Prophylactic and Therapeutic Effects of Oral Immunotherapy on Birch Pollen-Induced Allergic Conjunctivitis in Mice with a Rice-Based Edible Vaccine Expressing a Hypoallergenic Birch Pollen Allergen
Abstract
:1. Introduction
2. Materials and Methods
2.1. Mice
2.2. Transgenic Rice
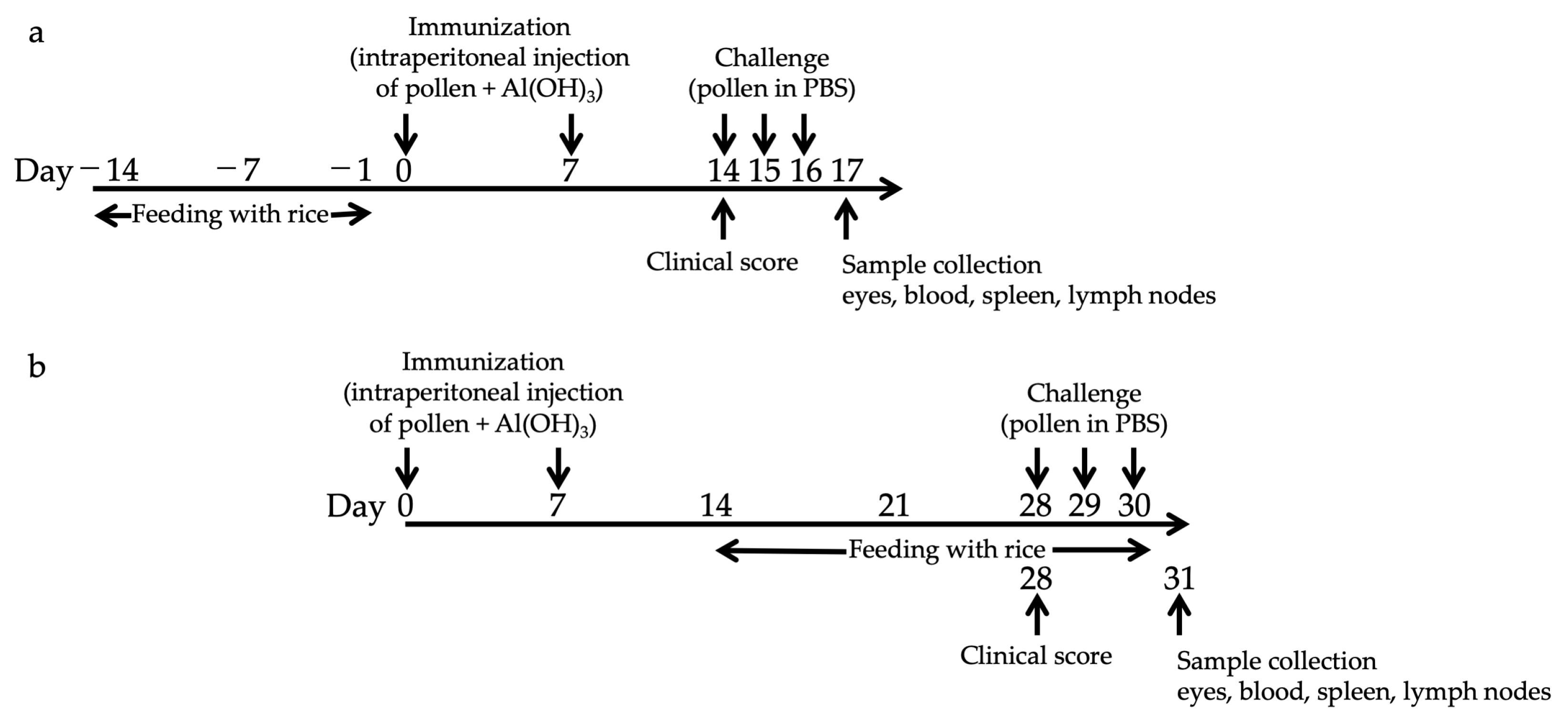
2.3. Feeding and Sensitization of Mice
2.4. Evaluation of Clinical Conjunctival Early Phase Reaction
2.5. Histological Evaluation of Conjunctiva
2.6. Measurement of Total and Bet v 1 Specific-IgE
2.7. Mesurement of Cytokine Release by Splenocytes
2.8. Flow Cytometric Analysis of CD4+CD25+Foxp3+ Expression by Lymphocytes from Lymph Nodes and the Spleen
2.9. Statistical Analysis
3. Results
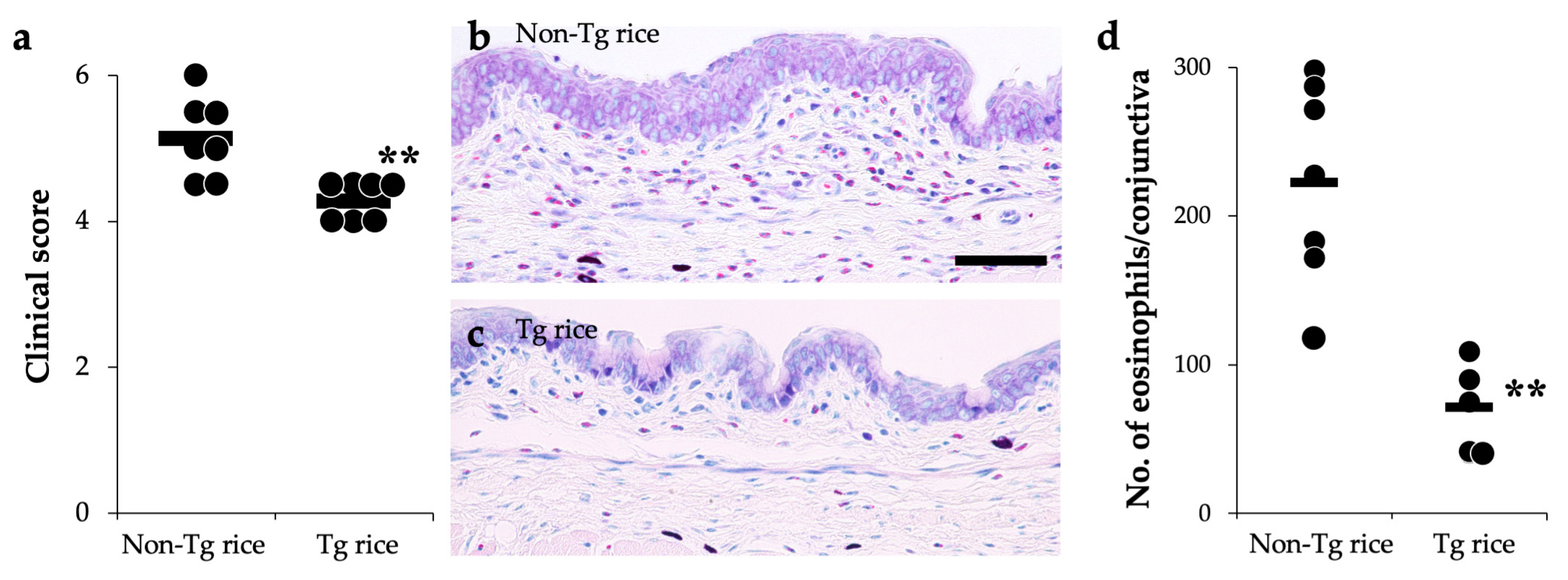
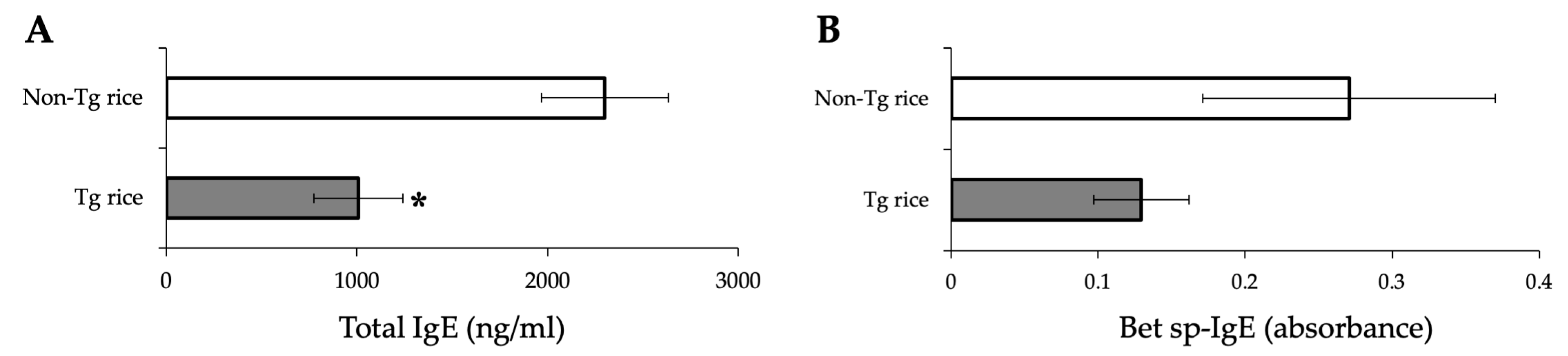
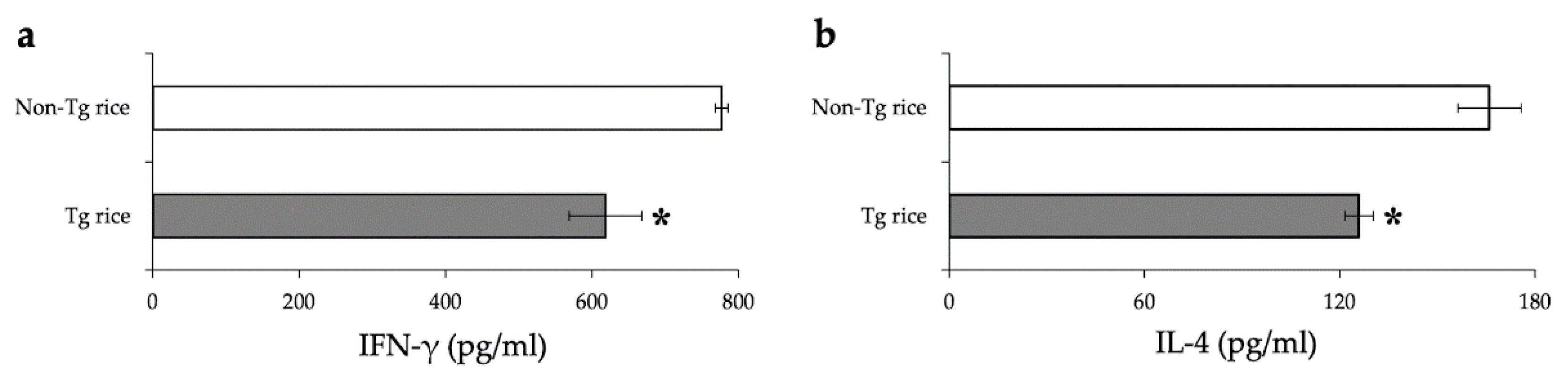
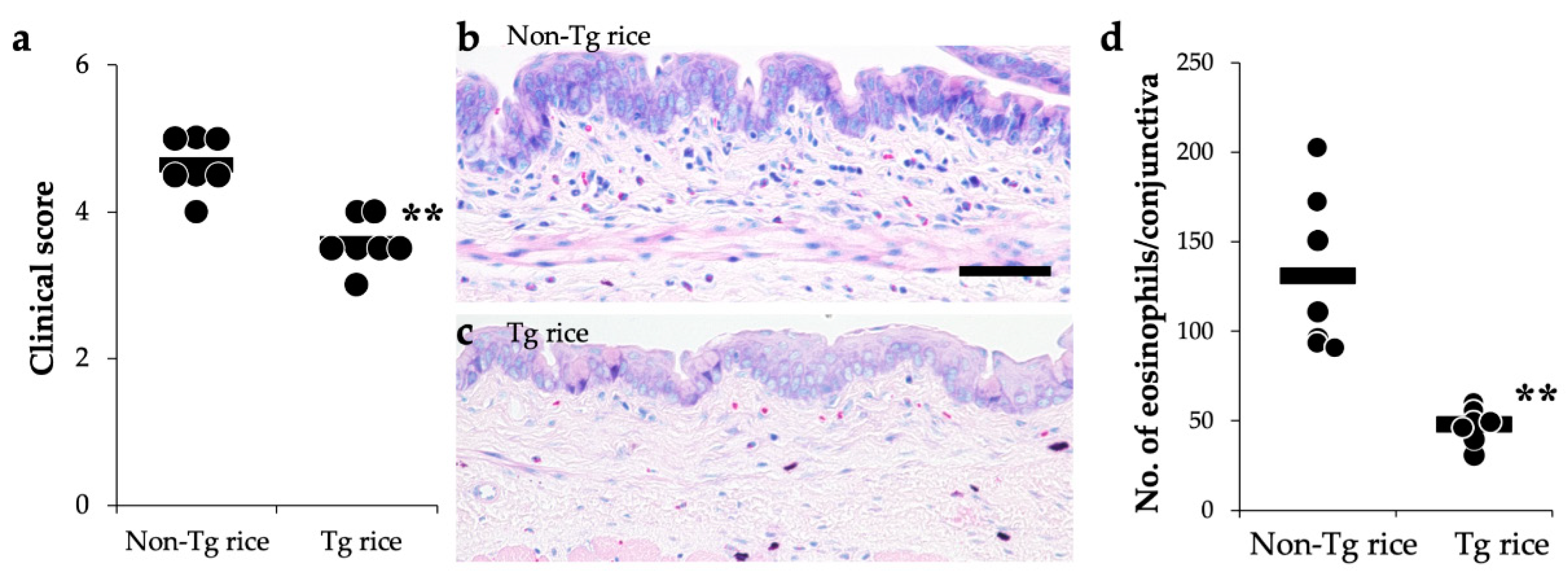
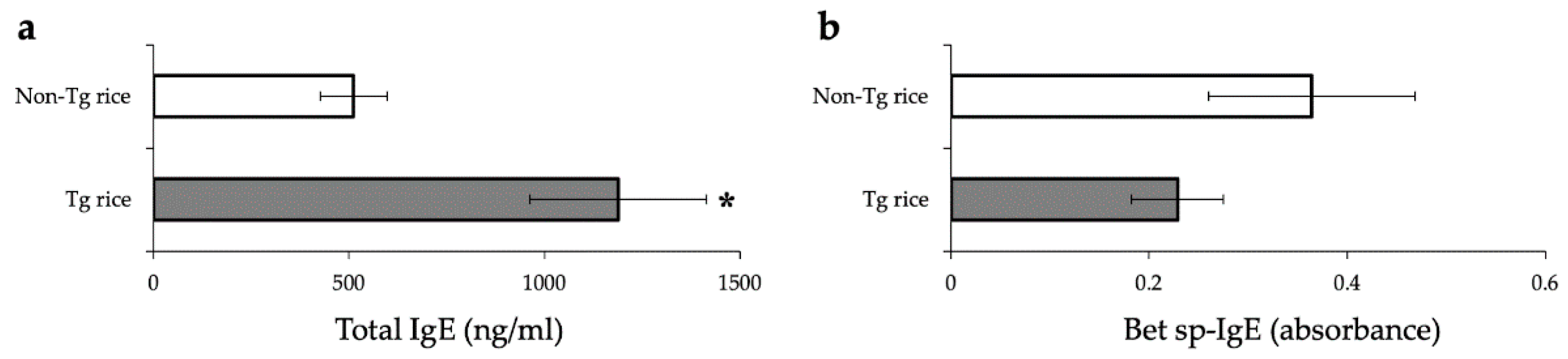
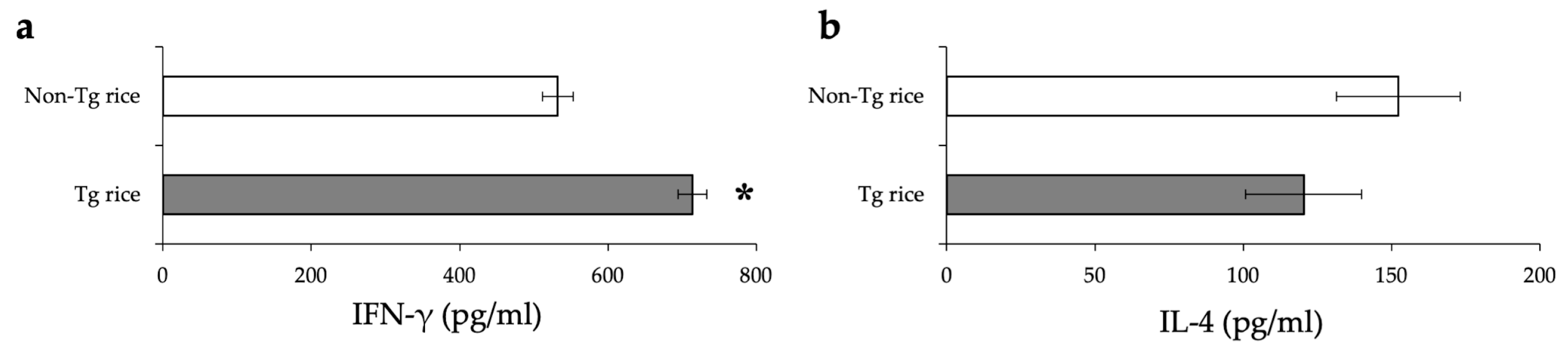
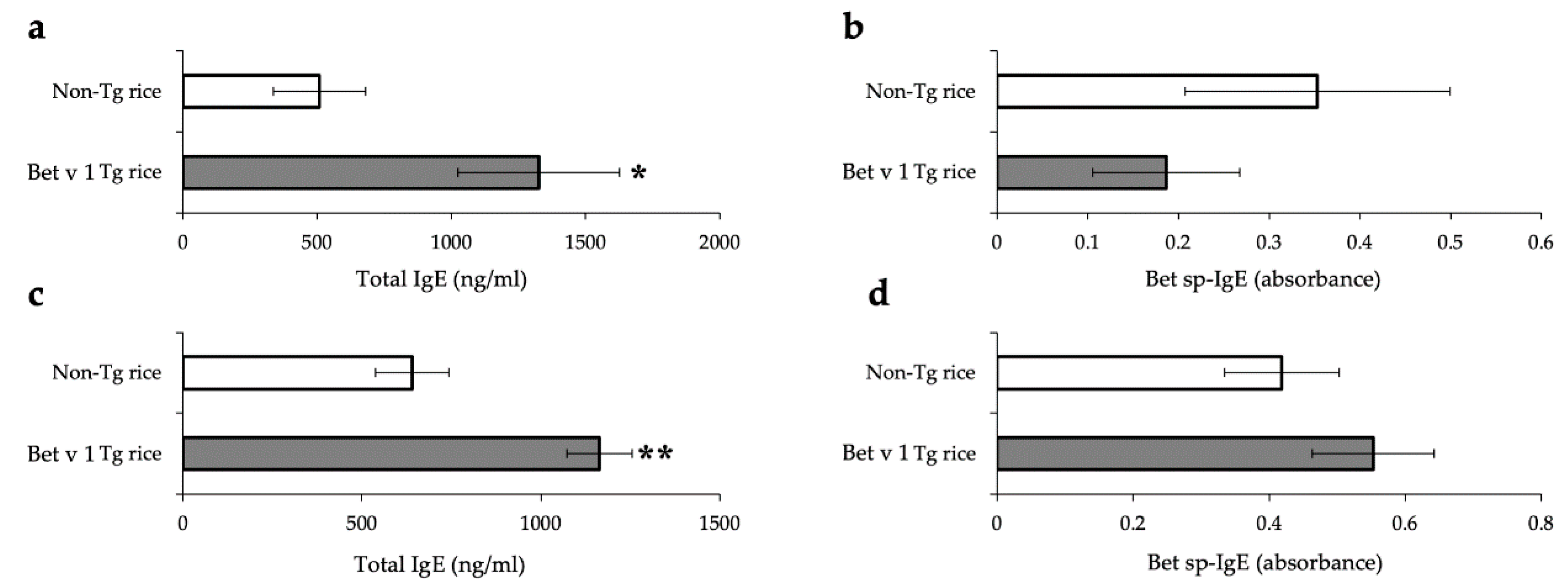
3.1. Prophylactic Administration with Tg Rice Suppresses the Development of EAC
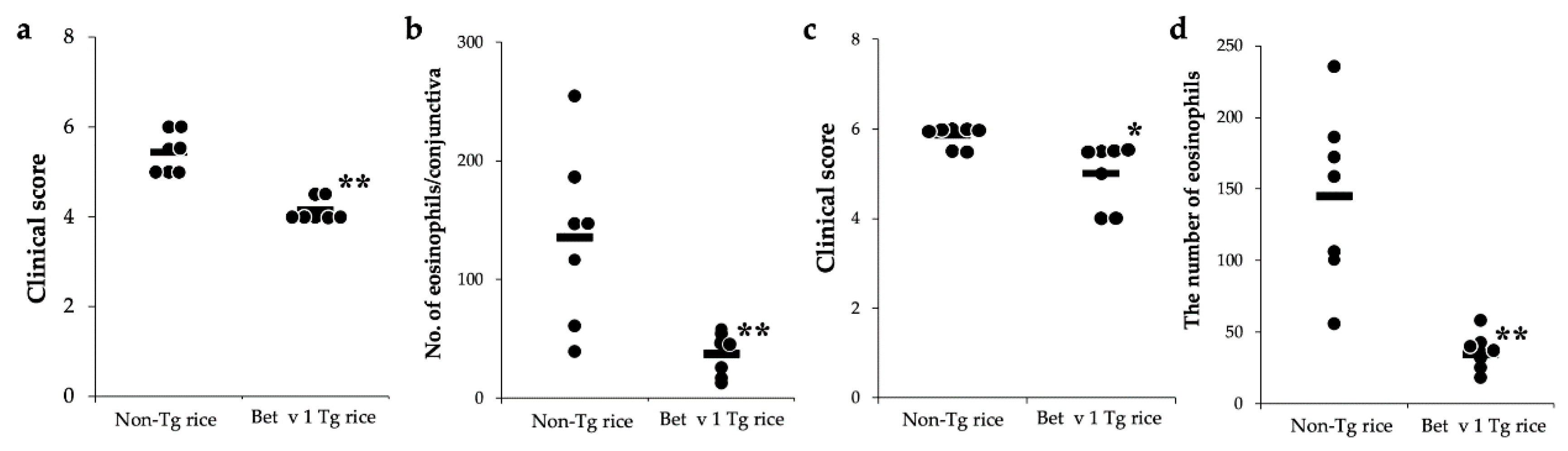
3.2. Therapeutic Administration with Tg Rice Suppresses the Established EAC
3.3. Effects of Prophylactic and Therapeutic Oral Administration of Native Bet v 1-Expressing Tg Rice on EAC
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Platts-Mills, T.A. The allergy epidemics: 1870–2010. J. Allergy Clin. Immunol. 2015, 136, 3–13. [Google Scholar] [CrossRef] [Green Version]
- Wang, X.-Y.; Ma, T.; Zhuang, Y.; Ning, H.-Y.; Shi, H.-Y.; Yu, R.-L.; Yan, D.; Huang, H.-D.; Bai, Y.-F.; Shan, G.-L.; et al. Prevalence of pollen-induced allergic rhinitis with high pollen exposure in grasslands of northern China. Allergy 2018, 73, 1232–1243. [Google Scholar] [CrossRef] [PubMed]
- Fukuda, K.; Ohbayashi, M.; Morohoshi, K.; Zhang, L.; Liu, F.-T.; Ono, S.J. Critical role of IgE-dependent mast cell activation in a murine model of allergic conjunctivitis. J. Allergy Clin. Immunol. 2009, 124, 827–833.e2. [Google Scholar] [CrossRef]
- Xie, Z.; Yin, J. Chinese Birch Pollen Allergy and Immunotherapy in Mice. Inflammation 2019, 42, 961–972. [Google Scholar] [CrossRef] [Green Version]
- Rojo, J.; Oteros, J.; Picornell, A.; Maya-Manzano, J.M.; Damialis, A.; Zink, K.; Werchan, M.; Werchan, B.; Smith, M.; Menzel, A.; et al. Effects of future climate change on birch abundance and their pollen load. Glob. Chang. Biol. 2021, 27, 5934–5949. [Google Scholar] [CrossRef]
- Erhard, S.M.; Bellach, J.; Yürek, S.; Tschirner, S.; Trendelenburg, V.; Grabenhenrich, L.B.; Fernandez-Rivas, M.; van Ree, R.; Keil, T.; Beyer, K. Primary and pollen-associated hazelnut allergy in school-aged children in Germany: A birth cohort study. Allergol. Int. Off. J. Jpn. Soc. Allergol. 2021, 70, 463–470. [Google Scholar] [CrossRef]
- Uotila, R.; Kukkonen, A.K.; Pelkonen, A.; Mäkelä, M. Cross-sensitization profiles of edible nuts in a birch-endemic area. Allergy 2016, 71, 514–521. [Google Scholar] [CrossRef]
- Bousquet, J.; Lockey, R.; Malling, H.J. Allergen immunotherapy: Therapeutic vaccines for allergic diseases. A WHO position paper. J. Allergy Clin. Immunol. 1998, 102 Pt 1, 558–562. [Google Scholar] [CrossRef]
- Noon, L. Prophylactic inoculation against hayfever. Lancet 1911, 4, 1572–1573. [Google Scholar] [CrossRef] [Green Version]
- Dhami, S.; Kakourou, A.; Asamoah, F.; Agache, I.; Lau, S.; Jutel, M.; Muraro, A.; Roberts, G.; Akdis, C.A.; Bonini, M.; et al. Allergen immunotherapy for allergic asthma: A systematic review and meta-analysis. Allergy 2017, 72, 1825–1848. [Google Scholar] [CrossRef]
- Roberts, G.; Pfaar, O.; Akdis, C.A.; Ansotegui, I.J.; Durham, S.R.; Van Wijk, R.G.; Halken, S.; Linnemann, D.L.; Pawankar, R.; Pitsios, C.; et al. EAACI Guidelines on Allergen Immunotherapy: Allergic rhinoconjunctivitis. Allergy 2017, 73, 765–798. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Canonica, G.W.; Cox, L.; Pawankar, R.; Baena-Cagnani, C.E.; Blaiss, M.; Bonini, S.; Bousquet, J.; Calderón, M.; Compalati, E.; Durham, S.R.; et al. Sublingual immunotherapy: World Allergy Organization position paper 2013 update. World Allergy Organ. J. 2014, 7, 1–6. [Google Scholar] [CrossRef] [Green Version]
- Dahl, R.; Kapp, A.; Colombo, G.; de Monchy, J.G.; Rak, S.; Emminger, W.; Riis, B.; Grønager, P.M.; Durham, S.R. Sublingual grass allergen tablet immunotherapy provides sustained clinical benefit with progressive immunologic changes over 2 years. J. Allergy Clin. Immunol. 2008, 121, 512–518.e2. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Senna, G.; Lombardi, C.; Canonica, G.W.; Passalacqua, G. How adherent to sublingual immunotherapy prescriptions are patients? The manufacturers’ viewpoint. J. Allergy Clin. Immunol. 2010, 126, 668–669. [Google Scholar] [CrossRef] [PubMed]
- O’BHourihane, J.; Beyer, K.; Abbas, A.; Fernández-Rivas, M.; Turner, P.J.; Blumchen, K.; Nilsson, C.; Ibáñez, M.D.; Deschildre, A.; Muraro, A.; et al. Efficacy and safety of oral immunotherapy with AR101 in European children with a peanut allergy (ARTEMIS): A multicentre, double-blind, randomised, placebo-controlled phase 3 trial. Lancet Child Adolesc Health 2020, 4, 728–739. [Google Scholar] [CrossRef]
- Kulis, M.D.; Patil, S.U.; Wambre, E.; Vickery, B.P. Immune mechanisms of oral immunotherapy. J. Allergy Clin. Immunol. 2018, 141, 491–498. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ishida, W.; Fukuda, K.; Harada, Y.; Sumi, T.; Taguchi, O.; Tsuda, M.; Yagita, H.; Fukushima, A. Oral administration of Ag suppresses Ag-induced allergic conjunctivitis in mice: Critical timing and dose of Ag. Br. J. Ophthalmol. 2013, 97, 492–497. [Google Scholar] [CrossRef] [PubMed]
- Takaiwa, F. A Rice-Based Edible Vaccine Expressing Multiple T-Cell Epitopes to Induce Oral Tolerance and Inhibit Allergy. Immunol. Allergy Clin. N. Am. 2007, 27, 129–139. [Google Scholar] [CrossRef] [PubMed]
- Takaiwa, F. Seed-based oral vaccines as allergen-specific immunotherapies. Hum. Vaccines 2011, 7, 357–366. [Google Scholar] [CrossRef] [Green Version]
- Takaiwa, F. Update on the use of transgenic rice seeds in oral immunotherapy. Immunotherapy 2013, 5, 301–312. [Google Scholar] [CrossRef]
- Takaiwa, F.; Wakasa, Y.; Takagi, H.; Hiroi, T. Rice seed for delivery of vaccines to gut mucosal immune tissues. Plant Biotechnol. J. 2015, 13, 1041–1055. [Google Scholar] [CrossRef]
- Wakasa, Y.; Takagi, H.; Hirose, S.; Yang, L.; Saeki, M.; Nishimura, T.; Kaminuma, O.; Hiroi, T.; Takaiwa, F. Oral immunotherapy with transgenic rice seed containing destructed Japanese cedar pollen allergens, Cry j 1 and Cry j 2, against Japanese cedar pollinosis. Plant Biotechnol. J. 2012, 11, 66–76. [Google Scholar] [CrossRef] [PubMed]
- Fukuda, K.; Ishida, W.; Harada, Y.; Wakasa, Y.; Takagi, H.; Takaiwa, F.; Fukushima, A. Prevention of allergic conjunctivitis in mice by a rice-based edible vaccine containing modified Japanese cedar pollen allergens. Br. J. Ophthalmol. 2015, 99, 705–709. [Google Scholar] [CrossRef] [PubMed]
- Fukuda, K.; Ishida, W.; Harada, Y.; Wakasa, Y.; Takagi, H.; Takaiwa, F.; Fukushima, A. Efficacy of oral immunotherapy with a rice-based edible vaccine containing hypoallergenic Japanese cedar pollen allergens for treatment of established allergic conjunctivitis in mice. Allergol. Int. 2017, 67, 119–123. [Google Scholar] [CrossRef] [PubMed]
- Saito, S.; Takagi, H.; Wakasa, Y.; Ozawa, K.; Takaiwa, F. Safety and efficacy of rice seed-based oral allergy vaccine for Japanese cedar pollinosis in Japanese monkeys. Mol. Immunol. 2020, 125, 63–69. [Google Scholar] [CrossRef] [PubMed]
- Ogo, Y.; Takahashi, H.; Wang, S.; Takaiwa, F. Generation mechanism of novel, huge protein bodies containing wild type or hypoallergenic derivatives of birch pollen allergen Bet v 1 in rice endosperm. Plant Mol. Biol. 2014, 86, 111–123. [Google Scholar] [CrossRef]
- Wang, S.; Takahashi, H.; Kajiura, H.; Kawakatsu, T.; Fujiyama, K.; Takaiwa, F. Transgenic Rice Seeds Accumulating Recombinant Hypoallergenic Birch Pollen Allergen Bet v 1 Generate Giant Protein Bodies. Plant Cell Physiol. 2013, 54, 917–933. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wallner, M.; Stöcklinger, A.; Thalhamer, T.; Bohle, B.; Vogel, L.; Briza, P.; Breiteneder, H.; Vieths, S.; Hartl, A.; Mari, A.; et al. Allergy multivaccines created by DNA shuffling of tree pollen allergens. J. Allergy Clin. Immunol. 2007, 120, 374–380. [Google Scholar] [CrossRef] [PubMed]
- Magone, M.T.; Chan, C.C.; Rizzo, L.V.; Kozhich, A.T.; Whitcup, S.M. A novel murine model of allergic conjunctivitis. Clin. Immunol. Immunopathol. 1998, 87, 75–84. [Google Scholar] [CrossRef] [PubMed]
- Fukuda, K.; Kuo, C.-H.; Morohoshi, K.; Liu, F.-T.; Ono, S.J. The murine CCR3 receptor regulates both eosinophilia and hyperresponsiveness in IgE-mediated allergic conjunctivitis. Br. J. Ophthalmol. 2012, 96, 1132–1136. [Google Scholar] [CrossRef]
- Takagi, H.; Hiroi, T.; Yang, L.; Tada, Y.; Yuki, Y.; Takamura, K.; Ishimitsu, R.; Kawauchi, H.; Kiyono, H.; Takaiwa, F. From The Cover: A rice-based edible vaccine expressing multiple T cell epitopes induces oral tolerance for inhibition of Th2-mediated IgE responses. Proc. Natl. Acad. Sci. USA 2005, 102, 17525–17530. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Breiteneder, H.; Pettenburger, K.; Bito, A.; Valenta, R.; Kraft, D.; Rumpold, H.; Scheiner, O.; Breitenbach, M. The gene coding for the major birch pollen allergen Betv1, is highly homologous to a pea disease resistance response gene. EMBO J. 1989, 8, 1935–1938. [Google Scholar] [CrossRef] [PubMed]
- Gajhede, M.; Osmark, P.; Poulsen, F.M.; Ipsen, H.; Larsen, J.N.; Joost van Neerven, R.J.; Schou, C.; Løwenstein, H.; Spangfort, M.D. X-ray and NMR structure of Bet v 1, the origin of birch pollen allergy. Nat. Struct. Biol. 1996, 3, 1040–1045. [Google Scholar] [CrossRef] [PubMed]
- Weber, R.W. Patterns of pollen cross-allergenicity. J. Allergy Clin. Immunol. 2003, 112, 229–239. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Radauer, C.; Breiteneder, H. Pollen allergens are restricted to few protein families and show distinct patterns of species distribution. J. Allergy Clin. Immunol. 2006, 117, 141–147. [Google Scholar] [CrossRef]
- Vieths, S.; Scheurer, S.; Ballmer-Weber, B. Current Understanding of Cross-Reactivity of Food Allergens and Pollen. Ann. N. Y. Acad. Sci. 2006, 964, 47–68. [Google Scholar] [CrossRef] [PubMed]
- Kucuksezer, U.C.; Ozdemir, C.; Cevhertas, L.; Ogulur, I.; Akdis, M.; Akdis, C.A. Mechanisms of allergen-specific immunotherapy and allergen tolerance. Allergol. Int. Off. J. Jpn. Soc. Allergol. 2020, 69, 549–560. [Google Scholar] [CrossRef]
- Ciprandi, G.; Fenoglio, D.; DI Gioacchino, M.; Ferrera, A.; Ferrera, F.; Sormani, M.P.; Marseglia, G.L. Sublingual immunotherapy provides an early increase of interferon-gamma production. J. Biol. Regul. Homeost. Agents 2008, 22, 169–173. [Google Scholar] [PubMed]
- Potter, P.C.; Baker, S.; Fenemore, B.; Nurse, B. Clinical and cytokine responses to house dust mite sublingual immunotherapy. Ann. Allergy Asthma Immunol. Off. Publ. Am. Coll. Allergy Asthma Immunol. 2015, 114, 327–334. [Google Scholar] [CrossRef] [PubMed]
- Wambre, E.; DeLong, J.H.; James, E.A.; LaFond, R.E.; Robinson, D.; Kwok, W.W. Differentiation stage determines pathologic and protective allergen-specific CD4+ T-cell outcomes during specific immunotherapy. J. Allergy Clin. Immunol. 2012, 129, 544–551.e7. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jutel, M.; Pichler, W.J.; Skrbic, D.; Urwyler, A.; Dahinden, C.; Müller, U.R. Bee venom immunotherapy results in decrease of IL-4 and IL-5 and increase of IFN-gamma secretion in specific allergen-stimulated T cell cultures. J. Immunol. 1995, 154, 4187–4194. [Google Scholar] [PubMed]
- Bellinghausen, I.; Metz, G.; Enk, A.H.; Christmann, S.; Knop, J.; Saloga, J. Insect venom immunotherapy induces interleukin-10 production and a Th2-to-Th1 shift, and changes surface marker expression in venom-allergic subjects. Eur. J. Immunol. 1997, 27, 1131–1139. [Google Scholar] [CrossRef] [PubMed]
- Fukushima, A.; Jian, Z.; Ishida, W.; Fukata, K.; Ozaki, A.; Ueno, H. Interleukin-4-mediated infiltration of eosinophils into the conjunctiva and its suppression by interferon-gamma. Curr. Eye Res. 2005, 30, 115–122. [Google Scholar] [CrossRef] [PubMed]
- Santos, A.; James, L.; Bahnson, H.T.; Shamji, M.H.; Couto-Francisco, N.C.; Islam, S.; Houghton, S.; Clark, A.T.; Stephens, A.; Turcanu, V.; et al. IgG4 inhibits peanut-induced basophil and mast cell activation in peanut-tolerant children sensitized to peanut major allergens. J. Allergy Clin. Immunol. 2015, 135, 1249–1256. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Spergel, J.M.; Elci, O.U.; Muir, A.B.; Liacouras, C.A.; Wilkins, B.J.; Burke, D.; Lewis, M.O.; Brown-Whitehorn, T.; Cianferoni, A. Efficacy of Epicutaneous Immunotherapy in Children With Milk-Induced Eosinophilic Esophagitis. Clin. Gastroenterol. Hepatol. 2019, 18, 328–336.e7. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Senti, G.; Freiburghaus, A.U.; Larenas-Linnemann, D.; Hoffmann, H.J.; Patterson, A.M.; Klimek, L.; Di Bona, D.; Pfaar, O.; Ahlbeck, L.; Akdis, M.; et al. Intralymphatic Immunotherapy: Update and Unmet Needs. Int. Arch. Allergy Immunol. 2019, 178, 141–149. [Google Scholar] [CrossRef] [Green Version]
| Non-Tg Rice | Tg Rice | |
|---|---|---|
| Mesenteric lymph node | 4.82 ± 0.36 | 4.32 ± 0.12 |
| Spleen | 2.26 ± 0.21 | 1.88 ± 0.07 |
| Non-Tg Rice | Tg Rice | |
|---|---|---|
| Mesenteric lymph node | 4.50 ± 0.24 | 4.43 ± 0.36 |
| Spleen | 2.39 ± 0.07 | 2.25 ± 0.13 |
| Prophylactic Administration | Therapeutic Administration | |||
|---|---|---|---|---|
| Non-Tg Rice | Bet v 1 Tg Rice | Non-Tg Rice | Bet v 1 Tg Rice | |
| Mesenteric lymph node | 4.97 ± 0.10 | 4.62 ± 0.26 | 3.64 ± 0.16 | 5.23 ± 0.47 * |
| Spleen | 2.53 ± 0.07 | 2.50 ± 0.25 | 2.52 ± 0.09 | 2.73 ± 0.05 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ishida, W.; Kishimoto, T.; Takaiwa, F.; Fukuda, K. Prophylactic and Therapeutic Effects of Oral Immunotherapy on Birch Pollen-Induced Allergic Conjunctivitis in Mice with a Rice-Based Edible Vaccine Expressing a Hypoallergenic Birch Pollen Allergen. Cells 2021, 10, 3361. https://doi.org/10.3390/cells10123361
Ishida W, Kishimoto T, Takaiwa F, Fukuda K. Prophylactic and Therapeutic Effects of Oral Immunotherapy on Birch Pollen-Induced Allergic Conjunctivitis in Mice with a Rice-Based Edible Vaccine Expressing a Hypoallergenic Birch Pollen Allergen. Cells. 2021; 10(12):3361. https://doi.org/10.3390/cells10123361
Chicago/Turabian StyleIshida, Waka, Tatsuma Kishimoto, Fumio Takaiwa, and Ken Fukuda. 2021. "Prophylactic and Therapeutic Effects of Oral Immunotherapy on Birch Pollen-Induced Allergic Conjunctivitis in Mice with a Rice-Based Edible Vaccine Expressing a Hypoallergenic Birch Pollen Allergen" Cells 10, no. 12: 3361. https://doi.org/10.3390/cells10123361
APA StyleIshida, W., Kishimoto, T., Takaiwa, F., & Fukuda, K. (2021). Prophylactic and Therapeutic Effects of Oral Immunotherapy on Birch Pollen-Induced Allergic Conjunctivitis in Mice with a Rice-Based Edible Vaccine Expressing a Hypoallergenic Birch Pollen Allergen. Cells, 10(12), 3361. https://doi.org/10.3390/cells10123361






