Nerve Growth Factor Neutralization Promotes Oligodendrogenesis by Increasing miR-219a-5p Levels
Abstract
1. Introduction
2. Materials and Methods
2.1. Anti-NGF AD11 Mouse Model
2.2. RNA Isolation, Amplification and Labeling
2.3. Microarray Analysis
2.4. Microarray Data Analysis
2.5. RNA Isolation and Reverse Transcription-PCR
2.6. Brain Dissection and Tissue Processing
2.7. Neural Stem Cell Cultures
2.8. Oligodendrocytes Primary Cultures
2.9. NGF and Anti-NGF αD11 Antibody Treatment
2.10. Immunofluorescence on Brain Section and OLs
2.11. Design, Production and Transduction of Lentiviral Vectors
3. Results
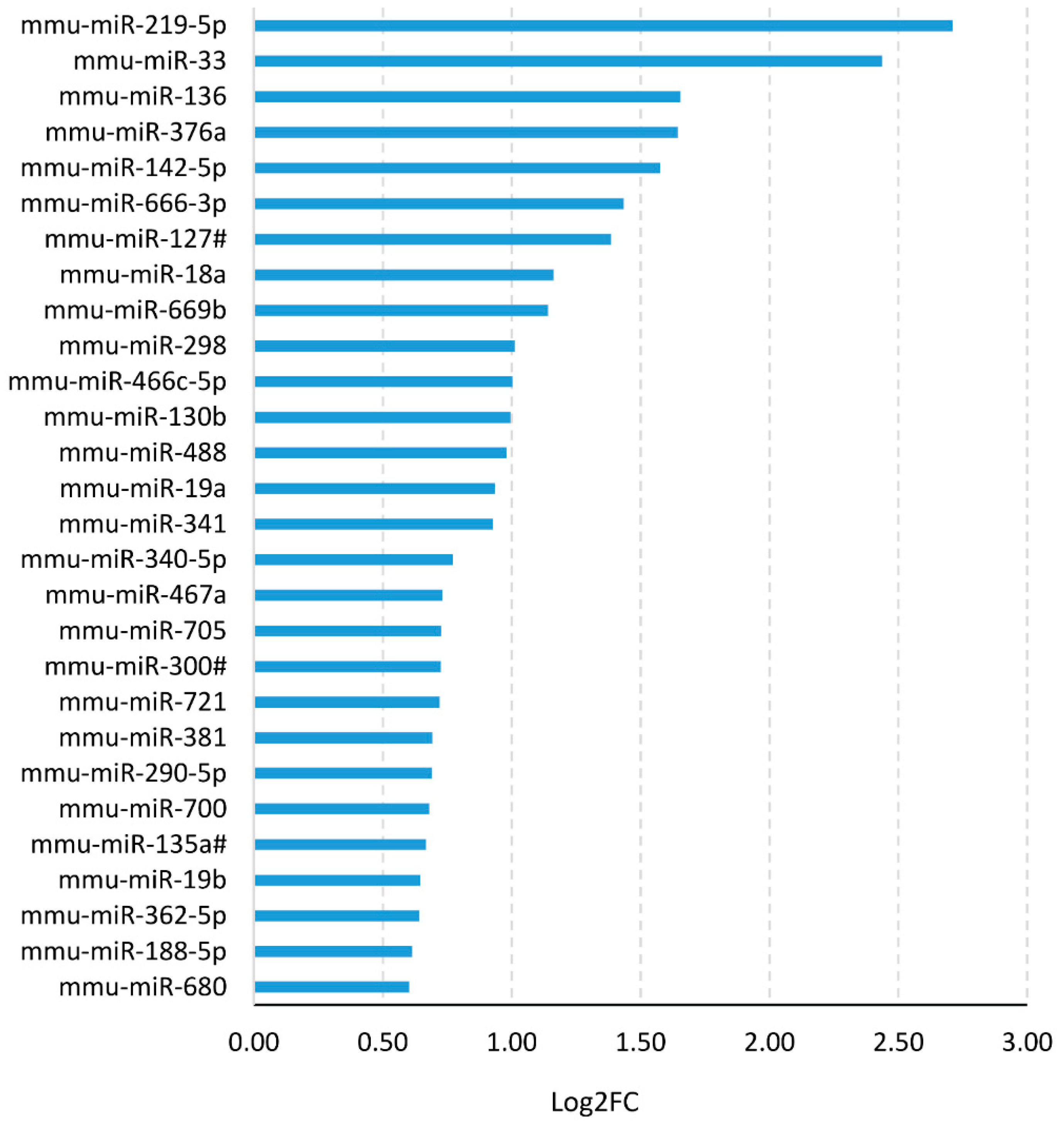
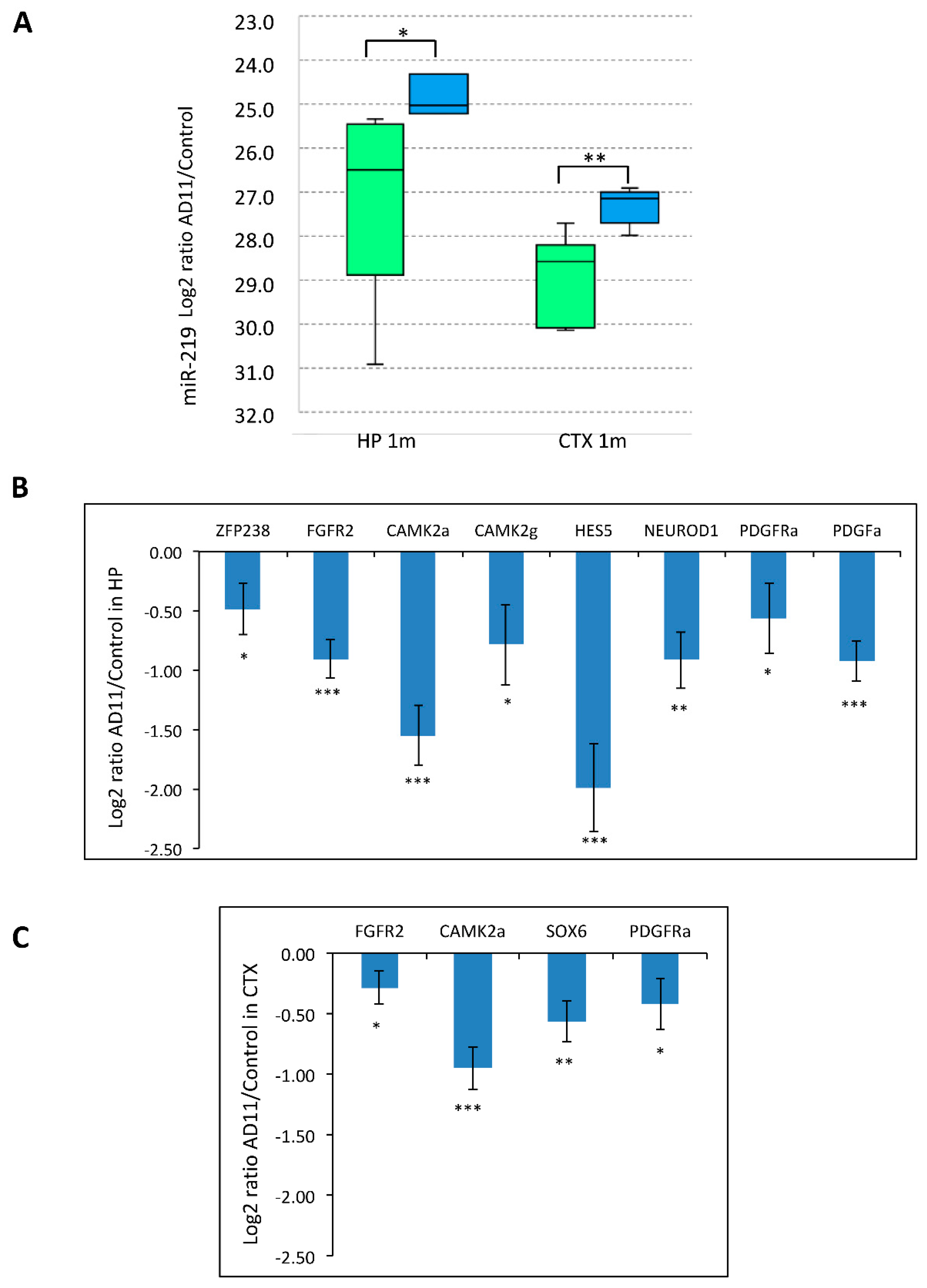
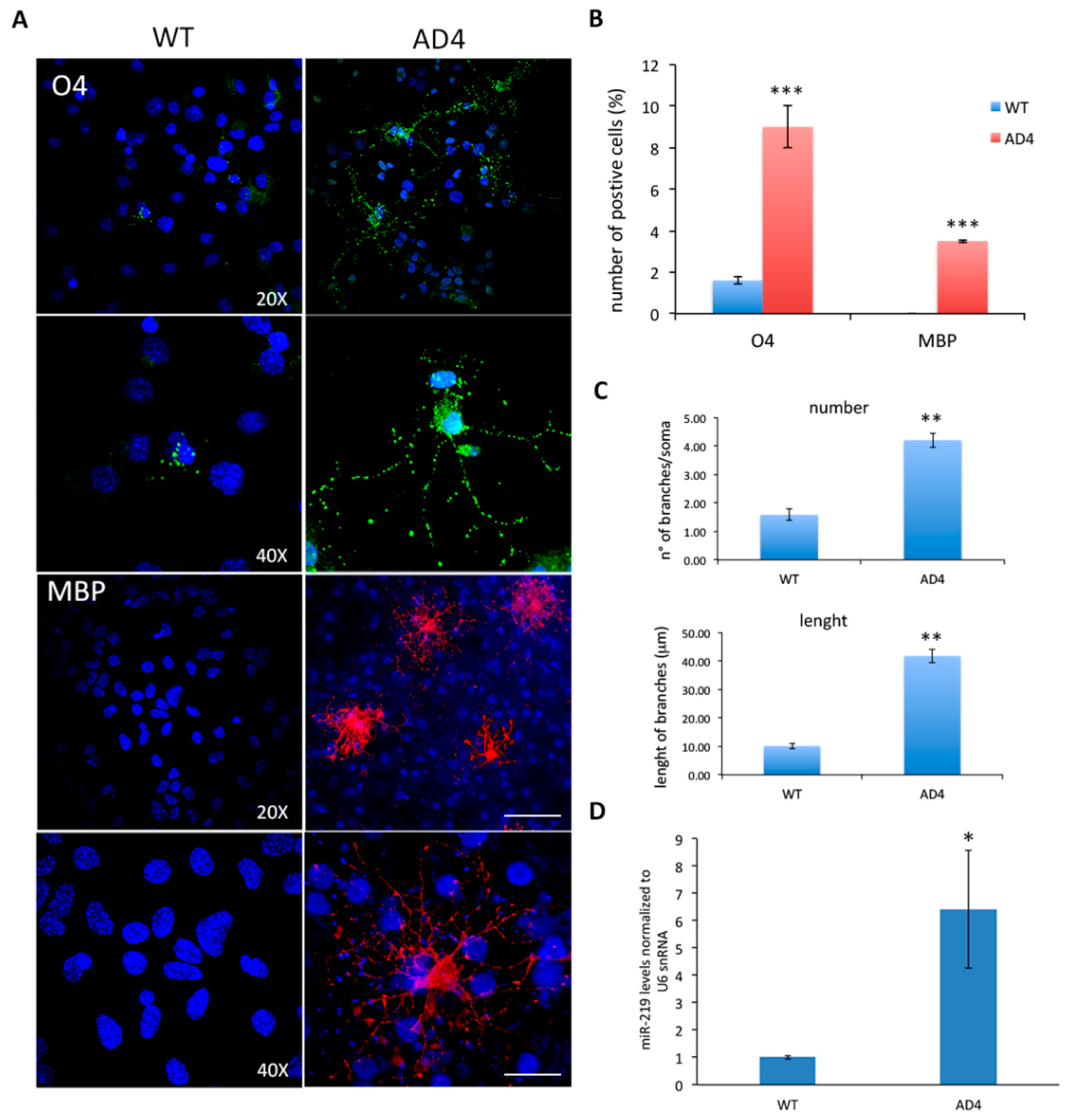
3.1. NGF Neutralization Promotes Oligodendrogenesis and miR-219 Up-Regulation in Vivo
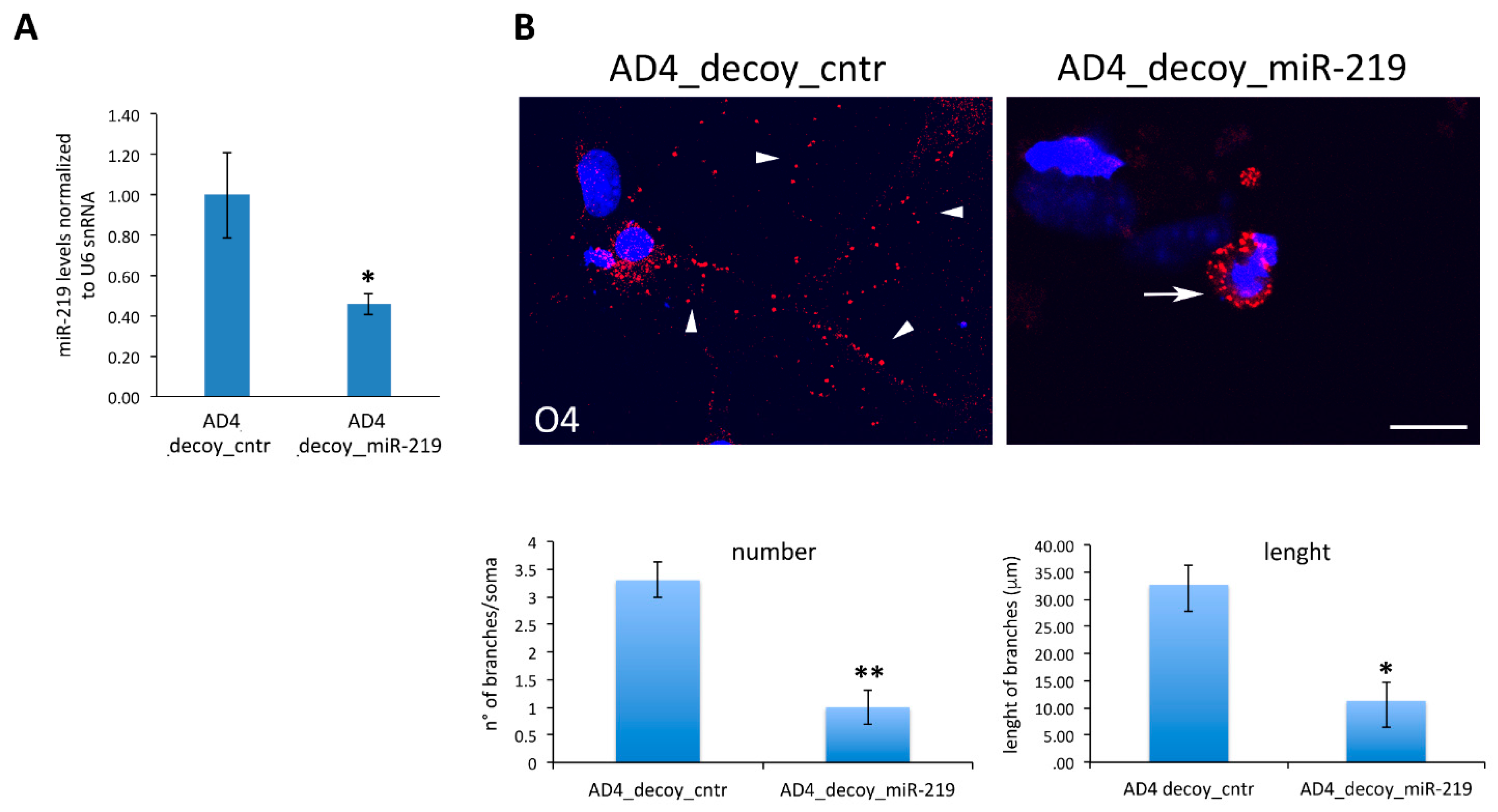
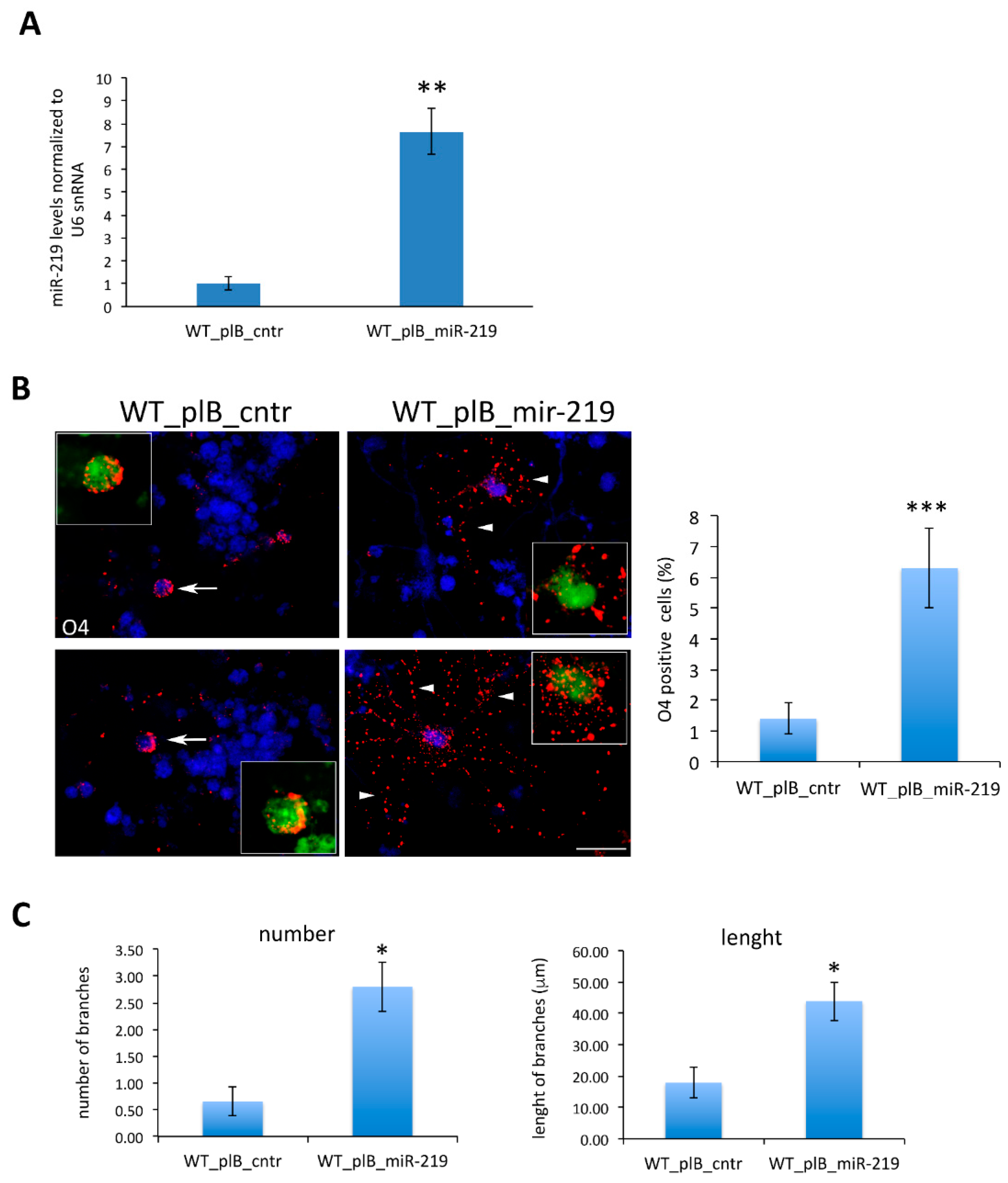
3.2. Oligodendrogenesis in AD4 Neurosphere Is Dependent on miR-219
3.3. Modulation of miR-219 Expression Is Dependent on NGF
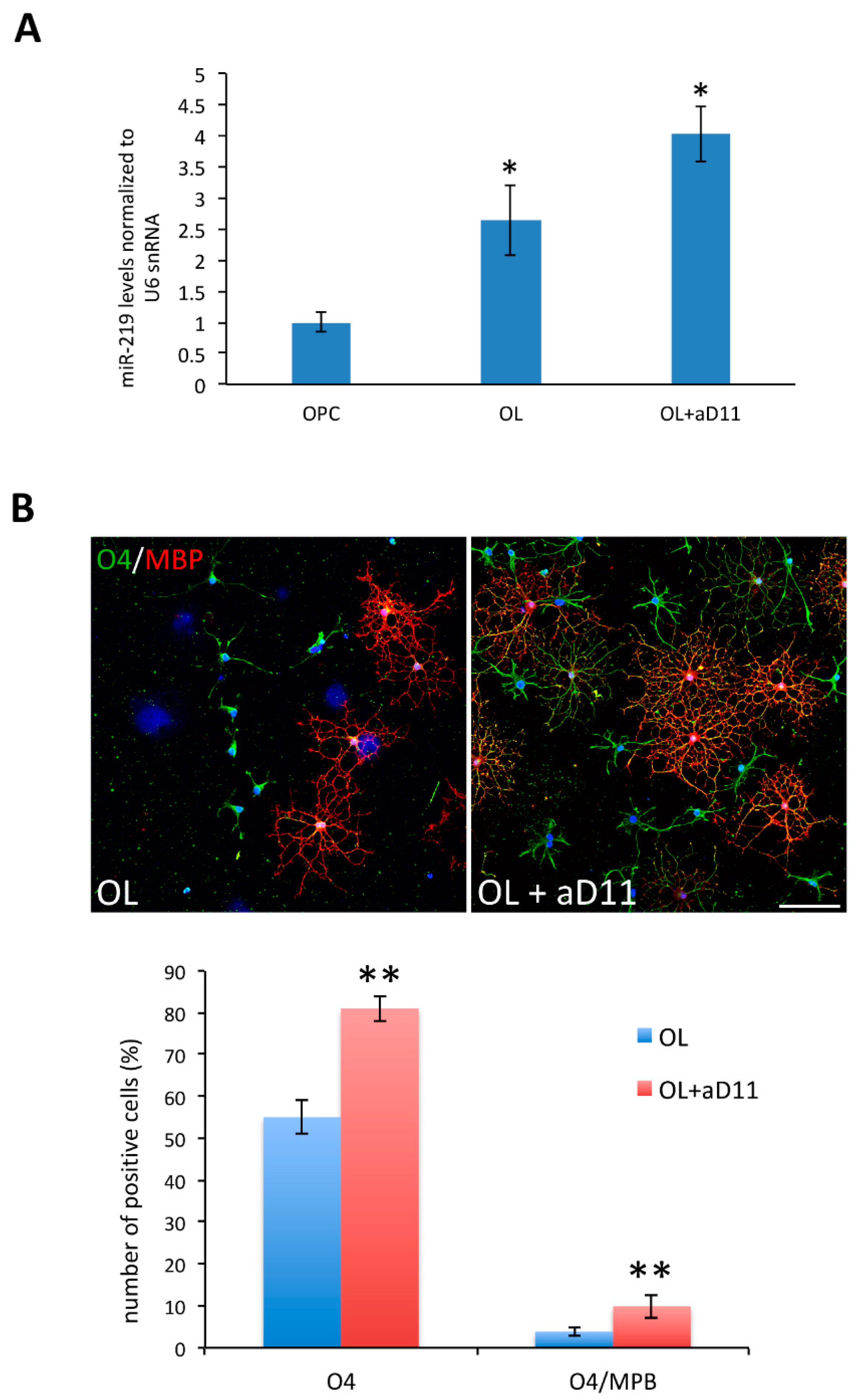
3.4. Anti-NGF αD11 Neutralization of NGF Increases OL Differentiation of Rat Primary OPCs
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Van Tilborg, E.; De Theije, C.G.M.; Van Hal, M.; Wagenaar, N.; De Vries, L.S.; Benders, M.J.; Rowitch, D.H.; Nijboer, C.H. Origin and dynamics of oligodendrocytes in the developing brain: Implications for perinatal white matter injury. Glia 2017, 66, 221–238. [Google Scholar] [CrossRef]
- Gallo, V.; Deneen, B. Glial Development: The Crossroads of Regeneration and Repair in the CNS. Neuron 2014, 83, 283–308. [Google Scholar] [CrossRef]
- Duncan, I.D.; Brower, A.; Kondo, Y.; Curlee, J.F.; Schultz, R.D. Extensive remyelination of the CNS leads to functional recovery. Proc. Natl. Acad. Sci. USA 2009, 106, 6832–6836. [Google Scholar] [CrossRef] [PubMed]
- Mei, F.; Lehmann-Horn, K.; Shen, Y.A.; Rankin, K.A.; Stebbins, K.J.; Lorrain, D.S.; Pekarek, K.; Sagan, S.A.; Xiao, L.; Teuscher, C.; et al. Accelerated remyelination during inflammatory demyelination prevents axonal loss and improves functional recovery. eLife 2016, 5, e18246. [Google Scholar] [CrossRef] [PubMed]
- Zawadzka, M.; Rivers, L.E.; Fancy, S.P.; Zhao, C.; Tripathi, R.; Jamen, F.; Young, K.; Goncharevich, A.; Pohl, H.; Rizzi, M.; et al. CNS-Resident Glial Progenitor/Stem Cells Produce Schwann Cells as well as Oligodendrocytes during Repair of CNS Demyelination. Cell Stem Cell 2010, 6, 578–590. [Google Scholar] [CrossRef]
- Goldschmidt, T.; Antel, J.; Konig, F.B.; Bruck, W.; Kuhlmann, T. Remyelination capacity of the MS brain decreases with disease chronicity. Neurology 2009, 72, 1914–1921. [Google Scholar] [CrossRef] [PubMed]
- Neumann, B.; Segel, M.; Chalut, K.J.; Franklin, R.J. Remyelination and ageing: Reversing the ravages of time. Mult. Scler. J. 2019, 25, 1835–1841. [Google Scholar] [CrossRef]
- Sim, F.J.; Zhao, C.; Penderis, J.; Franklin, R.J. The age-related decrease in CNS remye-lination efficiency is attributable to an impairment of both oligodendrocyte progenitor re-cruitment and differentiation. J. Neurosci. 2002, 22, 2451–2459. [Google Scholar] [CrossRef] [PubMed]
- Franklin, R.J.; Frisén, J.; Lyons, D.A. Revisiting remyelination: Towards a consensus on the regeneration of CNS myelin. Semin. Cell Dev. Biol. 2020. [Google Scholar] [CrossRef] [PubMed]
- Skaper, S.D. The Neurotrophin Family of Neurotrophic Factors: An Overview. Methods Mol. Biol. 2012, 846, 1–12. [Google Scholar] [CrossRef]
- Chao, M.V. Neurotrophins and their receptors: A convergence point for many signalling pathways. Nat. Rev. Neurosci. 2003, 4, 299–309. [Google Scholar] [CrossRef]
- Cellerino, A.; Kohler, K. Brain-derived neurotrophic factor/neurotrophin-4 receptor TrkB is localized on ganglion cells and dopaminergic amacrine cells in the vertebrate retina. J. Comp. Neurol 1997, 386, 149–160. [Google Scholar] [CrossRef]
- Wong, A.W.; Xiao, J.; Kemper, D.; Kilpatrick, T.J.; Murray, S.S. Oligodendroglial Expression of TrkB Independently Regulates Myelination and Progenitor Cell Proliferation. J. Neurosci. 2013, 33, 4947–4957. [Google Scholar] [CrossRef] [PubMed]
- Chan, J.R.; Cosgaya, J.M.; Wu, Y.J.; Shooter, E.M. Neurotrophins are key mediators of the myelination program in the peripheral nervous system. Proc. Natl. Acad. Sci. USA 2001, 98, 14661–14668. [Google Scholar] [CrossRef]
- Cosgaya, J.M.; Chan, J.R.; Shooter, E.M. The Neurotrophin Receptor p75NTR as a Positive Modulator of Myelination. Science 2002, 298, 1245–1248. [Google Scholar] [CrossRef] [PubMed]
- Woolley, A.G.; Tait, K.J.; Hurren, B.J.; Fisher, L.; Sheard, P.W.; Duxson, M.J. Developmental loss of NT-3in vivo results in reduced levels of myelin-specific proteins, a reduced extent of myelination and increased apoptosis of Schwann cells. Glia 2007, 56, 306–317. [Google Scholar] [CrossRef] [PubMed]
- Capsoni, S.; Covaceuszach, S.; Marinelli, S.; Ceci, M.; Bernardo, A.; Minghetti, L.; Ugolini, G.; Pavone, F.; Cattaneo, A. Taking Pain Out of NGF: A “Painless” NGF Mutant, Linked to Hereditary Sensory Autonomic Neuropathy Type V, with Full Neurotrophic Activity. PLoS ONE 2011, 6, e17321. [Google Scholar] [CrossRef]
- Chan, J.R.; Watkins, T.A.; Cosgaya, J.M.; Zhang, C.; Chen, L.; Reichardt, L.F.; Shooter, E.M.; Barres, B.A. NGF Controls Axonal Receptivity to Myelination by Schwann Cells or Oligodendrocytes. Neuron 2004, 43, 183–191. [Google Scholar] [CrossRef] [PubMed]
- Lee, X.; Yang, Z.; Shao, Z.; Rosenberg, S.S.; Levesque, M.; Pepinsky, R.B.; Qiu, M.; Miller, R.H.; Chan, J.R.; Mi, S. NGF Regulates the Expression of Axonal LINGO-1 to Inhibit Oligodendrocyte Differentiation and Myelination. J. Neurosci. 2007, 27, 220–225. [Google Scholar] [CrossRef]
- Malerba, F.; Paoletti, F.; Ercole, B.B.; Materazzi, S.; Nassini, R.; Coppi, E.; Patacchini, R.; Capsoni, S.; Lamba, D.; Cattaneo, A. Functional Characterization of Human ProNGF and NGF Mutants: Identification of NGF P61SR100E as a “Painless” Lead Investigational Candidate for Therapeutic Applications. PLoS ONE 2015, 10, e0136425. [Google Scholar] [CrossRef]
- D’Onofrio, M.; Arisi, I.; Brandi, R.; Di Mambro, A.; Felsani, A.; Capsoni, S.; Cattaneo, A. Early inflammation and immune response mRNAs in the brain of AD11 anti-NGF mice. Neurobiol. Aging 2011, 32, 1007–1022. [Google Scholar] [CrossRef] [PubMed]
- Duffy, C.P.; McCoy, C.E. The Role of MicroRNAs in Repair Processes in Multiple Sclerosis. Cells 2020, 9, 1711. [Google Scholar] [CrossRef] [PubMed]
- Kocerha, J.; Kauppinen, S.; Wahlestedt, C. microRNAs in CNS Disorders. Neuromol. Med. 2009, 11, 162–172. [Google Scholar] [CrossRef] [PubMed]
- Li, J.-S.; Yao, Z.-X. MicroRNAs: Novel Regulators of Oligodendrocyte Differentiation and Potential Therapeutic Targets in Demyelination-Related Diseases. Mol. Neurobiol. 2012, 45, 200–212. [Google Scholar] [CrossRef]
- Zhao, X.; He, X.; Han, X.; Yu, Y.; Ye, F.; Chen, Y.; Hoang, T.; Xu, X.; Mi, Q.-S.; Xin, M.; et al. MicroRNA-Mediated Control of Oligodendrocyte Differentiation. Neuron 2010, 65, 612–626. [Google Scholar] [CrossRef] [PubMed]
- Ruberti, F.; Capsoni, S.; Comparini, A.; Di Daniel, E.; Franzot, J.; Gonfloni, S.; Rossi, G.; Berardi, N.; Cattaneo, A. Phenotypic Knockout of Nerve Growth Factor in Adult Transgenic Mice Reveals Severe Deficits in Basal Forebrain Cholinergic Neurons, Cell Death in the Spleen, and Skeletal Muscle Dystrophy. J. Neurosci. 2000, 20, 2589–2601. [Google Scholar] [CrossRef] [PubMed]
- Capsoniab, S.; Giannottaa, S.; Cattaneo, A. β-Amyloid Plaques in a Model for Sporadic Alzheimer’s Disease Based on Transgenic Anti-Nerve Growth Factor Antibodies. Mol. Cell. Neurosci. 2002, 21, 15–28. [Google Scholar] [CrossRef]
- Capsoni, S.; Ugolini, G.; Comparini, A.; Ruberti, F.; Berardi, N.; Cattaneo, A. Alzheimer-like neurodegeneration in aged antinerve growth factor transgenic mice. Proc. Natl. Acad. Sci. USA 2000, 97, 6826–6831. [Google Scholar] [CrossRef]
- Origlia, N.; Capsoni, S.; Domenici, L.; Cattaneo, A. Time window in cholinomimetic ability to rescue long-term potentiation in neurodegenerating anti-nerve growth factor mice. J. Alzheimer’s Dis. 2006, 9, 59–68. [Google Scholar] [CrossRef]
- Pesavento, E.; Capsoni, S.; Domenici, L.; Cattaneo, A. Acute cholinergic rescue of synaptic plasticity in the neurodegenerating cortex of anti-nerve-growth-factor mice. Eur. J. Neurosci. 2002, 15, 1030–1036. [Google Scholar] [CrossRef]
- Bruinsma, I.B.; Van Dijk, M.; Bridel, C.; Van De Lisdonk, T.; Haverkort, S.Q.; Runia, T.F.; Steinman, L.; Hintzen, R.Q.; Killestein, J.; Verbeek, M.M.; et al. Regulator of oligodendrocyte maturation, miR-219, a potential biomarker for MS. J. Neuroinflamm. 2017, 14, 1–7. [Google Scholar] [CrossRef]
- Dugas, J.C.; Cuellar, T.L.; Scholze, A.; Ason, B.; Ibrahim, A.; Emery, B.; Zamanian, J.L.; Foo, L.C.; McManus, M.T.; Barres, B.A. Dicer1 and miR-219 Are Required for Normal Oligodendrocyte Differentiation and Myelination. Neuron 2010, 65, 597–611. [Google Scholar] [CrossRef]
- Lau, P.; Verrier, J.D.; Nielsen, J.A.; Johnson, K.R.; Notterpek, L.; Hudson, L.D. Identification of Dynamically Regulated MicroRNA and mRNA Networks in Developing Oligodendrocytes. J. Neurosci. 2008, 28, 11720–11730. [Google Scholar] [CrossRef]
- Wang, H.; Moyano, A.L.; Ma, Z.; Deng, Y.; Lin, Y.; Zhao, C.; Zhang, L.; Jiang, M.; He, X.; Ma, Z.; et al. miR-219 Cooperates with miR-338 in Myelination and Promotes Myelin Repair in the CNS. Dev. Cell 2017, 40, 566–582. [Google Scholar] [CrossRef]
- Cattaneo, A.; Rapposelli, B.; Calissano, P. Three Distinct Types of Monoclonal Antibodies After Long-Term Immunization of Rats with Mouse Nerve Growth Factor. J. Neurochem. 1988, 50, 1003–1010. [Google Scholar] [CrossRef] [PubMed]
- Paoletti, F.; Covaceuszach, S.; Konarev, P.V.; Gonfloni, S.; Malerba, F.; Schwarz, E.; Svergun, D.I.; Cattaneo, A.; Lamba, D. Intrinsic structural disorder of mouse proNGF. Proteins: Struct. Funct. Bioinform. 2008, 75, 990–1009. [Google Scholar] [CrossRef] [PubMed]
- Agarwal, V.; Bell, G.W.; Nam, J.-W.; Bartel, D.P. Predicting effective microRNA target sites in mammalian mRNAs. eLife 2015, 4, e05005. [Google Scholar] [CrossRef] [PubMed]
- Da Huang, W.; Sherman, B.T.; Lempicki, R.A. Systematic and integrative analysis of large gene lists using DAVID bioinformatics resources. Nat. Protoc. 2009, 4, 44–57. [Google Scholar] [CrossRef]
- Huang, D.W.; Sherman, B.T.; Zheng, X.; Yang, J.; Imamichi, T.; Stephens, R.; Lempicki, R.A. Extracting Biological Meaning from Large Gene Lists with DAVID. Curr. Protoc. Bioinform. 2009, 27, 13.11.1–13.11.13. [Google Scholar] [CrossRef]
- Edgar, R.; Domrachev, M.; Lash, A.E. Gene Expression Omnibus: NCBI gene expression and hybridization array data repository. Nucleic Acids Res. 2002, 30, 207–210. [Google Scholar] [CrossRef]
- Corvaglia, V.; Cilli, D.; Scopa, C.; Brandi, R.; Arisi, I.; Malerba, F.; La Regina, F.; Scardigli, R.; Cattaneo, A. ProNGF Is a Cell-Type-Specific Mitogen for Adult Hippocampal and for Induced Neural Stem Cells. Stem Cells 2019, 37, 1223–1237. [Google Scholar] [CrossRef]
- Amadio, S.; Parisi, C.; Montilli, C.; Carrubba, A.S.; Apolloni, S.; Volonté, C. P2Y12Receptor on the Verge of a Neuroinflammatory Breakdown. Mediat. Inflamm. 2014, 2014, 1–15. [Google Scholar] [CrossRef]
- Scardigli, R.; Capelli, P.; Vignone, D.; Brandi, R.; Ceci, M.; La Regina, F.; Piras, E.; Cintoli, S.; Berardi, N.; Capsoni, S.; et al. Neutralization of Nerve Growth Factor Impairs Proliferation and Differentiation of Adult Neural Progenitors in the Subventricular Zone. Stem Cells 2014, 32, 2516–2528. [Google Scholar] [CrossRef] [PubMed]
- Marangon, D.; Boda, E.; Parolisi, R.; Negri, C.; Giorgi, C.; Montarolo, F.; Perga, S.; Bertolotto, A.; Buffo, A.; Abbracchio, M.P.; et al. In vivo silencing of miR-125a-3p promotes myelin repair in models of white matter demyelination. Glia 2020, 68, 2001–2014. [Google Scholar] [CrossRef] [PubMed]
- Besnard, F.; Perraud, F.; Sensenbrenner, M.; Labourdette, G. Platelet-derived growth factor is a mitogen for glial but not for neuronal rat brain cells in vitro. Neurosci. Lett. 1987, 73, 287–292. [Google Scholar] [CrossRef]
- Stolt, C.C.; Schlierf, A.; Lommes, P.; Hillgärtner, S.; Werner, T.; Kosian, T.; Sock, E.; Kessaris, N.; Richardson, W.D.; Lefebvre, V.; et al. SoxD Proteins Influence Multiple Stages of Oligodendrocyte Development and Modulate SoxE Protein Function. Dev. Cell 2006, 11, 697–709. [Google Scholar] [CrossRef]
- Diao, H.J.; Low, W.C.; Milbreta, U.; Lu, Q.R.; Chew, S.Y. Nanofiber-mediated microRNA delivery to enhance differentiation and maturation of oligodendroglial precursor cells. J. Control. Release 2015, 208, 85–92. [Google Scholar] [CrossRef]
- Fan, H.-B.; Chen, L.-X.; Qu, X.-B.; Ren, C.-L.; Wu, X.-X.; Dong, F.-X.; Zhang, B.-L.; Gao, D.-S.; Yao, R.-Q. Transplanted miR-219-overexpressing oligodendrocyte precursor cells promoted remyelination and improved functional recovery in a chronic demyelinated model. Sci. Rep. 2017, 7, srep41407. [Google Scholar] [CrossRef]
- Liu, S.; Ren, C.; Qu, X.; Wu, X.; Dong, F.; Chand, Y.K.; Fan, H.; Yao, R.; Geng, D. miR-219 attenuates demyelination in cuprizone-induced demyelinated mice by regulating monocarboxylate transporter 1. Eur. J. Neurosci. 2017, 45, 249–259. [Google Scholar] [CrossRef] [PubMed]
- Nazari, B.; Soleimani, M.; Ebrahimi-Barough, S.; Enderami, S.E.; Kazemi, M.; Negahdari, B.; Sadroddiny, E.; Ai, J. Overexpression of miR-219 promotes differentiation of human induced pluripotent stem cells into pre-oligodendrocyte. J. Chem. Neuroanat. 2018, 91, 8–16. [Google Scholar] [CrossRef]
- Osorio-Querejeta, I.; Carregal-Romero, S.; Ayerdi-Izquierdo, A.; Mäger, I.; Nash, L.A.; Wood, M.; Egimendia, A.; Betanzos, M.; Alberro, A.; Iparraguirre, L.; et al. MiR-219a-5p Enriched Extracellular Vesicles Induce OPC Differentiation and EAE Improvement More Efficiently Than Liposomes and Polymeric Nanoparticles. Pharmaceutics 2020, 12, 186. [Google Scholar] [CrossRef] [PubMed]
- Pusic, A.D.; Kraig, R.P. Youth and environmental enrichment generate serum exosomes containing miR-219 that promote CNS myelination. Glia 2014, 62, 284–299. [Google Scholar] [CrossRef] [PubMed]
- Mi, S.; Miller, R.H.; Lee, X.; Scott, M.L.; Shulag-Morskaya, S.; Shao, Z.; Chang, J.; Thill, G.; Levesque, M.; Zhang, M.; et al. LINGO-1 negatively regulates myelination by oligodendrocytes. Nat. Neurosci. 2005, 8, 745–751. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.M.; Michel, K.; Jokhi, V.; Nedivi, E.; Arlotta, P. Neuron class-specific responses govern adaptive myelin remodeling in the neocortex. Science 2020, 370, eabd2109. [Google Scholar]
- Xin, W.; Chan, J.R. Myelin plasticity: Sculpting circuits in learning and memory. Nat. Rev. Neurosci. 2020, 21, 682–694. [Google Scholar] [CrossRef]
- Mazziotti, R.; Baroncelli, L.; Ceglia, N.; Chelini, G.; Della Sala, G.; Magnan, C.; Napoli, D.; Putignano, E.; Silingardi, D.; Tola, J.; et al. Mir-132/212 is required for maturation of binocular matching of orientation preference and depth perception. Nat. Commun. 2017, 8, 15488. [Google Scholar] [CrossRef]
- Berardi, N.; Cellerino, A.; Domenici, L.; Fagiolini, M.; Pizzorusso, T.; Cattaneo, A.; Maffei, L. Monoclonal antibodies to nerve growth factor affect the postnatal development of the visual system. Proc. Natl. Acad. Sci. USA 1994, 91, 684–688. [Google Scholar] [CrossRef]
- Love, S. Demyelinating diseases. J. Clin. Pathol. 2006, 59, 1151–1159. [Google Scholar] [CrossRef]
- Minnone, G.; De Benedetti, F.; Bracci-Laudiero, L. NGF and Its Receptors in the Regulation of Inflammatory Response. Int. J. Mol. Sci. 2017, 18, 1028. [Google Scholar] [CrossRef]
- Laudiero, L.; Aloe, L.; Levi-Montalcini, R.; Buttinelli, C.; Schilter, D.; Gillessen, S.; Otten, U. Multiple sclerosis patients express increased levels of β-nerve growth factor in cerebrospinal fluid. Neurosci. Lett. 1992, 147, 9–12. [Google Scholar] [CrossRef]
- Monteleone, F.; Nicoletti, C.G.; Iezzi, E.; Buttari, F.; Furlan, R.; Finardi, A.; Marfia, G.A.; Centonze, D.; Mori, F.; Bassi, M.S. Nerve growth factor is elevated in the CSF of patients with multiple sclerosis and central neuropathic pain. J. Neuroimmunol. 2018, 314, 89–93. [Google Scholar] [CrossRef]
- Malerba, F.; Paoletti, F.; Cattaneo, A. NGF and proNGF Reciprocal Interference in Immunoassays: Open Questions, Criticalities, and Ways Forward. Front. Mol. Neurosci. 2016, 9, 63. [Google Scholar] [CrossRef] [PubMed]
- Marques, S.; Zeisel, A.; Codeluppi, S.; Van Bruggen, D.; Falcão, A.M.; Xiao, L.; Li, H.; Häring, M.; Hochgerner, H.; Romanov, R.A.; et al. Oligodendrocyte heterogeneity in the mouse juvenile and adult central nervous system. Science 2016, 352, 1326–1329. [Google Scholar] [CrossRef]
- Jäkel, S.; Agirre, E.; Falcão, A.M.; Van Bruggen, D.; Lee, K.W.; Knuesel, I.; Malhotra, D.; Ffrench-Constant, C.; Williams, A.; Castelo-Branco, G. Altered human oligodendrocyte heterogeneity in multiple sclerosis. Nat. Cell Biol. 2019, 566, 543–547. [Google Scholar] [CrossRef]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Brandi, R.; Fabiano, M.; Giorgi, C.; Arisi, I.; La Regina, F.; Malerba, F.; Turturro, S.; Storti, A.E.; Ricevuti, F.; Amadio, S.; et al. Nerve Growth Factor Neutralization Promotes Oligodendrogenesis by Increasing miR-219a-5p Levels. Cells 2021, 10, 405. https://doi.org/10.3390/cells10020405
Brandi R, Fabiano M, Giorgi C, Arisi I, La Regina F, Malerba F, Turturro S, Storti AE, Ricevuti F, Amadio S, et al. Nerve Growth Factor Neutralization Promotes Oligodendrogenesis by Increasing miR-219a-5p Levels. Cells. 2021; 10(2):405. https://doi.org/10.3390/cells10020405
Chicago/Turabian StyleBrandi, Rossella, Marietta Fabiano, Corinna Giorgi, Ivan Arisi, Federico La Regina, Francesca Malerba, Sabrina Turturro, Andrea Ennio Storti, Flavia Ricevuti, Susanna Amadio, and et al. 2021. "Nerve Growth Factor Neutralization Promotes Oligodendrogenesis by Increasing miR-219a-5p Levels" Cells 10, no. 2: 405. https://doi.org/10.3390/cells10020405
APA StyleBrandi, R., Fabiano, M., Giorgi, C., Arisi, I., La Regina, F., Malerba, F., Turturro, S., Storti, A. E., Ricevuti, F., Amadio, S., Volontè, C., Capsoni, S., Scardigli, R., D’Onofrio, M., & Cattaneo, A. (2021). Nerve Growth Factor Neutralization Promotes Oligodendrogenesis by Increasing miR-219a-5p Levels. Cells, 10(2), 405. https://doi.org/10.3390/cells10020405






