-B7-H4 (h1D11 TDC) | PBD linked to engineered cysteines of an anti-B7-H4 mAb via a protease-labile linker. | Preclinical: This ADC induced durable regression in different mouse models of TNBC. | [153] |
| anti-TIM-1 (CDX-014) | Human anti-TIM-1 linked to MMAE | Clinical: NCT02837991, phase I in RCC, development now discontinued. | [139] |
| Anti-PD-L1 (STM-108) | Mab anti-glycosylated PD-L1 linked to MMAE | Preclinical: Induced bystander killing on PD-L1- tumor cells. | [154] |
| Anti-PD-L1 | scFv-PD-L1 linked to the maytansinoid DM1 | Preclinical: Specific binding to PD-L1+ tumor cells and antiproliferative activity in vitro. | [155] |
| Anti-PD-L1 (atezolizumab) | Atezolizumab linked to doxorubicin | Preclinical: Induced cell killing, disruption of tumor spheroids and induced apoptosis in a breast cancer cell line. | [156] |
| Bispecific Antibodies | |||
| Anti-B7-H3xanti-CD3 | Preclinical: MEK inhibitor trametinib augmented expression of B7-H3. Combined therapy (trametinib + bispecific mAb) increased T cell infiltration and significantly suppressed tumor growth. | [30] | |
| Anti-B7-H3xanti-CD3 | Preclinical: On hematological tumor cells, redirected T cells exhibited significant cytotoxicity, secreted more cytokines and granzyme B and expressed higher levels of activating marker CD69 compared to non-redirected T cells. | [157] | |
| Anti-B7-H3xanti-CD3 | Preclinical: On cells of ENKTCL redirected T cells killed tumor cells in vitro and suppressed the growth of NKTCL tumors in mouse models. | [158] | |
| Anti-B7-H4xanti-CD3 (mAb clone #25xOKT3) | Two constructs: one Fab (anti-B7-H4) xscFv (anti-CD3),one scFvxscRv | Preclinical: In a humanized mouse model of breast cancer the bispecific Ab had strong antitumor activity and promoted the infiltration of CD8+ CTLs into the tumor without any adverse effects over the long term. | [159] |
| Anti-CD155xanti-CD3 | Preclinical | [160,161] | |
| CAR T or NK Cells | |||
| Anti-PD-L1 CAR T cells | T cells expressing theextracellular domain of human PD-1 or the scFv of an anti-PD-L1 | Preclinical: Induced regression of established PDAC cancer by >80% in both xenograft and orthotopic models. | [162] |
| Anti-B7-H3 CAR T cells (376.96 mAb) | Preclinical: Control of the growth of PDAC, ovarian cancer and neuroblastoma in xenograft mouse models and in a syngeneic tumor model without toxicity. | [132] | |
| Anti-B7-H3 CAR T cells | scFv from an anti-B7-H3 mAb + PD-1 decoy receptor. | Preclinical: Potent antitumor activity in B7-H3+/B7-H1+ tumors in vivo. | [163] |
| Anti-B7-H3 CAR T cells | scFv derived from the anti-B7-H3 mAb enoblituzumab | Preclinical: Regression of established solid tumors in xenograft models. Efficacy dependent upon high surface antigen density on tumor tissues. | [164] |
| Anti-B7-H3 CAR NK cells (CAR-NK-92MI) | Preclinical: Inhibition of tumor growth in mouse xenografts of NSCLC and prolonged survival of mice. | [165] | |
| Anti-B7-H4 CAR T cells | Preclinical: Inhibition of growth of B7-H4+ human ovarian tumor xenografts, but lethal toxicity was observed 6-8 weeks after therapy due to expression of B7-H4 in ductal and mucosal epithelial cells in normal tissues. | [166] | |
| Anti-CD47 CAR NK cells | scFv from an anti-CD47 mAb | Preclinical: Inhibition of pancreatic xenograft tumor growth after intratumoral injection in mice. | [167] |
| Anti-CD70 CAR T cells | Anti-human and -mouse CD70 CAR T cells | Preclinical: Both human and mouse anti-CAR T cells induced regression of established GBM in xenograft and syngeneic models. | [168] |
| Anti-CD70 CAR T cells | Truncated CD27, the CD70 receptor, is the CD70 binder | Preclinical: Elimination of CD70-positive HNSCC cells. | [169] |
| Anti-CD70 CAR T cells | Truncated CD27, the CD70 receptor, was used as CD70 binder | Preclinical: Adoptively transferred anti-CD70 CAR T cells induced regression of established murine xenografts. | [170] |
| Anti-CD70 CAR T cells | Clinical: NCT04662294, phase I in AML, MM, NHL | N.A. | |
| Anti-CD70 CAR T cells | Clinical: NCT03125577, NCT04429438, phase I/II in hematological B-cell malignancies. | N.A. | |
| Anti-CD70 CAR T cells (CTX130) | Anti-CD70 allogeneic T cells. | Clinical: NCT04502446, phase I in relapsed or refractory T or B cell malignancies. | N.A. |
| Antibodies Inducing Cell Death Independently of Effector Functions | |||
| Anti-CD40 mAb (dacetuzumab) | Preclinical: Dacetuzumab + anti-CD20 mAb rituximab gave synergistic apoptotic effects on NHL cells through distinct, but complementary apoptotic signal transduction pathways. | [142] | |
| Anti-CD47 (mAb AO-176) | Clinical: NCT03834948, phase I/II, alone or with paclitaxel in solid tumors; NCT04445701, phase I/II alone or with bortezomib in MM.Preclinical: Induced tumor cell phagocytosis and cytotoxicity on human tumor cells but not normal cells. | [171] | |
| Anti-CD47 mAb Ad22 | Preclinical: Ad22 induced apoptosis of Jurkat cells and preactivated PBMC | [172] | |
| Anti-CD47 (mAb CC2C6) | Preclinical: Soluble CC2C6 induced apoptosis of T-ALL cells, restored phagocytosis, and synergized with low-dose chemotherapeutics to induce apoptosis. | [173] | |
| Anti-CD47 (mAb B6H12.2) | Preclinical: Enhanced phagocytosis of a set of human pancreatic CSCs and directly induced apoptosis in the absence of macrophages. | [174] | |
| Anti-galectin 9 (mAb P4D2) | Preclinical: Induced MM cell apoptosis, inhibited tumor growth and reduced tumor infiltration of M2 macrophages. | [64] | |
| Aptamers | |||
| Anti-PD-L1 aptamer-drug conjugate | Aptamer-paclitaxel conjugate | Preclinical: The anti-PD-L1 aptamer inhibited PD-1/PD-L1 interaction and restored T-cell function. The conjugate inhibited proliferation of PD-L1-overexpressing TNBC cells. | [175] |
| Oncolytic Virus | |||
| Oncolytic virus binding to CD155 | Neuroattenuated poliovirus strain PVSRIPO that replicates in and kills only tumor cells | Clinical: NCT03564782, NCT03712358, NCT02986178, NCT03043391, NCT01491893, phase I/II in invasive breast cancer, melanoma, GBM; NCT04479241, NCT04577807, NCT04690699, phase II plus anti-PD-1or -PD-L1 in GBM, melanoma or other solid tumors. | [176,177,178] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Marcucci, F.; Rumio, C. Depleting Tumor Cells Expressing Immune Checkpoint Ligands—A New Approach to Combat Cancer. Cells 2021, 10, 872. https://doi.org/10.3390/cells10040872
Marcucci F, Rumio C. Depleting Tumor Cells Expressing Immune Checkpoint Ligands—A New Approach to Combat Cancer. Cells. 2021; 10(4):872. https://doi.org/10.3390/cells10040872
Chicago/Turabian StyleMarcucci, Fabrizio, and Cristiano Rumio. 2021. "Depleting Tumor Cells Expressing Immune Checkpoint Ligands—A New Approach to Combat Cancer" Cells 10, no. 4: 872. https://doi.org/10.3390/cells10040872
APA StyleMarcucci, F., & Rumio, C. (2021). Depleting Tumor Cells Expressing Immune Checkpoint Ligands—A New Approach to Combat Cancer. Cells, 10(4), 872. https://doi.org/10.3390/cells10040872







